State of the Art in the Development and Properties of Protein-Based Films and Coatings and Their Applicability to Cellulose Based Products: An Extensive Review
Abstract
:1. Introduction
2. Proteins with Suitable Functionalities as Food Packaging Materials
| Origin | Plant Protein | Animal Protein | Fungi |
|---|---|---|---|
| Globular proteins | Wheat gluten | Whey protein | Hydrophobins |
| Rice (bran) protein | Egg albumin | ||
| Corn zein | |||
| Soy protein | |||
| Pea protein | |||
| Phaseolin | |||
| Peanut protein | |||
| Winged beans protein | |||
| Pistachios protein | |||
| Lupin protein | |||
| Sunflower protein | |||
| Cottonseed protein | |||
| Amaranth protein | |||
| Sorghum kafirin | |||
| Potato protein | |||
| Fibrillar proteins | Casein | ||
| Gelatin/collagen | |||
| Keratin | |||
| Fish myofibrillar protein |
3. Protein Processing and Purification
4. Protein Characterization and Functional Properties
5. Protein Film-Formation
6. Protein Modification to Control and Optimize Film-Formation
7. Different Technologies for Processing the Coating
7.1. Wet Coating
7.2. Extrusion
| Protein | L/D Ratio | Speed Screw Range (rpm) | Temperature Profile (°C) | Reference |
|---|---|---|---|---|
| Whey protein | 30:1 | 250 | 20–130 | [93,95] |
| Soy protein | – | 20–25 | 120–160 | [97] |
| Gelatin | 25:1 | 100–200 | 65–105 | [90] |
| Keratin | 25:1 | 25 | 120 | [98] |
| Sunflower protein | 25:1 | 20–200 | 85–160 | [88] |
8. Coating Formulations Based on Widely Available Proteins
8.1. Casein and Caseinates
8.1.1. Barrier Properties
8.1.2. Mechanical Properties
8.1.3. Surface Properties
8.1.4. End of Life
8.2. Whey Proteins
8.2.1. Barrier Properties
8.2.2. Mechanical Properties
8.2.3. Surface
8.2.4. End of Life
8.3. Gelatin
8.3.1. Barrier Properties
8.3.2. Mechanical Properties
8.3.3. Surface Properties
8.3.4. End of Life
8.4. Wheat Gluten
8.4.1. Barrier Properties
8.4.2. Mechanical Properties
8.4.3. Surface Properties
8.4.4. End of Life
8.5. Soy Protein
8.5.1. Barrier Properties
8.5.2. Mechanical Properties
8.5.3. Surface Properties
8.5.4. End of Life
8.6. Corn Zein
8.6.1. Barrier Properties
8.6.2. Mechanical Properties
8.6.3. Surface Properties
8.6.4. End of Life
9. Proteins with Low Availability or Not yet Developed
9.1. Emerging Proteins from Animal Kingdom
9.2. Emerging Proteins from Plant Kingdom
9.3. Emerging Proteins from Fungi Kingdom
| Origin | Proteins | References and Applications | Preparation/Applications |
|---|---|---|---|
| Animal | feathers | Dou, Huang, Zhang, He, Yin and Cui, 2015 [241] | Films by solution casting (cross-linker) |
| egg albumin | Gennadios, Handa, Froning, Weller and Hanna, 1998 [247] | Films by solution casting alkaline solutions (plasticizer) | |
| fish myofibrills | Cuq, Aymard, Cuq and Guilbert, 1995 [19]; Shiku, Hamaguchi, and Tanaka, 2003 [242]; Sobral, Santos and Garcia, 2005 [243]; | Edible and transparent films by solution casting from acidic or alkaline solutions (plasticizer) | |
| hake | Pires, Ramos, Teixeira, Batista, Nunes and Marques, 2013 [244] | Edible but yellowish films by solution casting from alkaline solutions (plasticizer) | |
| mussel | Lee, Scherer and Messersmith, 2006; Meredith and Wilker, 2015 [245] | Under study the industrial production of protein by recombinant approaches | |
| Plant | potato | Du, Chen, Zhang, Rempel, Thomson and Liu, 2015 [248] | Compression molded films |
| rice | Shih, 1996 [249] | Edible films containing pullulan | |
| amaranth | Denavi, Tapia-Placido, Anon, Sobral, Mauri and Menegalli, 2005 [251] | Films by solution casting (plasticizer) | |
| pea | Choi and Han, 2001 [254] | Films by solution casting (plasticizer), properties similar to soy and whey films | |
| phaseolin | Giosafatto, Di Pierro, Gunning, Mackie, Porta and Mariniello, 2014 [257] | Edible films by solution casting (plasticizer) containing also polysaccharides | |
| chickpea | Ionescu, Aprodu, Daraba, Gurau, Baciu and Nichita, 2009 [260] | Edible films by solution casting (plasticizer) | |
| faba bean | Saremnezhad, Azizi, Barzegar, Abbasi and Ahmadi, 2011 [22] | Edible films by solution casting of alkaline solutions (plasticizer) | |
| lupine | Chango, Villaume, Bau, Nicolas and Mejean, 1995 [262] | Edible films by solution casting of alkaline solutions | |
| pistachio | Zahedi, Ghanbarzadeh and Sedaghat, 2010 [265] | Edible films by solution casting of solutions (in cellulose based edible films) | |
| peanut | Jangchud and Chinnan, 1999 [266] | Edible films and coating by solution casting | |
| sunflower | Salgado, Lopez-Caballero, Gomez-Guillen, Mauri and Montero, 2013 [253] | Edible films by solution casting with antioxidant properties and antimicrobial properties | |
| cotton seed | Grevellec, Marquié, Ferry, Crespy and Vialettes, 2001 [268] | Films for packaging by solution casting or compression molding (plasticizer) | |
| sorghum | Buffo, Weller and Gennadios, 1997 [270] | Films by solution casting with properties similar to corn zein films but more colored | |
| Fungi | hydrophobin | Gabor, Buthe, Eck, Kaufmann, Koch and Meurer, 2007 [276] | Nano or molecular films by solution casting on cellulose products |
10. Combinations of Properties of Protein Coatings and Films
| Protein | Barrier Properties | Mechanical Properties | Adhesion | ||
|---|---|---|---|---|---|
| Oxygen | Water Vapor | Tensile Strength | Elongation at Break | ||
| Casein/caseinates | 3 | 1 | 1 | 2 | 3 |
| Whey | 3 | 1 | 1 | 2 | – |
| Gelatin | 1 | 1 | 2 | 1 | 3 |
| Wheat | 2 | 2 | 1 | 1 | 2 |
| Soy | 3 | 1 | 1 | 2 | 2 |
| Corn zein | 3 | 1 | 1 | 1 | 2 |
| Feathers | – | 2 | 2 | 1 | – |
| Egg albumin | – | – | 2 | 2 | – |
| Fish myofibrills | – | 2 | 2 | 1 | – |
| Hake | – | 2 | 1 | 2 | – |
| Mussel | – | – | – | – | 3 |
| Potato | – | – | 2 | 1 | 2 |
| Rice | – | 1 | 2 | 0 | – |
| Amaranth | 3 | 2 | 0 | 2 | – |
| Pea | – | 1 | 1 | 1 | – |
| Phaseolin | 3 | 1 | 1 | 1 | – |
| Chickpea | – | – | – | – | – |
| Faba bean | – | 1 | 1 | 2 | – |
| Lupine | – | – | – | – | – |
| Pistachio | – | 0 | 2 | 1 | – |
| Peanut | 0 | 0 | 1 | 2 | – |
| Sunflower | – | 1 | 0 | 1 | – |
| Cotton seed | – | – | – | – | – |
| Sorghum | – | 1 | 1 | 2 | – |
| Hydrophobins | – | – | – | - | – |
11. Application to Cellulose Based Products
11.1. Barrier Properties
| Protein Used | Process (Wet/Extrusion) | WVTR | OTR | Reference |
|---|---|---|---|---|
| whey | wet | 134 × 10−11 g·m/m2·s·Pa | – | [8] |
| soy | wet | 6.6 × 10−14 kg·m/m2·s·Pa | 2.4 × 10−18 m3·m/m2·s·Pa | [214] |
| caseinates | wet | 5.4 g∙mm/m2·d·kPa | 3.2 cm3·μm/m2·d·kPa | [115] |
| wet | 938 g·m/m2·d·kPa | – | [121] | |
| wet | 0.25 g·mm/m2·d·kPa | – | [2] | |
| wheat gluten | wet or compr. molded | – | 0.38 mL/mm·day·m2·atm | [32] |
| wet | 3.27 × 10−11 mol·m−1·s−1·Pa−1 | 49,689 × 10−18 mol·m−1·s−1·Pa−1 | [283] | |
| corn zein | wet | 881 g/m2·d | – | [235] |
| wet | 70 g/100 in2·d | 16.000 cm3/100 in2·day | [37] |
| Material | O2(g·m/m2·s·Pa) | H2O Vapor (g·m/m2·s·Pa) |
|---|---|---|
| Synthetic polymer | ||
| Polyester | 0.38 × 10−15 | 3.6 × 10−13 |
| Polypropylene (PP) | 0.38 × 10−19 | 6.5 × 10−13 |
| Polyvinyl chloride (PVC) | 0.36 × 10−21 | 2.16 × 10−11 |
| Polyethylene terephthalate (PET) | 0.15 × 10−21 | – |
| Low-density polyethylene (LDPE) | 0.16 × 10−19 | 8.1 × 10−13 |
| High-density polyethylene (HDPE) | 0.35 × 10−20 | 2.52 × 10−13 |
| Edible coating material | ||
| Methylcellulose (MC) | 0.27 × 10−8 | 9.35 × 10−11 |
| Hydroxypropyl cellulose (HPC) | 0.22 × 10−8 | 5.55 × 10−70 |
| Sucrose polyester | 0.15 × 10−20 | 4.2 × 10−13 |
| Zein | 0.5 × 10−21 | 1.17 × 10−10 |
| Chitosan | 0.98 × 10−24 | 4.9 × 10−10 |
| Wheat gluten | 0.21 × 10−19 | 9.18 × 10−11 |
| Whey protein isolate (WPI) | 0.79 × 10−21 | 1.1 × 10−9 |
| Soy protein | 0.22 × 10−21 | 3.49 × 10−10 |
11.2. Mechanical Properties
| Protein Used | Process (Wet/Extrusion) | Young Modulus (MPa) | Tensile Strength (MPa) | Elongation at Break (%) | Reference |
|---|---|---|---|---|---|
| whey | wet | 140 | – | 7 | [8] |
| wet | 4200 | 0.08 | 4.2 | [32] | |
| soy | wet | – | 17 * | 177 * | [214] |
| wet | – | 40 Nm/g | – | [304] | |
| caseinates | wet | – | 16 ** | 30 ** | [115] |
| wet | – | 5 kN/m | 4 | [2] | |
| wheat gluten | compression molded | 121 | 106 | 6 | [32] |
| Film | Tensile Strength (MPa) | Elongation (%) |
|---|---|---|
| UF-TMP a:Gly b (4:1) | 10.1 | 5.2 |
| β-casein:Gly (2:1) | 6–9 | 250 |
| αs1-casein:Gly (49:1) | 4.1 | 38 |
| αs1-casein:Gly (49:1), Tgase c | 10.6 | 77 |
| β-Lg d:Gly (2.6:1), Tgase | 141.2 | – |
| NaCas e:Gly (4:1) | 17.4–26.7 | 10.5 |
| NaCas:Gly (2:1) | 2.98 | 29.9 |
| NaCas:PEG f (4:1) | 10.9–16.35 | 5.3 |
| NaCas:PEG f (4:1) | 10.9–16.35 | 5.3 |
| NaCas g:Gly (2:1) | 4.25 | 1.4 |
| KCas h:Gly (2:1) | 2.97 | 42.8 |
| WPI i:Gly (2:1) | 5.76 | 22.7 |
| WPI:Gly (2.3:1) | 13.9 | 30.8 |
| WPI:Gly (5.7:1) | 29.1 | 4.1 |
| WPI:Sor j (1:1) | 14.7 | 8.7 |
| WPI:Sor (2.3:1) | 14.0 | 1.6 |
| WPC k:Gly (2:1) | 3.49 | 20.8 |
| NaCas:AM l:Gly (2:2:1) | 1.32 | 27.4 |
| CaCas:AM:Gly (2:2:1) | 2.14 | 13.4 |
| KCas:AM:Gly (2:2:1) | 1.66 | 17.7 |
| WPI:AM:Gly (2:2:1) | 3.14 | 10.8 |
| WPC:AM:Gly (2:2:1) | 1.08 | 13.6 |
| LDPE m | 13 | 500 |
| LDPE n | 26 | 300 |
11.3. Binding and Adhesion Properties
12. Food Safety Aspects, Migration, Legal and Regulatory Aspects
13. Biodegradation, Environmental Degradation and Other Environmental Aspects
13.1. Biodegradation and Composting of Protein Based Films
- EN 13431:2000 Packaging. Requirements for Packaging Recoverable in the Form of Energy Recovery Including Specification of Minimum Inferior Calorific Value.
