Hair Care Cosmetics: From Traditional Shampoo to Solid Clay and Herbal Shampoo, A Review
Abstract
1. Introduction
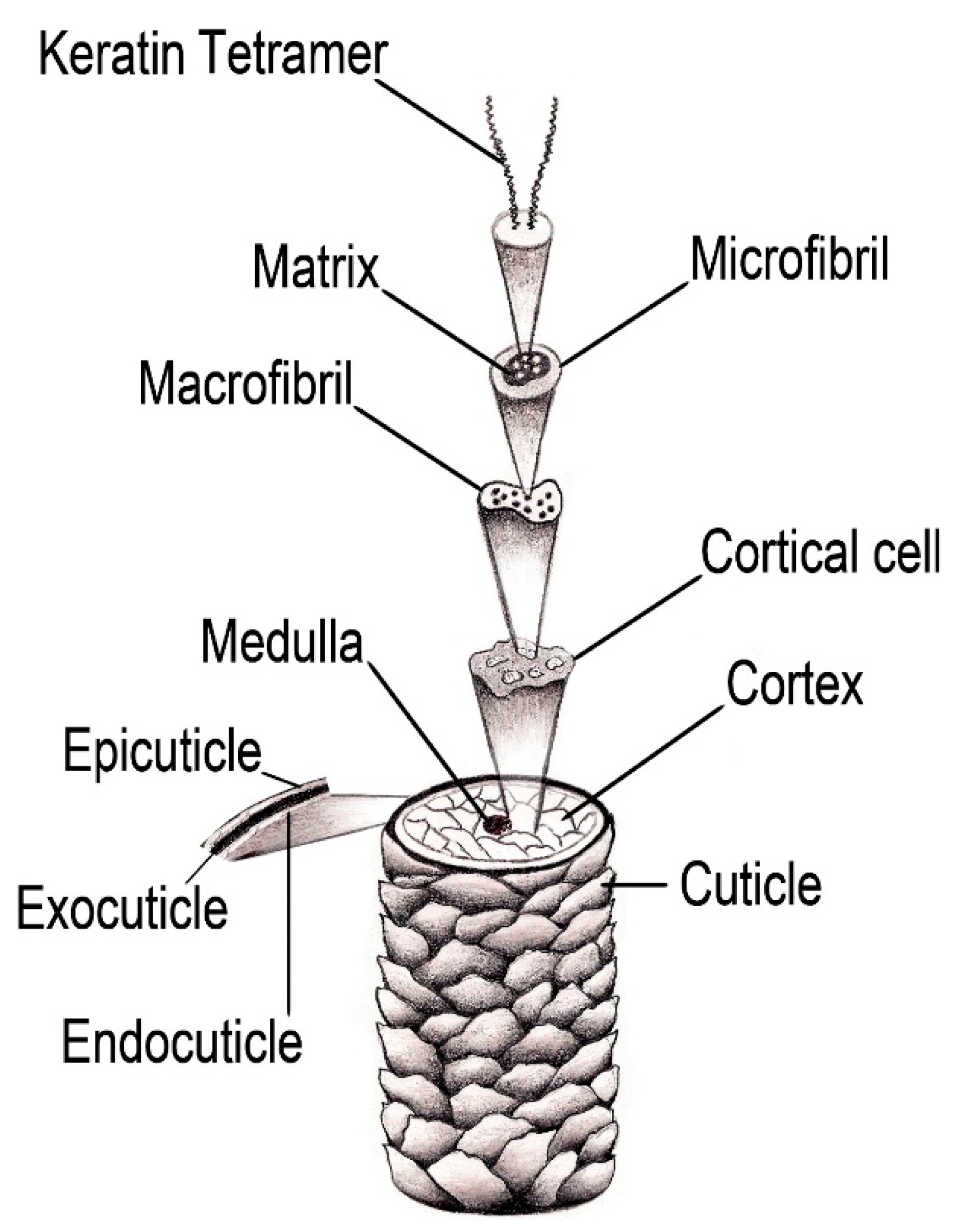
2. Hair
2.1. Hair Structure
2.2. Hair Care
3. Shampoos
3.1. Traditional Shampoos
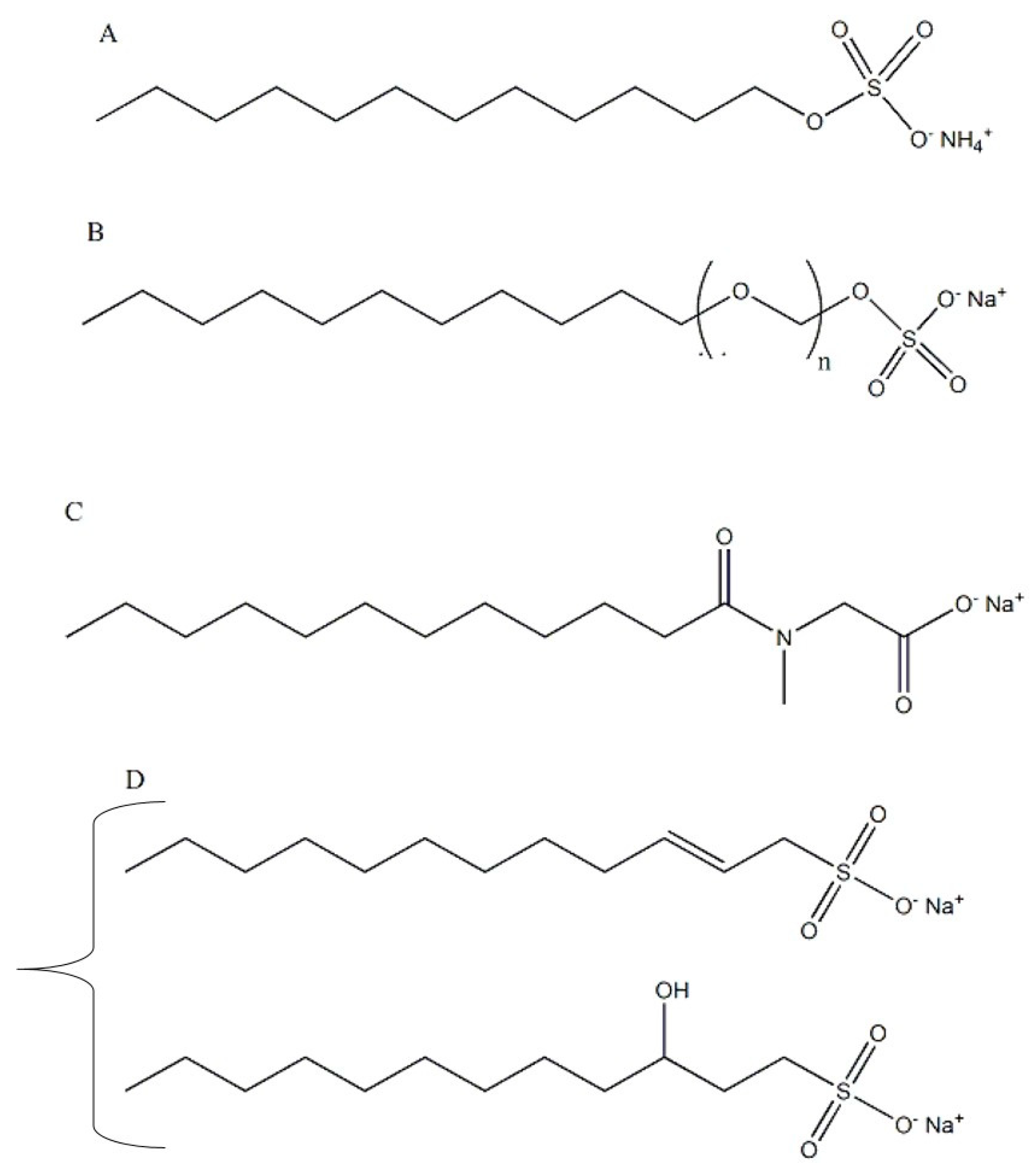
- Anionic surfactants are characterized by a negatively-charged hydrophilic polar group. Among them, ammonium lauryl sulfate, sodium laureth sulfate, sodium lauroyl sarcosinate, SDS, alpha-olefin sulfonate and ammonium laureth sulfate can be cited (Figure 2). Anionic surfactants are very good in removing sebum and dirt; however, they are strong cleaners and may induce an increase of the electrical negative charges on the hair surface, creating frizz and friction. Therefore, in order to reduce damages and to have a mild detergency, secondary surfactants such as nonionic and amphoteric surfactants are usually added in the formulation [4].
