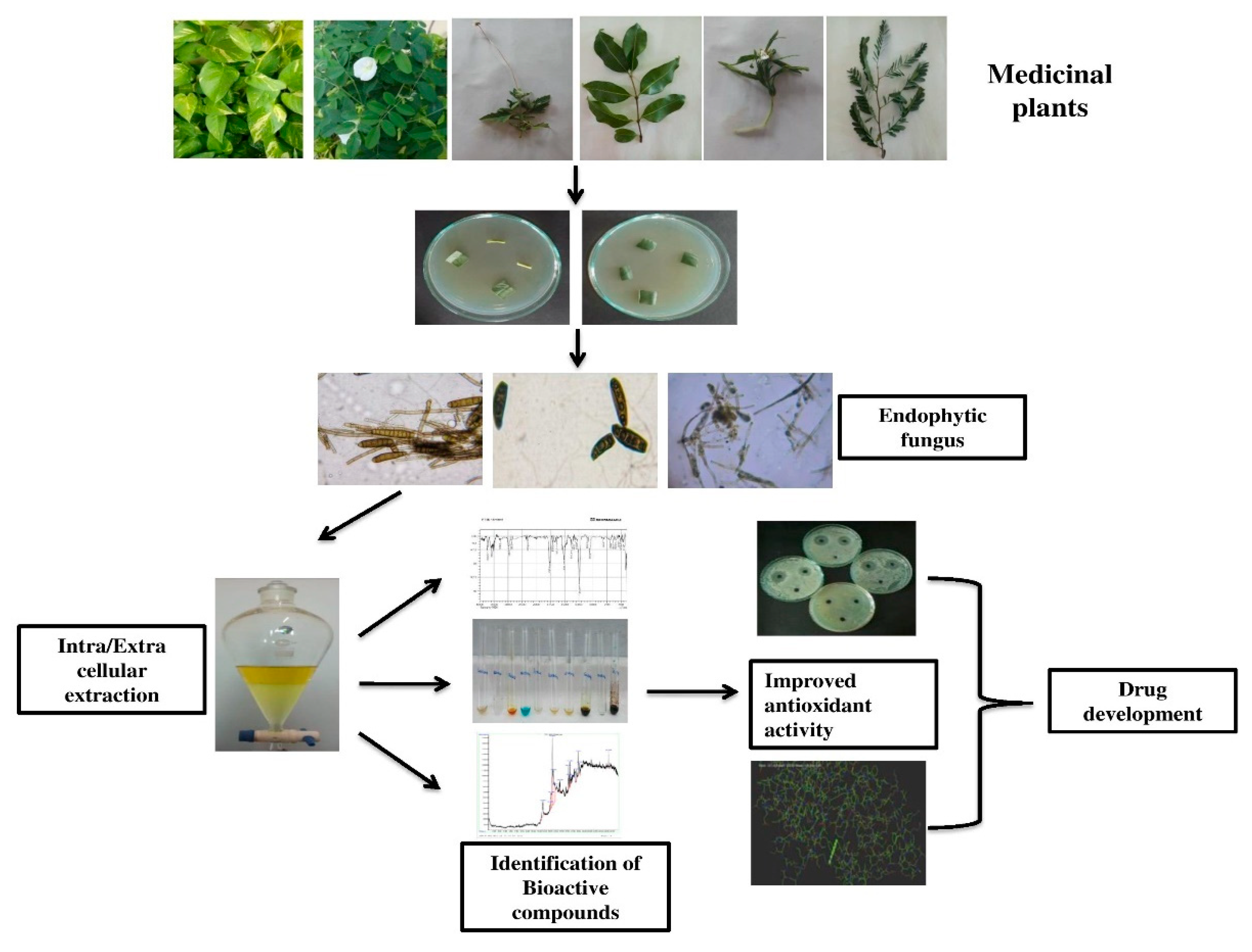
Bioactive Components and Health Potential of Endophytic Micro-Fungal Diversity in Medicinal Plants
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Surface Sterilization
2.3. Sample Inoculation
2.4. Morphological Identification of Endophytic Fungi
2.5. Lactophenol Cotton Blue Mounting
2.6. Diversity Analysis
2.7. Production of Fermentation Medium
2.8. Extraction of Metabolites
2.9. FTIR Analysis
2.10. GC-MS Analysis
2.11. Antibacterial Assay
2.12. Phytochemical Analysis
2.13. Flavonoids Test
2.13.1. Alkaline
2.13.2. Lead Acetate
2.13.3. Ferric Chloride
2.13.4. Sulphuric Acid
2.13.5. Saponins
2.14. Tannins Test
2.14.1. Ferric Chloride
2.14.2. Gelatin
2.14.3. Lead Acetate
2.15. Proteins Test
2.15.1. Millon
2.15.2. Biuret
2.15.3. Ninhydrin
2.15.4. Aminoacids
2.16. Carbohydrate Test
2.16.1. Benedicts
2.16.2. Iodine
2.16.3. Fehlings
2.16.4. Terpenoids
2.16.5. Steroids
2.16.6. Glycosides
2.16.7. Alkaloids
Mayer Test
2.16.8. Phenols
Ferric Chloride Test
Litmus Paper Test
2.17. Antioxidant Analysis
2.17.1. DPPH Radial Scavenging Activity
2.17.2. H2O2 Radical Scavenging Activity
2.17.3. Nitric Oxide Scavenging Activity
2.18. Molecular Docking
2.19. FT-IR Spectrophotometer Analysis
2.20. GC–MS Analysis
2.21. Statistical Analysis
3. Results and Discussion
3.1. Endophytic Microfungal Isolation
3.2. FTIR Analysis
3.3. GC-MS Analysis for Bioactive Compound Screening
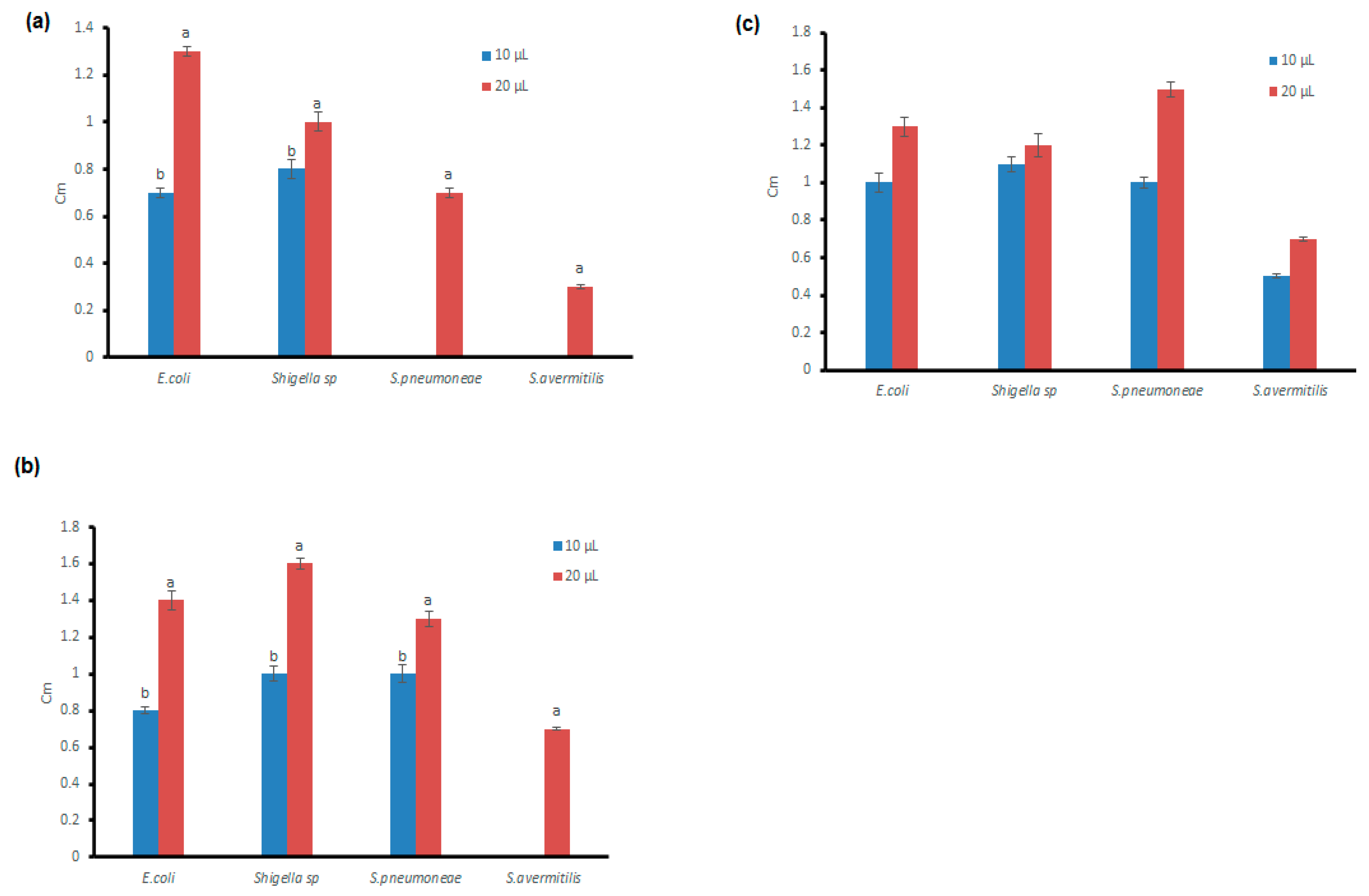
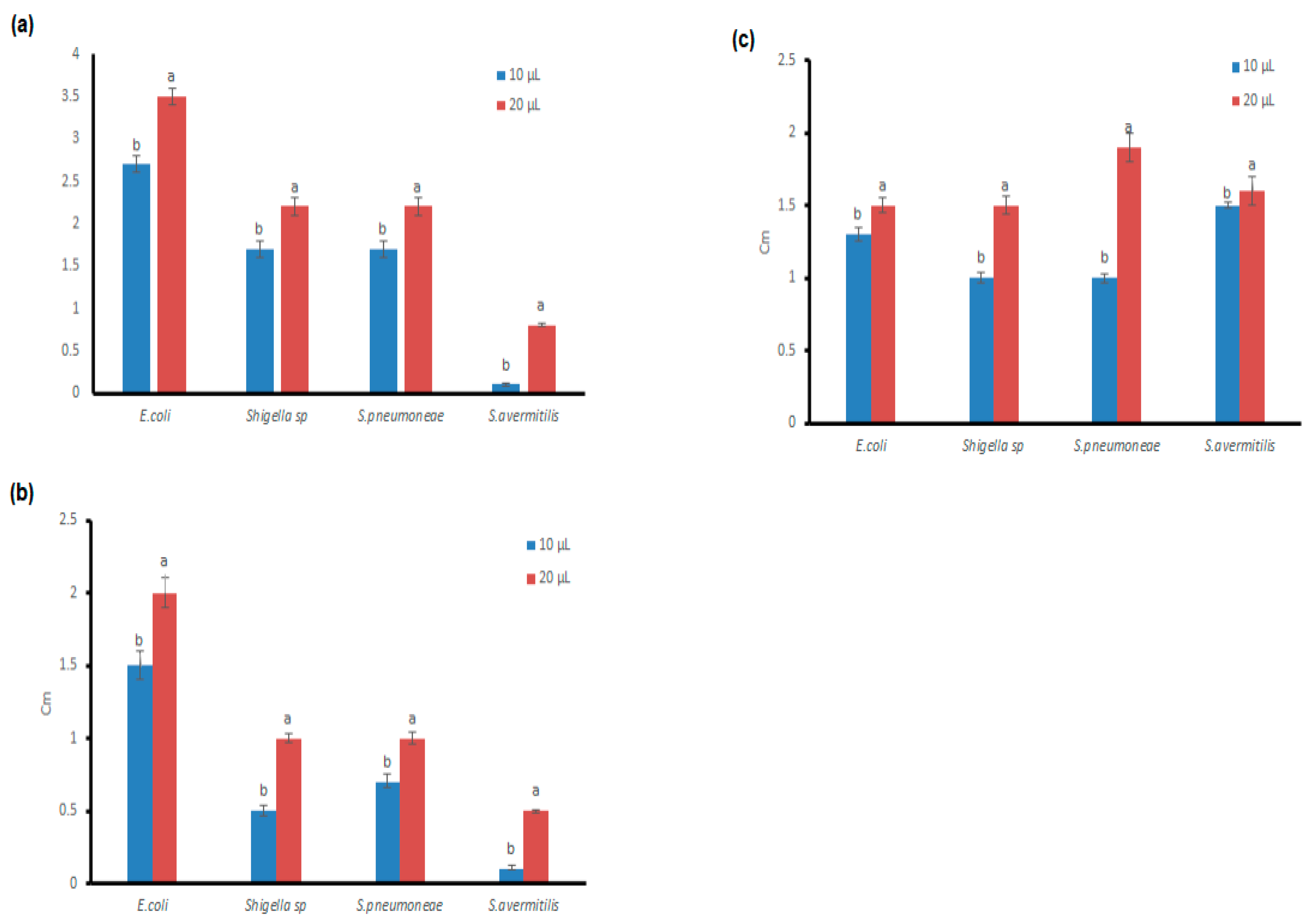
3.4. Antibacterial Potential of the Microfungal Extract
3.5. Phytochemical Analysis of the Endophytic Microfungal Extract
3.6. Antioxidant Activity of Endophytic Microfungal Extracts
3.6.1. DPPH Antioxidant Scavenging Activity of Fungal Extracts
3.6.2. H2O2 and Nitric Oxide Antioxidant Scavenging Activity of Fungal Extracts
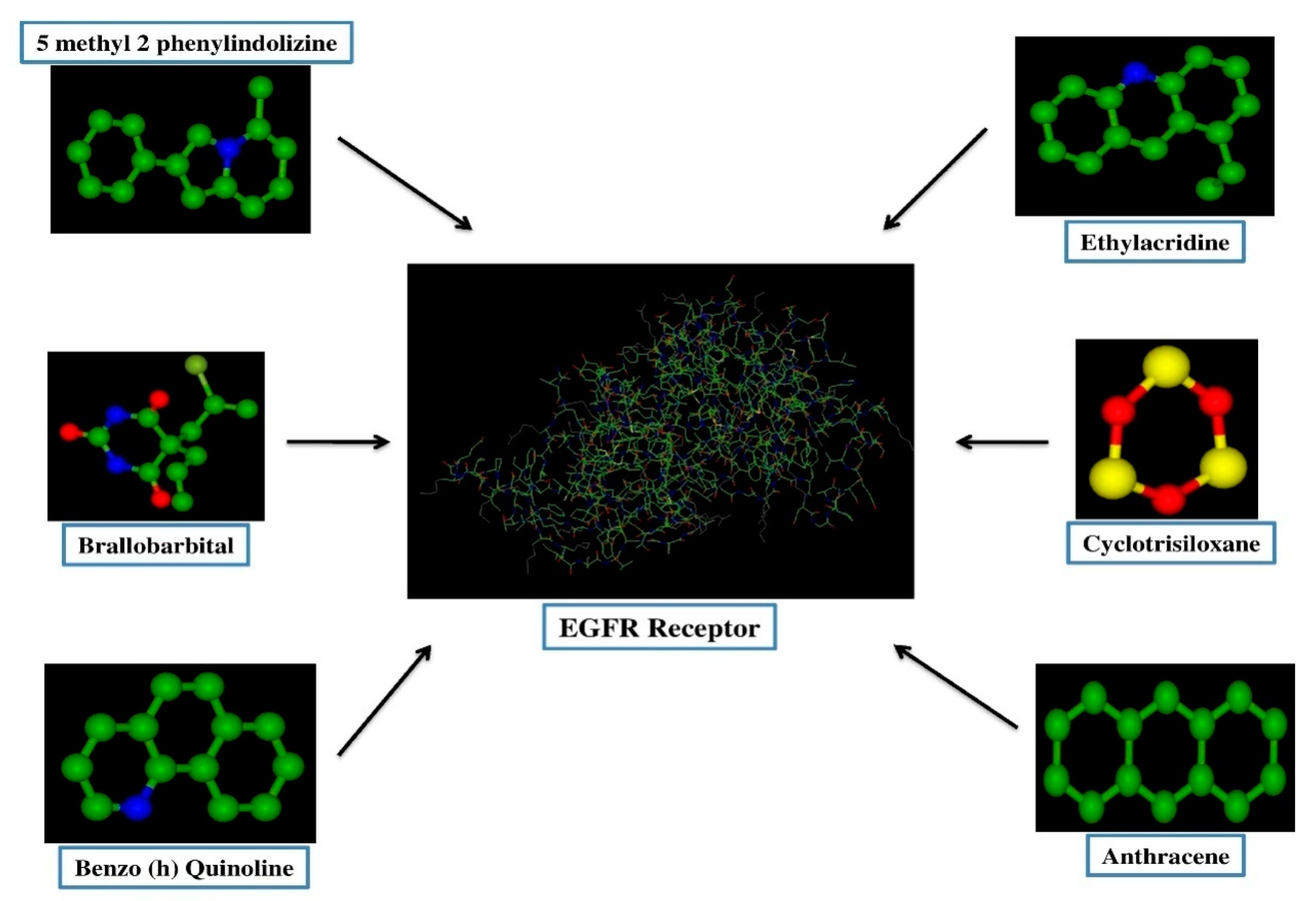
3.6.3. Molecular Docking Study
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pavithra, G.; Bindal, S.; Rana, M.; Srivastava, S. Role of endophytic microbes against plant pathogens: A review. Asian J. Plant Sci. 2020, 19, 54–62. [Google Scholar]
- Palanichamy, P.; Krishnamoorthy, G.; Kannan, S.; Marudhamuthu, M. Bioactive potential of secondary metabolites derived from medicinal plant endophytes. Egypt. J. Basic Appl. Sci. 2018, 5, 303–312. [Google Scholar] [CrossRef] [Green Version]
- Safwan, S.; Hsiao, G.; Lee, T.-H.; Lee, C.-K. Bioactive compounds from an endophytic fungi Nigrospora aurantiaca. Bot. Stud. 2021, 62, 18. [Google Scholar] [CrossRef]
