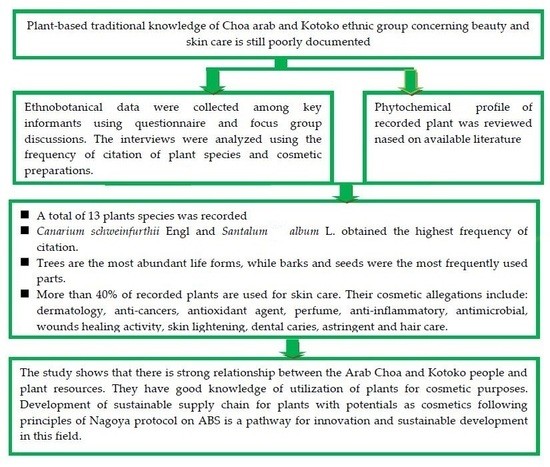
Herbal Cosmetics Knowledge of Arab-Choa and Kotoko Ethnic Groups in the Semi-Arid Areas of Far North Cameroon: Ethnobotanical Assessment and Phytochemical Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Description of the Study Site
2.2. Research Method
3. Results
3.1. Plants Used As Traditional Cosmetics
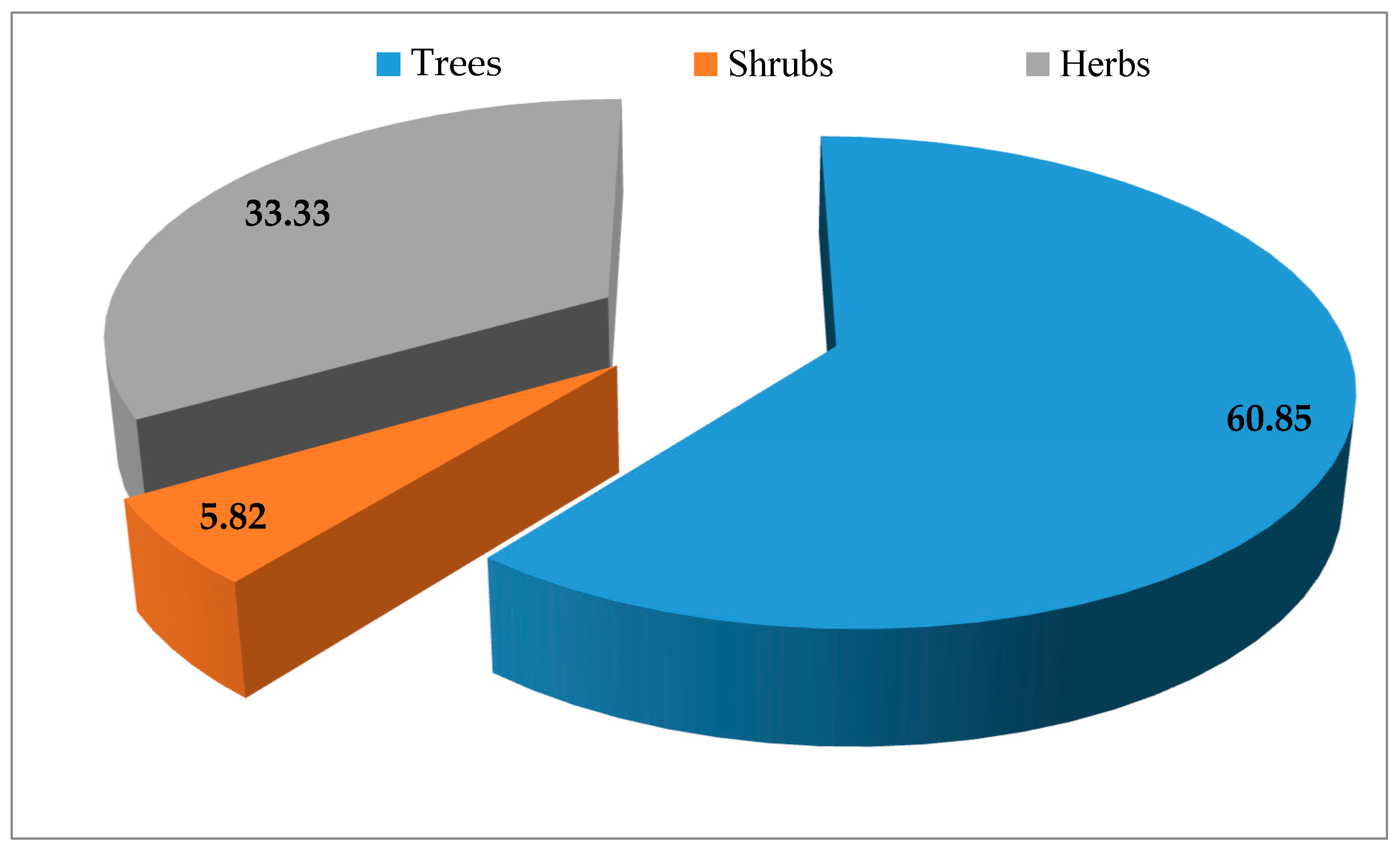
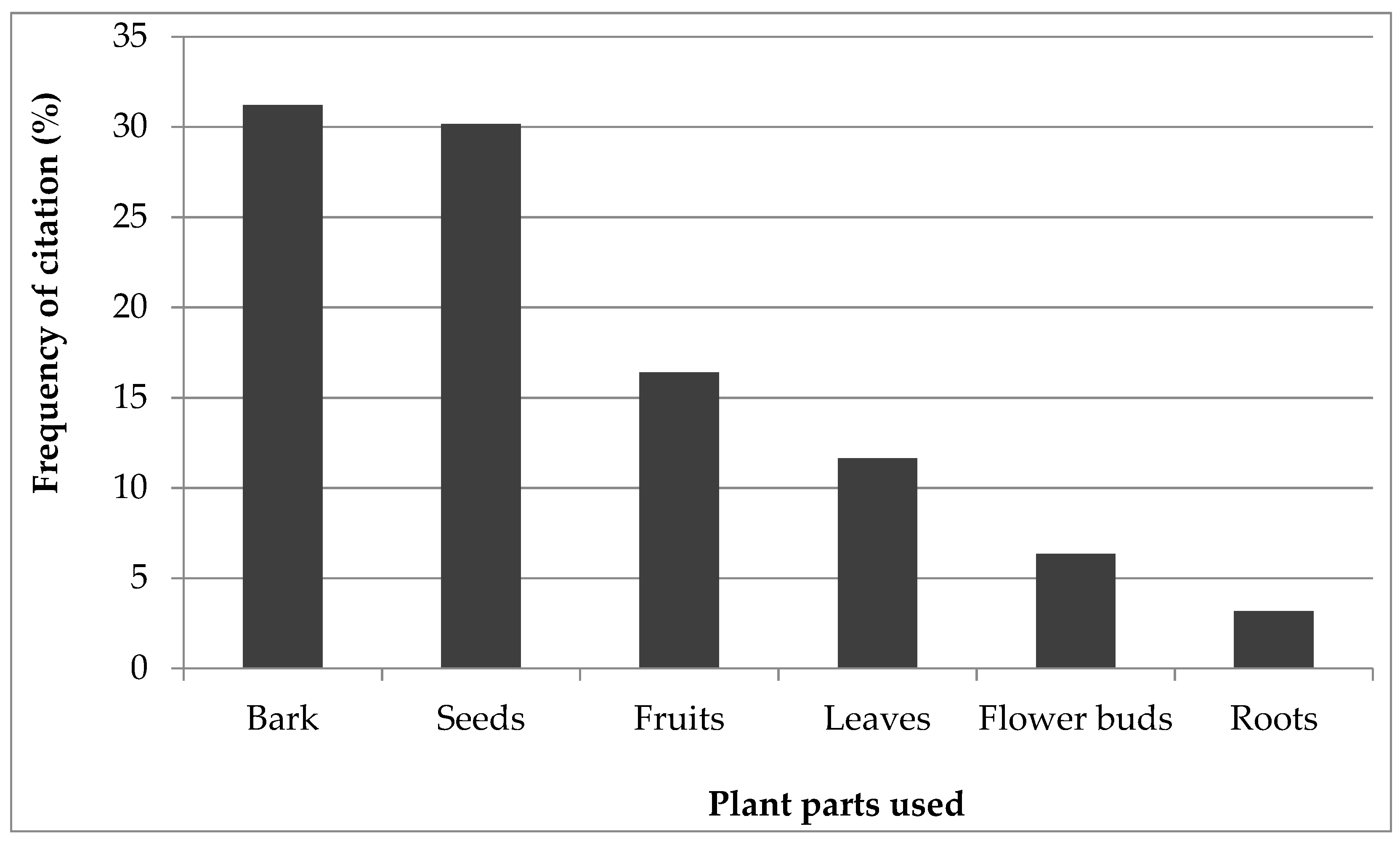
3.2. Biological Forms and Part Used
3.3. Cosmetic Use of the Recorded Plants
3.4. Phytochemical Review
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Olson, D.M.; Dinerstein, E.; Wikramanayake, E.D.; Burgess, N.D.; Powell, G.V.N.; Underwood, E.C.; D’Amico, J.A.; Itoua, I.; Strand, H.E.; Morrison, J.C.; et al. Terrestrial ecoregions of the world: A new map of life on Earth. BioScience 2001, 51, 933–938. [Google Scholar] [CrossRef]
- Thornthwaite, C.W. An approach toward a rational classification of climate. Geogr. Rev. 1948, 38, 55–94. [Google Scholar] [CrossRef]
- Wessels, K.J. Dryland Systems, Ecosystems and Human Well-Being: Current State and Trends; Island Press: Washington, DC, USA, 2006; Chapter 22; pp. 623–662. [Google Scholar]
- Maestre, F.T.; Salguero-Gómez, R.; Quero, J.L. It is getting hotter in here: Determining and projecting the impacts of global environmental change on drylands. Philos. Trans. R. Soc. Biol. Sci. 2012, 367, 3062–3075. [Google Scholar] [CrossRef] [PubMed]
