Nanocelluloses and Related Materials Applicable in Thermal Management of Electronic Devices: A Review
Abstract
1. Introduction
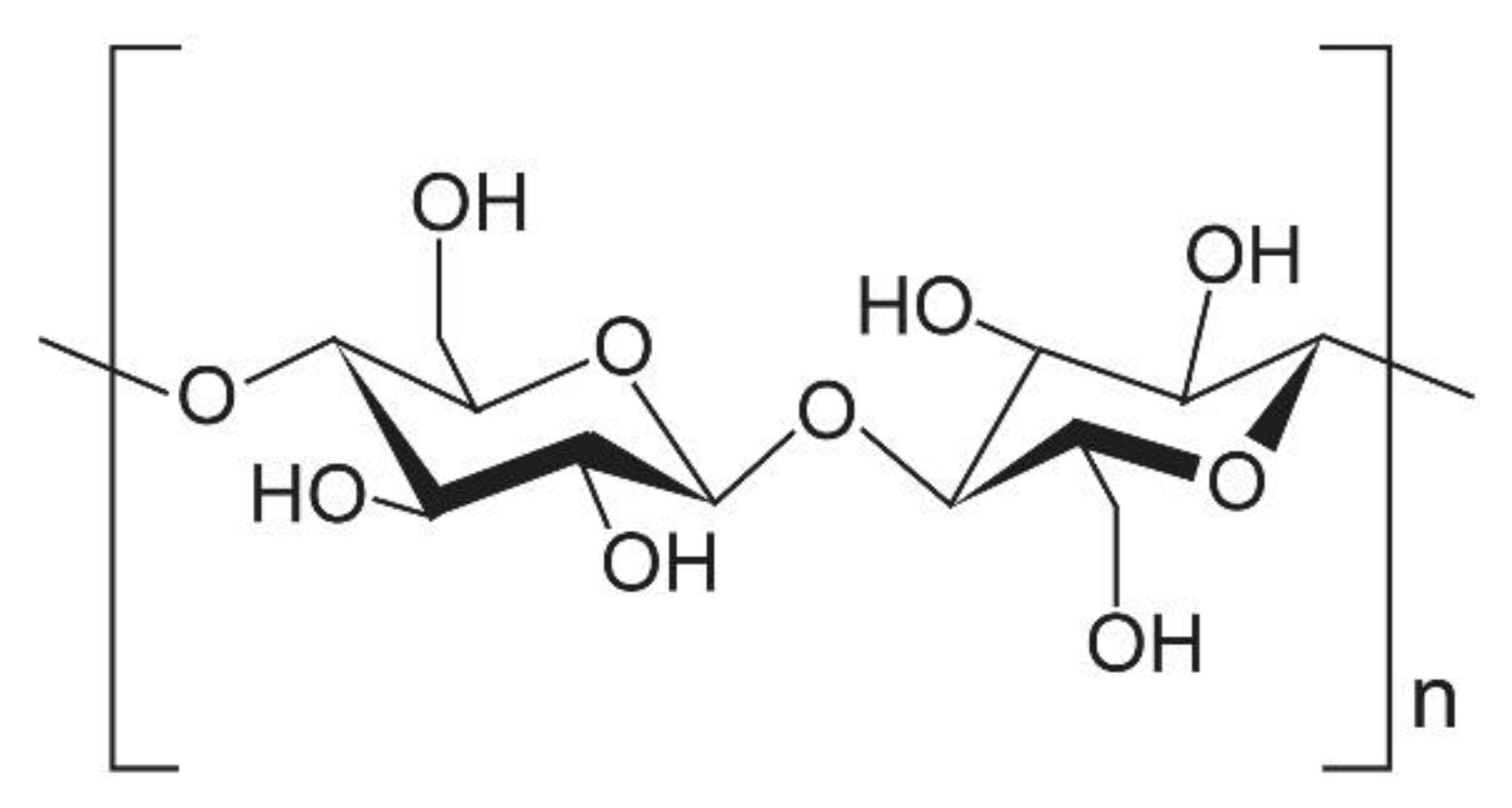
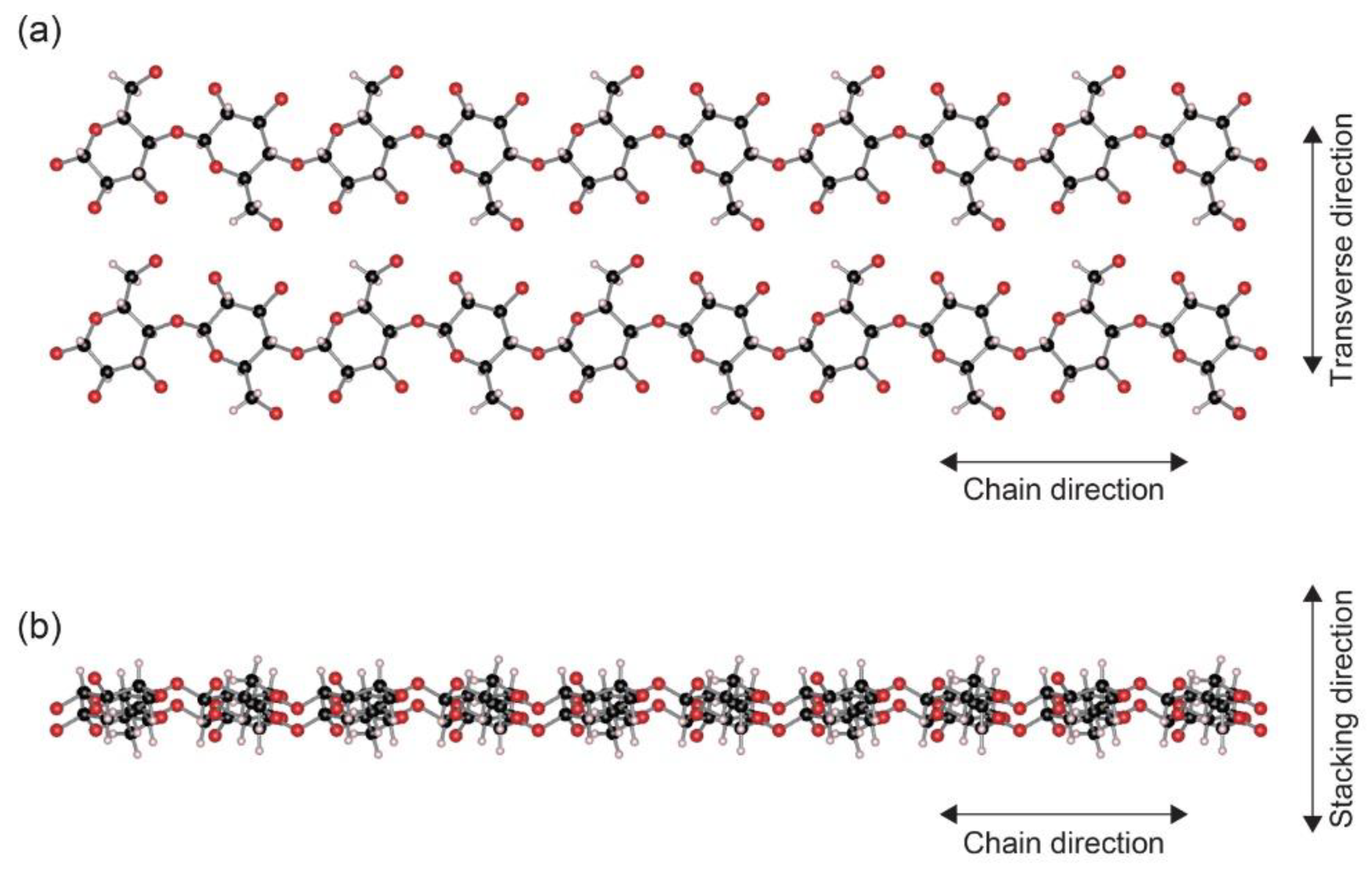
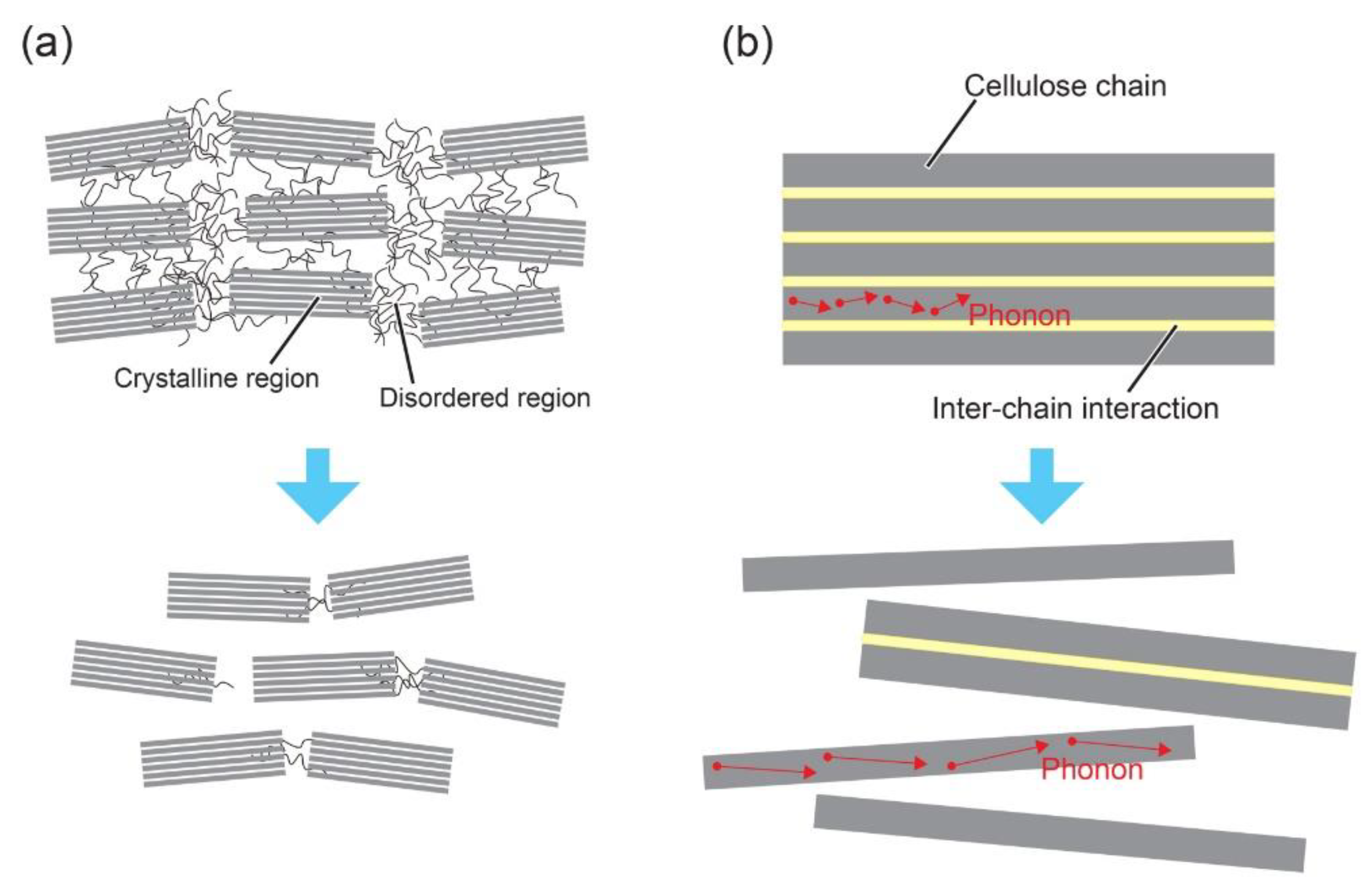
2. Heat Conduction in Nanocelluloses
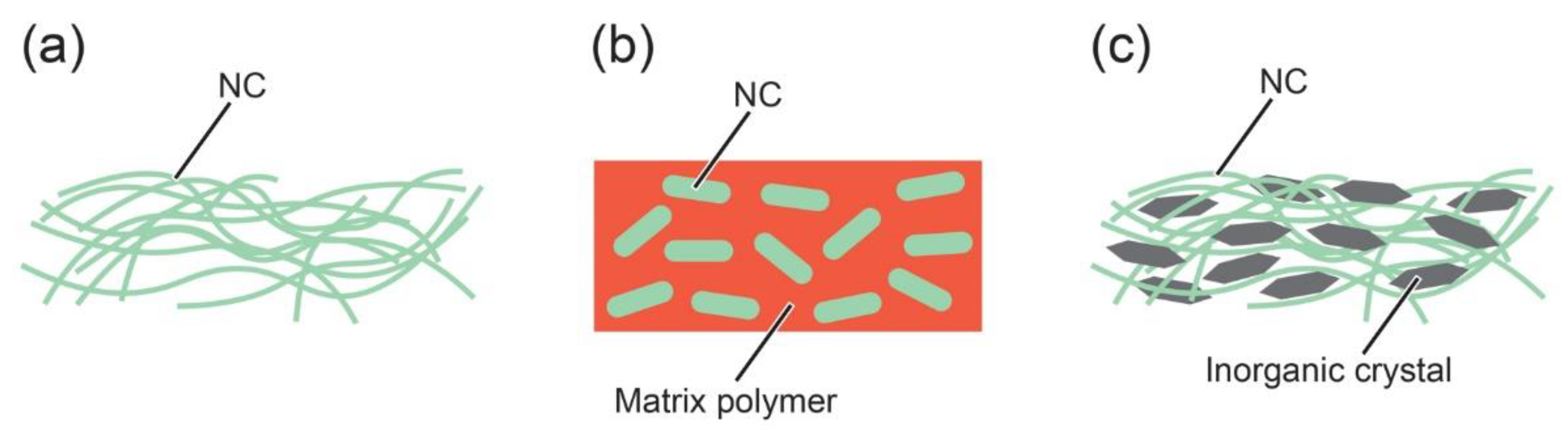
3. Thermally Conductive Materials Containing Nanocelluloses
3.1. Neat Nanocelluloses
3.2. Nanocelluloses as Heat-Conducting Fillers
3.3. Nanocelluloses as Composite Scaffolds
4. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Thompson, S.E.; Parthasarathy, S. Moore’s law: The future of Si microelectronics. Mater. Today 2006, 9, 20–25. [Google Scholar] [CrossRef]
- Pop, S. Energy Dissipation and Transport in Nanoscale Devices. Nano Res. 2010, 3, 147–169. [Google Scholar] [CrossRef]
- Waldrop, M.M. The chips are down for Moore’s law. Nature 2016, 530, 144–147. [Google Scholar] [CrossRef] [PubMed]
- Moore, A.L.; Shi, L. Emerging challenges and materials for thermal management of electronics. Mater. Today 2014, 17, 163–174. [Google Scholar] [CrossRef]
- Li, S.; Yu, S.; Feng, Y. Progress in and prospects for electrical insulating materials. High Volt. 2016, 1, 122–129. [Google Scholar] [CrossRef]
- Huang, X.; Jiang, P.; Tanaka, T. A Review of Dielectric Polymer Composites with High Thermal Conductivity. IEEE Electr. Insul. Mag. 2011, 27, 8–16. [Google Scholar] [CrossRef]
- Ngo, I.L.; Jeon, S.; Byon, C. Thermal conductivity of transparent and flexible polymers containing fillers: A literature review. Int. J. Heat Mass Tran. 2016, 98, 219–226. [Google Scholar] [CrossRef]
- Chen, H.; Ginzburg, V.V.; Yang, J.; Yang, Y.; Liu, W.; Huang, Y.; Du, L.; Chen, B. Thermal conductivity of polymer-based composites: Fundamentals and applications. Prog. Polym. Sci. 2016, 59, 41–85. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, F.; Wang, H. Novel Organic-Inorganic Composites with High Thermal Conductivity for Electronic Packaging Applications: A Key Issue Review. Polym. Compos. 2017, 38, 803–813. [Google Scholar] [CrossRef]
- Mehra, N.; Mu, L.; Ji, T.; Yang, X.; Kong, J.; Gu, J.; Zhu, J. Thermal transport in polymeric materials and across composite interfaces. Appl. Mater. Today 2018, 12, 92–130. [Google Scholar] [CrossRef]
- Zhang, Y.; Heo, Y.J.; Son, Y.R.; In, I.; An, K.H.; Kim, B.J.; Park, S.J. Recent advanced thermal interfacial materials: A review of conducting mechanisms and parameters of carbon materials. Carbon 2019, 142, 445–460. [Google Scholar] [CrossRef]
- Henry, A. Thermal Transport in Polymers. Annu. Rev. Heat Transf. 2014, 17, 485–520. [Google Scholar] [CrossRef]
- Ziman, J.M. Electrons and Phonons; Oxford University Press: New York, NY, USA, 1960; pp. 1–61. [Google Scholar]
- Ziman, J.M. Principles of the Theory of Solids, 2nd ed.; Cambridge University Press: New York, NY, USA, 1972; pp. 27–76. [Google Scholar]
- Kaviany, M. Heat Transfer Physics, 2nd ed.; Cambridge University Press: New York, NY, USA, 2014; pp. 1–49. [Google Scholar]
- Siró, I.; Plackett, D. Microfibrillated cellulose and new nanocomposite materials: A review. Cellulose 2010, 17, 459–494. [Google Scholar] [CrossRef]
