Upcycling Waste Lard Oil into Vertical Graphene Sheets by Inductively Coupled Plasma Assisted Chemical Vapor Deposition
Abstract
:1. Introduction
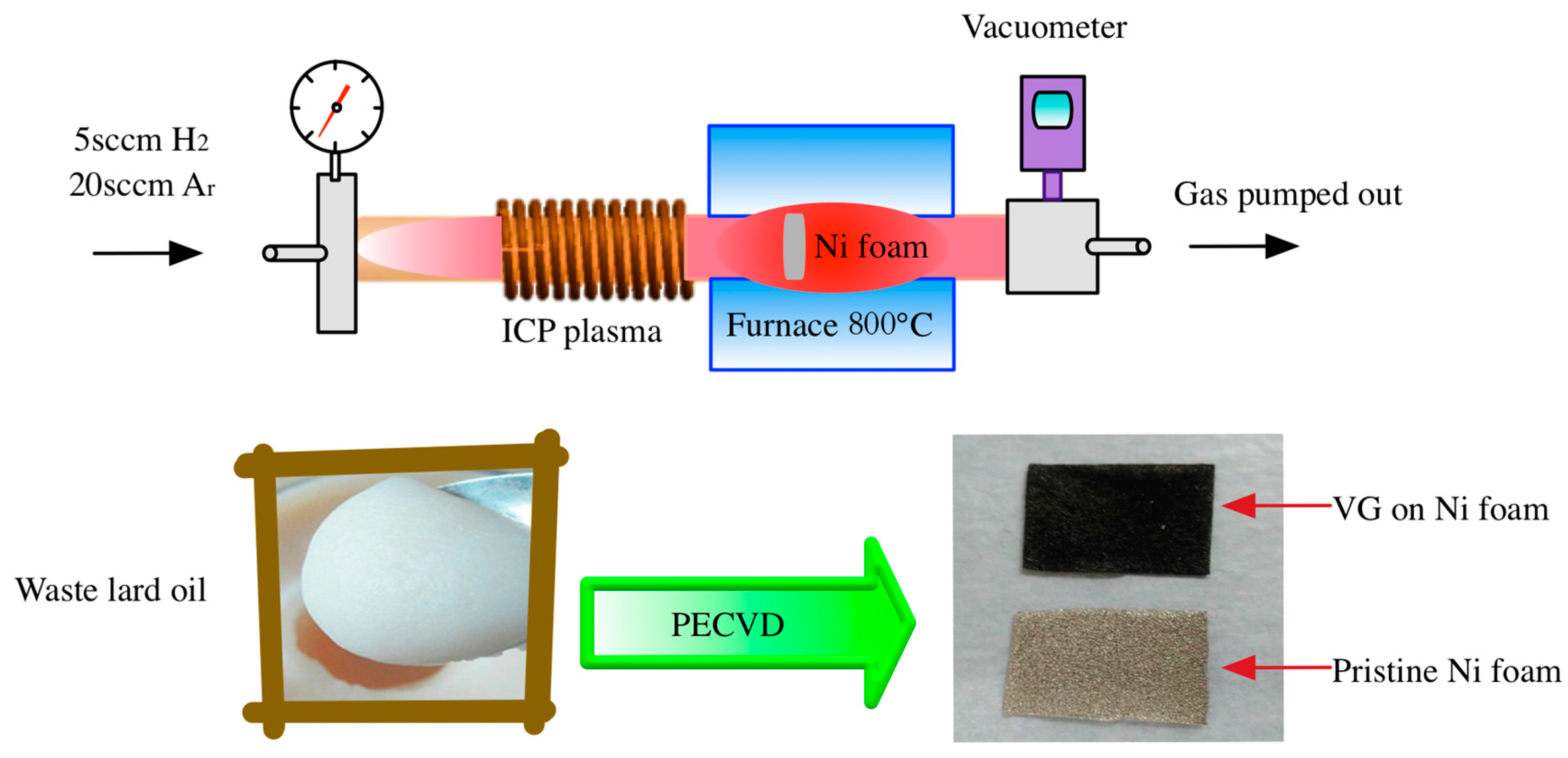
2. Materials and Methods
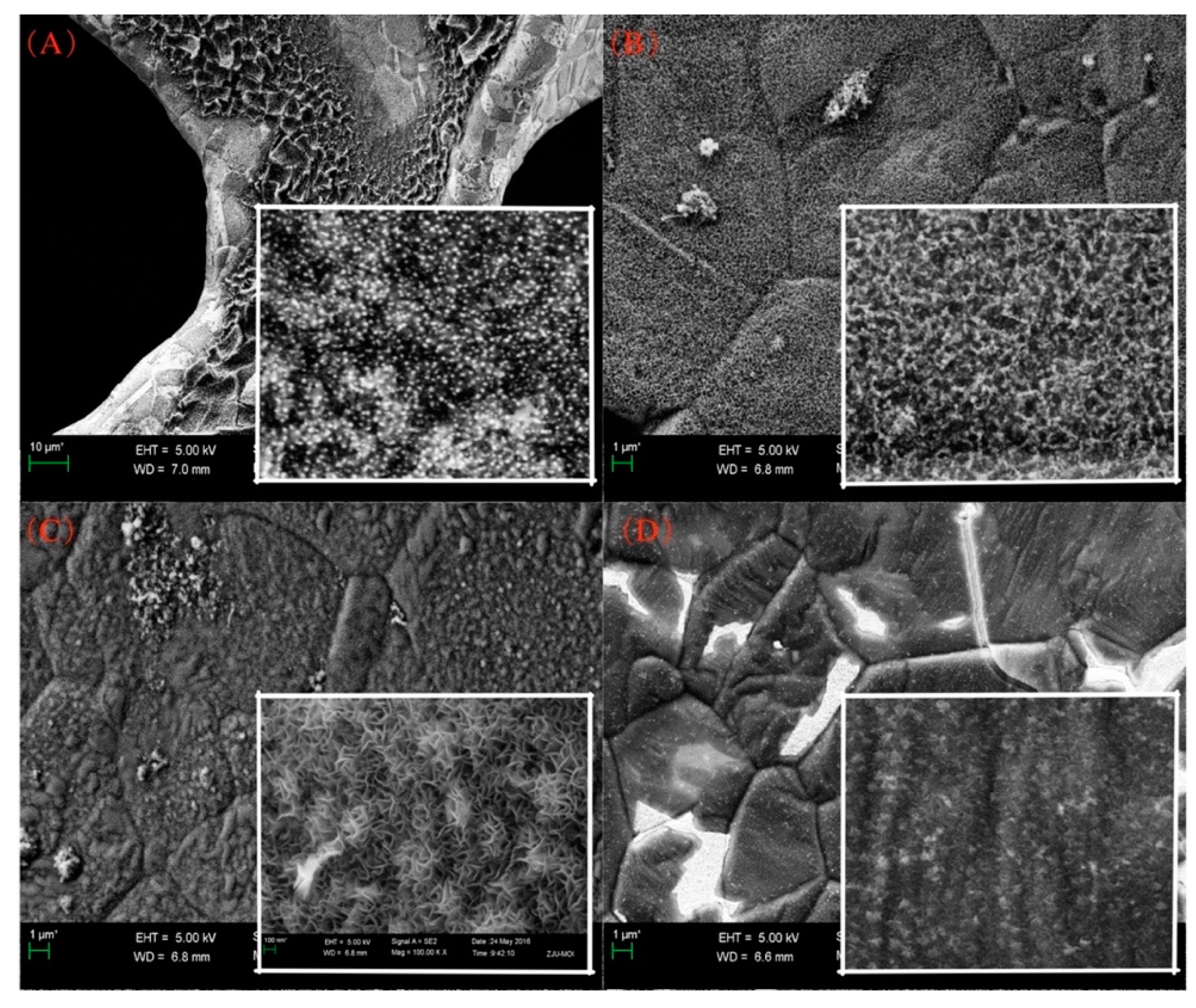
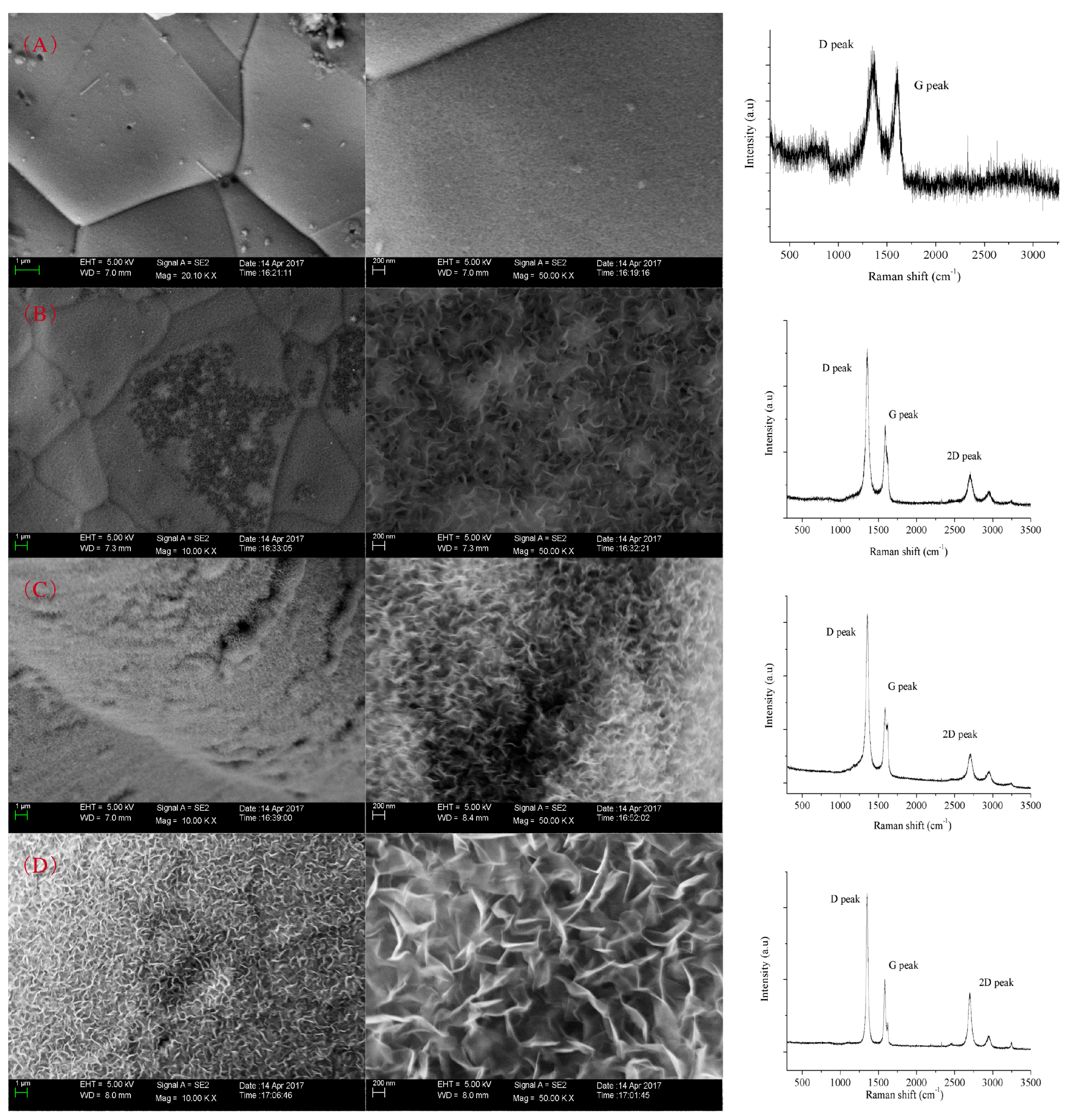
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Geim, A.K.; Novoselov, K. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, F.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Materials and methods: Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Seo, H.K.; Kim, T.S.; Park, C.; Xu, W.; Baek, K.; Baw, S.H.; Ahn, J.H.; Kim, K.; Choi, H.C.; Lee, T.W. Value-added synthesis of graphene: Recycling industrial carbon waste into electrodes for high-performance electronic device. Sci. Rep. 2015, 5, 16710. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.H.; Rider, A.E.; Kumar, S.; Randeniya, K.L.; Ostrikov, K. Vertical graphene gas- and bio-sensors via catalyst-free, reactive plasma reforming of natural honey. Carbon 2013, 60, 221–228. [Google Scholar] [CrossRef]
- Pumera, M. Graphene-based nanomaterials for energy storage. Energy Environ. Sci. 2011, 4, 668–674. [Google Scholar] [CrossRef]
- Bo, Z.; Yang, Y.; Chen, J.H.; Yu, K.H.; Yan, J.H.; Cen, K.F. Plasma-enhanced chemical vapor deposition synthesis of vertically oriented graphene nanosheets. Nanoscale 2013, 5, 5180–5204. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yuan, Q.; Li, J.; Ding, F. The transition metal surface dependent methane decomposition in graphene chemical vapor decomposition growth. Nanoscale 2017, 9, 11584–11589. [Google Scholar] [CrossRef] [PubMed]
