Thin Film Fabrication by Pulsed Laser Deposition from TiO2 Targets in O2, N2, He, or Ar for Dye-Sensitized Solar Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of PLD Bilayers
2.2. Solar Cell Fabrication
2.3. Physicochemical Characterization
3. Results and Discussion
3.1. Thin-Film Morphology and Topology
3.1.1. Top and Cross-Section SEM
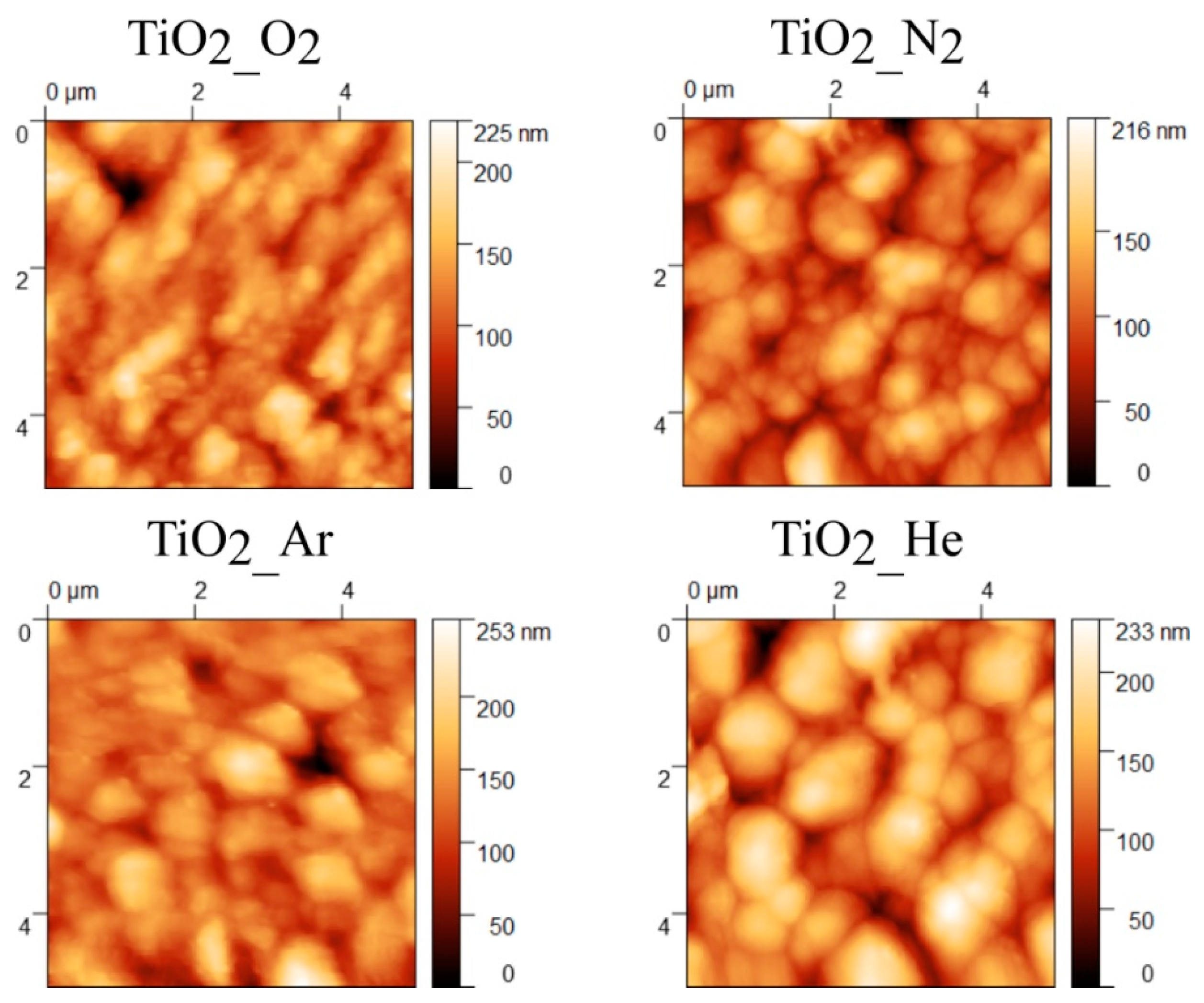
3.1.2. AFM
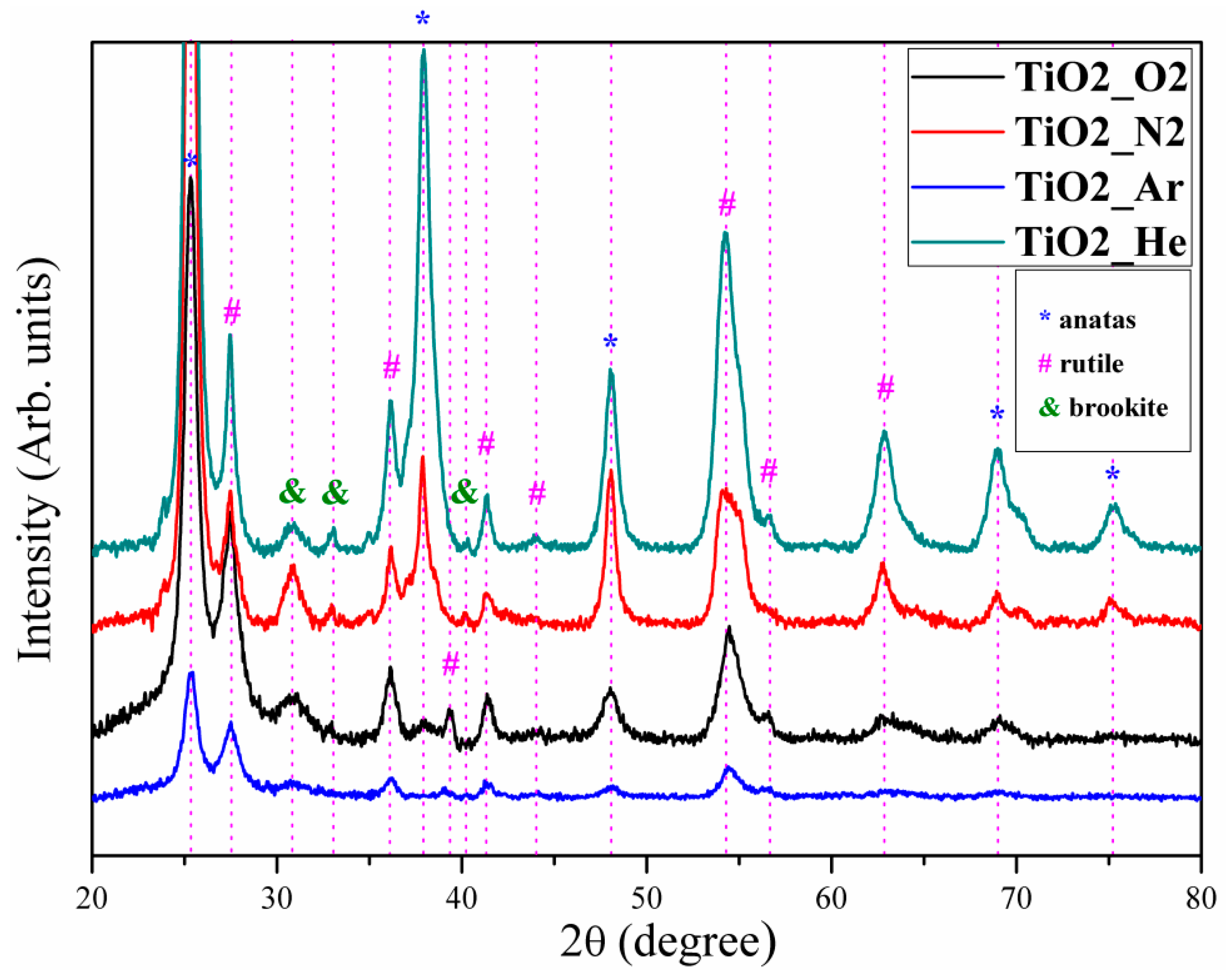
3.2. Thin-Film Structure and Composition
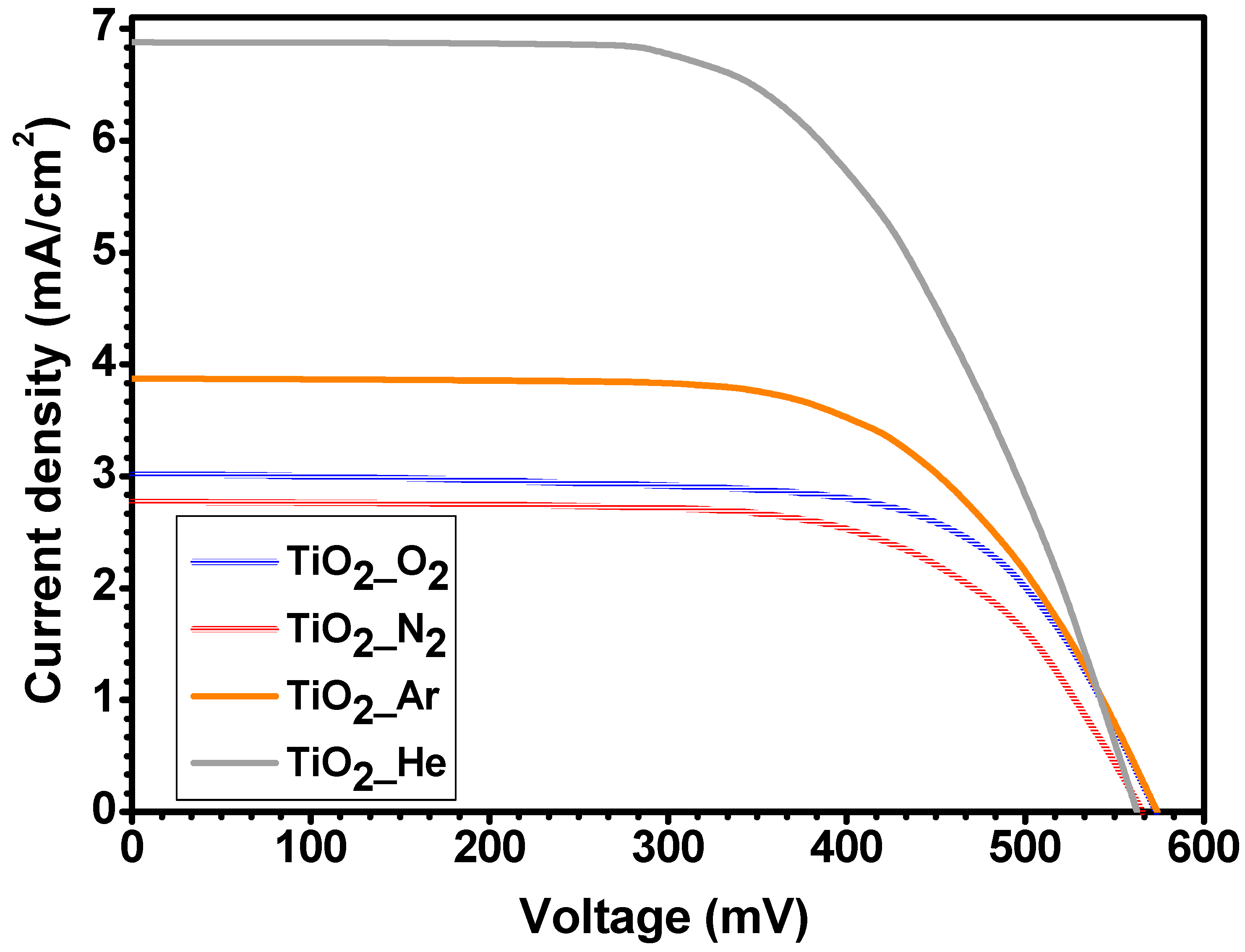
3.3. PV Characteristics of Prepared Solar Cells
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Grätzel, M. Photoelectrochemical cells. Nature 2001, 414, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Bahnemann, D. Photocatalytic water treatment: Solar energy applications. Sol. Energy 2004, 77, 445–459. [Google Scholar] [CrossRef]
- Perera, F. Pollution from Fossil-Fuel Combustion is the Leading Environmental Threat to Global Pediatric Health and Equity: Solutions Exist. Int. J. Environ. Res. Public Health 2018, 15, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chapin, D.M.; Fuller, C.S.; Pearson, G.L. A New Silicon p-n Junction Photocell for Converting Solar Radiation into Electrical Power. J. Appl. Phys. 1954, 25, 676–677. [Google Scholar] [CrossRef]
- O’Regan, B.; Grätzel, M. A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 1991, 353, 737–740. [Google Scholar] [CrossRef]
- Tong, Z.; Peng, T.; Sun, W.; Liu, W.; Guo, S.; Zhao, X.-Z. Introducing an Intermediate Band into Dye-Sensitized Solar Cells by W6+ Doping into TiO2 Nanocrystalline Photoanodes. J. Phys. Chem. C 2014, 118, 16892–16895. [Google Scholar] [CrossRef]
- Kakiage, K.; Aoyama, Y.; Yano, T.; Otsuka, T.; Kyomen, T.; Unno, M.; Hanaya, M. An achievement of over 12 percent efficiency in an organic dye-sensitized solar cell. Chem. Commun. 2014, 50, 6379–6381. [Google Scholar] [CrossRef]
- Graetzel, M.; Janssen, R.A.J.; Mitzi, D.B.; Sargent, E.H. Materials interface engineering for solution-processed photovoltaics. Nature 2012, 488, 304–312. [Google Scholar] [CrossRef]
