Ammonium Ion Enhanced V2O5-WO3/TiO2 Catalysts for Selective Catalytic Reduction with Ammonia
Abstract
:1. Introduction
2. Materials and Methods
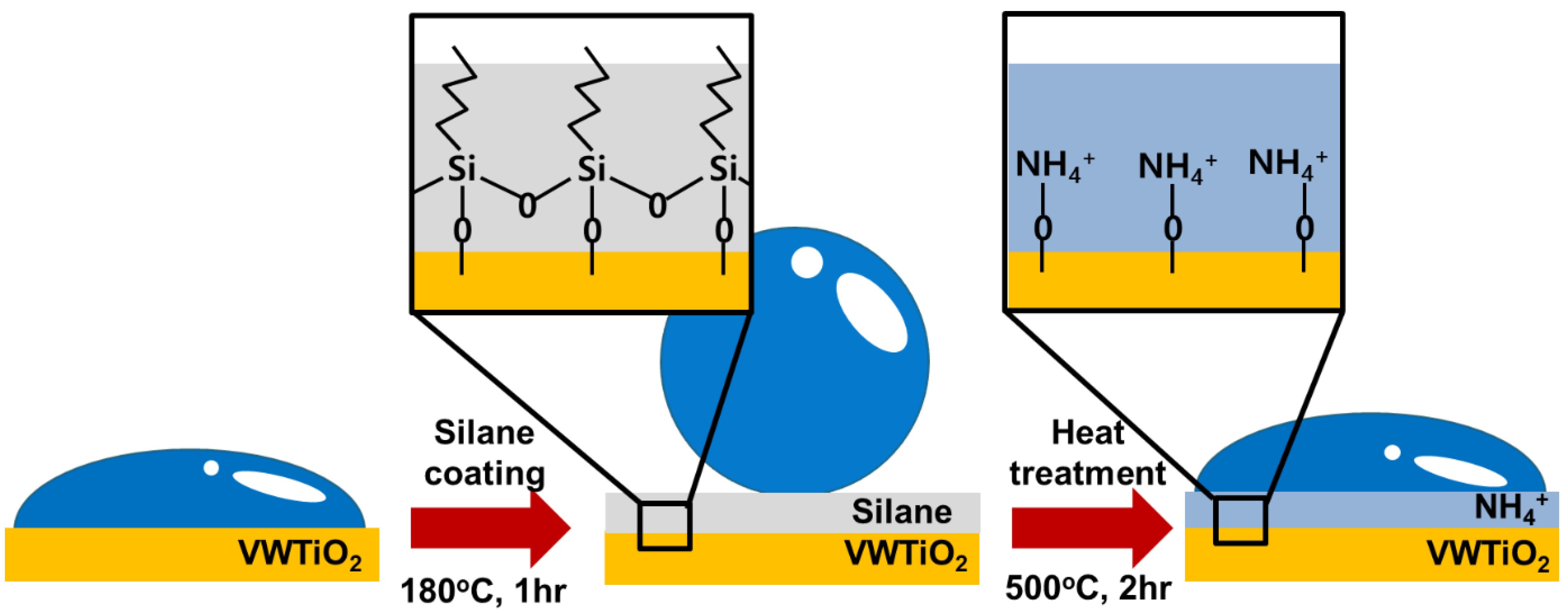
2.1. Catalyst Preparation
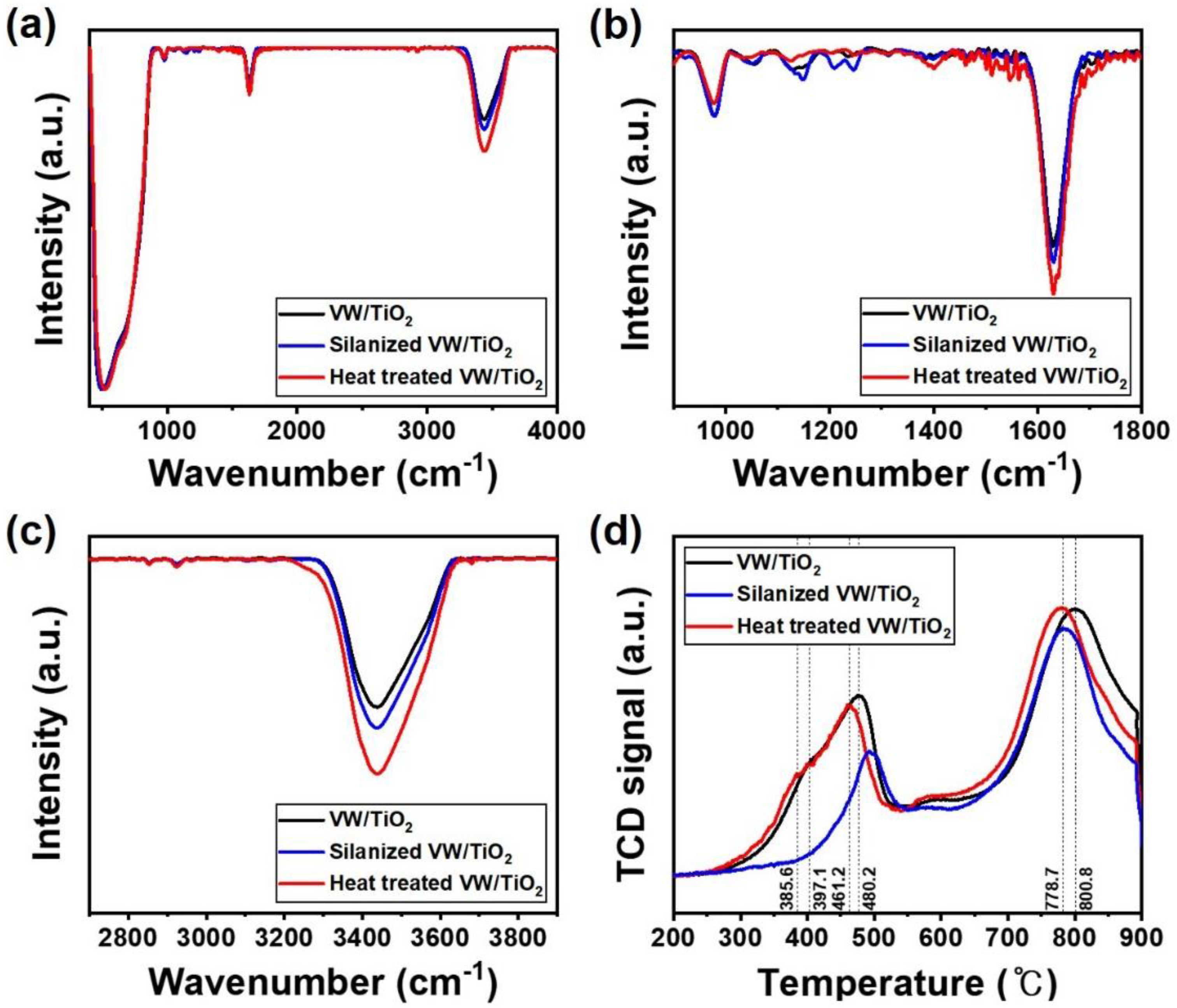
2.2. Catalyst Characterization
2.3. Catalytic Activity Measurement
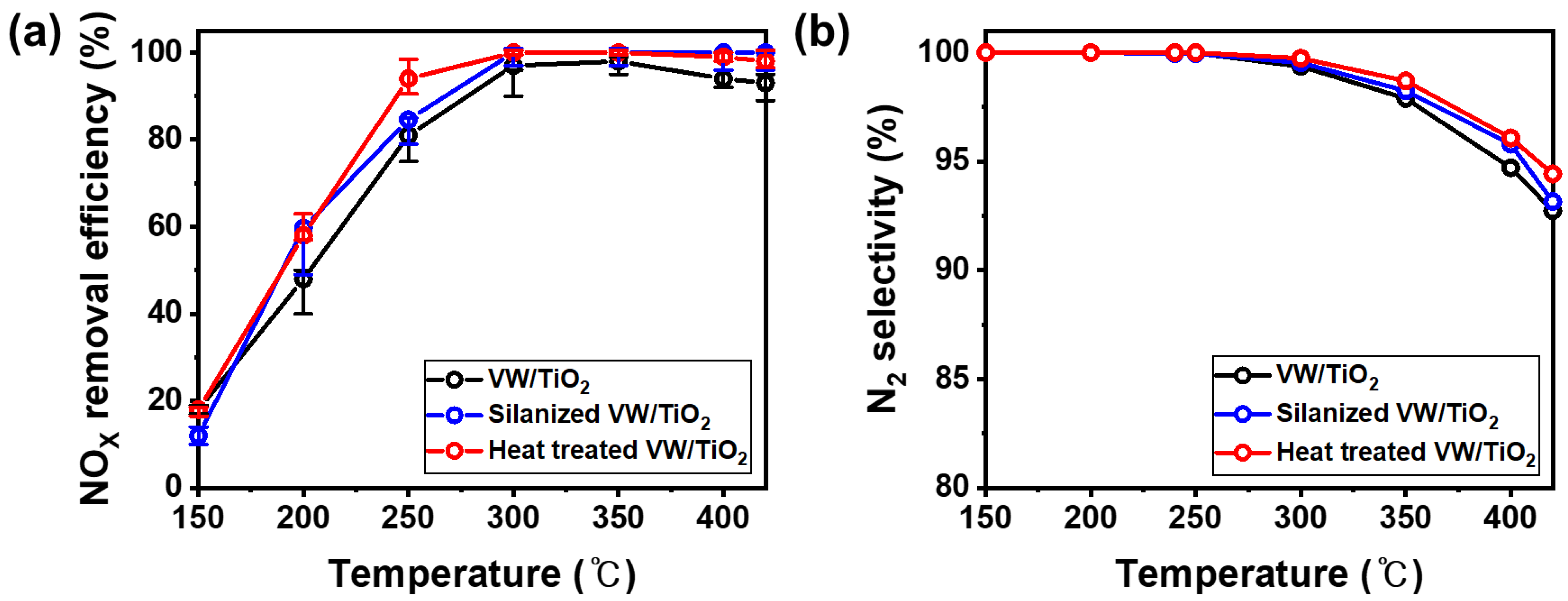
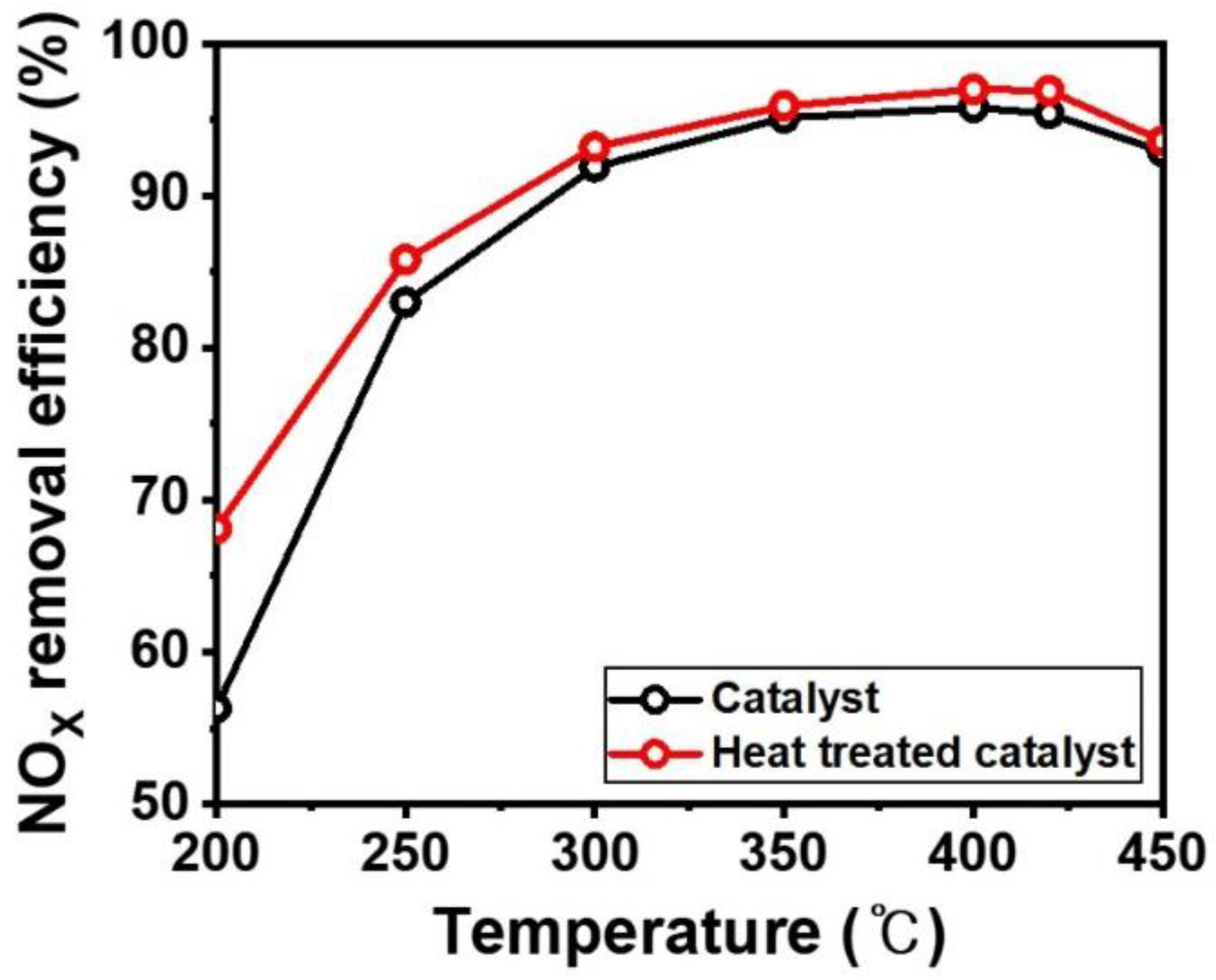
3. Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Damma, D.; Ettireddy, P.R.; Reddy, B.M.; Smirniotis, P.G. A review of low temperature NH3-SCR for removal of NOX. Catalysts 2019, 9, 349. [Google Scholar] [CrossRef] [Green Version]
- Lee, M.J.; Kim, D.H.; Lee, M.; Ye, B.; Jeong, B.; Lee, D.; Kim, H.D.; Lee, H. Enhanced NOX removal efficiency for SCR catalyst of well-dispersed Mn-Ce nanoparticles on hexagonal boron nitride. Environ. Sci. Pollut. Res. 2019, 26, 36107–36116. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, B.; Liu, B.; Sun, S. A review of Mn-containing oxide catalysts for low temperature selective catalytic reduction of NOX with NH3: Reaction mechanism and catalyst deactivation. RSC Adv. 2017, 7, 26226–26242. [Google Scholar] [CrossRef] [Green Version]
- Fu, M.; Li, C.; Lu, P.; Qu, L.; Zhang, M.; Zhou, Y.; Yu, M.; Fang, Y. A review on selective catalytic reduction of NOX by supported catalysts at 100–300 C—catalysts, mechanism, kinetics. Catal. Sci. Technol. 2014, 4, 14–25. [Google Scholar] [CrossRef]
