Fabrication of Water Absorbing Nanofiber Meshes toward an Efficient Removal of Excess Water from Kidney Failure Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
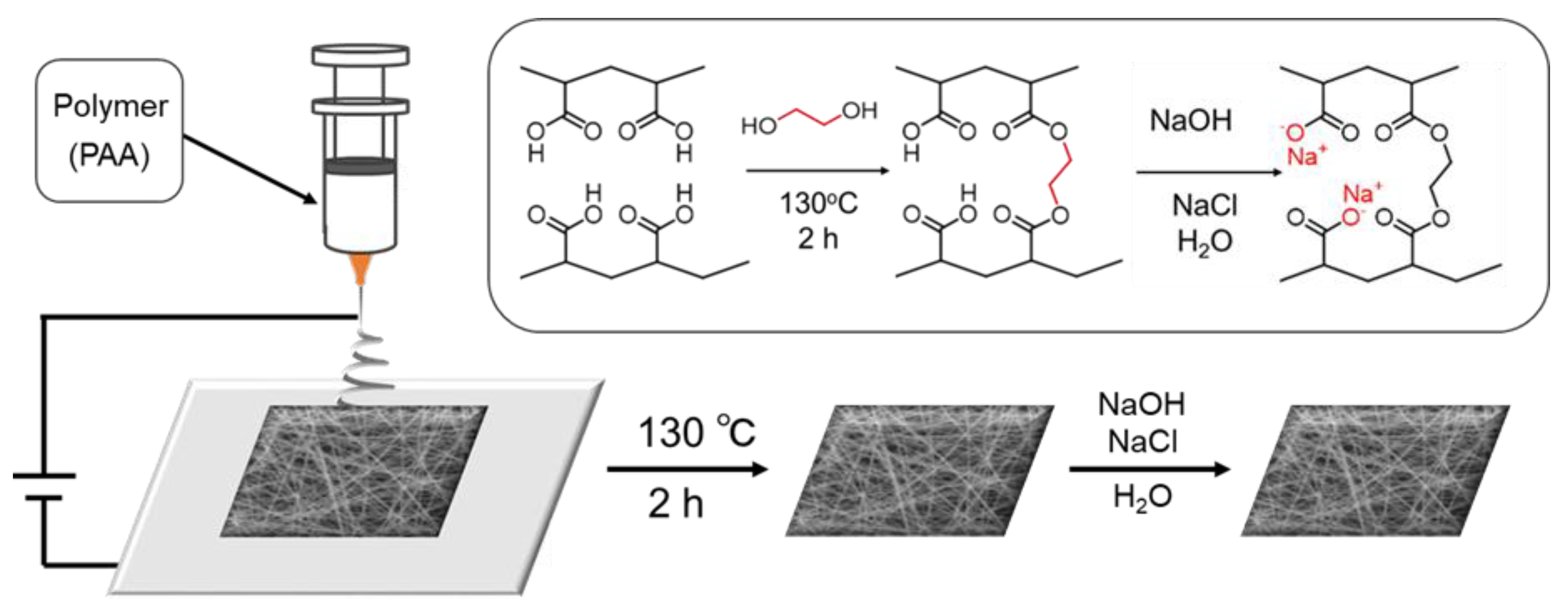
2.2. Fiber Fabrication
2.3. Fiber Characterization
2.4. Swelling Behavior of Nanofiber Meshes and Films
3. Results and Discussion
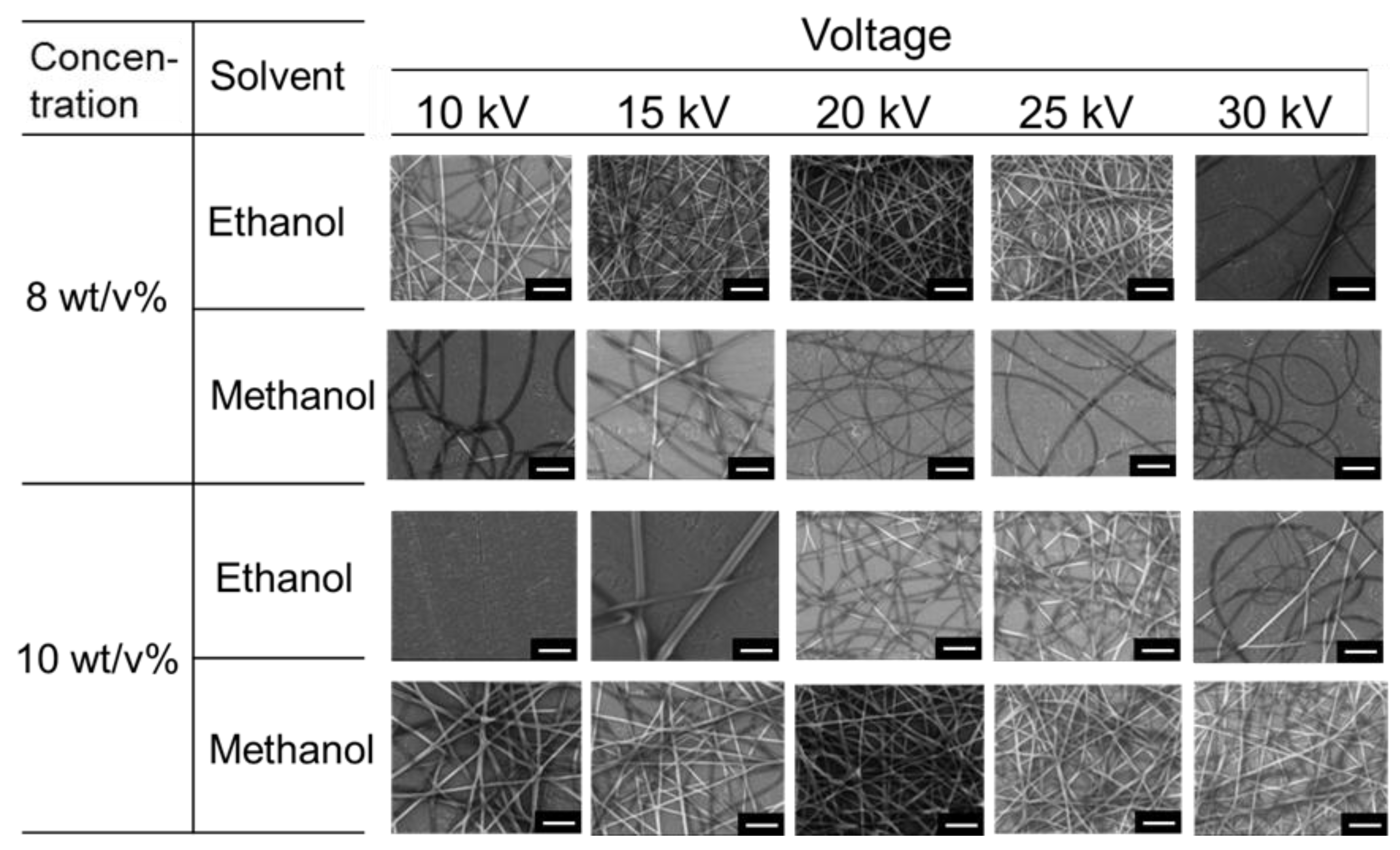
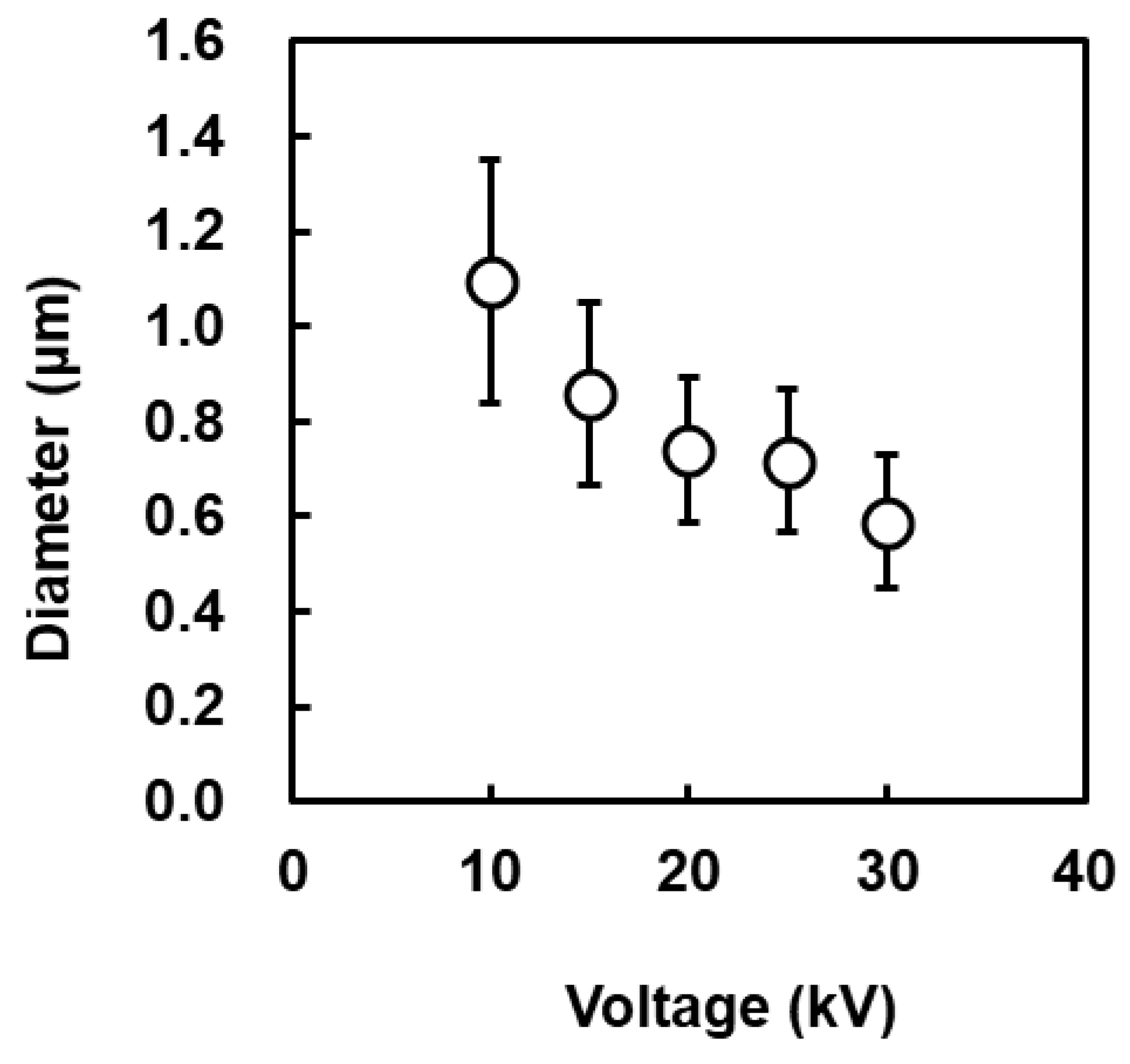
3.1. Fabrication of PSA Nanofiber Meshes
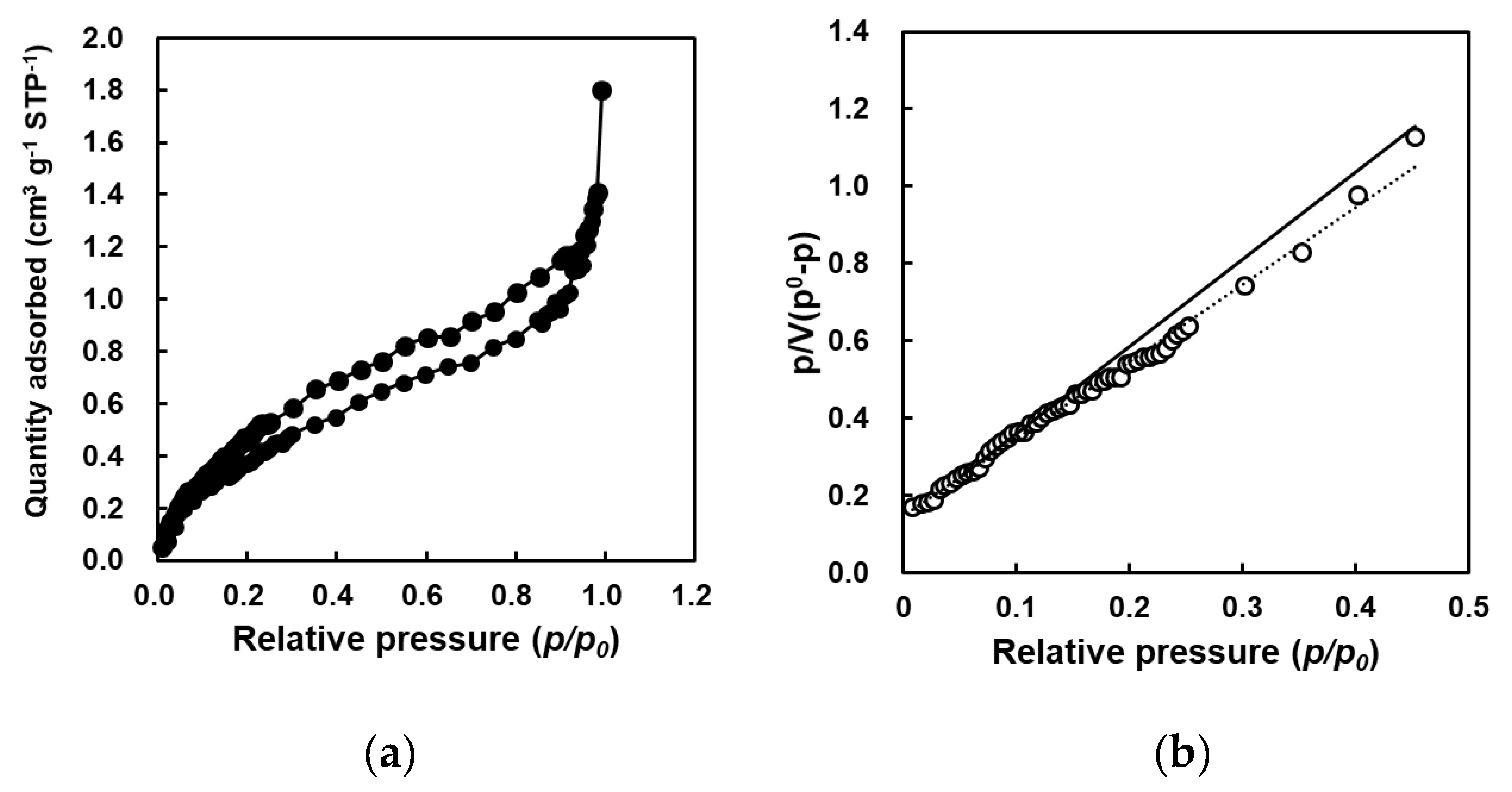
3.2. Porous Structure Analysis of PSA Nanofiber Meshes
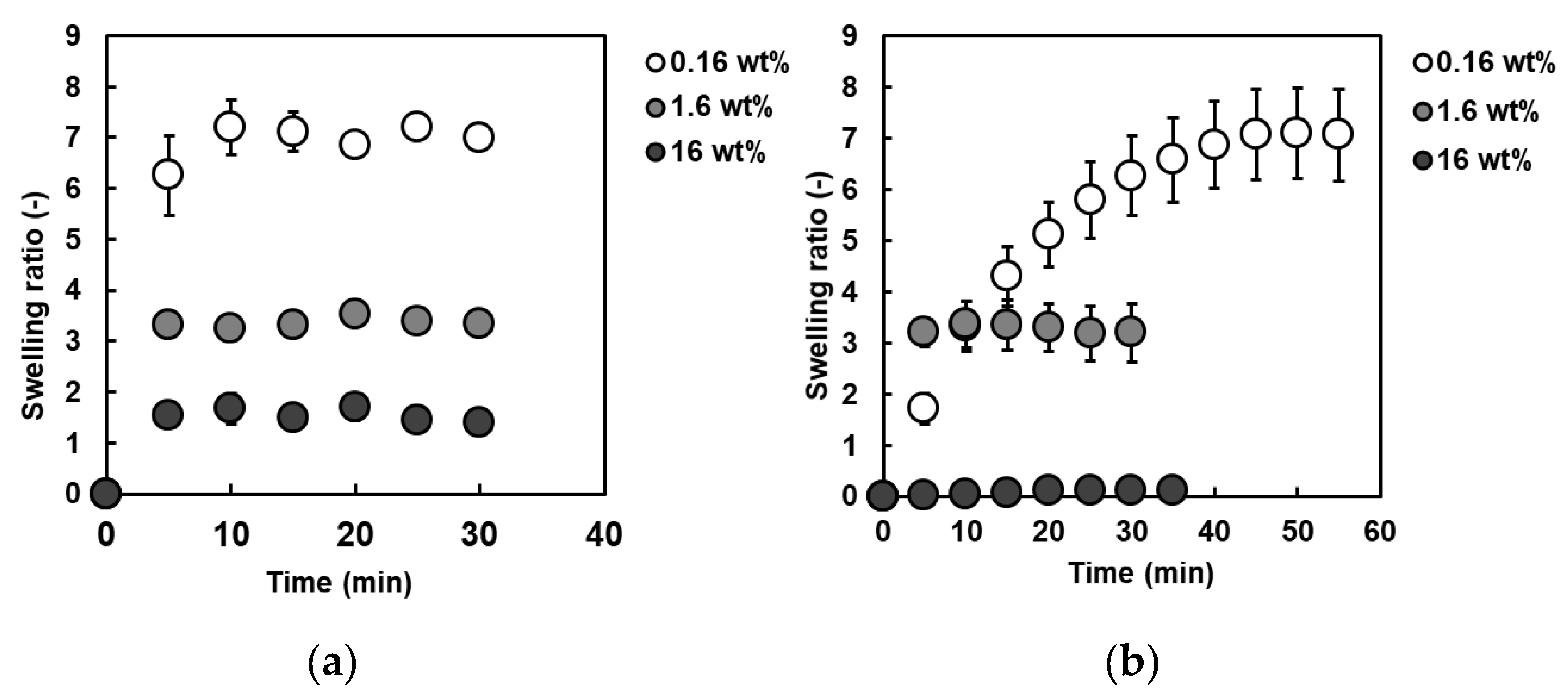
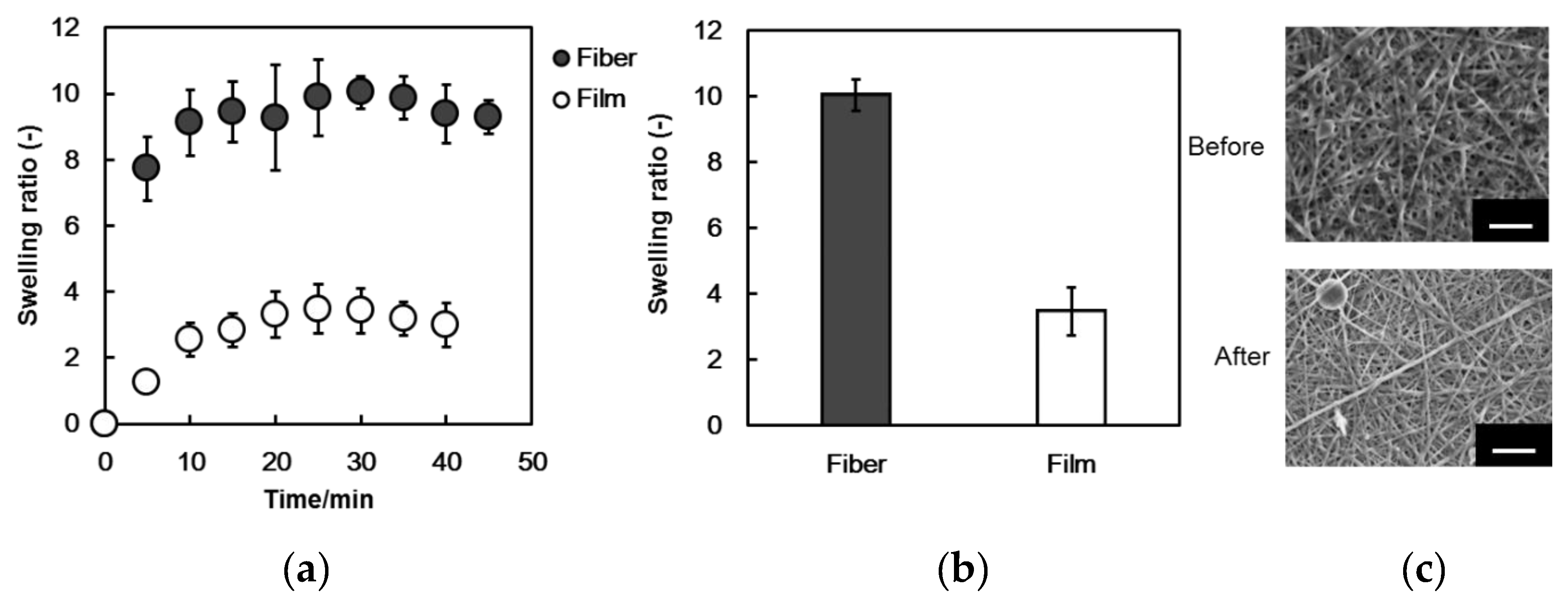
3.3. Water Absorption Test on PSA Nanofiber Meshes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Couser, W.G.; Remuzzi, G.; Mendis, S.; Tonelli, M. The contribution of chronic kidney disease to the global burden of major noncommunicable diseases. Kidney Int. 2011, 80, 1258–1270. [Google Scholar] [CrossRef] [PubMed]
