Abstract
Rosmarinus officinalis and Myrtus communis essential oils (EOs) are well-known for their ethno-pharmaceutical properties. In the present study, we have analyzed the chemical composition of both EOs by gas chromatography-mass spectrometry. Then we assessed their antibacterial, antibiofilm, and anti-virulence actions against the opportunistic pathogen Staphylococcus aureus. The cytotoxic effect of agents tested against this bacterium was investigated by monitoring reactive oxygen-species (ROS) generation and antioxidant-enzyme (catalase) production. Regarding the antistaphylococcal effects, our results showed antibacterial efficacy of both Eos and their combination, where the minimum inhibitory concentrations ranged between 0.7 and 11.25 mg/mL. A combination of tested agents showed the highest anti-hemolytic and anti-protease effects. Additionally, association between EOs displayed more potency against the development of biofilm performed by S. aureus, with percentage of removal reaching 74%. The inhibitory impacts of EOs on S. aureus virulence factors were discovered to be concentration-dependent. Furthermore, our results provide insight on the abilities of R. officinalis and M. communis EOs, as well as their potential in combination, to generate ROS and affect oxidative stress enzyme catalase in S. aureus, leading to their antagonistic effect against this pathogen.
1. Introduction
Staphylococcus aureus is one of the major pathogens credited as being responsible for nosocomial and community acquired infections in humans [1,2]. Colonizing the normal commensal flora of humans and many animals, this opportunistic bacterium is considered the main cause of hospital-acquired infections, generating a variety of symptoms, from mild localized infection to potentially fatal sepsis [3]. S. aureus is characterized by a variety of virulence factors, such as secretion of various enzymes and toxins [4], allowing it to avoid the host’s immune defense system and participate in tissue invasion and destruction, along with its ability to form biofilms on medical devices and biomaterials [5].
S. aureus is one of the biofilm-producing bacteria with high ability to survive in hostile environments and medical or industrial systems [6]. Inside this biological matrix, bacteria became resistant to different classes of antibiotics and ether-disinfectants, making biofilm-related infections more prone to relapse [7,8]. Across years, synthetic antibiotics are used to overcome S. aureus antimicrobial resistance. However, such agents showed high toxicity as well as several side effects in patients [9]. This is one of the factors making antibacterial resistance an increasingly serious threat to global public health. In clinical practice, the ineffectiveness of single-component medications in treating microbiological infections caused by resistant bacteria can be avoided using a combination of two or more antibiotics, which increases their potency and aids in treatment [10,11].
Antibacterial drugs’ principal modes of action include inhibition of cell-wall formation and interference with the ribosome, which cause inhibition of protein synthesis. Antibiotics also alter cell membrane activities, interfere with nucleic acid production (inhibition of DNA gyrase and RNA polymerase), and impede metabolic pathways [12]. Changes in the bacterial genome are responsible for bacterial antibiotic resistance [13]: primarily genetic mutations that modify genomic DNA, result in new resistant strains, as well as transfer of genetic material such as plasmids or mobile genetic components containing antibacterial-resistance genes.
The search for new anti-infection agents from natural resources, with different modes of action and competitive effects, has become a necessity [14]. Numerous lines of research are developing new classes of molecules aimed at new “targets” of action in bacteria, in order to circumvent bacterial resistance mechanisms [15]. For instance, Quorum sensing (QS) has been identified as one of the alternate approaches for combating multidrug-resistant pathogens [16]. Through the QS process, several pathogenic bacteria monitor their own population density and control their virulence factors, antibiotic production, biofilm formation, mobility, and swarming [17].
Essential oils (EOs) from medicinal and aromatic plants are of major interest due to their rich bioactive compounds and potent antimicrobial activity [18]. For instance, Rosmarinus officinalis is well known for is medicinal properties against respiratory diseases, headaches, illnesses, and neuropsychiatric disorders [19]. It was largely used for culinary purposes [20] and food preservation [21]. Moreover, Myrtus communis is a wild aromatic plant with a wide range of biological properties such as antioxidant, anti-inflammatory, and antimicrobial [22].
Given the large number of chemical compounds present in EOs, there are several targets in the bacterial cell exposed to these molecules [23,24]. Closely related to their chemical composition, numerous mechanisms of action of EOs were documented, including: alteration of the cell wall, degradation of the cytoplasmic membrane and alteration of membrane proteins, leakage of cell contents, coagulation of the cytoplasm, depletion of the proton motility force, and induction of oxidative stress in the bacterial cell [25,26]. It was postulated that EOs induce oxidative stress in treated cells [19]. EOs inhibit bacterial growth by enhancing the production of reactive oxygen species (ROS) in the cells, which consequently inhibit certain essential biological processes [20]. In this context, the present study was undertaken to evaluate the antibacterial properties and mode of action of R. officinalis and M. communis EOs against the opportunistic pathogen S. aureus.
2. Results
2.1. Chemical Characterization of EOs
The results of chemical analysis of R. officinalis and M. communis EOs by GC/MS and GC/FID are presented in Table 1.

Table 1.
Chemical composition (%) of R. officinalis and M. communis essential oils.
A total of 56 compounds are identified, representing 96.39% and 96.57% of tested EOs. The major constituent of R. officinalis EO was found to be 1,8-cineole with a prevalence of 37.56%. This molecule belongs to the class of monoterpene oxides. The major compound of M. communis EO was the α-Pinene, with a percentage of 45.3%. This compound belongs to the class of monoterpene hydrocarbons.
2.2. Antibacterial Activity
The antibacterial effects of both EOs and their combination are reported as ‘in vitro’ activity as MIC and MBC and summarized in Table 2.

Table 2.
Antibacterial activity of R. officinalis (EO1) and M. communis (EO2) essential oils and their combination (EO1 + EO2) against S. aureus ATCC 25923.
Both tested agents exerted a bacteriostatic effect against the S. aureus strain, with MICs values ranging between 0.7 and 11.25 mg/mL, for R. officinalis and M. communis respectively (p < 0.05). MIC obtained from the combination of EOs was better than that of R. officinalis EOs. All tested substances showed bacteriostatic activity against S. aureus (MBC/MIC > 4).
2.3. Antibiofilm Activity
To evaluate the antibiofilm effect of EOs, an established S. aureus biofilm was treated with various concentrations (MIC, MIC × 2, MIC × 4) of tested agents (Figure 1).
