Skin Retention of Sorbates from an After Sun Formulation for a Broad Photoprotection
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
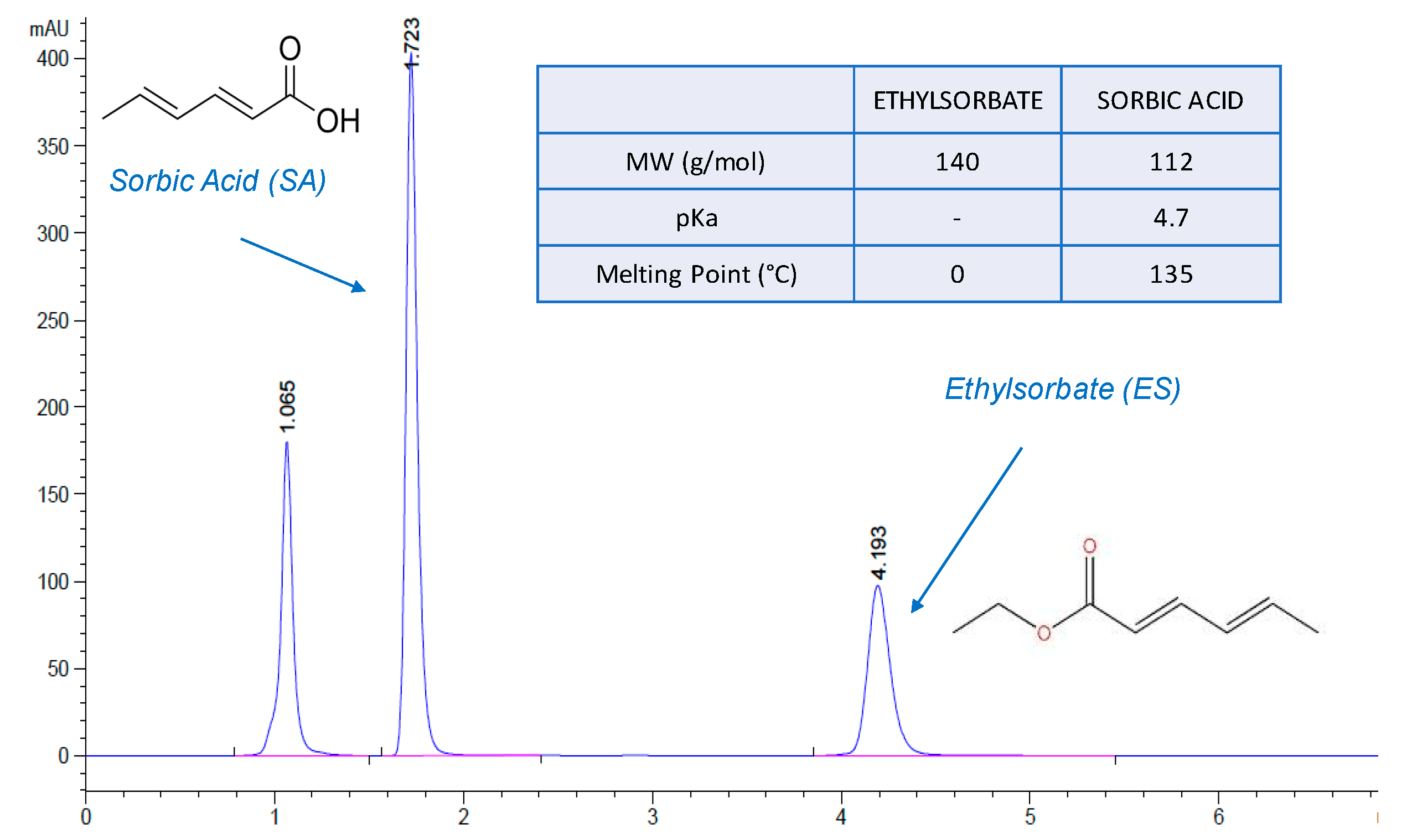
2.2. HPLC analysis
2.3. Determination of the Partition/Distribution Coefficient of Sorbic Acid and Ethylsorbate
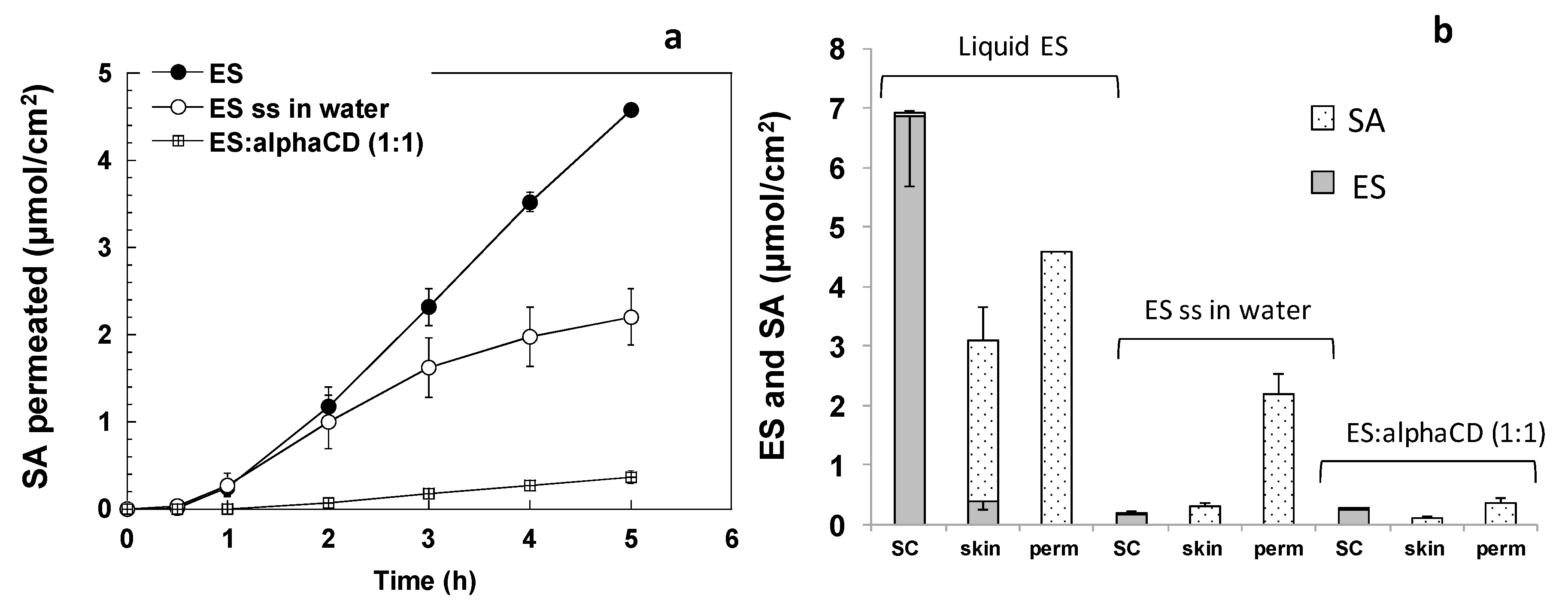
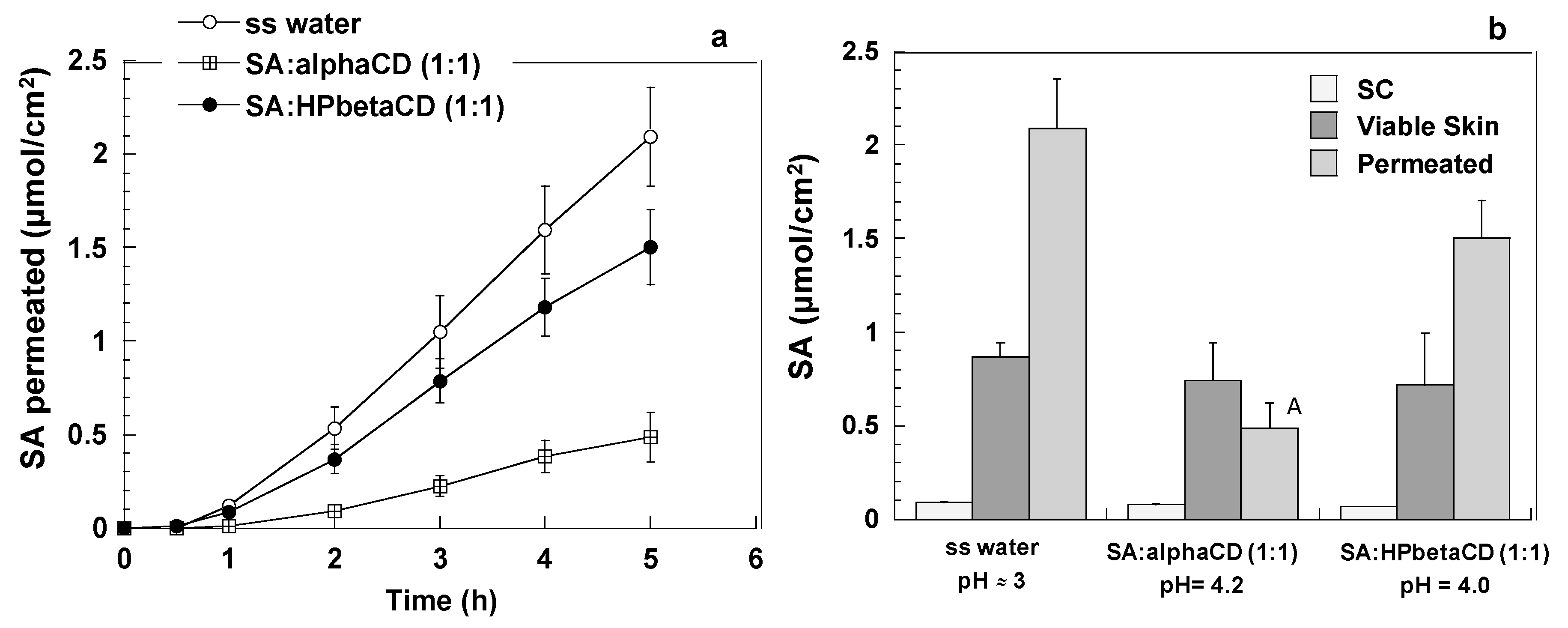