- Pei, S.; Cheng, H.M. The reduction of graphene oxide. Carbon 2012, 50, 3210–3228. [Google Scholar] [CrossRef]
- Bazaka, K.; Jacob, M.V.; Ostrikov, K. Sustainable life cycles of natural-precursor-derived nanocarbons. Chem. Rev. 2016, 116, 163–214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prasad, K.; Bandara, C.D.; Kumar, S.; Singh, G.P.; Brockhoff, B.; Bazaka, K.; Ostrikov, K. Effect of precursor on antifouling efficacy of vertically-oriented graphene nanosheets. Nanomaterials 2017, 7, 170. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.J.; Li, X.D.; Yan, J.H.; Yang, J.; Du, C.M.; Zhu, F.S.; Qian, J.Y. Co-generation of hydrogen and carbon aerosol from coalbed methane surrogate using rotating gliding arc plasma. Appl. Energy 2017, 195, 67–79. [Google Scholar] [CrossRef]
- Seo, D.H.; Han, Z.J.; Kumar, S.; Ostrikov, K. Structure-controlled, vertical graphene-based, binder-free electrodes from plasma-reformed butter enhance supercapacitor performance. Adv. Energy Mater. 2013, 3, 1316–1323. [Google Scholar] [CrossRef]
- Ma, G.S.; Hao, L.N.; Li, X.P.; Hu, X.Q.; He, Y.N.; Zhai, F.Y.; Yang, X.G.; Kong, Y.Z. Cooking oil consumption of adults in China. Food Nutr. China 2008, 9, 30–32. [Google Scholar]