- Mariotti, N.; Bonomo, M.; Fagiolari, L.; Barbero, N.; Gerbaldi, C.; Bella, F.; Barolo, C. Recent advances in eco-friendly and cost-effective materials towards sustainable dye-sensitized solar cells. Green Chem. 2020, 22, 7168–7218. [Google Scholar] [CrossRef]
- Yeoh, M.-E.; Chan, K.-Y. Recent advances in photo-anode for dye-sensitized solar cells: A review. Int. J. Energy Res. 2017, 41, 2446–2467. [Google Scholar] [CrossRef]
- Scarabino, S.; Nonomura, K.; Vlachopoulos, N.; Hagfeldt, A.; Wittstock, G. Effect of TiO2 Photoanodes Morphology and Dye Structure on Dye-Regeneration Kinetics Investigated by Scanning Electrochemical Microscopy. Electrochem 2020, 1, 329–343. [Google Scholar] [CrossRef]
- Toe, M.Z.; Matsuda, A.; Han, S.S.; Yaacob, K.A.; Pung, S.-Y. Effect of annealing temperature on the performance of ZnO thin film-based dye sensitized solar cell. AIP Conf. Proc. 2020, 2267, 20010. [Google Scholar] [CrossRef]
- Dhamodharan, P.; Chen, J.; Manoharan, C. Fabrication of dye-sensitized solar cells with ZnO nanorods as photoanode and natural dye extract as sensitizer. J. Mater. Sci. Mater. Electron. 2021, 32, 13418–13429. [Google Scholar] [CrossRef]
- Sufyan, M.; Mehmood, U.; Gill, Y.Q.; Nazar, R.; Khan, A.U.H. Hydrothermally synthesize zinc oxide (ZnO) nanorods as an effective photoanode material for third-generation Dye-sensitized solar cells (DSSCs). Mater. Lett. 2021, 297, 130017. [Google Scholar] [CrossRef]
- Lee, T.-H.; Sue, H.-J.; Cheng, X. Solid-state dye-sensitized solar cells based on ZnO nanoparticle and nanorod array hybrid photoanodes. Nanoscale Res. Lett. 2011, 6, 517. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yildiz, Z.K.; Atilgan, A.; Atli, A.; Özel, K.; Altinkaya, C.; Yildiz, A. Enhancement of efficiency of natural and organic dye sensitized solar cells using thin film TiO2 photoanodes fabricated by spin-coating. J. Photochem. Photobiol. A Chem. 2019, 368, 23–29. [Google Scholar] [CrossRef]
- Gnida, P.; Jarka, P.; Chulkin, P.; Drygała, A.; Libera, M.; Tański, T.; Schab-Balcerzak, E. Impact of TiO2 Nanostructures on Dye-Sensitized Solar Cells Performance. Materials 2021, 14, 1633. [Google Scholar] [CrossRef]