2.4. Skin Retention and Permeation Experiments
2.5. Validation of the Extraction Procedure
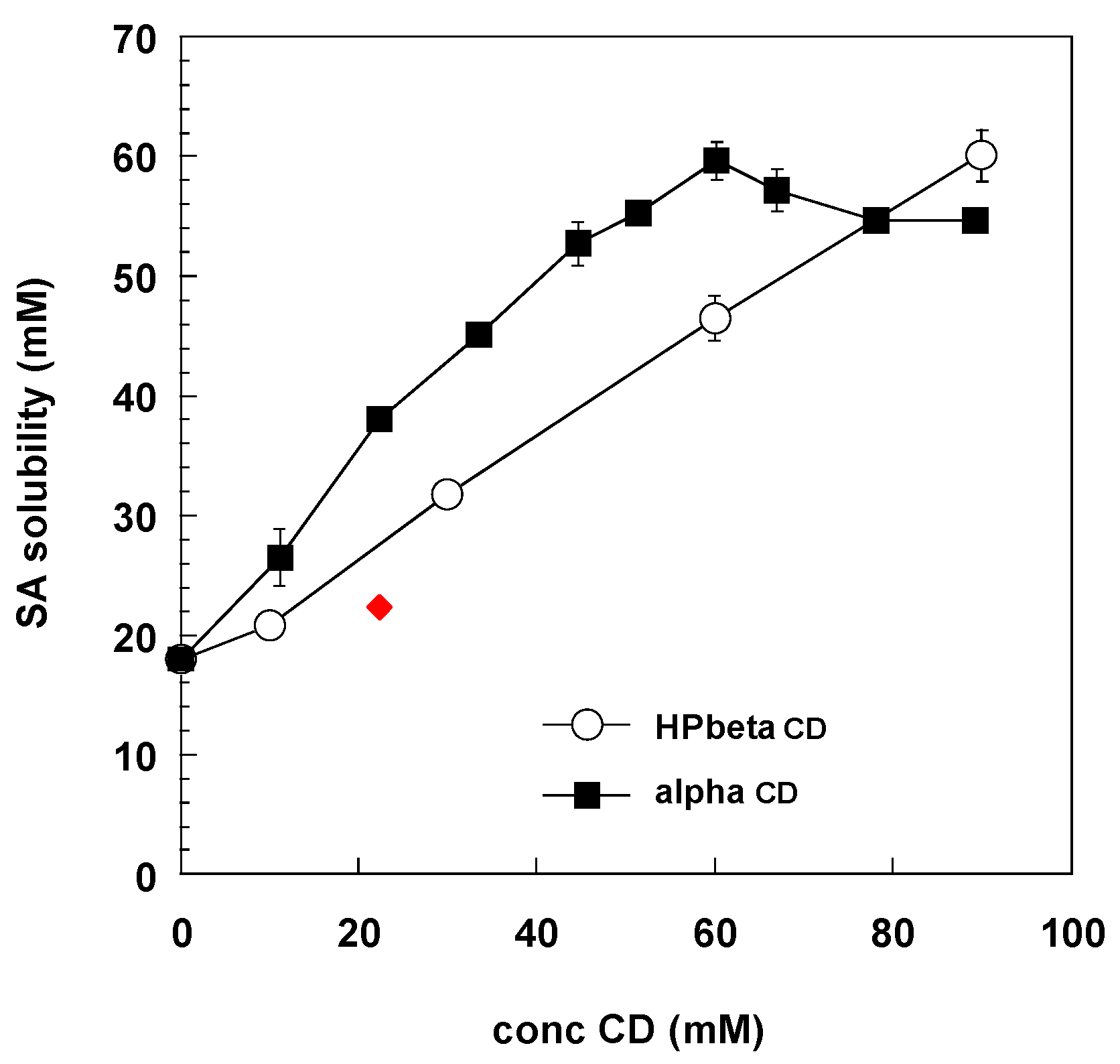
2.6. Solubility and Phase Solubility Diagram
2.7. Statistical Analysis
3. Results and Discussion
3.1. Basic Studies
3.1.1. Partition Coefficient
3.1.2. Solubility
3.1.3. Validation of Skin Extraction