- Kumari, M.; Taritla, S.; Sharma, A.; Jayabaskaran, C. Antiproliferative and antioxidative bioactive compounds in extracts of marine-derived endophytic fungus Talaromyces purpureogenus. Front. Microbiol. 2018, 9, 1777. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ukwatta, K.M.; Lawrence, J.L.; Wijayarathne, C. Antimicrobial, anticancer, anti-filarial and anti-inflammatory activities of Cowabenzophenone A extracted from the endophytic fungus Aspergillus terreus isolated from a mangrove plant Bruguiera gymnorrhyza. Mycology 2020, 11, 297–305. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.; Choi, K.; Chung, D. 3.35—Sample preparation for capillary electrophoretic applications. In Comprehensive Sampling and Sample Preparation; Pawliszyn, J., Ed.; Academic Press: Cambridge, MA, USA, 2012; Volume 3, pp. 701–721. [Google Scholar]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for invitro evaluating antimicrobial activity: Ar eview. J. Pharm. Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bethune, G.; Bethune, D.; Ridgway, N.; Xu, Z. Epidermal growth factor receptor (EGFR) in lung cancer: An overview and update. J. Thorac. Dis. 2010, 2, 48. [Google Scholar]
- Sharma, D.; Pramanik, A.; Agrawal, P.K. Evaluation of bioactive secondary metabolites from endophytic fungus Pestalotiopsis neglecta BAB-5510 isolated from leaves of Cupressus torulosa D.Don. 3Biotech 2016, 6, 210. [Google Scholar] [CrossRef] [Green Version]
- ALKahtani, M.D.; Fouda, A.; Attia, K.A.; Al-Otaibi, F.; Eid, A.M.; Ewais, E.E.-D.; Hijri, M.; St-Arnaud, M.; Hassan, S.E.-D.; Khan, N. Isolation and characterization of plant growth promoting endophytic bacteria from desert plants and their application as bioinoculants for sustainable agriculture. Agronomy 2020, 10, 1325. [Google Scholar] [CrossRef]
- Olusola, O.O.; Adebola, O.A.; Samuel, O.O. Evaluation of micro-fungi associated with leafspot of Allanblackia floribunda Oliv.in Southern Nigeria. Afr. J. Microbiol. Res. 2020, 14, 380–387. [Google Scholar] [CrossRef]
- Barron, G. The genera of hyphomycetes from soil. Soil Sci. 1972, 106, 477. [Google Scholar] [CrossRef]
- Domsch, K.H.; Gams, W.; Anderson, T.-H. Compendium of Soil Fungi; Academic Press Ltd.: London, UK, 1980; p. 1. [Google Scholar]
- Hajjaj, H.; Blanc, P.; Goma, G.; Francois, J. Sampling techniques and comparative extraction procedures for quantitative determination of intra- and extracellular metabolites in filamentous fungi. FEMS Microbiol. Lett. 1998, 164, 195–200. [Google Scholar] [CrossRef]