- Kathirvel, S.; Chen, H.-S.; Su, C.; Wang, H.-H.; Li, C.-Y.; Li, W.-R. Preparation of Smooth Surface TiO2 Photoanode for High Energy Conversion Efficiency in Dye-Sensitized Solar Cells. J. Nanomater. 2013, 2013, 367510. [Google Scholar] [CrossRef] [Green Version]
- Tsai, J.K.; Hsu, W.D.; Wu, T.C.; Meen, T.H.; Chong, W.J. Effect of compressed TiO2 nanoparticle thin film thickness on the performance of dye-sensitized solar cells. Nanoscale Res. Lett. 2013, 8, 459. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, M.S.; Pandey, A.K.; Rahim, N.A. Towards the plasmonic effect of Zn nanoparticles on TiO2 monolayer photoanode for dye sensitized solar cell applications. Mater. Lett. 2017, 195, 62–65. [Google Scholar] [CrossRef]
- Tyagi, J.; Gupta, H.; Purohit, L.P. Mesoporous ZnO/TiO2 photoanodes for quantum dot sensitized solar cell. Opt. Mater. 2021, 115, 111014. [Google Scholar] [CrossRef]
- Zhang, Q.; Li, C. TiO2 Coated ZnO Nanorods by Mist Chemical Vapor Deposition for Application as Photoanodes for Dye-Sensitized Solar Cells. Nanomaterials 2019, 9, 1339. [Google Scholar] [CrossRef] [Green Version]
- Bhogaita, M.; Devaprakasam, D. Hybrid photoanode of TiO2-ZnO synthesized by co-precipitation route for dye-sensitized solar cell using phyllanthus reticulatas pigment sensitizer. Sol. Energy 2021, 214, 517–530. [Google Scholar] [CrossRef]
- Kumi-Barimah, E.; Penhale-Jones, R.; Salimian, A.; Upadhyaya, H.; Hasnath, A.; Jose, G. Phase evolution, morphological, optical and electrical properties of femtosecond pulsed laser deposited TiO2 thin films. Sci. Rep. 2020, 10, 10144. [Google Scholar] [CrossRef] [PubMed]
- Deepa, H.A.; Madhu, G.M.; Venkatesham, V. Performance evaluation of DSSC’s fabricated employing TiO2 and TiO2-ZnO nanocomposite as the photoanodes. Mater. Today Proc. 2021, 46, 4579–4586. [Google Scholar] [CrossRef]
- Lungu, J.; Socol, G.; Stan, G.E.; Ştefan, N.; Luculescu, C.; Georgescu, A.; Popescu-Pelin, G.; Prodan, G.; Gîrţu, M.A.; Mihăilescu, I.N. Pulsed laser fabrication of TiO2 buffer layers for dye sensitized solar cells. Nanomaterials 2019, 9, 746. [Google Scholar] [CrossRef] [Green Version]
