Electrophoretic Deposition and Characterization of Functional Coatings Based on an Antibacterial Gallium (III)-Chitosan Complex
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Preparation of EPD Suspensions
2.3. Deposition Procedure
2.4. Suspension Characterization
2.5. SEM and EDX Spectroscopy
2.6. FTIR Analysis
2.7. Contact Angle Measurements
2.8. Swelling Study
2.9. In Vitro Degradation Study
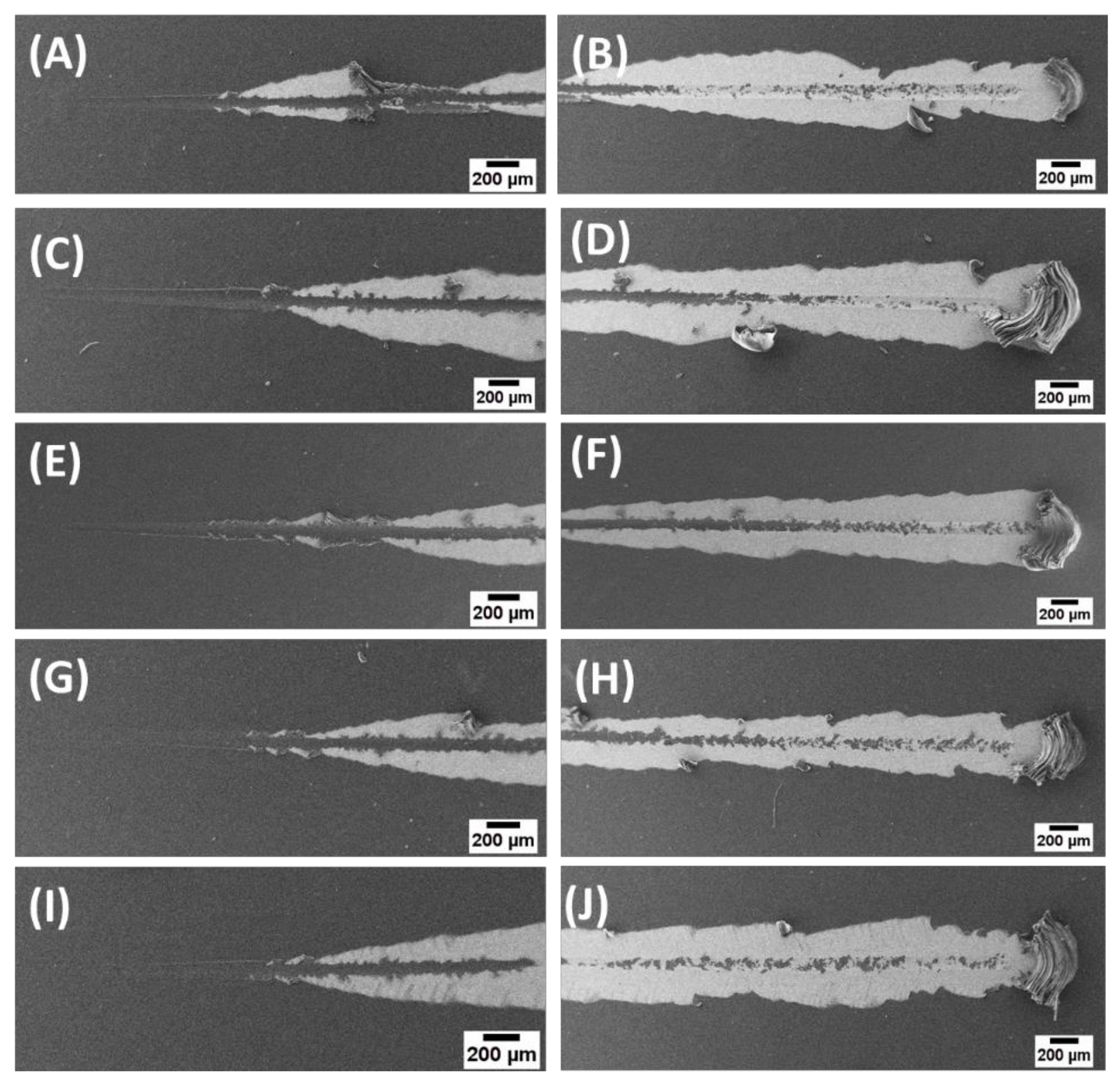
2.10. Mechanical Characterization
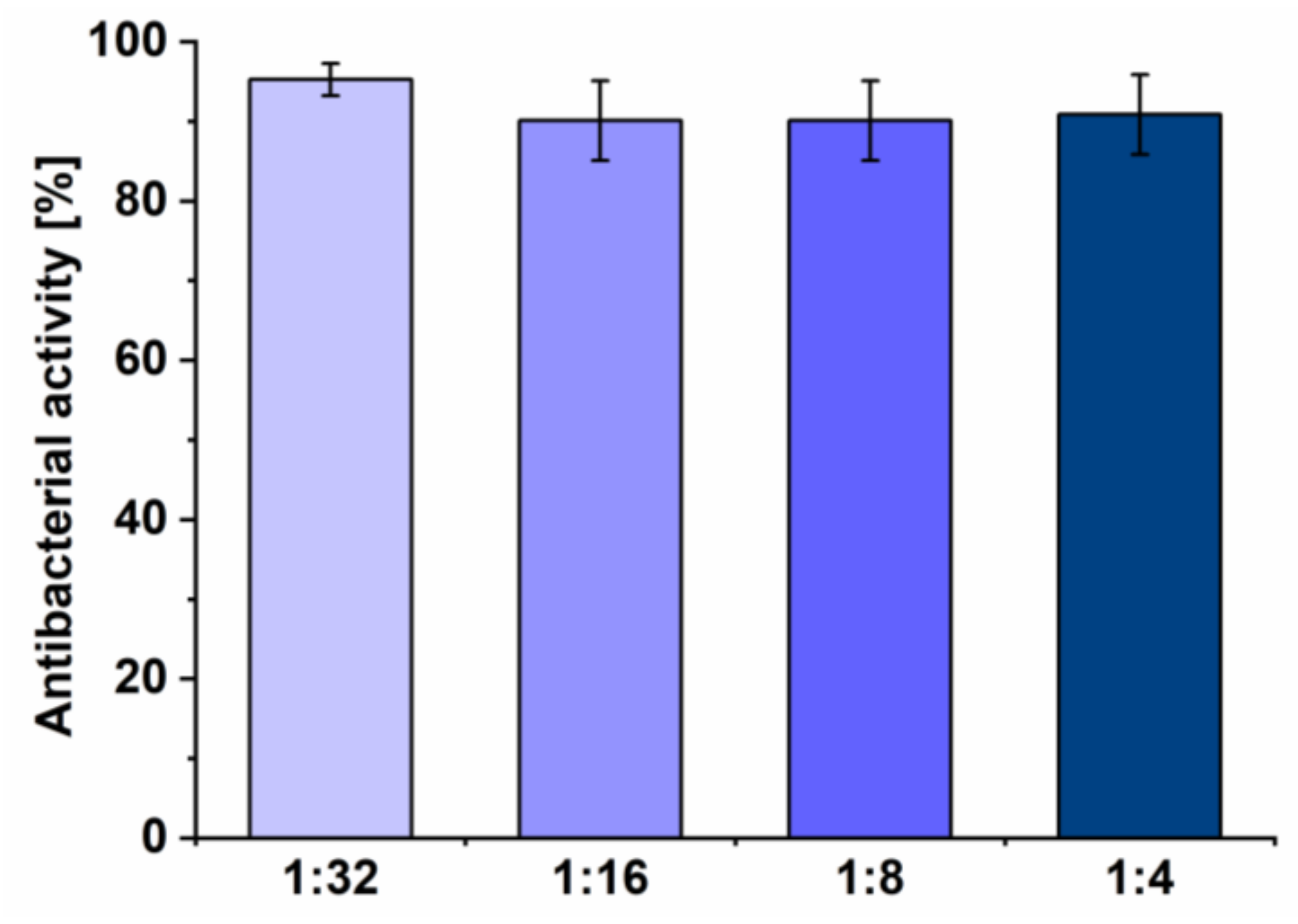
2.11. Antibacterial Assay
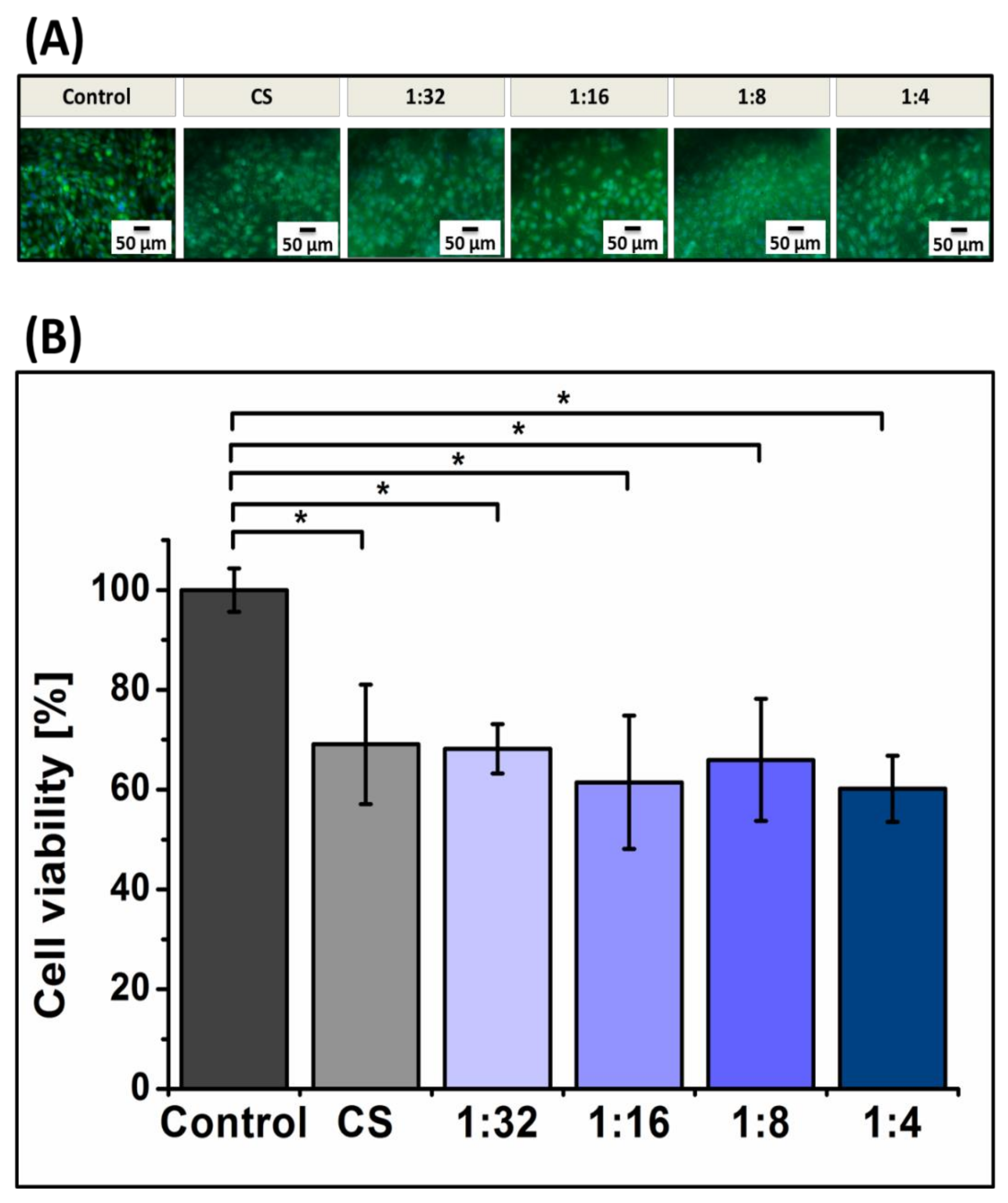
2.12. In Vitro Cellular Test
2.12.1. Cell Culture and Seeding
2.12.2. Cell Viability
2.12.3. Cell Morphology and Attachment
2.13. Statistical Analysis
3. Results and Discussion
3.1. Influence of Ga (III) Concentration on Suspension Properties
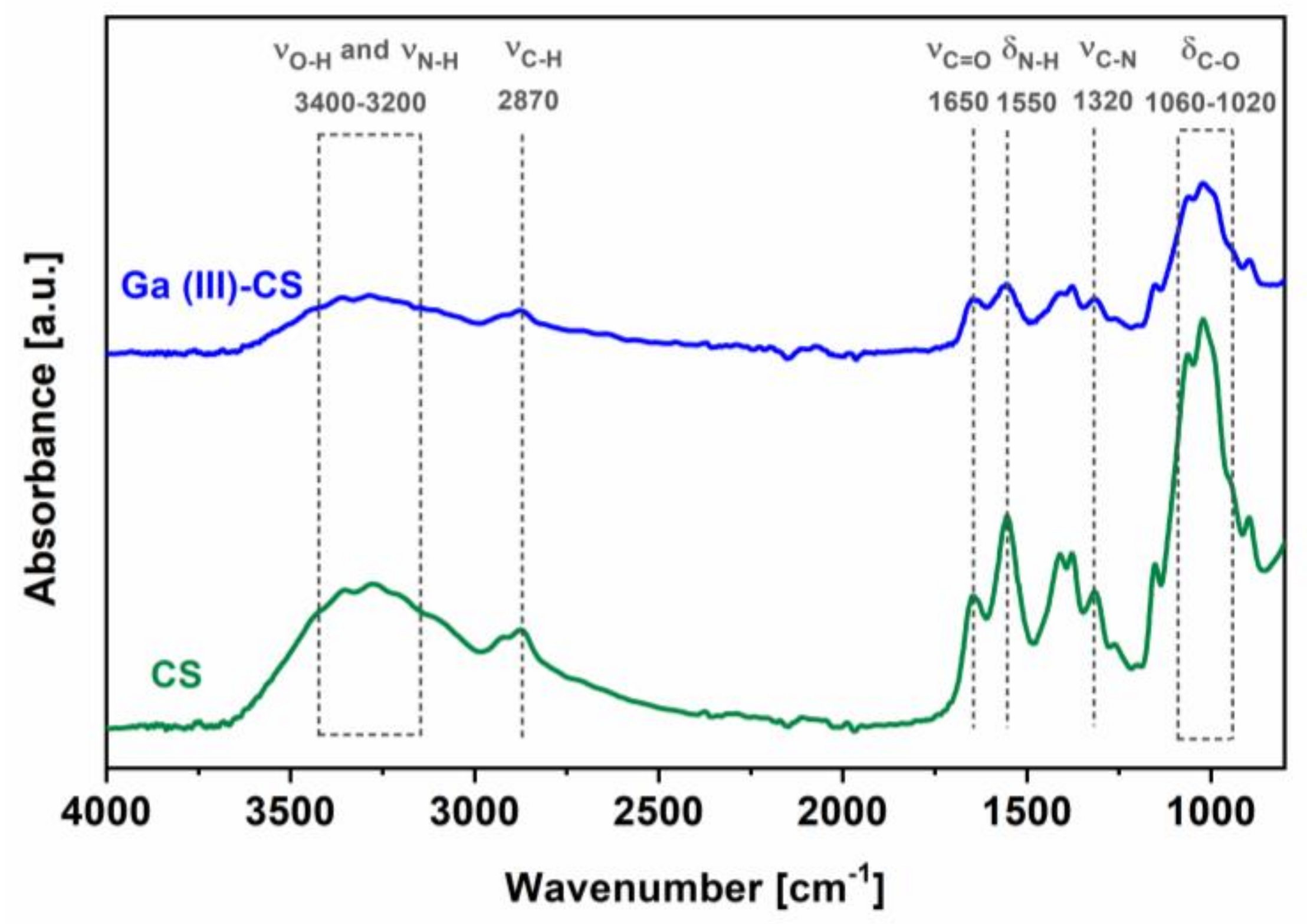
3.2. Morphological and Compositional Characterization
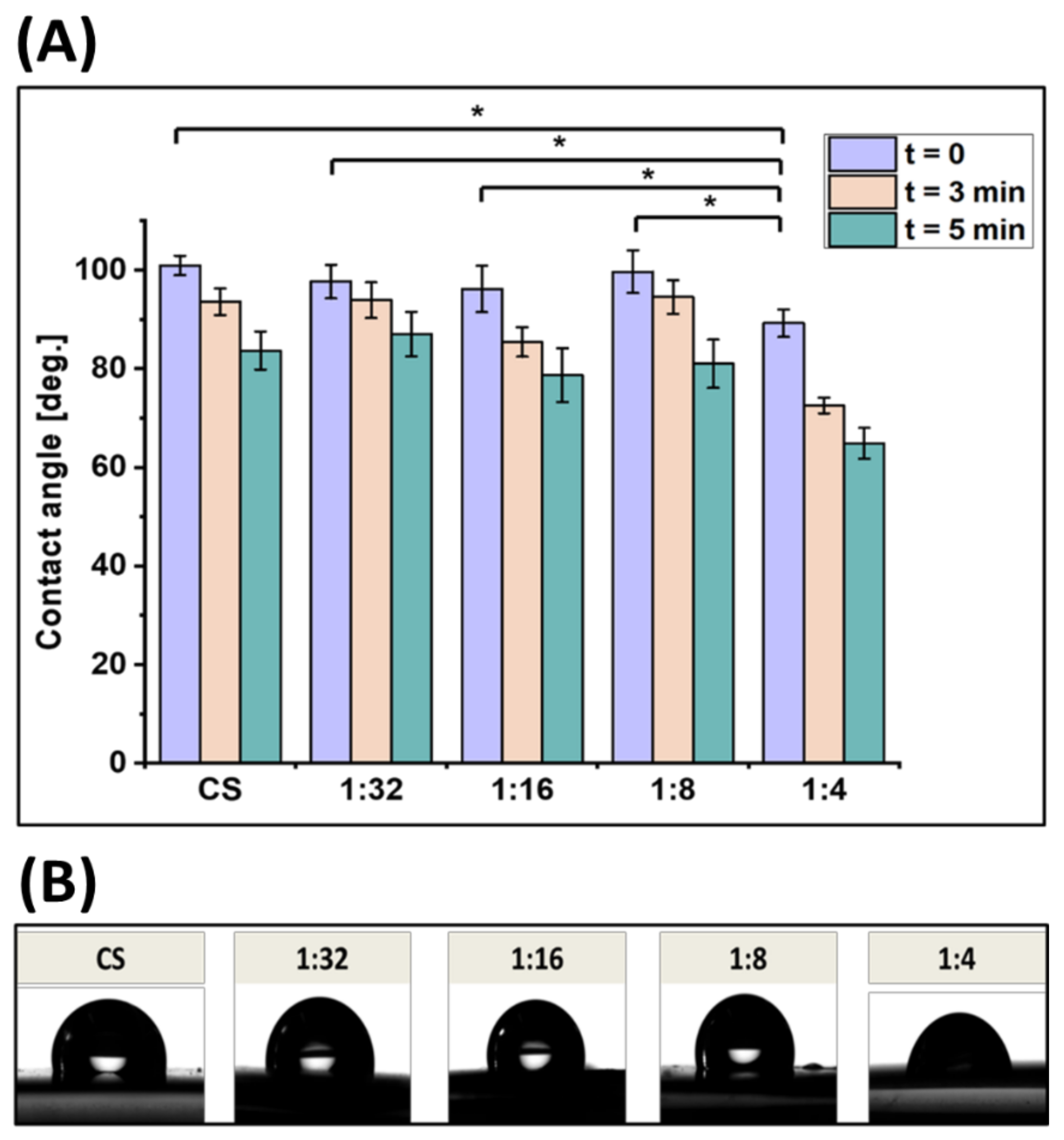
3.3. Wettability
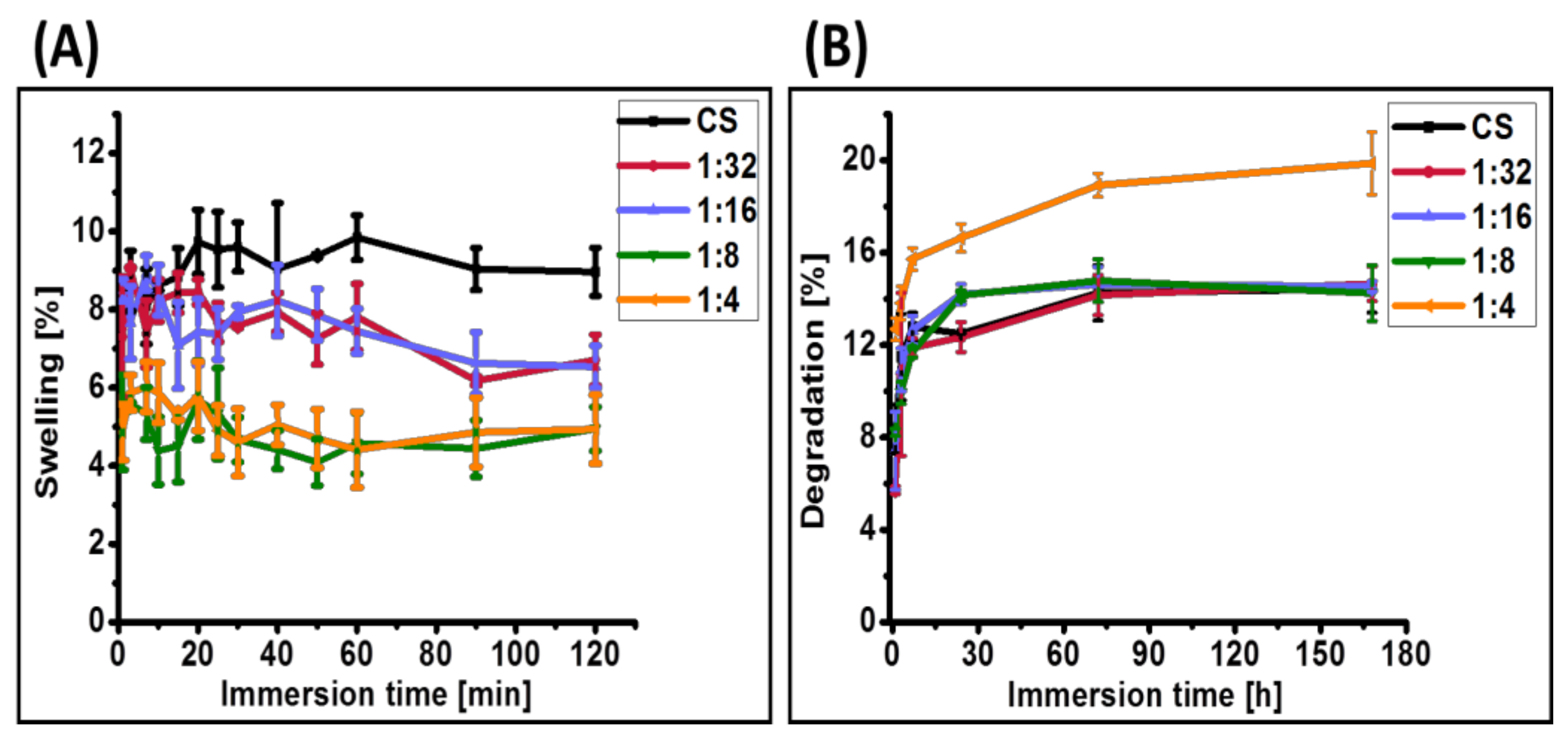
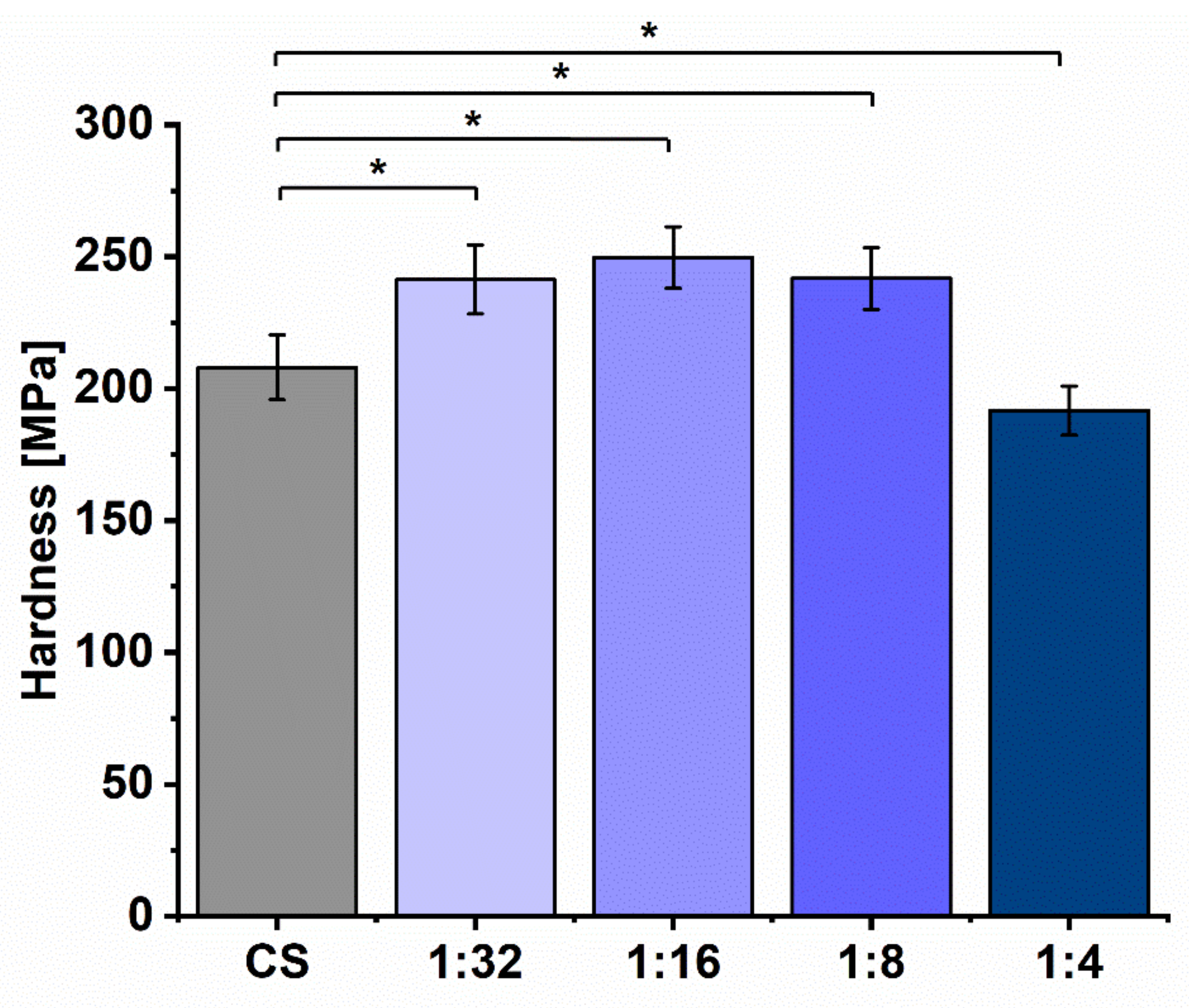
3.4. Physical and Mechanical Properties
3.5. Antibacterial Assay
3.6. Biological Characterization
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Miola, M.; Vernè, E.; Ciraldo, F.E.; Cordero-Arias, L.; Boccaccini, A.R. Electrophoretic Deposition of Chitosan/45S5 Bioactive Glass Composite Coatings Doped with Zn and Sr. Front. Bioeng. Biotechnol. 2015, 3, 1211. [Google Scholar] [CrossRef] [PubMed]
- Pishbin, F.; Simchi, A.A.; Ryan, M.P.; Boccaccini, A. A study of the electrophoretic deposition of Bioglass® suspensions using the Taguchi experimental design approach. J. Eur. Ceram. Soc. 2010, 30, 2963–2970. [Google Scholar] [CrossRef]
- Prasad, K.; Bazaka, O.; Chua, M.; Rochford, M.; Fedrick, L.; Spoor, J.; Symes, R.; Tieppo, M.; Collins, C.; Cao, A.; et al. Metallic Biomaterials: Current Challenges and Opportunities. Material 2017, 10, 884. [Google Scholar] [CrossRef] [PubMed]
