The Nanofication and Functionalization of Bacterial Cellulose and Its Applications
Abstract
1. Introduction
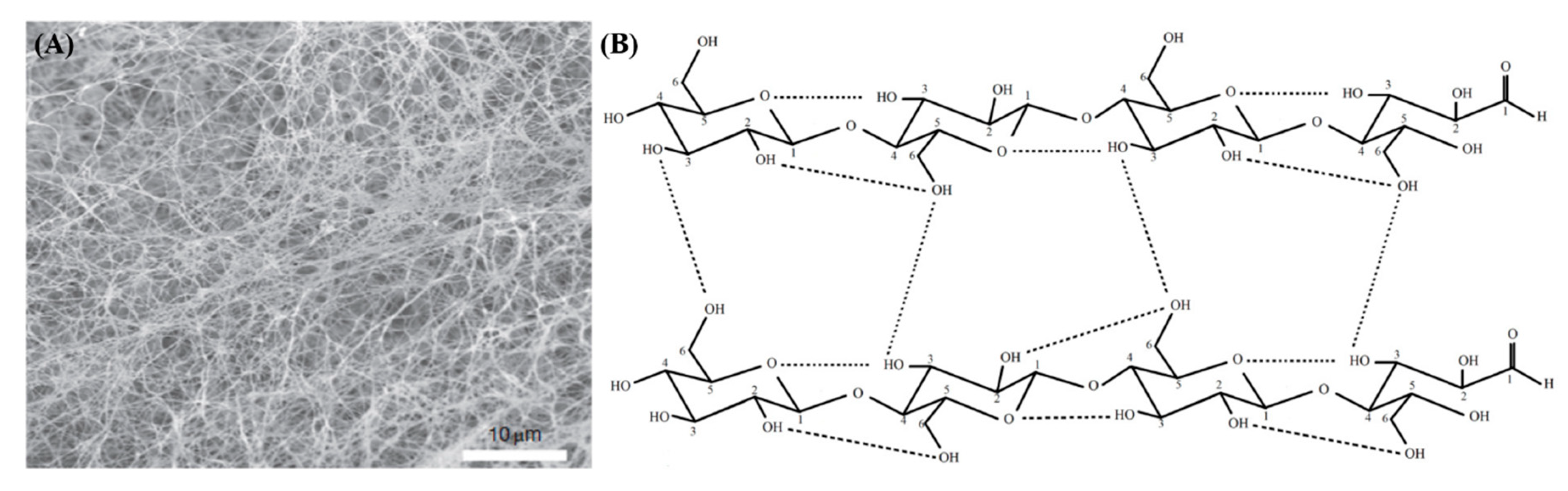
1.1. Structure
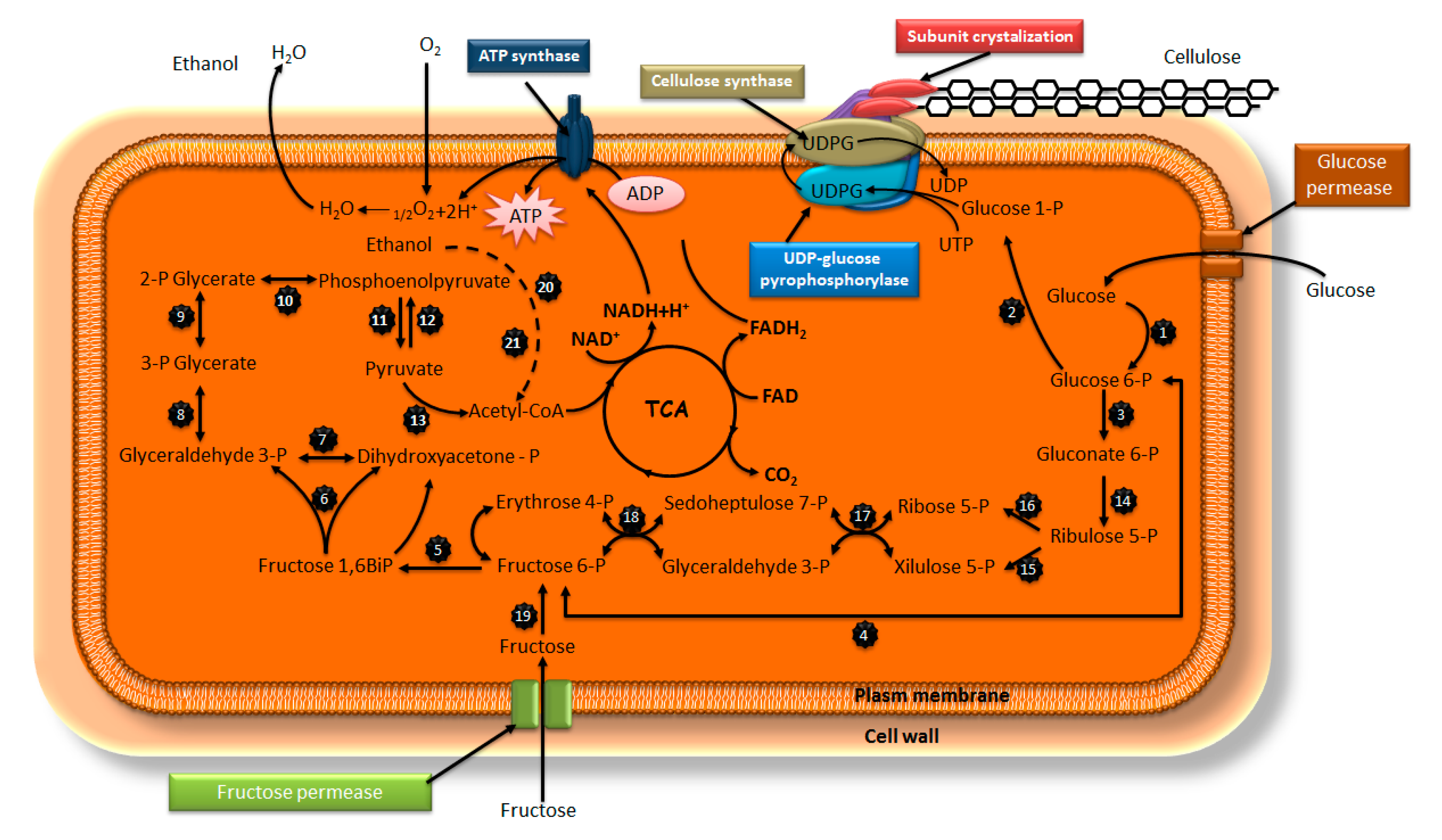
1.2. Biosynthesis and Its Mechanism
1.3. Cultivation Mode
2. The Nanofication of Bacterial Cellulose (BC)
2.1. Acid Hydrolysis
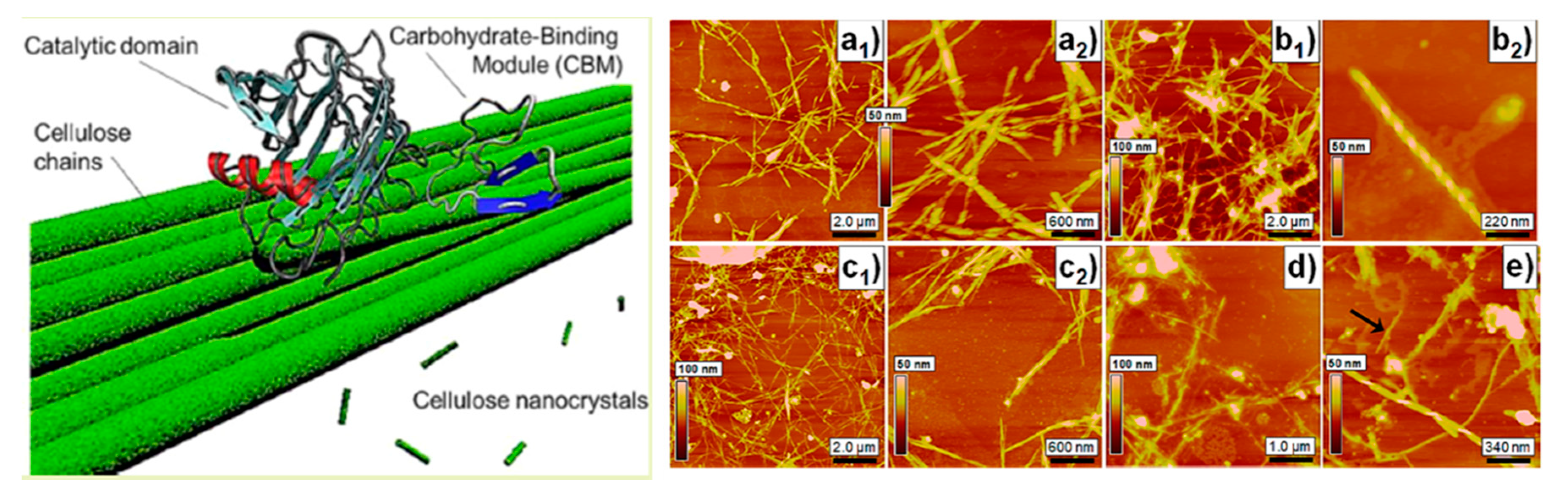
2.2. Enzymatic Hydrolysis
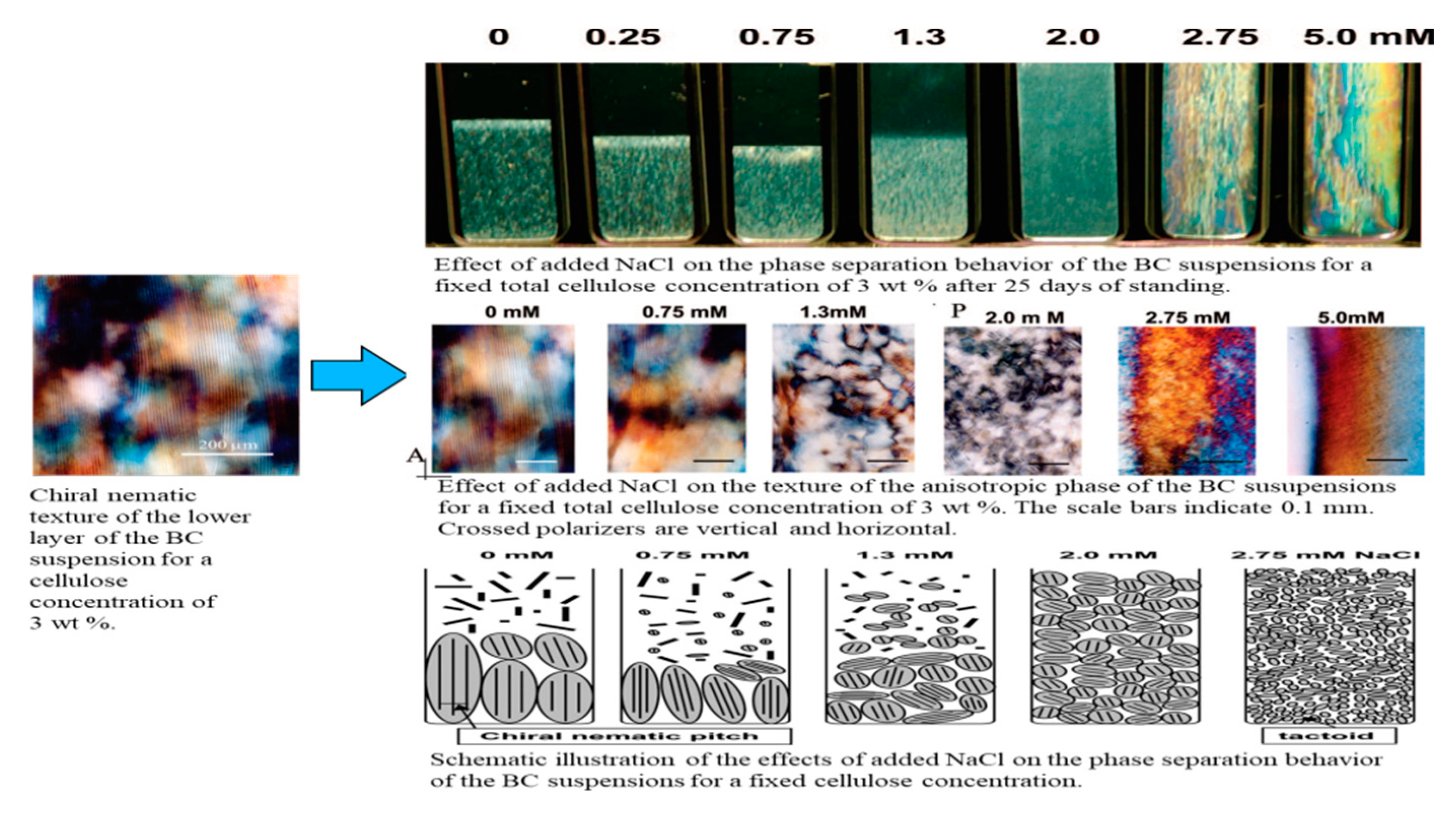
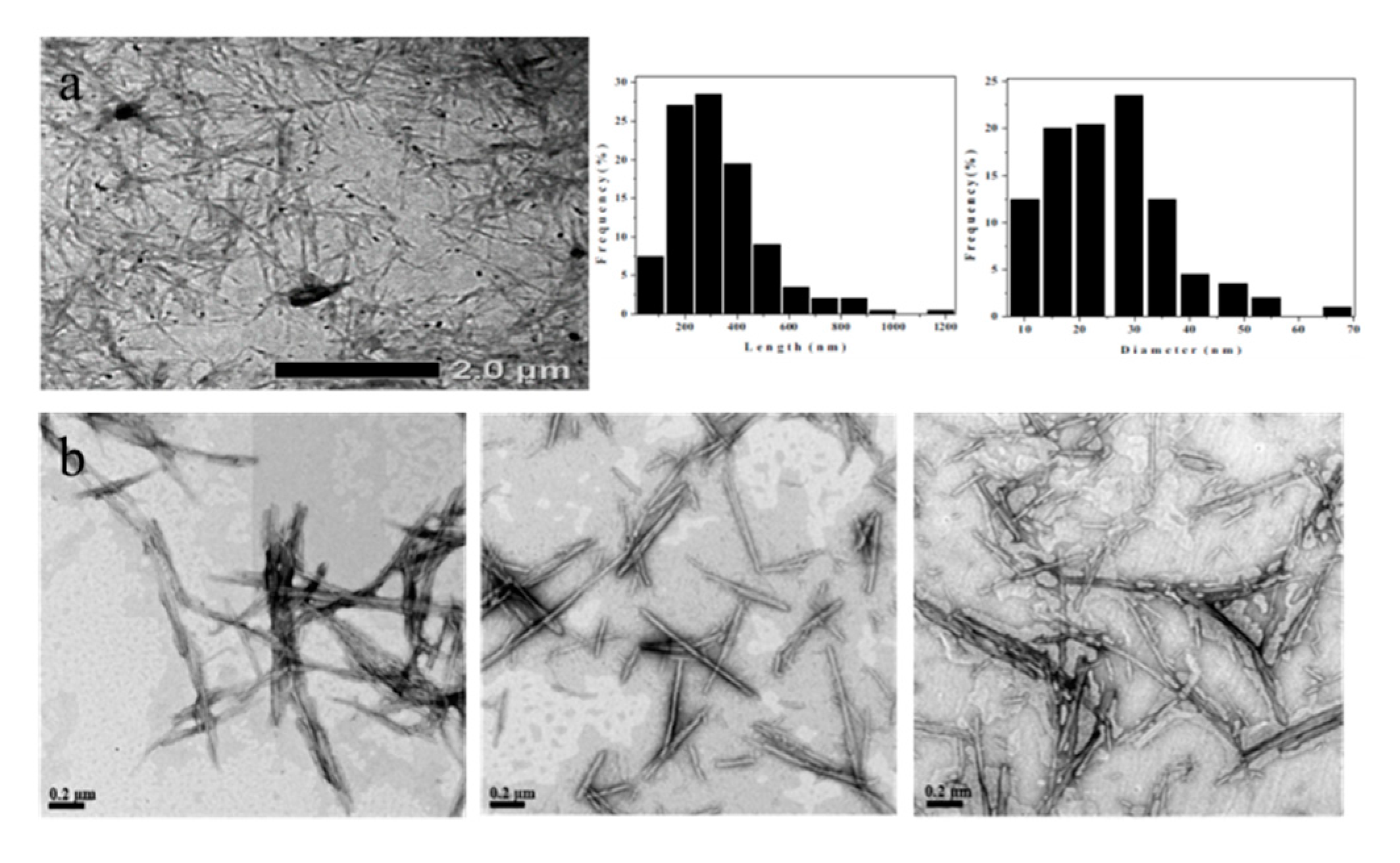
2.3. Shape of Hydrolyzed Bacterial Cellulose (BC) Nanocrystals
2.4. Nanoficated Bacterial Cellulose (BC)-Based Nanocomposites
2.4.1. Bacterial Cellulose (BC) Nanofibers-Based Nanocomposites
2.4.2. Bacterial Cellulose (BC) Nanocrystals-Based Nanocomposites
2.5. Applications of Nanoficated Bacterial Cellulose (BC)-Based Nanocomposites
3. Functionalization of Bacterial Cellulose (BC)
3.1. Suface Modification
3.2. Fuctionalization and Hybridization
3.3. Applications of Fuctionalized BC
4. Conclusions and Overview
Funding
Conflicts of Interest
References
- Costanza, R.; Fioramonti, L.; Kubiszewski, I. The UN sustainable development goals and the dynamics of well-being. Front. Ecol. Environ. 2016, 14, 59. [Google Scholar] [CrossRef]
- Akinsemolu, A.A. The role of microorganisms in achieving the sustainable development goals. J. Clean. Prod. 2018, 182, 139–155. [Google Scholar] [CrossRef]
- Lee, J.W.; Na, D.; Park, J.M.; Lee, J.; Choi, S.; Lee, S.Y. Systems metabolic engineering of microorganisms for natural and non-natural chemicals. Nat. Chem. Biol. 2012, 8, 536–546. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Song, C.W.; Shin, J.H.; Lee, S.Y. Biorefineries for the production of top building block chemicals and their derivatives. Metab. Eng. 2015, 28, 223–239. [Google Scholar] [CrossRef]
- Choi, S.Y.; Park, S.J.; Kim, W.J.; Yang, J.E.; Lee, H.; Shin, J.; Lee, S.Y. One-step fermentative production of poly(lactate-co-glycolate) from carbohydrates in Escherichia coli. Nat. Biotechnol. 2015, 34, 435–440. [Google Scholar] [CrossRef]
- Ullah, H.; Wahid, F.; Santos, A.H.; Khan, T. Advances in biomedical and pharmaceutical applications of functional bacterial cellulose-based nanocomposites. Carbohydr. Polym. 2016, 150, 330–352. [Google Scholar] [CrossRef]
- Khan, T.; Park, J.K.; Kwon, J.H. Functional biopolymers produced bybiochemical technology considering applications in food engineering. Korean J. Chem. Eng. 2017, 24, 816–826. [Google Scholar] [CrossRef]
- Petersen, N.; Gatenholm, P. Bacterial cellulose-based materials andmedical devices: current state and perspectives. Appl. Microbiol. Biotechnol. 2011, 91, 1277–1286. [Google Scholar] [CrossRef]
- Shezad, O.; Khan, S.; Khan, T.; Park, J.K. Physicochemical and mechanicalcharacterization of bacterial cellulose produced with an excellent productivityin static conditions using a simple fed-batch cultivation strategy. Carbohydr. Polym. 2010, 82, 173–180. [Google Scholar] [CrossRef]
- Cann, M.C. Nanocellulose and Sustainability: Production, Properties, Applications, and Case Studies. In Sustainability: Contributions through Science and Technology; Lee, K.Y., Ed.; CRC Press: Baca Raton, FL, USA, 2018; p. 132. [Google Scholar]
- Brown, A.J. XIX.-The chemical action of pure cultivations of bacterium aceti. J. Chem. Soc. Trans. 1886, 49, 172–187. [Google Scholar] [CrossRef]
- Brown, A.J. XLIII.-On an acetic ferment which forms cellulose. J. Chem. Soc. Trans. 1886, 49, 432–439. [Google Scholar] [CrossRef]
- Matsutani, M.; Ito, K.; Azuma, Y.; Ogino, H.; Shirai, M.; Yakushi, T.; Matsushita, K. Adaptive mutation related to cellulose producibility in Komagataeibactermedellinensis (Gluconacetobacter xylinus) NBRC 3288. Appl. Microbiol. Biotechnol. 2015, 99, 7229–7240. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Yukphan, P.; Lan Vu, H.T.; Muramatsu, Y.; Ochaikul, D.; Tanasupawat, S.; Nakagawa, Y. Description of Komagataeibacter gen. nov., with proposals ofnew combinations (Acetobacteraceae). J. Gen. Appl. Microbiol. 2012, 58, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.; Chen, S.; Yang, J.; Li, Z.; Wang, H. Functionalized bacterial cellulose derivatives and nanocomposites. Carbohydr. Polym. 2014, 101, 1043–1060. [Google Scholar] [CrossRef]
- Cacicedo, M.L.; Castro, M.C.; Servetas, I.; Bosnea, L.; Boura, K.; Tsafrakidou, P.; Dima, A.; Terpou, A.; Koutinas, A.; Castro, G.R. Progress in bacterial cellulose matrices for biotechnological applications. Bioresour. Technol. 2016, 213, 172–180. [Google Scholar] [CrossRef]
- Gao, M.; Li, J.; Bao, Z.; Hu, M.; Nian, R.; Feng, D.; An, D.; Li, X.; Xian, M.; Zhang, H. A natural in situ fabrication method of functional bacterial cellulose using a microorganism. Nat. Commun. 2019, 10, 437. [Google Scholar] [CrossRef]
