Organic Thin Films Deposited by Matrix-Assisted Pulsed Laser Evaporation (MAPLE) for Photovoltaic Cell Applications: A Review
Abstract
:1. Introduction
2. Organic Materials (Small Molecules Compounds, Oligomers and Polymers) for Photovoltaic Applications
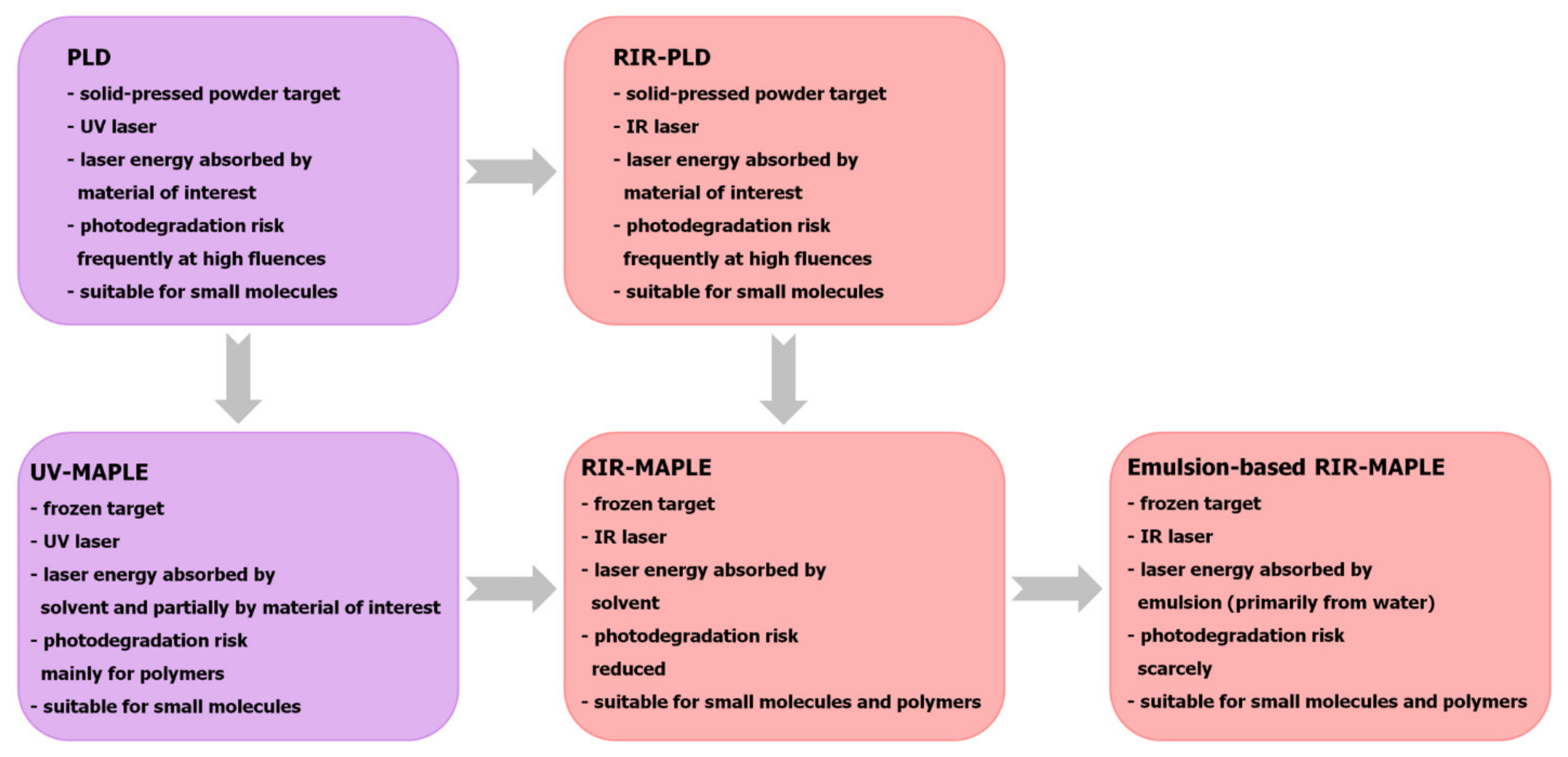
3. Organic Thin Films Deposited Using MAPLE-Based Techniques for OPV Applications
3.1. Layers Based on Small Molecule Compounds
3.1.1. Influence of the Laser Fluence
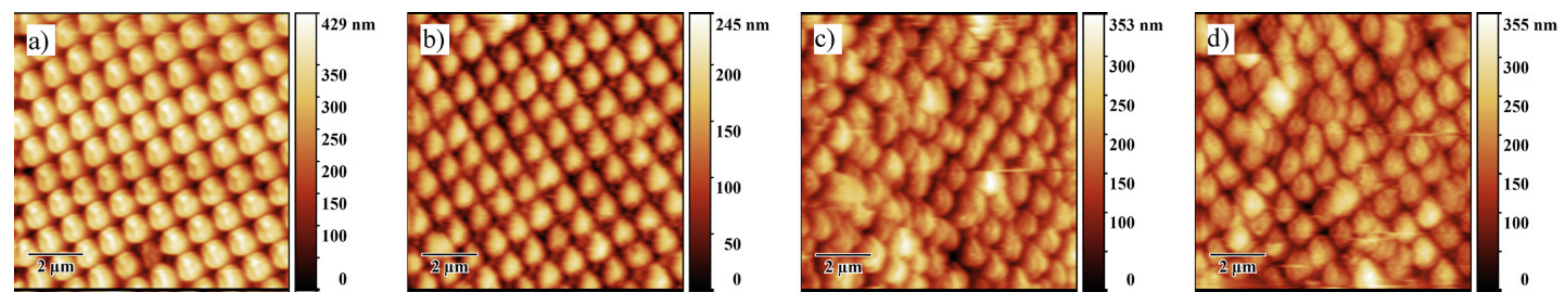
3.1.2. Influence of the Deposition Substrate
3.2. Single Layers Based on Polymers
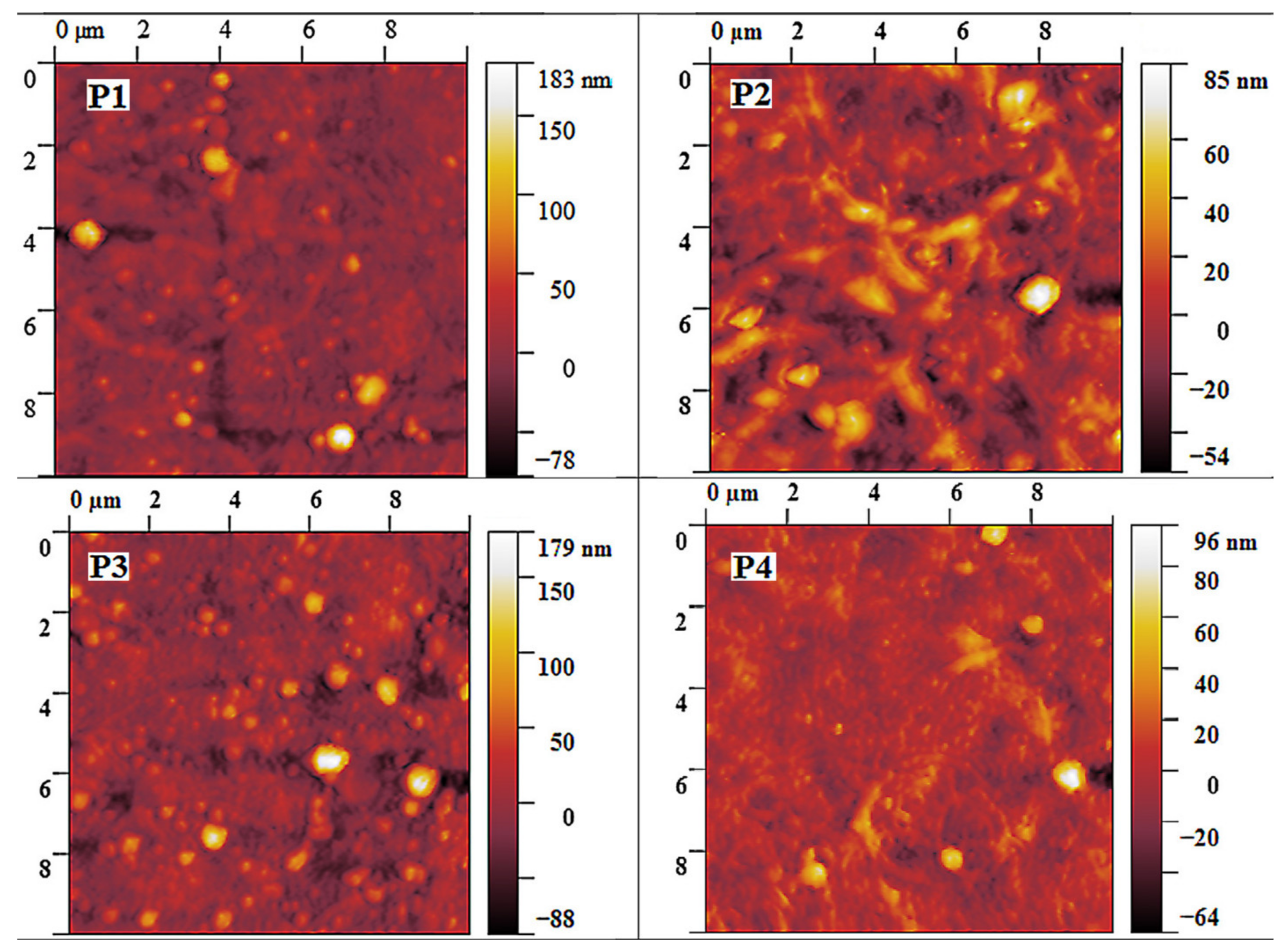
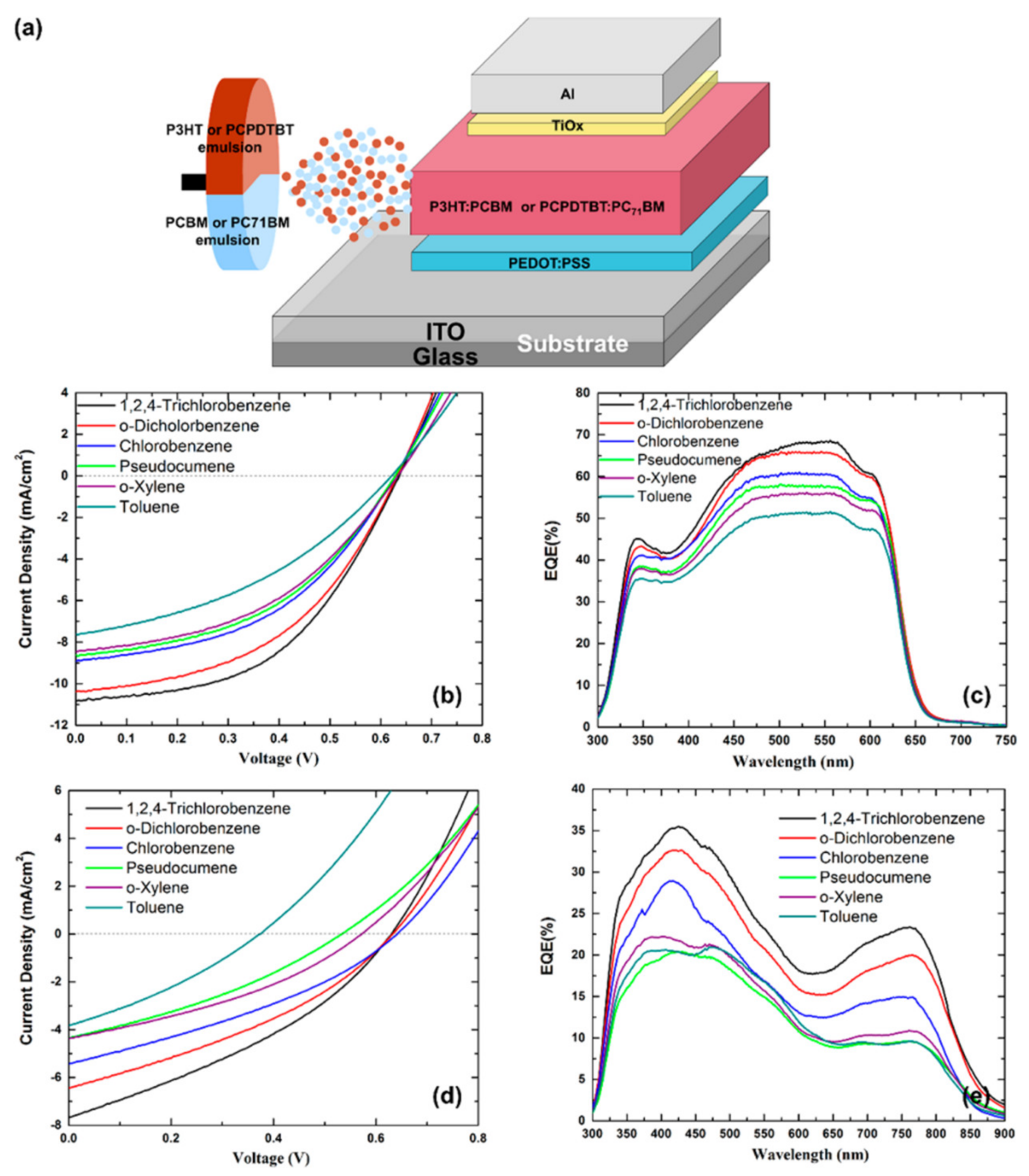
3.2.1. Influence of the Solvent/Emulsion
3.2.2. Influence of the Deposition Substrate
3.2.3. Influence of the Polymer Molecular Weight
3.3. Multi/Mixed Layers Based on Oligomers or Polymers and Fullerenes or Their Derivatives
3.3.1. Influence of the Deposition Configuration
3.3.2. Influence of the Solvent/Emulsion
3.3.3. Influence of the Deposition Substrate
3.4. Organic Thin Films Deposited Using MAPLE-Based Techniques for Photovoltaic Applications—Summary
4. Conclusions and Challenges
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Trending Science: In a first, the EU Produced More Energy from Renewables Than Fossil Fuels in 2020. Available online: https://cordis.europa.eu/article/id/428997-in-a-first-the-eu-produced-more-energy-from-renewables-than-fossil-fuels-in-2020 (accessed on 2 September 2021).
- 10 Interesting Things about Energy. Available online: https://climate.nasa.gov/news/2444/10-interesting-things-about-energy/ (accessed on 2 September 2021).
- Ohl, R.S. Light-Sensitive Electric Device including Silicon. US Patent 2443542, 15 June 1948. (filed 27 May 1941). [Google Scholar]
- Chapin, D.M.; Fuller, C.S.; Pearson, G.L. A new silicon p-n junction photocell for converting solar radiation into electrical power. J. Appl. Phys. 1954, 25, 676–677. [Google Scholar] [CrossRef]
- Luceño-Sánchez, J.A.; Díez-Pascual, A.M.; Capilla, R.P. Materials for photovoltaics: State of art and recent developments. Int. J. Mol. Sci. 2019, 20, 976. [Google Scholar] [CrossRef] [Green Version]
- Shalan, A.E.; Barhoum, A.; Elseman, A.M.; Rashad, M.M.; Lira-Cantú, M. Nanofibers as Promising Materials for New Generations of Solar Cells. In Handbook of Nanofibers, 1st ed.; Barhoum, A., Bechelany, M., Makhlouf, A.S.H., Eds.; Springer Nature Switzerland AG: Cham, Switzerland, 2019; pp. 1–33. [Google Scholar] [CrossRef]