- Ye, B.; Lee, M.; Jeong, B.; Kim, J.; Lee, D.H.; Baik, J.M.; Kim, H.D. Partially reduced graphene oxide as a support of Mn-Ce/TiO2 catalyst for selective catalytic reduction of NOX with NH3. Catal. Today 2019, 328, 300–306. [Google Scholar] [CrossRef]
- Lee, M.S.; Kim, S.I.; Lee, M.J.; Ye, B.; Kim, T.; Kim, H.D.; Lee, J.W.; Lee, D.H. Effect of Catalyst Crystallinity on V-Based Selective Catalytic Reduction with Ammonia. Nanomaterials 2021, 11, 1452. [Google Scholar] [CrossRef]
- Zhang, X.; Diao, Q.; Hu, X.; Wu, X.; Xiao, K.; Wang, J. Modification of V2O5-WO3/TiO2 Catalyst by Loading of MnOx for Enhanced Low-Temperature NH3-SCR Performance. Nanomaterials 2020, 10, 1900. [Google Scholar] [CrossRef] [PubMed]
- Jeong, B.; Ye, B.; Kim, E.-S.; Kim, H.-D. Characteristics of selective catalytic reduction (SCR) catalyst adding graphene-tungsten nanocomposite. Catal. Commun. 2017, 93, 15–19. [Google Scholar] [CrossRef]
- Lai, J.-K.; Wachs, I.E. A Perspective on the Selective Catalytic Reduction (SCR) of NO with NH3 by Supported V2O5-WO3/TiO2 Catalysts. ACS Catal. 2018, 8, 6537–6551. [Google Scholar] [CrossRef]
- Djerad, S.; Tifouti, L.; Crocoll, M.; Weisweiler, W. Effect of vanadia and tungsten loadings on the physical and chemical characteristics of V2O5-WO3/TiO2 catalysts. J. Mol. Catal. A Chem. 2004, 208, 257–265. [Google Scholar] [CrossRef]