3.2. Transdermal Penetration and Skin Retention of Ethylsorbate and Sorbic Acid from Solutions
3.2.1. Ethylsorbate (ES)
3.2.2. Sorbic Acid (SA)
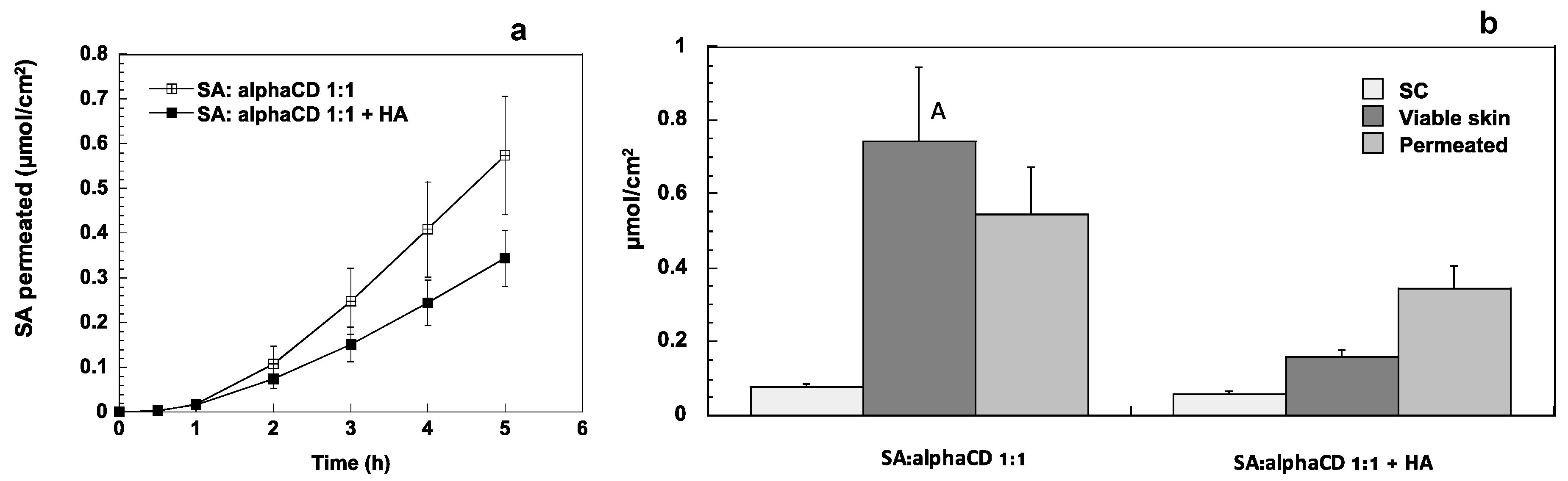
3.3. Formulation of Sorbic Acid: AlphaCD Complex in an After Sun Gel
3.4. Relevance of the SA Skin Concentration Obtained
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schadendorf, D.; Fisher, D.E.; Garbe, C.; Gershenwald, J.E.; Grob, J.J.; Halpern, A.; Herlyn, M.; Marchetti, M.A.; McArthur, G.; Ribas, A.; et al. Melanoma. Nat. Rev. Dis. Primers 2015, 1, 15003. [Google Scholar] [CrossRef] [PubMed]
- Ultraviolet Radiation and the INTERSUN Programme, FAQ Relating to Skin Cancers. Available online: https://www.who.int/uv/faq/skincancer/en/index1.html (accessed on 28 January 2019).