- Socol, G.; Gnatyuk, Y.; Stefan, N.; Smirnova, N.; Djokić, V.; Sutan, C.; Malinovschi, V.; Stanculescu, A.; Korduban, O.; Mihailescu, I.N. Photocatalytic activity of pulsed laser deposited TiO2 thin films in N2, O2 and CH4. Thin Solid Films 2010, 518, 4648–4653. [Google Scholar] [CrossRef]
- Chrisey, D.B.; Hubler, G.K. Pulsed Laser Deposition of Thin Films; Wiley: Hoboken, NJ, USA, 1994; ISBN 9780471592181. [Google Scholar]
- Eason, R. Pulsed Laser Deposition of Thin Films: Applications-Led Growth of Functional Materials; Wiley: Hoboken, NJ, USA, 2007; ISBN 9780470052112. [Google Scholar]
- Fusi, M.; Maccallini, E.; Caruso, T.; Casari, C.S.; Li Bassi, A.; Bottani, C.E.; Rudolf, P.; Prince, K.C.; Agostino, R.G. Surface electronic and structural properties of nanostructured titanium oxide grown by pulsed laser deposition. Surf. Sci. 2011, 605, 333–340. [Google Scholar] [CrossRef] [Green Version]
- Mihailescu, I.N.; Lita, A.; Teodorescu, V.S.; Luches, A.; Martino, M.; Perrone, A.; Gartner, M. Pulsed laser deposition of silicon nitride thin films by laser ablation of a Si target in low pressure ammonia. J. Mater. Sci. 1996, 31, 2839–2847. [Google Scholar] [CrossRef]
- Gyorgy, E.; Mihailescu, I.N.; Κομπίτσας, Μ.; Giannoudakos, A. Particulates-free Ta thin films obtained by pulsed laser deposition: The role of a second laser in the laser-induced plasma heating. Appl. Surf. Sci. 2002, 195, 270–276. [Google Scholar] [CrossRef]
- Amoruso, S. 6—Plume characterization in pulsed laser deposition of metal oxide thin films. In Metal Oxides; Pryds, N., Esposito, V., Korotcenkov, G., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 133–160. ISBN 978-0-12-811166-6. [Google Scholar]
- Yap, S.S.; Yong, T.K.; Nee, C.H.; Tou, T.Y. Pulsed Laser Deposition of ITO: From Films to Nanostructures. In Applications of Laser Ablation—Thin Film Deposition, Nanomaterial Synthesis and Surface Modification; Yong, T.K., Ed.; IntechOpen: Rijeka, Croatia, 2016; p. 4. ISBN 978-953-51-2812-0. [Google Scholar]