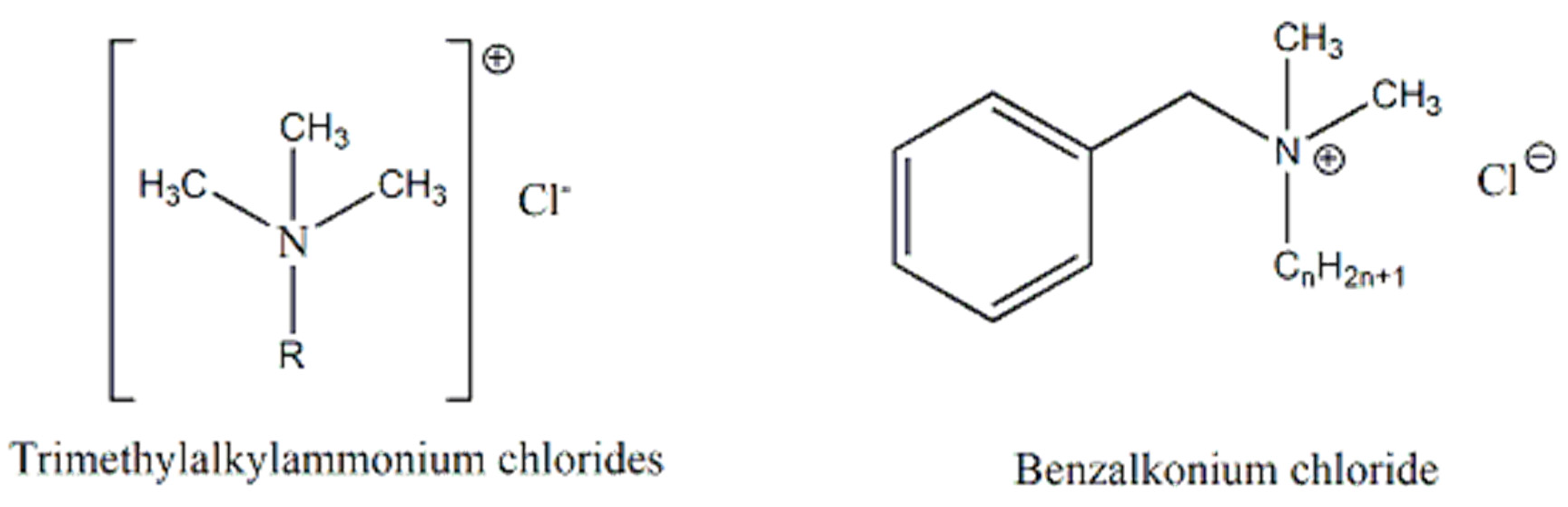
- Cationic surfactants have a positively charged hydrophilic end are used mainly as conditioners, being able to balance hair negative charges after washing, reducing frizz. Indeed, they are efficient softeners and substantive for hair because of the hair’s low isoelectric point (pH 2.15–3.17) [14]. Typical examples are trimethylalkylammonium chlorides, benzalkonium chloride or bromide, and so on (Figure 3) [4]. Due to their chemical nature, they also have bacteriostatic properties [4].
- Amphoteric surfactants are characterized by the charge control through the solution pH values. In other words, they are anionic or cationic surfactants in alkaline or acid solutions, respectively. They are considered very mild and thus dermatologically compatible. Furthermore, they have a good foaming, detergent and wetting properties. For these reasons, they are used to reduce the aggressiveness of anionic surfactants. Two types of amphoteric compounds can be mentioned: alkyl iminodipropionate and (amido) betaines (Figure 3) [4].
- Nonionic surfactants not exhibit electric charge in aqueous solutions due to the absence of hydrophilic dissociable groups. They are less aggressive than other surfactants and, thanks to their eudermic properties, are widely used as emulsifiers and solubilizers in cosmetic formulations. They are often derived from plants, such as glucosides, citrates, sulfosuccinates and hydrolysates proteins. Alcohols, having long chains, exhibit some surfactant properties. Among these, the fatty alcohols, cetyl alcohol, stearyl alcohol, and cetostearyl alcohol (consisting predominantly of cetyl and stearyl alcohols), and oleyl alcohol are very important (Figure 3) [4].
