Fabrication and Deposition of Copper and Copper Oxide Nanoparticles by Laser Ablation in Open Air
Abstract
1. Introduction
2. Materials and Methods
2.1. Laser Ablation
2.2. Sample Preparation and Characterization Technics
2.3. Antimicrobial Activity
2.4. Statistical Analysis
3. Results and Discussion
3.1. Ablation Rate
3.2. Physicochemical Characterization of the Film
3.3. Characterization of the Obtained Nanoparticles
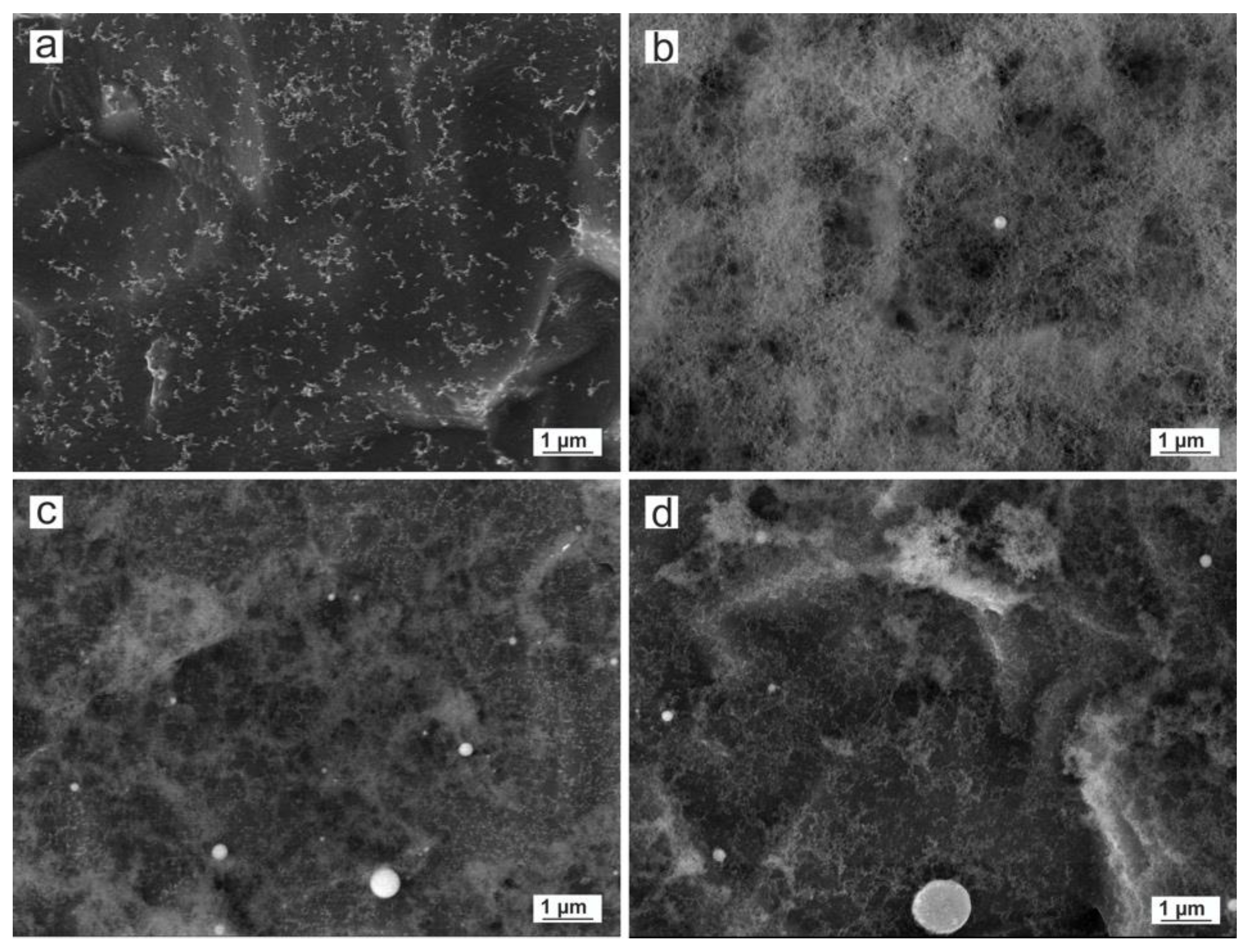
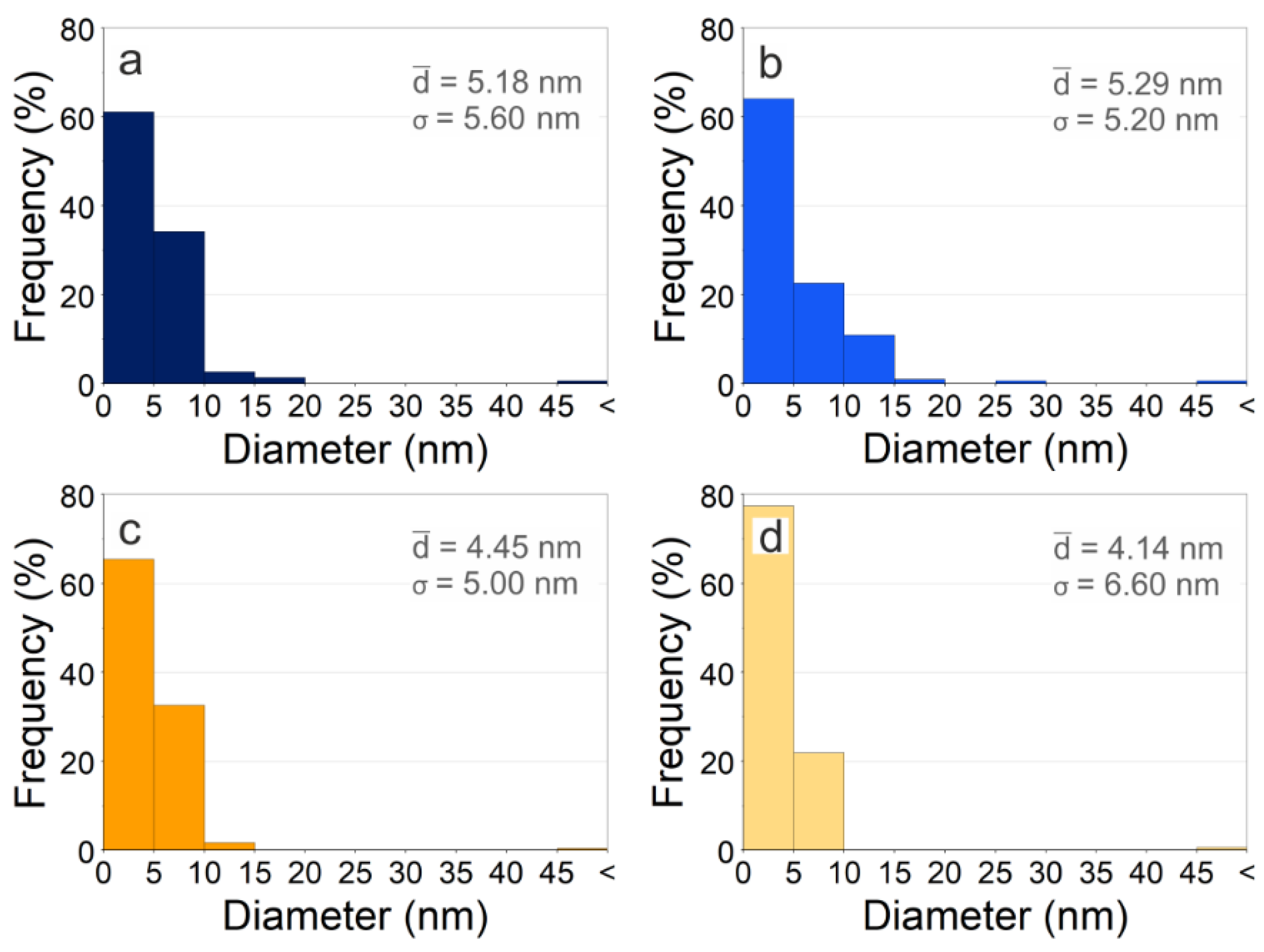
3.3.1. Size and Morphology
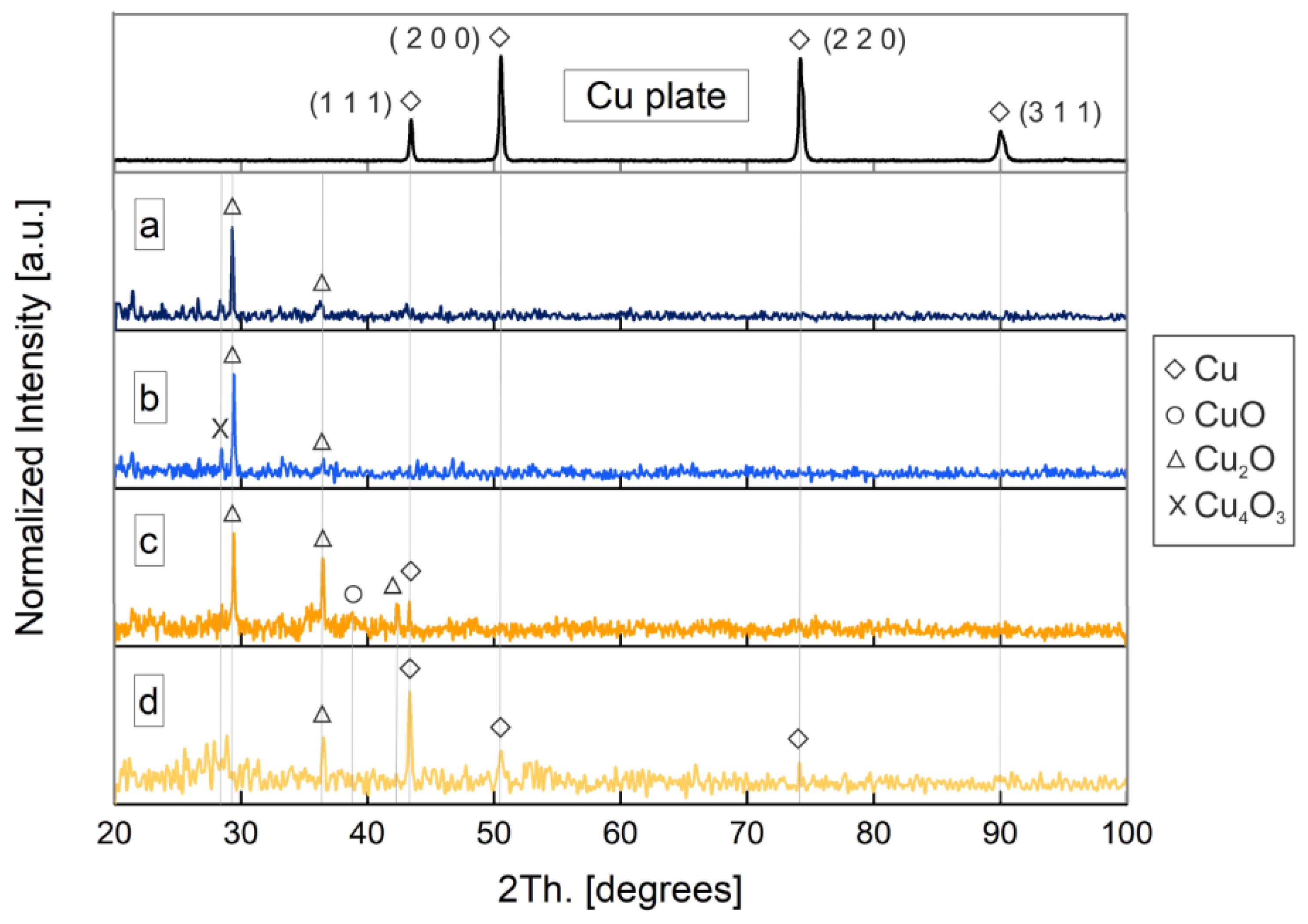
3.3.2. Composition and Crystallography
HRTEM, FFT and SAED
XRD
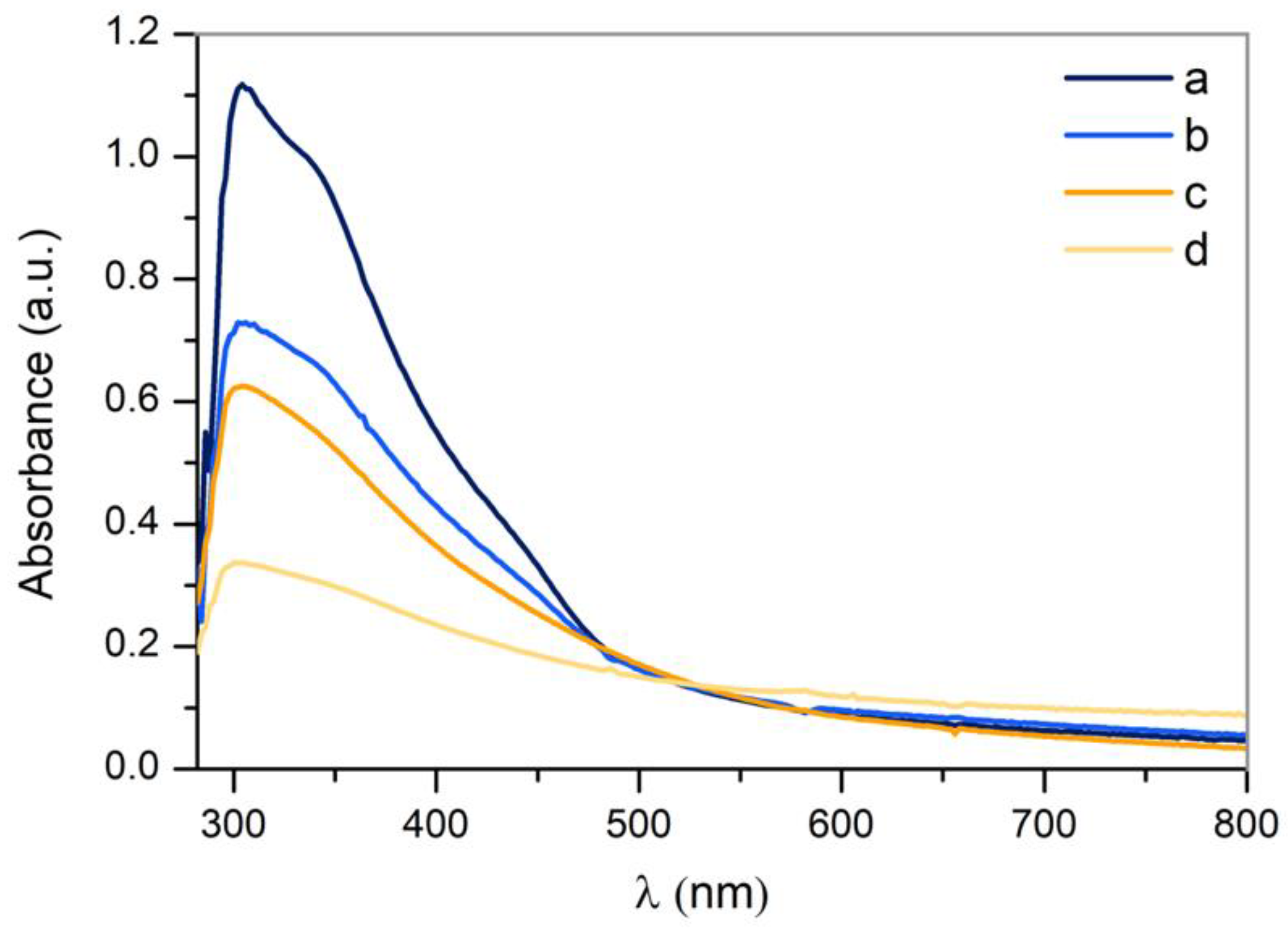
3.3.3. UV-VIS Absorption
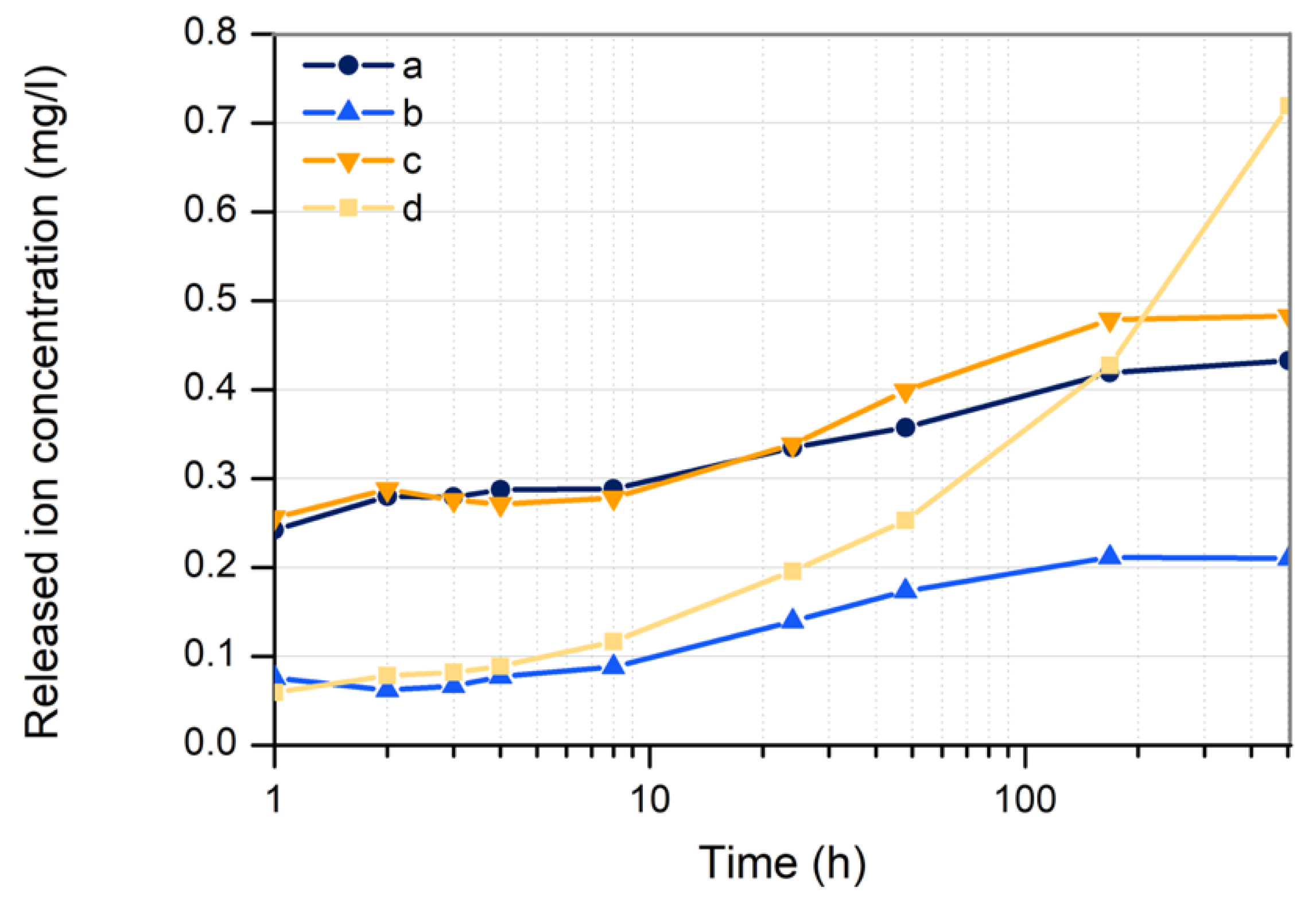
3.3.4. Copper Ion Release
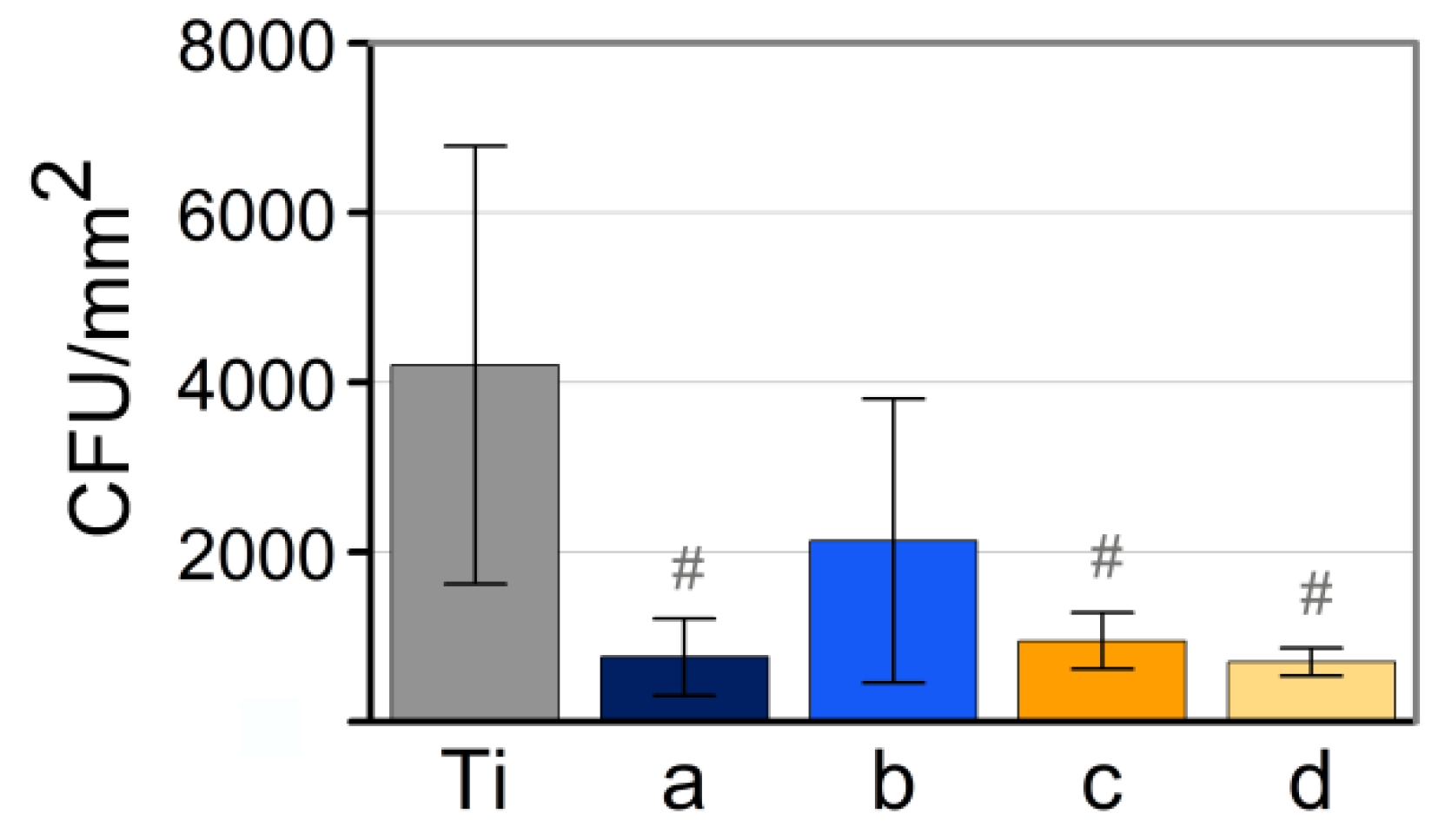
3.4. Analysis of Antimicrobial Activity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Reardon, S. WHO warns against ‘post-antibiotic’ era. Nature 2014. [Google Scholar] [CrossRef]
- Willyard, C. Drug-resistant bacteria ranked. Nature 2017, 543, 7643. [Google Scholar] [CrossRef]
- Ondusko, D.S.; Nolt, D. Staphylococcus aureus. Pediatr. Rev. 2018, 39, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Goudouri, O.; Kontonasaki, E.; Lohbauer, U.; Boccaccini, A.R. Antibacterial properties of metal and metalloid ions in chronic periodontitis and peri-implantitis therapy. Acta Biomater. 2014, 10, 3795–3810. [Google Scholar] [CrossRef] [PubMed]