- Ludwicka, K.; Jedrzejczak-Krzepkowska, M.; Kubiak, K.; Kolodziejczyk, M.; Pankiewicz, T.; Bielecki, S. Medical and cosmetic applications of bacterial nanocellulose. Bacterial NanoCellulose 2016, 145–165. [Google Scholar]
- Keshk, S.M. Bacterial cellulose production and its industrial applications. J. Bioprocess. Biotech. 2014, 4, 1–10. [Google Scholar] [CrossRef]
- Ng, C.C.; Shyu, Y.T. Development and production ofcholesterol-lowering Monascus-nata complex. World J. Microbiol. Biotechnol. 2004, 20, 875–879. [Google Scholar] [CrossRef]
- Budhiono, A.; Rosidi, B.; Taher, H.; Iguchi, M. Kinetic aspects of bacterialcellulose formation in nata-de-coco culture system. Carbohydr. Polym. 1999, 40, 137–143. [Google Scholar] [CrossRef]
- Bodin, A.; Backdahl, H.; Petersen, N.; Gatenholm, P. Comprehensive Biomaterials II. In Bacterial Cellulose as Biomaterial; Healy, K., Hutmacher, D.W., Grainger, D.W., Kirkpatrick, C.J., Eds.; Elsevier: Amsterdam, Nederland, 2017; Volume 2, pp. 505–511. [Google Scholar]
- Esa, F.; Tasirin, S.M.; Rahman, N.A. Overview of bacterial cellulose production and application. Agric. Agric. Sci. Procedia 2014, 2, 113–119. [Google Scholar] [CrossRef]
- Bielecki, S.; Krystynowicz, A.; Turkiewicz, M.; Kalinowska, H. Bacterial cellulose. In Biopolymers; Vandamme, E.J., De Baerts, S., Steinbuechel, A., Eds.; Wiley-VCH Verlag GmbH: Weinheim, Germany, 2002; Volume 5, p. 37. [Google Scholar]
- Chawla, P.R.; Bajaj, I.B.; Survase, S.A.; Singhal, R.S. Microbial Cellulose: Fermentative Production and Applications. Food Technol. Biotechnol. 2009, 47, 107–124. [Google Scholar]
- Gayathry, G.; Gopalaswamy, G. Production and Characterization of Microbial Cellulosic Fibre From Acetobacter Xylinum. Indian J. Fibre Text. 2014, 39, 93–96. [Google Scholar]
- Sani, A.; Dahman, Y. Improvements in the production of bacterial synthesized biocellulose nanofibres using different culture methods. J. Chem. Technol. Biotechnol. 2010, 85, 151–164. [Google Scholar] [CrossRef]
- Ross, P.; Mayer, R.; Benziman, M. Cellulose biosynthesis and function in bacteria. Microbiol. Rev. 1991, 55, 35–58. [Google Scholar] [CrossRef] [PubMed]
- Lustri, W.R.; de Oliveira Barud, H.G.; Barud, H.d.S.; Peres, M.F.S.; Gutierrez, J.; Tercjak, A.; de Oliveira Junior, O.B.; Lima Ribeiro, S.J. Microbial cellulose-Biosynthesis mechanisms and medical applications. In Cellululose-Fundamental Aspects and Current Trends; Poletto, M., Ed.; IntechOpen: London, UK, 2015; pp. 133–157. [Google Scholar]
- Morgan, J.L.; Strumillo, J.; Zimmer, J. Crystallographic snapshot of cellulose synthesis and membrane translocation. Nature 2013, 493, 181–186. [Google Scholar] [CrossRef]
- Brown, R.M. Cellulose structure and biosynthesis: what is in store for the 21st century? J. Polym. Sci. Polym. Chem. 2004, 42, 487–495. [Google Scholar] [CrossRef]
- Dahman, Y. Nanostructured Biomaterials and Biocomposites from Bacterial Cellulose Nanofibers. J. Nanosci. Nanotechnol. 2009, 9, 5105–5122. [Google Scholar] [CrossRef]
- Jang, W.D.; Hwang, J.H.; Kim, H.U.; Ryu, J.Y.; Lee, S.Y. Bacterial cellulose as an example product for sustainable production and consumption. Microb. Biotechnol. 2017, 10, 1181–1185. [Google Scholar] [CrossRef]
- Fijałkowski, K.; Zywicka, A.; Drozd, R.; Niemczyk., A.; Junka, A.F.; Peitler, D.; Kordas, M.; Konopacki, M.; Szymczyk, P.; Fray, M.E.; et al. Modification of bacterial cellulose through exposure to the rotatingmagnetic field. Carbohydr. Polym. 2015, 133, 52–60. [Google Scholar]
- Forng, E.R.; Anderson, S.M.; Cannon, R.E. Synthetic medium for Acetobacter xylinum that can be used for isolation of auxotrophic mutants and study of cellulose biosynthesis. Appl. Environ. Microbiol. 1989, 55, 1317–1319. [Google Scholar] [CrossRef] [PubMed]
- Koyama, M.; Helbert, W.; Imai, T.; Sugiyama, J.; Henrissat, B. Parallel upstructure evidences the molecular directionality during biosynthesis of bacterial cellulose. Proc. Natl. Acad. Sci. USA 1997, 94, 9091–9095. [Google Scholar] [CrossRef] [PubMed]
- Zugenmaier, P. Conformation and packing of various crystalline cellulose fibers. Prog. Polym. Sci. 2001, 26, 1341–1417. [Google Scholar] [CrossRef]
- De Olyveira, G.M.; Manzine Costa, L.M.; dos Santos Riccardi, C.; dos Santos, M.L.; Daltro, P.B.; Basmaji, P.; de Cerqueira Daltro, G.; Carlos Guastaldi, A. Bacterial cellulose for advanced medical materials. In Nanobiomaterials in Soft Tissue Engineering; Grumezescu, A.M., Ed.; William Andrew: New York, NY, USA, 2016; Volume 5, pp. 57–82. [Google Scholar]
- Azeredo, H.M.C.; Barud, H.; Farinas, C.S.; Vasconcellos, V.M.; Claro, A.M. Bacterial cellulose as a raw material for food and food packaging applications. Front. Sustain. Food Syst. 2019, 7, 1–14. [Google Scholar] [CrossRef]