- Eichhorn, S.J.; Dufresne, A.; Aranguren, M.; Marcovich, N.E.; Capadona, J.R.; Rowan, S.J.; Weder, C.; Thielemans, W.; Roman, M.; Renneckar, S.; et al. Review: Current international research into cellulose nanofibres and nanocomposites. J. Mater. Sci. 2010, 45, 1–33. [Google Scholar] [CrossRef]
- Klemm, D.; Kramer, F.; Moritz, S.; Lindström, T.; Ankerfors, M.; Gray, D.; Dorris, A. Nanocelluloses: A New Family of Nature-Based Materials. Angew. Chem. Int. Ed. 2011, 50, 5438–5466. [Google Scholar] [CrossRef]
- Moon, R.J.; Martini, A.; Nairn, J.; Simonsen, J.; Youngblood, J. Cellulose nanomaterials review: Structure, properties and nanocomposites. Chem. Soc. Rev. 2011, 40, 3941–3994. [Google Scholar] [CrossRef]
- Eichhorn, S.J. Cellulose nanowhiskers: Promising materials for advanced applications. Soft Matter 2011, 7, 303–315. [Google Scholar] [CrossRef]
- Tingaut, P.; Zimmermann, T.; Sèbe, G. Cellulose nanocrystals and microfibrillated cellulose as building blocks for the design of hierarchical functional materials. J. Mater. Chem. 2012, 22, 20105–20111. [Google Scholar] [CrossRef]
- Isogai, A.; Bergström, L. Preparation of cellulose nanofibers using green and sustainable chemistry. Curr. Opin. Green Sus. Chem. 2018, 12, 15–21. [Google Scholar] [CrossRef]
- Balea, A.; Fuente, E.; Monte, M.C.; Merayo, N.; Campano, C.; Negro, C.; Blanco, A. Industrial Application of Nanocelluloses in Papermaking: A Review of Challenges, Technical Solutions, and Market Perspectives. Molecules 2020, 25, 526. [Google Scholar] [CrossRef]
- Hermanson, G.T. Bioconjugate Techniques; Academic Press: San Diego, CA, USA, 1996; pp. 34–38. [Google Scholar]
- Uetani, K.; Hatori, K. Thermal conductivity analysis and applications of nanocellulose materials. Sci. Technol. Adv. Mater. 2017, 18, 877–892. [Google Scholar] [CrossRef] [PubMed]
- Chen, G. Nanoscale Energy Transport and Conversion; Oxford University Press: New York, NY, USA, 2005. [Google Scholar]
- Chen, G. Phonon heat conduction in nanostructures. Int. J. Therm. Sci. 2000, 39, 471–480. [Google Scholar] [CrossRef]
- Cahill, D.G.; Ford, W.K.; Goodson, K.E.; Mahan, G.D.; Majumdar, A.; Maris, H.J.; Merlin, R.; Phillpot, S.R. Nanoscale thermal transport. Appl. Phys. Rev. 2003, 93, 793–818. [Google Scholar] [CrossRef]
- Volz, S.G.; Chen, G. Molecular dynamics simulation of thermal conductivity of silicon nanowires. Appl. Phys. Lett. 1999, 75, 2056–2058. [Google Scholar] [CrossRef]
- Mingo, N.; Yang, L.; Li, D.; Majumdar, A. Predicting the Thermal Conductivity of Si and Ge Nanowires. Nano Lett. 2003, 3, 1713–1716. [Google Scholar] [CrossRef]
- Li, D.; Wu, Y.; Kim, P.; Shi, L.; Yang, P.; Majumdar, A. Thermal conductivity of individual silicon nanowires. Appl. Phys. Lett. 2003, 83, 2934–2936. [Google Scholar] [CrossRef]
- Hippalgaonkar, K.; Huang, B.; Chen, R.; Sawyer, K.; Ercius, P.; Majumdar, A. Fabrication of Microdevices with Integrated Nanowires for Investigating Low-Dimensional Phonon Transport. Nano Lett. 2010, 10, 4341–4348. [Google Scholar] [CrossRef]
- Shen, S.; Henry, A.; Tong, J.; Zheng, R.; Chen, G. Polyethylene nanofibres with very high thermal conductivities. Nat. Nanotechnol. 2010, 5, 251–255. [Google Scholar] [CrossRef]
- Liu, J.; Xu, Z.; Cheng, Z.; Xu, S.; Wang, X. Thermal Conductivity of Ultrahigh Molecular Weight Polyethylene Crystal: Defect Effect Uncovered by 0 K Limit Phonon Diffusion. ACS Appl. Mater. Interfaces 2015, 7, 27279–27288. [Google Scholar] [CrossRef]