- Elango, D.; Manikandan, V.; Jayanthi, P.; Velmurugan, P.; Balamuralikrishnan, B.; Ravi, A.V.; Shivakumar, M.S. Selection and characterization of extracellular enzyme production by an endophytic fungi Aspergillu ssojae and its bio-efficacy analysis against cotton leaf worm, Spodopteralitura. Curr. Plant Biol. 2020, 23, 100153. [Google Scholar] [CrossRef]
- Fouda, A.; Hassan, S.E.-D.; Abdo, A.M.; El-Gamal, M.S. Antimicrobial, antioxidant and larvicidal activities of spherical silver nanoparticles synthesized by endophytic Streptomyces spp. Biol. Trace Elem. Res. 2020, 195, 707–724. [Google Scholar] [CrossRef] [PubMed]
- Roghini, R.; Vijayalakshmi, K. Phytochemica lscreening, quantitative analysis of flavonoids and minerals in ethanolic extract of Citrus paradisi. Int. J. Pharm. Sci. Res. 2018, 9, 4859–4864. [Google Scholar]
- Senguttuvan, J.; Paulsamy, S.; Karthika, K. Phytochemical analysis and evaluation of leaf and root parts of the medicinal herb, Hypochaeris radicata L. for in vitro antioxidant activities. Asian Pac. J. Trop. Biomed. 2014, 4, S359–S367. [Google Scholar] [CrossRef]
- Shaik, N.A.; Al-Kreathy, H.M.; Ajabnoor, G.M.; Verma, P.K.; Banaganapalli, B. Molecular designing, virtual screening and docking study of novel curcumin analogue a smutation (S769LandK846R) selective inhibitor for EGFR. SaudiJ. Biol. Sci. 2019, 26, 439–448. [Google Scholar] [CrossRef]
- Khalil, A.M.A.; Hassan, S.E.-D.; Alsharif, S.M.; Eid, A.M.; Ewais, E.E.-D.; Azab, E.; Gobouri, A.A.; Elkelish, A.; Fouda, A. Isolation and characterization of fungal endophyte sisolated from medicinal plant Ephedra pachyclada as plant growth-promoting. Biomolecules 2021, 11, 140. [Google Scholar] [CrossRef]
- Saroj, A.; Alam, M.; Qamar, N.; Abdul-Khaliq; Sattar, A. First report of Bipolaris australiensis causing pod rot of senna in India. New Dis. Rep. 2011, 23, 28. [Google Scholar] [CrossRef] [Green Version]
- Jagannath, S.; Konappa, N.; Lokesh, A.; Bhuvaneshwari; Dasegowda, T.; Udayashankar, A.C.; Chowdappa, S.; Cheluviah, M.; Satapute, P.; Jogaiah, S. Bioactive compounds guided diversity of endophytic fungi from Baliospermum montanum and their potential extracellular enzymes. Anal. Biochem. 2021, 614, 114024. [Google Scholar] [CrossRef]
- Sundar, R.D.V.; Anitha, K.; Arunachalam, S. In vitro Antioxidant and Phytochemical analysis of crude extracts of endophytic fungi (Cladosporium sp.) from Boerhaavia diffusa Linn. Res. J. Pharm. Technol. 2021, 14, 202–206. [Google Scholar] [CrossRef]