- Karim, Z.; Afrin, S. Bacterial cellulose: Preparation and characterization. In Cellulose-Reinforced Nanofibre Composites; Jawaid, M., Boufi, S., Khalil, A., Eds.; Woodhead Publishing: Cambridge, England, 2017; pp. 327–340. [Google Scholar]
- Kralisch, D.; Hessler, N.; Klemm, D.; Erdmann, R.; Schmidt, W. White biotechnology for cellulose manufacturing: The HoLiR concept. Biotechnol. Bioeng. 2010, 105, 740–747. [Google Scholar] [PubMed]
- Yan, Z.; Chen, S.; Wang, H.; Wang, B.; Jiang, J. Biosynthesis of bacterial cellulose/multi-walled carbon nanotubes in agitated culture. Carbohydr. Polym. 2008, 74, 659–665. [Google Scholar] [CrossRef]
- Tse, M.L.V.; Chung, K.M.; Dong, L.; Thomas, B.K.; Fu, L.B.; Cheng, K.C.D.; Lu, C.; Tam, H.Y. Observation of symmetrical reflection sidebands in a silica suspended-core fiber Bragg grating. Opt. Express. 2010, 18, 17373–17381. [Google Scholar] [CrossRef]
- Wang, J.; Tavakoli, J.; Tang, Y. Bacterial cellulose production, properties and applications with different culture methods-A review. Carbohydr. Polym. 2019, 219, 63–76. [Google Scholar] [CrossRef]
- Lin, S.P.; Calvar, I.L.; Catchmark, J.F.; Liu, J.R.; Demirci, A.; Cheng, K.C. Biosynthesis, production and applications of bacterial cellulose. Cellulose 2013, 20, 2191–22119. [Google Scholar] [CrossRef]
- Ul-Islam, M.; Khan, T.; Ullah, M.W.; Park, J.K. Bacterial cellulose composites: Synthetic strategies and multiple applications in bio-medical and electro-conductive field. Biotechnol. J. 2015, 10, 1847–1861. [Google Scholar] [CrossRef]
- Kouda, T.; Naritomi, T.; Yano, H.; Yoshinaga, F. Inhibitory effect of carbon dioxide on bacterial cellulose production by Acetobacter in agitated culture. J. Ferment. Bioeng. 1998, 85, 318–321. [Google Scholar] [CrossRef]
- Kouda, T.; Naritomi, T.; Yano, H.; Yoshinaga, F. Effects of oxygen and carbon dioxide pressures on bacterial cellulose production by Acetobacter in aerated and agitated culture. J. Ferment. Bioeng. 1998, 84, 124–127. [Google Scholar] [CrossRef]
- Kouda, T.; Yano, H.; Yoshinaga, F. Effect of agitator configuration on bacterial cellulose productivity in aerated and agitated culture. J. Ferment. Bioeng. 1997, 83, 371–376. [Google Scholar] [CrossRef]
- Hu, Y.; Catchmark, J.M.; Vogler, E.A. Factors impacting the formation of sphere-like bacterial cellulose particles and their biocompatibility for human osteoblast growth. Biomacromolecules 2013, 14, 3444–3452. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Tabuchi, M.; Morinaga, Y.; Yoshinaga, F. Structural features and properties of bacterial cellulose produced in agitated culture. Cellulose 1998, 5, 187–200. [Google Scholar] [CrossRef]
- Shoda, M.; Sugano, Y. Recent advances in bacterial cellulose production. Biotechnol. Bioprocess Eng. 2005, 10, 1–8. [Google Scholar] [CrossRef]
- Wu, S.C.; Li, M.H. Production of bacterial cellulose membranes in a modified airlift bioreactor by Gluconacetobacter xylinus. J. Biosci. Bioeng. 2015, 120, 444–449. [Google Scholar] [CrossRef]
- Gratton, S.E.A.; Ropp, P.A.; Pohlhaus, P.D.; Luft, J.C.; Madden, V.J.; Napier, M.E.; DeSimone, J.M. The Effect of Particle Design on Cellular Internalization Pathways. Proc. Natl. Acad. Sci. USA 2008, 105, 11613–11618. [Google Scholar] [CrossRef]
- Lin, X.; Zhao, N.; Yan, P.; Hu, H.; Xu, F.J. The Shape and Size Effects of Polycation Functionalized Silica Nanoparticles on Gene Transfection. Acta Biomater. 2015, 11, 381–392. [Google Scholar] [CrossRef]
- Vasconcelos, N.F.; Feitosa, J.P.A.; da Gama, F.M.P.; Morais, J.P.S.; Andrade, F.K.; de Souza, M.d.S.M.; de Freitas Rosa, M. Bacterial cellulose nanocrystals produced under different hydrolysisconditions: Properties and morphological features. Carbohydr. Polym. 2017, 155, 425–431. [Google Scholar] [CrossRef]
- Revol, J.F.; Bradford, H.; Giasson, J.; Marchessault, R.H.; Gray, D.G. Helicoidal self-ordering of cellulose microfibrils in aqueous suspension. Int. J. Biol. Macromol. 1992, 14, 170–172. [Google Scholar] [CrossRef]
- George, J.; Ramana, K.V.; Sabapathy, S.N.; Jagannath, J.H.; Bawa, A.S. Characterization of chemically treated bacterial (Acetobacter xylinum) biopolymer: some thermo-mechanical properties. Int. J. Biol. Macromol. 2005, 37, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Brandes, R.; de Souza, L.; Carminatti, C.; Recouvreux, D. Production with a High Yield of Bacterial Cellulose Nanowhiss by Enzymatic Hydrolysis. Int. J. Nanosci. 2020, 19, 1950015–1950023. [Google Scholar]
- Moriana, R.; Vilaplana, F.; Ek, M. Cellulose nanocrystals from forestresidues as reinforcing agents for composites: A study from macro- tonano-dimensions. Carbohydr. Polym. 2016, 139, 139–149. [Google Scholar] [CrossRef]
- Raghuwanshi, V.S.; Cohen, Y.; Garnier, G.; Garvey, C.J.; Russell, R.A.; Darwish, T.; Garnier, G. Cellulose Dissolution in Ionic Liquid: Ion Binding Revealed by Neutron Scattering. Macromolecules 2018, 51, 7649–7655. [Google Scholar] [CrossRef]