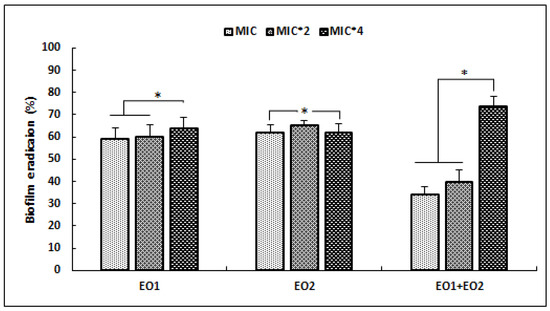
Figure 1.
Effects of R. officinalis (EO1) and M. communis (EO2) essential oils and their combination (EO1 + EO2) on the reduction of preformed biofilm of S. aureus ATCC 25923, expressed as eradication percentages (%) and evaluated by the Crystal Violet staining assay. Values are the average of at least three independent determinations. Error bars represent standard deviations. (*) Differences were considered significant at p < 0.05.
Our results showed that EO1 and EO2 were more effective against the development of preformed biofilm at low concentrations (MIC and 2 × MIC), with percentage reduction values higher than 50%. Whereas, the combination of EOs was more active against S. aureus biofilm, with percentage reduction value exceeding 74% at high concentration of 4 × MIC (p < 0.05). At the same dose (4 × MIC) EO1 and EO2 showed lower antibiofilm activities (p < 0.05) that did not exceed 50%.
2.4. Antivirulence Activities
The effects of tested EOs against S. aureus hemolysin, DNase, and protease production was evaluated by determining the diameter of inhibition halos, in comparison with the untreated strain (Table 3).

Table 3.
Anti-virulence activities of R. officinalis (EO1) and M. communis (EO2) essential oils and their combination (EO1 + EO2) against S. aureus ATCC 25923.
Our results showed that the most significant anti-hemolytic effect was obtained with a combination of EOs, resulting in an inhibition diameter equal to 9 ± 0.8 mm after treatment with a concentration of MIC × 4 (p < 0.05). At the same concentration, both EO1 and EO2 exhibited lower anti-hemolytic activities (p > 0.05).
Regarding the anti-DNase activity, EO of M. communis showed more potent effects when compared to other agents without significant difference (p > 0.05). Furthermore, tested EOs were found to be effective in the inhibition of S. aureus protease activities with a concentration-dependent manner. The most significant anti-protease activity was obtained with the combination of EOs (MIC × 4), with an inhibition diameter reaching 11.6 ± 0.5 mm (p < 0.05). At the same concentration, EO1 was found to be less effective against the protease activity of S. aureus (p < 0.05).
2.5. ROS Generation
In this part of our study, we tested the implication of EOs in S. aureus oxidative stress through ROS generation (Figure 2).
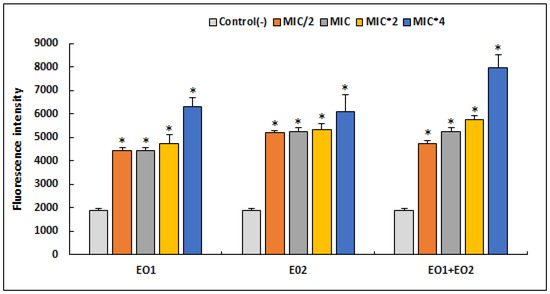
Figure 2.
Quantitation of intracellular ROS production by S. aureus ATCC 25923 after 24 h treatment with different concentrations of R. officinalis (EO1) and M. communis (EO2) essential oils and their combination (EO1 + EO2), using the DCFA-DA probe. Results are expressed as mean fluorescence intensity ± SD. Asterisks represent significant difference (p < 0.05) between each treatment with the negative control.
We have found that tested EOs as well as their association caused an increase in the production of reactive oxygen species compared to the control (p < 0.05). It was deduced that equal proportion of R. officinalis (50%) and M. communis (50%) strongly induced oxidative stress in an S. aureus strain treated with a MIC × 4, triggering a high production of ROS in a dose-dependent manner. At the same concentration (MIC × 4), both EO1 and EO2 showed high production of ROS (p > 0.05) when compared to their combination (EO1 + EO2).
2.6. Catalase Activity
After treatment of an S. aureus strain with different concentrations of EOs, we measured the catalase activity, since this enzyme is involved in the defense of the bacterial cell against oxidative stress. Results of this test are presented in Figure 3.
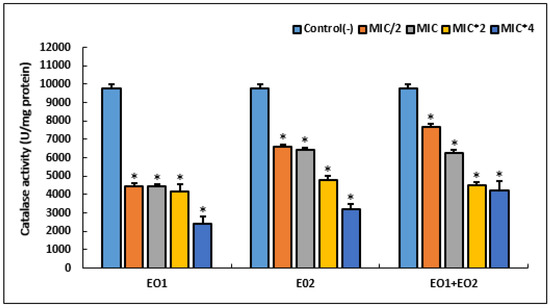
Figure 3.
Effect of different concentrations of R. officinalis (EO1) and M. communis (EO2) essential oils and their combination (EO1 + EO2) on the activity of oxidative stress enzyme (catalase) in S. aureus ATCC 25923. Bacteria were incubated with various concentrations of EOs for 24 h. An asterisk represents a significant difference (p < 0.05) between each treatment and the negative control.
Our results showed that the highest catalase activity (9750 U/mg protein) was observed in an untreated cell. Treatment with various MIC of EOs and their combination revealed a significant decrease in this anti-oxidant activity, in a dose dependent manner (p < 0.05). At high concentration (MIC × 4) of tested agents, EO1 showed the lowest catalase activity (p < 0.05).
3. Discussion
The first part of our investigation was conducted to analyze the chemical composition of R. officinalis and M. communis EOs. Our results showed that the major constituent of R. officinalis EO was found to be 1,8-cineole, which is in agreement with the study of Badreddine et al. [27], having reported a similar value of major compounds of R. officinalis EO, which is 1,8-cineole (34.8%). Another recent study conducted by Moumni et al. [28], showed that Tunisian R. officinalis EOs are characterized with a high percentage of 1,8-cineole (37.6% to 47.2). Regarding the M. communis EO, it was shown that its major compound was found to be the α-Pinene (35.9%) [29], while the study of Cherrat et al. [30] showed the presence of another major compound, Myrtenyl acetate (49.3%). This variability of the chemical composition, even among the same specimens of EOs, is often related to the site of collection as well as geographical provenance. In fact, all these components play a major role in the plant adaptation to the ecology and the environment [31].
