The Potential Application of Spring Sargassum glaucescens Extracts in the Moisture-Retention of Keratinocytes and Dermal Fibroblast Regeneration after UVA-Irradiation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Lines and Chemicals
2.2. Preparation of Extracts
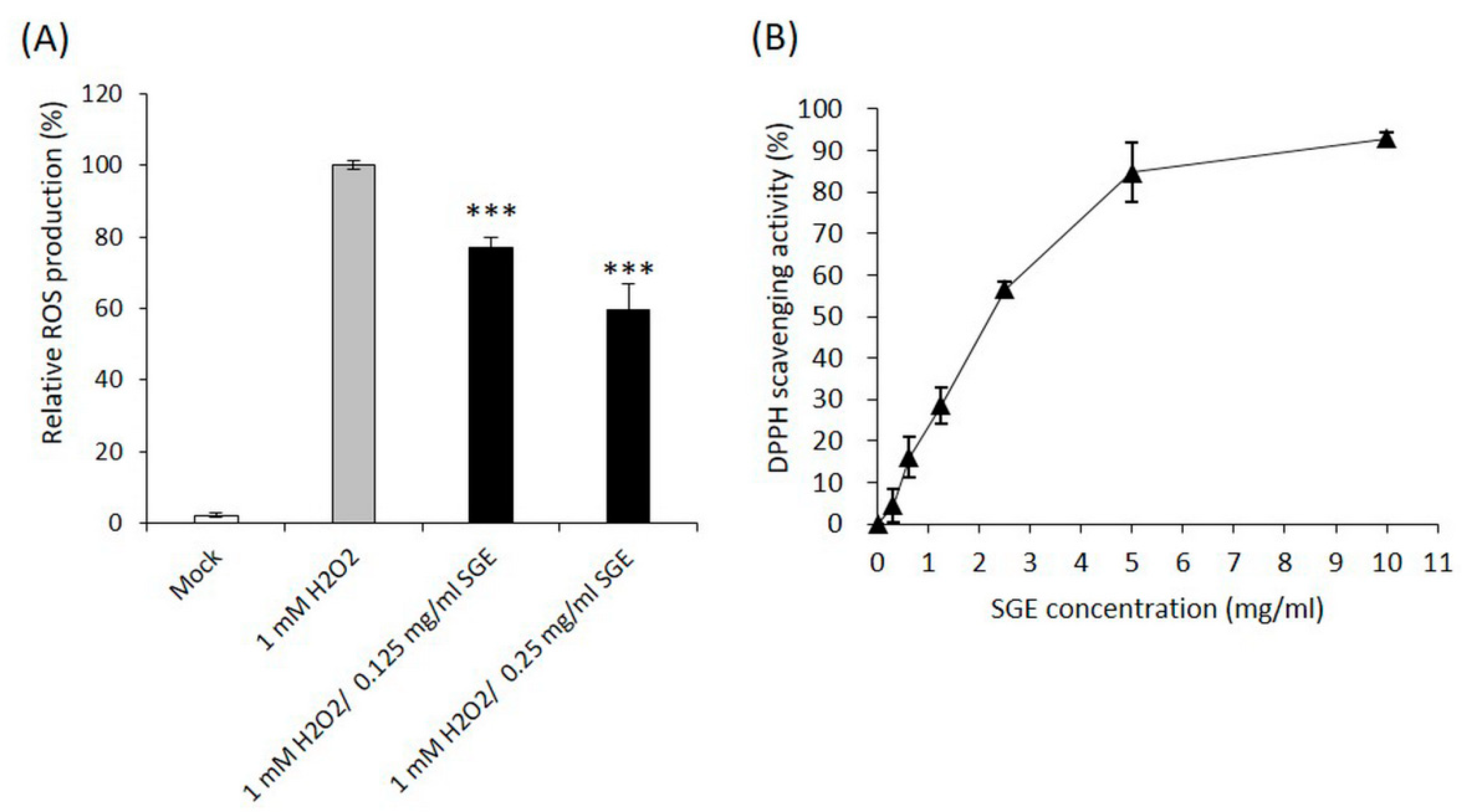
2.3. Quantification of Intracellular Reactive Oxygen Species (ROS) Levels
2.4. DPPH Radical Scavenging Activity
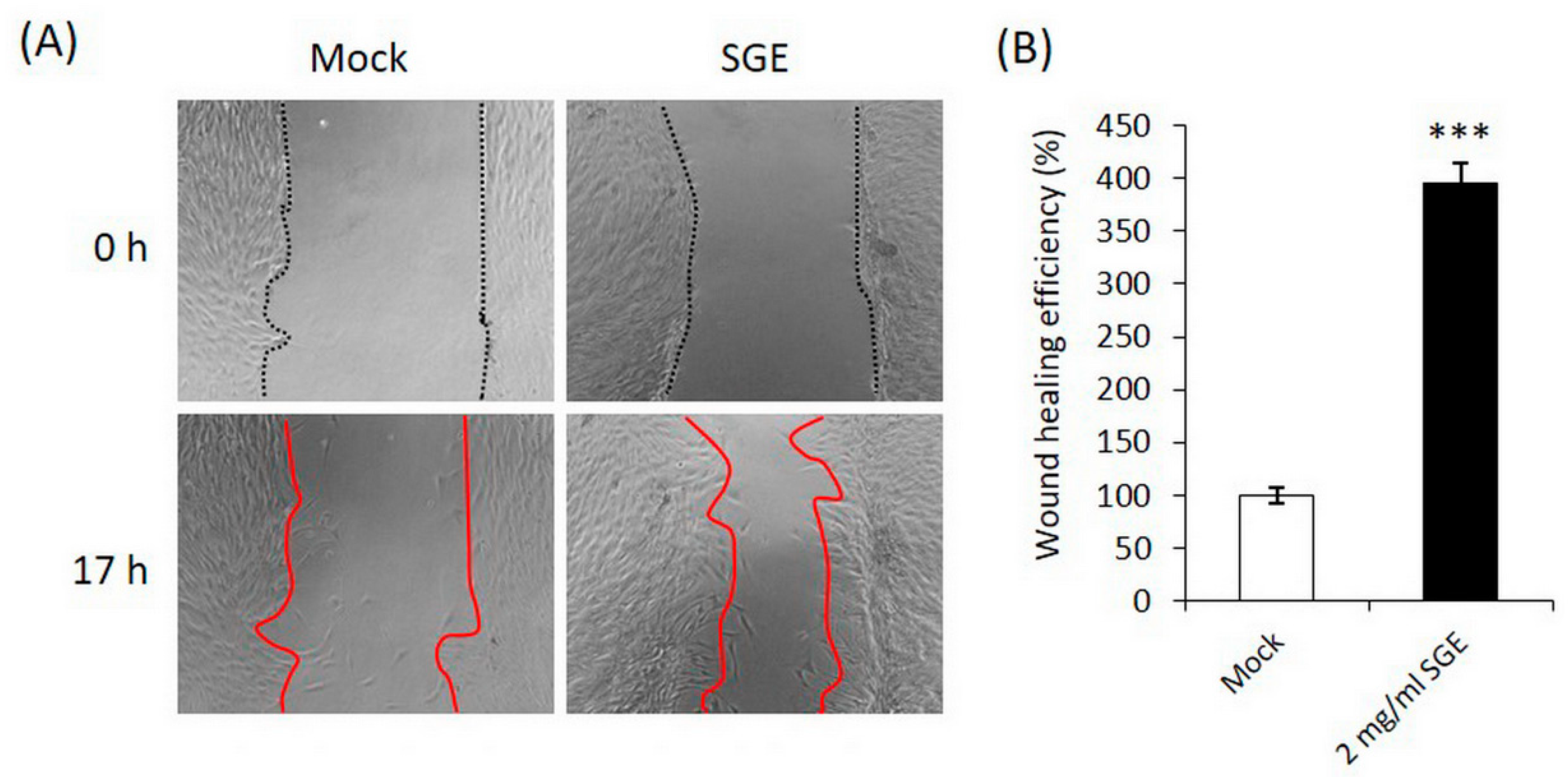
2.5. Wound Healing Assay
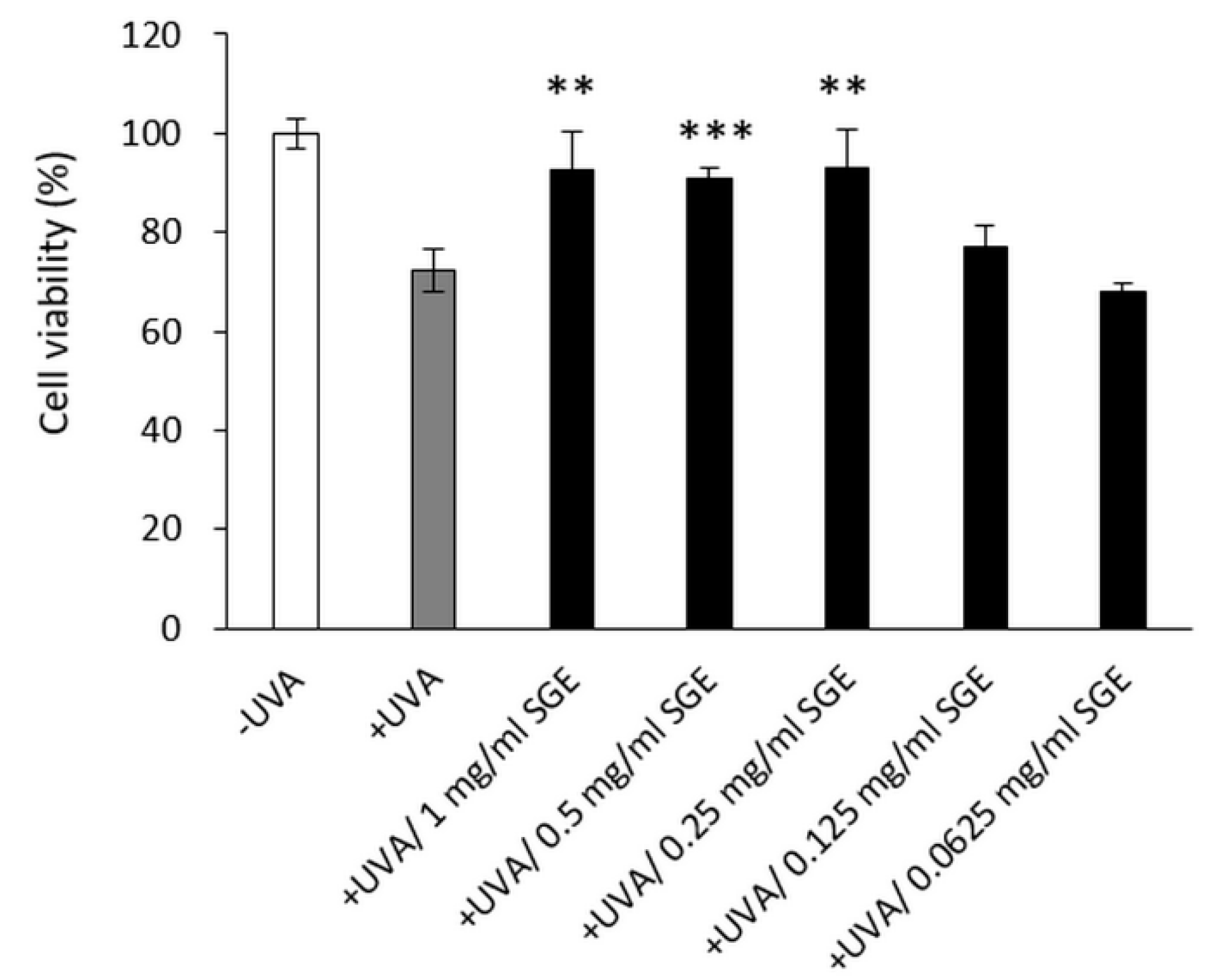
2.6. Cell Viability Assay
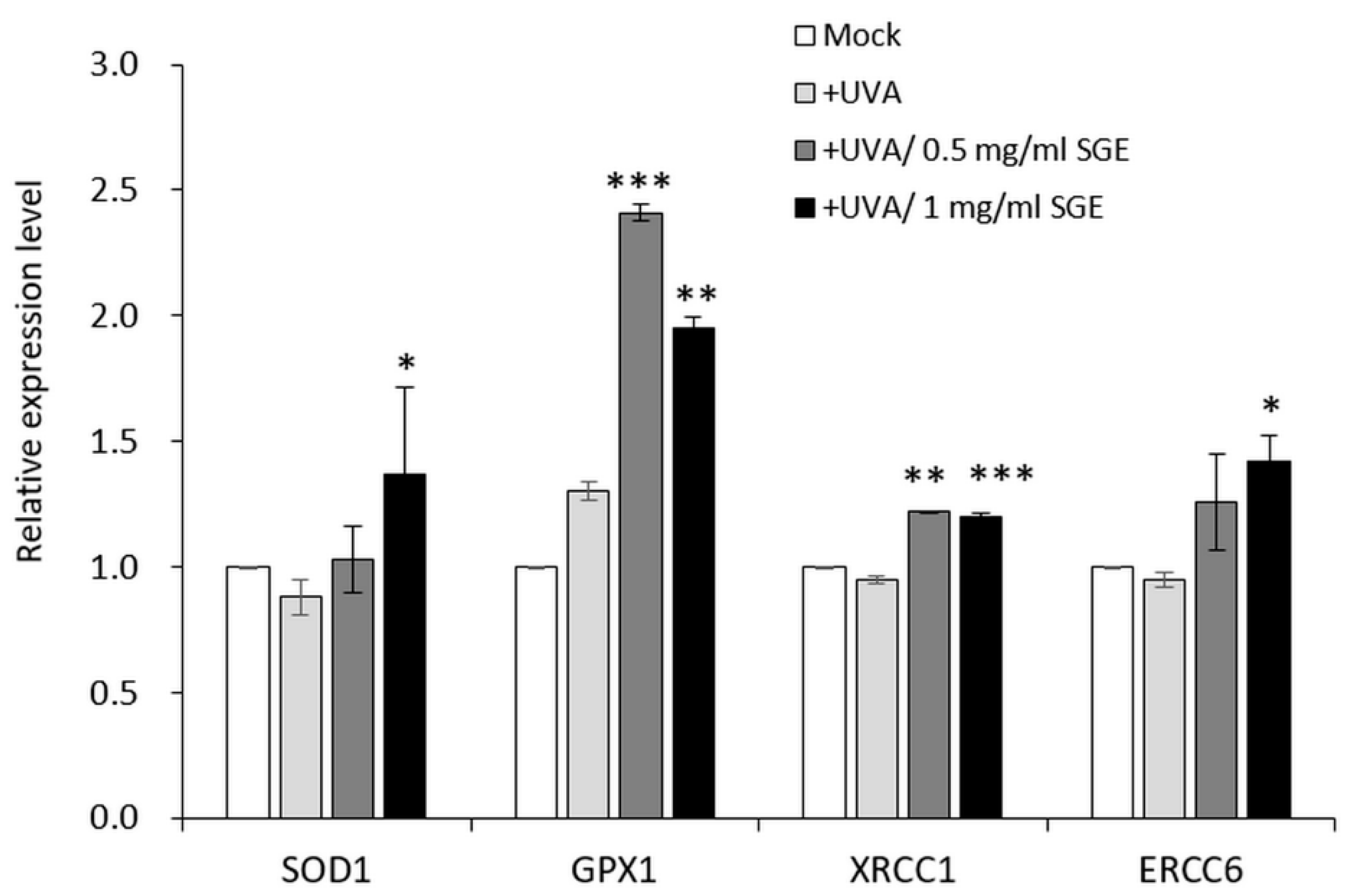
2.7. Quantification of Gene Expressions by Real-Time PCR
2.8. Statistical Analysis
3. Results and Discussion
3.1. Antioxidant and Wound Healing Activities of SGE
3.1.1. The Free Radical Scavenging Activities of SGE
3.1.2. SGE Stimulated CCD-966SK Fibroblast Migration in the Wound Healing Assay
3.2. Anti-Photoaging Activities Of SGE in Dermal Fibroblasts
3.2.1. SGE Recovered CCD-966SK Cell Regeneration after UVA Irradiation
3.2.2. SGE Induced Antioxidant Gene Expressions in CCD-966SK Cell after UVA Irradiation
3.3. Protective Effects of SGE in Keratinocytes and Skin Barrier
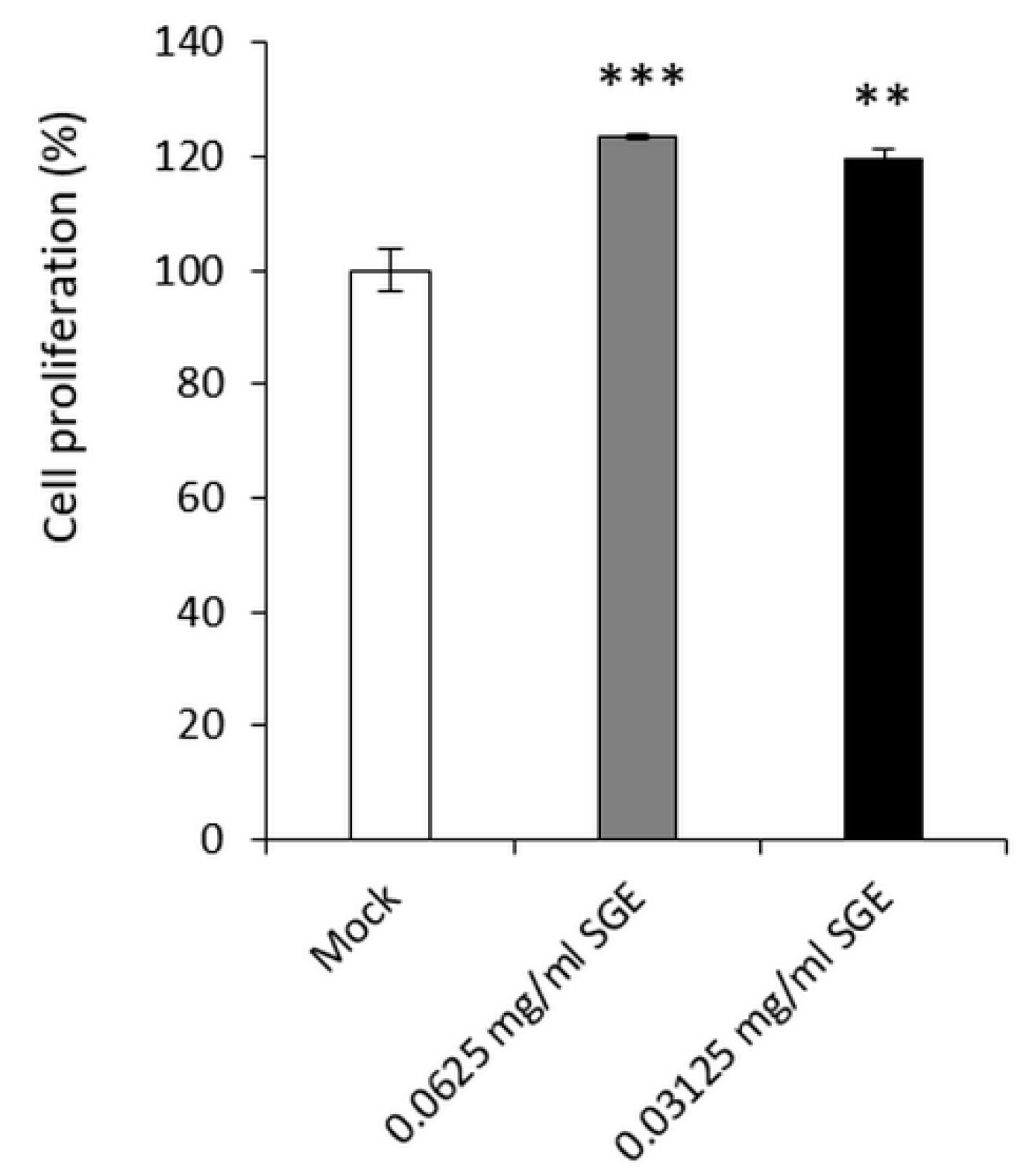
3.3.1. SGE Stimulated the Cell Proliferation of Human Primary Epidermal Keratinocytes
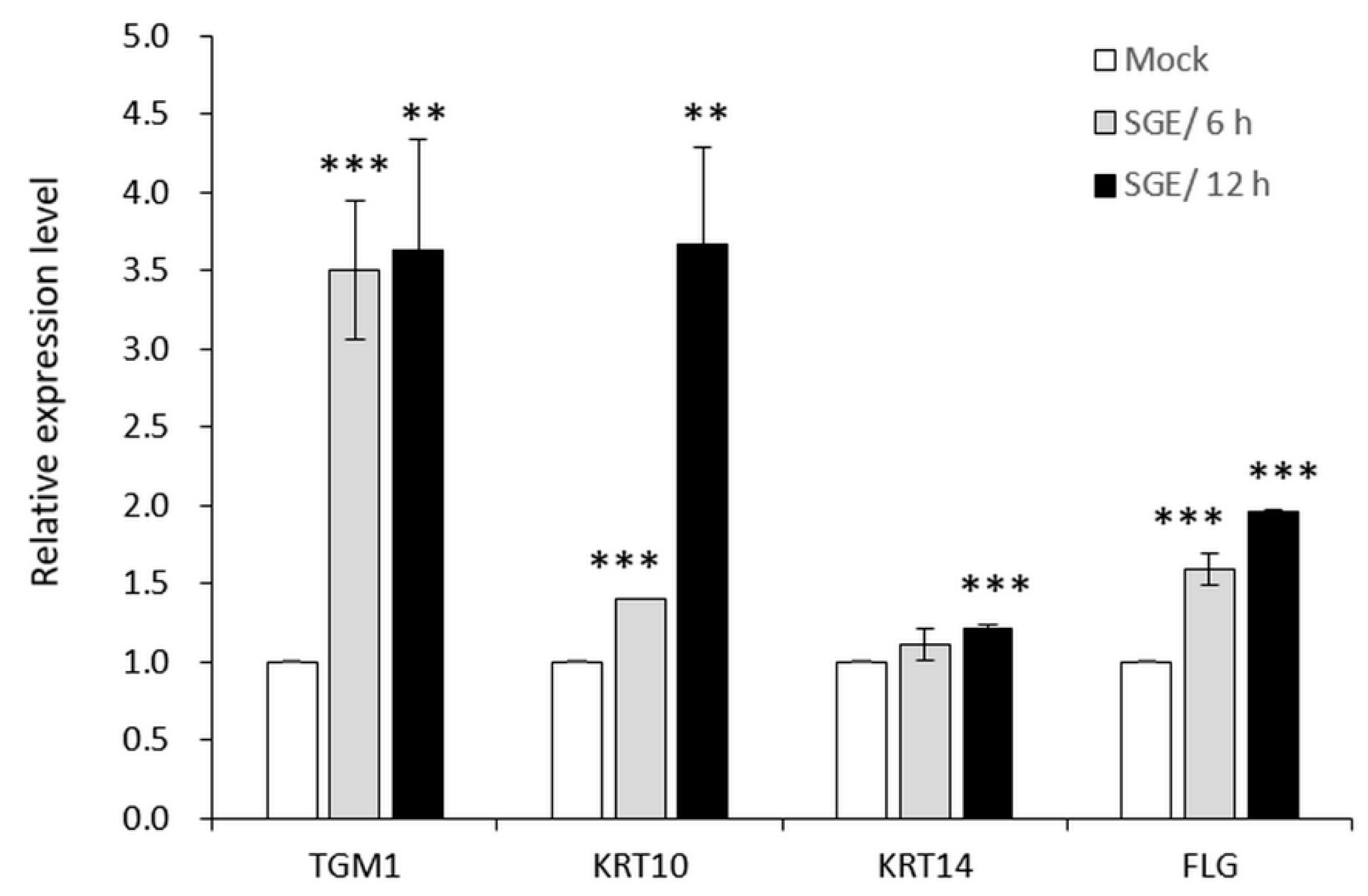
3.3.2. SGE Induced Expressions of Skin Barrier-Related Genes in Human Primary Epidermal Keratinocytes
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Berthon, J.Y.; Nachat-Kappes, R.; Bey, M.; Cadoret, J.P.; Renimel, I.; Filaire, E. Marine algae as attractive source to skin care. Free Radic. Res. 2017, 51, 555–567. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, T.; Fukuda, M.; Mikami, A.; Mizuno, S.; Kantachumpoo, A.; Tanoue, H.; Kawamiya, M. Possible change in distribution of seaweed, Sargassum horneri, in northeast Asia under A2 scenario of global warming and consequent effect on some fish. Mar. Pollut. Bull. 2014, 85, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Meenakshi, S.; Umayaparvathi, S.; Arumugam, M.; Balasubramanian, T. In vitro antioxidant properties and ftir analysis of two seaweeds of gulf of mannar. Asian Pac. J. Trop. Med. 2011, 1, S66–S70. [Google Scholar] [CrossRef]
- Manav, M.; Su, J.; Hughes, K.; Lee, H.P.; Ong, C.N. ω-3 fatty acids and selenium as coronary heart disease risk modifying factors in Asian Indian and Chinese males. Nutrition 2004, 20, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Farage, M.A.; Miller, K.W.; Elsner, P.; Maibach, H.I. Intrinsic and extrinsic factors in skin ageing: A review. Int. J. Cosmet. Sci. 2008, 30, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Han, A.; Chien, A.L.; Kang, S. Photoaging. Dermatol. Clin. 2014, 32, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Polefka, T.G.; Meyer, T.A.; Agin, P.P.; Bianchini, R.J. Effects of solar radiation on the skin. J. Cosmet. Dermatol. 2012, 11, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Fuentealba, D.; Galvez, M.; Alarcon, E.; Lissi, E.; Silva, E. Photosensitizing activity of advanced glycation endproducts on tryptophan, glucose 6-phosphate dehydrogenase, human serum albumin and ascorbic acid evaluated at low oxygen pressure. Photochem. Photobiol. 2007, 83, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.A.; Ahn, B.N.; Kong, C.S.; Kim, S.K. Protective effect of chromene isolated from Sargassum horneri against UV-A-induced damage in skin dermal fibroblasts. Exp. Dermatol. 2012, 21, 630–631. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.A.; Ahn, B.N.; Kong, C.S.; Kim, S.K. The chromene sargachromanol E inhibits ultraviolet A-induced ageing of skin in human dermal fibroblasts. Br. J. Dermatol. 2013, 168, 968–976. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Park, G.H.; Ahn, E.M.; Park, C.I.; Jang, J.H. Sargassum fulvellum protects HaCaT cells and BALB/c mice from UVB-induced proinflammatory responses. Evid. Based Complement. Alternat. Med. 2013, 2013, 747846. [Google Scholar] [CrossRef] [PubMed]
- Shao, P.; Chen, X.; Sun, P. Improvement of antioxidant and moisture-preserving activities of Sargassum horneri polysaccharide enzymatic hydrolyzates. Int. J. Biol. Macromol. 2015, 74, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.Y.; Wu, S.J.; Yang, W.N.; Kuan, A.W.; Chen, C.Y. Antioxidant activities of crude extracts of fucoidan extracted from Sargassum glaucescens by a compressional-puffing-hydrothermal extraction process. Food Chem. 2016, 197, 1121–1129. [Google Scholar] [CrossRef] [PubMed]
- Collins, K.G.; Fitzgerald, G.F.; Stanton, C.; Ross, R.P. Looking beyond the terrestrial: The potential of seaweed derived bioactives to treat non-communicable diseases. Mar. Drugs 2016, 14, 60. [Google Scholar] [CrossRef] [PubMed]
- Vijayabaskar, P.; Vaseela, N.; Thirumaran, G. Potential antibacterial and antioxidant properties of a sulfated polysaccharide from the brown marine algae Sargassum swartzii. Chin. J. Nat. Med. 2012, 10, 421–428. [Google Scholar] [CrossRef]
- Ale, M.T.; Maruyama, H.; Tamauchi, H.; Mikkelsen, J.D.; Meyer, A.S. Fucoidan from Sargassum sp. and fucus vesiculosus reduces cell viability of lung carcinoma and melanoma cells in vitro and activates natural killer cells in mice in vivo. Int. J. Biol. Macromol. 2011, 49, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Cong, Q.; Chen, H.; Liao, W.; Xiao, F.; Wang, P.; Qin, Y.; Dong, Q.; Ding, K. Structural characterization and effect on anti-angiogenic activity of a fucoidan from Sargassum fusiforme. Carbohydr. Polym. 2016, 136, 899–907. [Google Scholar] [CrossRef] [PubMed]