- Massa, M.A.; Covarrubias, C.; Bittner, M.; Fuentevilla, I.A.; Capetillo, P.; von Marttens, A.; Carvajal, J.C. Synthesis of new antibacterial composite coating for titanium based on highly ordered nanoporous silica and silver nanoparticles. Mater. Sci. Eng. C 2014, 45, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Palza, H.; Quijada, R.; Delgado, K. Antimicrobial polymer composites with copper micro- and nanoparticles: Effect of particle size and polymer matrix. J. Bioact. Compat. Polym. 2015, 30, 1–15. [Google Scholar] [CrossRef]
- Khalid, H.; Shamaila, S.; Zafar, N.; Sharif, R.; Nazir, J.; Rafique, M.; Ghani, S.; Saba, H. Antibacterial Behavior of Laser-Ablated Copper Nanoparticles. Acta Metall. Sin. Engl. Lett. 2016, 29, 748–754. [Google Scholar] [CrossRef]
- World Health Organization. Trace Elements in Human Nutrition and Health; World Health Organization: Geneva, Switzerland, 1996; pp. 1–360. [Google Scholar]
- Karlin, K.D. Metalloenzymes, Structural Motifs, and Inorganic Models. Science 1993, 261, 701–708. [Google Scholar] [CrossRef]
- Skalnaya, M.G.; Skalny, A.V. Essential Trace Elements in Human Health: A Physician’S View; Publishing House of Tomsk State University: Tomsk, Russian, 2018. [Google Scholar]