In the second part of our investigation, we evaluated the anti-staphylococcal activities of both EOs and their combination. The obtained results showed that all tested substances showed bacteriostatic activity against S. aureus. This finding was confirmed by previous studies having shown that EO of R. officinalis, with a high amount of 1,8-cineole, excreted a bacteriostatic effect against S. aureus strains [32]. Additionally, the same effect was previously reported with M. communis EO against Escherichia coli and S. aureus strains [33]. EOs exert various cytotoxic actions on bacterial strains, acting on several cellular structures. Thus, by crossing the bacterial wall and the plasma membrane, the aromatic compounds of EOs permeabilize the cell membrane and disrupt its function. They can make it permeable to protons and various ions, and inhibit the production of ATP. Eventually, this can lead to lysis of the bacteria. EOs can also reduce membrane fluidity, which impairs the proper functioning of the bacteria [34]. It has been shown that Gram-positive bacteria are more sensitive to EOs than Gram-negative ones. Indeed, it seems that the outer membrane of Gram-negative bacteria, rich in lipopolysaccharides, is more complex and represents an obstacle for aromatic molecules to reach the cytoplasm of this bacterium. However, these active compounds can more easily integrate into the cytoplasm of Gram-positive bacteria given the absence of the complex extra-membrane system, resulting in higher antibacterial activity [35].
Since microbial biofilms increase bacterial resistance to various antimicrobial agents, investigation of antibiofilm effects of bioactive substances from natural resources, such as EOs, remain of interest. Our results revealed that tested EOs and their association displayed high potency against the development of biofilm performed by S. aureus. Previously, the effectiveness EOs against performed methicilin-resistant S. aureus (MRSA) biofilms was reported [36,37]. Generally, the effect of EOs on bacterial biofilm depends on their compositions and on the bacterial strains tested. For example, a previous study reported that M. communis EO exhibited significant anti-biofilm activity against S. aureus strains, which could be attributed to their α-Pinene richness [38]. Another study conducted by Jardak et al. [39]. showed that Tunisian R. officinalis EO exerted a significant S. epidermidis biofilm eradication percentage of 67%. Similarly, another study showed that two varieties of Tunisian Laurus nobilis EO are capable of inhibiting biofilms of oral S. aureus strains, with eradication percentages ranging from 50 to 79% [31]. EOs affect biofilm formation through damage to the outer envelope of this bacterial structure, resulting in the loss of integrity of this layer. Similarly, EOs can also cause inhibition of biofilm synthesis proteins, preventing the development and maturation phase [40]. Within a biofilm, cell-cell interactions and communications have been described. These interactions involve chemical signals, such as quorum sensing (QS) self-inducers [41].
QS is an intercellular communication system that plays an essential role in biofilm formation and virulence-factor production in several bacterial species [17]. Thus, this communication mechanism is used by these microbes to express various survival or virulence traits leading to increased resistance of bacteria [40]. Numerous biosynthetic pathways are regulated by QS, including the production of metabolites [42], biosurfactants [43], and antimicrobials [44]. Interestingly, molecules that interfere with the QS system attenuate bacterial pathogenicity [45]. The activity of tested EOs against S. aureus hemolysin production showed that the most significant anti-hemolytic effect was obtained with combination of EOs. Oher findings reported the same hemolytic activity of two EOs of Dennettia tripetala on sheep red blood cells [46]. In addition, EOs from Lippia origanoides and Thymus vulgaris has been demonstrated to have significant effects on hemolytic activity of S. aureus ATCC 29213 [47]. Apart their roles of increasing the ability of the infection to establish and remain in humans, various types of S. arures hemolysins are produced and associated with a possible activation of QS, prior to biofilm formation [48,49]. Furthermore, our results showed the effectiveness of tested EOs in the inhibition of S. aureus DNase and protease activities with a concentration-dependent manner. Therefore, valorization of EOs seems to be an effective strategy to control virulence factors of pathogenic bacteria such as S. aureus.
The Implication of EOs in S. aureus oxidative stress through ROS generation was investigated. We have found that tested EOs, as well as their association, caused an increase in the production of reactive oxygen species compared to the control, in a dose-dependent manner. Our results are in agreement with a recent study showing that Chamomile EO generated oxidative stress in S. aureus ATCC 29213, which may be the main mode of anti-staphylococcal action of this oil [50]. Similarly, oxidative stress was also detected with an increased level of ROS in bacterial cells of Klebsiella pneumoniae BAA-1705 and E. coli ATCC 25922 treated with Lavender EO [25]. The generation of ROS, including superoxide anions (O2−), hydrogen peroxide (H2O2), and hydroxyl radicals (OH) that are highly reactive, and can lead to oxidative stress if the cell’s antioxidant mechanisms are overcome by pro-oxidant agents [51]. These species are highly reactive, cause oxidative damage and alter the structure and function of macromolecules, such as DNA/RNA, lipids and proteins [52,53]. Even for untreated bacterial cells, ROS production is a natural side effect of aerobic respiration [54], which explains their presence in weak proportions in negative controls.
To protect against the damaging effect of ROS, bacteria are able to produce enzymes such as catalase (CAT) and superoxide dismutase (SOD), to detoxify ROS and accelerate the spontaneous dismutation reaction of H2O2, along with regulatory mechanisms to counteract their damage [55,56]. After treatment of the S. aureus strain with different concentrations of EOs, we measured the catalase activity. Results of this test showed that the highest catalase activity was registered in an untreated cell, while treatment with various MIC of EOs revealed significant decrease in this anti-oxidant activity. In line with our findings, other studies showed decrease in catalase activity in S. aureus bacterium exposed to various phyto-compounds such as Leonurus cardiaca extract [57], allylpyrocatechol [58], silibin [59], and Catechin [60]. Catalase produced by bacteria facilitates cellular detoxification that allows them to repair or escape oxidative damage from H2O2 [61]. Reduction of catalase activity caused by biologically active substances might result in increased H2O2 level and lead to oxidative stress-mediated toxicity in bacterial cells [60]. However, a recent study conducted by Mohammed et al. [62] showed antioxidant capabilities of Artemisia judaica EO increasing the content of CAT and SOD enzymes in treated bacteria. It has been reported that a single chemical compound can function as both an antioxidant and a prooxidant [63]. Furthermore, it has been shown in different bacterial species that a short exposure to various antimicrobial agents leads to an increase of catalase enzyme activity in response to this external stress. Whereas, with increasing exposure time (12 to 24 h), bacteria lose their ability to detoxify these antibacterial agents and mitigate the induced stress, leading to a decreases of catalase activity [64,65].