- Soudi, N.; Nanayakkara, S.; Jahed, N.M.S.; Naahidi, S. Rise of nature-inspired solar photovoltaic energy convertors. Sol. Energy 2020, 208, 31–45. [Google Scholar] [CrossRef]
- Katiyar, N.K.; Goel, G.; Hawi, S.; Goel, S. Nature-inspired materials: Emerging trends and prospects. NPG Asia Mater. 2021, 13, 56. [Google Scholar] [CrossRef]
- Senthil, R.; Yuvaraj, S. A comprehensive review on bioinspired solar photovoltaic cells. Int. J. Energy Res. 2018, 43, 1068–1081. [Google Scholar] [CrossRef]
- Tianze, L.; Hengwei, L.; Chuan, J.; Luan, H.; Xia, Z. Application and design of solar photovoltaic system. J. Phys. Conf. Ser. 2011, 276, 012175. [Google Scholar] [CrossRef]
- Minnaert, B.; Veelaert, P. The Suitability of Organic Solar Cells for Different Indoor Conditions. Adv. Sci. Technol. 2010, 74, 170–175. [Google Scholar] [CrossRef]
- Apostolou, G.; Reinders, A.; Verwaal, M. Comparison of the indoor performance of 12 commercial PV products by a simple model. Energy Sci. Eng. 2016, 4, 69–85. [Google Scholar] [CrossRef] [Green Version]
- Krebs, F.C.; Biancardo, M.; Winther-Jensen, B.; Spanggard, H.; Alstrup, J. Strategies for incorporation of polymer photovoltaics into garments and textiles. Sol. Energy Mater. Sol. Cells 2006, 90, 1058–1067. [Google Scholar] [CrossRef]
- Lee, M.R.; Eckert, R.D.; Forberich, K.; Dennler, G.; Brabec, C.J.; Gaudiana, R.A. Solar Power Wires Based on Organic Photovoltaic Materials. Science 2009, 324, 232–235. [Google Scholar] [CrossRef]
- The Key to a Sustainable Building Façade. Available online: https://www.heliatek.com/en/news/detail/the-key-to-a-sustainable-building-facade/ (accessed on 3 September 2021).
- Ryu, H.; Park, S.Y.; Lee, T.H.; Kim, J.Y.; Woo, H.Y. Recent Progress in Indoor Organic Photovoltaics. Nanoscale 2020, 12, 5792–5804. [Google Scholar] [CrossRef]
- Kim, S.H.; Saeed, M.A.; Lee, S.Y.; Shim, J.W. Investigating the Indoor Performance of Planar Heterojunction Based Organic Photovoltaics. IEEE J. Photovolt. 2021, 11, 997–1003. [Google Scholar] [CrossRef]
- Kearns, D.; Calvin, M. Photovoltaic effect and photoconductivity in laminated organic systems. J. Chem. Phys. 1958, 29, 950–951. [Google Scholar] [CrossRef] [Green Version]
- Vanguard Satellite. 1958. Available online: https://www.nasa.gov/content/vanguard-satellite-1958 (accessed on 10 September 2021).