- Liyanage, T.; Ninomiya, T.; Jha, V.; Neal, B.; Patrice, H.M.; Okpechi, I.; Zhao, M.; Lv, J.; Garg, A.X.; Knight, J.; et al. Worldwide access to treatment for end-stage kidney disease: A systematic review. Lancet 2015, 385, 1975–1982. [Google Scholar] [CrossRef]
- Glassock, R.J.; Winearls, C. The global burden of chronic kidney disease: How valid are the estimates? Nephron Clin. Pract. 2008, 110, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Harris, A.; Cooper, B.A.; Li, J.J.; Bulfone, L.; Branley, P.; Collins, J.F.; Craig, J.C.; Fraenkel, M.B.; Johnson, D.W.; Kesselhut, J.; et al. Cost-effectiveness of initiating dialysis early: A randomized controlled trial. Am. J. Kidney Dis. 2011, 57, 707–715. [Google Scholar] [CrossRef]
- Baboolal, K.; McEwan, P.; Sondhi, S.; Spiewanowski, P.; Wechowski, J.; Wilson, K. The cost of renal dialysis in a UK setting—A multicentre study. Nephrol. Dial. Transplant. 2008, 23, 1982–1989. [Google Scholar] [CrossRef]
- Young, B.A.; Chan, C.; Blagg, C.; Lockridge, R.; Golper, T.; Finkelstein, F.; Shaffer, R.; Mehrotra, R. How to overcome barriers and establish a successful home HD program. Clin. J. Am. Soc. Nephrol. 2012, 7, 2023–2032. [Google Scholar] [CrossRef]