- Shrestha, R.; Li, P.; Chatterjee, B.; Zheng, T.; Wu, X.; Liu, Z.; Luo, T.; Choi, S.; Hippalgaonkar, K.; de Boer, M.P.; et al. Crystalline polymer nanofibers with ultra-high strength and thermal conductivity. Nat. Commun. 2018, 9, 1664. [Google Scholar] [CrossRef]
- Xu, Y.; Kraemer, D.; Song, B.; Jiang, Z.; Zhou, J.; Loomis, J.; Wang, J.; Li, M.; Ghasemi, H.; Huang, X.; et al. Nanostructured polymer films with metal-like thermal conductivity. Nat. Commun. 2019, 10, 1771. [Google Scholar] [CrossRef] [PubMed]
- Henry, A.; Chen, G.; Plimpton, S.J.; Thompson, A. 1D-to-3D transition of phonon heat conduction in polyethylene using molecular dynamics simulations. Phys. Rev. B 2010, 82, 144308. [Google Scholar] [CrossRef]
- Berber, S.; Kwon, Y.; Tománek, D. Unusually High Thermal Conductivity of Carbon Nanotubes. Phys. Rev. Lett. 2000, 84, 4613–4616. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, J.; Hashimoto, T. Thermal Diffusivity Measurement of Papers by an AC Joule Heating Method. Polym. Int. 1998, 45, 207–210. [Google Scholar] [CrossRef]
- Suleiman, B.M.; Larfeldt, J.; Leckner, B.; Gustavsson, M. Thermal conductivity and diffusivity of wood. Wood Sci. Technol. 1999, 33, 465–473. [Google Scholar] [CrossRef]
- Shimazaki, Y.; Miyazaki, Y.; Takezawa, Y.; Nogi, M.; Abe, K.; Ifuku, S.; Yano, H. Excellent Thermal Conductivity of Transparent Cellulose Nanofiber/Epoxy Resin Nanocomposites. Biomacromolecules 2007, 8, 2976–2978. [Google Scholar] [CrossRef]
- O’sullivan, A.C. Cellulose: The structure slowly unravels. Cellulose 1997, 4, 173–207. [Google Scholar]
- Jones, A.O.F.; Resel, R.; Schrode, B.; Machado-Charry, E.; Röthel, C.; Kunert, B.; Salzmann, I.; Kontturi, E.; Reishofer, D.; Spirk, S. Structural Order in Cellulose Thin Films Prepared from a Trimethylsilyl Precursor. Biomacromolecules 2020, 21, 653–659. [Google Scholar] [CrossRef]
- Atalla, R.H.; VanderHart, D.L. Native Celluloses: A Composite of Two Distinct Crystalline Forms. Science 1984, 223, 283–285. [Google Scholar] [CrossRef]
- Nishiyama, Y.; Langan, P.; Chanzy, H. Crystal Structure and Hydrogen-Bonding System in Cellulose Iβ from Synchrotron X-ray and Neutron Fiber Diffraction. J. Am. Chem. Soc. 2002, 124, 9074–9082. [Google Scholar] [CrossRef]
- Nishiyama, Y.; Sugiyama, J.; Chanzy, H.; Langan, P. Crystal Structure and Hydrogen Bonding System in Cellulose Iα from Synchrotron X-ray and Neutron Fiber Diffraction. J. Am. Chem. Soc. 2003, 125, 14300–14306. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Ijuin, A.; Hotta, Y. Thermal conductivity enhancement of alumina/polyamide composites via interfacial modification. Ceram. Int. 2015, 41, 10314–10318. [Google Scholar] [CrossRef]
- Diaz, J.A.; Ye, Z.; Wu, X.; Moore, A.L.; Moon, R.J.; Martini, A.; Boday, D.J.; Youngblood, J.P. Thermal Conductivity in Nanostructured Films: From Single Cellulose Nanocrystals to Bulk Films. Biomacromolecules 2014, 15, 4096–4101. [Google Scholar] [CrossRef] [PubMed]
- Imai, T.; Putaux, J.L.; Sugiyama, J. Geometric phase analysis of lattice images from algal cellulose microfibrils. Polymer 2003, 44, 1871–1879. [Google Scholar] [CrossRef]