- Tang, C.W. Two-layer organic photovoltaic cell. Appl. Phys. Lett. 1986, 48, 183–185. [Google Scholar] [CrossRef]
- Yu, G.; Gao, J.; Hummelen, J.C.; Wudi, F.; Heeger, A.J. Polymer photovoltaic cells: Enhanced efficiencies via a network of internal donor-acceptor heterojunctions. Science 1995, 270, 1789–1791. [Google Scholar] [CrossRef] [Green Version]
- Green, M.; Dunlop, E.; Hohl-Ebinger, J.; Yoshita, M.; Kopidakis, N.; Hao, X. Solar cell efficiency tables (version 57). Prog. Photovolt. Res. Appl. 2021, 29, 3–15. [Google Scholar] [CrossRef]
- Jin, K.; Xiao, Z.; Ding, L.M. 18.69% PCE from organic solar cells. J. Semicond. 2021, 42, 060502. [Google Scholar] [CrossRef]
- Liu, F.; Zhou, L.; Liu, W.; Zhou, Z.; Yue, Q.; Zheng, W.; Sun, R.; Liu, W.; Xu, S.; Fan, H.; et al. Organic Solar Cells with 18% Efficiency Enabled by an Alloy Acceptor: A Two-in-One Strategy. Adv. Mater. 2021, 33, 2100830. [Google Scholar] [CrossRef]
- Lin, Y.; Magomedov, A.; Firdaus, Y.; Kaltsas, D.; El-Labban, A.; Faber, H.; Naphade, D.R.; Yengel, E.; Zheng, X.; Yarali, E.; et al. 18.4% Organic Solar Cells Using a High Ionization Energy Self-Assembled Monolayer as Hole-Extraction Interlayer. ChemSusChem 2021, 14, 3569–3578. [Google Scholar] [CrossRef]
- Zhang, M.; Zhu, L.; Zhou, G.; Hao, T.; Qiu, C.; Zhao, Z.; Hu, Q.; Larson, B.W.; Zhu, H.; Ma, Z.; et al. Single-layered organic photovoltaics with double cascading charge transport pathways: 18% efficiencies. Nat. Commun. 2021, 12, 309. [Google Scholar] [CrossRef]
- Liang, Z.; Zhang, Q.; Jiang, L.; Cao, G. ZnO cathode buffer layers for inverted polymer solar cells. Energy Environ. Sci. 2015, 8, 3442–3476. [Google Scholar] [CrossRef] [Green Version]
- Lattante, S. Electron and Hole Transport Layers: Their Use in Inverted Bulk Heterojunction Polymer Solar Cells. Electronics 2014, 3, 132–164. [Google Scholar] [CrossRef]
- Hashemi, S.A.; Ramakrishna, S.; Aberle, A. Recent Progress in Flexible-Wearable Solar Cells for Self-Powered Electronic Devices. Energy Environ. Sci. 2020, 13, 685–743. [Google Scholar] [CrossRef]
- Dhar, A.; Alforda, T.L. Optimization of TiO2/Cu/TiO2 Multilayer as Transparent Composite Electrode (TCE) Deposited on Flexible Substrate at Room Temperature. ECS Solid State Lett. 2014, 3, N33–N36. [Google Scholar] [CrossRef]
- Xu, H.; Yuan, F.; Zhou, D.; Liao, X.; Chen, L.; Chen, Y. Hole Transport Layers for Organic Solar Cells: Recent Progress and Perspectives. J. Mater. Chem. A 2020, 8, 11478–11492. [Google Scholar] [CrossRef]
- Kim, T.; Lim, J.; Song, S. Recent Progress and Challenges of Electron Transport Layers in Organic–Inorganic Perovskite Solar Cells. Energies 2020, 13, 5572. [Google Scholar] [CrossRef]
- Kong, T.; Wang, R.; Zheng, D.; Yu, J. Modification of the SnO2 Electron Transporting Layer by Using Perylene Diimide Derivative for Efficient Organic Solar Cells. Front. Chem. 2021, 9, 703561. [Google Scholar] [CrossRef]
- Xu, T.; Yu, L. How to design low bandgap polymers for highly efficient organic solar cells. Mater. Today 2014, 17, 11–15. [Google Scholar] [CrossRef]
- Krebs, F.C. Fabrication and processing of polymer solar cells: A review of printing and coating techniques. Sol. Energy Mater. Sol. Cells 2009, 93, 394–412. [Google Scholar] [CrossRef]
- Schwartz, B.J. Conjugated Polymers as Molecular Materials: How Chain Conformation and Film Morphology Influence Energy Transfer and Interchain Interactions. Annu. Rev. Phys. Chem. 2003, 54, 141–172. [Google Scholar] [CrossRef] [Green Version]
- Dong, B.X.; Strzalka, J.; Jiang, Z.; Li, H.; Stein, G.E.; Green, P.F. Crystallization Mechanism and Charge Carrier Transport in MAPLE-Deposited Conjugated Polymer Thin Films. ACS Appl. Mater. Interfaces 2017, 9, 44799–44810. [Google Scholar] [CrossRef]
- Caricato, A.P.; Cesaria, M.; Gigli, G.; Loiudice, A.; Luches, A.; Martino, M.; Resta, V.; Rizzo, A.; Taurino, A. Poly-(3-hexylthiophene)/[6,6]-phenyl-C61-butyric-acid-methyl-ester bilayer deposition by matrix-assisted pulsed laser evaporation for organic photovoltaic applications. Appl. Phys. Lett. 2012, 100, 073306. [Google Scholar] [CrossRef]
- Ge, W.; Atewologun, A.; Stiff-Roberts, A.D. Hybrid nanocomposite thin films deposited by emulsion-based resonant infrared matrix-assisted pulsed laser evaporation for photovoltaic applications. Org. Electron. 2015, 22, 98–107. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Rasoga, O.; Breazu, C.; Stavarache, I.; Stanculescu, F.; Socol, G.; Gherendi, F.; Grumezescu, V.; Stefan, N.; et al. Flexible heterostructures based on metal phthalocyanines thin films obtained by MAPLE. Appl. Surf. Sci. 2016, 374, 403–410. [Google Scholar] [CrossRef]
- McGill, R.A.; Chrisey, D.B. Method of Producing a Film Coating by Matrix Assisted Pulsed Laser Deposition. U.S. Patent 6025036, 15 February 2000. (filed 28 May 1997). [Google Scholar]
- Krebs, H.-U.; Weisheit, M.; Faupel, J.; Süske, E.; Scharf, T.; Fuhse, C.; Störmer, M.; Sturm, K.; Seibt, M.; Kijewski, H.; et al. Pulsed Laser Deposition (PLD)—A Versatile Thin Film Technique. In Advances in Solid State Physics, 1st ed.; Kramer, B., Ed.; Springer: Berlin/Heidelberg, Germany, 2003; pp. 505–518. [Google Scholar] [CrossRef]
- Novotny, M.; Sebera, J.; Bensalah-Ledoux, A.; Guy, S.; Bulir, J.; Fitl, P.; Vlcek, J.; Zakutna, D.; Maresova, E.; Hubik, P.; et al. The growth of zinc phthalocyanine thin films by pulsed laser deposition. J. Mater. Res. 2015, 31, 163–172. [Google Scholar] [CrossRef]
- Yamaguchi, J.; Itaka, K.; Hayakawa, T.; Arai, K.; Yamashiro, M.; Yaginuma, S.; Koinuma, H. Combinatorial Pulsed Laser Deposition of Pentacene Films for Field Effect Devices. Macromol. Rapid Commun. 2004, 25, 334–338. [Google Scholar] [CrossRef]
- Blanchet, G.B.; Fincher, C.R.; Jackson, C.L.; Shah, S.I.; Gardner, K.H. Laser Ablation and the Production of Polymer Films. Science 1993, 262, 719–721. [Google Scholar] [CrossRef]
- Hansen, S.G.; Robitaille, T.E. Formation of polymer films by pulsed laser evaporation. Appl. Phys. Lett. 1988, 52, 81–83. [Google Scholar] [CrossRef]
- Blanchet, G.B. Deposition of Poly(methyl methacrylate) Films by UV Laser Ablation. Macromolecules 1995, 28, 4603–4607. [Google Scholar] [CrossRef]
- Park, H.K.; Schriver, K.E.; Haglund, R.F. Resonant infrared laser deposition of polymer-nanocomposite materials for optoelectronic applications. Appl. Phys. A 2011, 105, 583–592. [Google Scholar] [CrossRef]