- Premi, S.; Wallisch, S.; Mano, C.M.; Weiner, A.B.; Bacchiocchi, A.; Wakamatsu, K.; Bechara, E.J.; Halaban, R.; Douki, T.; Brash, D.E. Photochemistry. Chemiexcitation of melanin derivatives induces DNA photoproducts long after UV exposure. Science 2015, 347, 842–847. [Google Scholar] [CrossRef] [PubMed]
- Delinasios, G.J.; Karbaschi, M.; Cooke, M.S.; Young, A.R. Vitamin E inhibits the UVAI induction of “light” and “dark” cyclobutane pyrimidine dimers, and oxidatively generated DNA damage, in keratinocytes. Sci. Rep. 2018, 8, 423. [Google Scholar] [CrossRef] [PubMed]
- Loftsson, T.; Vogensen, S.B.; Brewster, M.E.; Konradsdottir, F. Effects of cyclodextrins on drug delivery through biological membranes. J. Pharm. Sci. 2007, 96, 2532–2546. [Google Scholar] [CrossRef] [PubMed]
- CFR-Code of Federal Regulation, Title 21: Food and Drug, Part 172: Food Additives Permitted for Direct Addition to Food for Human Consumption. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?CFRPart=172showFR=1 (accessed on 28 January 2019).
- Surber, C.; Wilhelm, K.P.; Hori, M.; Maibach, H.I.; Guy, R.H. Optimization of topical therapy: Partitioning of drugs into stratum corneum. Pharm. Res. 1990, 7, 1320–1324. [Google Scholar] [CrossRef] [PubMed]
- Hansch, C.; Leo, A.; Hoekman, D. Exploring QSAR—Hydrophobic, Electronic, and Steric Constants; ACS: Washington, DC, USA, 1995. [Google Scholar]
- Vaughan, C.D. Using solubility parameters in cosmetics formulation. J. Soc. Cosmet. Chem. 1985, 36, 319–333. [Google Scholar]
- Sloan, K.B.; Koch, S.A.M.; Siver, K.G.; Flowers, F.P. Use of Solubility Parameters of Drug and Vehicle to Predict Flux Through Skin. J. Investig. Dermatol. 1986, 87, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Yalkowsky, S.H.; Dannenfelser, R.M. Aquasol Database of Aqueous Solubility; College of Pharmacy, University of Arizona: Tucson, AZ, USA, 1992. [Google Scholar]
- Higuchi, T.; Connors, K.A. Phase-Solubility Techniques. Adv. Anal. Chem. Instr. 1965, 4, 117–122. [Google Scholar]
- Kuo, K.L.; Hsieh, Y.Z. Determination of preservatives in food products by cyclodextrin modified capillary electrophoresis with multiwavelength detection. J. Chromatogr. A 1997, 768, 334–341. [Google Scholar] [CrossRef]
- Velosa, A.C.; Baader, W.J.; Stevani, C.V.; Mano, C.M.; Bechara, E.J. 1,3-diene probes for detection of triplet carbonyls in biological systems. Chem. Res. Toxicol. 2007, 20, 1162–1169. [Google Scholar] [CrossRef] [PubMed]
- Regulation (EC) No 1223/2009 of the European Parliament and of the Council. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32009R1223 (accessed on 28 January 2019).