- Nishiyama, Y.; Johnson, G.P.; French, A.D.; Forsyth, V.T.; Langan, P. Neutron Crystallography, Molecular Dynamics, and Quantum Mechanics Studies of the Nature of Hydrogen Bonding in Cellulose Iβ. Biomacromolecules 2008, 9, 3133–3140. [Google Scholar] [CrossRef]
- Sinko, R.; Mishra, S.; Ruiz, L.; Brandis, N.; Keten, S. Dimensions of Biological Cellulose Nanocrystals Maximize Fracture Strength. ACS Macro Lett. 2014, 3, 64–69. [Google Scholar] [CrossRef]
- Momma, K.; Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 2011, 44, 1272–1276. [Google Scholar] [CrossRef]
- Akatsuka, M.; Takezawa, Y. Study of High Thermal Conductive Epoxy Resins Containing Controlled High-Order Structures. J. Appl. Polym. Sci. 2003, 89, 2464–2467. [Google Scholar] [CrossRef]
- Song, S.; Katagi, H.; Takezawa, Y. Study on high thermal conductivity of mesogenic epoxy resin with spherulite structure. Polymer 2012, 53, 4489–4492. [Google Scholar] [CrossRef]
- Uetani, K.; Okada, T.; Oyama, H.T. Crystallite Size Effect on Thermal Conductive Properties of Nonwoven Nanocellulose Sheets. Biomacromolecules 2015, 16, 2220–2227. [Google Scholar] [CrossRef]
- Uetani, K.; Okada, T.; Oyama, H.T. In-Plane Anisotropic Thermally Conductive Nanopapers by Drawing Bacterial Cellulose Hydrogels. ACS Macro Lett. 2017, 6, 345–349. [Google Scholar] [CrossRef]
- Bahar, E.; Ucar, N.; Onen, A.; Wang, Y.; Oksuz, M.; Ayaz, O.; Ucar, M.; Demir, A. Thermal and Mechanical Properties of Polypropylene Nanocomposite Materials Reinforced with Cellulose Nano Whiskers. J. Appl. Polym. Sci. 2012, 125, 2882–2889. [Google Scholar] [CrossRef]
- Uetani, K.; Okada, T.; Oyama, H.T. Thermally conductive and optically transparent flexible films with surface-exposed nanocellulose skeletons. J. Mater. Chem. C 2016, 4, 9697–9703. [Google Scholar] [CrossRef]
- Chowdhury, R.A.; Rai, A.; Glynn, E.; Morgan, P.; Moore, A.L.; Youngblood, J.P. Superior, processing dependent thermal conductivity of cellulose Nanocrystal-Poly(vinyl alcohol) composite films. Polymer 2019, 164, 17–25. [Google Scholar] [CrossRef]
- Althues, H.; Henle, J.; Kaskel, S. Functional inorganic nanofillers for transparent polymers. Chem. Soc. Rev. 2007, 36, 1454–1456. [Google Scholar] [CrossRef]
- Imai, Y.; Terahara, A.; Hakuta, Y.; Matsui, K.; Hayashi, H.; Ueno, N. Transparent poly(bisphenol A carbonate)-based nanocomposites with high refractive index nanoparticles. Eur. Polym. J. 2009, 45, 630–638. [Google Scholar] [CrossRef]
- Imai, Y.; Terahara, A.; Hakuta, Y.; Matsui, K.; Hayashi, H.; Ueno, N. Synthesis and characterization of high refractive index nanoparticle/poly(arylene ether ketone) nanocomposites. Polym. J. 2010, 42, 179–184. [Google Scholar] [CrossRef]
- Cranston, E.D.; Gray, D.G. Birefringence in spin-coated films containing cellulose nanocrystals. Colloid Surf. A 2008, 325, 44–51. [Google Scholar] [CrossRef]
- Yano, H.; Nakahara, S. Bio-composites produced from plant microfiber bundles with a nanometer unit web-like network. J. Mater. Sci. 2004, 39, 1635–1638. [Google Scholar] [CrossRef]
- Nogi, M.; Iwamoto, S.; Nakagaito, A.N.; Yano, H. Optically Transparent Nanofiber Paper. Adv. Mater. 2009, 21, 1595–1598. [Google Scholar] [CrossRef]
