From Hollow to Solid Carbon Spheres: Time-Dependent Facile Synthesis
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of Mesoporous Silica Spheres
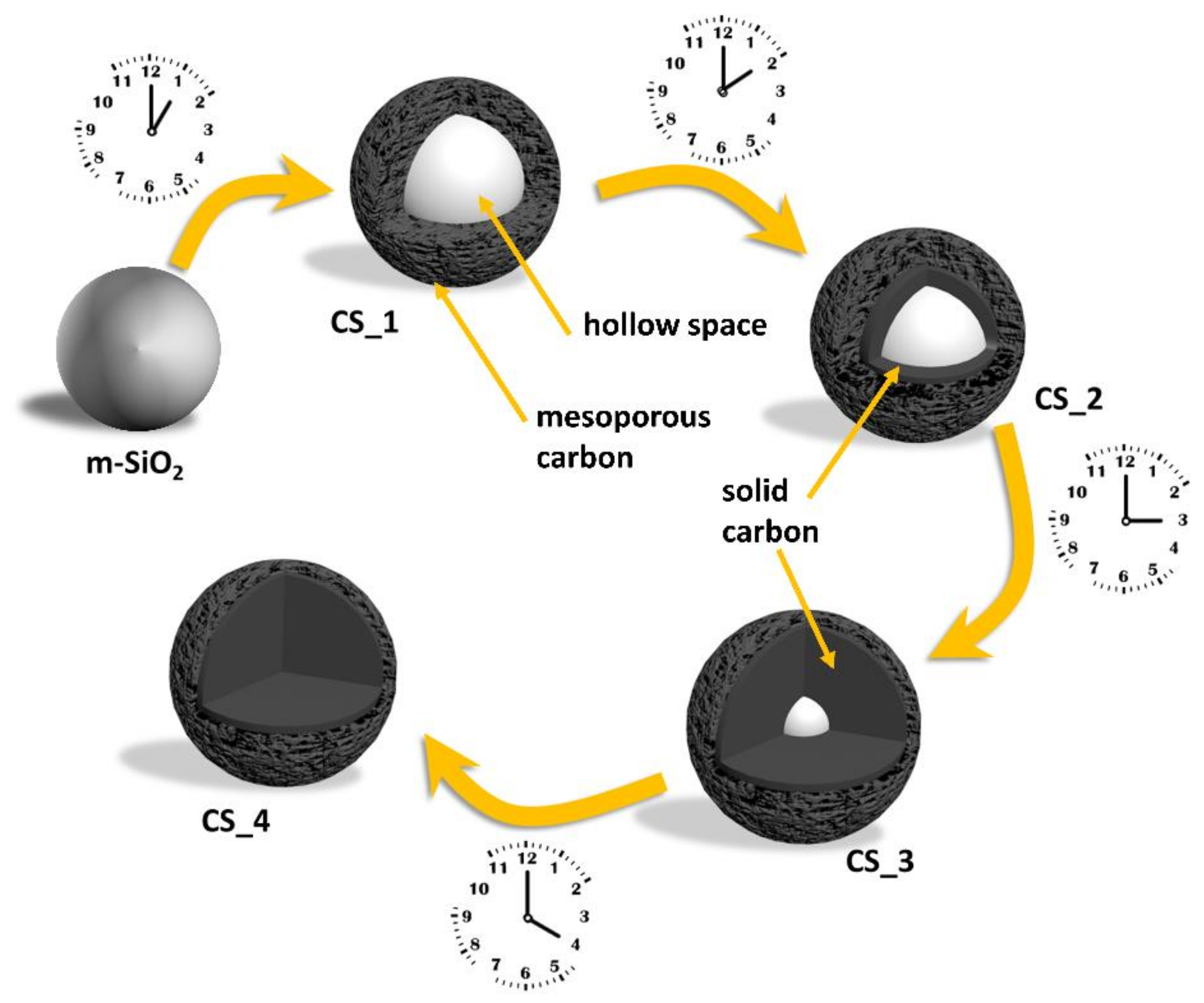
2.2. Synthesis of Carbon Spheres with Different Shell Thickness (from Hollow to Solid Carbon Spheres)
2.3. Characterization
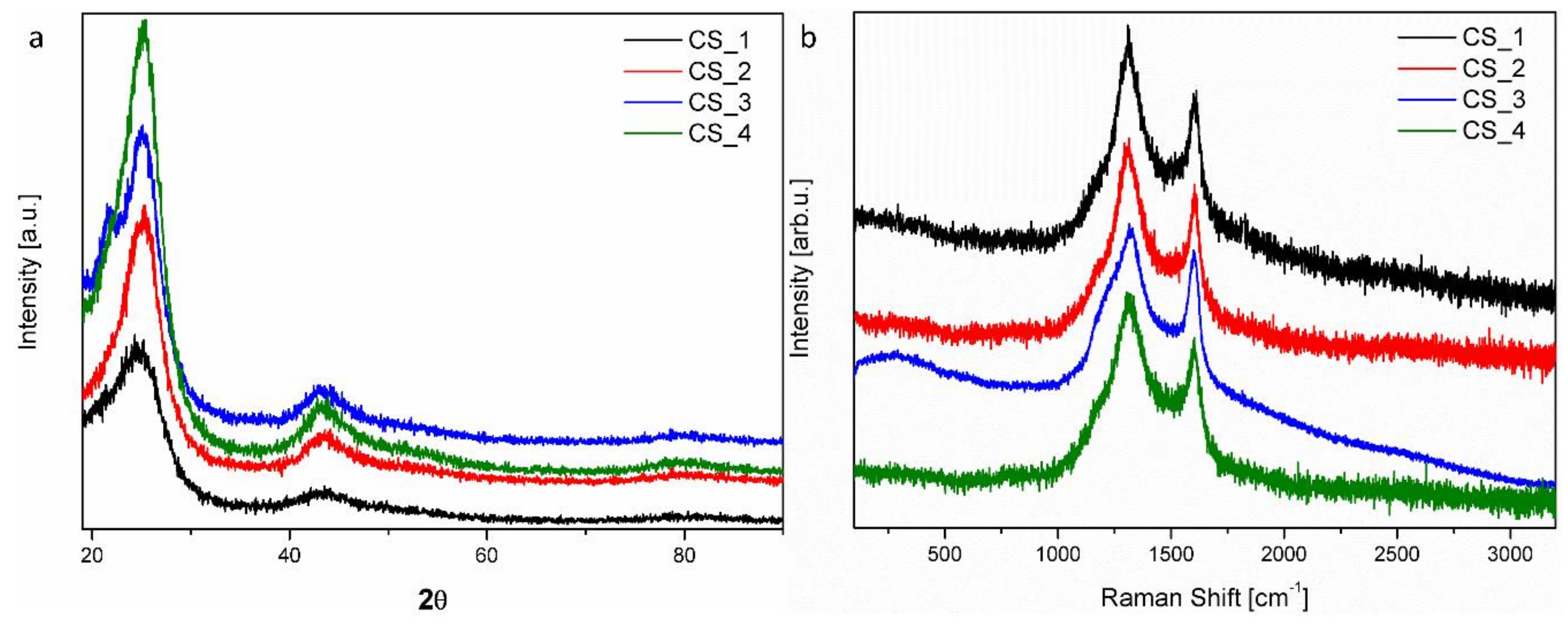
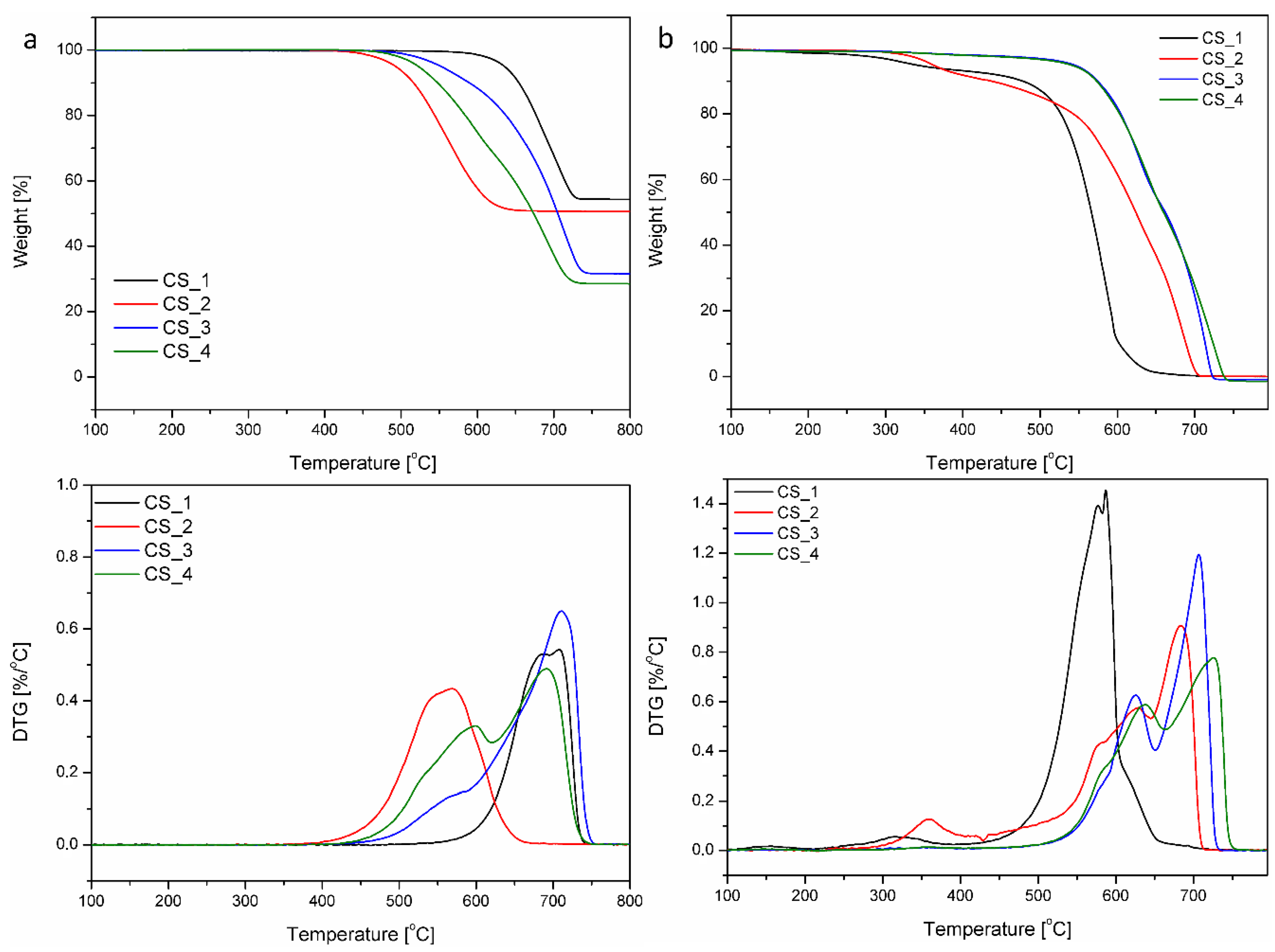
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, Q.; Yan, J.; Wang, Y.; Wei, T.; Zhang, M.; Jing, X.; Fan, Z. Three-dimensional flower-like and hierarchical porous carbon materials as high-rate performance electrodes for supercapacitors. Carbon 2014, 67, 119–127. [Google Scholar] [CrossRef]
- Li, Q.; Jiang, R.; Dou, Y.; Wu, Z.; Huang, T.; Feng, D.; Yang, J.; Yu, A.; Zhao, D. Synthesis of mesoporous carbon spheres with a hierarchical pore structure for the electrochemical double-layer capacitor. Carbon 2011, 49, 1248–1257. [Google Scholar] [CrossRef]
- Wang, Z.L.; Kang, Z.C. Pairing of pentagonal and heptagonal carbon rings in the growth of nanosize carbon spheres synthesized by a mixed-valent oxide-catalytic carbonization process. J. Phys. Chem. 1996, 100, 17725–17731. [Google Scholar] [CrossRef]
- Wang, Z.L.; Kang, Z.C. Graphitic structure and surface chemical activity of nanosize carbon spheres. Carbon 1997, 35, 419–426. [Google Scholar] [CrossRef]
- Sharon, M.; Mukhopadhyay, K.; Yase, K.; Iijima, S.; Ando, Y.; Zhao, X. Spongy carbon nanobeads—A new material. Carbon 1998, 36, 507–511. [Google Scholar] [CrossRef]
- Lou, X.W.; Li, C.M.; Archer, L.A. Designed synthesis of coaxial SnO2@carbon hollow nanospheres for highly reversible lithium storage. Adv. Mater. 2009, 21, 2536–2539. [Google Scholar] [CrossRef]