- Zimmerli, W. Clinical presentation and treatment of orthopaedic implant-associated infection. J. Intern. Med. 2014, 276, 111–119. [Google Scholar] [CrossRef]
- Khosravi, A.D.; Ahmadi, F.; Salmanzadeh, S.; Dashtbozor, A.; Abbasi-Montazeri, E. Study of Bacteria Isolated from Orthopedic Implant Infections and their Antimicrobial Susceptibility Pattern. Res. J. Microbiol. 2009, 4, 158–163. [Google Scholar] [CrossRef]
- Li, B.; Webster, T.J. Bacteria antibiotic resistance: New challenges and opportunities for implant-associated orthopedic infections. J. Orthop. Res. 2017, 36, 22–32. [Google Scholar] [CrossRef]
- De Sanctis, J.; Teixeira, L.; Van Duin, D.; Odio, C.; Hall, G.; Tomford, J.W.; Perez, F.; Rudin, S.D.; Bonomo, R.A.; Barsoum, W.K.; et al. Complex prosthetic joint infections due to carbapenemase-producing Klebsiella pneumoniae: A unique challenge in the era of untreatable infections. Int. J. Infect. Dis. 2014, 25, 73–78. [Google Scholar] [CrossRef]
- Aboltins, C.; Dowsey, M.M.; Buising, K.; Peel, T.N.; Daffy, J.; Choong, P.F.M.; Stanley, P. Gram-negative prosthetic joint infection treated with debridement, prosthesis retention and antibiotic regimens including a fluoroquinolone. Clin. Microbiol. Infect. 2011, 17, 862–867. [Google Scholar] [CrossRef]
- Hsieh, P.-H.; Lee, M.S.; Hsu, K.; Chang, Y.-H.; Shih, H.; Ueng, S.W. Gram?Negative Prosthetic Joint Infections: Risk Factors and Outcome of Treatment. Clin. Infect. Dis. 2009, 49, 1036–1043. [Google Scholar] [CrossRef]
- Croisier, F.; Jérôme, C. Chitosan-based biomaterials for tissue engineering. Eur. Polym. J. 2013, 49, 780–792. [Google Scholar] [CrossRef]
- Cheung, R.C.F.; Ng, T.B.; Wong, J.H.; Chan, W.-Y. Chitosan: An Update on Potential Biomedical and Pharmaceutical Applications. Mar. Drugs 2015, 13, 5156–5186. [Google Scholar] [CrossRef] [PubMed]