- Toftmann, B.; Papantonakis, M.R.; Auyeung, R.C.Y.; Kim, W.; O’Malley, S.M.; Bubb, D.M.; Horwitz, J.S.; Schou, J.; Johansen, R.F.; Haglund, R.F. UV and RIR matrix assisted pulsed laser deposition of organic MEH-PPV films. Thin Solid Films 2004, 453–454, 177–181. [Google Scholar] [CrossRef]
- Darwish, A.; Sarkisov, S. Method and Apparatus for Open-Air Pulsed Laser Deposition. US Patent 10316403 B2, 11 June 2019. [Google Scholar]
- Nikov, R.G.; Dikovska, A.O.; Nedyalkov, N.N.; Avdeev, G.V.; Atanasov, P.A. Au nanostructure fabrication by pulsed laser deposition in open air: Influence of the deposition geometry. Beilstein J. Nanotechnol. 2017, 8, 2438–2445. [Google Scholar] [CrossRef] [Green Version]
- Bonjakhi, M.; Mahdieh, M.H. Fabrication of silver nanoparticle films by pulsed laser deposition in flowless open air and studying the effects of laser fluence and number of pulses. Colloids Surf. A Physicochem. Eng. Asp. 2021, 626, 126990. [Google Scholar] [CrossRef]
- Nikov, R.G.; Dikovska, A.O.; Nedyalkov, N.N.; Atanasov, P.A.; Atanasova, G.; Hirsch, D.; Rauschenbach, B. ZnO nanostructures produced by pulsed laser deposition in open air. Appl. Phys. A 2017, 123, 657. [Google Scholar] [CrossRef]
- Atanasova, G.; Dikovska, A.O.; Dilova, T.; Georgieva, B.; Avdeev, G.V.; Stefanov, P.; Nedyalkov, N.N. Metal-oxide nanostructures produced by PLD in open air for gas sensor applications. Appl. Surf. Sci. 2018, 470, 861–869. [Google Scholar] [CrossRef]
- Nikov, R.G.; Dikovska, A.O.; Atanasova, G.B.; Avdeev, G.V.; Nedyalkov, N.N. Magnetic-field-assisted formation of oriented nanowires produced by pld in open air. Appl. Surf. Sci. 2020, 458, 273–280. [Google Scholar] [CrossRef]
- Darwish, A.M.; Sarkisov, S.S.; Wilson, S.; Wilson, J.; Collins, E.; Patel, D.N.; Cho, K.; Giri, A.; Koplitz, B.; Hui, D. Polymer nanocomposite sunlight spectrum down-converters made by open-air PLD. Nanotechnol. Rev. 2020, 9, 1044–1058. [Google Scholar] [CrossRef]
- Stiff-Roberts, A.D.; Ge, W. Organic/hybrid thin films deposited by matrix-assisted pulsed laser evaporation (MAPLE). Appl. Phys. Rev. 2017, 4, 041303. [Google Scholar] [CrossRef]
- Wu, P.K.; Ringeisen, B.R.; Krizman, D.B.; Frondoza, C.G.; Brooks, M.; Bubb, D.M.; Auyeung, R.C.Y.; Pique, A.; Spargo, B.; McGill, R.A.; et al. Laser transfer of biomaterials: Matrix-assisted pulsed laser evaporation (MAPLE) and MAPLE Direct Write. Rev. Sci. Instrum. 2003, 74, 2546–2557. [Google Scholar] [CrossRef]
- Caricato, A.P. MAPLE and MALDI: Theory and Experiments. In Lasers in Materials Science, 1st ed.; Castillejo, M., Ossi, P., Zhigilei, L., Eds.; Springer International Publishing: Cham, Switzerland, 2014; pp. 295–323. [Google Scholar] [CrossRef]
- Shepard, K.B.; Priestley, R.D. MAPLE Deposition of Macromolecules. Macromol. Chem. Phys. 2013, 214, 862–872. [Google Scholar] [CrossRef]
- Pate, R.; Lantz, K.R.; Stiff-Roberts, A.D. Tabletop resonant infrared matrix-assisted pulsed laser evaporation of light-emitting organic thin films. IEEE J. Sel. Top. Quantum Electron. 2008, 14, 1022–1030. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, J. Matrix-Assisted Pulsed Laser Evaporation (MAPLE) technique for deposition of hybrid nanostructures. Front. Nanosci. Nanotech. 2017, 3, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Ogugua, S.N.; Ntwaeaborwa, O.M.; Swart, H.C. Latest Development on Pulsed Laser Deposited Thin Films for Advanced Luminescence Applications. Coatings 2020, 10, 1078. [Google Scholar] [CrossRef]
- Caricato, A.P.; Ge, W.; Stiff-Roberts, A.D. UV- and RIR-MAPLE: Fundamentals and Applications, 1st ed.; Springer Series in Materials Science; Springer Nature Switzerland: Cham, Switzerland, 2018; pp. 275–308. [Google Scholar] [CrossRef]
- Axente, E.; Sima, L.E.; Sima, F. Biomimetic coatings obtained by combinatorial laser technologies. Coatings 2020, 10, 463. [Google Scholar] [CrossRef]
- Caricato, A.P.; Luches, A. Applications of the matrix-assisted pulsed laser evaporation method for the deposition of organic, biological and nanoparticle thin films: A review. Appl. Phys. A 2011, 105, 565–582. [Google Scholar] [CrossRef]
- Bloisi, F.; Barra, M.; Cassinese, A.; Vicari, L.R.M. Matrix-Assisted Pulsed Laser Thin Film Deposition by Using Nd:YAG Laser. J. Nanomater. 2012, 2012, 395436. [Google Scholar] [CrossRef] [Green Version]
- Plidschun, M.; Chemnitz, M.; Schmidt, M.A. Low-loss deuterated organic solvents for visible and near-infrared photonics. Opt. Mater. Express 2017, 7, 1122. [Google Scholar] [CrossRef]
- Ge, W.; McCormick, R.D.; Nyikayaramba, G.; Stiff-Roberts, A.D. Bulk Heterojunction PCPDTBT:PC71BM Organic Solar Cells Deposited by Emulsion-Based, Resonant Infrared Matrix-Assisted Pulsed Laser Evaporation. Appl. Phys. Lett. 2014, 104, 223901. [Google Scholar] [CrossRef]
- Ge, W.; Li, N.K.; McCormick, R.D.; Lichtenberg, E.; Yingling, Y.G.; Stiff-Roberts, A.D. Emulsion-Based RIR-MAPLE Deposition of Conjugated Polymers: Primary Solvent Effect and Its Implications on Organic Solar Cell Performance. ACS Appl. Mater. Interfaces 2016, 8, 19494–19506. [Google Scholar] [CrossRef] [Green Version]
- Piqué, A. The Matrix-Assisted Pulsed Laser Evaporation (MAPLE) process: Origins and future directions. Appl. Phys. A 2011, 105, 517–528. [Google Scholar] [CrossRef]
- Bubb, D.M.; Wu, P.K.; Horwitz, J.S.; Callahan, J.H.; Galicia, M.; Vertes, A.; McGill, R.A.; Houser, E.J.; Ringeisen, B.R.; Chrisey, D.B. The effect of the matrix on film properties in matrix-assisted pulsed laser evaporation. J. Appl. Phys. 2002, 91, 2055–2058. [Google Scholar] [CrossRef]
- Visan, A.; Cristescu, R.; Stefan, N.; Miroiu, M.; Nita, C.; Socol, M.; Florica, C.; Rasoga, O.; Zgura, I.; Sima, L.E.; et al. Antimicrobial polycaprolactone/polyethylene glycol embedded lysozyme coatings of Ti implants for osteoblast functional properties in tissue engineering. Appl. Surf. Sci. 2017, 417, 234–243. [Google Scholar] [CrossRef]
- Grumezescu, V.; Negut, I.; Cristescu, R.; Grumezescu, A.M.; Holban, A.M.; Iordache, F.; Chifiriuc, M.C.; Narayan, R.J.; Chrisey, D.B. Isoflavonoid-Antibiotic Thin Films Fabricated by MAPLE with Improved Resistance to Microbial Colonization. Molecules 2021, 26, 3634. [Google Scholar] [CrossRef] [PubMed]