- Youngs, T.G.A.; Holbrey, J.D.; Mullan, C.L.; Norman, S.E.; Lagunas, M.C.; D’Agostino, C.; Mantle, M.D.; Gladden, L.F.; Bowron, D.T.; Hardacre, C. Neutron diffraction, NMR and molecular dynamics study of glucose dissolved in the ionic liquid 1-ethyl-3-methylimidazolium acetate. Chem. Sci. 2011, 2, 1594–1605. [Google Scholar] [CrossRef]
- Bowron, D.T.; D’Agostino, C.; Gladden, L.F.; Hardacre, C.; Holbrey, J.D.; Lagunas, M.C.; McGregor, J.; Mantle, M.D.; Mullan, C.L.; Youngs, T.G.A. Structure and Dynamics of 1-Ethyl-3-methylimidazolium Acetate via Molecular Dynamics and Neutron Diffraction. J. Phys. Chem. B 2010, 114, 7760–7768. [Google Scholar] [CrossRef]
- Matos Ruiz, M.; Cavaillé, J.Y.; Dufresne, A.; Gérard, J.F.; Graillat, C. Processing and Characterization of New Thermoset Nanocomposites Based on Cellulose Whiskers. Compos. Compos. Interfaces 2000, 7, 117–131. [Google Scholar] [CrossRef]
- Hirai, A.; Inui, O.; Horii, F.; Tsuji, M. Phase Separation Behavior in Aqueous Suspensions of Bacterial Cellulose Nanocrystals Prepared by Sulfuric Acid Treatment. Langmuir 2009, 25, 497–502. [Google Scholar] [CrossRef]
- Singhsa, P.; Narain, R.; Manuspiya, H. Bacterial Cellulose Nanocrystals (BCNC) Preparation and Characterization from Three Bacterial Cellulose Sources and Development of Functionalized BCNCs as Nucleic Acid Delivery Systems. ACS Appl. Nano Mater. 2018, 1, 209–221. [Google Scholar] [CrossRef]
- Martínez-Sanz, M.; Lopez-Rubio, A.; Lagaron, J.M. Optimization of the nanofabrication by acid hydrolysis of bacterial cellulose nanowhiskers. Carbohydr. Polym. 2011, 85, 228–236. [Google Scholar] [CrossRef]
- Pirich, C.L.; de Freitas, R.A.; Woehl, M.A.; Picheth, G.F.; Petri, D.F.S.; Sierakowski, M.R. Bacterial cellulose nanocrystals: impact of the sulfate content on the interaction with xyloglucan. Cellulose 2015, 22, 1773–1787. [Google Scholar] [CrossRef]
- Winter, H.T.; Cerclier, C.; Delorme, N.; Bizot, H.; Quemener, B.; Cathala, B. Improved Colloidal Stability of Bacterial Cellulose Nanocrystal Suspensions for the Elaboration of Spin-Coated Cellulose-Based Model Surfaces. Biomacromolecules 2010, 11, 3144–3151. [Google Scholar] [CrossRef] [PubMed]
- Roman, M.; Winter, W.T. Effect of sulfate groups from sulfuric acid hydrolysis on the thermal degradation behavior of bacterial cellulose. Biomacromolecules 2004, 5, 1671–1677. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, T.; Pohler, E.; Geiger, T. Cellulose fibrils for polymer reinforcement. Adv. Eng. Mater. 2004, 6, 754–761. [Google Scholar] [CrossRef]
- Li, Y.; Liu, Y.; Chen, W.; Wang, Q.; Liu, Y.; Li, J.; Yu, H. Facile extraction of cellulose nanocrystals from wood using ethanol and peroxide solvothermal pretreatment followed by ultrasonic nanofibrillation. Green Chem. 2016, 18, 1010–1018. [Google Scholar] [CrossRef]
- Ahola, S.; Turon, X.; Osterberg, M.; Laine, J.; Rojas, O.J. Enzymatic Hydrolysis of Native Cellulose Nanofibrils and Other Cellulose Model Films: Effect of Surface Structure. Langmuir 2008, 24, 11592–11599. [Google Scholar] [CrossRef]
- Himmel, M.E.; Ding, S.Y.; Johnson, D.K.; Andey, W.S.; Nimlos, M.R.; Brady, J.W.; Foust, T.D. Biomass recalcitrance: engineering plants and enzymes for biofuels production. Science 2007, 315, 804–807. [Google Scholar] [CrossRef]
- Väljamäe, P.; Sild, V.; Nutt, A.; Pettersson, G.; Johansson, G. Acid hydrolysis of bacterial cellulose reveals different modes of synergistic action between cellobiohydrolase I and endoglucanase I. Eur. J. Biochem. 1999, 266, 327–334. [Google Scholar]
- Rabinovich, M.L.; Melnick, M.S.; Bolobova, A.V. The structure and mechanism of action of cellulolytic enzymes. Biochem. (Mosc) 2002, 67, 850–871. [Google Scholar] [CrossRef]
- Santa-Maria, M.; Jeoh, T. Molecular-scale investigations of cellulose microstructure during enzymatic hydrolysis. Biomacromolecules 2010, 11, 2000–2007. [Google Scholar] [CrossRef] [PubMed]
- Domingues, A.A.; Pereira, F.V.; Sierakowski, M.R.; Rojas, O.J.; Petri, D.F.S. Interfacial properties of cellulose nanoparticles obtained from acid and enzymatic hydrolysis of cellulose. Cellulose 2016, 23, 2421–2437. [Google Scholar] [CrossRef]
- Rovera, C.; Ghaani, M.; Santo, N.; Trabattoni, S.; Olsson, R.T.; Romano, D.; Farris, S. Enzymatic Hydrolysis in the Green Production of Bacterial Cellulose Nanocrystals. ACS Sustain. Chem. Eng. 2018, 6, 7725–7734. [Google Scholar] [CrossRef]
- Boisset, C.; Fraschini, C.; Schulein, M.; Henrissat, B.; Chanzy, H. Imaging the Enzymatic Digestion of Bacterial Cellulose Ribbons Reveals the Endo Character of the Cellobiohydrolase Cel6A from Humicola insolens and Its Mode of Synergy with Cellobiohydrolase Cel7A. Appl. Environ. Microbiol. 2000, 66, 1444–1452. [Google Scholar] [CrossRef]