- EN 13432:2000 Packaging. Requirements for Packaging Recoverable Through Composting and Biodegradation. Test Scheme and Evaluation Criteria for the Final Acceptance of Packaging.
- CR 13695-1 Packaging. Requirements for Measuring and Verifying the Four Heavy Metals (Cr, Rd, Hg, Pb) and Their Release into the Environment, and Other Dangerous Substances Present in Packaging.
- EN 13427:2000 Packaging. Requirements for the Use of European Standards in the Field of Packaging Waste (“Umbrella Norm”).
- EN 13428:2000 Packaging. Requirements Specific to Manufacturing and Composition. Prevention by Source Reduction.
- EN 13429:2000 Packaging. Reuse.
- EN 13430:2000 Packaging. Requirements for Packaging Recoverable by Material Recycling.
- The product must contain at least 50% organic matter and may not exceed the heavy metal limits specified in the standard.
- The products should mineralize for at least 90%, adopting the mineralization of a similar sample of cellulose as 100% positive control, within 6 months under controlled composting conditions, where mineralization is defined as the conversion of the organic C to CO2 and biomass, this characteristic is linked to the chemical composition of the sample.
- The product, in the form which enters the market, should, within a timeframe of 12 weeks, fragment in parts smaller than 2 mm under controlled composting conditions. It has to be outlined that this requirement refers to the physical form of the product instead of to the chemical composition. Consequently, this characteristic is linked to the thickness and the physical construction (e.g., laminate, coating, etc.) of the sample, and can result tricky to be met also for packaging based on biodegradable materials.
- The compost obtained at the end of the composting trial, that can also contain some no degraded residuals from the product, must not have any negative effect to the germination and growth of plants.
13.2. Integration of Protein Based Technologies in Current Waste Management System
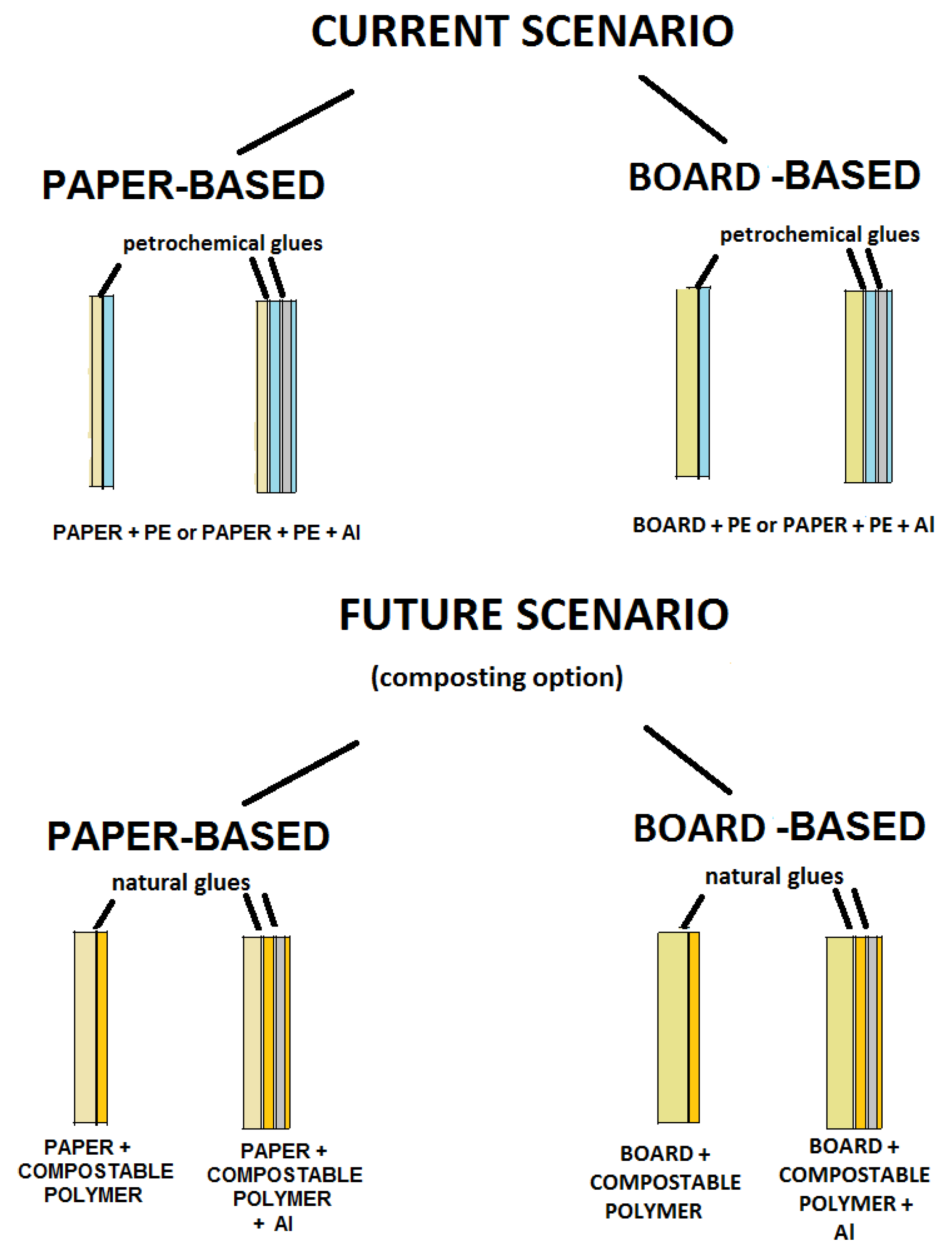
14. Conclusions and Industrial Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
Acronyms
| CGM | corn gluten meal |
| DAS | dialdehyde starch |
| EDTA | ethylendiaminetetraacetic acid |
| EVA | poly(ethylene-co-vinyl acetate) |
| EVOH | poly(ethylene-co-vinyl alcohol) |
| GHCl | guanidine hydrochloride |
| HPMC | hydroxypropylmethylcellulose |
| LDPE | low density poly(ethylene) |
| MAP | modified atmosphere packaging |
| MMT | montmorillonite |
| NaCAS | sodium caseinate |
| OCC | old corrugated containers |
| OPP | oriented poly(propylene) |
| OTR | oxygen transmission rate |
| PCL | poly(e-caprolactone) |
| PE | poly(ethylene) |
| PLA | poly(lactic acid) |
| PVA | poly(vinyl alcohol) |
| PVC | poly(vinyl chloride) |
| PVDC | poly(vinylidene chloride) |
| RH | relative humidity |
| SME | specific mechanical energy |
| SPH | sodium hypophosphite |
| SPI | soy protein isolate |
| WPI | whey protein isolate |
| WVP | water vapor permeability |
| WVTR | water vapor transmission |
References
- Gastaldi, E.; Chalier, P.; Guillemin, A.; Gontard, N. Microstructure of protein-coated paper as affected by physico-chemical properties of coating solutions. Colloids Surf. A Physicochem. Eng. Aspects 2007, 301, 301–310. [Google Scholar] [CrossRef]
- Khwaldia, K. Water vapor barrier and mechanical properties of paper-sodium caseinate and paper-sodium caseinate-paraffin wax films. J. Food Biochem. 2010, 34, 998–1013. [Google Scholar] [CrossRef]
- Aloui, H.; Khwaldia, K.; Ben Slama, M.; Hamdi, M. Effect of glycerol and coating weight on functional properties of biopolymer-coated paper. Carbohydr. Polym. 2011, 86, 1063–1072. [Google Scholar] [CrossRef]
- Ottenio, D.; Escabasse, J.-Y.; Podd, B. Packaging Materials Vol. 6. Paper and Board for Food Packaging Applications; ILSI: Brussels, Belgium, 2004; pp. 1–28. [Google Scholar]
- Dixon, J. Packaging Materials Vol. 9. Multilayer Packaging for Food and Beverages; ILSI: Brussels, Belgium, 2011; pp. 1–43. [Google Scholar]
- Farris, S.; Schaich, K.M.; Liu, L.; Piergiovanni, L.; Yam, K.L. Development of polyion-complex hydrogels as an alternative approach for the production of bio-based polymers for food packaging applications: A review. Trends Food Sci. Technol. 2009, 20, 316–332. [Google Scholar] [CrossRef]
- Khwaldia, K.; Arab-Tehrany, E.; Desobry, S. Biopolymer coatings on paper packaging materials. Compr. Rev. Food Sci. Food Saf. 2010, 9, 82–91. [Google Scholar] [CrossRef]
- Han, J.H.; Krochta, J.M. Physical properties and oil absorption of whey-protein-coated paper. J. Food Sci. 2001, 66, 294–299. [Google Scholar] [CrossRef]
- Lin, S.Y.; Krochta, J.M. Plasticizer effect on grease barrier and color properties of whey-protein coatings on paperboard. J. Food Sci. 2003, 68, 229–233. [Google Scholar] [CrossRef]
- Park, S.K.; Hettiarachchy, N.S.; Were, L. Degradation behavior of soy protein-wheat gluten films in simulated soil conditions. J. Agric. Food Chem. 2000, 48, 3027–3031. [Google Scholar] [CrossRef] [PubMed]
- Peelman, N.; Ragaert, P.; de Meulenaer, B.; Adons, D.; Peeters, R.; Cardon, L.; Devlieghere, F. Application of bioplastics for food packaging. Trends Food Sci. Technol. 2013, 32, 128–141. [Google Scholar] [CrossRef]
- Debeaufort, F.; Quezada-Gallo, J.A.; Voilley, A. Edible films and coatings: Tomorrow’s packagings: A review. Crit. Rev. Food Sci. Nutr. 1998, 38, 299–313. [Google Scholar] [CrossRef] [PubMed]
- Andersson, C. New ways to enhance the functionality of paperboard by surface treatment—A review. Packag. Technol. Sci. 2008, 21, 339–373. [Google Scholar] [CrossRef]
- Oh, J.H.; Wang, B.; Field, P.D.; Aglan, H.A. Characteristics of edible films made from dairy proteins and zein hydrolysate cross-linked with transglutaminase. Int. J. Food Sci. Technol. 2004, 39, 287–294. [Google Scholar] [CrossRef]
- Gennadios, A.; Weller, C.L.; Gooding, C.H. Measurement errors in water-vapor permeability of highly permeable, hydrophilic edible films. J. Food Eng. 1994, 21, 395–409. [Google Scholar] [CrossRef]
- Krochta, J.M.; DeMulder-Johnston, C. Edible and biodegradable polymer films: Challenges and opportunities. Food Technol. 1997, 51, 61–74. [Google Scholar]
- Pol, H.; Dawson, P.; Acton, J.; Ogale, A. Soy protein isolate/corn-zein laminated films: Transport and mechanical properties. J. Food Sci. 2002, 67, 212–217. [Google Scholar] [CrossRef]
- Weber, C.J. Biobased Packaging Materials for the Food Industry: Status and Perspectives; KVL: Frederiksberg, Denmark, 2000. [Google Scholar]
- Cuq, B.; Aymard, C.; Cuq, J.L.; Guilbert, S. Edible packaging films based on fish myofibrillar proteins: Formulation and functional properties. J. Food Sci. 1995, 60, 1369–1374. [Google Scholar] [CrossRef]
- Rhim, J.-W. Potential use of biopolymer-based nanocomposite films in food packaging applications. Food Sci. Biotechnol. 2007, 16, 691–709. [Google Scholar]
- Cuq, B.; Gontard, N.; Guilbert, S. Proteins as agricultural polymers for packaging production. Cereal Chem. 1998, 75, 1–9. [Google Scholar] [CrossRef]
- Saremnezhad, S.; Azizi, M.H.; Barzegar, M.; Abbasi, S.; Ahmadi, E. Properties of a new edible film made of faba bean protein isolate. J. Agric. Sci. Technol. 2011, 13, 181–192. [Google Scholar]
- Viroben, G.; Barbot, J.; Mouloungui, Z.; Gueguen, J. Preparation and characterization of films from pea protein. J. Agric. Food Chem. 2000, 48, 1064–1069. [Google Scholar] [CrossRef] [PubMed]
- Brandenburg, A.H.; Weller, C.L.; Testin, R.F. Edible films and coatings from soy protein. J. Food Sci. 1993, 58, 1086–1089. [Google Scholar] [CrossRef]
- Bamdad, F.; Goli, A.H.; Kadivar, M. Preparation and characterization of proteinous film from lentil (Lens culinaris): Edible film from lentil (Lens culinaris). Food Res. Int. 2006, 39, 106–111. [Google Scholar] [CrossRef]
- Cutter, C.N.; Susan, S.S. Application of Edible Coatings on Muscle Foods. In Protein Based Films and Coatings; Gennadios, A., Ed.; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
- Gennadios, A.; Weller, C.L.; Testin, R.F. Modification of physical and barrier properties of edible wheat gluten-based films. Cereal Chem. 1993, 70, 426–429. [Google Scholar]
- Tang, C.-H.; Xiao, M.-L.; Chen, Z.; Yang, X.-Q. Influence of succinylation on the properties of cast films from red bean protein isolate at various plasticizer levels. J. Appl. Polym. Sci. 2011, 120, 1934–1941. [Google Scholar] [CrossRef]
- Tsoukala, A.; Papalamprou, E.; Makri, E.; Doxastakis, G.; Braudo, E.E. Adsorption at the air-water interface and emulsification properties of grain legume protein derivatives from pea and broad bean. Coll. Surf. B Biointerfaces 2006, 53, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Lange, J.; Wyser, Y. Recent innovations in barrier technologies for plastic packaging—A review. Packag. Technol. Sci. 2003, 16, 149–158. [Google Scholar] [CrossRef]
- Farris, S.; Cozzolino, C.A.; Introzzi, L.; Piergiovanni, L. Development and characterization of a gelatin-based coating with unique sealing properties. J. Appl. Polym. Sci. 2010, 118, 2969–2975. [Google Scholar] [CrossRef]
- Gallstedt, M.; Brottman, A.; Hedenqvist, M.S. Packagin-related properties of protein- and chitosan-coated paper. Packag. Technol. Sci. 2005, 18, 161–170. [Google Scholar] [CrossRef]
- Han, J.H.; Krochta, J.M. Wetting properties and water vapor permeability of whey-protein-coated paper. Trans. ASAE 1999, 42, 1375–1382. [Google Scholar] [CrossRef]
- Hong, S.I.; Krochta, J.M. Oxygen barrier properties of whey protein isolate coatings on polypropylene films. J. Food Sci. 2003, 68, 224–228. [Google Scholar] [CrossRef]
- Hong, S.I.; Krochta, J.M. Whey protein isolate coating on LDPE film as a novel oxygen barrier in the composite structure. Packag. Technol. Sci. 2004, 17, 13–21. [Google Scholar] [CrossRef]
- Farris, S.; Introzzi, L.; Piergiovanni, L. Evaluation of a bio-coating as a solution to improve barrier, friction and optical properties of plastic films. Packag. Technol. Sci. 2009, 22, 69–83. [Google Scholar] [CrossRef]
- Trezza, T.A.; Wiles, J.L.; Vergano, P.J. Water vapor and oxygen barrier properties of corn zein coated paper. Tappi J. 1998, 81, 171–176. [Google Scholar]