3.2. Non-Traditional Shampoos
3.2.1. Herbal Shampoo
3.2.2. Solid Shampoos
Clay Minerals as Natural Ingredients
General Uses of Clays
Clay Minerals and Clays
Chemical Composition and Impurities
Clay Minerals in Cosmetic and Health Care
- Clay minerals can be topically applied as dermatological protectors or for cosmetic reasons [29]. Creams, powders, emulsions used as cosmetic products, applied on the external parts of the body, embellish, modify physical appearance, and/or preserve the physico-chemical skin conditions. For example, as reported by Carretero et al. [26], clays with a high sorption capacity are present in creams, powders, emulsions for giving opacity, removing shine, and covering physical skin imperfections. Moreover, the ability of adhering to skin forming a protective film makes these materials able to absorb the excess sebum. For this purpose, the phyllosilicates group (such as palygorskite and sepiolite, in liquid preparations, kaolinite, smectites, and talc) is considered the most suitable [26]. In the case of pharmaceutical applications as dermatological protectors, clays are generally used in the form of powders, creams and ointments to protect skin against external agents, and also in the case of exudations and liquid excretions. The most commonly used clays are kaolinite, talc and smectites, due to their high absorbent power [26,29]. Indeed, these minerals are capable of forming a film by adhering to skin and protecting it against external physical or chemical agents [29]. By absorbing the skin’s secretions, they also have a refreshing action, producing a large surface for the evaporation, together with a gentle antiseptic action thanks to a water-poor environment, unfavorable for the development of bacteria. In this context, fibrous minerals (palygorskite and sepiolite) were considered the most suitable materials, but since there are doubts existing concerning the possible carcinogenic effect of palygorskite, if inhaled, their use as a dermatological protector is not desirable [26]. However, some authors report that these minerals are neither toxic nor dangerous [29].
- As functional ingredients in hair care cosmetics and in skin care mask formulations, clay minerals are used due to their high absorbency level of substances such as greases, toxins, etc.) [29]. Therefore, they are recommended for treating cutaneous inflammatory processes, such as seborrhoeic dermatitis, psoriasis, chronic eczemas or acne [26,29]. In particular, in the case of hair care applications, the addition of sulfur-containing minerals in shampoo formulations is considered an effective remedy against dandruff and seborrhoea [26].
- Clays and clay minerals are potential candidates as natural UV-protection agents in sunscreen formulations through mechanisms of absorption or reflection of UV radiation. Hoang-Minh et al. [30] studied the protective role of kaolin, smectite, mixed-layer series-dominated clay and mica-dominated clay against ultraviolet (UV) radiation, in the range 250–400 nm, due to the bulk Fe2O3 content that lowers the UV-transmission level. Additionally, the UV-protection depends on the expandability of the clay or the combination of clay mineral with mixed ointment [30]. Besides the direct use of clays as sunscreen, clays and clay minerals being used as delivery systems in cosmetic products, in order to improve the stability of an organic sunscreen like PABA (p-amino benzoic acid, an UV-B absorber in the range 200–313 nm), these new systems also open a novel horizon in the use of clays in this field as a promising, good, efficient way to protect other chemical filters. Moreover, as reported by Perioli et al. [31], these formulations allow a very low sunscreen release avoiding the close contact between skin and filter preventing at least cutaneous reactions and allergy problems.
- A clear distinction must be made between “healing clays” and those we have identified as “antibacterial clays”. The antiseptic and disinfectant activity of these minerals is mainly due to a high astringent capacity, largely dependent on their concentration. It is worth underlining that high concentrations of these materials can be considered highly toxic to organisms. For this reason, their continuous application over large skin areas or over non-intact skin should be avoided [26]. While clays may heal various illnesses by means of their unique physical properties (e.g., high absorbance, high surface area, high heat capacity, high exchange capacity, etc.), only a few natural clays that kill pathogenic bacteria are reported. For example, among the healing clays, the researcher’s attention was drawn to a clinical use of French green clay (rich in Fe-smectite) for healing Buruli ulcers, a necrotizing fasciitis caused by Mycobacterium ulcers [32]. On the other hand, white, gray and yellow clays have an antibacterial effect against Staphylococcus aureus (bacterial viability, colony-forming unit, CFU, of around 0 after 24 h) but have no effect against Pseudomonas aeruginosa exhibiting bacterial viability CFU from 80 to 100. Instead, under the same condition, pink clay explained an antibacterial effect against Pseudomonas aeruginosa (bacterial viability CFU of around 0) and has a low effect against Staphylococcus aureus exhibiting bacterial viability CFU of around 40 [33]. In spite of the use of geological mineral clay to heal skin bacterial infections having been evident since ancient times [33], the antibacterial process displayed by the identified clays is not yet well known [32]. Therefore, studies should be the focus in this direction since we are in the era of bacteria developing antibiotic resistance to existing pharmacological agents, and the discovery of new antibacterial agents, such as natural clay minerals, against pathogenic bacteria, could bring peculiar and great advantages [32]. For example, Williams et al. [32] reported that, under their conditions, the complete killing of E. coli, S. typhimurium, P. aeruginosa, and M. marinum by the investigated clays was observed. Thus, their use could provide an inexpensive treatment for skin infections, especially in areas with limited access to hospitals and medical resources [32].