- Rands, M.R.; Adams, W.M.; Bennun, L.; Butchart, S.H.; Clements, A.; Coomes, D.; Entwistle, A.; Hodge, I.; Kapos, V.; Scharlemann, J.P.; et al. Biodiversity Conservation: Challenges Beyond 2010. Science 2010, 329, 1298–1303. [Google Scholar] [CrossRef] [PubMed]
- European Parliament and Council of the European Union. Regulation (EC) N° 1223/2009 of the European Parliament and of the Council of 30 November 2009 on cosmetic products (recast). Off. J. Eur. Union 2009, L342/59, 151. [Google Scholar]
- Hetta, M.H. Phytocosmetics in Africa. Int. J. Phytocosmet. Nat. Ingred. 2016, 3, 1. [Google Scholar] [CrossRef]
- Mahomoodally, F.M.; Ramjuttun, P. Phytocosmetics from the African Herbal Pharmacopeia. Int. J. Phytocosmet. Nat. Ingred. 2017, 4, 4. [Google Scholar] [CrossRef]
- Oumeish, Y. The cultural and philosophical concepts of cosmetics in beauty and art through the medical history of mankind. Clin Dermatol. 2001, 19, 375–386. [Google Scholar] [CrossRef]
- DeLancey, M.D.; Mbuh, R.N.; DeLancey, M.W. Historical Dictionaries of Africa; No. 113; The Scarecrow Press, Inc.: Lanham, MD, USA; Toronto, ON, Canada; Plymouth, UK, 2010; 531p. [Google Scholar]
- Todou, G.; Froumsia, M.; Souaré, K.; Nnanga, J.F. Woody plants diversity and type of vegetation in non cultivated plain of Moutourwa, Far North, Cameroon. J. Agric. Environ. Int. Dev. 2016, 110, 217–227. [Google Scholar]
- Haiwa, G.; Tchobsala; Ngakou, A. Ecological characterization of the Vegetation in the Sudano-Sahelian Zone of Cameroon. Acad. J. Biosci. 2017, 5, 164–173. [Google Scholar]