- Xiang, Z.; Jin, X.; Liu, Q.; Chen, Y.; Li, J.; Lu, F. The reinforcement mechanism of bacterial cellulose on paper made from woody and non-woody fiber sources. Cellulose 2017, 24, 5147–5156. [Google Scholar] [CrossRef]
- Xiang, Z.; Liu, Q.; Chen, Y.; Lu, F. Effects of physical and chemical structures of bacterial cellulose on its enhancement to paper physical properties. Cellulose 2017, 24, 3513–3523. [Google Scholar] [CrossRef]
- Zhang, M.; Wu, X.; Hu, Z.; Xiang, Z.; Song, T.; Lu, F. A Highly efficient and durable fluorescent paper produced from bacterial cellulose/Eu complex and cellulosic fibers. Nanomaterials 2019, 9, 1322. [Google Scholar] [CrossRef]
- Mihranyan, A. Cellulose from Cladophorales Green Algae: From Environmental Problem to High-Tech Composite Materials. J. Appl. Polym. Sci. 2011, 119, 2449–2460. [Google Scholar] [CrossRef]
- Nogi, M.; Ifuku, S.; Abe, K.; Handa, K.; Nakagaito, A.N.; Yano, H. Fiber-content dependency of the optical transparency and thermal expansion of bacterial nanofiber reinforced composites. Appl. Phys. Lett. 2006, 88, 133124. [Google Scholar] [CrossRef]
- Gilkes, R.N.; Jervis, E.; Henrissat, B.; Tekant, B.; Miller, C.R.; Warren, J.A.R.; Kilburn, G.D. The adsorption of a bacterial cellulase and its two isolated domains to crystalline cellulose. J. Biol. Chem. 1992, 267, 6734–6749. [Google Scholar]
- Conner, A.H. Size exclusion chromatography of cellulose and cellulose derivatives. In Handbook of Size Exclusion Chromatography, Chromatographic Science Series; Wu, C.S., Ed.; Marcel Dekker Inc.: New York, NY, USA, 1995; Volume 69, pp. 331–352. [Google Scholar]
- Klemm, D.; Heublein, B.; Fink, H.P.; Bohn, A. Cellulose: Fascinating biopolymer and sustainable raw material. Angew. Chem. Int. Ed. 2005, 44, 3358–3393. [Google Scholar] [CrossRef] [PubMed]
- El Azm, N.A.; Fleita, D.; Rifaat, D.; Mpingirika, E.Z.; Amleh, A.; El-Sayed, M.M.H. Production of Bioactive Compounds from the Sulfated Polysaccharides Extracts of Ulva lactuca: Post-extraction Enzymatic Hydrolysis Followed by Ion-exchange Chromatographic Fractionation. Molecules 2019, 24, 2132. [Google Scholar] [CrossRef] [PubMed]
- Czaja, W.; Romanovicz, D.; Brown, R.M. Structural investigations of microbial cellulose produced in stationary and agitated culture. Cellulose 2004, 11, 403–411. [Google Scholar] [CrossRef]
- Elazzouzi-Hafraoui, S.; Nishiyama, Y.; Putaux, J.L.; Heux, L.; Dubreuil, F.; Rochas, C. The Shape and Size Distribution of Crystalline Nanoparticles Prepared by Acid Hydrolysis of Native Cellulose. Biomacromolecules 2008, 9, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.S.; Koo, W.M.; Kim, H.D. Preparation and properties of new regenerated cellulose fibers. Textile Res. J. 2003, 73, 998–1004. [Google Scholar] [CrossRef]
- Anwar, B.; Bundjali, B.; Arcana, I.M. Isolation of cellulose nanocrystals from bacterial cellulose produced from pineapple peel waste juice as culture medium. Procedia Chem. 2015, 16, 279–284. [Google Scholar] [CrossRef]
- Rahmad, M.; Rahmad, D. Synthesis of nano bacterial cellulose using acid hydrolysisultrasonication Treatment. J. Phys. Conf. 2019, 1185, 012028–012037. [Google Scholar]
- Azizi Samir, M.A.S.; Alloin, F.; Dufresne, A. Review of recent research into cellulosic whiskers, their properties and their application in nanocomposite field. Biomacromolecules 2005, 6, 612–626. [Google Scholar] [CrossRef]
- Eichhorn, S.J.; Dufresne, A.; Aranguren, M.; Marcovich, N.E.; Capadona, J.R.; Rowan, S.J.; Weder, C.; Thielemans, W.; Roman, M.; Renneckar, S.; et al. Review: Current international research into cellulose nanofibers and nanocomposites. J. Mater. Sci. 2010, 45, 1–33. [Google Scholar] [CrossRef]
- Juntaro, J.; Pommet, M.; Kalinka, G.; Mantalaris, A.; Shaffer, M.S.P.; Bismarck, A. Creating Hierarchical Structures in Renewable Composites by Attaching Bacterial Cellulose onto Sisal Fibers. Adv. Mater. 2008, 20, 3122–3126. [Google Scholar] [CrossRef]
- Vu, C.M.; Nguyen, D.D.; Sinh, L.H.; Pham, T.D.; Pham, L.T.; Choi, H.J. Environmentally benign green composites based on epoxy resin/bacterial cellulose reinforced glass fiber: Fabrication and mechanical characteristics. Polym. Test. 2017, 61, 150–161. [Google Scholar] [CrossRef]
- Phomrak, S.; Phisalaphong, M. Reinforcement of Natural Rubber with Bacterial Cellulose via a Latex Aqueous Microdispersion Process. J. Nanomat. 2017, 2017, 4739793. [Google Scholar] [CrossRef]
- Wang, H.; Zhu, E.; Yang, J.; Zhou, P.; Sun, D.; Tang, W. Bacterial Cellulose Nanofiber-Supported Polyaniline Nanocomposites with Flake-Shaped Morphology as Supercapacitor Electrodes. J. Phys. Chem. C 2012, 116, 13013–13019. [Google Scholar] [CrossRef]