- Tihminlioglu, F.; Atik, I.D.; Ozen, B. Water vapor and oxygen-barrier performance of corn-zein coated polypropylene films. J. Food Eng. 2010, 96, 342–347. [Google Scholar] [CrossRef]
- Schmid, M.; Cinelli, P.; Wildner, J.; Bazzichi, A.; Lazzeri, A. Whey protein based barrier layers to enhance the barrier properties in polylactic acid films while maintaining biodegradability. In Proceeding of the Bipoco 2012 International Conference on Bio-Based Polymers and Composites, Siofok, Hungary, 27–31 May 2012.
- Fuss, C.; Palmaz, J.; Sprague, E. Fibrinogen: Structure, function, and surface interactions. J. Vasc. Int. Radiol. 2001, 12, 677–682. [Google Scholar] [CrossRef]
- Walter, B.K.; Bainbridge, N.Y. Casein Adhesive Composition and Laminated Structure Utilizing the Same. US Patent 2754240, 10 July 1956. [Google Scholar]
- Cheng, E.; Sun, X.; Karr, G.S. Adhesive properties of modified soybean flour in wheat straw particle board. Compos. Part A Appl. Sci. Manufac. 2004, 35, 297–302. [Google Scholar] [CrossRef]
- Kalapathy, U.; Hettiarachchy, N.; Myers, D.; Hanna, M. Modification of soy proteins and their adhesive properties on woods. J. Am. Oil Chem. Soc. 1995, 72, 507–510. [Google Scholar] [CrossRef]
- Wang, G.; Guo, M. Property and storage stability of whey protein-sucrose based safe paper glue. J. Appl. Polym. Sci. 2014, 131, 39710:1–39710:8. [Google Scholar] [CrossRef]
- Gennadios, A.; Hanna, M.A.; Kurth, L.B. Application of edible coatings on meats, poultry and seafoods: A review. LWT-Food Sci. Technol. 1997, 30, 337–350. [Google Scholar] [CrossRef]
- Baldwin, E.A.; Hagenmaier, R.D.; Bai, J. Edible Coatings and Films to Improve Food Quality, 2nd Edition ed; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Hettiarachchy, N.S.; Ziegler, G.R. Protein Functionality in Food Systems; Marcel Dekker: New York, NY, USA, 1994. [Google Scholar]
- Shih, F.F. Film-forming properties and edible films of plant proteins. Food/Nahrung 1998, 42, 254–256. [Google Scholar] [CrossRef]
- Gennadios, A. Protein-Based Films and Coatings; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
- Bourtoom, T. Review Article. Edible protein films: Properties enhancement. Int. Food Res. J. 2009, 16, 1–9. [Google Scholar]
- Scopes, R.K. Protein Purification: Principles and Practice; Springer-Verlag: New York, NY, USA, 1994. [Google Scholar]
- Cheftel, J.C. Nutritional effects of extrusion-cooking. Food Chem. 1986, 20, 263–283. [Google Scholar] [CrossRef]
- Damodaran, S.; Kinsella, J.E. Effect of conglycinin on the thermal aggregation of glycinin. J. Agric. Food Chem. 1982, 30, 812–817. [Google Scholar] [CrossRef]
- Barone, J.R.; Schmidt, W.F. Nonfood applications of proteinaceous renewable materials. J. Chem. Educ. 2006, 83, 1003. [Google Scholar] [CrossRef]
- Schmid, M. Properties of cast films made from different ratios of whey protein isolate, hydrolysed whey protein isolate and glycerol. Materials 2013, 6, 3254–3269. [Google Scholar] [CrossRef]
- Schmid, M.; Dallmann, K.; Bugnicourt, E.; Cordoni, D.; Wild, F.; Lazzeri, A.; Noller, K. Properties of whey-protein-coated films and laminates as novel recyclable food packaging materials with excellent barrier properties. Int. J. Polym. Sci. 2012, 2012, 562381:1–562381:7. [Google Scholar] [CrossRef]
- Schmid, M.; Noller, K.; Wild, F.; Bugnicourt, E. Whey Protein Coated Films. Pantent WO2013014493 A1, 2013. [Google Scholar]
- Lieberma, E.R.; Gilbert, S.G. Gas permeation of collagen films as affected by crosslinkage, moisture, and plasticizer content. J. Polym. Sci. Part C Polym. Symp. 1973, 41, 33–43. [Google Scholar] [CrossRef]
- Tanaka, M.; Iwata, K.; Sanguandeekul, R.; Handa, A.; Ishizaki, S. Influence of plasticizers on the properties of edible films prepared from fish water-soluble proteins. Fish. Sci. 2001, 67, 346–351. [Google Scholar] [CrossRef]
- McHugh, T.H.; Krochta, J.M. Water-vapor permeability properties of edible whey protein-lipid emulsion films. J. Am. Oil Chem. Soc. 1994, 71, 307–312. [Google Scholar] [CrossRef]
- Martin-Polo, M.; Mauguin, C.; Voilley, A. Hydrophobic films and their efficiency against moisture transfer. 1. Influence of the film preparation technique. J. Agric. Food Chem. 1992, 40, 407–412. [Google Scholar] [CrossRef]
- Bosquez-Molina, E.; Guerrero-Legarreta, I.; Vernon-Carter, E.J. Moisture barrier properties and morphology of mesquite gum-candelilla wax based edible emulsion coatings. Food Res. Int. 2003, 36, 885–893. [Google Scholar] [CrossRef]
- Kamper, S.L.; Fennema, O. Use of an edible film to maintain water vapor gradients in foods. J. Food Sci. 1985, 50, 382–384. [Google Scholar] [CrossRef]
- Weller, C.L.; Gennadios, A.; Saraiva, R.A. Edible bilayer films from Zein and grain sorghum wax or carnauba wax. LWT-Food Sci. Technol. 1998, 31, 279–285. [Google Scholar] [CrossRef]
- Chick, J.; Hernandez, R.J. Physical, thermal, and barrier characterization of casein-wax-based edible films. J. Food Sci. 2002, 67, 1073–1079. [Google Scholar] [CrossRef]
- Rodriguez-Turienzo, L.; Cobos, A.; Diaz, O. Effects of microbial transglutaminase added edible coatings based on heated or ultrasound-treated whey proteins in physical and chemical parameters of frozen Atlantic salmon (Salmo salar). J. Food Eng. 2013, 119, 433–438. [Google Scholar] [CrossRef]
- Vachon, C.; Yu, H.L.; Yefsah, R.; Alain, R.; St-Gelais, D.; Lacroix, M. Mechanical and structural properties of milk protein edible films cross-linked by heating and gamma-irradiation. J. Agric. Food Chem. 2000, 48, 3202–3209. [Google Scholar] [CrossRef] [PubMed]
- Winkler, H.; Vorwerg, W.; Schmid, M. Synthesis of hydrophobic whey protein isolate by acylation with fatty acids. Eur. Polym. J. 2015, 62, 10–18. [Google Scholar] [CrossRef]
- Santos, C.V.; Tomasula, P.M. Acylation and solubility of casein precipitated by carbon dioxide. J. Food Sci. 2000, 65, 227–230. [Google Scholar] [CrossRef]
- Orliac, O.; Rouilly, A.; Silvestre, F.; Rigal, L. Effects of various plasticizers on the mechanical properties, water resistance and aging of thermo-moulded films made from sunflower proteins. Ind. Crops Prod. 2003, 18, 91–100. [Google Scholar] [CrossRef]
- Bertoldo, M.; Bronco, S.; Gragnoli, T.; Ciardelli, F. Modification of Gelatin by Reaction with 1,6-Diisocyanatohexane. Macromol. Biosci. 2007, 7, 328–338. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Muñoz, P.; Villalobos, R.; Chiralt, A. Effect of cross-linking using aldehydes on properties of glutenin-rich films. Food Hydrocoll. 2004, 18, 403–411. [Google Scholar] [CrossRef]
- DeJong, G.A.H.; Koppelman, S.J. Transglutaminase catalyzed reactions: Impact on food applications. J. Food Sci. 2002, 67, 2798–2806. [Google Scholar] [CrossRef]
- Porta, R.; Mariniello, L.; di Pierro, P.; Sorrentino, A.; Giosafatto, C.V.L. Transglutaminase crosslinked pectin- and chitosan-based edible films: A review. Crit. Rev. Food Sci. Nutr. 2011, 51, 223–238. [Google Scholar] [CrossRef] [PubMed]
- Mariniello, L.; Porta, R. Transglutaminases as Biotechnological Tools. In Transglutaminases: Family of Enzymes with Diverse Functions; Mehta, K., Eckert, R., Eds.; Karger: Basel, Switzerland, 2003; pp. 174–191. [Google Scholar]
- Gaber, M.H. Effect of gamma-irradiation on the molecular properties of bovine serum albumin. J. Biosci. Bioeng. 2005, 100, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Durchschlag, H.; Fochler, L.C.; Feser, I.B.; Hausmann, L.S.; Seroneit, T.; Swlentek, J.M.; Swoboda, L.E.; Wlnklmair, L.A.; WlEkL, L.C.; Zipper, P. Effects of X- and UV-irradiation on proteins. Radiat. Phys. Chem. 1996, 47, 501–505. [Google Scholar] [CrossRef]
- Tracton, A.A. Coatings Technology Handbook; Taylor and Francis: Boca Raton, FL, USA, 2005. [Google Scholar]
- Embuscado, M.E.; Huber, K.C. Edible Films and Coatings for Food Applications; Springer: Dordrecht, The Netherlands; New York, NY, USA, 2009. [Google Scholar]
- Perez-Gago, M.B.; Krochta, J.M. Drying temperature effect on water vapor permeability and mechanical properties of whey protein-lipid emulsion films. J. Agric. Food Chem. 2000, 48, 2687–2692. [Google Scholar] [CrossRef] [PubMed]
- Perez-Gago, M.B.; Krochta, J.M. Denaturation time and temperature effects on solubility, tensile properties, and oxygen permeability of whey protein edible films. J. Food Sci. 2001, 66, 705–710. [Google Scholar] [CrossRef]
- Plackett, D. Biopolymers: New Materials for Sustainable Films and Coatings; Wiley: Chichester, West Sussex, UK; Hoboken, NJ, USA, 2011. [Google Scholar]
- Lent, L.E.; Vanasupa, L.S.; Tong, P.S. Whey protein edible film structures determined by atomic force microscope. J. Food Sci. 1998, 63, 824–827. [Google Scholar] [CrossRef]
- Mahmoud, R.; Savello, P.A. Solubility and hydrolyzability of films produced by transglutaminase catalytic cross-linking of whey-protein. J. Dairy Sci. 1993, 76, 29–35. [Google Scholar] [CrossRef]
- Bugnicourt, E.; Schmid, M.; Mc Nerney, O.; Wildner, J.; Smykala, L.; Lazzeri, A.; Cinelli, P. Processing and validation of whey-protein-coated films and laminates at semi-industrial scale as novel recyclable food packaging materials with excellent barrier properties. Adv. Mater. Sci. Eng. 2013, 2013, 496207:1–496207:10. [Google Scholar] [CrossRef]
- Kowalczyk, D.; Baraniak, B. Effects of plasticizers, pH and heating of film-forming solution on the properties of pea protein isolate films. J. Food Eng. 2011, 105, 295–305. [Google Scholar] [CrossRef]
- Verbeek, C.J.R.; van den Berg, L.E. Extrusion processing and properties of protein-based thermoplastics. Macromol. Mater. Eng. 2010, 295, 10–21. [Google Scholar] [CrossRef]
- Rouilly, A.; Mériaux, A.; Geneau, C.; Silvestre, F.; Rigal, L. Film extrusion of sunflower protein isolate. Polym. Eng. Sci. 2006, 46, 1635–1640. [Google Scholar] [CrossRef] [Green Version]
- Hanani, Z.A.N.; Beatty, E.; Roos, Y.H.; Morris, M.A.; Kerry, J.P. Manufacture and characterization of gelatin films derived from beef, pork and fish sources using twin screw extrusion. J. Food Eng. 2012, 113, 606–614. [Google Scholar] [CrossRef]
- Hanani, Z.A.N.; Beatty, E.; Roos, Y.H.; Kerry, J.P. Manufacture of Gelatin-Based Films Using Extrusion: Assessment of Extrusion Parameters on Film Properties. In Presented at the 11th International Congress on Engineering and Food, Athens, Greece, 22–26 May 2011.