- Johnson, DW.; Hayes, B.; Gray, NA.; Hawley, C.; Hole, J.; Mantha, M. Renal services disaster planning: Lessons learnt from the 2011 Queensland floods and North Queensland cyclone experiences. Nephrology 2013, 18, 41–46. [Google Scholar] [CrossRef] [PubMed]
- See, E.J.; Alrukhaimi, M.; Ashuntantang, G.E.; Bello, A.K.; Bellorin-Font, E.; Gharbi, M.B.; Braam, B.; Feehally, J.; Harris, D.C.; Jha, V.; et al. Global coverage of health information systems for kidney disease: Availability, challenges, and opportunities for development. Kidney Int. Suppl. 2018, 8, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Murakami, N.; Siktel, H.B.; Lucido, D.; Winchester, J.F.; Harbord, N.B. Disaster preparedness and awareness of patients on hemodialysis after Hurricane Sandy. Clin. J. Am. Soc. Nephrol. 2015, 10, 1389–1396. [Google Scholar] [CrossRef] [PubMed]
- Gorham, G.; Howard, K.; Togni, S.; Lawton, P.; Hughes, J.; Majoni, S.W.; Brown, S.; Barnes, S.; Cass, A. Economic and quality of care evaluation of dialysis service models in remote Australia: Protocol for a mixed methods study. BMC Health Serv. Res. 2017, 17, 320. [Google Scholar] [CrossRef] [PubMed]
- Sever, M.S.; Erek, E.; Vanholder, R.; Kalkan, A.; Guney, N.; Usta, N.; Yilmaz, C.; Kutanis, C.; Turgut, R.; Lameire, N. Features of chronic hemodialysis practice after the Marmara Earthquake. J. Am. Soc. Nephrol. 2004, 15, 1071–1076. [Google Scholar] [CrossRef] [PubMed]
- Nangaku, M.; Akizawa, T. Diary of a Japanese nephrologist during the present disaster. Kidney Int. 2011, 79, 1037–1039. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Namekawa, K.; Schreiber, M.T.; Aoyagi, T.; Ebara, M. Fabrication of zeolite–polymer composite nanofibers for removal of uremic toxins from kidney failure patients. Biomater. Sci. 2014, 2, 674–679. [Google Scholar] [CrossRef]
- Takai, R.; Kurimoto, R.; Nakagawa, Y.; Kotsuchibashi, Y.; Namekawa, K.; Ebara, M. Towards a rational design of zeolite-polymer composite nanofibers for efficient adsorption of creatinine. J. Nanomater. 2016, 2016, 1–7. [Google Scholar] [CrossRef]