- Mapongmetsem, P.M.; Kapchie, V.N.; Tefempa, B.H. Diversity of local fruit trees and their contribution in sustaining the rural livelihood in the northern Cameroon. Ethiop. J. Environ. Stud. Manag. 2012, 5, 32–46. [Google Scholar] [CrossRef]
- Betti, J.L.; Mebere Yemefa’a, S.R. An ethnobotanical study of medicinal plants used in the Kalamaloué National Park, Cameroon. J. Med. Plants Res. 2011, 5, 1447–1458. [Google Scholar]
- Betti, J.L.; Mebere Yemefa’a, S.R.; Nchembi, F.T. Contribution to the knowledge of non wood forest products of the far north region of Cameroon: Medicinal plants sold in the Kousséri market. J. Ecol. Nat. Environ. 2011, 3, 241–254. [Google Scholar]
- UNESCO. Les Plantes Médicinales des Régions Arides; UNESCO: noy, Paris-7, France, 1960; 97p. [Google Scholar]
- Fongnzossie, E.F.; Tize, Z.; Fogang Nde, P.J.; Nyangono Biyegue, C.F.; Bouelet Ntsama, I.S.; Dibong, S.D.; Nkongmeneck, B.A. Ethnobotany and pharmacognostic perspective of plant species used as traditional cosmetics and cosmeceuticals among the Gbaya ethnic group in Eastern Cameroon. S. Afr. J. Bot. 2017, 112, 29–39. [Google Scholar] [CrossRef]
- L’Hote, Y. Climatologie et Agroclimatologie de la Province Extrême Nord (PEN) du Cameroun. Préparation de l’Atlas de la PEN; Laboratoire d’Hydrologie, 1998; 31p, Available online: http://horizon.documentation.ird.fr/exl-doc/pleins_textes/pleins_textes_6/griseli1/010014730.pdf (accessed on 2 May 2018).