- Krishna, M.; Nindo, C.I.; Min, S.C. Development of fish gelatin edible films using extrusion and compression molding. J. Food Eng. 2012, 108, 337–344. [Google Scholar] [CrossRef]
- Hernandez-Izquierdo, V.M. Thermal transitions, extrusion, and heat-sealing of whey protein edible films. Ph.D. Thesis, University of California, Davis, CA, USA, 2007. [Google Scholar]
- Hernandez-Izquierdo, V.M.; Krochta, J.M. Thermoplastic processing of proteins for film formation—A review. J. Food Sci. 2008, 73, R30–R39. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Izquierdo, V.M.; Krochta, J.M. Thermal transitions and heat-sealing of glycerol-plasticized whey protein films. Packag. Technol. Sci. 2009, 22, 255–260. [Google Scholar] [CrossRef]
- Hernandez-Izquierdo, V.M.; Reid, D.S.; McHugh, T.H.; De J. Berrios, J.; Krochta, J.M. Thermal transitions and extrusion of glycerol-plasticized whey protein mixtures. J. Food Sci. 2008, 73, E169–E175. [Google Scholar] [CrossRef] [PubMed]
- Dangaran, K.; Tomasula, P.M.; Qi, P. Structure and function of protein-based edible films and coatings. In Edible Films and Coatings for Food Applications; Embuscado, M.E., Kerry, C.H., Eds.; Springer: Dordrecht, The Netherlands; New York, NY, USA, 2009; pp. 25–56. [Google Scholar]
- Zhang, H.; Mittal, G. Biodegradable protein-based films from plant resources: A review. Environ. Prog. Sustain. Energy 2010, 29, 203–220. [Google Scholar] [CrossRef]
- Barone, J.R.; Schmidt, W.F.; Gregoire, N.T. Extrusion of feather keratin. J. Appl. Polym. Sci. 2006, 100, 1432–1442. [Google Scholar] [CrossRef]
- Redl, A.; Morel, M.H.; Bonicel, J.; Guilbert, S.; Vergnes, B. Rheological properties of gluten plasticized with glycerol: Dependence on temperature, glycerol content and mixing conditions. Rheol. Acta 1999, 38, 311–320. [Google Scholar] [CrossRef]
- Tostoguzov, V.B. Some physico-chemical aspects of protein processing in foods. Multicomponent gels. Food Hydrocoll. 1995, 9, 317–332. [Google Scholar] [CrossRef]
- Sothornvit, R.; Krochta, J.M. Plasticizer effect on oxygen permeability of beta-lactoglobulin films. J. Agric. Food Chem. 2000, 48, 6298–6302. [Google Scholar] [CrossRef] [PubMed]
- Sothornvit, R.; Krochta, J.M. Plasticizer effect on mechanical properties of beta-lactoglobulin films. J. Food Eng. 2001, 50, 149–155. [Google Scholar] [CrossRef]
- Nguyen, H.A.; Wong, M.; Anema, S.G.; Havea, P.; Guyomarch, F. Effects of adding low levels of a disulfide reducing agent on the disulfide interactions of β-lactoglobulin and κ-casein in skim milk. J. Agric. Food Chem. 2012, 60, 2337–2342. [Google Scholar] [CrossRef] [PubMed]
- Jansens, K.J.A.; Bruyninckx, K.; Redant, L.; Lagrain, B.; Brijs, K.; Goderis, B.; Smet, M.; Delcour, J.A. Importance of cross-linking and disulfide bridge reduction for the mechanical properties of rigid wheat gluten bioplastics compression molded with thiol and/or disulfide functionalized additives. J. Appl. Polym. Sci. 2014, 131. [Google Scholar] [CrossRef]
- Schmid, M.; Müller, K.; Sängerlaub, S.; Stäbler, A.; Starck, V.; Ecker, F.; Noller, K. Mechanical and barrier properties of thermoplastic whey protein isolate/ethylene vinyl acetate blends. J. Appl. Polym. Sci. 2014, 131. [Google Scholar] [CrossRef]
- Aithani, D.; Mohanty, A.K. Value-added new materials from byproduct of corn based ethanol industries: Blends of plasticized corn gluten meal and poly(β-caprolactone). Ind. Eng. Chem. Res. 2006, 45, 6147–6152. [Google Scholar] [CrossRef]
- Mariani, P.D.S.C.; Allganer, K.; Oliveira, F.B.; Cardoso, E.J.B.N.; Innocentini-Mei, L.H. Effect of soy protein isolate on the thermal, mechanical andmorphological properties of poly (β-caprolactone) and corn starch blends. Polym. Test. 2009, 28, 824–829. [Google Scholar] [CrossRef]
- Ghosh, A.; Azam Ali, M.; Selvanesan, L.; Dias, G.J. Structure–function characteristics of the biomaterials based on milk-derived proteins. Int. J. Biol. Macromol. 2010, 46, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Leman, J.; Kinsella, J.E. Surface-activity, film formation, and emulsifying properties of milk-proteins. Crit. Rev. Food Sci. Nutr. 1989, 28, 115–138. [Google Scholar] [CrossRef] [PubMed]
- Mark, H.F.; Kroschwitz, J.I. Encyclopedia of Polymer Science and Engineering; Wiley: New York, NY, USA, 1985. [Google Scholar]
- Avena-Bustillos, R.J.; Krochta, J.M. Water-vapor permeability of caseinate-based edible films as affected by pH, calcium cross-linking and lipid-content. J. Food Sci. 1993, 58, 904–907. [Google Scholar] [CrossRef]
- Brault, D.; Daprano, G.; Lacroix, M. Formation of free-standing sterilized edible films from irradiated caseinates. J. Agric. Food Chem. 1997, 45, 2964–2969. [Google Scholar] [CrossRef]
- Mchugh, T.H.; Krochta, J.M. Permeability Properties of Edible Films. In Edible Coatings and Films to Improve Food Quality; Baldwin, E.A., Hegenmaier, R., Bai, J., Eds.; CRC Press: Boca Raton, FL, USA, 1994; pp. 139–187. [Google Scholar]
- Guilbert, S.; Gontard, N.; Cuq, B. Technology and applications of edible protective films. Packag. Technol. Sci. 1995, 8, 339–346. [Google Scholar] [CrossRef]
- Khwaldia, K.; Banon, S.; Desobry, S.; Hardy, J. Mechanical and barrier properties of sodium caseinate-anhydrous milk fat edible films. Int. J. Food Sci. Technol. 2004, 39, 403–411. [Google Scholar] [CrossRef]
- Miller, K.S.; Krochta, J.M. Oxygen and aroma barrier properties of edible films: A review. Trends Food Sci. Technol. 1997, 8, 228–237. [Google Scholar] [CrossRef]
- Pereda, M.; Aranguren, M.I.; Marcovich, N.E. Caseinate films modified with tung oil. Food Hydrocoll. 2010, 24, 800–808. [Google Scholar] [CrossRef]
- Fabra, M.J.; Talens, P.; Chiralt, A. Effect of alginate and lambda-carrageenan on tensile properties and water vapor permeability of sodium caseinate-lipid based films. Carbohydr. Polym. 2008, 74, 419–426. [Google Scholar] [CrossRef]
- Fabra, J.M.; Talens, P.; Chiralt, A. Influence of calcium on tensile, optical and water vapor permeability properties of sodium caseinate edible films. J. Food Eng. 2010, 96, 356–364. [Google Scholar] [CrossRef]
- Aliheidari, N.; Fazaeli, M.; Ahmadi, R.; Ghasemlou, M.; Emam-Djomeh, Z. Comparative evaluation on fatty acid and Matricaria recutita essential oil incorporated into casein-based film. International. J. Biol. Macromol. 2013, 56, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Khwaldia, K.; Linder, M.; Banon, S.; Desobry, S. Effects of mica, carnauba wax, glycerol, and sodium caseinate concentrations on water vapor barrier and mechanical properties of coated paper. J. Food Sci. 2005, 70, E192–E197. [Google Scholar] [CrossRef]
- Chick, J.; Ustunol, Z. Mechanical and barrier properties of lactic acid and rennet precipitated casein-based edible films. J. Food Sci. 1998, 63, 1024–1027. [Google Scholar] [CrossRef]
- Banerjee, R.; Chen, H.; Wu, J. Milk protein-based edible film mechanical strength changes due to ultrasound process. J. Food Sci. 1996, 61, 824–828. [Google Scholar] [CrossRef]
- Gennadios, A.; McHugh, T.H.; Weller, C.L.; Krochta, J.M. Edible Coatings and Films Based on Proteins. In Edible Coating and Films to Improve Food Quality; Krochta, J.M., Baldwin, E.A., Nisperos-Carriedo, M.O., Eds.; Technomic Publ. Co.: Lancaster, PA, USA, 1994; pp. 201–277. [Google Scholar]
- Bruno, M.; Giancone, T.; Torrieri, E.; Masi, P.; Moresi, M. Engineering properties of edible transglutaminase cross-linked caseinate-based films. Food Bioprocess Technol. 2008, 1, 393–404. [Google Scholar] [CrossRef]
- Mezgheni, E.; D’Aprano, G.; Lacroix, M. Formation of sterilized edible films based on caseinates: Effects of calcium and plasticizers. J. Agric. Food Chem. 1998, 46, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Ressouany, M.; Vachon, C.; Lacroix, M. Irradiation dose and calcium effect on the mechanical properties of cross-linked caseinate films. J. Agric. Food Chem. 1998, 46, 1618–1623. [Google Scholar] [CrossRef]
- Fabra, M.J.; Talens, P.; Chiralt, A. Microstructure and optical properties of sodium caseinate films containing oleic acid-beeswax mixtures. Food Hydrocoll. 2009, 23, 676–683. [Google Scholar] [CrossRef]
- Mezgheni, E.; Vachon, C.; Lacroix, M. Biodegradability behavior of cross-linked calcium caseinate films. Biotechnol. Prog. 1998, 14, 534–536. [Google Scholar] [CrossRef] [PubMed]
- Seydim, A.C.; Sarikus, G. Antimicrobial activity of whey protein based edible films incorporated with oregano, rosemary and garlic essential oils. Food Res. Int. 2006, 39, 639–644. [Google Scholar] [CrossRef]
- Herrmann, P.S.D.; Yoshida, C.M.P.; Antunes, A.J.; Marcondes, J.A. Surface evaluation of whey protein films by atomic force microscopy and water vapor permeability analysis. Packag. Technol. Sci. 2004, 17, 267–273. [Google Scholar] [CrossRef]
- Boland, M. Whey Proteins. In Handbook of Food Proteins; Phillips, G.O., Williams, P.A., Eds.; Woodhead Publishing: Cambridge, UK, 2011; pp. 30–55. [Google Scholar]
- Elgar, D.F.; Norris, C.S.; Ayers, J.S.; Pritchard, M.; Otter, D.E.; Palmano, K.P. Simultaneous separation and quantitation of the major bovine whey proteins including proteose peptone and caseinomacropeptide by reversed-phase high-performance liquid chromatography on polystyrene–divinylbenzene. J. Chromatogr. A 2000, 878, 183–196. [Google Scholar] [CrossRef]
- Dunnill, P.; Green, D.W. Sulphydryl groups and conformational change in β-lactoglobulin. J. Mol. Biol. 1966, 15, 147–151. [Google Scholar] [CrossRef]
- Priestley, R.J. Effects of Heating on Foodstuffs; Applied Science Publishers: London, UK, 1979. [Google Scholar]