- Malpede, A. Ingredienti Cosmetici: Le Classi Chimiche, Fisiche e Funzionali. In Manuale del Cosmetologo; D’Agostinis, G., Mignini, E., Eds.; Tecniche Nuove: Milano, Italy, 2015; pp. 7–136. [Google Scholar]
- Herman, A. Antimicrobial Ingredients as Preservative Booster and Components of Self-Preserving Cosmetic Products. Curr. Microbiol. 2018, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kocevar Glavac, N.; Lunder, M. Preservative efficacy of selected antimicrobials of natural origin in a cosmetic emulsion. Int. J. Cosmet. Sci. 2018, 40, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Saviuc, C.; Ciubuca, B.; Dinca, G.; Bleotu, C.; Drumea, V.; Chifiriuc, M.C.; Popa, M.; Pircalabioru, G.G.; Marutescu, L.; Lazar, V. Development and Sequential Analysis of a New Multi-Agent, Anti-Acne Formulation Based on Plant-Derived Antimicrobial and Anti-Inflammatory Compounds. Int. J. Mol. Sci. 2017, 18, 175. [Google Scholar] [CrossRef] [PubMed]
- Milani, M.; Sparavigna, A. The 24-hour skin hydration and barrier function effects of a hyaluronic 1%, glycerin 5%, and Centella asiatica stem cells extract moisturizing fluid: An intra-subject, randomized, assessor-blinded study. Clin. Cosmet. Investig. Dermatol. 2017, 10, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Ferrillo, M.; Vastarella, M.; Cantelli, M.; Mazzella, C.; Fabbrocini, G. Instrumental, clinical and subjective evaluation of the efficacy of a cosmetic treatment for home use. J. Cosmet. Laser Ther. 2018, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.B.; Jones, S.A. Hyaluronic acid: A unique topical vehicle for the localized delivery of drugs to the skin. J. Eur. Acad. Dermatol. Venereol. 2005, 19, 308–318. [Google Scholar] [CrossRef] [PubMed]
- Trommer, H.; Wartewig, S.; Bottcher, R.; Poppl, A.; Hoentsch, J.; Ozegowski, J.H.; Neubert, R.H. The effects of hyaluronan and its fragments on lipid models exposed to UV irradiation. Int. J. Pharm. 2003, 254, 223–234. [Google Scholar] [CrossRef]
| Compound | Concentration | Cyclodextrin | HA | pH |
|---|---|---|---|---|
| ES | 100% | - | - | n.d. |
| 0.6 mg/mL | - | - | n.d. | |
| 2.5 mg/mL | Alpha CD (1:1) 2 | - | n.d. | |
| SA | 2 mg/mL 1 | - | - | 2.7–3.0 |
| 2.5 mg/mL | Alpha CD (1:1) 2 | - | 4.2 | |
| 2.5 mg/mL | HPbeta CD (1:1) 2 | - | 4.0 | |
| 2.5 mg/mL | Alpha CD (1:1) 2 | 1.5% | 4.0 |
| MW | Melting Point (°C) | Water Solubility (mg/mL) | LogK OCT/Saline | LogK IPM/Saline | |
|---|---|---|---|---|---|
| Sorbic Acid | 112 | 135 | 2.00 ± 0.10 | 0.56 ± 0.13 | 0.31 ± 0.02 |
| Ethyl Sorbate | 140 | 0 | 1.40 ± 0.30 1 | 2.39 ± 0.01 | 2.38 ± 0.03 |
| Matrix | Extraction Mixture (v:v) | Volume (mL) | T °C | Contact Time | % Recovery SA |
|---|---|---|---|---|---|
| SC | MeOH:PBS 70:30 | 3 | 25 stirring | 2 h | 96.8 ± 3.2 |
| Stripped skin | MeOH:PBS 50:50 | 2 | 25 | overnight | 93.5 ± 1.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Padula, C.; Pescina, S.; Grolli Lucca, L.; Demurtas, A.; Santi, P.; Nicoli, S. Skin Retention of Sorbates from an After Sun Formulation for a Broad Photoprotection. Cosmetics 2019, 6, 14. https://doi.org/10.3390/cosmetics6010014
Padula C, Pescina S, Grolli Lucca L, Demurtas A, Santi P, Nicoli S. Skin Retention of Sorbates from an After Sun Formulation for a Broad Photoprotection. Cosmetics. 2019; 6(1):14. https://doi.org/10.3390/cosmetics6010014
Chicago/Turabian StylePadula, Cristina, Silvia Pescina, Leticia Grolli Lucca, Anna Demurtas, Patrizia Santi, and Sara Nicoli. 2019. "Skin Retention of Sorbates from an After Sun Formulation for a Broad Photoprotection" Cosmetics 6, no. 1: 14. https://doi.org/10.3390/cosmetics6010014
APA StylePadula, C., Pescina, S., Grolli Lucca, L., Demurtas, A., Santi, P., & Nicoli, S. (2019). Skin Retention of Sorbates from an After Sun Formulation for a Broad Photoprotection. Cosmetics, 6(1), 14. https://doi.org/10.3390/cosmetics6010014