- Dawood, M.S.; Hamdan, A.; Margot, J. Influence of surrounding gas, composition and pressure on plasma plume dynamics of nanosecond pulsed laser-induced aluminum plasmas. AIP Adv. 2015, 5, 107143. [Google Scholar] [CrossRef] [Green Version]
- Hagfeldt, A.; Boschloo, G.; Sun, L.; Kloo, L.; Pettersson, H. Dye-Sensitized Solar Cells. Chem. Rev. 2010, 110, 6595–6663. [Google Scholar] [CrossRef] [PubMed]
- Nazeeruddin, M.K.; Kay, A.; Rodicio, I.; Humphry-Baker, R.; Mueller, E.; Liska, P.; Vlachopoulos, N.; Graetzel, M. Conversion of light to electricity by cis-X2bis(2,2’-bipyridyl-4,4’-dicarboxylate)ruthenium(II) charge-transfer sensitizers (X = Cl−, Br−, I−, CN−, and SCN−) on nanocrystalline titanium dioxide electrodes. J. Am. Chem. Soc. 1993, 115, 6382–6390. [Google Scholar] [CrossRef]
- Smestad, G.; Gratzel, M. Demonstrating Electron Transfer and Nanotechnology: A Natural Dye-Sensitized Nanocrystalline Energy Converter. J. Chem. Educ. 1998, 75, 75. [Google Scholar] [CrossRef]
- Millington, K.R.; Fincher, K.W.; King, A.L. Mordant dyes as sensitisers in dye-sensitised solar cells. Sol. Energy Mater. Sol. Cells 2007, 91, 1618–1630. [Google Scholar] [CrossRef]
- Georgescu, A.; Damache, G.; Gîtu, M.A. Class A small area solar simulator for dye-sensitized solar cell testing. J. Optoelectron. Adv. Mater. 2008, 10, 3003–3007. [Google Scholar]
- Hafez, H.S.; Shenouda, S.S.; Fadel, M. Photovoltaic characteristics of natural light harvesting dye sensitized solar cells. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2018, 192, 23–26. [Google Scholar] [CrossRef]
- Wen, K.; Liu, M.; Liu, X.; Deng, C.; Zhou, K. Deposition of Photocatalytic TiO2 Coating by Modifying the Solidification Pathway in Plasma Spraying. Coatings 2017, 7, 169. [Google Scholar] [CrossRef] [Green Version]
- Iliescu, M.; Nelea, V.; Werckmann, J.; Mihailescu, I.N.; Socol, G.; Bigi, A.; Bracci, B. Electron microscopy studies of octa-calcium phosphate thin films obtained by pulsed laser deposition. Thin Solid Films 2004, 453–454, 157–161. [Google Scholar] [CrossRef]