- Spirescu, V.A.; Suhan, R.; Niculescu, A.-G.; Grumezescu, V.; Negut, I.; Holban, A.M.; Oprea, O.-C.; Bîrca, A.C.; Vasile, B.S.; Grumezescu, A.M.; et al. Biofilm-Resistant Nanocoatings Based on ZnO Nanoparticles and Linalool. Nanomaterials 2021, 11, 2564. [Google Scholar] [CrossRef] [PubMed]
- Holban, A.M.; Grumezescu, V.; Grumezescu, A.M.; Vasile, B.Ş.; Truşcă, R.; Cristescu, R.; Socol, G.; Iordache, F. Antimicrobial nanospheres thin coatings prepared by advanced pulsed laser technique. Beilstein J. Nanotechnol. 2014, 5, 872–880. [Google Scholar] [CrossRef] [Green Version]
- Huang, G.; Chen, Y.; Zhang, J. Nanocomposited coatings produced by laser-assisted process to prevent silicone hydrogels from protein fouling and bacterial contamination. Appl. Surf. Sci. 2016, 360, 383–388. [Google Scholar] [CrossRef]
- Oprea, A.; Pandel, L.; Dumitrescu, A.; Andronescu, E.; Grumezescu, V.; Chifiriuc, M.; Mogoanta, L.; Balseanu, T.A.; Mogosanu, G.D.; Socol, G.; et al. Bioactive ZnO Coatings Deposited by MAPLE—An Appropriate Strategy to Produce Efficient Anti-Biofilm Surfaces. Molecules 2016, 21, 220. [Google Scholar] [CrossRef] [Green Version]
- Bloisi, F.; Vicari, L.; Papa, R.; Califano, V.; Pedrazzani, R.; Bontempi, E.; Depero, L.E. Biomaterial thin film deposition and characterization by means of MAPLE technique. Mater. Sci. Eng. C 2007, 27, 1185–1190. [Google Scholar] [CrossRef]
- Yang, S.; Tse, W.H.; Zhang, J. Deposition of antibody modified upconversion nanoparticles on glass by a laser-assisted method to improve the performance of cell culture. Nanoscale Res. Lett. 2019, 14, 101. [Google Scholar] [CrossRef]
- Icriverzi, M.; Rusen, L.; Brajnicov, S.; Bonciu, A.; Dinescu, M.; Cimpean, A.; Evans, R.W.; Dinca, V.; Roseanu, A. Macrophage in vitro response on hybrid coatings obtained by matrix assisted pulsed laser evaporation. Coatings 2019, 9, 236. [Google Scholar] [CrossRef] [Green Version]
- Marturano, V.; Abate, F.; Ambrogi, V.; Califano, V.; Cerruti, P.; Pepe, G.P.; Vicari, L.R.M.; Ausanio, G. Smart Coatings Prepared via MAPLE Deposition of Polymer Nanocapsules for Light-Induced Release. Molecules 2021, 26, 2736. [Google Scholar] [CrossRef]
- Visan, A.I.; Ristoscu, C.; Popescu-Pelin, G.; Sopronyi, M.; Matei, C.E.; Socol, G.; Chifiriuc, M.C.; Bleotu, C.; Grossin, D.; Brouillet, F.; et al. Composite Drug Delivery System Based on Amorphous Calcium Phosphate–Chitosan: An Efficient Antimicrobial Platform for Extended Release of Tetracycline. Pharmaceutics 2021, 13, 1659. [Google Scholar] [CrossRef] [PubMed]
- Puiu, R.A.; Balaure, P.C.; Constantinescu, E.; Grumezescu, A.M.; Andronescu, E.; Oprea, O.-C.; Vasile, B.S.; Grumezescu, V.; Negut, I.; Nica, I.C.; et al. Anti-Cancer Nanopowders and MAPLE-Fabricated Thin Films Based on SPIONs Surface Modified with Paclitaxel Loaded β-Cyclodextrin. Pharmaceutics 2021, 13, 1356. [Google Scholar] [CrossRef]
- Stanculescu, A.; Breazu, C.; Socol, M.; Rasoga, O.; Preda, N.; Petre, G.; Solonaru, A.M.; Grigoras, M.; Stanculescu, F.; Socol, G.; et al. Effect of ITO electrode patterning on the properties of organic heterostructures based on non-fullerene acceptor prepared by MAPLE. Appl. Surf. Sci. 2020, 509, 145351. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Petre, G.; Costas, A.; Rasoga, O.; Popescu-Pelin, G.; Mihailescu, A.; Stanculescu, A.; Socol, G. MAPLE Deposition of Binary and Ternary Organic Bulk Heterojunctions Based on Zinc Phthalocyanine. Coatings 2020, 10, 956. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Breazu, C.; Costas, A.; Petre, G.; Stanculescu, A.; Popescu-Pelin, G.; Mihailescu, A.; Socol, G. Organic Thin Films Based on DPP-DTT:C60 Blends Deposited by MAPLE. Nanomaterials 2020, 10, 2366. [Google Scholar] [CrossRef] [PubMed]
- Stanculescu, F.; Rasoga, O.; Catargiu, A.M.; Vacareanu, L.; Socol, M.; Breazu, C.; Preda, N.; Socol, G.; Stanculescu, A. MAPLE prepared heterostructures with arylene based polymer active layer for photovoltaic applications. Appl. Surf. Sci. 2015, 336, 240–248. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Costas, A.; Breazu, C.; Stanculescu, A.; Rasoga, O.; Popescu-Pelin, G.; Mihailescu, A.; Socol, G. Hybrid organic-inorganic thin films based on zinc phthalocyanine and zinc oxide deposited by MAPLE. Appl. Surf. Sci. 2020, 503, 144317. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Costas, A.; Borca, B.; Popescu-Pelin, G.; Mihailescu, A.; Socol, G.; Stanculescu, A. Thin films based on cobalt phthalocyanine:C60 fullerene:ZnO hybrid nanocomposite obtained by laser evaporation. Nanomaterials 2020, 10, 468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferguson, S.; Williams, C.V.; Mohapi, B.; Stiff-Roberts, A.D. Deposition of β-Polyfluorene by Resonant Infrared Matrix-Assisted Pulsed Laser Evaporation. J. Electron. Mater. 2019, 48, 3388–3398. [Google Scholar] [CrossRef]
- Mariano, F.; Caricato, A.P.; Accorsi, G.; Leo, C.; Cesaria, M.; Carallo, S.; Genco, A.; Simeone, D.; Tunno, T.; Martino, M.; et al. White multi-layered polymer light emitting diode through matrix assisted pulsed laser evaporation. J. Mater. Chem. C 2016, 4, 7667–7674. [Google Scholar] [CrossRef]
- Guo, Y.; Ren, S. Bilayer PMMA antireflective coatings via microphase separation and MAPLE. J. Polym. Eng. 2021, 41, 164–173. [Google Scholar] [CrossRef]
- Ionita, I.; Bercea, A.; Brajnicov, S.; Matei, A.; Ion, V.; Marascu, V.; Mitu, B.; Constantinescu, C. Second harmonic generation (SHG) in pentacene thin films grown by matrix assisted pulsed laser evaporation (MAPLE). Appl. Surf. Sci. 2019, 480, 212–218. [Google Scholar] [CrossRef]
- Constantinescu, C.; Matei, A.; Ion, V.; Mitu, B.; Ionita, I.; Dinescu, M.; Luculescu, C.R.; Vasiliu, C.; Emandi, A. Ferrocene carboxaldehyde thin films grown by matrix-assisted pulsed laser evaporation for non linear optical applications. Appl. Surf. Sci. 2014, 302, 83–86. [Google Scholar] [CrossRef]
- Stanculescu, A.; Rasoga, O.; Mihut, L.; Socol, M.; Stanculescu, F.; Ionita, I.; Albu, A.-M.; Socol, G. Preparation and characterization of polar aniline functionalized copolymers thin films for optical non-linear applications. Ferroelectrics 2009, 389, 159–173. [Google Scholar] [CrossRef]
- Ajnsztajn, A.; Ferguson, S.; Thostenson, J.O.; Ngaboyamahina, E.; Parker, C.B.; Glass, J.T.; Stiff-Roberts, A.D. Transparent MXene-Polymer Supercapacitive Film Deposited Using RIR-MAPLE. Crystals 2020, 10, 152. [Google Scholar] [CrossRef] [Green Version]