- Kim, S.; Shibata, E.; Sergiienko, R.; Nakamura, T. Purification and separation of carbon nanocapsules as a magnetic carrier for drug delivery systems. Carbon 2008, 46, 1523–1529. [Google Scholar] [CrossRef]
- Ma, Y.; Hu, Z.; Huo, K.; Lu, Y.; Hu, Y.; Liu, Y.; Hu, J.; Chen, Y. A practical route to the production of carbon nanocages. Carbon 2005, 43, 1667–1672. [Google Scholar] [CrossRef]
- Chen, X.; Kierzek, K.; Cendrowski, K.; Pelech, I.; Zhao, X.; Feng, J.; Kalenczuk, R.J.; Tang, T.; Mijowska, E. CVD generated mesoporous hollow carbon spheres as supercapacitors. Colloids Surf. A 2012, 396, 246–250. [Google Scholar] [CrossRef]
- Lazzara, G.; Cavallaro, G.; Panchal, A.; Fakhrullin, R.; Stavitskaya, A.; Vinokurov, V.; Lvov, Y. An assembly of organic-inorganic composites using halloysite clay nanotubes. Curr. Opin. Colloid Interface Sci. 2018, 35, 42–50. [Google Scholar] [CrossRef]
- Hu, J.; Chen, M.; Fang, X.; Wu, L. Fabrication and application of inorganic hollow spheres. Chem. Soc. Rev. 2011, 40, 5472–5491. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.B.; Sohn, K.; Kim, J.Y.; Shin, C.-H.; Yu, J.-S.; Hyeon, T. Fabrication of carbon capsules with hollow macroporous core/mesoporous shell structures. Adv. Mater. 2002, 14, 19–21. [Google Scholar] [CrossRef]
- Xia, Y.; Mokaya, R. Ordered mesoporous carbon hollow spheres nanocast using mesoporous silica via chemical vapor deposition. Adv. Mater. 2004, 16, 886–891. [Google Scholar] [CrossRef]
- Liang, C.; Li, Z.; Dai, S. Mesoporous carbon materials: Synthesis and modification. Angew. Chem. Int. Ed. 2008, 47, 3696–3717. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Rubner, M.F.; Cohen, R.E. Formation of nanoparticle-loaded microcapsules based on hydrogen-bonded multilayers. Chem. Mater. 2005, 17, 1099–1105. [Google Scholar] [CrossRef]