- Vimbela, G.V.; Ngo, S.M.; Fraze, C.; Yang, L.; Stout, D.A. Antibacterial properties and toxicity from metallic nanomaterials. Int. J. Nanomed. 2017, 12, 3941–3965. [Google Scholar] [CrossRef]
- Wagener, P.; Jakobi, J.; Rehbock, C.; Chakravadhanula, V.S.K.; Thede, C.; Wiedwald, U.; Bartsch, M.; Kienle, L.; Barcikowski, S. Solvent-surface interactions control the phase structure in laser-generated iron-gold core-shell nanoparticles. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef]
- Boutinguiza, M.; Pou, J.; Lusquiños, F.; Comesaña, R.; Riveiro, A. Laser-assisted production of tricalcium phosphate nanoparticles from biological and synthetic hydroxyapatite in aqueous medium. Appl. Surf. Sci. 2011, 257, 5195–5199. [Google Scholar] [CrossRef]
- Boutinguiza, M.; Lusquiños, F.; Riveiro, A.; Comesaña, R.; Pou, J. Hydroxylapatite nanoparticles obtained by fiber laser-induced fracture. Appl. Surf. Sci. 2009, 255, 5382–5385. [Google Scholar] [CrossRef]
- Boutinguiza, M.; Comesaña, R.; Lusquiños, F.; Riveiro, A.; del Val, J.; Pou, J. Production of silver nanoparticles by laser ablation in open air. Appl. Surf. Sci. 2014, 336, 108–111. [Google Scholar] [CrossRef]