- Dore, C.M.; das, C.F.A.M.G.; Will, L.S.; Costa, T.G.; Sabry, D.A.; de Souza Rego, L.A.; Accardo, C.M.; Rocha, H.A.; Filgueira, L.G.; Leite, E.L. A sulfated polysaccharide, fucans, isolated from brown algae Sargassum vulgare with anticoagulant, antithrombotic, antioxidant and anti-inflammatory effects. Carbohydr. Polym. 2013, 91, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Morya, V.K.; Kim, J.; Kim, E.K. Algal fucoidan: Structural and size-dependent bioactivities and their perspectives. Appl. Microbiol. Biotechnol. 2012, 93, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Wells, M.L.; Potin, P.; Craigie, J.S.; Raven, J.A.; Merchant, S.S.; Helliwell, K.E.; Smith, A.G.; Camire, M.E.; Brawley, S.H. Algae as nutritional and functional food sources: Revisiting our understanding. J. Appl. Phycol. 2017, 29, 949–982. [Google Scholar] [CrossRef] [PubMed]
- Marinho-Soriano, E.; Fonseca, P.C.; Carneiro, M.A.; Moreira, W.S. Seasonal variation in the chemical composition of two tropical seaweeds. Bioresour. Technol. 2006, 97, 2402–2406. [Google Scholar] [CrossRef] [PubMed]
- Murugaiyan, K.; Sivakumar, K. Seasonal variation in elemental composition of Stoechospermum marginatum (Ag.) Kutz and Sargassum wightii (Greville Mscr.) J.G. Agardh in relation to chemical composition of seawater. Colloids Surf. B Biointerfaces 2008, 64, 140–144. [Google Scholar] [CrossRef] [PubMed]
- Hwang, P.A.; Hung, Y.L.; Tsai, Y.K.; Chien, S.Y.; Kong, Z.L. The brown seaweed Sargassum hemiphyllum exhibits alpha-amylase and alpha-glucosidase inhibitory activity and enhances insulin release in vitro. Cytotechnology 2015, 67, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Shimada, K.; Fujikawa, K.; Yahara, K.; Nakamura, T. Antioxidative properties of xanthan on the autoxidation of soybean oil in cyclodextrin emulsion. J. Agric. Food Chem. 1992, 40, 945–948. [Google Scholar] [CrossRef]