- Gill, I.; Ballesteros, A. Encapsulation of biologicals within silicate, siloxane, and hybrid sol-gel polymers: An efficient and generic approach. J. Am. Chem. Soc. 1998, 120, 8587–8598. [Google Scholar] [CrossRef]
- Liu, H.J.; Cui, W.J.; Jin, L.H.; Wang, C.X.; Xia, Y.Y. Preparation of three-dimensional ordered mesoporous carbon sphere arrays by a two-step templating route and their application for supercapacitors. J. Mater. Chem. 2009, 19, 3661–3667. [Google Scholar] [CrossRef]
- Huang, X.; Kim, S.; Heo, M.S.; Kim, J.E.; Suh, H.; Kim, I. Easy synthesis of hierarchical carbon spheres with superior capacitive performance in supercapacitors. Langmuir 2013, 29, 12266–12274. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Feng, S.; Song, Y.; Li, W.; Gao, W.; Elzatahry, A.A.; Aldhayan, D.; Xia, Y.; Zhao, D. Synthesis of hierarchically porous carbon spheres with yolk-shell structure for high performance supercapacitors. Catal. Today 2015, 243, 199–208. [Google Scholar] [CrossRef]
- Demir Cakan, R.; Titirici, M.-M.; Antonietti, M.; Cui, G.; Maier, J.; Hu, Y.-S. Hydrothermal carbon spheres containing silicon nanoparticles: Synthesis and lithium storage performance. Chem. Commun. 2008, 3759. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.-M.; Hu, J.-S.; Guo, Y.-G.; Zheng, S.-F.; Zhong, L.-S.; Song, W.-G.; Wan, L.-J. Tin-nanoparticles encapsulated in elastic hollow carbon spheres for high-performance anode material in lithium-Ion batteries. Adv. Mater. 2008, 20, 1160–1165. [Google Scholar] [CrossRef]
- Zheng, G.; Lee, S.W.; Liang, Z.; Lee, H.-W.; Yan, K.; Yao, H.; Wang, H.; Li, W.; Chu, S.; Cui, Y. Interconnected hollow carbon nanospheres for stable lithium metal anodes. Nat. Nanotechnol. 2014, 9, 618–623. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, J.; Yang, S.; Sun, Q.; Zhu, L.; Wu, Q.; Zhang, H.; Meng, X.; Xiao, F.-S. Sulfonated hollow sphere carbon as an efficient catalyst for acetalisation of glycerol. J. Mater. Chem. A 2013, 1, 9422. [Google Scholar] [CrossRef]