- Dewit, J.N.; Klarenbeek, G. Effects of various heat-treatments on structure and solubility of whey proteins. J. Dairy Sci. 1984, 67, 2701–2710. [Google Scholar] [CrossRef]
- Shimada, K.; Cheftel, J.C. Sulphydryl group/disulfide bond interchange reactions during heat-induced gelation of whey protein isolate. J. Agric. Food Chem. 1989, 37, 161–168. [Google Scholar] [CrossRef]
- Langton, M.; Hermansson, A.M. Fine-stranded particulate gels of β-lactoglobulin and whey protein at varying pH. Food Hydrocoll. 1992, 5, 523–539. [Google Scholar] [CrossRef]
- Belitz, H.D.; Grosch, W.; Schieberle, P. Lehrbuch der Lebensmittelchemie; Springer: Berlin, Germany, 2007. (In German) [Google Scholar]
- Onwulata, C.; Huth, P. Whey Processing, Functionality and Health Benefits; Wiley-Blackwell: Ames, IA, USA, 2008. [Google Scholar]
- Cinelli, P.; Schmid, M.; Bugnicourt, E.; Wildner, J.; Bazzichi, A.; Anguillesi, I.; Lazzeri, A. Whey protein layer applied on biodegradable packaging film to improve barrier properties while maintaining biodegradability. Polym. Degrad. Stable 2014, 108, 151–157. [Google Scholar] [CrossRef]
- Hamman, F.; Schmid, M. Determination and quantification of molecular interactions in protein films: A review. Materials 2014, 7, 7975–7996. [Google Scholar] [CrossRef]
- Schmid, M.; Eib, S.; Reinlet, M. Modeling, simulation, and experimental validation of drying and denaturation behavior of whey protein isolate-based coatings. Dry. Technol. 2015, 33, 1382–1395. [Google Scholar] [CrossRef]
- Schmid, M.; Hinz, L.-V.; Wild, F.; Noller, K. Effects of hydrolysed whey proteins on the techno-functional characteristics of whey protein-based films. Materials 2013, 6, 927–940. [Google Scholar] [CrossRef]
- Schmid, M.; Reichert, K.; Hammann, F.; Stäbler, A. Storage time-dependent alteration of molecular interaction—Property relationships of whey protein isolate-based films and coatings. J. Mater. Sci. 2015, 12, 4396–4404. [Google Scholar] [CrossRef]
- Schmid, M.; Herbst, C.; Müller, K.; Stäbler, A.; Schlemmer, D.; Coltelli, M.B.; Lazzeri, A. Effect of potato pulp filler on the mechanical properties and water vapor transmission rate of thermoplastic WPI/PBS blends. Polym.-Plast. Technol. Eng. 2015. [Google Scholar] [CrossRef]
- Rossi Marquez, G.; di Pierro, P.; Esposito, M.; Mariniello, L.; Porta, R. Application of transglutaminase-crosslinked whey protein/pectin films as water barrier coatings in fried and baked foods. Food Bioprocess Technol. 2014, 7, 447–455. [Google Scholar] [CrossRef]
- Perez-Gago, M.B.; Krochta, J.M. Water vapor permeability of whey protein emulsion films as affected by pH. J. Food Sci. 1999, 64, 695–698. [Google Scholar] [CrossRef]
- Shellhammer, T.H.; Krochta, J.M. Whey protein emulsion film performance as affected by lipid type and amount. J. Food Sci. 1997, 62, 390–394. [Google Scholar] [CrossRef]
- Chan, M.A.; Krochta, J.M. Grease and oxygen barrier properties of whey protein isolate coated paperboard. TAPPI Solut. 2001, 84, 1–11. [Google Scholar]
- Lin, S.Y.D.; Krochta, J.M. Whey protein coating efficiency on surfactant-modified hydrophobic surfaces. J. Agric. Food Chem. 2005, 53, 5018–5023. [Google Scholar] [CrossRef] [PubMed]
- Shaw, N.B.; Monahan, F.J.; O’Riordan, E.D.; O’Sullivan, M. Physical properties of WPI films plasticized with glycerol, xylitol, or sorbitol. J. Food Sci. 2002, 67, 164–167. [Google Scholar] [CrossRef]
- Ustunol, Z.; Mert, B. Water solubility, mechanical, barrier, and thermal properties of cross-linked whey protein isolate-based films. J. Food Sci. 2004, 69, E129–E133. [Google Scholar] [CrossRef]
- Galietta, G.; di Gioia, L.; Guilbert, S.; Cuq, B. Mechanical and thermomechanical properties of films based on whey proteins as affected by plasticizer and cross-linking agents. J. Dairy Sci. 1998, 81, 3123–3130. [Google Scholar] [CrossRef]
- Fairley, P.; Monahan, F.J.; German, J.B.; Krochta, J.M. Mechanical properties and water vapor permeability of edible films from whey protein isolate and sodium dodecyl sulfate. J. Agric. Food Chem. 1996, 44, 438–443. [Google Scholar] [CrossRef]
- Wihodo, M.; Moraru, C.I. Physical and chemical methods used to enhance the structure and mechanical properties of protein films: A review. J. Food Eng. 2013, 114, 292–302. [Google Scholar] [CrossRef]
- Tang, C.-H.; Jiang, Y. Modulation of mechanical and surface hydrophobic properties of food protein films by transglutaminase treatment. Food Res. Int. 2007, 40, 504–509. [Google Scholar] [CrossRef]
- Li, J.C.; Chen, H.D. Biodegradation of whey protein-based edible films. J. Polym. Environ. 2000, 8, 135–143. [Google Scholar] [CrossRef]
- Gómez-Guillén, M.C.; Giménez, B.; López-Caballero, M.E.; Montero, M.P. Functional and bioactive properties of collagen and gelatin from alternative sources: A review. Food Hydrocoll. 2011, 25, 1813–1827. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Auty, M.A.E.; Rau, A.; Kerry, J.F.; Kerry, J.P. Effect of pH and addition of corn oil on the properties of gelatin-based biopolymer films. J. Food Eng. 2009, 90, 11–19. [Google Scholar] [CrossRef]
- Pulieri, E.; Chiono, V.; Ciardelli, G.; Vozzi, G.; Ahluwalia, A.; Domenici, C.; Vozzi, F.; Giusti, P. Chitosan/gelatin blends for biomedical applications. J. Biomed. Mater. Res. Part A 2008, 86, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Avena-Bustillos, R.J.; Chiou, B.; Olsen, C.W.; Bechtel, P.J.; Olson, D.A.; McHugh, T.H. Gelation, oxygen permeability, and mechanical properties of mammalian and fish gelatin films. J. Food Sci. 2011, 76, E519–E524. [Google Scholar] [CrossRef] [PubMed]
- Arvanitoyannis, I.; Psomiadou, E.; Nakayama, A.; Aiba, S.; Yamamoto, N. Edible films made from gelatin, soluble starch and polyols, Part 3. Food Chem. 1997, 60, 593–604. [Google Scholar] [CrossRef]
- Rubin, A.L.; Riggio, R.R.; Nachman, R.L.; Schwartz, G.H.; Miyata, T.; Stenzeel, K.H. Collagen materials in dialysis and implantation. Trans. Am. Soc. Artif. Intern. Organs 1968, 14, 169–175. [Google Scholar] [PubMed]
- Gennadios, A.; Rhim, J.W.; Handa, A.; Weller, C.L.; Hanna, M.A. Ultraviolet radiation affects physical and molecular properties of soy protein films. J. Food Sci. 1998, 63, 225–228. [Google Scholar] [CrossRef]
- Lim, L.T.; Mine, Y.; Tung, M.A. Barrier and tensile properties of transglutaminase cross-linked gelatin films as affected by relative humidity, temperature, and glycerol content. J. Food Sci. 1999, 64, 616–622. [Google Scholar] [CrossRef]
- De Carvalho, R.A.; Grosso, C.R.F. Characterization of gelatin based films modified with transglutaminase, glyoxal and formaldehyde. Food Hydrocoll. 2004, 18, 717–726. [Google Scholar] [CrossRef]
- Ahmad, M.; Benjakul, S.; Prodpran, T.; Agustini, T.W. Physico-mechanical and antimicrobial properties of gelatin film from the skin of unicorn leatherjacket incorporated with essential oils. Food Hydrocoll. 2012, 28, 189–199. [Google Scholar] [CrossRef]
- Amiza, M.A.; Wong, Y.P. Effect of Palm Oil Incorporation on the Properties of Biodegradable Cobia (Rachycentron Canadum) Skin Gelatin Films. In Gums and Stabilisers for the Food Industry 16; Williams, P.A., Phillips, G.O., Eds.; RSC Publishing: Cambridge, UK, 2012; pp. 269–277. [Google Scholar]
- Andreuccetti, C.; Carvalho, R.A.; Grosso, C.R.F. Effect of hydrophobic plasticizers on functional properties of gelatin-based films. Food Res. Int. 2009, 42, 1113–1121. [Google Scholar] [CrossRef]
- Condés, M.C.; Añon, M.C.; Mauri, A.N. Amaranth protein films from thermally treated proteins. J. Food Eng. 2013, 119, 573–579. [Google Scholar] [CrossRef]
- Alves, P.M.A.; Carvalho, R.A.; Moraes, I.C.F.; Luciano, C.G.; Bittante, A.M.Q.B.; Sobral, P.J.A. Development of films based on blends of gelatin and poly(vinyl alcohol) cross linked with glutaraldehyde. Food Hydrocoll. 2011, 25, 1751–1757. [Google Scholar] [CrossRef]
- Chambi, H.; Grosso, C. Edible films produced with gelatin and casein cross-linked with transglutaminase. Food Res. Int. 2006, 39, 458–466. [Google Scholar] [CrossRef]
- Maria, T.M.C.; de Carvalho, R.A.; Sobral, P.J.A.; Habitante, A.M.B.Q.; Solorza-Feria, J. The effect of the degree of hydrolysis of the PVA and the plasticizer concentration on the color, opacity, and thermal and mechanical properties of films based on PVA and gelatin blends. J. Food Eng. 2008, 87, 191–199. [Google Scholar] [CrossRef]
- Rawdkuen, S.; Suthiluk, P.; Kamhangwong, D.; enjakul, S. Mechanical, physico-chemical, and antimicrobial properties of gelatin-based film incorporated with catechin-lysozyme. Chem. Cent. J. 2012, 6. [Google Scholar] [CrossRef] [PubMed]
- Cao, N.; Yang, X.; Fu, Y. Effects of various plasticizers on mechanical and water vapor barrier properties of gelatin films. Food Hydrocoll. 2009, 23, 729–735. [Google Scholar] [CrossRef]
- Vanin, F.M.; Sobral, P.J.A.; Menegalli, F.C.; Carvalho, R.A.; Habitante, A. Effects of plasticizers and their concentrations on thermal and functional properties of gelatin-based films. Food Hydrocoll. 2005, 19, 899–907. [Google Scholar] [CrossRef]
- Frohberg, P.; Pietzsch, M.; Ulrich, J. Effect of crystalline substances in biodegradable films. Chem. Eng. Res. Des. 2010, 88, 1148–1152. [Google Scholar] [CrossRef]
- Sartori, S.; Rechichi, A.; Vozzi, G.; D’Acunto, M.; Heine, E.; Giusti, P.; Ciardelli, G. Surface modification of a synthetic polyurethane by plasma glow discharge: Preparation and characterization of bioactive monolayers. React. Funct. Polym. 2008, 68, 809–821. [Google Scholar] [CrossRef]