- Boutrais, J.; Boulet, J.; Beauvilain, A.; Gubry, P.; Barreteau, D.; Die’j, M.; Breton, R.; Seignobos, C.; Pontie, G.; Marguerat, Y.; et al. “Le Milieu Naturel”—Le Nord du Cameroun: Des hommes, une Région; ORSTOM (Collection Memoires n°102): Paris, France, 1984; pp. 19–100. [Google Scholar]
- Arbonier, M. Arbres Arbustes et Lianes des Zones Sèches d’Afrique de l’Ouest; MNHN-Quae: Paris, France, 2009; 571p. [Google Scholar]
- Atawodi, S.E. Polyphenol composition and in vitro antioxidant potential of Nigerian Canarium schweinfurthii Engl. Oil. Adv. Biol. Res. 2010, 4, 314–322. [Google Scholar]
- Ngbolua, K.N.; Moke, L.E.; Lumande, J.K.; Mpiana, P.T. Canarium schweinfurthii Engl. (Burseraceae): An updated review and future direction for sickle cell disease. J. Adv. Med. Life Sci. 2015, 1–5. Available online: https://pdfs.semanticscholar.org/e81c/8b67699994fddda149ff106f84cbb5fedc8f.pdf?_ga=2.43720102.2019391631.1525225105-1418532351.1513041768 (accessed on 2 May 2018).
- Tcheghebe, T.O.; Seukep, A.J.; Ngouafong Tatong, F. A Review on Traditional Uses, Phytochemical Composition and Pharmacological Profile of Canarium Schweinfurthii Eng. Nat. Sci. 2016, 14, 17–22. [Google Scholar]
- Rakesh, K.S.; Upma, A.K.; Sahil, A. Santalum album Linn: A review on morphology, phytochemistry and pharmacological aspects. Int. J. PharmTech Res. 2010, 2, 914–919. [Google Scholar]
- Borade, A.S.; Kale, B.N.; SheteWagini, R.V. A phytopharmacological review on Lawsonia inermis (Linn.). Int. J. Pharm. Life Sci. 2011, 2, 536–541. [Google Scholar]
- Wagini, N.H.; Soliman, A.; Abbas, M.S.; Hanafy, Y.A.; Badawy, E.S.M. Phytochemicl analysis of Nigerian nd Egyptian henna (Lawsonia inermis L.) leaves using TLC, FTIR and GCMS. Plant 2014, 2, 27–32. [Google Scholar] [CrossRef]
- Yin, N.S.; Abdullah, Y.; Phin, C.K. Phytochemical constituents from leaves of Elaeis guineensis and their antioxidant and antimicrobial activities. Int. J. Pharm. Pharm. Sci. 2013, 5, 137–140. [Google Scholar]
- Kalyana, S.; Ravigadevi, S.; Yew-Ai, T. Palm fruit chemistry and nutrition. Asia Pac. J. Clin. Nutr. 2003, 12, 355–362. [Google Scholar]
- Sreenivasan, S.; Nilawatyi, R.; Rathinam, X.; Lachimanan Yoga, L.; Amala, R. Wound healing potential of Elaeis guineensis Jacq leaves in an infected albino rat model. Molecules 2010, 15, 3186–3199. [Google Scholar]
- Rabasco, A.A.M.; González, R.M.L. Lipids in pharmaceutical and cosmetic preparations. Grasas y Aceites 2000, 51, 74–96. [Google Scholar]
- Galgut, J.M.; Ali, S.A. Effect and mechanism of action of resveratrol: A novel melanolytic compound from the peanut skin of Arachis hypogaea. J. Recept. Signal Transduct. Res. 2011, 31, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Sim, E.W.; Lai, S.Y.; Chang, Y.P. Antioxidant capacity, nutritional and phytochemical content of peanut (Arachis hypogaea L.) shells and roots. Afr. J. Biotechnol. 2012, 11, 11547–11551. [Google Scholar]
- Prabasheela1, B.; Venkateshwari, R.; Nivetha, S.; Mohana Priya, P.; Jayashree, T.; Vimala, R.; Karthik, K. Phytochemical analysis and antioxidant activity of Arachis hypogeal. J. Chem. Pharm. Res. 2015, 7, 116–121. [Google Scholar]
- Nuwan, V.G.S.I.; Ediriweera, E.R.H.S.S.; Wasana, R.K.R. Medicinal and nutritional value of Panicum miliaceum L. (Meneri): A review. In Proceedings of the National Research Symposium: Fundamentals of Indigenous Medicine: Challenges and New Perspectives, Colombo, Sri Lanka, 22 December 2016. [Google Scholar]