- Mehdi, M.; Alawi, A.; Thabet, F.; Alarabi, Y.S.; Omar, G.N.; Pradhan, V. Analysis of bioactive chemical compounds of leaves extracts from Tamarindus indica using FT-IR and GC-MS spectroscopy. Asian J. Res. Biochem. 2021, 8, 22–34. [Google Scholar] [CrossRef]
- Tran, T.-M.-D.; Nguyen, V.-K.; Duong, T.-H.; Tran, T.-N.; Nguyen, N.-H.; Devi, A.P.; Chavasiri, W.; Dinh, M.-H.; Tran, N.-M.-A.; Sichaem, J. Maydisone, a novel oxime polyketide from the cultures of Bipolarismaydis. Nat. Prod. Res. 2021, 36, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Strobel, G.; Daisy, B. Bioprospecting for microbial endophytes and their natural products. Microbiol. Mol. Biol. Rev. 2003, 67, 491–502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bharadwaj, R.; Cunningham, K.-M.; Zhang, K.; Lloyd, T.-E. FIG4 regulates lysosome membrane homeostasis independent of phosphatase function. Hum. Mol. Genet. 2016, 25, 681–692. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weber, R.-W.; Stenger, E.; Meffert, A.; Hahn, M. Brefeldin A production by Phoma medicaginis in dead pre-colonized plant tissue: A strategy for habitat conquest? Mycol. Res. 2004, 108, 662–671. [Google Scholar] [CrossRef]
- Peláez, F. Biological activities of fungal metabolites. In Handbook of Industrial Mycology; An, Z., Ed.; MarcelDekker: NewYork, NY, USA, 2005; pp. 49–92. [Google Scholar]
- Bacon, C.-W.; White, J.-F. Microbial Endophytes; MarcelDeker: NewYork, NY, USA, 2000. [Google Scholar]
- Kanjana, M.; Kanimozhi, G.; Udayakumar, R.; Panneerselvam, A. GC-MS analysis of bioactive compounds of endophytic fungi Chaetomium globosum, Cladosporium tenuissimum and Penicillium janthinellum. J. Biomed. Pharm. Sci. 2019, 2, 123. [Google Scholar]
- Hateet, R.R. GC-MS Analysis of extract for Endophytic fungus Acremonium coenophialum and its Antimicrobial and Antidiabetic. Res. J. Pharm. Technol. 2020, 13, 119–123. [Google Scholar] [CrossRef]
- Amelia, P.; Ivada, P.A.K.; Fitriana, N.; Komala, I.; Bahri, S.; Hanafi, M. Antioxidant and antimicrobial activity of secondary metabolite produced by endophytic fungi isolated from Lannea coromandelica (Houtt.) Merr. Int. J. Pharm. Sci. Res. 2021, 12, 1588–1592. [Google Scholar]
- Ding, L.; Qin, S.; Li, F.; Chi, X.; Laatsch, H. Isolation, Antimicrobial activity, and metabolites of fungus Cladosporium sp. Associated with Red Alga Porphyrayezoensis. Curr. Microbiol. 2008, 56, 229–235. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Debnath, S.; Saha, A.K. Evaluation of antioxidant activity of two endophytic fungi isolated from Ananus comosus L. Eur. J. Biomed. Pharm. 2018, 5, 896–899. [Google Scholar]