- Boutinguiza, M.; Fernández-Arias, M.; del Val, J.; Buxadera-Palomero, J.; Rodríguez, D.; Lusquiños, F.; Gil, F.J.; Pou, J. Synthesis and deposition of silver nanoparticles on cp Ti by laser ablation in open air for antibacterial effect in dental implants. Mater. Lett. 2018, 231, 126–129. [Google Scholar] [CrossRef]
- Fernández-Arias, M.; Boutinguiza, M.; del Val, J.; Covarrubias, C.; Bastias, F.; Gómez, L.; Maureira, M.; Arias-González, F.; Riveiro, A.; Pou, J. Copper nanoparticles obtained by laser ablation in liquids as bactericidal agent for dental applications. Appl. Surf. Sci. 2019, 507, 145032. [Google Scholar] [CrossRef]
- Vadillo, J.M.; Fernández, J.M.; Rodríguez, C.; Laserna, J.J. Effect of Plasma Shielding on Laser Ablation Rate of Pure Metals at Reduced Pressure. Surf. Interface Anal. 1999, 27, 1009–1015. [Google Scholar] [CrossRef]
- Gottfried, J.L. Influence of exothermic chemical reactions on laser-induced shock waves. Phys. Chem. Chem. Phys. 2014, 16, 21452–21466. [Google Scholar] [CrossRef]
- Maina, M.; Okamoto, Y.; Inoue, R.; Nakashiba, S.; Okada, A.; Sakagawa, T. Influence of Surface State in Micro-Welding of Copper by Nd:YAG Laser. Appl. Sci. 2018, 8, 2364. [Google Scholar] [CrossRef]