- Jabrane, T.; Dube, M.; Griffiths, M.; Mangin, P.J. Towards a commercial production of phage-based bioactive paper. J. Sci. Technol. For. Prod. Process. 2011, 1, 6–13. [Google Scholar]
- Abrusci, C.; Martın-Gonzalez, A.; Del Amo, A.; Corrales, T.; Catalina, F. Biodegradation of type-B gelatine by bacteria isolated from cinematographic films. A viscometric study. Polym. Degrad. Stab. 2004, 86, 283–291. [Google Scholar] [CrossRef]
- Gennadios, A.; Weller, C.L. Edible films and coatings from wheat and corn proteins. Food Technol. 1990, 44, 63–69. [Google Scholar]
- Kieffer, R.; Schurer, F.; Köhler, P.; Wieser, H. Effect of hydrostatic pressure and temperature on the chemical and functional properties of wheat gluten: Studies on gluten, gliadin and glutenin. J. Cereal Sci. 2007, 45, 285–292. [Google Scholar] [CrossRef]
- Reddy, N.; Yang, Y. Novel protein fibers from wheat gluten. Biomacromolecules 2007, 8, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Shewry, P.R.; Tatham, A.S. Disulphide Bonds in wheat gluten proteins. J. Cereal Sci. 1997, 25, 207–227. [Google Scholar] [CrossRef]
- Wieser, H. Chemistry of gluten proteins. Food Microbiol. 2007, 24, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Leon, A.; Rosell, C.A.; de Barber, C.B. A differential scanning calorimetry study of wheat proteins. Eur. Food Res. Technol. 2003, 217, 13–16. [Google Scholar] [CrossRef]
- Van Der Borght, A.; Goesaert, H.; Veraverbeke, W.S.; Delcour, J.A. Fractionation of wheat and wheat flour into starch and gluten: Overview of the main processes and the factors involved. J. Cereal Sci. 2005, 41, 221–237. [Google Scholar] [CrossRef]
- Gontard, N.; Guilbert, S.; Cuq, J.L. Edible Wheat Gluten Films Influence of The Main Process And Environmental Conditions On Thermal, Mechanical And Barrier Properties. Abstr. Pap. Am. Chem. Soc. 1992, 204. [Google Scholar]
- Sun, S.; Song, Y.; Zheng, Q. Morphologies and properties of thermo-molded biodegradable plastics based on glycerol-plasticized wheat gluten. Food Hydrocoll. 2007, 21, 1005–1013. [Google Scholar] [CrossRef]
- Derksen, J.T.P.; Cuperus, F.P.; Kolster, P. Renewable resources in coatings technology: A review. Prog. Org. Coat 1996, 27, 45–53. [Google Scholar] [CrossRef]
- Micard, V.; Belamri, R.; Morel, M.H.; Guilbert, S. Properties of chemically and physically treated wheat gluten films. J. Agric. Food Chem. 2000, 48, 2948–2953. [Google Scholar] [CrossRef] [PubMed]
- Pochat-Bohatier, C.; Sanchez, J.; Gontard, N. Influence of relative humidity on carbon dioxide sorption in wheat gluten films. J. Food Eng. 2006, 77, 983–991. [Google Scholar] [CrossRef]
- Lens, J.P.; de Graaf, L.A.; Stevels, W.M.; Dietz, C.; Verhelst, K.C.S.; Vereijken, J.M.; Kolster, P. Influence of processing and storage conditions on the mechanical and barrier properties of films cast from aqueous wheat gluten dispersions. Ind. Crops Prod. 2003, 17, 119–130. [Google Scholar] [CrossRef]
- Pommet, M.; Redl, A.; Guilbert, S.; Morel, M.H. Intrinsic influence of various plasticizers on functional properties and reactivity of wheat gluten thermoplastic materials. J. Cereal Sci. 2005, 42, 81–91. [Google Scholar] [CrossRef]
- Sanchez, A.C.; Popineau, Y.; Mangavel, C.; Larre, C.; Gueguen, J. Effect of different plasticizers on the mechanical and surface properties of wheat gliadin films. J. Agric. Food Chem. 1998, 46, 4539–4544. [Google Scholar] [CrossRef]
- Gennadios, A.; Brandenburg, A.H.; Weller, C.L.; Testin, R.F. Effect of pH on properties of wheat gluten and soy protein isolate films. J. Agric. Food Chem. 1993, 41, 1835–1839. [Google Scholar] [CrossRef]
- Utsumi, S.; Matsumura, Y.; Mori, T. Structure-Function Relationships of Soy Proteins. In Food Proteins and Their Applications; Damodaran, S., Paraf, A., Eds.; Marcel Dekker: New York, NY, USA, 1997; pp. 257–291. [Google Scholar]
- Wang, G.; Zhang, T.; Ahmad, S.; Cheng, J.; Guo, M. Physicochemical and adhesive properties, microstructure and storage stability of whey protein-based paper glue. Int. J. Adhes. 2013, 41, 198–205. [Google Scholar] [CrossRef]
- Staswick, P.E.; Hermodson, M.A.; Nielsen, N.C. Identification of the cystines which link the acidic and basic components of the glycinin subunits. J. Biol. Chem. 1984, 259, 13431–13435. [Google Scholar] [PubMed]
- Renkema, J.M.S.; Knabben, J.H.M.; van Vliet, T. Gel formation by β-conglycinin and glycinin and their mixtures. Food Hydrocoll. 2001, 15, 407–414. [Google Scholar] [CrossRef]
- Chao, Z.; Yue, M.; Xiaoyan, Z.; Dan, M. Development of soybean protein-isolate edible films incorporated with beeswax, Span 20, and glycerol. J. Food Sci. 2010, 75, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Song, F.; Tang, D.-L.; Wang, X.-L.; Wang, Y.-Z. Biodegradable soy protein isolate-based materials: A review. Biomacromolecules 2011, 12, 3369–3380. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.M.; Hwang, K.T.; Weller, C.L.; Hanna, M.A. Preparation and characterization of soy protein isolate films modified with sorghum wax. J. Am. Oil Chem. Soc. 2002, 79, 615–619. [Google Scholar] [CrossRef]
- Ogale, A.A.; Cunningham, P.; Dawson, P.L.; Acton, J.C. Viscoelastic, thermal, and microstructural characterization of soy protein isolate films. J. Food Sci. 2000, 65, 672–679. [Google Scholar] [CrossRef]
- Di Pierro, P.; Mariniello, L.; Giosafatto, C.V.L.; Masi, P.; Porta, R. Solubility and permeability properties of edible pectin-soy flour films obtained in the absence or presence of transglutaminase. Food Biotechnol. 2005, 19, 37–49. [Google Scholar] [CrossRef]
- Mariniello, L.; di Pierro, P.; Esposito, C.; Sorrentino, A.; Masi, P.; Porta, R. Preparation and mechanical properties of edible pectin-soy flour films obtained in the absence or presence of transglutaminase. J. Biotechnol. 2003, 102, 191–198. [Google Scholar] [CrossRef]
- Wan, V.C.H.; Kim, M.S.; Lee, S.Y. Water vapor permeability and mechanical properties of soy protein isolate edible films composed of different plasticizer combinations. J. Food Sci. 2005, 70, E387–E391. [Google Scholar] [CrossRef]
- Kim, K.M.; Marx, D.B.; Weller, C.L.; Hanna, M.A. Influence of sorghum wax, glycerin, and sorbitol on physical properties of soy protein isolate films. J. Am. Oil Chem. Soc. 2003, 80, 71–76. [Google Scholar] [CrossRef]
- Stuchell, Y.M.; Krochta, J.M. Enzymatic treatments and thermal effects on edible soy protein films. J. Food Sci. 1994, 59, 1332–1337. [Google Scholar] [CrossRef]
- Ghorpade, V.M.; Li, H.; Gennadios, A.; Hanna, M.A. Chemically modified soy protein films. Trans. Asae 1995, 38, 1805–1808. [Google Scholar] [CrossRef]
- Tang, C.H.; Jiang, Y.; Wen, Q.B.; Yang, X.Q. Effect of transglutaminase treatment on the properties of cast films of soy protein isolates. J. Biotechnol. 2005, 120, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Mauri, A.N.; Anon, M.C. Mechanical and physical properties of soy protein films with pH-modified microstructures. Food Sci. Technol. Int. 2008, 14, 119–125. [Google Scholar] [CrossRef]
- Rhim, J.-W.; Lee, J.-H.; Hong, S.-I. Water resistance and mechanical properties of biopolymer (alginate and soy protein) coated paperboards. LWT-Food Sci. Technol. 2006, 39, 806–813. [Google Scholar] [CrossRef]
- Gonzalez, A.; Strumia, M.; Alvarez Igarzabal, C.I. Cross-linked soy protein as material for biodegradable films: Synthesis, characterization and biodegradation. J. Food Eng. 2011, 106, 331–338. [Google Scholar] [CrossRef]
- Malumba Kamba, P.; Vanderghem, C.; Deroanne, C.; Bera, F. Influence of drying temperature on the solubility, the purity of isolates and the electrophoretic patterns of corn proteins. Food Chem. 2008, 101, 564–572. [Google Scholar] [CrossRef]
- Lawton, J.W. Plasticizers for zein: Their effect on tensile properties and water absorption of zein films. Cereal Chem. 2004, 81, 1–5. [Google Scholar] [CrossRef]
- Dickey, L.C.; Parris, N.; Craig, J.C.; Kurantz, M.J. Ethanolic extraction of zein from maize. Ind. Crops Prod. 2001, 13, 67–76. [Google Scholar] [CrossRef]
- Sessa, D.J.; Selling, G.W.; Willett, J.L.; Palmquist, D.E. Viscosity control of zein processing with sodium dodecyl sulfate. Ind. Crops Prod. 2006, 23, 15–22. [Google Scholar] [CrossRef]
- Lawton, J.W. Viscoelasticity of zein-starch doughs. Cereal Chem. 1992, 69, 351–355. [Google Scholar]
- Bai, J.; Alleyne, V.; Hagenmaier, R.D.; Mattheis, J.P.; Baldwin, E.A. Formulation of zein coatings for apples (Malus domestica Borkh). Postharvest Biol. Technol. 2003, 28, 259–268. [Google Scholar] [CrossRef]
- Selling, G.W.; Sessa, D.J.; Palmquist, D.E. Effect of water and tri(ethylene) glycol on the rheological properties of zein. Polymer 2004, 45, 4249–4255. [Google Scholar] [CrossRef]
- Ha, T.T.; Padua, G.W. Effect of extrusion processing on properties of zein-fatty acids sheets. Trans. Asae 2001, 44, 1223–1228. [Google Scholar]
- Lai, H.M.; Padua, G.W. Properties and microstructure of plasticized zein films. Cereal Chem. 1997, 74, 771–775. [Google Scholar] [CrossRef]
- Wang, Y.; Filho, F.L.; Geil, P.; Padua, G.W. Effects of processing on the structure of zein/oleic acid films investigated by X-ray diffraction. Macromol. Biosci. 2005, 5, 1200–1208. [Google Scholar] [CrossRef] [PubMed]
- Ghanbarzadeh, B.; Oromiehie, A.R.; Musavi, M.; Falcone, P.M.; D-Jomeh, Z.E.; Rad, E.R. Study of mechanical properties, oxygen permeability and AFM topography of zein films plasticized by polyols. Packag. Technol. Sci. 2007, 20, 155–163. [Google Scholar] [CrossRef]