- Zhu, H.; Li, Y.; Fang, Z.; Xu, J.; Cao, F.; Wan, J.; Preston, C.; Yang, B.; Hu, L. Highly Thermally Conductive Papers with Percolative Layered Boron Nitride Nanosheets. ACS Nano 2014, 8, 3606–3613. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Ching, W. Calculation of ground-state and optical properties of boron nitrides in the hexagonal, cubic, and wurtzite structures. Phys. Rev. B 1991, 44, 7787–7798. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Feng, Y.; Shen, Z. Structural and electronic properties of h-BN. Phys. Rev. B 2003, 68, 104102. [Google Scholar] [CrossRef]
- Sato, K.; Horibe, H.; Shirai, T.; Hotta, Y.; Nakano, H.; Nagai, H.; Mitsuishi, K.; Watari, K. Thermally conductive composite films of hexagonal boron nitride and polyimide with affinity-enhanced interfaces. J. Mater. Chem. 2010, 20, 2749–2752. [Google Scholar] [CrossRef]
- Tanimoto, M.; Yamagata, T.; Miyata, K.; Ando, S. Anisotropic Thermal Diffusivity of Hexagonal Boron Nitride-Filled Polyimide Films: Effects of Filler Particle Size, Aggregation, Orientation, and Polymer Chain Rigidity. ACS Appl. Mater. Interfaces 2013, 5, 4374–4382. [Google Scholar] [CrossRef]
- Kim, K.; Kim, J. Fabrication of thermally conductive composite with surface modified boron nitride by epoxy wetting method. Ceram. Int. 2014, 40, 5181–5189. [Google Scholar] [CrossRef]
- Gu, J.; Lv, Z.; Wu, Y.; Guo, Y.; Tian, L.; Qiu, H.; Li, W.; Zhang, Q. Dielectric thermally conductive boron nitride/polyimide composites with outstanding thermal stabilities via in-situ polymerization-electrospinning-hot press method. Compos. Part A 2017, 94, 209–216. [Google Scholar] [CrossRef]
- Gorbachev, R.V.; Riaz, I.; Nair, R.R.; Jalil, R.; Britnell, L.; Belle, B.D.; Hill, E.W.; Novoselov, K.S.; Watanabe, K.; Taniguchi, T.; et al. Hunting for Monolayer Boron Nitride: Optical and Raman Signatures. Small 2011, 7, 465–468. [Google Scholar] [CrossRef]
- Chen, X.; Dobson, J.F.; Raston, C.L. Vortex fluidic exfoliation of graphite and boron nitride. Chem. Commun. 2012, 48, 3703–3705. [Google Scholar] [CrossRef]
- Tominaga, Y.; Sato, K.; Shimamoto, D.; Imai, Y.; Hotta, Y. Wet-jet milling-assisted exfoliation of h-BN particles with lamination structure. Ceram. Int. 2015, 41, 10512–10519. [Google Scholar] [CrossRef]
- Morishita, T.; Okamoto, H.; Katagiri, Y.; Matsushita, M.; Fukumori, K. A high-yield ionic liquid-promoted synthesis of boron nitride nanosheets by direct exfoliation. Chem. Commun. 2015, 51, 12068–12071. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Tominaga, Y.; Hotta, Y. High-Throughput Dimensional Evaluation of Hexagonal Boron Nitride 2D Nanomaterials. Cryst. Res. Technol. 2019, 54, 1800249. [Google Scholar] [CrossRef]
- Lin, Y.; Connell, J.W. Advances in 2D boron nitride nanostructures: Nanosheets, nanoribbons, nanomeshes, and hybrids with graphene. Nanoscale 2012, 4, 6908–6939. [Google Scholar] [CrossRef] [PubMed]
- Meziani, M.J.; Song, W.; Wang, P.; Lu, F.; Hou, Z.; Anderson, A.; Maimaiti, H.; Sun, Y. Boron Nitride Nanomaterials for Thermal Management Applications. ChemPhysChem 2015, 16, 1339–1346. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Wang, Y.; Hitz, E.; Lin, Y.; Yang, B.; Hu, L. Solution Processed Boron Nitride Nanosheets: Synthesis, Assemblies and Emerging Applications. Adv. Funct. Mater. 2017, 27, 1701450. [Google Scholar] [CrossRef]