- Hermann, J.; Gerhard, C.; Axente, E.; Dutouquet, C. Comparative investigation of laser ablation plumes in air and argon by analysis of spectral line shapes: Insights on calibration-free laser-induced breakdown spectroscopy. Spectrochim. Acta Part B At. Spectrosc. 2014, 100, 189–196. [Google Scholar] [CrossRef]
- Hamad, A.H. Laser Ablation in Different Environments and Generation of Nanoparticles Generation. Appl. Laser Ablation Thin Film Depos. Nanomater. Synth. Surf. Modif. 2016, 177–196. [Google Scholar] [CrossRef]
- Yang, G.W. Laser ablation in liquids: Applications in the synthesis of nanocrystals. Prog. Mater. Sci. 2007, 52, 648–698. [Google Scholar] [CrossRef]
- Ingham, B. X-Ray Diffraction for Characterizing Metallic Films. In Metallic Films for Electronic, Optical and Magnetic Applications; Woodhead Publishing: Sawston, UK, 2014; pp. 3–38. [Google Scholar] [CrossRef]
- Raghav, R.; Aggarwal, P.; Srivastava, S. Tailoring oxides of copper-Cu2O and CuO nanoparticles and evaluation of organic dyes degradation. AIP Conf. Proc. 2016, 1724. [Google Scholar] [CrossRef]
- Zhu, J.; Li, D.; Chen, H.; Yang, X.; Lu, L.; Wang, X. Highly dispersed CuO nanoparticles prepared by a novel quick-precipitation method. Mater. Lett. 2004, 58, 3324–3327. [Google Scholar] [CrossRef]
- Khashan, K.S.; Jabir, M.S.; Abdulameer, F.A. Carbon Nanoparticles decorated with cupric oxide Nanoparticles prepared by laser ablation in liquid as an antibacterial therapeutic agent. Mater. Res. Express 2018, 5, 035003. [Google Scholar] [CrossRef]