- Rakotonirainy, A.M.; Padua, G.W. Effects of lamination and coating with drying oils on tensile and barrier properties of zein films. J. Agric. Food Chem. 2001, 49, 2860–2863. [Google Scholar] [CrossRef] [PubMed]
- Rakotonirainy, A.M.; Wang, Q.; Padua, G.W. Evaluation of zein films as modified atmosphere packaging for fresh broccoli. J. Food Sci. 2001, 66, 1108–1111. [Google Scholar] [CrossRef]
- Wang, Q.; Padua, G.W. Properties of zein films coated with drying oils. J. Agric. Food Chem. 2005, 53, 3444–3448. [Google Scholar] [CrossRef] [PubMed]
- Santosa, F.X.B.; Padua, G.W. Tensile properties and water absorption of zein sheets plasticized with oleic and linoleic acids. J. Agric. Food Chem. 1999, 47, 2070–2074. [Google Scholar] [CrossRef]
- Padgett, T.; Han, I.Y.; Dawson, P.L. Effect of lauric acid addition on the antimicrobial efficacy and water permeability of corn zein films containing nisin. J. Food Process. Preserv. 2000, 24, 423–432. [Google Scholar] [CrossRef]
- Parris, N.; Coffin, D.R. Composition factors affecting the water vapor permeability and tensile properties of hydrophilic zein films. J. Agric. Food Chem. 1997, 45, 1596–1599. [Google Scholar] [CrossRef]
- Ghanbarzadeh, B.; Musavi, M.; Oromiehie, A.R.; Rezayi, K.; Rad, E.R.; Milani, J. Effect of plasticizing sugars on water vapor permeability, surface energy and microstructure properties of zein films. LWT-Food Sci. Technol. 2007, 40, 1191–1197. [Google Scholar] [CrossRef]
- Trezza, T.A.; Vergano, P.J. Grease resistance of corn zein coated paper. J. Food Sci. 1994, 59, 912–915. [Google Scholar] [CrossRef]
- Parris, N.; Dickey, L.C.; Wiles, J.L.; Moreau, R.A.; Cooke, P.H. Enzymatic hydrolysis, grease permeation, and water barrier properties of zein isolate coated paper. J. Agric. Food Chem. 2000, 48, 890–894. [Google Scholar] [CrossRef] [PubMed]
- Soliman, E.A.; Mohy Eldin, M.S.; Furuta, M. Biodegradable zein-based films: Influence of γ-irradiation on structural and functional properties. J. Agric. Food Chem. 2009, 57, 2529–2535. [Google Scholar] [CrossRef] [PubMed]
- Ghanbarzadeh, B.; Oromiehie, A.R.; Musavi, M.; D-Jomeh, Z.E.; Rad, E.R.; Milani, W. Effect of plasticizing sugars on rheological and thermal properties of zein resins and mechanical properties of zein films. Food Res. Int. 2006, 39, 882–890. [Google Scholar] [CrossRef]
- Gillgren, T.; Stading, M. Mechanical and barrier properties of avenin, kafirin, and zein films. Food Biophys. 2008, 3, 287–294. [Google Scholar] [CrossRef]
- Ghanbarzadeh, B.; Oromiehi, A.R. Biodegradable biocomposite films based on whey protein and zein: Barrier, mechanical properties and AFM analysis. Int. J. Biol. Macromol. 2008, 43, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Parris, N.; Vergano, P.J.; Dickey, L.C.; Cooke, P.H.; Craig, J.C. Enzymatic hydrolysis of zein-wax-coated paper. J. Agric. Food Chem. 1998, 46, 4056–4059. [Google Scholar] [CrossRef]
- Dou, Y.; Haung, X.; Zhang, B.; He, M.; Yin, G.; Cui, Y. Preparation and characterization of a dialdehyde starch crosslinked feather keratin film for food packaging application. RSC Adv. 2015, 5, 27168–27174. [Google Scholar] [CrossRef]
- Shiku, Y.; Hamaguchi, P.Y.; Tanaka, M. Effect of pH on the preparation of edible films based on fish myofibrillar proteins. Fish. Sci. 2003, 69, 1026–1032. [Google Scholar] [CrossRef]
- Sobral, P.J.A.; Dos Santos, J.S.; Garcia, F.T. Effect of protein and plasticizer concentrations in film forming solutions on physical properties of edible films based on muscle proteins of a Thai Tilapia. J. Food Eng. 2005, 70, 93–100. [Google Scholar] [CrossRef]
- Pires, C.; Ramos, C.; Teixeira, B.; Batista, I.; Nunes, M.L.; Marques, A. Hake proteins edible films incorporated with essential oils: Physical, mechanical, antioxidant and antibacterial properties. Food Hydrocoll. 2013, 30, 224–231. [Google Scholar] [CrossRef]
- Lee, H.; Scherer, N.F.; Messersmith, P.B. Single-molecule mechanics of mussel adhesion. PNAS 2006, 103, 12999–13003. [Google Scholar] [CrossRef] [PubMed]
- Meredith, H.J.; Wilker, J.J. The interplay of modulus, strength, and ductility in adhesive design using biomimetic polymer chemistry. Adv. Funct. Mater. 2015, 25, 5057–5065. [Google Scholar] [CrossRef]
- Gennadios, A.; Handa, A.; Froning, G.W.; Weller, C.L.; Hanna, M.A. Physical properties of egg white-dialdehyde starch films. J. Agric. Food Chem. 1998, 46, 1297–1302. [Google Scholar] [CrossRef]
- Du, Y.; Chen, F.; Zhang, Y.; Rempel, C.; Thompson, M.R.; Liu, Q. Potato protein isolate-based biopolymers. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Shih, F.F. Edible films from rice protein concentrate and pullulan. Cereal Chem. 1996, 73, 406–409. [Google Scholar]
- Scilingo, A.A.; Molina Ortiz, S.A.; Martinez, E.N.; Anon, M.C. Amaranth protein isolates modified by hydrolytic and thermal treatments. Relationship between structure and solubility. Food Res. Int. 2002, 35, 855–862. [Google Scholar] [CrossRef]
- Denavi, G.; Tapia-Placido, D.R.; Anon, M.C.; Sobral, P.J.A.; Mauri, A.N.; Menegalli, F.C. Effects of drying conditions on some physical properties of soy protein films. J. Food Eng. 2009, 90, 341–349. [Google Scholar] [CrossRef]
- Avanza, M.V.; Puppo, M.C.; Anon, M.C. Rheological characterization of amaranth protein gels. Food Hydrocoll. 2005, 19, 889–898. [Google Scholar] [CrossRef]
- Salgado, P.R.; Lopez-Caballero, M.E.; Gomez-Guillen, M.C.; Mauri, A.N.; Montero, M.P. Sunflower protein films incorporated with clove essential oil have potential application for the preservation of fish patties. Food Hydrocoll. 2013, 33, 74–84. [Google Scholar] [CrossRef]
- Choi, W.S.; Han, J.H. Physical and mechanical properties of pea-protein-based edible films. J. Food Sci. 2001, 66, 319–322. [Google Scholar] [CrossRef]
- Mariniello, L.; Giosafatto, C.V.L.; Moschetti, G.; Aponte, M.; Masi, P.; Sorrentino, A.; Porta, R. Fennel waste-based films suitable for protecting cultivations. Biomacromolecules 2007, 8, 3008–3014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mariniello, L.; Giosafatto, C.V.L.; di Pierro, P.; Sorrentino, A.; Porta, R. Swelling, mechanical and barrier properties of albedo-based films prepared in the presence of phaseolin crosslinked or not by transglutaminase. Biomacromolecules 2010, 11, 2394–2398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giosafatto, C.V.L.; di Pierro, P.; Gunning, P.; Mackie, A.; Porta, R.; Mariniello, L. Characterization of Citrus pectin edible films containing transglutaminase-modified phaseolin. Carbohydr. Polym. 2014, 106, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Giosafatto, C.V.L.; di Pierro, P.; Gunning, P.; Mackie, A.; Porta, R.; Mariniello, L. Trehalose-containing hydrocolloid edible films prepared in the presence of transglutaminase. Biopolymers 2014, 101, 931–937. [Google Scholar] [CrossRef] [PubMed]
- Porta, R.; di Pierro, P.; Rossi-Marquez, G.; Mariniello, L.; Kadivar, M.; Arabestani, A. Microstructure and properties of bitter vetch (Vicia. ervilia) protein films reinforced by microbial transglutaminase. Food Hydrocoll. 2015, 50, 102–107. [Google Scholar] [CrossRef]
- Ionescu, A.; Aprodu, I.; Daraba, A.; Gurau, G.; Baciu, C.; Nichita, A. Chemical and Functional Characterization of Chickpea Protein Derivates. In The Annals of the University Dunarea de Jos of Galati Fascicle VI—Food Technology, New Series Year III (XXXIII), Proceeding of the International Symposium Euro—Aliment 2009, Galati, Romania, 9–10 October 2009; pp. 16–27.
- D’agostina, A.; Antonioni, C.; Resta, D.; Arnoldi, A.; Bez, J.; Kanuf, U.; Wasche, A. Optimization of a pilot-scale process for producing lupin protein isolates with valuable technological properties and minimum thermal damage. J. Agric. Food Chem. 2006, 54, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Chango, A.; Villaume, C.; Bau, H.M.; Nicolas, J.P.; Mejean, L. Fractionation by thermal coagulation of lupin proteins: Physicochemical characteristics. Food Res. Int. 1995, 28, 9l–99. [Google Scholar] [CrossRef]
- Schindler, S.; Wittig, M.; Zelena, K.; Krings, U.; Bez, J.; Eisner, P.; Berger, R.G. Lactic fermentation to improve the aroma of protein extracts of sweet lupin (Lupinus angustifolius). Food Chem. 2011, 128, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Ayranci, E.; Cetin, E. The effect of protein isolate of pistacia terebinthus L. on moisture transfer properties of cellulose-based edible films. Lebensm. Wiss. Technol. 1995, 28, 241–244. [Google Scholar] [CrossRef]
- Zahedi, Y.; Ghanbarzadeh, B.; Sedaghat, N. Physical properties of edible emulsified films based on pistachio globulin protein and fatty acids. J. Food Eng. 2010, 100, 102–108. [Google Scholar] [CrossRef]
- Jangchud, A.; Chinnan, M.S. Properties of peanut protein film: Sorption isotherm and plasticizer effect. LWT-Food Sci. Technol. 1999, 32, 89–94. [Google Scholar] [CrossRef]
- Marquié, C.; Aymard, C.; Cuq, J.-L.; Guilbert, S. Biodegradable packaging made from cottonseed flour: Formation and improvement by chemical treatments with gossypol, formaldehyde and glutaraldehyde. J. Agric. Food Chem. 1995, 43, 2762–2767. [Google Scholar] [CrossRef]
- Grevellec, J.; Marquié, C.; Ferry, L.; Crespy, A.; Vialettes, V. Processability of cottonseed proteins into biodegradable materials. Biomacromolecules 2001, 2, 1104–1109. [Google Scholar] [CrossRef] [PubMed]
- Hamaker, B.R.; Bugusu, B.A. Overview: Sorghum Proteins and Food Quality. Available online: http://www.afripro.org.uk/papers/paper08hamaker.pdf (accessed on 4 December 2015).