- Guerra, V.; Wan, C.; McNally, T. Thermal conductivity of 2D nano-structured boron nitride (BN) and its composites with polymers. Prog. Mater. Sci. 2019, 100, 170–186. [Google Scholar] [CrossRef]
- Hu, Z.; Wang, S.; Chen, G.; Zhang, Q.; Wu, K.; Shi, J.; Liang, L.; Lu, M. An aqueous-only, green route to exfoliate boron nitride for preparation of high thermal conductive boron nitride nanosheet/cellulose nanofiber flexible film. Compos. Sci. Technol. 2018, 168, 287–295. [Google Scholar] [CrossRef]
- Wang, Z.; Wen, Y.; Zhao, S.; Zhang, W.; Ji, Y.; Zhang, S.; Li, J. Soy protein as a sustainable surfactant to functionalize boron nitride nanosheets and its application for preparing thermally conductive biobased composites. Ind. Crop. Prod. 2019, 137, 239–247. [Google Scholar] [CrossRef]
- Chen, J.; Huang, X.; Zhu, Y.; Jiang, P. Cellulose Nanofiber Supported 3D Interconnected BN Nanosheets for Epoxy Nanocomposites with Ultrahigh Thermal Management Capability. Adv. Funct. Mater. 2017, 27, 1604754. [Google Scholar] [CrossRef]
- Rubio, A.; Corkill, J.L.; Cohen, M.L. Theory of graphitic boron nitride nanotubes. Phys. Rev. B 1994, 49, 5081–5084. [Google Scholar] [CrossRef]
- Chopra, N.G.; Luyken, R.J.; Cherrey, K.; Crespi, V.H.; Cohen, M.L.; Louie, S.G.; Zettl, A. Boron Nitride Nanotubes. Science 1995, 269, 966–967. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.W.; Fennimore, A.M.; Afanasiev, A.; Okawa, D.; Ikuno, T.; Garcia, H.; Li, D.; Majumdar, A.; Zettl, A. Isotope Effect on the Thermal Conductivity of Boron Nitride Nanotubes. Phys. Rev. Lett. 2006, 97, 085901. [Google Scholar] [CrossRef] [PubMed]
- Stewart, D.A.; Savić, I.; Mingo, N. First-Principles Calculation of the Isotope Effect on Boron Nitride Nanotube Thermal Conductivity. Nano Lett. 2009, 9, 81–84. [Google Scholar] [CrossRef] [PubMed]
- Zhi, C.; Bando, Y.; Terao, T.; Tang, C.; Kuwahara, H.; Golberg, D. Towards Thermoconductive, Electrically Insulating Polymeric Composites with Boron Nitride Nanotubes as Fillers. Adv. Funct. Mater. 2009, 19, 1857–1862. [Google Scholar] [CrossRef]
- Huang, X.; Zhi, C.; Jiang, P.; Golberg, D.; Bando, Y.; Tanaka, T. Polyhedral Oligosilsesquioxane-Modified Boron Nitride Nanotube Based Epoxy Nanocomposites: An Ideal Dielectric Material with High Thermal Conductivity. Adv. Funct. Mater. 2013, 23, 1824–1831. [Google Scholar] [CrossRef]
- Zeng, X.; Sun, J.; Yao, Y.; Sun, R.; Xu, J.; Wong, C. A Combination of Boron Nitride Nanotubes and Cellulose Nanofibers for the Preparation of a Nanocomposite with High Thermal Conductivity. ACS Nano 2017, 11, 5167–5178. [Google Scholar] [CrossRef]
- Horváth, L.; Magrez, A.; Golberg, D.; Zhi, C.; Bando, Y.; Smajda, R.; Horváth, E.; Forró, L.; Schwaller, B. In Vitro Investigation of the Cellular Toxicity of Boron Nitride Nanotubes. ACS Nano 2011, 5, 3800–3810. [Google Scholar] [CrossRef]
- Balandin, A.A. Thermal properties of graphene and nanostructured carbon materials. Nat. Mater. 2011, 10, 569–581. [Google Scholar] [CrossRef]
- Shahil, K.M.F.; Balandin, A.A. Graphene-Multilayer Graphene Nanocomposites as Highly Efficient Thermal Interface Materials. Nano Lett. 2012, 12, 861–867. [Google Scholar] [CrossRef]