- Owoyele, B.V.; Negedu, M.N.; Olaniran, S.O.; Onasanwo, S.A.; Oguntoye, S.O.; Sanya, J.O.; Oyeleke, S.A.; Ibidapo, A.J.; Soladoye, A.O. Analgesic and anti-inflammatory effects of aqueous extract of Zea mays. Husk in male Wistar rats. J. Med. Food 2010, 13, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Mann, A.; Yahaya, Y.; Banso, A.; John, F. Phytochemical and antimicrobial activity of Terminalia avicennioides extracts against some bacteria pathogens associated with patients suffering from complicated respiratory tract diseases. J. Med. Plants Res. 2008, 2, 94–97. [Google Scholar]
- Azeez, M.A.; Yekeen, T.A.; Animasaun, D.A.; Durodola, F.A.; Bello, O.B. Terminalia avicennioides as a potential candidate for pharmaceutical industry: A Review. Res. J. Pharm. Biol. Chem. Sci. 2015, 6, 748–754. [Google Scholar]
- Salatino, A.; Maria, L.; Salatino, F.; Giuseppina, N. Traditional uses, Chemistry and Pharmacology of Croton species (Euphorbiaceae). J. Braz. Chem. Soc. 2007, 18, 11–33. [Google Scholar] [CrossRef]
- Maruti, J.D.; Chidamber, B.J.; Jai, S.G.; Kailash, D.S. Study of Antimicrobial activity of Lemon (Citrus lemon) Peel extract. Br. J. Pharmacol. Toxicol. 2011, 2, 119–122. [Google Scholar]
- Campêlo, L.M.L.; Gonçalves, F.C.M.; Feitosa, C.M.; Freitas, R.M. Antioxidant activity of Citrus limon essential oil in mice hippocampus. Pharm. Biol. 2011, 49, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Shoib, A.B.; Aubid, H.M.; Zahoor, A.W.; Tabasum, M.; Zeeshan, S.; Nazia, A.; Nasheeman, A. Phytochemical analysis and antioxidant activity of different tissue types of Crocus sativus and oxidative stress alleviating potential of saffron extract in plants, bacteria, and yeast. S. Afr. J. Bot. 2015, 99, 80–87. [Google Scholar]
- Bhargava, V.K. Medicinal uses and pharmacological properties of Crocus sativus Linn (Saffron). Int. J. Pharm. Pharm. Sci. 2011, 3, 22–26. [Google Scholar]
- Javadi, B.; Sahebkar, A.; Emami, S.A. A Survey on Saffron in Major Islamic Traditional Medicine Books. Iran. J. Basic Med. Sci. 2013, 16, 1–11. [Google Scholar] [PubMed]
- Mittal, M.; Gupta, N.; Parashar, P.; Mehra, V.; Khatri, M. Phytochemical evaluation and pharmacological activity of Syzygium aromaticum: A comprehensive review. Int. J. Pharm. Pharm. Sci. 2014, 6, 67–72. [Google Scholar]
- Diego, F.C.R.; de Souza Fernandes, C.R.; Oliveira, W.P. Clove (Syzygium aromaticum): A precious spice. Asian Pac. J. Trop. Biomed. 2014, 4, 90–96. [Google Scholar]
- Huang, X.W.; Feng, Y.C.; Huang, Y.; Li, H.L. Chemical composition, antioxidant and the possible use as skin-care ingredient of clove oil (Syzygium aromaticum (L.) Merr. & Perry) and citronella oil (Cymbopogon goeringii) from China. J. Essent. Oil Res. 2013, 25, 315–323. [Google Scholar]
- Ansel, J.L.; Butaud, J.F.; Raharivelomanana, P. Bibliographical analysis of main lignified taxons used in tropical cosmetopoiea. C. R. Chim. 2016, 19, 1035–1048. [Google Scholar] [CrossRef]
- Sharma, R.K.; Goel, A.; Bhatia, A.K. Lawsonia inermis Linn: A plant with cosmetic and medical benefits. Int. J. Appl. Sci. Biotechnol. 2016, 4, 15–20. [Google Scholar] [CrossRef]
- Dawang, S.N.; Danahap, T.S.; Makvereng, S.S.; Nyam, M. Preliminary Survey of the Indigenous Knowledge of Canarium schweinfurthii Engl. (Atile) In Some Parts of Plateau State, Nigeria. IOSR J. Pharm. Biol. Sci. (IOSR-JPBS) 2016, 11, 76–78. [Google Scholar]
- Linnea. Zea mays Unsaponifiable Fraction. 2012. Available online: https://www.linnea.ch/media/4857/lin_scheda-zea-mays.pdf (accessed on 23 February 2018).
- Chinese Herbs Healing. Sandalwood (Santalum Album, Tan Xiang). 2012. Available online: http://www.chineseherbshealing.com/sandalwood/ (accessed on 23 February 2018).