- Díaz-Visurraga, J.; Daza, C.; Pozo, C.; Becerra, A.; von Plessing, C.; García, A. Study on antibacterial alginate-stabilized copper nanoparticles by FT-IR and 2D-IR correlation spectroscopy. Int. J. Nanomed. 2012, 7, 3597–3612. [Google Scholar] [CrossRef]
- Goncharova, D.; Lapin, I.; Svetlichnyi, V. Structure and optical properties of nanoparticles obtained by pulsed laser ablation of copper in gases. J. Phys. Conf. Ser. 2019, 1145, 1–9. [Google Scholar] [CrossRef]
- Mafune, F.; Kohno, J.; Takeda, Y.; Kondow, T. Formation and Size Control of Silver Nanoparticles by Laser Ablation in Aqueous Solution. J. Phys. Chem. B 2000, 104, 9111–9117. [Google Scholar] [CrossRef]
- Dadras, S.; Torkamany, M.J.; Jafarkhani, P. Analysis and optimization of silver nanoparticles laser synthesis with emission spectroscopy of induced plasma. J. Nanosci. Nanotechnol. 2012, 12, 3115–3122. [Google Scholar] [CrossRef]
- Truong, V.K.; Lapovok, R.; Estrin, Y.S.; Rundell, S.; Wang, J.Y.; Fluke, C.J.; Crawford, R.J.; Ivanova, E.P. The influence of nano-scale surface roughness on bacterial adhesion to ultrafine-grained titanium. Biomaterials 2010, 31, 3674–3683. [Google Scholar] [CrossRef]