4. Materials and Methods
4.1. Tested Agents and Bacterial Strain
Rosmarinus officinalis and Myrtus communis essential oils were purchased commercially from a local producer (KG Flower, Diar ben Salem Béni khiar, Tunisia) after hydrodistillation of fresh aerial parts. For each species, 3 samples of the obtained EO were stored at 4 °C until analysis was attempted. The bacterial strain Staphylococcus aureus ATCC 25923 was obtained from the American Type Culture Collection (ATCC). To ensure optimal growth, the bacterial strain was sub-cultured twice, in Brain heart infusion (BHI) broth, and incubated at 37 °C for 24 h before each treatment.
4.2. Chemical Characterization of Essential Oils
Quantitative and qualitative analyses of all the chemical composition of studied essential oils were determined by Gas chromatography with flame-ionization detector (GC/FID) and Gas chromatography-mass-spectrometry (GC/MS) as previously described [28].
4.3. Antibacterial Activity of EOs and Their Combinations
The minimum inhibitory concentration (MIC) of R. officinalis (EO1) and M. communis (EO2) EOs against the S. aureus strain, as well as their combination (50% EO1 + 50% EO2), was determined by the broth dilution method according to standard protocols [66]. Various concentrations ranging between 0.05 mg/mL and 50 mg/mL of the tested agents were aseptically prepared in 96-well microtiter plates containing Muller Hinton broth (MH) and dimethyl sulfoxide (DMSO). Then inocula (0.5 McFarland) of the tested S. aureus strain was added to each well. To determine the minimum bactericidal concentration (MBC) values, MH plates were inoculated with 10 µL from each well medium that had no apparent growth and then incubated for 24 h at 37 °C. MBC was defined as the lowest concentration that killed 99% of the treated bacteria [67].
4.4. Antibiofilm Activity
The antibiofilm activity of EO1 and EO2, as well as their combination (EO1 + EO2), were assessed by crystal violet (CV) staining test as described previously [31]. An established S. aureus biofilm (48 h) on a sterile 96 microtiter plate was treated with various concentrations of tested agents (1 × MIC, 2 × MIC, and 4 × MIC per well), prepared in DMSO and BHI broth. After incubation for 24 h, the plate was stained with CV (1%) and the biofilm’s biomass was quantified at 570 nm using the microplate reader. The percentage of biofilm eradication was determined by the following formula: [(OD growth control − OD sample)/OD growth control] × 100. Where control is untreated biofilm with EOs or their combination.
4.5. Anti-Hemolysin, Anti-DNase and Anti-Protease Activities
The hemolytic activity of the treated S. aureus strain was assessed on bacteriological agar supplemented with 5% sheep’s blood for alpha or beta-hemolysin production, and DNase Test Agar Base (DTAB) for the detection of deoxyribonuclease activity [68]. The overnight bacterial culture grown in trypticase soy broth (TSB, Bio-Rad) was diluted (1:100) with the new TSB medium. Then 100 µL of these dilutions were introduced into uniform wells of 6 mm diameter, which were aseptically perforated in the blood and DTAB agar. Following 24 h incubation of each plate at 37 °C, the diameters of the clear zones around the wells were determined [69]. The anti-hemolysin and anti-DNase activities were performed as described below, with the addition of EOs at different concentrations (1 × MIC, 2 × MIC, and 4 × MIC) per well. All the assays were carried out in triplicate.
To check the inhibition of the protein-digesting enzyme protease, the bacterial cells were incubated with varying concentrations (MIC/2; MIC; MIC × 2 and MIC × 4) of EOs and their combination. Then, 10 µL from each treated bacterial cultures were spotted on Bacto agar containing casein (5%), and incubated at 37 °C for 24 h [70]. The cleared zone surrounding the colony was measured and compared to the measured zone obtained from the control (untreated cells).
4.6. Reactive Oxygen Species (ROS) Generation
The production of ROS by the S. aureus strain exposed to EOs was performed using a peroxynitrite indicator, 20–70-dichlorodihydrofluorescein diacetate (DCFH-DA) (SigmaAldrich, UK), which can detect a broad range of ROS [70]. The adjusted bacterial culture (0.5 McF) was treated with different concentrations of EOs and their combination (corresponding to MIC/2, MIC, 2 × MIC, and 4 × MIC), in presence of DCFH-DA at a final concentration of 5 mM in 0.85% saline, and incubated at 37 °C aerobically for 24 h. Untreated bacterial culture was served as a negative control. The fluorescence emission of DCFH-DA was measured at 525 nm using a Tecan microtiter plate reader with an excitation wavelength of 485 nm [71]. Experiment was carried out in triplicate.
4.7. Antioxidant Enzyme Activity
The catalase (CAT) enzyme activity was determined after treatment of overnight S. aureus cultures with different concentrations of EOs and their combination (MIC/2, MIC, 2 × MIC, and 4 × MIC). Following incubation for 24 h at 37 °C, the treated bacterial culture was centrifuged at 3000 rpm for 10 min, and the resultant pellet was washed twice with PBS. For enzyme assay, the bacterial extract was prepared by resuspending the pellet in 500 μL of cell lysate buffer (10 Mm Tris-HCl, 1 mMEDTA, 0.1% Triton-X-100 and 150 mM NaCl). After incubation at 37 °C for 1 h, the contents were then centrifuged (3000 rpm for 10 min) and the supernatant was collected for enzyme assay [72]. CAT activity in the bacterial extract was determined according to Acuna et al. [73]. In a quartz cuvette, 780 µL of phosphate buffer (KH2PO2/K2HPO4, pH7) were introduced to 200 µL H2O2 (20 mM), to which 20 µL of bacterial cell lysate was added. Then, optical density of the mixture in each bacterial cell was monitored for 60 s (t = 0 s and = 60 s) at a wavelength of 240 nm. One unit (U) of enzyme activity is defined as the amount of enzyme required to convert 1 µmol of H2O2 in one second.
4.8. Statistical Analysis
All the experiments were carried out in triplicate and the data obtained were presented as means ± standard deviations. Data were further analyzed using the one-way analysis of variance (ANOVA) test to calculate the significance of the results: p values less than 0.05 were considered significantly statistically different.