- Vasita, R.; Katti, D.S. Nanofibers and their applications in tissue engineering. Int. J. Nanomed. 2006, 1, 15–30. [Google Scholar] [CrossRef]
- Zhou, F.-L.; Gong, R.-H. Manufacturing technologies of polymeric nanofibres and nanofibre yarns. Polym. Int. 2008, 57, 837–845. [Google Scholar] [CrossRef]
- Greiner, A.; Wendorff, J.H. Electrospinning: A fascinating method for the preparation of ultrathin fibers. Angew. Chem. Int. Ed. 2007, 46, 5670–5703. [Google Scholar] [CrossRef]
- Doshi, J.; Reneker, D.H. Electrospinning process and applications of electrospun fibers. J. Electrost. 1995, 35, 151–160. [Google Scholar] [CrossRef]
- Reneker, D.H.; Yarin, A.L.; Fong, H.; Koombhongse, S. Bending instability of electrically charged liquid jets of polymer solutions in electrospinning. J. Appl. Phys. 2000, 87, 4531–4547. [Google Scholar] [CrossRef]
- Thompson, C.J.; Chase, G.G.; Yarin, A.L.; Reneker, D.H. Effects of parameters on nanofiber diameter determined from electrospinning model. Polymer 2007, 48, 6913–6922. [Google Scholar] [CrossRef]