- Bian, X.; Zhu, J.; Liao, L.; Scanlon, M.D.; Ge, P.; Ji, C.; Girault, H.H.; Liu, B. Nanocomposite of MoS2 on ordered mesoporous carbon nanospheres: A highly active catalyst for electrochemical hydrogen evolution. Electrochem. Commun. 2012, 22, 128–132. [Google Scholar] [CrossRef]
- Wickramaratne, N.P.; Jaroniec, M. Importance of small micropores in CO2 capture by phenolic resin-based activated carbon spheres. J. Mater. Chem. A 2013, 1, 112–116. [Google Scholar] [CrossRef]
- Wickramaratne, N.P.; Jaroniec, M. Activated carbon spheres for CO2 adsorption. ACS Appl. Mater. Interfaces 2013, 5, 1849–1855. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Zheng, G.; Yang, J.; Tang, H.; Zhang, Y.; Kong, B.; Lv, Y.; Xu, C.; Asiri, A.M.; Zi, J.; et al. Dual-pore mesoporous carbon@silica composite core-shell nanospheres for multidrug delivery. Angew. Chem. Int. Ed. 2014, 53, 5366–5370. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xu, P.; Wu, M.; Meng, Q.; Chen, H.; Shu, Z.; Wang, J.; Zhang, L.; Li, Y.; Shi, J. Colloidal RBC-shaped, hydrophilic, and hollow mesoporous carbon nanocapsules for highly efficient biomedical engineering. Adv. Mater. 2014, 26, 4294–4301. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Hu, Z.; Xu, J.; Zhao, Y. Therapeutic applications of low-toxicity spherical nanocarbon materials. NPG Asia Mater. 2014, 6, e84. [Google Scholar] [CrossRef]