- Song, N.; Jiao, D.; Ding, P.; Cui, S.; Tang, S.; Shi, L. Anisotropic thermally conductive flexible films based on nanofibrillated cellulose and aligned graphene nanosheets. J. Mater. Chem. C 2016, 4, 305–314. [Google Scholar] [CrossRef]
- Song, N.; Cui, S.; Jiao, D.; Hou, X.; Ding, P.; Shi, L. Layered nanofibrillated cellulose hybrid films as flexible lateral heat spreaders: The effect of graphene defect. Carbon 2017, 115, 338–346. [Google Scholar] [CrossRef]
- Song, N.; Jiao, D.; Cui, S.; Hou, X.; Ding, P.; Shi, L. Highly Anisotropic Thermal Conductivity of Layer-by-Layer Assembled Nanofibrillated Cellulose/Graphene Nanosheets Hybrid Films for Thermal Management. ACS Appl. Mater. Interfaces 2017, 9, 2924–2932. [Google Scholar] [CrossRef] [PubMed]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Zheng, R.; Jiang, J.; Yu, J.; Dai, K.; Yan, C. Enhanced thermal conductivity and retained electrical insulation of heat spreader by incorporating alumina-deposited graphene filler in nano-fibrillated cellulose. Compos. Part. B-Eng. 2019, 178, 107489. [Google Scholar] [CrossRef]
- Ōsawa, E. Recent progress and perspectives in single-digit nanodiamond. Diamond Relat. Mater. 2007, 16, 2018–2022. [Google Scholar] [CrossRef]
- Mochalin, V.N.; Shenderova, O.; Ho, D.; Gogotsi, Y. The properties and applications of nanodiamonds. Nat. Nanotechnol. 2012, 7, 11–23. [Google Scholar] [CrossRef]
- Kumar, S.; Nehra, M.; Kedia, D.; Dilbaghi, N.; Tankeshwar, K.; Kim, K. Nanodiamonds: Emerging face of future nanotechnology. Carbon 2019, 143, 678–699. [Google Scholar] [CrossRef]
- Schelling, P.K.; Phillpot, S.R.; Keblinski, P. Comparison of atomic-level simulation methods for computing thermal conductivity. Phys. Rev. B 2002, 65, 144306. [Google Scholar] [CrossRef]
- Song, N.; Cui, S.; Hou, X.; Ding, P.; Shi, L. Significant Enhancement of Thermal Conductivity in Nanofibrillated Cellulose Films with Low Mass Fraction of Nanodiamond. ACS Appl. Mater. Interfaces 2017, 9, 40766–40773. [Google Scholar] [CrossRef]
- Sato, K.; Tominaga, Y.; Hotta, Y.; Shibuya, H.; Sugie, M.; Saruyama, T. Cellulose nanofiber/nanodiamond composite films: Thermal conductivity enhancement achieved by a tuned nanostructure. Adv. Powder Technol. 2018, 29, 972–976. [Google Scholar] [CrossRef]
- Tominaga, Y.; Sato, K.; Hotta, Y.; Shibuya, H.; Sugie, M.; Saruyama, T. Improvement of thermal conductivity of composite film composed of cellulose nanofiber and nanodiamond by optimizing process parameters. Cellulose 2018, 25, 3973–3983. [Google Scholar] [CrossRef]
- Tominaga, Y.; Sato, K.; Hotta, Y.; Shibuya, H.; Sugie, M.; Saruyama, T. Effect of the addition of Al2O3 and h-BN fillers on the thermal conductivity of a cellulose nanofiber/nanodiamond composite film. Cellulose 2019, 26, 5281–5289. [Google Scholar] [CrossRef]
- Watari, K. High Thermal Conductivity Non-Oxide Ceramics. J. Ceram. Soc. Jpn. 2001, 109, S7–S16. [Google Scholar] [CrossRef]
- Slack, G.A. Nonmetallic Crystals with High Thermal Conductivity. J. Phys. Chem. Solids 1973, 34, 321–335. [Google Scholar] [CrossRef]
- Slack, G.A.; Tanzilli, R.A.; Pohl, R.O.; Vandersande, J.W. The Intrinsic Thermal Conductivity of AlN. J. Phys. Chem. Solids 1987, 48, 641–647. [Google Scholar] [CrossRef]