- Rajala, J.W.; Shin, H.U.; Lolla, D.; Chase, G.G. Core–Shell electrospun hollow aluminum oxide ceramic fibers. Fibers 2015, 3, 450–462. [Google Scholar] [CrossRef]
- Bou, S.; Ellis, A.V.; Ebara, M. Synthetic stimuli-responsive ‘smart’ fibers. Curr. Opin. Biotechnol. 2016, 39, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Garrett, R.; Niiyama, E.; Kotsuchibashi, Y.; Uto, K.; Ebara, M. Biodegradable nanofiber for delivery of immunomodulating agent in the treatment of basal cell carcinoma. Fibers 2015, 3, 478–490. [Google Scholar] [CrossRef]
- Li, A.; Zhang, J.; Wang, A. Synthesis, characterization and water absorbency properties of poly(acrylic acid)/sodium humate superabsorbent composite. Polym. Adv. Technol. 2005, 16, 675–680. [Google Scholar] [CrossRef]
- Chen, Y.; Tan, H. Crosslinked carboxymethylchitosan-g-poly(acrylic acid) copolymer as a novel superabsorbent polymer. Carbohydr. Res. 2006, 341, 887–896. [Google Scholar] [CrossRef] [PubMed]
- Omidian, H.; Hashemi, S.A.; Sammes, P.G.; Meldrum, I. Modified acrylic-based superabsorbent polymers (dependence on particle size and salinity). Polymer 1999, 40, 1753–1761. [Google Scholar] [CrossRef]
- Fu, Q.; Wang, X.; Si, Y.; Liu, L.; Yu, J.; Ding, B. Scalable fabrication of electrospun nanofibrous membranes functionalized with citric acid for high-performance protein adsorption. Appl. Mater. Interfaces 2016, 8, 11819–11829. [Google Scholar] [CrossRef]
- Si, Y.; Wang, X.; Li, Y.; Chen, K.; Wang, J.; Yu, J.; Wang, H.; Ding, B. Optimized colorimetric sensor strip for mercury(II) assay using hierarchical nanostructured conjugated polymers. J. Mater. Chem. A 2014, 2, 645–652. [Google Scholar] [CrossRef]
- Barakat, N.A.M.; Khalil, K.A.; Mahmoud, I.H.; Kanjwal, M.A.; Sheikh, F.A.; Kim, H.Y. CoNi bimetallic nanofibers by electrospinning: Nickel-based soft magnetic material with improved magnetic properties. J. Phys. Chem. C 2010, 114, 15589–15593. [Google Scholar] [CrossRef]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsuge, M.; Takahashi, K.; Kurimoto, R.; Fulati, A.; Uto, K.; Kikuchi, A.; Ebara, M. Fabrication