- Chen, J.; Peng, G.; Liang, T.; Zhang, W.; Zheng, W.; Zhao, H.; Guo, L.; Wu, X. Catalytic Performances of Cu/MCM-22 Zeolites with Different Cu loadings in NH3-SCR. Nanomaterials 2020, 10, 2170. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhang, C.; Li, X.; Tan, P.; Zhou, A.; Fang, Q.; Chen, G. Ho-modified Mn-Ce/TiO2 for low-temperature SCR of NOX with NH3: Evaluation and characterization. Chin. J. Catal. 2018, 39, 1653–1663. [Google Scholar] [CrossRef]
- Huang, J.; Huang, H.; Jiang, H.; Liu, L. The promotional role of Nd on Mn/TiO2 catalyst for the low-temperature NH3-SCR of NOX. Catal. Today 2019, 332, 49–58. [Google Scholar] [CrossRef]
- Meng, D.; Zhan, W.; Guo, Y.; Guo, Y.; Wang, L.; Lu, G. A highly effective catalyst of Sm-MnOx for the NH3-SCR of NOX at low temperature: Promotional role of Sm and its catalytic performance. ACS Catal. 2015, 5, 5973–5983. [Google Scholar] [CrossRef]
- Sun, P.; Guo, R.-T.; Liu, S.-M.; Wang, S.-X.; Pan, W.-G.; Li, M.-Y. The enhanced performance of MnOx catalyst for NH3-SCR reaction by the modification with Eu. Appl. Catal. A Gen. 2017, 531, 129–138. [Google Scholar] [CrossRef]
- Kwak, J.H.; Tran, D.; Burton, S.D.; Szanyi, J.; Lee, J.H.; Peden, C.H.F. Effects of hydrothermal aging on NH3-SCR reaction over Cu/zeolites. J. Catal. 2012, 287, 203–209. [Google Scholar] [CrossRef]