Clay Minerals Safety Specifications
Toxicological Evaluation of Clays
Clays Tests
Clays’ Cleaning Properties
Rhassoul Clay
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Robbins, C.R. Chemical and Physical Behavior of Human Hair, 4th ed.; Springer: New York, NY, USA, 2002. [Google Scholar]
- Arora, P.; Arun, N.; Karan, M. Shampoos based on synthetic ingredients vis-à-vis shampoos based on herbal ingredients: A review. Int. J. Pharm. Sci. Rev. Res. 2011, 7, 41–46. [Google Scholar]
- Bellare, J.; Iyer, R.; Mainkar, A.R.; Jolly, C.I. A study on the conditioning effects of natural shampoos using the scanning electron microscope. Int. J. Cosmet. Sci. 2001, 23, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Gavazzoni Dias, M.F.R. Hair Cosmetics: An Overview. Int. J. Trichol. 2015, 7, 2–15. [Google Scholar] [CrossRef] [PubMed]
- Nayak, S.B.; Ann, C.Y.; Azhar, A.B.; Ling, E.C.S.; Yen, W.H.; Aithal, P.A. A Study on Scalp Hair Health and Hair Care Practices among Malaysian Medical Students. Int. J. Trichol. 2017, 9, 58–62. [Google Scholar]
- Perfitt, R.J.; Carimbocas, C.A.R. Dry Shampoo Composition. Patent US 9801793B2, 31 October 2017. [Google Scholar]
- Constantine, M.J.; Krysztal, S. Solid Shampoo Composition in Compact Needle Form with Water as a Binder. Patent U.S. 4,996,006, 26 February 1991. [Google Scholar]
- Naizet, S. Dess de Cosmetologie. Monographie. Les c Heveux Gras. Diplôme D’études Supérieures Spécialisées en Cosmétologie; Université du Québec à Chicoutimi: Chicoutimi, QC, Canade, 2016. [Google Scholar]
- Hordinsky, M.; Avancini Caramori, A.P.; Donovan, J.C. Hair Physiology and Grooming. In Cosmetic Dermatology: Products and Procedures; Draelos, Z.D., Ed.; Wiley-Blackwell: Chichester, West Sussex, UK, 2016; Chapter 29; pp. 222–226. [Google Scholar]
- Zhang, Y.; Alsop, R.J.; Soomro, A.; Yang, F.C.; Rheinstädter, M.C. Effect of shampoo, conditioner and permanent waving on the molecular structure of human hair. PeerJ 2015, 3, e1296. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, R.D. Healthy hair: What is it? J. Investig. Dermatol. Symp. Proc. 2007, 12, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Jaya Preethi, P.; Padmini, K.; Srikanth, J.; Lohita, M.; Swetha, K.; Vengal Rao, P. A Review on Herbal Shampoo and Its Evaluation. Asian J. Pharm. Anal. 2013, 3, 153–156. [Google Scholar]
- Surupsing, M.V.; Akash, D.P.; Harishchandra, M.Y.; Vipul, H.J.; Pawar, S.P. Formulation and Evaluation of Herbal Shampoo Powder. Int. J. Pharm. Chem. Res. 2017, 3, 492–498. [Google Scholar]
- Parreira, H.C. On the isoelectric point of human hair. J. Colloid Interface Sci. 1980, 75, 212–217. [Google Scholar] [CrossRef]
- Bovero, A. Dermocosmetologia. Dall’inestetismo al Trattamento Cosmetico; Tecniche Nuove: Milano, Italy, 2011. [Google Scholar]
- Vijetha, J.R.; Grace, X.F.; Shanmuganathan, S.; Chamundeeswari, D. Preparation and Evaluation of Polyherbal Shampoo Powder. Int. J. Pharm. Biol. Sci. 2013, 3, 60–66. [Google Scholar]
- Al Badi, K.; Khan, S.A. Formulation, evaluation and comparison of the herbal shampoo with the commercial shampoos. Beni-Suef Univ. J. Basic Appl. Sci. 2014, 3, 301–305. [Google Scholar] [CrossRef]