- Gritsch, L.; Lovell, C.; Goldmann, W.H.; Boccaccini, A. Fabrication and characterization of copper(II)-chitosan complexes as antibiotic-free antibacterial biomaterial. Carbohydr. Polym. 2018, 179, 370–378. [Google Scholar] [CrossRef] [PubMed]
- Hussein, M.H.M.; El-Hady, M.F.; Sayed, W.; Hefni, H. Preparation of some chitosan heavy metal complexes and study of its properties. Polym. Sci. Ser. A 2012, 54, 113–124. [Google Scholar] [CrossRef]
- Wang, X.; Du, Y.; Fan, L.; Liu, H.; Hu, Y. Chitosan- metal complexes as antimicrobial agent: Synthesis, characterization and Structure-activity study. Polym. Bull. 2005, 55, 105–113. [Google Scholar] [CrossRef]
- Li, P.; Zhang, X.; Xu, R.; Wang, W.; Liu, X.; Yeung, K.W.K.; Chu, P.K. Electrochemically deposited chitosan/Ag complex coatings on biomedical NiTi alloy for antibacterial application. Surf. Coat. Technol. 2013, 232, 370–375. [Google Scholar] [CrossRef]
- Gritsch, L.; Maqbool, M.; Mouriño, V.; Ciraldo, F.E.; Cresswell, M.; Jackson, P.R.; Lovell, C.; Boccaccini, A.R. Chitosan/hydroxyapatite composite bone tissue engineering scaffolds with dual and decoupled therapeutic ion delivery: Copper and strontium. J. Mater. Chem. B 2019, 7, 6109–6124. [Google Scholar] [CrossRef]
- Hijazi, S.; Visaggio, D.; Pirolo, M.; Frangipani, E.; Bernstein, L.; Visca, P. Antimicrobial Activity of Gallium Compounds on ESKAPE Pathogens. Front. Microbiol. 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Xu, Z.; Zhao, X.; Chen, X.; Chen, Z.; Xia, Z. Antimicrobial effect of gallium nitrate against bacteria encountered in burn wound infections. RSC Adv. 2017, 7, 52266–52273. [Google Scholar] [CrossRef]
- Guidon, P.T.; Salvatori, R.; Bockman, R.S. Gallium nitrate regulates rat osteoblast expression of osteocalcin protein and mRNA levels. J. Bone Miner. Res. 2009, 8, 103–112. [Google Scholar] [CrossRef]