- Mihailescu, I.N.; Teodorescu, V.S.; Gyorgy, E.; Luches, A.; Perrone, A.; Martino, M. About the nature of particulates covering the surface of thin films obtained by reactive pulsed laser deposition. J. Phys. D Appl. Phys. 1998, 31, 2236–2240. [Google Scholar] [CrossRef]
- Popescu, A.C.; Stan, G.E.; Duta, L.; Nita, C.; Popescu, C.; Surdu, V.-A.; Husanu, M.-A.; Bita, B.; Ghisleni, R.; Himcinschi, C.; et al. The Role of Ambient Gas and Pressure on the Structuring of Hard Diamond-Like Carbon Films Synthesized by Pulsed Laser Deposition. Materials 2015, 8, 3284–3305. [Google Scholar] [CrossRef] [Green Version]
- Bergström, D.; Powell, J.; Kaplan, A.F.H. The absorption of light by rough metal surfaces—A three-dimensional ray-tracing analysis. J. Appl. Phys. 2008, 103, 103515. [Google Scholar] [CrossRef]
- Bergström, D.; Powell, J.; Kaplan, A.F.H. A ray-tracing analysis of the absorption of light by smooth and rough metal surfaces. J. Appl. Phys. 2007, 101, 113504. [Google Scholar] [CrossRef] [Green Version]
- Abedi, H.; Hoseinpour Gollo, M. An experimental study of the effects of surface roughness and coating of Cr2O3 layer on the laser-forming process. Opt. Laser Technol. 2019, 109, 336–347. [Google Scholar] [CrossRef]
- Scholtz, L.; Ladanyi, L.; Mullerova, J. Influence of surface roughness on optical characteristics of multilayer solar cells. Adv. Electr. Electron. Eng. 2014, 12, 631–638. [Google Scholar] [CrossRef]
- Ding, J.; Li, Y.; Hu, H.; Bai, L.; Zhang, S.; Yuan, N. The influence of anatase-rutile mixed phase and ZnO blocking layer on dye-sensitized solar cells based on TiO2nanofiberphotoanodes. Nanoscale Res. Lett. 2013, 8, 9. [Google Scholar] [CrossRef] [Green Version]
- Hao, Q.; Fu, X.; Song, S.; Gibson, D.; Li, C.; Chu, H.O.; Shi, Y. Investigation of TiO2 Thin Film Deposited by Microwave Plasma Assisted Sputtering and Its Application in 3D Glasses. Coatings 2018, 8, 270. [Google Scholar] [CrossRef] [Green Version]
- Ba-Abbad, M.; Kadhum, A.; Mohamad, A.B.; Takriff, M.; Sopian, K. Synthesis and Catalytic Activity of TiO2 Nanoparticles for Photochemical Oxidation of Concentrated Chlorophenols under Direct Solar Radiation. Int. J. Electrochem. Sci. 2012, 7, 4871–4888. [Google Scholar]