- Yarrow, J.C.; Perlman, Z.E.; Westwood, N.J.; Mitchison, T.J. A high-throughput cell migration assay using scratch wound healing, a comparison of image-based readout methods. BMC Biotechnol. 2004, 4, 21. [Google Scholar] [CrossRef] [PubMed]
- Shindo, Y.; Hashimoto, T. Antioxidant defence mechanism of the skin against uv irradiation: Study of the role of catalase using acatalasaemia fibroblasts. Arch. Dermatol. Res. 1995, 287, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Yende, S.R.; Harle, U.N.; Chaugule, B.B. Therapeutic potential and health benefits of Sargassum species. Pharmacogn. Rev. 2014, 8, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Hwang, P.A.; Wu, C.H.; Gau, S.Y.; Chien, S.Y.; Hwang, D.F. Antioxidant and immune-stimulating activities of hot-water extract from seaweed Sargassum hemiphyllum. J. Mar. Sci. Technol. 2010, 18, 41–46. [Google Scholar]
- Park, P.J.; Heo, S.J.; Park, E.J.; Kim, S.K.; Byun, H.G.; Jeon, B.T.; Jeon, Y.J. Reactive oxygen scavenging effect of enzymatic extracts from Sargassum thunbergii. J. Agric. Food Chem. 2005, 53, 6666–6672. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.N.; Cheung, P.C.; Ooi, V.E.; Ang, P.O. Evaluation of antioxidative activity of extracts from a brown seaweed, Sargassum siliquastrum. J. Agric. Food Chem. 2002, 50, 3862–3866. [Google Scholar] [CrossRef] [PubMed]
- Wen, Z.S.; Liu, L.J.; OuYang, X.K.; Qu, Y.L.; Chen, Y.; Ding, G.F. Protective effect of polysaccharides from Sargassum horneri against oxidative stress in RAW264.7 cells. Int. J. Biol. Macromol. 2014, 68, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Hulikere, M.M.; Joshi, C.G.; Ananda, D.; Poyya, J.; Nivya, T. Antiangiogenic, wound healing and antioxidant activity of Cladosporium cladosporioides (Endophytic fungus) isolated from seaweed (Sargassum wightii). Mycology 2016, 7, 203–211. [Google Scholar] [CrossRef] [PubMed]