- Fu, J.; Xu, Q.; Chen, J.; Chen, Z.; Huang, X.; Tang, X. Controlled fabrication of uniform hollow core porous shell carbon spheres by the pyrolysis of core/shell polystyrene/cross-linked polyphosphazene composites. Chem. Commun. 2010, 46, 6563–6565. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Yin, H.; Wang, A.; Wu, Z.; Wu, G.; Jiang, T.; Shen, Y.; Jiang, T. Size-controlled preparation of hollow silica spheres and glyphosate release. Trans. Nonferrous Met. Soc. China 2012, 22, 1161–1168. [Google Scholar] [CrossRef]
- Yang, S.; Feng, X.; Zhi, L.; Cao, Q.; Maier, J.; Müllen, K. Nanographene-constructed hollow carbon spheres and their favorable electroactivity with respect to lithium storage. Adv. Mater. 2010, 22, 838–842. [Google Scholar] [CrossRef] [PubMed]
- Zielinska, B.; Michalkiewicz, B.; Mijowska, E.; Kalenczuk, R.J. Advances in Pd nanoparticle size decoration of mesoporous carbon spheres for energy application. Nanoscale Res. Lett. 2015, 10, 430. [Google Scholar] [CrossRef] [PubMed]
- Wickström, H.; Hilgert, E.; Nyman, J.; Desai, D.; Şen Karaman, D.; de Beer, T.; Sandler, N.; Rosenholm, J. Inkjet printing of drug-loaded mesoporous silica nanoparticles—A platform for drug development. Molecules 2017, 22, 2020. [Google Scholar] [CrossRef] [PubMed]
- Cavallaro, G.; Lazzara, G.; Milioto, S. Dispersions of nanoclays of different shapes into aqueous and solid biopolymeric matrices. extended physicochemical study. Langmuir 2011, 27, 1158–1167. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Hrymak, A.N.; Kamal, M.R. Effect of hybrid carbon fillers on the electrical and morphological properties of polystyrene nanocomposites in microinjection molding. Nanomaterials 2018, 8, 779–793. [Google Scholar] [CrossRef] [PubMed]
- Cavallaro, G.; Milioto, S.; Parisi, F.; Lazzara, G. Halloysite nanotubes loaded with calcium hydroxide: Alkaline fillers for the deacidification of waterlogged archeological woods. ACS Appl. Mater. Interfaces 2018, 10, 27355–27364. [Google Scholar] [CrossRef] [PubMed]