5. Conclusions
In the context of fighting against pathogenic bacteria, using biological methods, we investigated through this study the anti-staphylococcal activities of two Eos of R. officinalis and M. communis. Our results displayed the potent efficacy of both Eos and their combination on S. aureus, as a representative of Gram-positive bacteria. Additionally, we highlight the anti-virulence properties of tested agents, due to their active compounds. Of significance, we showed EOs exerting considerable oxidative stress internally within cells, which is coupled with reduction of catalase activity, contributing to their antagonistic effect against S. aureus. To overcome chemical degradation and prevent the volatilization of bioactive compounds of tested substances, their encapsulation in nanometric systems could offer a promising intervention.
Author Contributions
Conceptualization, A.M.; methodology, K.H.B., S.M., K.B.H.S., M.F.A. and A.M.; validation, K.B.H.S. and A.M.; writing—original draft preparation, A.M., K.H.B., S.M., K.B.H.S. and M.F.A.; writing—review and editing, A.M., K.B.H.S., S.M. and M.F.A.; supervision, A.M., K.B.H.S. and S.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
This work was supported by the Ministry of Higher Education and Scientific Research (MHESR) of Tunisia.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Fridkin, S.K.; Hageman, J.C.; Morrison, M.; Sanza, L.T.; Como-Sabetti, K.; Jernigan, J.A.; Harriman, K.; Harrison, L.H.; Lynfield, R.; Farley, M.M. Active Bacterial Core Surveillance Program of the Emerging Infections Program Network. Methicillin-resistant Staphylococcus aureus disease in three communities. N. Engl. J. Med. 2005, 352, 1436–1444. [Google Scholar] [CrossRef] [PubMed]
- Weiner, L.M.; Webb, A.K.; Limbago, B.; Dudeck, M.A.; Patel, J.; Kallen, A.J.; Edwards, J.R.; Sievert, D.M. Antimicrobial-Resistant Pathogens Associated With Healthcare-Associated Infections: Summary of Data Reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2011-2014. Infect. Control. Hosp. Epidemiol. 2016, 37, 1288–1301. [Google Scholar] [CrossRef]
- Egert, M.; Späth, K.; Weik, K.; Kunzelmann, H.; Horn, C.; Kohl, M.; Blessing, F. Bacteria on smartphone touchscreens in a German university setting and evaluation of two popular cleaning methods using commercially available cleaning products. Folia Microbiol. 2015, 60, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Prévost, G. Toxins in Staphylococcus aureus pathogenesis. In Molecular and Cellular Biology, Horizon Bioscience; Proft, Ed.; Microbial Toxins: Norfolk, UK, 2005; pp. 243–284. [Google Scholar]
- Lentino, J.R. Prosthetic joint infections: Bane of orthopedists, challenge for infectious disease specialists. Clin. Infect. Dis. 2003, 36, 1157–1161. [Google Scholar] [CrossRef] [PubMed]
- Akbari-Ayezloy, E.; Hosseini-Jazani, N.; Yousefi, S.; Habibi, N. Eradication of methicillin resistant S. aureus biofilm by the combined use of fosfomycin and β-chloro-L-alanine. Iran J. Microbiol. 2017, 9, 1–10. [Google Scholar]
- Cerqueira, G.M.; Peleg, A.Y. Insights into Acinetobacter baumannii pathogenicity. IUBMB Life 2011, 63, 1055–1060. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; Li, H.; Zhang, C.; Liang, B.; Li, J.; Wang, L.; Du, X.; Liu, X.; Qiu, S.; Song, H. Relationship between Antibiotic Resistance, Biofilm Formation, and Biofilm-Specific Resistance in Acinetobacter baumannii. Front. Microbiol. 2016, 7, 483. [Google Scholar] [CrossRef]
- Nguyen, H.M.; Graber, C.J. Limitations of antibiotic options for invasive infections caused by methicillin-resistant Staphylococcus aureus: Is combination therapy the answer? J. Antimicrob. Chemother. 2010, 65, 24–36. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Luo, J.; Deng, F.; Huang, Y.; Zhou, H. Antibiotic Combination Therapy: A Strategy to Overcome Bacterial Resistance to Aminoglycoside Antibiotics. Front. Pharmacol. 2022, 13, 839808. [Google Scholar] [CrossRef]
- Singh, N.; Yeh, P.J. Suppressive drug combinations and their potential to combat antibiotic resistance. J. Antibiot. 2017, 70, 1033–1042. [Google Scholar] [CrossRef]
- Abushaheen, M.A.; Muzaheed Fatani, A.J.; Alosaimi, M.; Mansy, W.; George, M.; Acharya, S.; Rathod, S.; Divakar, D.D.; Jhugroo, C.; Vellappally, S.; et al. Antimicrobial resistance, mechanisms and its clinical significance. Dis. Mon. 2020, 66, 100971. [Google Scholar] [CrossRef] [PubMed]
- Uddin, T.M.; Chakraborty, A.J.; Khusro, A.; Zidan, B.R.M.; Mitra, S.; Emran, T.B.; Dhama, K.; Ripon, M.K.H.; Gajdács, M.; Sahibzada, M.U.K.; et al. Antibiotic resistance in microbes: History, mechanisms, therapeutic strategies and future prospects. J. Infect. Public Health 2021, 14, 1750–1766. [Google Scholar] [CrossRef]
- Merghni, A.; Noumi, E.; Hadded, O.; Dridi, N.; Panwar, H.; Ceylan, O.; Mastouri, M.; Snoussi, M. Assessment of the antibiofilm and antiquorum sensing activities of Eucalyptus globulus essential oil and its main component 1,8-cineole against methicillin-resistant Staphylococcus aureus strains. Microb. Pathog. 2018, 118, 74–80. [Google Scholar] [CrossRef]