- Gunasekaran, S.; Sathiavelu, M.; Arunachalam, S. In vitro antioxidant and antibacterial activity of endophytic fungi isolated from Mussaenda luteola. J. Appl. Pharm. Sci. 2017, 7, 234–238. [Google Scholar]
- Meenambiga, S.; Rajagopal, K. Antibiofilm activity and molecular docking studies of bioactive secondary metabolites from endophytic fungus Aspergillus nidulans on oral Candida albicans. J. Appl. Pharm. Sci. 2018, 8, 037–045. [Google Scholar]


| Extracellular (Ethyl Acetate) Extract | Intracellular (Methanol Mat) Extract | ||||||
|---|---|---|---|---|---|---|---|
| Gaminicolous helminthosporium | Bipolaris australiensis | Cladosporium cladosporioides | Gaminicolous helminthosporium | Bipolaris australiensis | Cladosporium cladosporioides | ||
| Peak Range | Peak Range | Peak Range | Peak Range | Peak Range | Peak Range | Group | Compound |
| 517.85 | 517.85 | 456.13 | 516.89 | 516.89 | 516.89 | C-I stretching | halo compound |
| 580.53 | 547.75 | 517.85 | 596.93 | 595.96 | 593.07 | C-I stretching | halo compound |
| 635.50 | 592.11 | 592.11 | 640.48 | 648.04 | 648.04 | C-I stretching | halo compound |
| 692.40 | 636.47 | 635.50 | 678.90 | 679.89 | 678.90 | C-H bending | Monosubstituted |
| 806.19 | - | - | - | - | 874.66 | C=C bending | Alkene |
| 947.95 | 814.87 | - | - | 1007.74 | 940.23 | N-O stretching | Oxidized Nitrogen |
| 1056.92 | 1055.95 | 1057.88 | 1081.03 | 1056.92 | 1008.70 | CO-O-CO stretching | Anhydride |
| 1171.68 | 1101.28 | - | 1148.53 | 1147.57 | 1055.95 | P=O stretching | phosphine oxide |
| 1243.04 | 1243.04 | 1243.04 | 1384.79 | 1384.79 | 1147.57 | C-N stretching | Amine |
| - | 1315.36 | 1312.26 | 1454.23 | 1315.36 | 1213.14 | S=O stretching | Sulfonamide |
| 1371.29 | 1338.51 | 1394.44 | 1338.51 | 1488.94 | 1398.30 | N-O stretching | nitro compound |
| 1644.20 | 1510.16 | 1510.16 | 1511.12 | 1589.23 | 1511.12 | C=C stretching | α,β-unsaturated ketone |
| 1679.88 | 1643.24 | - | - | - | 1605.63 | C=O stretching | tertiary amide |
| 1712.67 | 1712.67 | 1712.67 | 1717.49 | - | - | C=O stretching | carboxylic acid |
| 1767.64 | 1767.64 | 1759.92 | - | 1800.43 | - | N=C=S stretching | Isothiocyanate |
| 2314.42 | 2312.49 | 2360.71 | 2309.60 | 2309.60 | 2309.60 | O=C=O stretching | carbon dioxide |
| 2816.84 | 2882.42 | 2882.42 | 2884.35 | 2867.95 | 2868.92 | C-H stretching | Aldehyde |