- Rose, O.L.; Bonciu, A.; Marascu, V.; Matei, A.; Liu, Q.; Rusen, L.; Dinca, V.; Dinu, C.Z. Thin Films of Metal-Organic Framework Interfaces Obtained by Laser Evaporation. Nanomaterials 2021, 11, 1367. [Google Scholar] [CrossRef]
- Sawczak, M.; Jendrzejewski, R.; Maskowicz, D.; Garcia, Y.; Dîrtu, M.; Kumar, V.; Śliwiński, G. Host–guest exchange contribution to transition temperature downshift in nanocrystalline Fe(pyrazine)[Pt(CN)4] thin films prepared by matrix-assisted pulsed laser evaporation. J. Appl. Phys. 2021, 129, 155308. [Google Scholar] [CrossRef]
- Sawczak, M.; Jendrzejewski, R.; Maskowicz, D.; Garcia, Y.; Ghosh, A.C.; Gazda, M.; Czechowski, J.; Śliwiński, G. Nanocrystalline Polymer impregnated [Fe(pz)Pt(CN)4] Thin Films prepared by Matrix-Assisted Pulsed Laser Evaporation. Eur. J. Inorg. Chem. 2019, 3249–3255. [Google Scholar] [CrossRef]
- Wang, Y.; Gu, K.; Monnier, X.; Jeong, H.; Chowdhury, M.; Cangialosi, D.; Loo, Y.-L.; Priestley, R.D. Tunable Properties of MAPLE-Deposited Thin Films in the Presence of Suppressed Segmental Dynamics. ACS Macro Lett. 2019, 8, 1115–1121. [Google Scholar] [CrossRef]
- Benetti, M.; Cannatà, D.; Verona, E.; Palla Papavlu, A.; Dinca, V.C.; Lippert, T.; Dinescu, M.; Di Pietrantonio, F. Highly selective surface acoustic wave e-nose implemented by laser direct writing. Sens. Actuator B–Chem. 2019, 283, 154–162. [Google Scholar] [CrossRef]
- Jelínek, M.; Remsa, J.; Kocourek, T.; Kubešová, B.; Schůrek, J.; Myslík, V. MAPLE activities and applications in gas sensors. Appl. Phys. A 2011, 105, 643–649. [Google Scholar] [CrossRef]
- Bonciu, A.; Vasilescu, A.; Dinca, V.; Peteu, S.F. Interfaces obtained by MAPLE for chemical and biosensors applications. Sens. Actuators Rep. 2021, 3, 100040. [Google Scholar] [CrossRef]
- 548 Results from Web of Science Core Collection for: Organic Wearable Electronic. Available online: https://www.webofscience.com/wos/woscc/summary/c7e8260f-21cb-4bbd-9719-dbe16f6c7229-11b384c7/relevance/1 (accessed on 15 September 2021).
- Schunemann, C.; Wynands, D.; Wilde, L.; Hein, M.P.; Pfutzner, S.; Elschner, C.; Eichhorn, K.-J.; Leo, K.; Riede, M. Phase separation analysis of bulk heterojunctions in small-molecule organic solar cells using zinc-phthalocyanine and C60. Phys. Rev. B 2012, 85, 245314. [Google Scholar] [CrossRef]
- Ghani, F.; Kristen, J.; Riegler, H. Solubility Properties of Unsubstituted Metal Phthalocyanines in Different Types of Solvents. J. Chem. Eng. Data 2012, 57, 439–449. [Google Scholar] [CrossRef]
- Ibrahim, N.M.; Hassan, E.K. Structural and Morphological of Pulsed Laser Deposited Magnesium Phthalocyanine (MgPc) Thin Film. Nano Hybrids Compos. 2020, 29, 15–21. [Google Scholar] [CrossRef]
- Park, S.H.; Jeong, J.G.; Kim, H.-J.; Park, S.-H.; Cho, M.-H.; Cho, S.W.; Yi, Y.; Heo, M.Y.; Sohn, H. The electronic structure of C60/ZnPc interface for organic photovoltaic device with blended layer architecture. Appl. Phys. Lett. 2010, 96, 013302. [Google Scholar] [CrossRef] [Green Version]
- Gao, W.; Kahn, A. Electronic structure and current injection in zinc phthalocyanine doped with tetrafluorotetracyanoquinodimethane: Interface versus bulk effects. Org. Electron. 2002, 3, 53–63. [Google Scholar] [CrossRef]
- Maruhashi, H.; Oku, T.; Suzuki, A.; Akiyama, T.; Yamasaki, Y. Fabrication and characterization of P3HT:PCBM-based thin film organic solar cells with zinc phthalocyanine. AIP Conf. Proc. 2015, 1649, 89–95. [Google Scholar] [CrossRef] [Green Version]
- Matsuo, Y.; Ogumi, K.; Jeon, I.; Wang, H.; Nakagawa, T. Recent progress in porphyrin- and phthalocyanine-containing perovskite solar cells. RSC Adv. 2020, 10, 32678–32689. [Google Scholar] [CrossRef]
- Stanculescu, A.; Stanculescu, F.; Tugulea, L.; Socol, M. Optical properties of 3,4,9,10-perylenetetracarboxylic dianhydride and 8-hydroxyquinoline aluminium salt films prepared by vacuum deposition. Mater. Sci. Forum 2006, 514–516, 956–960. [Google Scholar] [CrossRef]
- Stanculescu, F.; Stanculescu, A.; Socol, M. Effect of the metallic contact on the electrical properties of organic semiconductor film. J. Optoelectron. Adv. Mater. 2007, 9, 1352–1357. [Google Scholar]
- Stanculescu, A.; Socol, M.; Socol, G.; Mihailescu, I.N.; Girtan, M.; Stanculescu, F. MAPLE prepared organic heterostructures for photovoltaic applications. Appl. Phys. A 2011, 104, 921–928. [Google Scholar] [CrossRef] [Green Version]
- Reusswig, P.D.; Congreve, D.N.; Thompson, N.J.; Baldo, M.A. Enhanced external quantum efficiency in an organic photovoltaic cell via singlet fission exciton sensitizer. Appl. Phys. Lett. 2012, 101, 113304. [Google Scholar] [CrossRef] [Green Version]
- Jendrzejewski, R.; Majewska, N.; Majumdar, S.; Sawczak, M.; Ryl, J.; Sliwinski, G. Rubrene Thin Films with Viably Enhanced Charge Transport Fabricated by Cryo-Matrix-Assisted Laser Evaporation. Materials 2021, 14, 4413. [Google Scholar] [CrossRef]
- Liu, S.; Wu, H.; Zhang, X.; Hu, W. Research progress of rubrene as an excellent multifunctional organic semiconductor. Front. Phys. 2021, 16, 13304. [Google Scholar] [CrossRef]
- Ivan, T.; Vacareanu, L.; Grigoras, M. Synthesis and Optoelectronic Characterization of Some Star-Shaped Oligomers with Benzene and Triphenylamine Cores. ISRN Org. Chem. 2012, 2012, 976178. [Google Scholar] [CrossRef] [Green Version]
- Stanculescu, A.; Socol, G.; Grigoras, M.; Ivan, T.; Vacareanu, L.; Socol, M.; Rasoga, O.; Breazu, C.; Mihailescu, I.N.; Iordache, I.; et al. Laser prepared organic heterostructures based on star-shaped, arylenevinylene compounds. Appl. Phys. A 2014, 117, 261–268. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Vacareanu, L.; Grigoras, M.; Socol, G.; Mihailescu, I.N.; Stanculescu, F.; Jelinek, M.; Stanculescu, A.; Stoicanescu, M. Organic heterostructures based on arylenevinylene oligomers deposited by MAPLE. Appl. Surf. Sci. 2014, 302, 216–222. [Google Scholar] [CrossRef]
- Vacareanu, L.; Ivan, T.; Grigoras, M. New symmetrical conjugated thiophene-azomethines containing triphenylamine or carbazole units: Synthesis, thermal and optoelectrochemical properties. High Perform. Polym. 2012, 24, 717–729. [Google Scholar] [CrossRef]