- El Amri, J.; Elbadaoui, K.; Zair, T.; Bouharb, H.; Chakir, S.; Alaoui, T.I. Étude de l’activité antibactérienne des huiles essentielles de Teucrium capitatium L et l’extrait de Siléne vulgaris sur différentes souches testées. J. Appl. Biosci. 2014, 82, 7481. [Google Scholar] [CrossRef]
- Luis, Â.; Duarte, A.; Gominho, J.; Domingues, F.; Duarte, A.P. Chemical composition, antioxidant, antibacterial and anti-quorum sensing activities of Eucalyptus globulus and Eucalyptus radiata essential oils. Ind. Crops Prod. 2016, 79, 274–282. [Google Scholar] [CrossRef]
- Singh, B.N.; Singh, B.R.; Singh, R.L.; Prakash, D.; Sarma, B.K.; Singh, H.B. Antioxidant and anti-quorum sensing activities of green pod of Acacia nilotica L. Food Chem. Toxicol. 2009, 47, 778–786. [Google Scholar] [CrossRef]
- Al-Jabri, N.N.; Hossain, M.A. Comparative chemical composition and antimicrobial activity study of essential oils from two imported lemon fruits samples against pathogenic bacteria. Beni-Suef Univ. J. Basic Appl. Sci. 2014, 3, 247–253. [Google Scholar]
- Achour, M.; Mateos, R.; Ben Fredj, M.; Mtiraoui, A.; Bravo, L.; Saguem, S. A comprehensive characterisation of rosemary tea obtained from Rosmarinus officinalis L. collected in a sub-humid Area of Tunisia: Compounds from Rosmarinus officinalis tea. Phytochem Anal. 2018, 29, 87–100. [Google Scholar] [CrossRef]
- Ribeiro-Santos, R.; Carvalho-Costa, D.; Cavaleiro, C.; Costa, H.S.; Albuquerque, T.G.; Castilho, M.C.; Ramos, F.; Melo, N.R.; Sanches-Silva, A. A novel insight on an ancient aromatic plant: The rosemary (Rosmarinus officinalis L.). Trends Food Sci. Technol. 2015, 45, 355–368. [Google Scholar] [CrossRef]
- Sasikumar, B. Handbook of Herbs and Spices, 2nd ed.; Woodhead publishing series in food science, technology and nutrition; Woodhead Publishing: Cambridge, UK, 2012; Volume 1, pp. 452–468. [Google Scholar]
- Hennia, A.; Nemmiche, S.; Dandlen, S.; Miguel, M.G. Myrtus communis essential oils: Insecticidal, antioxidant and antimicrobial activities: A review. J. Essent. Oil Res. 2019, 31, 487–545. [Google Scholar] [CrossRef]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Andrade-Ochoa, S.; Chacón-Vargas, K.F.; Sánchez-Torres, L.E.; Rivera-Chavira, B.E.; Nogueda-Torres, B.; Nevárez-Moorillón, G.V. Differential Antimicrobial Effect of Essential Oils and Their Main Components: Insights Based on the Cell Membrane and External Structure. Membranes 2021, 11, 405. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.K.; Yusoff, K.; Thomas, W.; Akseer, R.; Alhosani, M.S.; Abushelaibi, A.; Lim, S.H.; Lai, K.S. Lavender essential oil induces oxidative stress which modifies the bacterial membrane permeability of carbapenemase producing Klebsiella pneumoniae. Sci. Rep. 2020, 10, 819. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Long, Y.; Li, D.; Shi, A.; Deng, J.; Ma, Y.; Wen, J.; Li, X.; Zhang, Y.; Liu, S.; et al. Natural essential oils efficacious in internal organs fibrosis treatment: Mechanisms of action and application perspectives. Pharmacol. Res. 2022, 182, 106339. [Google Scholar] [CrossRef]
- Badreddine, B.S.; Olfa, E.; Samir, D.; Hnia, C.; Lahbib, B.J. Chemical composition of Rosmarinus and Lavandula essential oils and their insecticidal effects on Orgyia trigotephras (Lepidoptera, Lymantriidae). Asian Pac. J. Trop. Med. 2015, 8, 98–103. [Google Scholar] [CrossRef]
- Moumni, S.; Elaissi, A.; Trabelsi, A.; Merghni, A.; Chraief, I.; Jelassi, B.; Chemli, R.; Ferchichi, S. Correlation between chemical composition and antibacterial activity of some Lamiaceae species essential oils from Tunisia. BMC Complement Med. Ther. 2020, 20, 103. [Google Scholar] [CrossRef]
- Bouzabata, A. Traditional Treatment of high blood pressure and diabetes in Souk Ahras District. J. Pharmacogn. Phytother. 2013, 5, 12–20. [Google Scholar]
- Cherrat, L.; Espina, L.; Bakkali, M.; García-Gonzalo, D.; Pagán, R.; Laglaoui, A. Chemical composition and antioxidant properties of Laurus nobilis L. and Myrtus communis L. essential oils from Morocco and evaluation of their antimicrobial activity acting alone or in combined processes for food preservation. J. Sci. Food Agric. 2014, 94, 1197–1204. [Google Scholar] [CrossRef]
- Merghni, A.; Marzouki, H.; Hentati, H.; Aouni, M.; Mastouri, M. Antibacterial and antibiofilm activities of Laurus nobilis L. essential oil against Staphylococcus aureus strains associated with oral infections. Pathol. Biol. 2015. Online ahead of print. [Google Scholar]
- Ait-Ouazzou, A.; Lorán, S.; Bakkali, M.; Laglaoui, A.; Rota, C.; Herrera, A.; Pagán, R.; Conchello, P. Chemical composition and antimicrobial activity of essential oils of Thymus algeriensis, Eucalyptus globulus and Rosmarinus officinalis from Morocco. J. Sci. Food Agric. 2011, 91, 2643–2651. [Google Scholar] [CrossRef]
- Djenane, D.; Yangüela, J.; Amrouche, T.; Boubrit, S.; Boussad, N.; Roncalés, P. Chemical composition and antimicrobial effects of essential oils of Eucalyptus globulus, Myrtus communis and Satureja hortensis against Escherichia coli O157, H7 and Staphylococcus aureus in minced beef. Food Sci. Technol. Int. 2011, 17, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Silou, T.; Nsikabaka, S.; Nombault Nienzy, J.P.; Baou, A.; Biassala, T.E.; Figuérédo, G.; Chalchat, J.C. Aromatic Plants from Plateau des Cataractes: Occurrence of the Citronella Chemotype of Cymbopogon flexuosus (Nees ex Steud.) W.Watson Acclimatized in Congo-Brazzaville. Chem. Biodivers. 2019, 16, e1800446. [Google Scholar] [CrossRef]