- Wiatrowski, A.; Mazur, M.; Obstarczyk, A.; Wojcieszak, D.; Kaczmarek, D.; Morgiel, J.; Gibson, D. Comparison of the Physicochemical Properties of TiO2 Thin Films Obtained by Magnetron Sputtering with Continuous and Pulsed Gas Flow. Coatings 2018, 8, 412. [Google Scholar] [CrossRef] [Green Version]
- Khan, A.F.; Mehmood, M.; Durrani, S.K.; Ali, M.L.; Rahim, N.A. Structural and optoelectronic properties of nanostructured TiO2 thin films with annealing. Mater. Sci. Semicond. Process. 2015, 29, 161–169. [Google Scholar] [CrossRef]
- Wold, A. Photocatalytic Properties of TiO2. Chem. Mater. 1993, 5, 280–283. [Google Scholar] [CrossRef]
- Hoffmann, M.R.; Martin, S.T.; Choi, W.; Bahnemann, D.W. Environmental Applications of Semiconductor Photocatalysis. Chem. Rev. 1995, 95, 69–96. [Google Scholar] [CrossRef]
- Sima, F.; Sugioka, K. Ultrafast laser manufacturing of nanofluidic systems. Nanophotonics 2021, 10, 2389–2406. [Google Scholar] [CrossRef]
- Bocquet, L. Nanofluidics coming of age. Nat. Mater. 2020, 19, 254–256. [Google Scholar] [CrossRef] [Green Version]
- Bocquet, L.; Charlaix, E. Nanofluidics, from bulk to interfaces. Chem. Soc. Rev. 2010, 39, 1073–1095. [Google Scholar] [CrossRef] [Green Version]
- Islam, S.Z.; Reed, A.; Kim, D.Y.; Rankin, S.E. N2/Ar plasma induced doping of ordered mesoporous TiO2 thin films for visible light active photocatalysis. Microporous Mesoporous Mater. 2016, 220, 120–128. [Google Scholar] [CrossRef] [Green Version]
- Yao, Z.; Jia, F.; Tian, S.; Li, C.; Jiang, Z.; Bai, X. Microporous Ni-Doped TiO2 film Photocatalyst by Plasma Electrolytic Oxidation. ACS Appl. Mater. Interfaces 2010, 2, 2617–2622. [Google Scholar] [CrossRef]


| Sample | Pressure (Pa) | Nature of the Gas | No. of Pulses |
|---|---|---|---|
| TiO2_O2 | 10/100 | Oxygen, O2 | 1000/20,000 |
| TiO2_N2 | 10/100 | Nitrogen, N2 | 1000/80,000 |
| TiO2_Ar | 8/80 | Argon, Ar | 1000/20,000 |
| TiO2_He | 5.9/5900 | Helium, He | 1000/40,000 |