- Stanculescu, A.; Rasoga, O.; Socol, M.; Vacareanu, L.; Grigoras, M.; Socol, G.; Stanculescu, F.; Breazu, C.; Matei, E.; Preda, N.; et al. MAPLE prepared heterostructures with oligoazomethine: Fullerene derivative mixed layer for photovoltaic applications. Appl. Surf. Sci. 2017, 417, 183–195. [Google Scholar] [CrossRef]
- Kroon, R.; Lenes, M.; Hummelen, J.C.; Blom, P.W.M.; de Boer, B. Small Bandgap Polymers for Organic Solar Cells (Polymer Material Development in the Last 5 Years). Polym. Rev. 2008, 48, 531–582. [Google Scholar] [CrossRef]
- Park, J.-M.; Cheng, D.; Patz, A.; Luo, L.; Liu, Z.; Fungura, F.; Shinar, S.; Ho, K.-M.; Shinar, J.; Wang, J. Ultrafast nonlinear transparency driven at a telecom wavelength in an organic semiconductor system. AIP Adv. 2019, 9, 025303. [Google Scholar] [CrossRef]
- Berger, P.R.; Kim, M. Polymer solar cells: P3HT:PCBM and beyond. J. Renew. Sustain. Energy 2018, 10, 013508. [Google Scholar] [CrossRef]
- Giro, R.; Caldas, M.J.; Galvão, D.S. Band gap engineering for poly(p-phenylene) and poly(p-phenylene vinylene) copoly-mers using the tight-binding approach. Int. J. Quantum Chem. 2005, 103, 588–596. [Google Scholar] [CrossRef]
- Pratyusha, T.; Sivakumar, G.; Yella, A.; Gupta, D. Novel Ternary Blend of PCDTBT, PCPDTBT and PC 70 BM for the Fabrication of Bulk Heterojunction Organic Solar Cells. Mater. Today Proc. 2017, 4, 5067–5073. [Google Scholar] [CrossRef]
- Li, J.; Zhao, Y.; Tan, H.S.; Guo, Y.; Di, C.-A.; Yu, G.; Liu, Y.; Lin, M.; Lim, S.H.; Zhou, Y.; et al. A stable solution-processed polymer semiconductor with record high-mobility for printed transistors. Sci. Rep. 2012, 2, 754. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; You, P.; Li, J.; Li, J.; Lee, C.-S.; Ong, B.S.; Surya, C.; Yan, F. Enhanced efficiency of polymer solar cells by adding a high-mobility conjugated polymer. Energy Environ. Sci. 2015, 8, 1463–1470. [Google Scholar] [CrossRef]
- Armin, A.; Hambsch, M.; Wolfer, P.; Jin, H.; Li, J.; Shi, Z.; Burn, P.L.; Meredith, P. Efficient, large area, and thick junction polymer solar cells with balanced mobilities and low defect densities. Adv. Energy Mater. 2014, 5, 1401221. [Google Scholar] [CrossRef]
- Lei, Y.; Wu, B.; Chan, W.-K.E.; Zhu, F.; Ong, B.S. Engineering gate dielectric surface properties for enhanced polymer field-effect transistor performance. J. Mater. Chem. C 2015, 3, 12267–12272. [Google Scholar] [CrossRef]
- Yu, G.; Heeger, A.J. Charge separation and photovoltaic conversion in polymer composites with internal donor/acceptor heterojunctions. J. Appl. Phys. 1995, 78, 4510–4515. [Google Scholar] [CrossRef]
- Braatz, C.R.; Öhl, G.; Jakob, P. Vibrational properties of the compressed and the relaxed 1,4,5,8-naphthalene-tetracarboxylic dianhydride monolayer on Ag(111). J. Chem. Phys. 2012, 136, 134706. [Google Scholar] [CrossRef] [PubMed]
- Stanculescu, A.; Socol, M.; Rasoga, O.; Mihailescu, I.N.; Socol, G.; Preda, N.; Breazu, C.; Stanculescu, F. Laser prepared organic hetrostructures on glass/AZO substrates. Appl. Surf. Sci. 2014, 302, 169–176. [Google Scholar] [CrossRef]
- Lassiter, B.E.; Wei, G.; Wang, S.; Zimmerman, J.D.; Diev, V.V.; Thompson, M.E.; Forrest, S.R. Organic photovoltaics incorporating electron conducting exciton blocking layers. Appl. Phys. Lett. 2011, 98, 243307. [Google Scholar] [CrossRef]
- Radu (Călugăr), A.I.; Antohe, V.-A.; Iftimie, S.; Radu, A.; Filipescu, M.; Ion, L.; Dinescu, M.; Antohe, Ş. On the physical and photo-electrical properties of organic photovoltaic cells based on 1,10-Phenanthroline and 5,10,15,20-Tetra(4-pyridyl)-21H,23H-porphine non-fullerene thin films. Appl. Surf. Sci. 2020, 531, 147332. [Google Scholar] [CrossRef]
- Sun, H.; Ryno, S.; Zhong, C.; Ravva, M.K.; Sun, Z.; Körzdörfer, T.; Brédas, J.-L. Ionization Energies, Electron Affinities, and Polarization Energies of Organic Molecular Crystals: Quantitative Estimations from a Polarizable Continuum Model (PCM)-Tuned Range-Separated Density Functional Approach. J. Chem. Theory Comput. 2016, 12, 2906–2916. [Google Scholar] [CrossRef] [Green Version]
- Canulescu, S.; Schou, J.; Nielsen, S.F. Processing of C60 thin films by Matrix-Assisted Pulsed Laser Evaporation (MAPLE). Appl. Phys. A 2011, 104, 775–780. [Google Scholar] [CrossRef]
- Canulescu, S.; Schou, J.; Fæster, S.; Hansen, K.V.; Conseil, H. Deposition of matrix-free fullerene films with improved morphology by matrix-assisted pulsed laser evaporation (MAPLE). Chem. Phys. Lett. 2013, 588, 119–123. [Google Scholar] [CrossRef]
- Majewska, N.; Gazda, M.; Jendrzejewski, R.; Majumdar, S.; Sawczak, M.; Sliwinski, G. Organic semiconductor rubrene thin films deposited by pulsed laser evaporation of solidified solutions. Proc. SPIE 2017, 10453, 104532H. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Breazu, C.; Stanculescu, A.; Costas, A.; Stanculescu, F.; Girtan, M.; Gherendi, F.; Popescu-Pelin, G.; Socol, G. Flexible organic heterostructures obtained by MAPLE. Appl. Phys. A 2018, 124, 602. [Google Scholar] [CrossRef]
- Pate, R.; Stiff-Roberts, A.D. The impact of laser-target absorption depth on the surface and internal morphology of matrix-assisted pulsed laser evaporated conjugated polymer thin films. Chem. Phys. Lett. 2009, 477, 406–410. [Google Scholar] [CrossRef]
- Liu, Y.; Atewologun, A.; Stiff-Roberts, A.D. Organic Semiconductor Thin Films Deposited by Resonant Infrared Matrix-Assisted Pulsed Laser Evaporation: A Fundamental Study of the Emulsion Target. Mater. Res. Soc. Symp. Proc. 2014, 1733. [Google Scholar] [CrossRef]
- Gutierrez-Llorente, A. Growth of polyalkylthiophene films by matrix assisted pulsed laser evaporation. Org. Electron. 2004, 5, 29–34. [Google Scholar] [CrossRef]
- Li, A.; Dong, B.X.; Green, P.F. Influence of morphological disorder on in- and out-of-plane charge transport in conjugated polymer films. MRS Commun. 2015, 5, 593–598. [Google Scholar] [CrossRef]
- Dong, B.X.; Li, A.; Strzalka, J.; Stein, G.E.; Green, P.F. Molecular organization in MAPLE-deposited conjugated polymer thin films and the implications for carrier transport characteristics. J. Polym. Sci. B Polym. Phys. 2017, 55, 39–48. [Google Scholar] [CrossRef]
- Wenderott, J.K.; Dong, B.X.; Green, P.F. Band bending in conjugated polymer films: Role of morphology and implications for bulk charge transport characteristics. J. Mater. Chem. C 2017, 5, 7446–7451. [Google Scholar] [CrossRef]