- Bockman, R.S.; Guidon, P.T.; Pan, L.C.; Salvatori, R.; Kawaguchi, A. Gallium nitrate increases type I collagen and fibronectin mRNA and collagen protein levels in bone and fibroblast cells. J. Cell. Biochem. 1993, 52, 396–403. [Google Scholar] [CrossRef]
- Chitambar, C.R. Medical Applications and Toxicities of Gallium Compounds. Int. J. Environ. Res. Public Health 2010, 7, 2337–2361. [Google Scholar] [CrossRef] [PubMed]
- Chitambar, C.R. Gallium nitrate for the treatment of non-Hodgkin’s lymphoma. Expert Opin. Investig. Drugs 2004, 13, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Chitambar, C.R. Gallium-containing anticancer compounds. Futur. Med. Chem. 2012, 4, 1257–1272. [Google Scholar] [CrossRef] [PubMed]
- Mouriño, V.; Newby, P.; Boccaccini, A. Preparation and Characterization of Gallium Releasing 3-D Alginate Coated 45S5 Bioglass® Based Scaffolds for Bone Tissue Engineering. Adv. Eng. Mater. 2010, 12, B283–B291. [Google Scholar] [CrossRef]
- Esfahani, A.G.; Lazazzera, B.; Draghi, L.; Farè, S.; Chiesa, R.; De Nardo, L.; Billi, F. Bactericidal activity of gallium-doped chitosan coatings against staphylococcal infection. J. Appl. Microbiol. 2018, 126, 87–101. [Google Scholar] [CrossRef]
- Bernstein, L.R. Mechanisms of therapeutic activity for gallium. Pharmacol. Rev. 1998, 50, 665–682. [Google Scholar]
- Praveen, A.S.; Arjunan, A. Effect of nano-Al2O3 addition on the microstructure and erosion wear of HVOF sprayed NiCrSiB coatings. Mater. Res. Express 2019, 7, 015006. [Google Scholar] [CrossRef]
- Nouri, A.; Wen, C. Introduction to surface coating and modification for metallic biomaterials. Surf. Coat. Modif. Met. Biomater. 2015. [Google Scholar] [CrossRef]
- Praveen, A.S.; Arjunan, A. Parametric optimisation of high-velocity oxy-fuel nickel-chromium-silicon-boron and aluminium-oxide coating to improve erosion wear resistance. Mater. Res. Express 2019, 6, 096560. [Google Scholar] [CrossRef]