- Yang, J.; Yu, J.; Fan, J.; Sun, D.; Tang, W.; Yang, X. Biotemplated preparation of CdS nanoparticles/bacterial cellulose hybrid nanofibers for photocatalysis application. J. Hazard. Mater. 2011, 189, 377–383. [Google Scholar] [CrossRef]
- George, J.; Ramana, K.V.; Bawa, A.S.; Siddaramaiah. Bacterial cellulose nanocrystals exhibiting high thermal stability and their polymer nanocomposites. Int. J. Biol. Macromol. 2011, 48, 50–57. [Google Scholar] [CrossRef]
- Grunert, M.; Winter, W.T. Nanocomposites of Cellulose Acetate Butyrate Reinforced with Cellulose Nanocrystals. J. Polym. Environ. 2002, 10, 27–30. [Google Scholar] [CrossRef]
- Seoane, I.T.; Manfredi, L.B.; Cyras, V.P.; Torre, L.; Fortunati, E.; Puglia, D. Effect of Cellulose Nanocrystals and Bacterial Cellulose on Disintegrability in Composting Conditions of Plasticized PHB Nanocomposites. Polymers 2017, 9, 561. [Google Scholar] [CrossRef] [PubMed]
- Georgea, J.; Kumar, R.; Sajeevkumar, V.A.; Ramana, K.V.; Rajamanickam, R.; Abhishek, V.; Nadanasabapathy, S.; Siddaramaiah. Hybrid HPMC nanocomposites containing bacterial cellulose nanocrystals and silver nanoparticles. Carbohydr. Polym. 2014, 105, 285–292. [Google Scholar] [CrossRef]
- Pommet, M.; Juntaro, J.; Heng, J.Y.; Mantalaris, A.; Lee, A.F.; Wilson, K.; Kalinka, G.; Shaffer, M.S.; Bismarck, A. Surface modification of natural fibers using bacteria: depositing bacterial cellulose onto natural fibers to create hierarchical fiber reinforced nanocomposites. Biomacromolecules 2008, 9, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Ho, K.K.; Schlufter, K.; Bismarck, A. Hierarchical composites reinforced with robust short sisal fibre preforms utilizing bacterial cellulose as binder. Compos. Sci. Technol. 2012, 72, 1479–1486. [Google Scholar] [CrossRef]
- Xu, X.; Liu, F.; Jiang, L.; Zhu, J.Y.; Haagenson, D.; Wiesenborn, D.P. Cellulose Nanocrystals vs. Cellulose Nanofibrils: A Comparative Study on Their Microstructures and Effects as Polymer Reinforcing Agents. ACS Appl. Mater. Interfaces 2013, 5, 2999–3009. [Google Scholar] [CrossRef]
- Ramsden, W. Separation of solids in the surface-layers of solutions and suspensions. Proc. R. Soc. Lond. 1904, 72, 156–164. [Google Scholar]
- Pickering, S.U. CXCVI.-Emulsions. J. Chem. Soc. Trans. 1907, 91, 2001–2021. [Google Scholar] [CrossRef]
- Hu, Z.; Ballinger, S.; Pelton, R.; Cranston, E.D. Surfactant-enhanced cellulose nanocrystal Pickering emulsions. J. Colloid Interface Sci. 2015, 439, 139–148. [Google Scholar] [CrossRef]
- Fujisawa, S.; Togawa, E.; Kuroda, K. Nanocellulose-stabilized Pickering emulsions and their applications. Sci. Technol. Adv. Mater. 2017, 18, 959–971. [Google Scholar] [CrossRef]
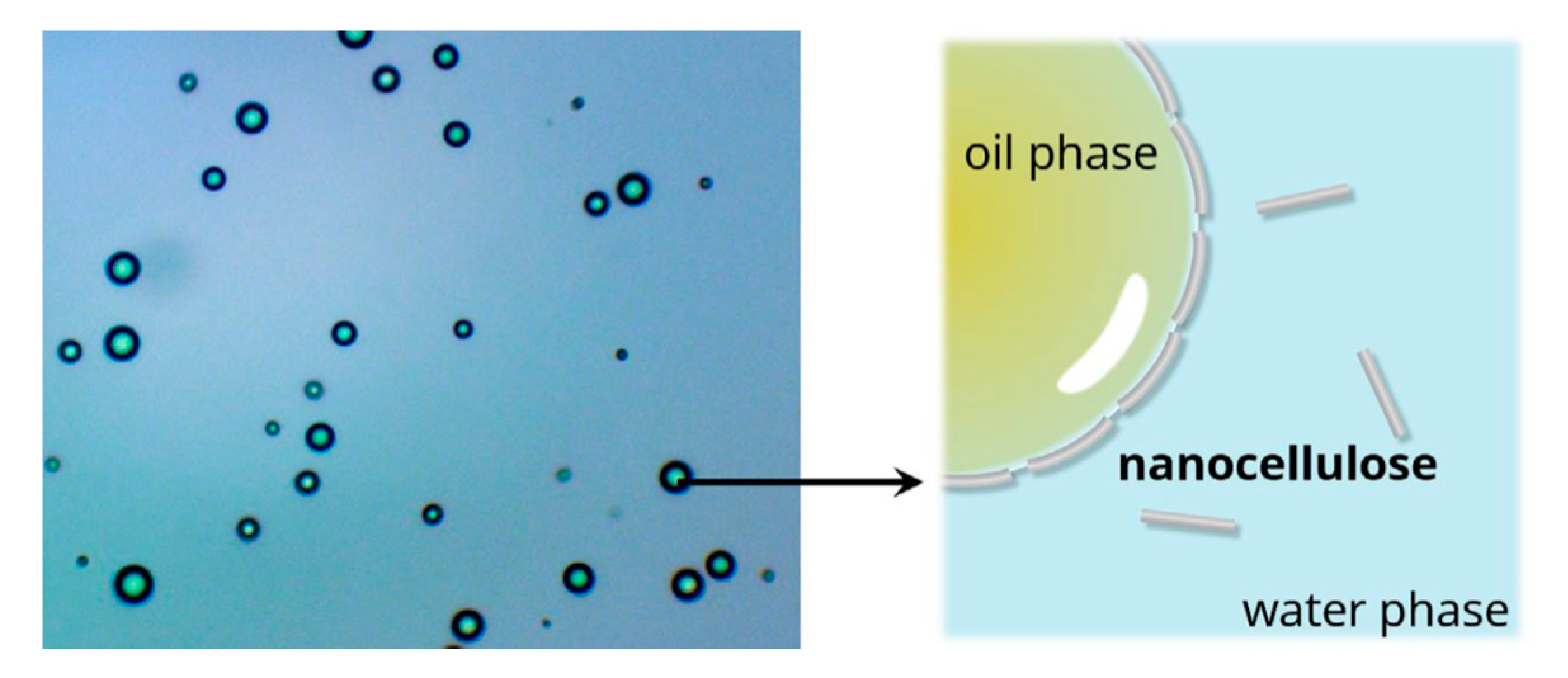
- Kalashnikova, I.; Bizot, H.; Cathala, B.; Capron, I. New Pickering Emulsions Stabilized by Bacterial Cellulose Nanocrystals. Langmuir 2011, 27, 7471–7479. [Google Scholar] [CrossRef]
- Abend, S.; Bonnke, N.; Gutschner, U.; Lagaly, G. Stabilization of emulsions by heterocoagulation of clay minerals and layered double hydroxides. Colloid Polym. Sci. 1998, 276, 730–737. [Google Scholar] [CrossRef]
- Horozov, T.S.; Binks, B.P. Particle-stabilized emulsions: A bilayer or abridging monolayer. Angew. Chem. Int. Ed. 2006, 45, 773–776. [Google Scholar] [CrossRef]
- Arditty, S.; Whitby, C.P.; Binks, B.P.; Schmitt, V.; Leal-Calderon, F. Some general features of limited coalescence in solid-stabilized emulsions. Eur. Phys. J. E 2003, 11, 273–281. [Google Scholar] [CrossRef]
- Binks, B.P.; Lumsdon, S.O. Stability of oil-in-water emulsions stabilised by silica particles. Phys. Chem. Chem. Phys. 1999, 1, 3007–3016. [Google Scholar] [CrossRef]
- Yan, H.; Chen, X.; Feng, M.; Shi, Z.; Zhang, W.; Wang, Y.; Ke, C.; Lin, Q. Entrapment of bacterial cellulose nanocrystals stabilized Pickering emulsions droplets in alginate beads for hydrophobic drug delivery. Colloids Surf. B 2019, 177, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Chen, X.; Song, H.; Li, J.; Feng, Y.; Shi, Z.; Wang, X.; Lin, Q. Synthesis of bacterial cellulose and bacterial cellulose nanocrystals for their applications in the stabilization of olive oil pickering emulsion. Food Hydrocoll. 2017, 72, 127–135. [Google Scholar] [CrossRef]
- Martinez-Sanz, M.; Lopez-Rubio, A.; Lagaron, J.M. High-barrier coated bacterial cellulose nanowhiskers with reduced moisture sensitivity. Carbohydr. Polym. 2013, 98, 1072–1082. [Google Scholar] [CrossRef]
- Fabra, M.J.; López-Rubio, A.; Ambrosio-Martín, J.; Lagaron, J.M. Improving the barrier properties of thermoplastic corn starch-based films containing bacterial cellulose nanowhiskers by means of PHA electrospun coatings of interest in food packaging. Food Hydrocoll. 2016, 61, 261–268. [Google Scholar] [CrossRef]