- D’Orazio, N.; Gemello, E.; Gammone, M.A.; de Girolamo, M.; Ficoneri, C.; Riccioni, G. Fucoxantin: A treasure from the sea. Mar. Drugs 2012, 10, 604–616. [Google Scholar] [CrossRef]
- Weydert, C.J.; Cullen, J.J. Measurement of superoxide dismutase, catalase and glutathione peroxidase in cultured cells and tissue. Nat. Protoc. 2010, 5, 51–66. [Google Scholar] [CrossRef]
- Newman, J.C.; Bailey, A.D.; Weiner, A.M. Cockayne syndrome group B protein (CSB) plays a general role in chromatin maintenance and remodeling. Proc. Natl. Acad. Sci. USA 2006, 103, 9613–9618. [Google Scholar] [CrossRef]
- Lake, R.J.; Fan, H.-Y. Structure, function and regulation of CSB: A multi-talented gymnast. Mech. Ageing Dev. 2013, 134, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Ramaniuk, V.P.; Nikitchenko, N.V.; Savina, N.V.; Kuzhir, T.D.; Rolevich, A.I.; Krasny, S.A.; Sushinsky, V.E.; Goncharova, R.I. Polymorphism of DNA repair genes OGG1, XRCC1, XPD and ERCC6 in bladder cancer in Belarus. Biomarkers 2014, 19, 509–516. [Google Scholar] [CrossRef]
- Kindleysides, S.; Quek, S.Y.; Miller, M.R. Inhibition of fish oil oxidation and the radical scavenging activity of New Zealand seaweed extracts. Food Chem. 2012, 133, 1624–1631. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Q.; Zhang, Z.; Song, H.; Li, P. Potential antioxidant and anticoagulant capacity of low molecular weight fucoidan fractions extracted from Laminaria japonica. Int. J. Biol. Macromol. 2010, 46, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Segre, J.A. Epidermal barrier formation and recovery in skin disorders. J. Clin. Investig. 2006, 116, 1150–1158. [Google Scholar] [CrossRef]
- Chamcheu, J.C.; Siddiqui, I.A.; Syed, D.N.; Adhami, V.M.; Liovic, M.; Mukhtar, H. Keratin gene mutations in disorders of human skin and its appendages. Arch. Biochem. Biophys. 2011, 508, 123–137. [Google Scholar] [CrossRef] [PubMed]