- De Cicco, D.; Asaee, Z.; Taheri, F. Use of nanoparticles for enhancing the interlaminar properties of fiber-reinforced composites and adhesively bonded joints—A review. Nanomaterials 2017, 7, 360. [Google Scholar] [CrossRef] [PubMed]
- Wilgosz, K.; Chen, X.; Kierzek, K.; Machnikowski, J.; Kalenczuk, R.J.; Mijowska, E. Template method synthesis of mesoporous carbon spheres and its applications as supercapacitors. Nanoscale Res. Lett. 2012, 7, 269. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Alsmeyer, D.C.; Mccreery, R.L. Raman spectroscopy of carbon materials: Structural basis of observed spectra. Carbon 1990, 2, 557–563. [Google Scholar] [CrossRef]
- Wang, J.; Liu, Q. An ordered mesoporous aluminosilicate oxynitride template to prepare N-incorporated ordered mesoporous carbon. J. Phys. Chem. C 2007, 111, 7266–7272. [Google Scholar] [CrossRef]
- Dunens, O.M.; Mackenzie, K.J.; Harris, A.T. Synthesis of multiwalled carbon nanotubes on fly ash derived catalysts. Environ. Sci. Technol. 2009, 43, 7889–7894. [Google Scholar] [CrossRef] [PubMed]
- Ajayan, P.M.; Ebbesen, T.W.; Ichihashi, T.; Iijima, S.; Tanigaki, K.; Hiura, H. Opening carbon nanotubes with oxygen and implications for filling. Nature 1993, 362, 522–525. [Google Scholar] [CrossRef]
- Rinzler, G.; Liu, J.; Dai, H.; Nikolaev, P.; Huffman, C.B.; Rodríguez-Macías, F.J.; Boul, P.J.; Lu, H.; Heymann, D.; Colbert, D.T.; et al. Large-scale purification of single-wall carbon nanotubes: Process, product, and characterization. Appl. Phys. A Mater. Sci. Process. 1998, 67, 29–37. [Google Scholar] [CrossRef]