| 2882.42 | 2980.78 | 2991.39 | 2981.74 | 2977.89 | 2942.21 | C-H stretching | Alkane |
| 2993.32 | 3290.33 | - | - | 3284.55 | 3336.62 | C-H stretching | Alkane |
| 3645.21 | 3588.32 | 3525.63 | - | - | - | N-H stretching | primary amine |
| 3746.47 | 3738.75 | 3739.72 | - | 3786.97 | 3723.32 | O-H stretching | Alcohol |
| Compounds | Extracellular (Ethyl Acetate) | Intracellular (Methanol Mat) | Application |
|---|---|---|---|
| Benzo [h] quinoline | Bipolaris australiensis, Cladosporium cladosporioides | Cladosporium cladosporioides | Wound healing, antibacterial, antioxidant, antimalarial, antiviral, anticonvulsant, anti-HIV |
| 2-Ethylacridine | Graminicolous helminthosporium, Bipolarisaustraliensis, Cladosporium cladosporioides | Bipolaris australiensis, Cladosporium cladosporioides | Antitumor Antioxidant |
| Cyclotrisiloxane | Graminicolous helminthosporium, Bipolaris australiensis, Cladosporium cladosporioides | Graminicolou shelminthosporium, Bipolaris australiensis, Cladosporium cladosporioides | Antibacterial, antiinflammatory Anticancer |
| Anthracene | Bipolaris australiensis, Cladosporium cladosporioides | - | Insecticidal, wood preservative, used as a preparation of cool-tar, fossil fuel, cosmetic, anticancer, antibacterial, anti-inflammatory |
| 5-methyl-2-phenylindolizine | - | Graminicolous helminthosporium, Bipolaris australiensis | Inhibitory act against TB |
| Indole-2-one | Graminicolous helminthosporium, Bipolaris australiensis | - | Enrich metabolism Anticancer Antiviral |
| Tris(tert-butyldimethylsilyloxy)arsane | Graminicolous helminthosporium, Cladosporium cladosporioides | Graminicoloushelminthosporium, Bipolarisaustraliensis, Cladosporium cladosporioides | Antioxidant Antibacterial Antifungal |
| 3-quinolinecarboxylic acid | Graminicolous helminthosporium | - | Antibacterial Antifungal |
| Benzene | Graminicolous helminthosporium, Bipolaris australiensis | - | Industrial manufacturing dyes, lubricants, drugs, fuel |
| n-Hexadeconic acid | Bipolaris australiensis | - | Antiinflammatory antioxidant, anticancer, Antinociceptive, cytotoxic compound |
| Oleic acid, Oxacyclotetradecan-2-one, Cis-Vaccenic acid | Bipolaris australiensis | - | Emulsifying agent, anticancer effect, reduces blood pressure, Reduces coronary heart disease (CHD), Antioxidant and antipolymerization agents, Used in asthma inhalers. |
| Oxacyclotetradecan-2-one | Bipolaris australiensis | - | Antibacterial activity |