of Water Absorbing Nanofiber Meshes toward an Efficient Removal of Excess Water from Kidney Failure Patients. Fibers 2019, 7, 39. https://doi.org/10.3390/fib7050039
Tsuge M, Takahashi K, Kurimoto R, Fulati A, Uto K, Kikuchi A, Ebara M. Fabrication of Water Absorbing Nanofiber Meshes toward an Efficient Removal of Excess Water from Kidney Failure Patients. Fibers. 2019; 7(5):39. https://doi.org/10.3390/fib7050039
Chicago/Turabian StyleTsuge, Mirei, Kanoko Takahashi, Rio Kurimoto, Ailifeire Fulati, Koichiro Uto, Akihiko Kikuchi, and Mitsuhiro Ebara. 2019. "Fabrication of Water Absorbing Nanofiber Meshes toward an Efficient Removal of Excess Water from Kidney Failure Patients" Fibers 7, no. 5: 39. https://doi.org/10.3390/fib7050039
APA StyleTsuge, M., Takahashi, K., Kurimoto, R., Fulati, A., Uto, K., Kikuchi, A., & Ebara, M. (2019). Fabrication of Water Absorbing Nanofiber Meshes toward an Efficient Removal of Excess Water from Kidney Failure Patients. Fibers, 7(5), 39. https://doi.org/10.3390/fib7050039