- Luk, Y.Y.; Kato, M.; Mrksich, M. Self-assembled monolayers of alkanethiolates presenting mannitol groups are inert to protein adsorption and cell attachment. Langmuir 2000, 16, 9604–9608. [Google Scholar] [CrossRef]
- McClary, K.B.; Ugarova, T.; Grainger, D.W. Modulating fibroblast adhesion, spreading, and proliferation using selfassembled monolayer films of alkylthiolates on gold. J. Biomed. Mater. Res. 2000, 50, 428–439. [Google Scholar] [CrossRef]
- Faucheux, N.; Schweiss, R.; Lu¨tzow, K.; Werner, C.; Groth, T. Self-assembled monolayers with different terminating groups as model substrates for cell adhesion studies. Biomaterials 2004, 25, 2721–2730. [Google Scholar] [CrossRef]
- Belgacem, M.N.; Salon-Brochier, M.C.; Krouit, M.; Bras, J. Recent advances in surface chemical modification of cellulose fibres. J. Adhes. Sci. Technol. 2010, 25, 661–684. [Google Scholar] [CrossRef]
- Andrade, F.K.; Moreira, S.M.G.; Domingues, L.; Gama, F.M.P. Improving the affinity of fibroblasts for bacterial cellulose using carbohydrate-binding modules fused to RGD. J. Biomed. Mater. Res. A 2010, 92, 9–17. [Google Scholar] [CrossRef]
- Cai, Z.; Kim, J. Bacterial cellulose/poly(ethylene glycol) composite: characterization and first evaluation of biocompatibility. Cellulose 2010, 17, 83–91. [Google Scholar] [CrossRef]
- Curran, J.M.; Chen, R.; Hunt, J.A. Controlling the phenotype and function of mesenchymal stem cells in vitro by adhesion to silane-modified clean glass surfaces. Biomaterials 2005, 26, 7057–7067. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Tabil, L.G.; Panigrahi, S. Chemical treatments of natural fiber for use in natural fiber-reinforced composites: a review. J. Polym. Environ. 2007, 15, 25–33. [Google Scholar] [CrossRef]
- Lee, K.Y.; Blaker, J.J.; Bismarck, A. Surface functionalization of bacterial cellulose as the route to produce green polylactide nanocomposites with improved properties. Compos. Sci. Technol. 2009, 69, 2724–2733. [Google Scholar] [CrossRef]
- Rodriguez-Chanfrau, J.E.; Santos, M.L.D.; Riccardi, C.D.S.; De Olyveira, G.M.; Hernandez-Escalona, M.; Basmaji, P.; Veranes-Pantoja, Y.; Guastaldi, A.C. Chemical modification of bacterial cellulose for use in regenerative medicine. Cellulose Chem. Technol. 2017, 51, 673–680. [Google Scholar]
- Rouabhia, M.; Asselin, J.; Tazi, N.; Messaddeq, Y.; Levinson, D.; Zhang, Z. Production of biocompatible and antimicrobial bacterial cellulose polymers functionalized by RGDC grafting groups and gentamicin. ACS Appl. Mater. Interfaces 2014, 6, 1439–1446. [Google Scholar] [CrossRef]
- Badshah, M.; Ullah, H.; Khan, A.R.; Khan, S.; Park, J.K.; Khan, T. Surface modification and evaluation of bacterial cellulose for drug delivery. Int. J. Biol. Macromol. 2018, 113, 526–533. [Google Scholar] [CrossRef]
- Pertile, R.A.N.; Andrade, F.K.; Alves Jr, C.; Gama, M. Surface modification of bacterial cellulose by nitrogen-containing plasma for improved interaction with cells. Carbohydr. Polym. 2010, 82, 692–698. [Google Scholar] [CrossRef]
- Bäckdahl, H.; Helenius, G.; Bodin, A.; Nannmark, U.; Johansson, B.; Risberg, B.; Gatenholm, P. Mechanical properties of bacterial cellulose and interactions withsmooth muscle cells. Biomaterials 2006, 27, 2141–2149. [Google Scholar] [CrossRef]
- Czaja, W.K.; Young, D.J.; Kawecki, M.; Brown, R.M. The future prospects ofmicrobial cellulose in biomedical applications. Biomacromolecules 2007, 8, 1–12. [Google Scholar] [CrossRef]
- Oliveira Barud, H.G.; Barud, H.d.S.; Cavicchioli, M.; Amaral, T.S.; de Oliveira Junior, O.B.; Santos, D.M.; Almeida Petersen, A.L.d.O.; Celes, F.; Borges, V.M.; de Oliveira, C.I.; et al. Preparation and characterization of a bacterial cellulose/silk fibroin sponge scaffold for tissue regeneration. Carbohydr. Polym. 2015, 128, 41–51. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, L.; Zhang, A.; Huang, Y.; Tavakoli, J.; Tang, Y. Novel bacterial cellulose/gelatin hydrogels as 3D scaffolds for tumor cell culture. Polymers 2018, 10, 581. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.C.; Lien, C.C.; Yeh, H.J.; Yu, C.M.; Hsu, S.H. Bacterial cellulose and bacterial cellulose-chitosan membranes for wound dressing applications. Carbohydr. Polym. 2013, 94, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Nisi, R.; Stoppa, M.; Licciulli, A. Silver-functionalized bacterial cellulose as antibacterial membrane for wound healing applications. ACS Omega 2017, 2, 3632–3639. [Google Scholar] [CrossRef] [PubMed]
- Katepetch, C.; Rujiravanit, R.; Tamura, H. Formation of nanocrystalline ZnO particles into bacterial cellulose pellicle by ultrasonic-assisted in situ synthesis. Cellulose 2013, 20, 1275–1292. [Google Scholar] [CrossRef]
- Zhijiang, C.; Ping, X.; Shiqi, H.; Cong, Z. Soy protein nanoparticles modified bacterial cellulose electrospun nanofiber membrane scaffold by ultrasound-induced self-assembly technique: Characterization and cytocompatibility. Cellulose 2019, 26, 6133–6150. [Google Scholar] [CrossRef]