| Cis-Vaccenic acid | Bipolaris australiensis | - | Antibacterial reduce sickle cell, reduction in the risk of coronary heart disease (CHD) |
| Tetrasiloxane | Bipolaris australiensis | Bipolaris australiensis, Cladosporium cladosporioides | Anticancer |
| Extracellular | Intracellular | |||||
|---|---|---|---|---|---|---|
| Cladosporium cladosporioides | Bipolaris australiensis | Graminicolous helminthosporium | Cladosporium cladosporioides | Bipolaris australiensis | Graminicolous helminthosporium | |
| Flavonoid | +++ | +++ | +++ | +++ | +++ | +++ |
| Saponin | ++ | - - | + | ++ | ++ | + |
| Tannin | ++ | - - | - - | +++ | +++ | +++ |
| Carbohydrate | - - | - - | - - | ++ | +++ | ++ |
| Phenol | +++ | +++ | +++ | + | + | +++ |
| Terpenoid | - - | +++ | +++ | + | ++ | ++ |
| Concentration of Samples (µL) | C. cladosporioides | G. helminthosporium | B. australiensis | |||
|---|---|---|---|---|---|---|
| Absorption Value (OD) | % of Scavenging | Absorption Value (OD) | % of Scavenging | Absorption Value (OD) | % of Scavenging | |
| 10 | 0.2 | 68.99 c | 0.209 | 67.6 c | 0.195 | 69.76 c |
| 30 | 0.195 | 69.76 b | 0.192 | 70.23 b | 0.192 | 70.23 b |
| 50 | 0.192 | 70.23 a | 0.187 | 71 a | 0.187 | 71 a |
| Concentration of Samples (µL) | C. cladosporioides | G. helminthosporium | B. australiensis | |||
|---|---|---|---|---|---|---|
| Absorption Value (OD) | % of Scavenging | Absorption Value (OD) | % of Scavenging | Absorption Value (OD) | % of Scavenging | |
| 10 | 0.187 | 70.01 c | 0.196 | 69.61 c | 0.132 | 79.53 c |
| 30 | 0.167 | 74.11 b | 0.189 | 70.7 b | 0.105 | 83.72 b |
| 50 | 0.147 | 77.21 a | 0.182 | 71.78 a | 0.026 | 95.97 a |
| Concentration of Samples (µL) | C. cladosporioides | G. helminthosporium | B. australiensis | |||
|---|---|---|---|---|---|---|
| Absorption Value (OD) | % of Scavenging | Absorption Value (OD) | % of Scavenging | Absorption Value (OD) | % of Scavenging | |
| 10 | 0.123 | 93.35 b | 0.17 | 90.82 c | 0.167 | 90.98 c |
| 30 | 0.092 | 95.01 a | 0.118 | 93.63 b | 0.091 | 95.03 b |
| 50 | 0.087 | 95.31 a | 0.06 | 96.76 a | 0.058 | 96.87 a |
| Concentration of Samples (µL) | C. cladosporioides | G. helminthosporium | B. australiensis | |||
|---|---|---|---|---|---|---|
| Absorption Value (OD) | % of Scavenging | Absorption Value (OD) | % of Scavenging | Absorption Value (OD) | % of Scavenging | |
| 10 | 0.266 | 85.63 c | 0.255 | 86.23 b | 0.273 | 85.25 c |
| 30 | 0.213 | 88.49 b | 0.203 | 29.03 c | 0.194 | 89.52 b |
| 50 | 0.107 | 94.23 a | 0.161 | 91.3 a | 0.15 | 91.9 a |