- Corni, I.; Ryan, M.P.; Boccaccini, A. Electrophoretic deposition: From traditional ceramics to nanotechnology. J. Eur. Ceram. Soc. 2008, 28, 1353–1367. [Google Scholar] [CrossRef]
- Boccaccini, A.; Keim, S.; Ma, R.; Li, Y.; Zhitomirsky, I. Electrophoretic deposition of biomaterials. J. R. Soc. Interface 2010, 7, S581–S613. [Google Scholar] [CrossRef] [PubMed]
- Avcu, E.; Bastan, F.E.; Abdullah, H.Z.; Rehman, M.A.U.; Avcu, Y.Y.; Boccaccini, A.R. Electrophoretic deposition of chitosan-based composite coatings for biomedical applications: A review. Prog. Mater. Sci. 2019, 103, 69–108. [Google Scholar] [CrossRef]
- Cai, X.; Cai, J.; Ma, K.; Huang, P.; Gong, L.; Huang, D.; Jiang, T.; Wang, Y. Fabrication and characterization of Mg-doped chitosan–gelatin nanocompound coatings for titanium surface functionalization. J. Biomater. Sci. Polym. Ed. 2016, 27, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.; Huang, D.; Cai, J.; Cai, X.; Gong, L.; Huang, P.; Wang, Y.; Jiang, T. Surface functionalization with strontium-containing nanocomposite coatings via EPD. Colloids Surf. B Biointerfaces 2016, 146, 97–106. [Google Scholar] [CrossRef]
- Ma, K.; Gong, L.; Cai, X.; Huang, P.; Cai, J.; Huang, D.; Jiang, T. A green single-step procedure to synthesize Ag-containing nanocomposite coatings with low cytotoxicity and efficient antibacterial properties. Int. J. Nanomed. 2017, 12, 3665–3679. [Google Scholar] [CrossRef]
- Huang, D.; Ma, K.; Cai, X.; Yang, X.; Hu, Y.; Huang, P.; Wang, F.; Jiang, T.; Wang, Y. Evaluation of antibacterial, angiogenic, and osteogenic activities of green synthesized gap-bridging copper-doped nanocomposite coatings. Int. J. Nanomed. 2017, 12, 7483–7500. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, B.; Gray, K.M.; Cheng, Y.; Kim, E.; Rubloff, G.; Bentley, W.; Wang, Q.; Payne, G.F. Electrodeposition of a weak polyelectrolyte hydrogel: Remarkable effects of salt on kinetics, structure and properties. Soft Matter 2013, 9, 2703. [Google Scholar] [CrossRef]