- Middha, S.K.; Goyal, A.K.; Lokesh, P.; Yardi, V.; Mojamdar, L.; Keni, D.S.; Babu, D.; Usha, T. Toxicological Evaluation of Emblica officinalis Fruit Extract and its Anti-inflammatory and Free Radical Scavenging Properties. Pharm. Mag. 2015, 11, S427–S433. [Google Scholar]
- Sandhyarani, G.; Ramesh, A.; Balaji, B. A review on shampoos. Acta Biomed. Sci. 2014, 1, 61–64. [Google Scholar]
- Chen, Y.F.; Yang, C.H.; Chang, M.S.; Ciou, Y.P.; Foam Huang, Y.C. Foam properties and detergent abilities of the saponins from Camellia oleifera. Int. J. Mol. Sci. 2010, 11, 4417–4425. [Google Scholar] [CrossRef]
- Utane, R.; Deo, S.; Itankar, P. Preparation of herbal shampoo (HS) by green method and their characterization. Int. J. Res. Soc. Sci. Inf. Stud. 2017, 5, 254–258. [Google Scholar]
- Dubey, S.; Nema, N.K.; Nayak, S. Preparation and Evaluation of Herbal Shampoo Powder. Anc. Sci. Life 2004, 24, 38–44. [Google Scholar]
- Neame, E. Dry Shampoo Composition for Hair. Patent WO2015071631A1, 21 May 2015. [Google Scholar]
- Carretero, M.I.; Gomes, C.S.F.; Tateo, F. Clays and human health. In Handbook of Clay Science. Developments in Clay Science; Bergaya, F., Theng, B.K.G., Lagaly, G., Eds.; Elsevier Ltd.: Amsterdam, The Netherlands, 2006; Volume 1, pp. 717–741. [Google Scholar]
- Viseras, C.; Aguzzi, C.; Cerezo, P.; Lopez-Galindo, A. Uses of clay minerals in semisolid health care and therapeutic products. Appl. Clay Sci. 2007, 36, 37–50. [Google Scholar] [CrossRef]
- Carretero, M.I.; Pozo, M. Clay and non-clay minerals in the pharmaceutical and cosmetic industries Part II. Active ingredients. Appl. Clay Sci. 2010, 47, 171–181. [Google Scholar] [CrossRef]
- Roselli, C.; Desideri, D.; Cantaluppi, C.; Mattioli, M.; Fasson, A.; Meli, M.A. Essential and toxic elements in clays for pharmaceutical and cosmetic use. J. Toxicol. Environ. Health A 2015, 78, 316–324. [Google Scholar] [CrossRef]
- Lopez-Galindo, A.; Viseras, C.; Cerezo, P. Compositional, technical and safety specifications of clays to be used as pharmaceutical and cosmetic products. Appl. Clay Sci. 2007, 36, 51–63. [Google Scholar] [CrossRef]
- Carretero, M.I. Clay minerals and their beneficial effects upon human health. A review. Appl. Clay Sci. 2002, 21, 151–163. [Google Scholar] [CrossRef]
- Hoang-Minh, T.; Le, T.T.L.; Kasbohm, J.; Gieré, R. UV-protection characteristics of some clays. Appl. Clay Sci. 2010, 48, 349–357. [Google Scholar] [CrossRef]
- Perioli, L.; Ambrogi, V.; Bertini, B.; Ricci, M.; Nocchetti, M.; Latterini, L.; Rossi, C. Anionic clays for sunscreen agent safe use: Photoprotection, photostability and prevention of their skin penetration. Eur. J. Pharm. Biopharm. 2006, 62, 185–193. [Google Scholar] [CrossRef]
- Williams, L.B.; Haydel, S.E. Evaluation of the medicinal use of clay minerals as antibacterial agents. Int. Geol. Rev. 2010, 52, 745–770. [Google Scholar] [CrossRef]
- Lafi, S.A.; Al-Dulaimy, M.R. Antibacterial Effect of some Mineral Clays In Vitro. Egypt. Acad. J. Biol. Sci. 2011, 3, 75–81. [Google Scholar] [CrossRef]