- Buffo, R.A.; Weller, C.L.; Gennadios, A. Films from laboratory extracted sorghum kafirin. Cereal Chem. 1997, 74, 473–475. [Google Scholar] [CrossRef]
- Grunbacher, A.; Throm, T.; Seidel, C.; Gutt, B.; Rohrig, J.; Strunk, T.; Vincze, P.; Walheim, S.; Schimmel, T.; Wenzel, W.; et al. Six hydrophobins are involved in hydrophobin rodlet formation in aspergillus nidulans and contribute to hydrophobicity of the spore surface. PLoS ONE 2014, 9, e94546. [Google Scholar] [CrossRef] [PubMed]
- Kwan, A.H.; Winefield, R.D.; Sunde, M.; Matthews, J.M.; Haverkamp, R.G.; Templeton, M.D. Structural basis for rodlet assembly in fungal hydrophobins. PNAS 2006, 103, 3621–3626. [Google Scholar] [CrossRef] [PubMed]
- Magarkar, A.; Mele, N.; Abdel-Rahman, N.; Butcher, S.; Torkkeli, M.; Serimaa, R.; Paananen, A.; Linder, M.; Bunker, A. Hydrophobin film structure for HFBI and HFBII and mechanism for accelerated film formation. PLoS Comput. Biol. 2014, 10, e13745. [Google Scholar] [CrossRef] [PubMed]
- Linder, M.B.; Szilvay, G.R.; Nakari-Setala, T.; Penttila, M.E. Hydrophobins: The protein-amphiphiles of filamentous fungi. FEMS Microbiol. Rev. 2005, 29, 877–896. [Google Scholar] [CrossRef] [PubMed]
- Koivu, K. A Method for Hydrophobin Production in Plants and Methods to Produce Hydrophobin Multimers in Plants and Microbes. Patent EP 2631296 A1, 28 August 2013. [Google Scholar]
- Gabor, E.; Buthe, A.; Eck, J.; Kaufmann, F.; Koch, H.J.; Meure, G. Method of Treating Cellulosic Materials with Hydrophobins. Patent CA 2679719 A1, 12 March 2007. [Google Scholar]
- Zhou, J.J.; Wang, S.Y.; Gunasekaran, S. Preparation and characterization of whey protein film incorporated with TiO2 nanoparticles. J. Food Sci. 2009, 74, N50–N56. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Kim, S.H.; Lim, S.T.; Shin, D.H.; Choi, S.Y.; Hwang, K.T. Grease resistance and mechanical properties of isolated soy protein-coated paper. J. Am. Oil Chem. Soc. 2000, 77, 269–273. [Google Scholar] [CrossRef]
- Ben Arfa, A.; Chrakabandhu, Y.; Preziosi-Belloy, L.; Chalier, P.; Gontard, N. Coating papers with soy protein isolates as inclusion matrix of carvacrol. Food Res. Int. 2006, 40, 22–32. [Google Scholar] [CrossRef]
- Ben Arfa, A.; Preziosi-Belloy, L.; Chalier, P.; Gontard, N. Antimicrobial paper based on a soy protein isolate or modified starch coating including carvacrol and cinnamaldehyde. J. Agric. Food Chem. 2007, 55, 2155–2162. [Google Scholar] [CrossRef] [PubMed]
- Chalier, P.; Ben Arfa, A.; Guillard, V.; Gontard, N. Moisture and temperature triggered release of a volatile active agent from soy protein coated paper: Effect of glass transition phenomena on carvacrol diffusion coefficient. J. Agric. Food Chem. 2009, 57, 658–665. [Google Scholar] [CrossRef] [PubMed]
- Chalier, P.; Ben Arfa, A.; Preziosi-Belloy, L.; Gontard, N. Carvacrol losses from soy protein coated papers as a function of drying conditions. J. Appl. Polym. Sci. 2007, 106, 611–620. [Google Scholar] [CrossRef]
- Guillaume, C.; Pinte, J.; Gontard, N.; Gastaldi, E. Wheat gluten-coated papers for bio-based food packaging: Structure, surface and transfer properties. Food Res. Int. 2010, 43, 1395–1401. [Google Scholar] [CrossRef]
- Chalier, P.; Peyches-Bach, A.; Gastaldi, E.; Gontard, N. Effect of concentration and relative humidity on the transfer of alkan-2-ones through paper coated with wheat gluten. J. Agric. Food Chem. 2007, 55, 867–875. [Google Scholar] [CrossRef] [PubMed]
- Khwaldia, K.; Perez, C.; Banon, S.; Desobry, S.; Hardy, J. Milk proteins for edible films and coatings. Crit. Rev. Food Sci. Nutr. 2004, 44, 239–251. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, J.; Atarés, L.; Vargas, M.; Chiralt, A. Edible films and coatings to prevent the detrimental effect of oxygen on food quality: Possibilities and limitations. J. Food Eng. 2012, 110, 208–213. [Google Scholar] [CrossRef]
- Hui, Y.H.; Guerrero Legarretta, I.; Lim, M.H.; Murrell, K.D.; Nip, W.K. Handbook of Frozen Food; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Lin, D.; Zhao, Y. Innovations in the development and application of edible coatings for fresh and minimally processed fruits and vegetables. Compr. Rev. Food Sci. Food Saf. 2007, 6, 60–75. [Google Scholar] [CrossRef]
- Parris, N.; Sykes, M.; Dickey, L.C.; Wiles, J.L.; Urbanik, T.J.; Cooke, P.H. Recyclable zein-coated kraft paper and linerboard. Progr. Pap. Recycl. 2002, 11, 24–29. [Google Scholar]
- Gällstedt, M. Films and Composites Based on Chitosan, Wheat Gluten or Whey Proteins—Their Packaging Related Mechanical and Barrier Properties. Available online: http://kth.diva-portal.org/smash/get/diva2:9580/FULLTEXT01.pdf (accessed on 4 December 2015).
- Kim, J.-Y.; Jung, C.-K.; Kim, D.-H.; Kim, S.-B. Properties of edible biofilm manufactured from yellowfin tuna (Thunnus albacares) skin gelatin. Korean J. Chem. Eng. 2012, 29, 786–791. [Google Scholar] [CrossRef]
- Chiou, B.-S.; Avena-Bustillos, R.J.; Bechtel, P.J.; Jafri, H.; Narayan, R.; Imam, S.H.; Orts, W.J. Cold water fish gelatin films: Effects of cross-linking on thermal, mechanical, barrier, and biodegradation properties. Eur. Polym. J. 2008, 44, 3748–3753. [Google Scholar] [CrossRef]
- Gómez-Guillén, M.C.; Pérez-Mateos, M.; Gómez-Estaca, J.; López-Caballero, E.; Giménez, B.; Montero, P. Fish gelatin: A renewable material for developing active biodegradable films. Trends Food Sci. Technol. 2009, 20, 3–16. [Google Scholar] [CrossRef]
- Hosseini, S.F.; Rezaei, M.; Zandi, M.; Ghavi, F.F. Preparation and functional properties of fish gelatin-chitosan blend edible films. Food Chem. 2013, 136, 1490–1495. [Google Scholar] [CrossRef] [PubMed]
- Al-Hassan, A.A.; Norziah, M.H. Starch–gelatin edible films: Water vapor permeability and mechanical properties as affected by plasticizers. Food Hydrocoll. 2012, 26, 108–117. [Google Scholar] [CrossRef]
- Rivero, S.; García, M.A.; Pinotti, A. Composite and bi-layer films based on gelatin and chitosan. J. Food Eng. 2009, 90, 531–539. [Google Scholar] [CrossRef]
- Ozcalik, O.; Tihminlioglu, F. Barrier properties of corn zein nanocomposite coated polypropylene films for food packaging applications. J. Food Eng. 2013, 114, 505–513. [Google Scholar] [CrossRef]
- Martelli, S.M.; Moore, G.; Paes, S.S.; Gandolfo, C.; Laurindo, J.B. Influence of plasticizers on the water sorption isotherms and water vapor permeability of chicken feather keratin films. LWT-Food Sci. Technol. 2006, 39, 292–301. [Google Scholar] [CrossRef]
- Guazzotti, V.; Marti, A.; Piergiovanni, L.; Limbo, S. Bio-based coatings as potential barriers to chemical contaminants from recycled paper and board for food packaging. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2014, 31, 402–413. [Google Scholar] [CrossRef] [PubMed]
- Coltelli, M.B.; Coiai, S.; Bronco, S.; Passaglia, E. Nanocomposites Based on Phyllosilicates: From Petrochemicals to Renewable Thermoplastic Matrices. In Advanced Nanomaterials; Geckeler, K.E., Nishide, H., Eds.; Wiley: Weinheim, Germany, 2010; pp. 403–458. [Google Scholar]
- Castiello, S.; Coltelli, M.B.; Conzatti, L.; Bronco, S. Comparative study about preparation of poly(lactide)/organophilic montmorillonites nanocomposites through melt blending or ring opening polymerization methods. J. Appl. Polym. Sci. 2011, 125, E413–E428. [Google Scholar] [CrossRef]
- Sothornvit, R.; Rhim, J.W.; Hong, S.I. Effect of nano-clay type on the physical and antimicrobial properties of whey protein isolate/clay composite films. J. Food Eng. 2009, 91, 468–473. [Google Scholar] [CrossRef]
- Kumar, P.; Sandeep, K.P.; Alawi, S.; Truong, V.D.; Gorga, R.E. Preparation and characterization of bio-nanocomposite films based on soy protein isolate and montmorillonite using melt extrusion. J. Food Eng. 2010, 100, 480–489. [Google Scholar] [CrossRef]
- Salam, A.; Lucia, L.A.; Jameel, H. A preliminary assay of the potential of soy protein isolate and its hydrolysates to provide interfiber bonding enhancements in lignocellulosic furnishes. React. Funct. Polym. 2014, 85, 228–234. [Google Scholar] [CrossRef]
- Flory, A.R.; Requesens, D.V.; Devaiah, S.P.; Teoh, K.T.; Mansfield, S.D.; Hood, E.E. Development of a green binder system for paper products. BMC Biotechnol. 2013, 13. [Google Scholar] [CrossRef] [PubMed]
- Mayer, F. Adhesive Based on Potato Pulp and Preparation and Use of this Adhesive. Patent EP0732386, 18 September 1996. [Google Scholar]
- Pervaiz, M.; Sain, M. Protein extraction from secondary sludge of paper mill wastewater and its utilization as a wood adhesive. BioResources 2011, 6, 961–970. [Google Scholar]
- Cutter, C.N. Opportunities for bio-based packaging technologies to improve the quality and safety of fresh and further processed muscle foods. Meat Sci. 2006, 74, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Johansson, C.; Bras, J.; Mondragon, I.; Nechita, P.; Plackett, D.; Simon, P.; Aucejo, S. Renewable fibers and bio-based materials for packaging applications—A review of recent developments. Bioresources 2012, 7, 2506–2552. [Google Scholar] [CrossRef]
- Jane, L.; Lim, S.T.; Paetau, I. Degradable Plastics Made of from Starch and Proteins. In Biodegradable Polymers and Packaging; Ching, C., Kaplan, D.L., Thomas, E.L., Eds.; Technomic: Lancaster, PA, USA, 1993; pp. 63–73. [Google Scholar]
- Schnabel, W. Polymer Degradation: Principles and Practical Applications; Macmillan Publishing Co. Inc.: New York, NY, USA, 1981. [Google Scholar]
- Fishman, M. Edible and biodegradable polymer films: Challenges and opportunities. Food Technol. 1997, 51, 16. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coltelli, M.-B.; Wild, F.; Bugnicourt, E.; Cinelli, P.; Lindner, M.; Schmid, M.; Weckel, V.; Müller, K.; Rodriguez, P.; Staebler, A.; et al. State of the Art in the Development and Properties of Protein-Based Films and Coatings and Their Applicability to Cellulose Based Products: An Extensive Review. Coatings 2016, 6, 1. https://doi.org/10.3390/coatings6010001
Coltelli M-B, Wild F, Bugnicourt E, Cinelli P, Lindner M, Schmid M, Weckel V, Müller K, Rodriguez P, Staebler A, et al. State of the Art in the Development and Properties of Protein-Based Films and Coatings and Their Applicability to Cellulose Based Products: An Extensive Review. Coatings. 2016; 6(1):1. https://doi.org/10.3390/coatings6010001
Chicago/Turabian StyleColtelli, Maria-Beatrice, Florian Wild, Elodie Bugnicourt, Patrizia Cinelli, Martina Lindner, Markus Schmid, Verena Weckel, Kerstin Müller, Pablo Rodriguez, Andreas Staebler, and et al. 2016. "State of the Art in the Development and Properties of Protein-Based Films and Coatings and Their Applicability to Cellulose Based Products: An Extensive Review" Coatings 6, no. 1: 1. https://doi.org/10.3390/coatings6010001