- Akhtar, M.; Ilyas, K.; Dlouhý, I.; Siska, F.; Boccaccini, A.R. Electrophoretic Deposition of Copper(II)–Chitosan Complexes for Antibacterial Coatings. Int. J. Mol. Sci. 2020, 21, 2637. [Google Scholar] [CrossRef]
- Ballarre, J.; Aydemir, T.; Liverani, L.; Roether, J.; Goldmann, W.; Boccaccini, A.R. Versatile bioactive and antibacterial coating system based on silica, gentamicin, and chitosan: Improving early stage performance of titanium implants. Surf. Coat. Technol. 2020, 381, 125138. [Google Scholar] [CrossRef]
- Stevanović, M.; Đošić, M.; Jankovic, A.; Kojić, V.; Vukašinović-Sekulić, M.; Stojanović, J.; Odović, J.; Sakač, M.C.; Rhee, K.Y.; Miskovic-Stankovic, V. Gentamicin-Loaded Bioactive Hydroxyapatite/Chitosan Composite Coating Electrodeposited on Titanium. ACS Biomater. Sci. Eng. 2018, 4, 3994–4007. [Google Scholar] [CrossRef]
- Ma, K.; Cai, X.; Zhou, Y.; Zhang, Z.; Jiang, T.; Wang, Y. Osteogenetic property of a biodegradable three-dimensional macroporous hydrogel coating on titanium implants fabricated via EPD. Biomed. Mater. 2014, 9, 15008. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Ouyang, L.; Luo, Y.; Sun, Z.; Yang, C.; Wang, J.; Liu, X.; Zhang, X. Multifunctional sulfonated polyetheretherketone coating with beta-defensin-14 for yielding durable and broad-spectrum antibacterial activity and osseointegration. Acta Biomater. 2019, 86, 323–337. [Google Scholar] [CrossRef] [PubMed]
- National Food Safety Standard Food Microbiological Examination: Aerobic Plate Count. GB4789.2. 2010. Available online: http://www.puntofocal.gov.ar/doc/nchn199.pdf (accessed on 15 August 2019).