- Tokarský, J. Ghassoul—Moroccan clay with excellent adsorption properties. Mater. Today Proc. 2018, 5, S78–S87. [Google Scholar] [CrossRef]
- El Fadeli, S.; Pineau, A.; Lekouch, N.; Sedki, A. Analysis of traditional pharmacopeia product from Morocco-Rhassoul. Anal. Chem. Indian J. 2011, 10, 60–61. [Google Scholar]
- Mattioli, M.; Giardini, L.; Roselli, C.; Desideri, D. Mineralogical characterization of commercial clays used in cosmetics and possible risk for health. Appl. Clay Sci. 2016, 119, 449–454. [Google Scholar] [CrossRef]
- Williams, G.L. Hair Care Composition. Patent US9107839B1, 18 August 2014. [Google Scholar]
- Benhammou, A.; Tanouti, B.; Nibou, L.; Yaacoubi, A.; Bonnet, J.-P. Mineralogical and physicochemical investigation of mg-smectite from jbel ghassoul, Morocco. Clays Clay Miner. 2009, 57, 264–270. [Google Scholar] [CrossRef]
- Rhouta, B.; Kaddami, H.; Elbarqy, J.; Amjoud, M.; Daoudi, L.; Maury, F.; Senocq, F.; Maazouz, A.; Gerard, J.-F. Elucidating the crystal-chemistry of Jbel Rhassoul stevensite (Morocco) by advanced analytical techniques. Clay Miner. 2008, 43, 393–404. [Google Scholar] [CrossRef]
| Evaluation Parameters of Herbal Powder Shampoos | |
|---|---|
| Organoleptic Evaluation | Color |
| Odor | |
| Taste | |
| Texture | |
| Particle Size | |
| General Powder Characters | Angle of Repose |
| Bulk Density | |
| Tapped Density | |
| Packaging Criteria | |
| Physicochemical Parameters (Evaluation) | Ash value |
| Total ash | |
| Acid insoluble ash | |
| Moisture content | |
| pH | |
| Cleaning action | |
| Foaming index (capacity) | |
| Dirt dispersion | |
| Wetting time | |
| Solubility | |
| Loss on drying | |
| Swelling index | |
| Nature of hair after wash | |
| Evaluation Parameters of Commercial Shampoos | |
| Physical appearance/visual inspection | Clarity |
| Determination of pH | |
| Determination % of solids contents | |
| Surface tension measurements | |
| Rheological evaluation | |
| Dirt dispersion | |
| Cleaning action | |
| Detergency ability | |
| Wetting time | |
| Foaming ability and foam stability | |
| Conditioning performance | |
| Stability study | |
| Eye irritation test | |
| Skin sensitization test | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gubitosa, J.; Rizzi, V.; Fini, P.; Cosma, P. Hair Care Cosmetics: From Traditional Shampoo to Solid Clay and Herbal Shampoo, A Review. Cosmetics 2019, 6, 13. https://doi.org/10.3390/cosmetics6010013
Gubitosa J, Rizzi V, Fini P, Cosma P. Hair Care Cosmetics: From Traditional Shampoo to Solid Clay and Herbal Shampoo, A Review. Cosmetics. 2019; 6(1):13. https://doi.org/10.3390/cosmetics6010013
Chicago/Turabian StyleGubitosa, Jennifer, Vito Rizzi, Paola Fini, and Pinalysa Cosma. 2019. "Hair Care Cosmetics: From Traditional Shampoo to Solid Clay and Herbal Shampoo, A Review" Cosmetics 6, no. 1: 13. https://doi.org/10.3390/cosmetics6010013
APA StyleGubitosa, J., Rizzi, V., Fini, P., & Cosma, P. (2019). Hair Care Cosmetics: From Traditional Shampoo to Solid Clay and Herbal Shampoo, A Review. Cosmetics, 6(1), 13. https://doi.org/10.3390/cosmetics6010013