- Zhang, K.; Tao, P.; Zhang, Y.; Liao, X.; Nie, S. Highly thermal conductivity of CNF/AlN hybrid films for thermal management of flexible energy storage devices. Carbohyd. Polym. 2019, 213, 228–235. [Google Scholar] [CrossRef]
- Zhang, K.; Lu, Y.; Hao, N.; Nie, S. Enhanced thermal conductivity of cellulose nanofibril/aluminum nitride hybrid films by surface modification of aluminum nitride. Cellulose 2019, 26, 8669–8683. [Google Scholar] [CrossRef]
- Qiu, J.; Hotta, Y.; Sato, K.; Watari, K.; Mitsuishi, K. Fabrication of Fine AlN Particles by Pulverizing with Very Small ZrO2 Beads. J. Am. Ceram. Soc. 2005, 88, 1676–1679. [Google Scholar] [CrossRef]
- Kocjan, A.; Krnel, K.; Kosmač, T. The influence of temperature and time on the AlN powder hydrolysis reaction products. J. Eur. Ceram. Soc. 2008, 28, 1003–1008. [Google Scholar] [CrossRef]
- Kocjan, A.; Dakskobler, A.; Krnel, K.; Kosmač, T. The course of the hydrolysis and the reaction kinetics of AlN powder in diluted aqueous suspensions. J. Eur. Ceram. Soc. 2011, 31, 815–823. [Google Scholar] [CrossRef]
- Teramoto, Y. Material development using the inherent features of nano-cellulose and nano-chitin: Necessity of simple processes and cross-disciplinary collaboration. Adv. Powder Technol. in press. [CrossRef]
- Kontturi, E.; Spirk, S. Ultrathin Films of Cellulose: A Materials Perspective. Front. Chem. 2019, 7, 488. [Google Scholar] [CrossRef] [PubMed]
- Volz, S.; Shiomi, J.; Nomura, M.; Miyazaki, K. Heat conduction in nanostructured materials. J. Therm. Sci. Technol. 2016, 11, 1–15. [Google Scholar] [CrossRef]
| Class | NCs Utilized | Matrix Polymer | Inorganic Crystals Incorporated | Thermal Conductivity (W·m−1·K−1) | References |
|---|---|---|---|---|---|
| Neat NCs | CNF | 2.5 | [55] | ||
| BNC | 2.1 | [56] | |||
| Fillers | CNF | Epoxy resin | 1.1 | [41] | |
| CNC | Polypropylene | 0.4 | [57] | ||
| CNF | Acrylic resin | 2.5 | [58] | ||
| CNC | Poly(vinyl alcohol) | 3.5 | [59] | ||
| Scaffolds | CNF | h-BN nanosheet | 145 | [66] | |
| CNF | h-BN nanosheet | 23 | [82] | ||
| CNF | h-BN nanosheet | 7 | [83] | ||
| CNF | Epoxy resin | h-BN nanosheet | 3.1 | [84] | |
| CNF | BNNT | 21 | [91] | ||
| CNF | Graphene | 12.6 | [95–97] | ||
| CNF | Al2O3, Graphene | 8.3 | [99] | ||
| CNF | ND | 11 | [104] | ||
| CNF | DND | 4.8 | [105,106] | ||
| CNF | DND, Al2O3, h-BN | 6.2 | [107] | ||
| CNF | AlN | 5.1 | [111,112] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sato, K.; Tominaga, Y.; Imai, Y. Nanocelluloses and Related Materials Applicable in Thermal Management of Electronic Devices: A Review. Nanomaterials 2020, 10, 448. https://doi.org/10.3390/nano10030448
Sato K, Tominaga Y, Imai Y. Nanocelluloses and Related Materials Applicable in Thermal Management of Electronic Devices: A Review. Nanomaterials. 2020; 10(3):448. https://doi.org/10.3390/nano10030448
Chicago/Turabian StyleSato, Kimiyasu, Yuichi Tominaga, and Yusuke Imai. 2020. "Nanocelluloses and Related Materials Applicable in Thermal Management of Electronic Devices: A Review" Nanomaterials 10, no. 3: 448. https://doi.org/10.3390/nano10030448
APA StyleSato, K., Tominaga, Y., & Imai, Y. (2020). Nanocelluloses and Related Materials Applicable in Thermal Management of Electronic Devices: A Review. Nanomaterials, 10(3), 448. https://doi.org/10.3390/nano10030448