- Fadel, H.; Benayache, F.; Chalchat, J.C.; Figueredo, G.; Chalard, P.; Hazmoune, H.; Benayache, S. Essential oil constituents of Juniperus oxycedrus L. and Cupressus sempervirens L. (Cupressaceae) growing in Aures region of Algeria. Nat. Prod. Res. 2021, 35, 2616–2620. [Google Scholar] [CrossRef]
- Rosato, A.; Sblano, S.; Salvagno, L.; Carocci, A.; Clodoveo, M.L.; Corbo, F.; Fracchiolla, G. Anti-Biofilm Inhibitory Synergistic Effects of Combinations of Essential Oils and Antibiotics. Antibiotics 2020, 9, 637. [Google Scholar] [CrossRef]
- Gómez-Sequeda, N.; Cáceres, M.; Stashenko, E.E.; Hidalgo, W.; Ortiz, C. Antimicrobial and Antibiofilm Activities of Essential Oils against Escherichia coli O157, H7 and Methicillin-Resistant Staphylococcus aureus (MRSA). Antibiotics 2020, 9, 730. [Google Scholar] [CrossRef]
- Akin, M.; Aktumsek, A.; Nostro, A. Antibacterial activity and composition of the essential oils of Eucalyptus camaldulensis Dehn and Myrtus communis L. growing in Northern Cyprus. Afr. J. Biotechnol. 2010, 9, 531–535. [Google Scholar]
- Jardak, M.; Elloumi-Mseddi, J.; Aifa, S.; Mnif, S. Chemical composition, anti-biofilm activity and potential cytotoxic effect on cancer cells of Rosmarinus officinalis L. essential oil from Tunisia. Lipids Health Dis. 2017, 16, 190. [Google Scholar] [CrossRef]
- Poli, J.P.; Guinoiseau, E.; de Rocca Serra, D.; Sutour, S.; Paoli, M.; Tomi, F.; Quilichini, Y.; Berti, L.; Lorenzi, V. Anti-Quorum Sensing Activity of 12 Essential Oils on chromobacterium violaceum and Specific Action of cis-cis-p-Menthenolide from Corsican Mentha suaveolens ssp. Insularis. Molecules 2018, 23, 2125. [Google Scholar] [CrossRef]
- Passerini, D.; Fécamp, F.; Marchand, L.; Kolypczuk, L.; Bonnetot, S.; Sinquin, C.; Verrez-Bagnis, V.; Hervio-Heath, D.; Colliec-Jouault, S.; Delbarre-Ladrat, C. Characterization of Biofilm Extracts from Two Marine Bacteria. Appl. Sci. 2019, 9, 4971. [Google Scholar] [CrossRef]
- Mitra, S.; Thawrani, D.; Banerjee, P.; Gachhui, R.; Mukherjee, J. Induced biofilm cultivation enhances riboflavin production by an intertidally derived Candida famata. Appl. Biochem. Biotechnol. 2012, 166, 1991–2006. [Google Scholar] [CrossRef]
- Victor, I.U.; Kwiencien, M.; Tripathi, L.; Cobice, D.; McClean, S.; Marchant, R.; Banat, I.M. Quorum sensing as a potential target for increased production of rhamnolipid biosurfactant in Burkholderia thailandensis E264. Appl. Microbiol. Biotechnol. 2019, 103, 6505–6517. [Google Scholar] [CrossRef]
- Schmidt, S.; Blom, J.F.; Pernthaler, J.; Berg, G.; Baldwin, A.; Mahenthiralingam, E.; Eberl, L. Production of the antifungal compound pyrrolnitrin is quorum sensing-regulated in members of the Burkholderia cepacia complex. Environ. Microbiol. 2009, 11, 1422–1437. [Google Scholar] [CrossRef]
- Issac Abraham, S.V.; Palani, A.; Ramaswamy, B.R.; Shunmugiah, K.P.; Arumugam, V.R. Antiquorum sensing and antibiofilm potential of Capparis spinosa. Arch. Med. Res. 2011, 42, 658–668. [Google Scholar] [CrossRef] [PubMed]
- Okoh, S.O.; Iweriegbor, B.C.; Okoh, O.O.; Nwodo, U.U.; IOkoh, A. Bactericidal and antioxidant properties of essential oils from the fruits Dennettia tripetala G. Baker. BMC Complement Altern. Med. 2016, 16, 486. [Google Scholar] [CrossRef] [PubMed]
- Martínez, A.; Manrique-Moreno, M.; Klaiss-Luna, M.C.; Stashenko, E.; Zafra, G.; Ortiz, C. Effect of Essential Oils on Growth Inhibition, Biofilm Formation and Membrane Integrity of Escherichia coli and Staphylococcus aureus. Antibiotics 2021, 10, 1474. [Google Scholar] [CrossRef]
- Kim, M.K.; Zhao, A.; Wang, A.; Brown, Z.Z.; Muir, T.W.; Stone, H.A.; Bassler, B.L. Surface-attached molecules control Staphylococcus aureus quorum sensing and biofilm development. Nat. Microbiol. 2017, 2, 17080. [Google Scholar] [CrossRef] [PubMed]
- Quave, C.L.; Horswill, A.R. Identification of Staphylococcal Quorum Sensing Inhibitors by Quantification of õ-Hemolysin with High Performance Liquid Chromatography. Methods Mol. Biol. 2018, 1673, 363–370. [Google Scholar] [PubMed]
- Das, S.; Horváth, B.; Šafranko, S.; Jokić, S.; Széchenyi, A.; Kőszegi, T. Antimicrobial Activity of Chamomile Essential Oil: Effect of Different Formulations. Molecules 2019, 24, 4321. [Google Scholar] [CrossRef] [PubMed]
- Correa-Llantén, D.N.; Amenábar, M.J.; Blamey, J.M. Antioxidant capacity of novel pigments from an Antarctic bacterium. J. Microbiol. 2012, 50, 374–379. [Google Scholar] [CrossRef]
- Sies, H. Oxidative stress: Oxidants and antioxidants. Exp. Physiol. 1997, 82, 291–295. [Google Scholar] [CrossRef]
- Imlay, J.A. Pathways of oxidative damage. Annu. Rev. Microbiol. 2003, 57, 395–418. [Google Scholar] [CrossRef]
- Ong, K.S.; Cheow, Y.L.; Lee, S.M. The role of reactive oxygen species in the antimicrobial activity of pyochelin. J. Adv. Res. 2017, 8, 393–398. [Google Scholar] [CrossRef]
- Van Acker, H.; Gielis, J.; Acke, M.; Cools, F.; Cos, P.; Coenye, T. The Role of Reactive Oxygen Species in Antibiotic-Induced Cell Death in Burkholderia cepacia Complex Bacteria. PLoS ONE 2016, 11, e0159837. [Google Scholar] [CrossRef]