- Guo, R.-T.; Li, M.-Y.; Sun, P.; Pan, W.-G.; Liu, S.-M.; Liu, J.; Sun, X.; Liu, S.-W. Mechanistic Investigation of the Promotion Effect of Bi Modification on the NH3–SCR Performance of Ce/TiO2 Catalyst. J. Phys. Chem. C 2017, 121, 27535–27545. [Google Scholar] [CrossRef]
- Hu, H.; Xie, J.L.; Fang, D.; He, F. Study of Co-Mn/TiO2 SCR catalyst at low temperature. In Advanced Materials Research; Trans Tech Publications Ltd.: Zürich, Switzerland, 2015; Volume 1102, pp. 11–16. [Google Scholar]
- Ou, X.; Chen, K.; Wei, L.; Deng, Y.; Li, J.; Li, B.; Dong, L. Effect of Co Doping on Magnetic and CO-SCR Properties of γ-Fe2O3. Ind. Eng. Chem. Res. 2021, 60, 5744–5757. [Google Scholar] [CrossRef]
- Shen, M.; Li, C.; Wang, J.; Xu, L.; Wang, W.; Wang, J. New insight into the promotion effect of Cu doped V2O5/WO3–TiO2 for low temperature NH3-SCR performance. RSC Adv. 2015, 5, 35155–35165. [Google Scholar] [CrossRef]
- Guo, R.T.; Sun, P.; Pan, W.G.; Li, M.Y.; Liu, S.M.; Sun, X.; Liu, S.W.; Liu, J. A Highly Effective MnNdOx Catalyst for the Selective Catalytic Reduction of NOX with NH3. Ind. Eng. Chem. Res. 2017, 56, 12566–12577. [Google Scholar] [CrossRef]
- Liu, K.; Liu, F.; Xie, L.; Shan, W.; He, H. DRIFTS study of a Ce–W mixed oxide catalyst for the selective catalytic reduction of NOX with NH3. Catal. Sci. Technol. 2015, 5, 2290–2299. [Google Scholar] [CrossRef]
- Forzatti, P.; Nova, I.; Tronconi, E. New enhanced NH3-SCR reaction for NOX emission control. Ind. Eng. Chem. Res. 2010, 49, 10386–10391. [Google Scholar] [CrossRef]