| Concentration of Samples (µL) | C. cladosporioides | G. helminthosporium | B. australiensis | |||
|---|---|---|---|---|---|---|
| Absorption Value (OD) | % of Scavenging | Absorption Value (OD) | % of Scavenging | Absorption Value (OD) | % of Scavenging | |
| 10 | 0.065 | 86.82 c | 0.071 | 85.6 c | 0.068 | 86.21 b |
| 30 | 0.061 | 87.63 b | 0.065 | 86.82 b | 0.064 | 87.01 a |
| 50 | 0.059 | 88.03 a | 0.063 | 87.22 a | 0.061 | 87.63 a |
| Concentration of Samples (µL) | C. cladosporioides | G. helminthosporium | B. australiensis | |||
|---|---|---|---|---|---|---|
| Absorption Value (OD) | % of Scavenging | Absorption Value (OD) | % of Scavenging | Absorption Value (OD) | % of Scavenging | |
| 10 | 0.273 | 44.62 c | 0.193 | 60.85 c | 0.32 | 35.1 c |
| 30 | 0.239 | 51.52 b | 0.073 | 85.19 a | 0.141 | 71.4 b |
| 50 | 0.065 | 86.82 a | 0.08 | 83.77 b | 0.092 | 81.34 a |
| Ligand | E min |
|---|---|
| Benzo [h] quinolone | −213.20 |
| Cyclotrisiloxane | −146.53 |
| Ethylacridine | −213.24 |
| Anthracene | −199.45 |
| 5 methyl 2 phenylindolizine | −224.85 |
| Brallobarbital | −227.89 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muthukrishnan, S.; Prakathi, P.; Sivakumar, T.; Thiruvengadam, M.; Jayaprakash, B.; Baskar, V.; Rebezov, M.; Derkho, M.; Zengin, G.; Shariati, M.A. Bioactive Components and Health Potential of Endophytic Micro-Fungal Diversity in Medicinal Plants. Antibiotics 2022, 11, 1533. https://doi.org/10.3390/antibiotics11111533
Muthukrishnan S, Prakathi P, Sivakumar T, Thiruvengadam M, Jayaprakash B, Baskar V, Rebezov M, Derkho M, Zengin G, Shariati MA. Bioactive Components and Health Potential of Endophytic Micro-Fungal Diversity in Medicinal Plants. Antibiotics. 2022; 11(11):1533. https://doi.org/10.3390/antibiotics11111533
Chicago/Turabian StyleMuthukrishnan, Sundaram, Paranivasakam Prakathi, Thangavel Sivakumar, Muthu Thiruvengadam, Bindhu Jayaprakash, Venkidasamy Baskar, Maksim Rebezov, Marina Derkho, Gokhan Zengin, and Mohammad Ali Shariati. 2022. "Bioactive Components and Health Potential of Endophytic Micro-Fungal Diversity in Medicinal Plants" Antibiotics 11, no. 11: 1533. https://doi.org/10.3390/antibiotics11111533
APA StyleMuthukrishnan, S., Prakathi, P., Sivakumar, T., Thiruvengadam, M., Jayaprakash, B., Baskar, V., Rebezov, M., Derkho, M., Zengin, G., & Shariati, M. A. (2022). Bioactive Components and Health Potential of Endophytic Micro-Fungal Diversity in Medicinal Plants. Antibiotics, 11(11), 1533. https://doi.org/10.3390/antibiotics11111533