- Wang, Z.P.; Ogata, H.; Morimoto, S.; Medina, J.O.; Fujishige, M.; Takeuchi, K.; Muramatsu, H.; Hayashi, T.; Terrones, M.; Hashimoto, Y.; et al. Nanocarbons from rice husk by microwave plasma irradiation: From graphene and carbon nanotubes to graphenated carbon nanotube hybrids. Carbon 2015, 94, 479–484. [Google Scholar] [CrossRef]
- Wu, A.J.; Li, X.D.; Chen, L.; Du, C.M.; Yan, J.H. Investigation of the physical properties in rotating gliding arc discharge with rapeseed oil. IEEE Trans. Plasma Sci. 2015, 43, 3219–3223. [Google Scholar] [CrossRef]
- Wu, A.; Li, X.; Chen, L.; Zhu, F.; Zhang, H.; Du, C.; Yan, J. Utilization of waste rapeseed oil by rotating gliding arc plasma. Int. J. Hydrogen Energy 2015, 40, 9039–9048. [Google Scholar] [CrossRef]
- Tsaneva, V.N.; Kwapinski, W.; Teng, X.; Glowacki, B.A. Assessment of structural evaluation of carbons from microwave plasma natural gas reforming and biomass pyrolysis using Raman spectroscopy. Carbon 2014, 80, 617–628. [Google Scholar] [CrossRef]
- Jacob, M.V.; Rawat, R.S.; Bo, Q.Y.; Bazaka, K.; Kumar, D.S.; Taguchi, D.; Iwamoto, M.; Neupane, R.; Varghese, O.K. Catalyst-free plasma enhanced growth of graphene from sustainbale sources. Nano Lett. 2015, 15, 5702–5708. [Google Scholar] [CrossRef] [PubMed]
- Qu, B.; Lian, X.B.; Wu, Q.H. Growth of three-dimensional graphene films on the Ni foil. Surf. Eng. 2016, 32, 1–5. [Google Scholar] [CrossRef]