| Sample Code | Thickness (µm) | Deposition Rate (Å/Pulse) | Average Crystallite Size (nm) | RMS (nm) |
|---|---|---|---|---|
| TiO2_O2 | 6.49 ± 0.25 | 3.1 ± 0.1 | 9.9 ± 2.4 | 32.3 ± 4 |
| TiO2_N2 | 4 ± 0.48 | 0.5 ± 0.06 | 12.3 ± 3.4 | 30.3 ± 5.8 |
| TiO2_Ar | 3 ± 0.25 | 1.4 ± 0.1 | 10.6 ± 2.7 | 40 ± 6.7 |
| TiO2_He | 6.55 ± 0.53 | 1.6 ± 0.1 | 12.5 ± 3.8 | 44 ± 2.9 |
| Sample | Isc (mA) | Voc (mV) | Jsc (mA/cm2) | Pmax (μW) | FF | η (%) |
|---|---|---|---|---|---|---|
| TiO2_O2 | 2.37 | 573.4 | 3.014 | 930 | 0.69 | 1.18 |
| TiO2_N2 | 2.18 | 565.7 | 2.771 | 802 | 0.65 | 1.02 |
| TiO2_Ar | 3.04 | 574.0 | 3.873 | 1117 | 0.64 | 1.42 |
| TiO2_He | 5.40 | 562.7 | 6.881 | 1825 | 0.60 | 2.32 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Albu, D.F.; Lungu, J.; Popescu-Pelin, G.; Mihăilescu, C.N.; Socol, G.; Georgescu, A.; Socol, M.; Bănică, A.; Ciupina, V.; Mihailescu, I.N. Thin Film Fabrication by Pulsed Laser Deposition from TiO2 Targets in O2, N2, He, or Ar for Dye-Sensitized Solar Cells. Coatings 2022, 12, 293. https://doi.org/10.3390/coatings12030293
Albu DF, Lungu J, Popescu-Pelin G, Mihăilescu CN, Socol G, Georgescu A, Socol M, Bănică A, Ciupina V, Mihailescu IN. Thin Film Fabrication by Pulsed Laser Deposition from TiO2 Targets in O2, N2, He, or Ar for Dye-Sensitized Solar Cells. Coatings. 2022; 12(3):293. https://doi.org/10.3390/coatings12030293
Chicago/Turabian StyleAlbu, Dorel F., Jeanina Lungu, Gianina Popescu-Pelin, Cristian N. Mihăilescu, Gabriel Socol, Adrian Georgescu, Marcela Socol, Alexandra Bănică, Victor Ciupina, and Ion N. Mihailescu. 2022. "Thin Film Fabrication by Pulsed Laser Deposition from TiO2 Targets in O2, N2, He, or Ar for Dye-Sensitized Solar Cells" Coatings 12, no. 3: 293. https://doi.org/10.3390/coatings12030293
APA StyleAlbu, D. F., Lungu, J., Popescu-Pelin, G., Mihăilescu, C. N., Socol, G., Georgescu, A., Socol, M., Bănică, A., Ciupina, V., & Mihailescu, I. N. (2022). Thin Film Fabrication by Pulsed Laser Deposition from TiO2 Targets in O2, N2, He, or Ar for Dye-Sensitized Solar Cells. Coatings, 12(3), 293. https://doi.org/10.3390/coatings12030293