- Keremidarska-Markova, M.; Radeva, E.; Eleršič, K.; Iglič, A.; Pramatarova, L.; Krasteva, N. Plasma deposited composite coatings to control biological response of osteoblast-like MG-63 cells. J. Phys. Conf. Ser. 2014, 558, 012057. [Google Scholar] [CrossRef]
- Simchi, A.A.; Pishbin, F.; Boccaccini, A.R. Electrophoretic deposition of chitosan. Mater. Lett. 2009, 63, 2253–2256. [Google Scholar] [CrossRef]
- Aznam, I.; Mah, J.C.; Muchtar, A.; Somalu, M.R.; Ghazali, M.J. A review of key parameters for effective electrophoretic deposition in the fabrication of solid oxide fuel cells. J. Zhejiang Univ. A 2018, 19, 811–823. [Google Scholar] [CrossRef]
- Unagolla, J.; Adikary, S. Adsorption characteristics of cadmium and lead heavy metals into locally synthesized Chitosan Biopolymer. Trop. Agric. Res. 2015, 26, 395. [Google Scholar] [CrossRef]
- Lapo, B.; Demey, H.; Zapata, J.; Romero, C.; Sastre, A. Sorption of Hg(II) and Pb(II) Ions on Chitosan-Iron(III) from Aqueous Solutions: Single and Binary Systems. Polymers 2018, 10, 367. [Google Scholar] [CrossRef]
- Gerente, C.; Lee, V.K.C.; Le Cloirec, P.; McKay, G. Application of Chitosan for the Removal of Metals From Wastewaters by Adsorption—Mechanisms and Models Review. Crit. Rev. Environ. Sci. Technol. 2007, 37, 41–127. [Google Scholar] [CrossRef]
- Qu, J.; Hu, Q.; Shen, K.; Zhang, K.; Li, Y.; Li, H.; Zhang, Q.; Wang, J.; Quan, W. The preparation and characterization of chitosan rods modified with Fe3+ by a chelation mechanism. Carbohydr. Res. 2011, 346, 822–827. [Google Scholar] [CrossRef]
- Besra, L.; Liu, M. A review on fundamentals and applications of electrophoretic deposition (EPD). Prog. Mater. Sci. 2007, 52, 1–61. [Google Scholar] [CrossRef]
- Cheng, Y.; Luo, X.; Betz, J.; Buckhout-White, S.; Bekdash, O.; Payne, G.F.; Bentley, W.E.; Rubloff, G. In situ quantitative visualization and characterization of chitosan electrodeposition with paired sidewall electrodes. Soft Matter 2010, 6, 3177. [Google Scholar] [CrossRef]
- Park, J.B.; Bronzino, J.D. Biomaterials: Principles and Applications; CRC Press: Boca Raton, FL, USA, 2002; pp. 1–17. [Google Scholar]
- Algarhy, M.; Hasaballah, S.; Rashed, U.; El-Sabbagh, M.; Soliman, H.; Saudy, A. Surface Processing of Stainless Steel Using the Channel Spark Device. Nat. Sci. 2015, 13, 127–131. [Google Scholar]
- Rogina, A.; Lončarević, A.; Antunovic, M.; Marijanovic, I.; Ivanković, M.; Ivanković, H. Tuning physicochemical and biological properties of chitosan through complexation with transition metal ions. Int. J. Boil. Macromol. 2019, 129, 645–652. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Shen, Y.; Shen, C.; Wen, Y.; Liu, W. Al-doping chitosan–Fe(III) hydrogel for the removal of fluoride from aqueous solutions. Chem. Eng. J. 2014, 248, 98–106. [Google Scholar] [CrossRef]
- Antonino, R.; Fook, B.R.P.L.; Lima, V.A.D.O.; Rached, R.; Ícaro, D.F.; Lima, E.P.N.; Lima, R.; Peniche-Covas, C.; Fook, M.V.L. Preparation and Characterization of Chitosan Obtained from Shells of Shrimp (Litopenaeus vannamei Boone). Mar. Drugs 2017, 15, 141. [Google Scholar] [CrossRef]
- Chen, G.; Zhang, S.; Xia, L.; Ding, C.; Wen, L.; Wan, W. Biocompatible nanocarriers that respond to oxidative environments via interactions between chitosan and multiple metal ions. Int. J. Nanomed. 2016, 11, 2769–2784. [Google Scholar] [CrossRef]
- Modrzejewska, Z.; Dorabialska, M. The Mechanism of Sorption of Ag+ Ions on Chitosan Microgranules: IR and NMR Studies. Prog. Chem. Chitin Deriv. 2009, 14, 49–64. [Google Scholar]
- Brunel, F.; El Gueddari, N.E.; Moerschbacher, B.M. Complexation of copper(II) with chitosan nanogels: Toward control of microbial growth. Carbohydr. Polym. 2013, 92, 1348–1356. [Google Scholar] [CrossRef]
- Thevenot, P.; Wenjing, H.; Tang, L. Surface Chemistry Implant Biocompatibility. Curr. Top. Med. Chem. 2008, 8, 270–280. [Google Scholar]
- Sierra, H.; Cordova, M.; Chen, C.-S.J.; Rajadhyaksha, M. Confocal imaging-guided laser ablation of basal cell carcinomas: An ex vivo study. J. Investig. Dermatol. 2014, 135, 612–615. [Google Scholar] [CrossRef]
- Menzies, K.L.; Jones, L. The Impact of Contact Angle on the Biocompatibility of Biomaterials. Optom. Vis. Sci. 2010, 87, 1. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, S.N.; Gonçalves, I.C.; Martins, M.C.L.; Barbosa, M.; Ratner, B.D. Fibrinogen adsorption, platelet adhesion and activation on mixed hydroxyl-/methyl-terminated self-assembled monolayers. Biomaterials 2006, 27, 5357–5367. [Google Scholar] [CrossRef] [PubMed]
- Sobahi, T.R.; Abdelaal, M.; Makki, M.S. Chemical modification of Chitosan for metal ion removal. Arab. J. Chem. 2014, 7, 741–746. [Google Scholar] [CrossRef]
- Yin, X. Metal-coordinating controlled oxidative degradation of chitosan and antioxidant activity of chitosan-metal complex. Arkivoc 2004, 2004, 66. [Google Scholar] [CrossRef]