- Schubert, A.; Wassmann, T.; Holtappels, M.; Kurbad, O.; Krohn, S.; Bürgers, R. Predictability of Microbial Adhesion to Dental Materials by Roughness Parameters. Coatings 2019, 9, 456. [Google Scholar] [CrossRef]
- Drelich, J.; Li, B.; Bowen, P.; Hwang, J.; Mills, O.; Hoffman, D. Vermiculite decorated with copper nanoparticles: Novel antibacterial hybrid material. Appl. Surf. Sci. 2011, 257, 9435–9443. [Google Scholar] [CrossRef]
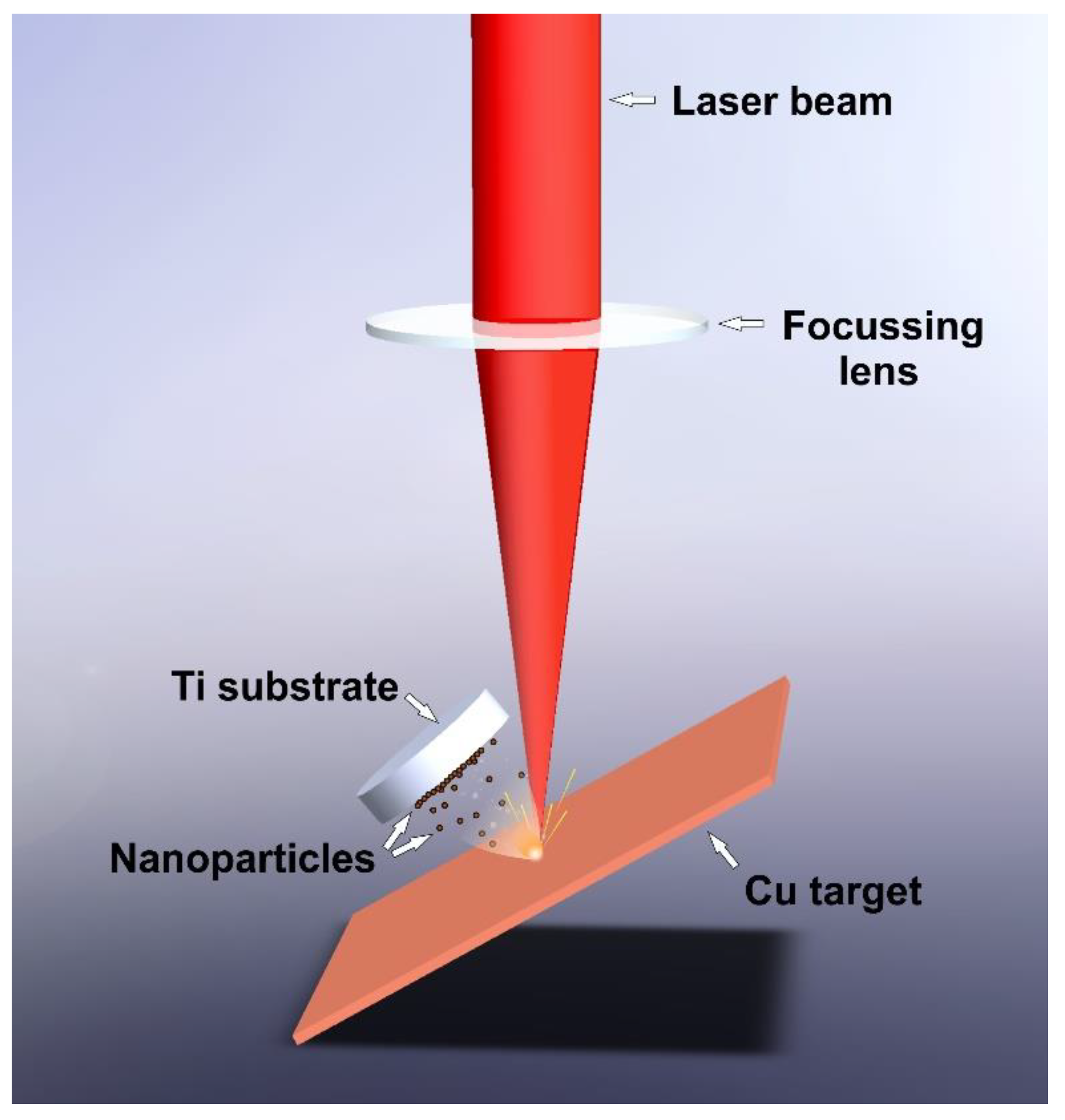





| Laser Source | Wavelength (nm) | Pulse Length (ns) | Pulse Frequency (kHz) | Pulse Energy (mJ) | Scanning Speed (mm/s) |
|---|---|---|---|---|---|
| Green-Nanosecond | 532 | 14 | 20 | 0.26 | 50 |
| IR-Picosecond | 1064 | 0.8 | 200 | 0.03 | 50 |
| Sample | Laser Source | Atmosphere | Time (min) |
|---|---|---|---|
| a | Green-Nanosecond | air | 5 |
| b | Green-Nanosecond | argon | 7 |
| c | IR-Picosecond | air | 2 |
| d | IR-Picosecond | argon | 3 |
| a | b | c | d | Cu (hkl) | CuO (hkl) | Cu2O (hkl) |
|---|---|---|---|---|---|---|
| 0.247 | 0.246 | 0.248 | 0.247 (111) | |||
| 0.234 | 0.231 (200) | |||||
| 0.208 | 0.209 (111) | |||||
| 0.214 | 0.216 | 0.213 | 0.214 (200) | |||
| 0.183 | 0.181 (200) | |||||
| 0.187 | 0.187 (−202) | |||||
| 0.152 | 0.151 | 0.151 | 0.151 | 0.150 (−113) | 0.151 (220) | |
| 0.129 | 0.132 | 0.129 | 0.128 | 0.128 (220) | 0.129 (311) | |