- Wenderott, J.K.; Green, P.F. Self-Assembled Monolayers at the Conjugated Polymer/Electrode Interface: Implications for Charge Transport and Band-Bending Behavior. ACS Appl. Mater. Interfaces 2018, 10, 21458–21465. [Google Scholar] [CrossRef]
- McCormick, R.D.; Lenhardt, J.; Stiff-Roberts, A.D. Effects of Emulsion-Based Resonant Infrared Matrix Assisted Pulsed Laser Evaporation (RIR-MAPLE) on the Molecular Weight of Polymers. Polymers 2012, 4, 341–354. [Google Scholar] [CrossRef] [Green Version]
- Dong, B.X.; Smith, M.; Strzalka, J.; Li, H.; McNeil, A.J.; Stein, G.E.; Green, P.F. Molecular weight dependent structure and charge transport in MAPLE-deposited poly(3-hexylthiophene) thin films. J. Polym. Sci. B Polym. Phys. 2018, 56, 652–663. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Stanculescu, A.; Breazu, C.; Florica, C.; Stanculescu, F.; Iftimie, S.; Girtan, M.; Popescu-Pelin, G.; Socol, G. Organic heterostructures deposited by MAPLE on AZO substrate. Appl. Surf. Sci. 2017, 417, 196–203. [Google Scholar] [CrossRef]
- Stanculescu, A.; Socol, G.; Vacareanu, L.; Socol, M.; Rasoga, O.; Breazu, C.; Girtan, M.; Stanculescu, F. MAPLE preparation and characterization of mixed arylenevinylene based oligomers:C60 layers. Appl. Surf. Sci. 2016, 374, 278–289. [Google Scholar] [CrossRef]
- Stiff-Roberts, A.D.; McCormick, R.D.; Ge, W. Material properties and applications of blended organic thin films with nanoscale domains deposited by RIR-MAPLE. Proc. SPIE 2015, 9350, 935007. [Google Scholar] [CrossRef]
- Yen, C.-P.; Yu, P.-F.; Wang, J.; Lin, J.-Y.; Chen, Y.-M.; Chen, S. Deposition of organic dyes for dye-sensitized solar cell by using matrix-assisted pulsed laser evaporation. AIP Adv. 2016, 6, 085011. [Google Scholar] [CrossRef] [Green Version]
- Dunlap-Shohl, W.A.; Barraza, E.T.; Barrette, A.; Gundogdu, K.; Stiff-Roberts, A.D.; Mitzi, D.B. MAPbI3 Solar Cells with Absorber Deposited by Resonant Infrared Matrix-Assisted Pulsed Laser Evaporation. ACS Energy Lett. 2018, 3, 270–275. [Google Scholar] [CrossRef]



| Organic Compounds | Solvent for Host Matrix | Concentration of the Organic Compound in the Primary Solvent | Laser Wavelength | Laser Fluence | Published Year | Ref. |
|---|---|---|---|---|---|---|
| MEH-PPV | THF * | 0.3 wt.% | 193 nm | <190 mJ/cm2 | 2004 | [49] |
| TOL * | ||||||
| THF | 0.3 wt.% | 248 nm | ||||
| TOL | ||||||
| CHL * | 8.2 μm | 0.5 J/cm2 | ||||
| P3HT | OX * | 0.8 wt.% | 266 nm | 200 mJ/cm2 | 2004 | [145] |
| MEH-CN-PPV | THF | 1 wt.% | 2.9 μm | 0.67–2 J/cm2 | 2008 | [61] |
| CB * | ||||||
| TOL | ||||||
| OX | ||||||
| CHL | ||||||
| PH *:THF | ||||||
| PH:DIW * | 0.25 wt.% | |||||
| MEH-PPV | THF | 1 wt.% | ||||
| TOL | ||||||
| CHL | ||||||
| PH:THF | ||||||
| PH:DIW | 0.25 wt.% | |||||
| MEH-CN-PPV | PH:DIW | 1 wt.% | 2.9 μm | 2 J/cm2 | 2009 | [143] |
| C60 | ANS * | 0.67 wt.% | 355 nm | 0.15–3.9 J/cm2 | 2011 | [139] |
| ZnPc | DMSO * | 2.5 g/L | 248 nm | 433 mJ/cm2 | 2011 | [115] |
| PTCDA | DMSO | 2.5 g/L or 4 g/L | 333–400 mJ/cm2 | |||
| Alq3 | CHL | 4 g/L | 166 or 433 mJ/cm2 | |||
| DMSO | 2.5 g/L | 350 mJ/cm2 | ||||
| PEDOT:PSS | NMP * | 1.2–1.4 wt.% | 3 µm | 2 J/cm2 | 2011 | [48] |
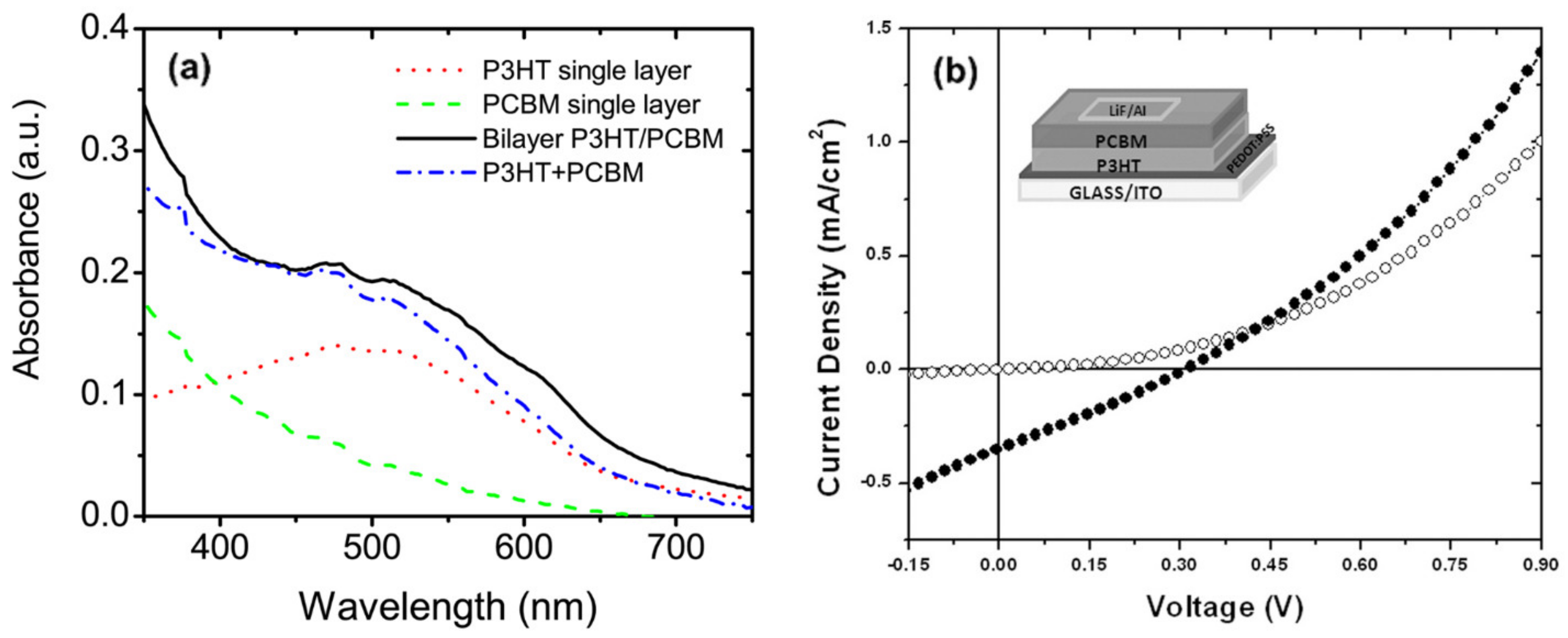
| P3HT/PCBM | TOL | 0.3 wt.% | 248 nm | 250 mJ/cm2 | 2012 | [38] |
| P3HT MEH-PPV | TOL:PH:DIW | 1 wt.% | 2.94 μm | Not mentioned | 2012 | [150] |
| C60 | ANS | 0.67 wt.% | 355 nm | 0.5–4 J/cm2 | 2013 | [140] |
| ZnPc NTCDA | DMSO | 2.5 g/L | 248 nm | 300 or 400 mJ/cm2 | 2014 | [135] |
| IT77:PCBB | CHL | 1 g/L or 3 g/L | 248 nm | 250 or 300 mJ/cm2 | 2014 | [120] |
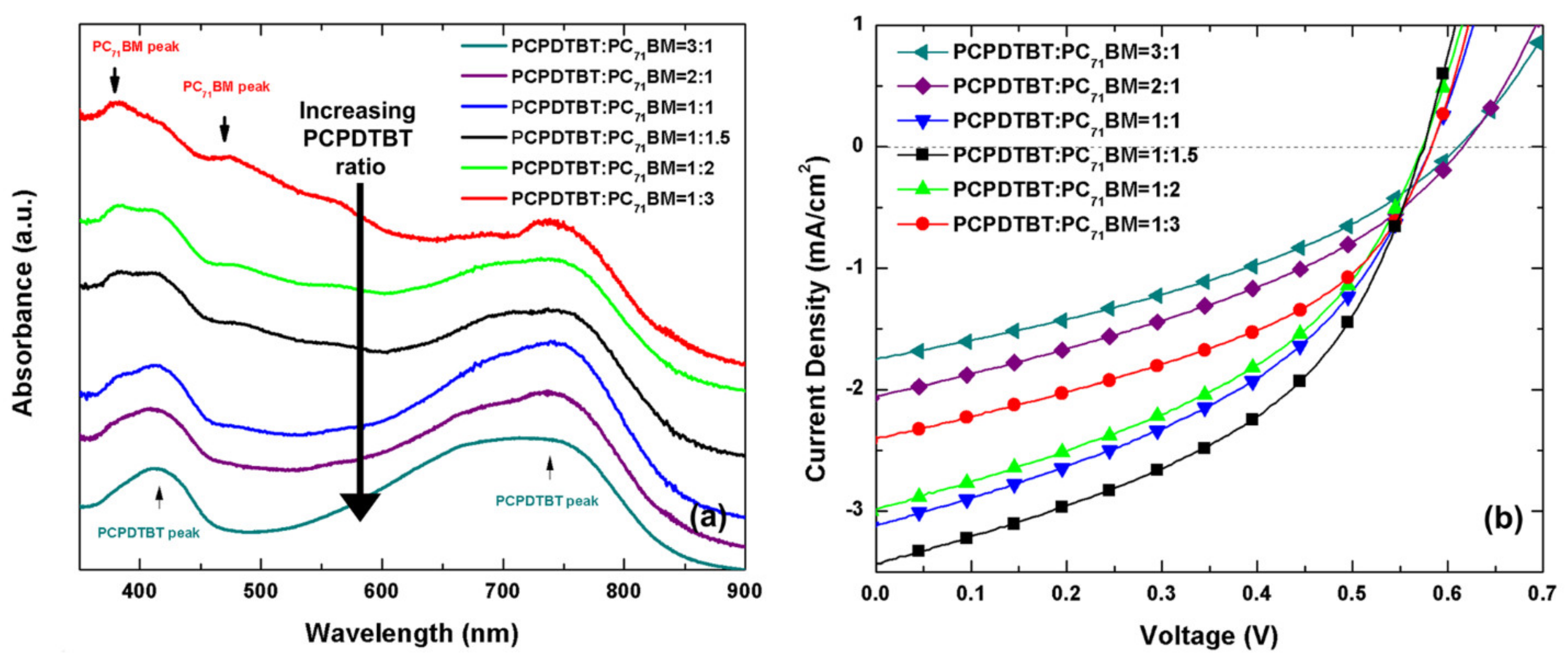
| PCPDTBT:PC71BM | CB *:PH: DIW:SLS | 1 wt.% | 2.94 μm | 2 J/cm2 | 2014 | [69] |
| P3HT | solvent:PH: alcohol:DIW | 0.5 wt.% | 2.9 μm | 1.46–1.6 J/cm2 | 2014 | [144] |
| AMC16 AMC22 AMC16:C60 AMC22:C60 | CHL | 3 g/L | 248 nm | 250 mJ/cm2 | 2015 | [88] |
| P3HT | ODCB *:BA *: DIW:SDS * | 5 mg/mL | 2.94 μm | 1.3 J/cm2 | 2015 | [146] |
| P3HT | ODCB:BA: DIW:SDS | 1 wt.% | 2.94 μm | 1.7 J/cm2 | 2015 | [154] |
| PCPDTBT | CB:PH: DIW:SDS | |||||
| ODCB:PH: DIW:SDS | ||||||
| TCB *:PH: DIW:SDS | ||||||
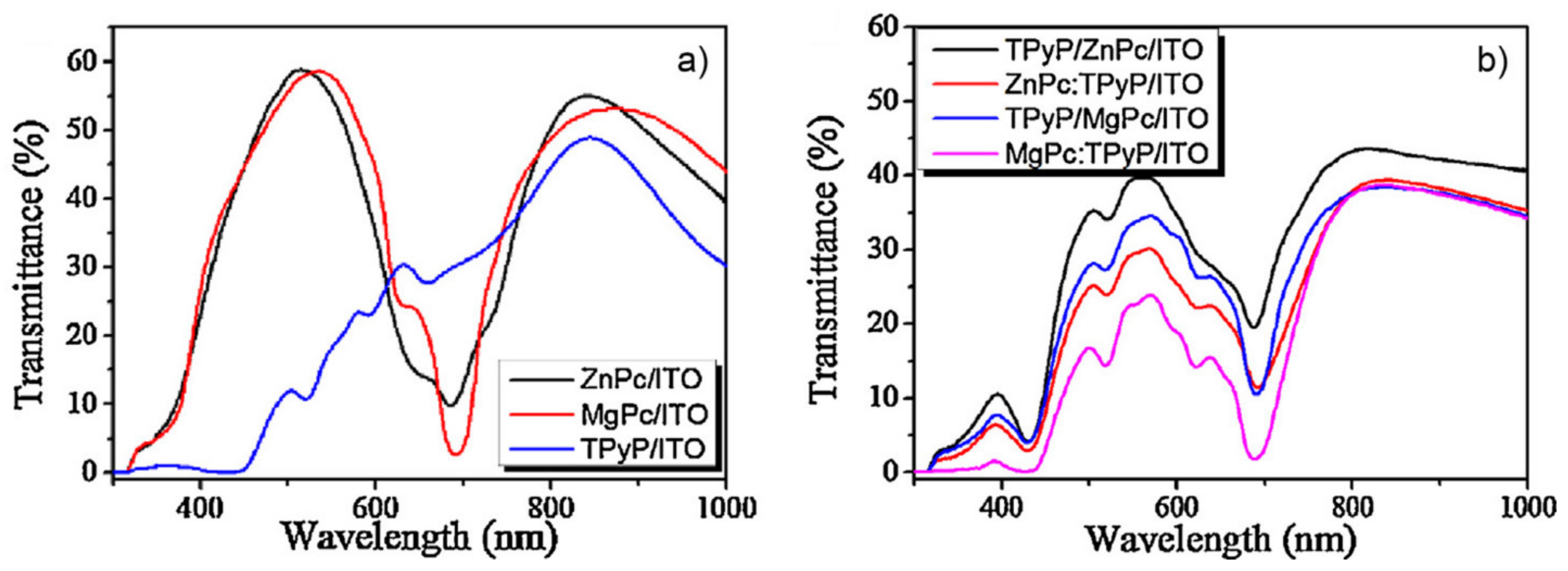
| ZnPc/TPyP ZnPc:TPyP MgPc/TPyP MgPc:TPyP | DMSO | 2.5 g/L | 248 nm | 300 mJ/cm2 | 2016 | [40] |
| LS13:C60 LS78:C60 | ODCB | 4 g/L | 248 nm | 300 mJ/cm2 | 2016 | [153] |
| P3HT:PCBM PCPDTBT:PC71BM | TL *:PH:DIW | 5 mg/mL | 2.94 μm | 1.8 J/cm2 | 2016 | [70] |
| OX:PH:DIW | ||||||
| PDC:PH:DIW | ||||||
| CB:PH:DIW | ||||||
| ODCB:PH:DIW | ||||||
| TCB:PH:DIW | ||||||
| rubrene | TOL | 0.23–1 wt.% | 266 nm or 1064 nm | 0.22–10.37 J/cm2 | 2017 | [141] |
| XL | ||||||
| DCM | ||||||
| DCE | ||||||
| LV4:C61 LV5:C61 | CHL | 3 g/L | 248 nm | 312 mJ/cm2 | 2017 | [123] |
| DMSO | ||||||
| P3HT:C60 | TOL | 3 g/L | 248 nm | 250 mJ/cm2 | 2017 | [152] |
| P3HT | ODCB:BA: DIW:SDS | 5 mg/mL | 2.94 μm | 1.3 J/cm2 | 2017 | [37,147,148] |
| ZnPc | DMSO | 3 g/L | 248 nm | 250 mJ/cm2 | 2018 | [142] |
| PTCDA | 3 g/L | |||||
| Alq3 | 1.5 g/L | |||||
| P3HT | ODCB:BA: DIW:SDS | 5 mg/mL | 2.94 μm | 1.3 J/cm2 | 2018 | [149,151] |
| ZnPc:C60 ZnPc:PC70BM ZnPc:PC70BM:rubrene | DMSO | 3 g/L | 248 nm | 300 mJ/cm2 | 2020 | [86] |
| IT77:AMC14 | DMSO | 3 g/L | 248 nm | 250 mJ/cm2 | 2020 | [85] |
| DPP-DTT:C60 | CHL | 3 g/L | 193 nm | 100 mJ/cm2 | 2020 | [87] |
| rubrene | DCE * | 0.3–0.7 wt.% | 1064 nm | 3–4.8 J/cm2 | 2021 | [117] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Socol, M.; Preda, N.; Socol, G. Organic Thin Films Deposited by Matrix-Assisted Pulsed Laser Evaporation (MAPLE) for Photovoltaic Cell Applications: A Review. Coatings 2021, 11, 1368. https://doi.org/10.3390/coatings11111368
Socol M, Preda N, Socol G. Organic Thin Films Deposited by Matrix-Assisted Pulsed Laser Evaporation (MAPLE) for Photovoltaic Cell Applications: A Review. Coatings. 2021; 11(11):1368. https://doi.org/10.3390/coatings11111368
Chicago/Turabian StyleSocol, Marcela, Nicoleta Preda, and Gabriel Socol. 2021. "Organic Thin Films Deposited by Matrix-Assisted Pulsed Laser Evaporation (MAPLE) for Photovoltaic Cell Applications: A Review" Coatings 11, no. 11: 1368. https://doi.org/10.3390/coatings11111368
APA StyleSocol, M., Preda, N., & Socol, G. (2021). Organic Thin Films Deposited by Matrix-Assisted Pulsed Laser Evaporation (MAPLE) for Photovoltaic Cell Applications: A Review. Coatings, 11(11), 1368. https://doi.org/10.3390/coatings11111368