- Hou, P.; Liu, C.; Tong, Y.; Xu, S.; Liu, M.; Cheng, H. Purification of single-walled carbon nanotubes synthesized by the hydrogen arc-discharge method. J. Mater. Res. 2001, 16, 2526–2529. [Google Scholar] [CrossRef]
- Lima, A.F.; Musumeci, A.; Liu, H.W.; Waclawik, E.; Silva, G. Purity evaluation and influence of carbon nanotubes on carbon nanotube/graphite thermal stability. J. Therm. Anal. Calorim. 2009, 97, 257–263. [Google Scholar] [CrossRef]


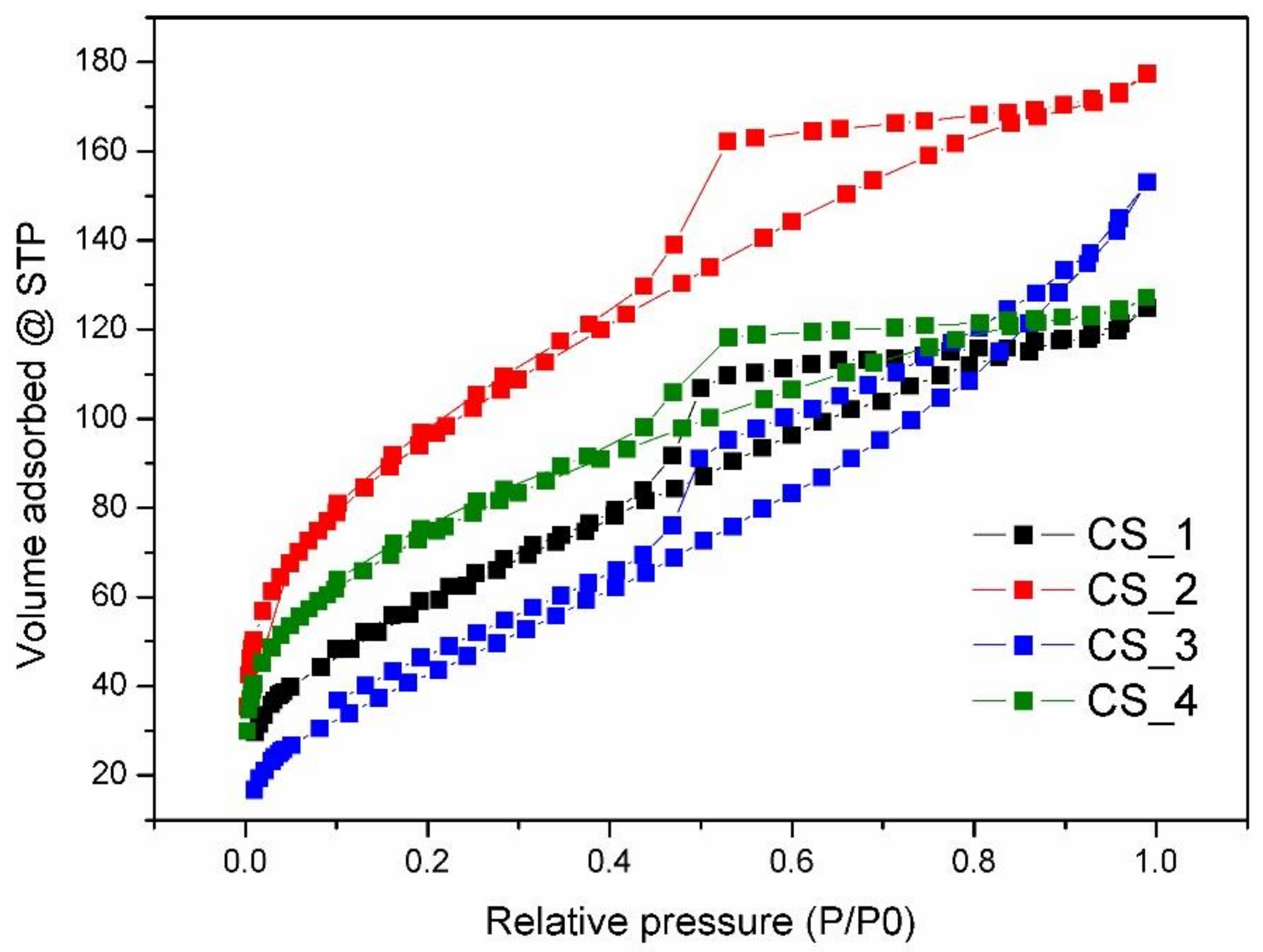
| Sample | SBET a (m2·g−1) | Vtotal b (cm3·g−1) | Pore Size c (nm) | Error d |
|---|---|---|---|---|
| m-SiO2 | 224.2 | 0.210 | 2.313 | 0.391% |
| CS_1 | 218.2 | 0.174 | 3.969 | 0.609% |
| CS_2 | 344.8 | 0.248 | 3.167 | 0.970% |
| CS_3 | 169.8 | 0.212 | 2.245 | 0.544% |
| CS_4 | 263.5 | 0.177 | 1.178 | 0.621% |
| Sample | Tstart (°C) | Tmax (°C) | Tend (°C) |
|---|---|---|---|
| CS_1 | 110 | 580 | 650 |
| CS_2 | 220 | 680 | 715 |
| CS_3 | 450 | 705 | 735 |
| CS_4 | 450 | 725 | 755 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kukułka, W.; Wenelska, K.; Baca, M.; Chen, X.; Mijowska, E. From Hollow to Solid Carbon Spheres: Time-Dependent Facile Synthesis. Nanomaterials 2018, 8, 861. https://doi.org/10.3390/nano8100861
Kukułka W, Wenelska K, Baca M, Chen X, Mijowska E. From Hollow to Solid Carbon Spheres: Time-Dependent Facile Synthesis. Nanomaterials. 2018; 8(10):861. https://doi.org/10.3390/nano8100861
Chicago/Turabian StyleKukułka, Wojciech, Karolina Wenelska, Martyna Baca, Xuecheng Chen, and Ewa Mijowska. 2018. "From Hollow to Solid Carbon Spheres: Time-Dependent Facile Synthesis" Nanomaterials 8, no. 10: 861. https://doi.org/10.3390/nano8100861
APA StyleKukułka, W., Wenelska, K., Baca, M., Chen, X., & Mijowska, E. (2018). From Hollow to Solid Carbon Spheres: Time-Dependent Facile Synthesis. Nanomaterials, 8(10), 861. https://doi.org/10.3390/nano8100861