- Gasser, V.; Baco, E.; Cunrath, O.; August, P.S.; Perraud, Q.; Zill, N.; Schleberger, C.; Schmidt, A.; Paulen, A.; Bumann, D.; et al. Catechol siderophores repress the pyochelin pathway and activate the enterobactin pathway in Pseudomonas aeruginosa: An opportunity for siderophore-antibiotic conjugates development. Environ. Microbiol. 2016, 18, 819–832. [Google Scholar] [CrossRef]
- Micota, B.; Sadowska, B.; Podsędek, A.; Paszkiewicz, M.; Sosnowska, D.; Różalska, B. Is it true that plant-derived polyphenols are always beneficial for the human? In vitro study on Leonurus cardiaca extract properties in the context of the pathogenesis of Staphylococcus aureus infections. J. Med. Microbiol. 2016, 65, 1171–1181. [Google Scholar] [CrossRef] [PubMed]
- Hussain, R.M.; Abdullah, N.F.; Amom, Z. Killing of Staphylococcus aureus by allylpyrocatechol is potentiated by induction of intracellular oxidative stress and inhibition of catalase activity. J. Integr. Med. 2016, 14, 456–464. [Google Scholar] [CrossRef]
- Cai, J.Y.; Li, J.; Hou, Y.N.; Ma, K.; Yao, G.D.; Liu, W.W.; Hayashi, T.; Itoh, K.; Tashiro, S.I.; Onodera, S.; et al. Concentration-dependent dual effects of silibinin on kanamycin-induced cells death in Staphylococcus aureus. Biomed Pharmacother. 2018, 102, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Sinsinwar, S.; Vadivel, V. Catechin isolated from cashew nut shell exhibits antibacterial activity against clinical isolates of MRSA through ROS-mediated oxidative stress. Appl. Microbiol. Biotechnol. 2020, 104, 8279–8297. [Google Scholar] [CrossRef]
- Golenia, A.; Leśkiewicz, M.; Regulska, M.; Budziszewska, B.; Szczęsny, E.; Jagiełła, J.; Wnuk, M.; Ostrowska, M.; Lasoń, W.; Basta-Kaim, A.; et al. Catalase activity in blood fractions of patients with sporadic ALS. Pharm. Rep. 2014, 66, 704–707. [Google Scholar] [CrossRef]
- Mohammed, H.A.; Qureshi, K.A.; Ali, H.M.; Al-Omar, M.S.; Khan, O.; Mohammed, S.A.A. Bio-Evaluation of the Wound Healing Activity of Artemisia judaica L. as Part of the Plant’s Use in Traditional Medicine; Phytochemical, Antioxidant, Anti-Inflammatory, and Antibiofilm Properties of the Plant’s Essential Oils. Antioxidants 2022, 11, 332. [Google Scholar] [CrossRef] [PubMed]
- Monsalves, M.T.; Ollivet-Besson, G.P.; Amenabar, M.J.; Blamey, J.M. Isolation of a Psychrotolerant and UV-C-Resistant Bacterium from Elephant Island, Antarctica with a Highly Thermoactive and Thermostable Catalase. Microorganisms 2020, 8, 95. [Google Scholar] [CrossRef]
- Barros, D.; Pradhan, A.; Mendes, V.M.; Manadas, B.; Santos, P.M.; Pascoal, C.; Cássio, F. Proteomics and antioxidant enzymes reveal different mechanisms of toxicity induced by ionic and nanoparticulate silver in bacteria. Environ. Sci. Nano 2019, 6, 1207–1218. [Google Scholar] [CrossRef]
- Hamida, R.S.; Ali, M.A.; Goda, D.A.; Khalil, M.I.; Al-Zaban, M.I. Novel Biogenic Silver Nanoparticle-Induced Reactive Oxygen Species Inhibit the Biofilm Formation and Virulence Activities of Methicillin-Resistant Staphylococcus aureus (MRSA) Strain. Front. Bioeng. Biotechnol. 2020, 8, 433. [Google Scholar] [CrossRef] [PubMed]
- Conforti, F.; Statti, G.A.; Tundis, R.; Menichini, F.; Houghton, P. Antioxidant activity of methanolic extract of Hypericum triquetrifolium Turra aerial part. Fitoterapia 2002, 73, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Magina, M.D.; Dalmarco, E.M.; Wisniewski AJr Simionatto, E.L.; Dalmarco, J.B.; Pizzolatti, M.G.; Brighente, I.M.C. Chemical composition and antibacterial activity of essen-tial oils of Eugenia species. J. Nat. Med. 2009, 63, 345–350. [Google Scholar] [CrossRef]
- Quinn, P.J.; Carter, M.E.; Markey, B.K.; Carter, G.R. The streptococci and related cocci. In Clinical Veterinary Microbiology; Quinn, P.J., Carter, M.E., Markey, B., Carter, G.R., Eds.; Mosby Ltd.: London, UK, 1994; p. 127e36. [Google Scholar]
- Kanemitsu, K.; Nishino, T.; Kunishima, H.; Okamura, N.; Takemura, H.; Yamamoto, H.; Kaku, M. Quantitative determination of gelatinase activity among enterococci. J. Microbiol. Methods 2001, 47, 11–16. [Google Scholar] [CrossRef]
- Arakha, M.; Pal, S.; Samantarrai, D.; Panigrahi, T.K.; Mallick, B.C.; Pramanik, K. Antimicrobial activity of iron oxide nanoparticle upon modulation of nanoparticle-bacteria interface. Sci. Rep. 2015, 5, 14813. [Google Scholar] [CrossRef]
- Han, L.; Patil, S.; Boehm, D.; Milosavljevic, V.; Cullen, P.J.; Bourke, P. Mechanisms of inactivation by high-voltage atmospheric cold plasma differ for Escherichia coli and Staphylococcus aureus. Appl. Environ. Microbiol. 2016, 82, 450–458. [Google Scholar] [CrossRef]
- Martins, D.; McKay, G.; Sampathkumar, G.; Khakimova, M.; English, A.M.; Nguyen, D. Superoxide dismutase activity confers (p)ppGppmediated antibiotic tolerance to stationary-phase Pseudomonas aeruginosa. Proc. Natl. Acad. Sci. USA 2018, 115, 9797–9802. [Google Scholar] [CrossRef]
- Acuña, L.G.; Calderón, I.L.; Elías, A.O.; Castro, M.E.; Vásquez, C.C. Expression of the yggE gene protects Escherichia coli from potassium tellurite-generated oxidative stress. Arch. Microbiol. 2009, 191, 473–476. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).