- Watanabe, H.; Kondo, H.; Sekine, M.; Hiramatsu, M.; Hori, M. Control of super hydrophobic and super hydrophilic surfaces of carbon nanowalls using atmospheric pressure plasma treatments. Jpn. J. Appl. Phys. 2012, 51, 109–117. [Google Scholar] [CrossRef]
- Seo, D.H.; Kumar, S.; Ostrikov, K. Control of morphology and electrical properties of self-organized graphenes in a plasma. Carbon 2011, 49, 4331–4339. [Google Scholar] [CrossRef]
- Wang, J.J.; Zhu, M.J.; Outlaw, R.A.; Zhao, X.; Manos, D.M.; Holloway, B.X. Synthesis of carbon nanosheets by inductively coupled radio-frequency plasma enhanced chemical vapor deposition. Carbon 2004, 42, 2867–2872. [Google Scholar] [CrossRef]
- Seo, D.H.; Yick, S.; Pineda, S.; Su, D.; Wang, G.; Han, Z.J.; Ostrikov, K. Single-step, plasma-enabled reforming of natural precursors into vertical graphene electrodes with high areal capacitance. ACS. Sustain. Chem. Eng. 2015, 3, 544–551. [Google Scholar] [CrossRef]
- Kurita, S.; Yoshimura, A.; Kawamoto, H.; Uchida, T.; Kojima, K.; Tachibana, M. Raman spectra of carbon nanowalls grown by plasma-enhanced chemical vapor deposition. J. Appl. Phys. 2005, 10, 104320. [Google Scholar] [CrossRef]
- Ni, Z.H.; Fan, H.M.; Feng, Y.P.; Shen, Z.X.; Yang, B.J.; Wu, Y.H. Raman spectroscopic investigation of carbon nanowalls. J. Chem. Phys. 2006, 124, 204703. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Qu, L.; Deng, J.H. High-efficiency field emission from pressed nickel foam-flat graphene-vertical graphene hybrids. Mater. Lett. 2016, 176, 165–168. [Google Scholar] [CrossRef]