- Lončarević, A.; Ivanković, M.; Rogina, A. Lysozyme-Induced Degradation of Chitosan: The Characterisation of Degraded Chitosan Scaffolds. J. Tissue Repair Regen. 2017, 1, 12–22. [Google Scholar] [CrossRef]
- Freier, T.; Koh, H.S.; Kazazian, K.; Shoichet, M.S. Controlling cell adhesion and degradation of chitosan films by N-acetylation. Biomaterial 2005, 26, 5872–5878. [Google Scholar] [CrossRef]
- Fahim, I.S.; Aboulkhair, N.; Everitt, N.M. Nanoindentation Investigation on Chitosan Thin Films with Different Types of Nano Fillers. J. Mater. Sci. Res. 2018, 7, 11. [Google Scholar] [CrossRef]
- Hernández, R.B.; Franco, A.P.; Yola, O.R.; López-Delgado, A.; Felcman, J.; Recio, M.A.L.; Mercê, A.L.R. Coordination study of chitosan and Fe3+. J. Mol. Struct. 2008, 877, 89–99. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Gómez-Fatou, M.A.; Ania, F.; Flores, A. Nanoindentation in polymer nanocomposites. Prog. Mater. Sci. 2015, 67, 1–94. [Google Scholar] [CrossRef]
- Jugowiec, D.; Kot, M.; Moskalewicz, T. Electrophoretic Deposition and Characterisation of Chitosan Coatings on Near-Β Titanium Alloy. Arch. Met. Mater. 2016, 61, 657–664. [Google Scholar] [CrossRef]
- Li, J.; Beres, W. Scratch Test for Coating/Substrate Systems—A Literature Review. Can. Met. Q. 2007, 46, 155–173. [Google Scholar] [CrossRef]
- Gugala, N.; Chatfield-Reed, K.; Turner, R.J.; Chua, G. Using a Chemical Genetic Screen to Enhance Our Understanding of the Antimicrobial Properties of Gallium against Escherichia coli. Genes 2019, 10, 34. [Google Scholar] [CrossRef] [PubMed]
- Shirosaki, Y.; Tsuru, K.; Hayakawa, S.; Osaka, A.; Lopes, M.A.; Santos, J.D.; Fernandes, M.H. In vitro cytocompatibility of MG63 cells on chitosan-organosiloxane hybrid membranes. Biomaterial 2005, 26, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Cimmino, C.; Rossano, L.; Netti, P.A.; Ventre, M. Spatio-Temporal Control of Cell Adhesion: Toward Programmable Platforms to Manipulate Cell Functions and Fate. Front. Bioeng. Biotechnol. 2018, 6. [Google Scholar] [CrossRef]
- Ducheyne, P.; Healy, K.; Hutmacher, D.W.; Grainger, D.W.; Kirkpatrick, C.J. Comprehensive Biomaterials, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 222–223. [Google Scholar]
- Campos, M.G.N.; Innocentini-Mei, L.H.; Junior, A.R.S. Sorbitol-Plasticized and Neutralized Chitosan Membranes as Skin Substitutes. Mater. Res. 2015, 18, 781–790. [Google Scholar] [CrossRef]
- Zhang, Z.; Cheng, X.; Yao, Y.; Luo, J.; Tang, Q.; Wu, H.; Lin, S.; Han, C.; Wei, Q.; Chen, L. Electrophoretic deposition of chitosan/gelatin coatings with controlled porous surface topography to enhance initial osteoblast adhesive responses. J. Mater. Chem. B 2016, 4, 7584–7595. [Google Scholar] [CrossRef]
- Cai, S.-J.; Li, C.-W.; Weihs, D.; Wang, G.-J. Control of cell proliferation by a porous chitosan scaffold with multiple releasing capabilities. Sci. Technol. Adv. Mater. 2017, 18, 987–996. [Google Scholar] [CrossRef]
- Schmalz, G.; Arenholt-Bindslev, D. Biocompatibility of Dental Materials; Springer: Berlin/Heidelberg, Germany, 2009; pp. 76–80. [Google Scholar] [CrossRef]

| CS | 1:32 | 1:16 | 1:8 | 1:4 | |
|---|---|---|---|---|---|
| Zeta-Potential [mV] | 32 ± 6 | 29 ± 7 | 27 ± 7 | 31 ± 7 | 28 ± 6 |
| Sample | 1:32 | 1:16 | 1:8 | 1:4 |
|---|---|---|---|---|
| GaL/Ck𝛼 ratio of Ga (III)-CS (%) | 3.1 ± 0.2 | 7.1 ± 0.3 | 10.5 ± 0.4 | 32.1 ± 0.9 |
| GaL/Ck𝛼 ratio of Ga (III)-CS coating (%) | 2.4 ± 0.1 | 5.1 ± 0.1 | 8.0 ± 2.9 | 24.2 ± 0.1 |
| Sample | CS | 1:32 | 1:16 | 1:8 | 1:4 |
|---|---|---|---|---|---|
| Critical Load (N) | 2.2 ± 0.1 | 3.1 ± 0.1 | 3.1 ± 0.2 | 2.7 ± 0.1 | 2.6 ± 0.1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akhtar, M.A.; Hadzhieva, Z.; Dlouhý, I.; Boccaccini, A.R. Electrophoretic Deposition and Characterization of Functional Coatings Based on an Antibacterial Gallium (III)-Chitosan Complex. Coatings 2020, 10, 483. https://doi.org/10.3390/coatings10050483
Akhtar MA, Hadzhieva Z, Dlouhý I, Boccaccini AR. Electrophoretic Deposition and Characterization of Functional Coatings Based on an Antibacterial Gallium (III)-Chitosan Complex. Coatings. 2020; 10(5):483. https://doi.org/10.3390/coatings10050483
Chicago/Turabian StyleAkhtar, Muhammad Asim, Zoya Hadzhieva, Ivo Dlouhý, and Aldo R. Boccaccini. 2020. "Electrophoretic Deposition and Characterization of Functional Coatings Based on an Antibacterial Gallium (III)-Chitosan Complex" Coatings 10, no. 5: 483. https://doi.org/10.3390/coatings10050483
APA StyleAkhtar, M. A., Hadzhieva, Z., Dlouhý, I., & Boccaccini, A. R. (2020). Electrophoretic Deposition and Characterization of Functional Coatings Based on an Antibacterial Gallium (III)-Chitosan Complex. Coatings, 10(5), 483. https://doi.org/10.3390/coatings10050483