- Dydak, K.; Junka, A.; Szymczyk, P.; Chodaczek, G.; Toporkiewicz, M.; Fijalkowski, K.; Dudek, B.; Bartoszewicz, M. Development and biological evaluation of Ti6Al7Nb scaffold implants coated with gentamycin-saturated bacterial cellulose biomaterial. PLoS ONE 2018, 13, e0205205. [Google Scholar] [CrossRef]


| Main Process | Purification | Treatment Procedure | Post-treatment | References |
|---|---|---|---|---|
| Acid hydrolysis | Washing, homogenization, drying, grinding | H2SO4/HCl mixture at 45 °C, dilution | Centrifugation, dialysis, ultrasonication | Vasconcelos et al. [56] Revol et al.[57] George et al.[58] |
| Enzymatic hydrolysis | Mechanically defibrillating | Cellulase and citrate buffer solution at 50 °C under shaking until colloidal suspension was observed | Centrifugation, dialysis | Brandes et al.[59] Moriana et al.[60] |
| Ionic liquid | Freeze-dried for 48 h | 1-ethyl-3-methylimidazolium acetate (EMIMAc) ILs at 90 °C under vacuum to remove water traces | Centrifugation | Raghuwanshet al. [61] Young et al. [62] Bowron et al.[63] |
| Acid Type | Raw Bacteria | Yield (%) | Crystallite Size (nm) * | Crystallinity Index (%) ** | Zeta Potential (mV) | References |
|---|---|---|---|---|---|---|
| H2SO4 | Komagataeibacter xylinus | 78.6~81.5 | 6.3 | 85~87 | −(31.5 ± 1) | [66] |
| Gluconacetobacter xylinum 7351 | - | 1.04~1.74 | 77~90 | - | [67] | |
| Nata de coco by Komagataeibacter xylinus | 14~63 | 5.11~5.93 | 79~92 | −(24.7 ± 2.1~ 53.6 ± 0.7) | [56] | |
| Acetobacter xylinum | - | 5.5~7.33 | 82 | −(46 ± 1) | [68] | |
| HCl | Acetobacter xylinum | - | 5.41~7.41 | 83 | −(5 ± 1) | [68] |
| Komagataeibacter xylinus | 84.4~85.6 | 6.5~6.7 | 87~89 | −11 | [66] | |
| Nata de coco by Komagataeibacter xylinus | 14 | 5.22 | 83 | −(43.9 ± 0.8) | [56] | |
| HCl + H2SO4 | Komagataeibacter xylinus | 80.5~82.4 | 6.4~6.6 | 87~89 | −(25 ± 1) | [66] |
| Acetobacter xylinum | - | 5.32~7.94 | 82 | −(40 ± 1) | [68] |
| Cellulose Source | Degree of Polymerization (DPw) | Length (nm) [4,5] | Cross-section (nm) | Crystallinity (%) |
|---|---|---|---|---|
| Algal [84] | 3–5 | >1000 | 10–20 | 95 |
| Bacterial [85,86,87,88,89] | 2000–6000 | 100–1500 | 5–10 by 30–50 | 84–89 |
| Cotton [87,88,89,90] | 8000–15000 | 200–350 | 5 | 40–60 |
| Tunicate [91,92] | 900–3500 | 100–1000 | 10–20 | 50–60 |
| Reinforcement | Substrate | Improved factor | Reference | ||
|---|---|---|---|---|---|
| Factors | Before Improvement | After Improvement | |||
| BC nanofiber covered sisal fibers | poly(L-lactic acid) (PLLA) | Tensile modulus (MPa) | 64 | 113.8 ± 8.10 | Juntaro et al. [97] |
| Young’s modulus (GPa) | 2.5 | 11.21 ± 0.69 | |||
| BC nanofiber | Glass fiber | Crack initiation (%) | 100 | 128.8 | Vu et al. [98] |
| Crack separation (%) | 100 | 111.0 | |||
| Natural rubber | Tensile modulus (MPa) | 0.8 ± 0.1 | 75.1 ± 27.1 | Phomrak et al. [99] | |
| Young’s modulus (GPa) | 0.0016 ± 0.4 | 4.13 ± 0.99 | |||
| Polyaniline (PANI) | Electrical conductivity (S/cm) | 1.61 × 10−4 | 5.1 | Wang et al. [100] | |
| CdS particles | Reaction rate of photocatalyst (min−1) | 0.00013 | 0.012 | Yang et al. [101] | |
| BC nanocrystals | Polyvinylalcohol (PVA) | Tensile strength (MPa) | 62.5 | 128 | George et al. [102] |
| Elastic modulus (GPa) | 2 | 3.4 | |||
| Melting temperature (°C) | 203.3 | 212.7 | |||
| Cellulose acetate butyrate | Melting temperature (°C) | 146.5 | 149.3 | Grunert et al. [103] | |
| Elastic modulus (GPa) at 81°C | 0.9 | 1.5 | |||
| Poly(hydroxybutyrate) (PHB) | Contact angle (o) | 76 | 72 | Seoane et al. [104] | |
| Disintegration at 14 days (%) | 18 | 50 | |||
| BC nanocrystals+ silver nanoparticles (AgNPs) | Hydroxypropyl methyl cellulose | Tensile strength (MPa) | 59 ± 5.3 | 78 ± 6.9 | Georgea et al. [105] |
| Tensile modulus (GPa) | 1.33 ± 0.25 | 2.28 ± 0.27 | |||
| Moisture sorption (%), Iglesias and Chirife model | 3.37 ± 0.91 | 2.18 ± 0.91 | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, S.M.; Shin, E.J. The Nanofication and Functionalization of Bacterial Cellulose and Its Applications. Nanomaterials 2020, 10, 406. https://doi.org/10.3390/nano10030406
Choi SM, Shin EJ. The Nanofication and Functionalization of Bacterial Cellulose and Its Applications. Nanomaterials. 2020; 10(3):406. https://doi.org/10.3390/nano10030406
Chicago/Turabian StyleChoi, Soon Mo, and Eun Joo Shin. 2020. "The Nanofication and Functionalization of Bacterial Cellulose and Its Applications" Nanomaterials 10, no. 3: 406. https://doi.org/10.3390/nano10030406
APA StyleChoi, S. M., & Shin, E. J. (2020). The Nanofication and Functionalization of Bacterial Cellulose and Its Applications. Nanomaterials, 10(3), 406. https://doi.org/10.3390/nano10030406