- Fallick, K. Relevance of the Chemical Constituency of East Indian Sandalwood Essential Oil to Therapeutic and Traditional Use; Australasian College of Natural Therapies: Sydney, Australia, 2009; 14p. [Google Scholar]
- Nikiema, A. Agroforestry Parkland Species Diversity: Uses and Management in Semi-Arid West Africa (Burkina Faso). Ph.D. Thesis, University of Wageningen, Wageningen, The Netherlands, 2005. [Google Scholar]
- Warra, A.A. Cosmetic potentials of African sheet nuts (Vitellaria paradoxa) butter. Curr. Res. Chem. 2011, 3, 80–86. [Google Scholar]
- Diop, M.; Kaya, B.; Niang, A.; Olivier, A. Les Espèces Ligneuses et Leurs Usages: Les preferences des paysans dans le Cercle de Ségou, au Mali; ICRAF Working Paper No. 9; World Agroforestry Centre Nairobi: Nairobi, Kenya, 2005. [Google Scholar]
- Lykke, A.M.; Kristensen, M.K.; Ganaba, S. Valuation of local use and dynamics of 56 woody species in the Sahel. Biodivers. Conserv. 2004, 13, 1961–1990. [Google Scholar] [CrossRef]
- Okullo, J.B.L.; Omujal, F.; Agea, J.G.; Vuzi, P.C.; Namutebi, A.; Okello, J.B.A.; Nyanzi, S.A. Physico-chemical characteristics of shea butter (Vitellaria paradoxa C.F. Gaertn.) oil from the shea districts of Uganda. Afr. J. Food Agric. Nutr. Dev. 2010, 10, 2070–2084. [Google Scholar] [CrossRef]
- Coulibaly, Y.; Ouedraogo, S.; Niculescu, N. Experimental study of shea butter extraction efficiency using a centrifugal process. ARPN J. Eng. Appl. Sci. 2009, 4, 14–19. [Google Scholar]
- Attipoe, L.; Van Andel, A.; Nyame, S.K. The Novella PROJECT. Developing a Sustainable Supply Chain for Allanblackia Oil; SNV Netherlands Development Organisation: Accra, Ghana, 2006; 11p. [Google Scholar]
- UNDP. A Bottom-Up Approach to ABS: Community Level Capacity Development for Successful Engagement in ABS Value Chains in Cameroon (Echinops giganteus and Mondia whitei); Cameroon Project Document; UNDP: Yaoundé, Cameroon, 2016; 120p. [Google Scholar]
- Buck, M.; Hamilton, C. The nagoya protocol on access to genetic resources and the fair and equitable sharing of benefits arising from their utilization to the convention on biological diversity. Matthias Buck Clare Hamilt. 2011, 20. [Google Scholar] [CrossRef]
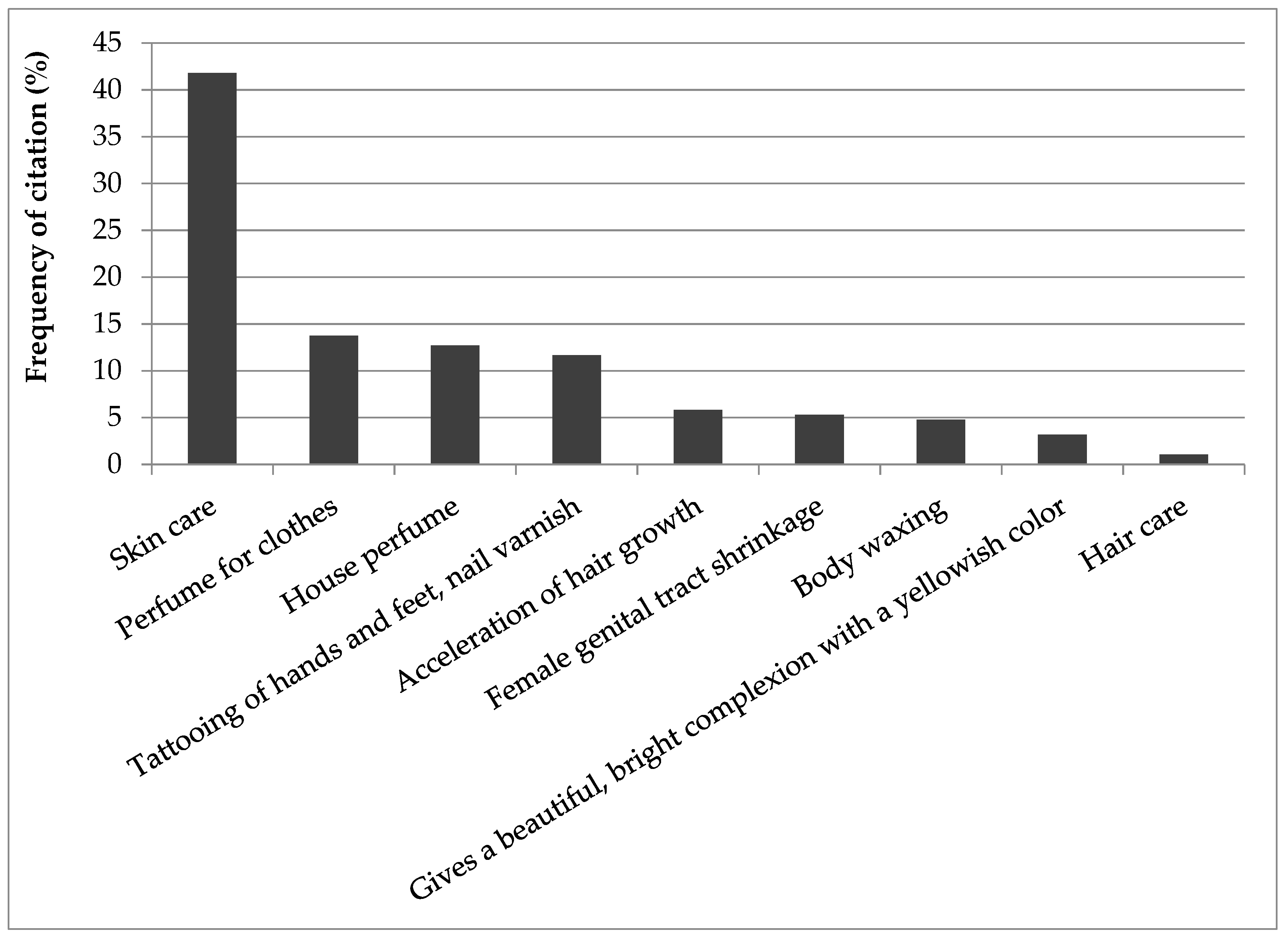
| N° | Species | Local Name | Part Used * | Usage | Frequency (%) |
|---|---|---|---|---|---|
| 1 | Canarium schweinfurthii Engl | Gamari | Bk, Fb, Rs | Barks, resins and flower buds are burned as incense to perfume the house | 12.7 |
| 2 | Santalum album L. | Sandal | Bk | Perfume for cloths | 12.17 |
| 3 | Lawsonia inermis L. | Hinna | Lv | Tattooing of hands and feet, nail varnish | 11.64 |
| 4 | Elaeis guineensis Jacq | Hamra | Fr | Gumming product for skin care | 10.58 |
| 5 | Arachis hypogae L. | Foul | S | Gumming product for skin care | 10.05 |
| 6 | Panicum miliaceum L. | Dourra amar | S | Gumming product for skin care | 10.05 |