- Magin, T.M.; Vijayaraj, P.; Leube, R.E. Structural and regulatory functions of keratins. Exp. Cell Res. 2007, 313, 2021–2032. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, J.; Bowden, P.E.; Coulombe, P.A.; Langbein, L.; Lane, E.B.; Magin, T.M.; Maltais, L.; Omary, M.B.; Parry, D.A.; Rogers, M.A.; et al. New consensus nomenclature for mammalian keratins. J. Cell Biol. 2006, 174, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Moll, R.; Divo, M.; Langbein, L. The human keratins: Biology and pathology. Histochem. Cell Biol. 2008, 129, 705–733. [Google Scholar] [CrossRef] [PubMed]
- Kezic, S.; Kemperman, P.M.; Koster, E.S.; de Jongh, C.M.; Thio, H.B.; Campbell, L.E.; Irvine, A.D.; McLean, W.H.; Puppels, G.J.; Caspers, P.J. Loss-of-function mutations in the filaggrin gene lead to reduced level of natural moisturizing factor in the stratum corneum. J. Investig. Dermatol. 2008, 128, 2117–2119. [Google Scholar] [CrossRef] [PubMed]
| Gene Name | Primer Name | Primer Sequence | Gene Function |
|---|---|---|---|
| SOD1 | SOD1-F | 5′- GGTGGGCCAAAGGATGAAGAG -3′ | anti-oxidative gene |
| SOD1-R | 5′- CCACAAGCCAAACGACTTCC -3′ | ||
| GPX1 | GPX1-F | 5′- CAGTCGGTGTATGCCTTCTCG -3′ | anti-oxidative gene |
| GPX1-R | 5′- GAGGGACGCCACATTCTCG -3′ | ||
| XRCC1 | XRCC1-F | 5′- TTCTTCCAGGGCAAGCACTT -3′ | DNA repair regulatory gene |
| XRCC1-R | 5′- GATCCCATTCCTGTGCTGTGA -3′ | ||
| ERCC6 | ERCC6-F | 5′- GTCCAAGATCACATAGTGCC -3′ | DNA repair regulatory gene |
| ERCC6-R | 5′- AAAAGGAGAAACTAATTCGA -3′ | ||
| TGM1 | TGM1-F | 5′- GATCGCATCACCCTTGAGTTAC -3′ | epidermal barrier structural genes |
| TGM1-R | 5′- GCAGGTTCAGATTCTGCCC -3′ | ||
| KRT10 | KRT10-F | 5′- TCCTACTTGGACAAAGTTCGGG -3′ | epidermal barrier structural genes |
| KRT10-R | 5′- CCCCTGATGTGAGTTGCCA -3′ | ||