- Bae, Y.K.; Kim, T.-W.; Kim, J.-R.; Kim, Y.; Ha, K.-S.; Chae, H.-J. Enhanced SO2 tolerance of V2O5-Sb2O3/TiO2 catalyst for NO reduction with co-use of ammonia and liquid ammonium nitrate. J. Ind. Eng. Chem. 2021, 96, 277–283. [Google Scholar] [CrossRef]
- Kobaku, S.P.R.; Kota, A.K.; Lee, D.H.; Mabry, J.M.; Tuteja, A. Patterned Superomniphobic-Superomniphilic Surfaces: Templates for Site-Selective Self-Assembly. Angew. Chem. 2012, 124, 10256–10260. [Google Scholar] [CrossRef]
- Golovin, K.; Lee, D.H.; Mabry, J.M.; Tuteja, A. Transparent, Flexible, Superomniphobic Surfaces with Ultra-Low Contact Angle Hysteresis. Angew. Chem. 2013, 125, 13245–13249. [Google Scholar] [CrossRef] [Green Version]
- Li, S.; Huang, W.; Xu, H.; Chen, T.; Ke, Y.; Qu, Z.; Yan, N. Alkali-induced deactivation mechanism of V2O5-WO3/TiO2 catalyst during selective catalytic reduction of NO by NH3 in aluminum hydrate calcining flue gas. Appl. Catal. B Environ. 2020, 270, 118872. [Google Scholar] [CrossRef]
- Kang, H.; Hong, S.; Lee, J.; Lee, K. Electrostatically Self-Assembled Nonconjugated Polyelectrolytes as an Ideal Interfacial Layer for Inverted Polymer Solar Cells. Adv. Mater. 2012, 24, 3005–3009. [Google Scholar] [CrossRef]
- Liu, G.; Tao, C.; Zhang, M.; Gu, X.; Meng, F.; Zhang, X.; Chen, Y.; Ruan, S. Effects of surface self-assembled NH4+ on the performance of TiO2-based ultraviolet photodetectors. J. Alloy. Compd. 2014, 601, 104–107. [Google Scholar] [CrossRef]