| 7 | Zea mays L. | Massara | S | Gumming product for skin care | 10.05 |
| 8 | Terminalia avicennioides Guill. & Perr | Dorot | Bk | Perfume for clothes, Female genital tract shrinkage | 6.88 |
| 9 | Croton Zambesicus Müell. Arg. | Chébé | Acceleration of hair growth | 5.82 | |
| 10 | Citrus limon (L.) Burm.f. | Lemon | Fr | Used for body waxing | 4.76 |
| 11 | Crocus sativus L. | Courcoum | Rt | Gives a beautiful, bright complexion with a yellowish color | 3.17 |
| 12 | Syzigium aromaticum (L.) Merr. & L.M.Perry | Grounfal | Fr | Skin care | 1.06 |
| 13 | Vitellaria paradoxa C.F. Gaertn. | Oum kouroum | S | Hair care | 1.06 |
| Species | Part Used | Phytochemical Constituents | Properties/Activities | References |
|---|---|---|---|---|
| Canarium schweinfurthii Engl | Seed | Seed kernel oil extract | chemoprevention of cancers and other oxidative damage-induced diseases | [21] |
| Resin | Essential oil | natural antioxidant agent | [22,23] | |
| Barks | Polyphenols, triterpenes and steroids | antibacterial and antifungal activities | ||
| Santalum album L. | wood | Tannins, terpenes, resins and waxes | Sandalwood oil useful in perfume industry | [24] |
| wood | Essential oils | Antibacterial and antifungal activity | ||
| wood | Α-Santalumol | skin cancer preventive effect | ||
| Lawsonia inermis L. | Leaves | Lawsone, flavonoids, coumarins, triterpenoids, steroids, xanthones | anti-inflammatory, antibacterial, antimicrobial, antifungal, antiviral, antidermatophytic, antioxidant | [25,26] |
| Elaeis guineensis Jacq | oil | Phenolic, tannin and flavonoid compounds, vitamine E isomers (Tocopherols and tocotrienols) | Antioxidant activity | [27,28] |
| Alkaloids, saponins, tannins, anthraquinones, steroids, flavonoids | Antimicrobial activity | |||
| Terpenoids and alkaloids | Wounds healing activity | [29] | ||
| Oil | Excipient in most cosmetic preparation like the production of soaps | [30] | ||
| Vitamine A | Slows the desquamation of the cells of the epidermis | [26] | ||
| Arachis hypogae L. | Seeds | Resveratrol, Tannins, phlobatannins, saponins, flavonoids, quinones, terpenoids and cardiac glycosides | Melanolytic and skin lightening activity, reducing cancer risks, inhibiting dental caries, antioxidant capacity | [31,32,33] |
| Panicum miliaceum | Seeds | Carbohydrates, protein, oleic acid, linoleic acid, stearic acid and essential minerals like phosphorus, manganese, calcium and magnesium and rich in B-complex vitamins | Applied as a poultice for abscesses and sores | [34] |
| Zea mays | Carotenoids, Phenolic compounds, Phytosterols, Selenium, vitamins A, C, and K, Anthocyanins | has the potential to alleviate pain and possess analgesic activity, antimicrobial, and anti-inflammatory properties | [35] | |