- Idem, R.O.; Katikaneni, S.P.R.; Bakhshi, N.N. Thermal cracking of canola oil: Reaction products in the presence and absence of steam. Energy Fuel 1996, 10, 1150–1162. [Google Scholar] [CrossRef]
- Maher, K.; Bressler, D. Pyrolysis of triglyceride materials for the production of renewable fuels and chemicals. Bioresour. Technol. 2007, 98, 2351–2368. [Google Scholar] [CrossRef] [PubMed]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, A.; Li, X.; Yang, J.; Du, C.; Shen, W.; Yan, J. Upcycling Waste Lard Oil into Vertical Graphene Sheets by Inductively Coupled Plasma Assisted Chemical Vapor Deposition. Nanomaterials 2017, 7, 318. https://doi.org/10.3390/nano7100318
Wu A, Li X, Yang J, Du C, Shen W, Yan J. Upcycling Waste Lard Oil into Vertical Graphene Sheets by Inductively Coupled Plasma Assisted Chemical Vapor Deposition. Nanomaterials. 2017; 7(10):318. https://doi.org/10.3390/nano7100318
Chicago/Turabian StyleWu, Angjian, Xiaodong Li, Jian Yang, Changming Du, Wangjun Shen, and Jianhua Yan. 2017. "Upcycling Waste Lard Oil into Vertical Graphene Sheets by Inductively Coupled Plasma Assisted Chemical Vapor Deposition" Nanomaterials 7, no. 10: 318. https://doi.org/10.3390/nano7100318
APA StyleWu, A., Li, X., Yang, J., Du, C., Shen, W., & Yan, J. (2017). Upcycling Waste Lard Oil into Vertical Graphene Sheets by Inductively Coupled Plasma Assisted Chemical Vapor Deposition. Nanomaterials, 7(10), 318. https://doi.org/10.3390/nano7100318