- Chen, P.; Jabłońska, M.; Weide, P.; Caumanns, T.; Weirich, T.E.; Muhler, M.; Moos, R.; Palkovits, R.; Simon, U. Formation and Effect of NH4+ Intermediates in NH3–SCR over Fe-ZSM-5 Zeolite Catalysts. ACS Catal. 2016, 6, 7696–7700. [Google Scholar] [CrossRef]
- Zhang, Y.; Guo, W.; Wang, L.; Song, M.; Yang, L.; Shen, K.; Xu, H.; Zhou, C. Characterization and activity of V2O5-CeO2/TiO2-ZrO2 catalysts for NH3-selective catalytic reduction of NOX. Chin. J. Catal. 2015, 36, 1701–1710. [Google Scholar] [CrossRef]
- Wu, X.; Yu, W.; Si, Z.; Weng, D. Chemical deactivation of V2O5-WO3/TiO2 SCR catalyst by combined effect of potassium and chloride. Front. Environ. Sci. Eng. 2013, 7, 420–427. [Google Scholar] [CrossRef]
- Lee, M.; Ye, B.; Jeong, B.; Chun, H.-Y.; Lee, D.H.; Park, S.-S.; Lee, H.; Kim, H.-D. Reduced graphene oxide supported V2O5-WO3-TiO2 catalysts for selective catalytic reduction of NOX. Korean J. Chem. Eng. 2018, 35, 1988–1993. [Google Scholar] [CrossRef]



| Sample | Chemical Composition (wt.%) | SBET (m2/g) | Pore Volume (cm3/g) | Pore Size (nm) | |||
|---|---|---|---|---|---|---|---|
| TiO2 | V2O5 | WO3 | SO3 | ||||
| VW/TiO2 | 87.53 | 2.03 | 10.04 | 0.40 | 68.86 | 0.28 | 15.94 |
| Silanized VW/TiO2 | 87.32 | 2.05 | 10.02 | 0.61 | 65.54 | 0.21 | 12.95 |
| Heat-treated VW/TiO2 | 87.53 | 1.98 | 9.99 | 0.50 | 66.39 | 0.24 | 14.17 |