| Terminalia avicennioides Guill. & Perr | Bark | Phenols, steroids, glycosides, flavonoids, tannins, ellagic acids, Anthraquinone, saponins, and terpenes | antimicrobial activity, Wound Healing Activity, Antioxidant activity, Antibacterial activity, Antifungal Activity | [36,37] |
| Croton zambesicus Müell. Arg. | Flavonoids, Saponins, Alkaloid, Tannins, Phenols, Triterpene, sesquiterpenes, Phytosterol, Anthraquinones and Carbohydrates, Labdanes, Trachylobanes, isopimaranes, Volatile Oils | antioxidant property | [38] | |
| Citrus limon (L.) Burm.f. | Lemon peel | Astringent and good antimicrobial agent. | [39] | |
| Phenolic compounds, sesamin, sesamol, sesamolin, and phytosterol | Antioxydant | [28,40] | ||
| Crocus sativus L. | Phenolics flavonoids and Apocarotenoids, safranal, crocin | Antioxidant activity | [41,42] | |
| Crocin, crocetin, diglucosylcrocetin, and dimethylcrocetin | Anti-carcinogenic effects | [43] | ||
| Syzigium aromaticum (L.) Merr. & L.M.Perry | oil | Monoterpenes, sesquiterpenes, phenolics and hydrocarbon compounds, eugenol, eugenyl acetate and β-caryophyllene | antibacterial, antifungal, insecticidal, antioxidant, anticarcinogenic capacities | [44,45,46] |
| topical analgesic in dentistry | ||||
| Vitellaria paradoxa C.F. Gaertn. | Seed oil | Provitamine A, allantoine, tocopherols | Antioxydant, antimicrobial, treatment of scars, burns and erythemas, skin lightening, dryness of the hair and produces a good lubrication of the hair | [47] |
| Triglycerides phospholipides palmitic, stearic, oleic, linoleique and linolenic acids |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fongnzossie Fedoung, E.; Zra, T.; Nyangono Biyegue, C.F.; Nouga Bissoue, A.; Baraye, S.; Tsabang, N. Herbal Cosmetics Knowledge of Arab-Choa and Kotoko Ethnic Groups in the Semi-Arid Areas of Far North Cameroon: Ethnobotanical Assessment and Phytochemical Review. Cosmetics 2018, 5, 31. https://doi.org/10.3390/cosmetics5020031
Fongnzossie Fedoung E, Zra T, Nyangono Biyegue CF, Nouga Bissoue A, Baraye S, Tsabang N. Herbal Cosmetics Knowledge of Arab-Choa and Kotoko Ethnic Groups in the Semi-Arid Areas of Far North Cameroon: Ethnobotanical Assessment and Phytochemical Review. Cosmetics. 2018; 5(2):31. https://doi.org/10.3390/cosmetics5020031
Chicago/Turabian StyleFongnzossie Fedoung, Evariste, Tize Zra, Christine Fernande Nyangono Biyegue, Achille Nouga Bissoue, Suzanne Baraye, and Nole Tsabang. 2018. "Herbal Cosmetics Knowledge of Arab-Choa and Kotoko Ethnic Groups in the Semi-Arid Areas of Far North Cameroon: Ethnobotanical Assessment and Phytochemical Review" Cosmetics 5, no. 2: 31. https://doi.org/10.3390/cosmetics5020031
APA StyleFongnzossie Fedoung, E., Zra, T., Nyangono Biyegue, C. F., Nouga Bissoue, A., Baraye, S., & Tsabang, N. (2018). Herbal Cosmetics Knowledge of Arab-Choa and Kotoko Ethnic Groups in the Semi-Arid Areas of Far North Cameroon: Ethnobotanical Assessment and Phytochemical Review. Cosmetics, 5(2), 31. https://doi.org/10.3390/cosmetics5020031