| KRT14 | KRT14-F | 5′- TTCTGAACGAGATGCGTGAC -3′ | epidermal barrier structural genes |
| KRT14-R | 5′- GCAGCTCAATCTCCAGGTTC -3′ | ||
| FLG | FLG-F | 5′- GGCAAATCCTGAAGAATCCA -3′ | epidermal barrier structural genes |
| FLG-R | 5′- TGCTTTCTGTGCTTGTGTCC -3′ | ||
| GAPDH | GAPDH-F | 5′- CTGGGCTACACTGAGCACC -3′ | housekeeping gene |
| GAPDH-R | 5′- AAGTGGTCGTTGAGGGCAATG -3′ |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Z.-y.; Yu, C.-H.; Lin, Y.-T.; Su, H.-L.; Kan, K.-W.; Liu, F.-C.; Chen, C.-T.; Lin, Y.-T.; Hsu, H.-F.; Lin, Y.-H. The Potential Application of Spring Sargassum glaucescens Extracts in the Moisture-Retention of Keratinocytes and Dermal Fibroblast Regeneration after UVA-Irradiation. Cosmetics 2019, 6, 17. https://doi.org/10.3390/cosmetics6010017
Li Z-y, Yu C-H, Lin Y-T, Su H-L, Kan K-W, Liu F-C, Chen C-T, Lin Y-T, Hsu H-F, Lin Y-H. The Potential Application of Spring Sargassum glaucescens Extracts in the Moisture-Retention of Keratinocytes and Dermal Fibroblast Regeneration after UVA-Irradiation. Cosmetics. 2019; 6(1):17. https://doi.org/10.3390/cosmetics6010017
Chicago/Turabian StyleLi, Zih-yi, Chin-Hsiu Yu, Yu-Ting Lin, Hsiang-Ling Su, Kai-Wen Kan, Fu-Chen Liu, Ciao-Ting Chen, Yi-Tsen Lin, Hsin-Fen Hsu, and Yung-Hsiang Lin. 2019. "The Potential Application of Spring Sargassum glaucescens Extracts in the Moisture-Retention of Keratinocytes and Dermal Fibroblast Regeneration after UVA-Irradiation" Cosmetics 6, no. 1: 17. https://doi.org/10.3390/cosmetics6010017
APA StyleLi, Z.-y., Yu, C.-H., Lin, Y.-T., Su, H.-L., Kan, K.-W., Liu, F.-C., Chen, C.-T., Lin, Y.-T., Hsu, H.-F., & Lin, Y.-H. (2019). The Potential Application of Spring Sargassum glaucescens Extracts in the Moisture-Retention of Keratinocytes and Dermal Fibroblast Regeneration after UVA-Irradiation. Cosmetics, 6(1), 17. https://doi.org/10.3390/cosmetics6010017