| 0.109 | 0.109 (311) | |||||
| 0.098 | 0.083 | 0.083 (331) | 0.098 (331) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Arias, M.; Boutinguiza, M.; del Val, J.; Riveiro, A.; Rodríguez, D.; Arias-González, F.; Gil, J.; Pou, J. Fabrication and Deposition of Copper and Copper Oxide Nanoparticles by Laser Ablation in Open Air. Nanomaterials 2020, 10, 300. https://doi.org/10.3390/nano10020300
Fernández-Arias M, Boutinguiza M, del Val J, Riveiro A, Rodríguez D, Arias-González F, Gil J, Pou J. Fabrication and Deposition of Copper and Copper Oxide Nanoparticles by Laser Ablation in Open Air. Nanomaterials. 2020; 10(2):300. https://doi.org/10.3390/nano10020300
Chicago/Turabian StyleFernández-Arias, Mónica, Mohamed Boutinguiza, Jesús del Val, Antonio Riveiro, Daniel Rodríguez, Felipe Arias-González, Javier Gil, and Juan Pou. 2020. "Fabrication and Deposition of Copper and Copper Oxide Nanoparticles by Laser Ablation in Open Air" Nanomaterials 10, no. 2: 300. https://doi.org/10.3390/nano10020300
APA StyleFernández-Arias, M., Boutinguiza, M., del Val, J., Riveiro, A., Rodríguez, D., Arias-González, F., Gil, J., & Pou, J. (2020). Fabrication and Deposition of Copper and Copper Oxide Nanoparticles by Laser Ablation in Open Air. Nanomaterials, 10(2), 300. https://doi.org/10.3390/nano10020300