| Sample | Element (at.%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| N | O | Si | F | C | S | V | W | Ti | |
| VW/TiO2 | 0.40 | 52.47 | - | - | 18.28 | 0.53 | 1.55 | 1.82 | 24.95 |
| Silanized VW/TiO2 | 0.31 | 45.90 | 0.24 | 12.89 | 19.91 | 0.24 | 1.28 | 1.36 | 17.89 |
| Heat-treated VW/TiO2 | 0.79 | 56.44 | - | - | 18.06 | 0.61 | 1.34 | 1.80 | 20.96 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, M.S.; Kim, S.-I.; Jeong, B.; Park, J.-W.; Kim, T.; Lee, J.W.; Kwon, G.; Lee, D.H. Ammonium Ion Enhanced V2O5-WO3/TiO2 Catalysts for Selective Catalytic Reduction with Ammonia. Nanomaterials 2021, 11, 2677. https://doi.org/10.3390/nano11102677
Lee MS, Kim S-I, Jeong B, Park J-W, Kim T, Lee JW, Kwon G, Lee DH. Ammonium Ion Enhanced V2O5-WO3/TiO2 Catalysts for Selective Catalytic Reduction with Ammonia. Nanomaterials. 2021; 11(10):2677. https://doi.org/10.3390/nano11102677
Chicago/Turabian StyleLee, Min Seong, Sun-I Kim, Bora Jeong, Jin-Woo Park, Taehyo Kim, Jung Woo Lee, Gibum Kwon, and Duck Hyun Lee. 2021. "Ammonium Ion Enhanced V2O5-WO3/TiO2 Catalysts for Selective Catalytic Reduction with Ammonia" Nanomaterials 11, no. 10: 2677. https://doi.org/10.3390/nano11102677
APA StyleLee, M. S., Kim, S.-I., Jeong, B., Park, J.-W., Kim, T., Lee, J. W., Kwon, G., & Lee, D. H. (2021). Ammonium Ion Enhanced V2O5-WO3/TiO2 Catalysts for Selective Catalytic Reduction with Ammonia. Nanomaterials, 11(10), 2677. https://doi.org/10.3390/nano11102677








