Innovative Smart Materials in Restorative Dentistry
Abstract
1. Introduction
2. Key Smart Materials in Restorative Dentistry
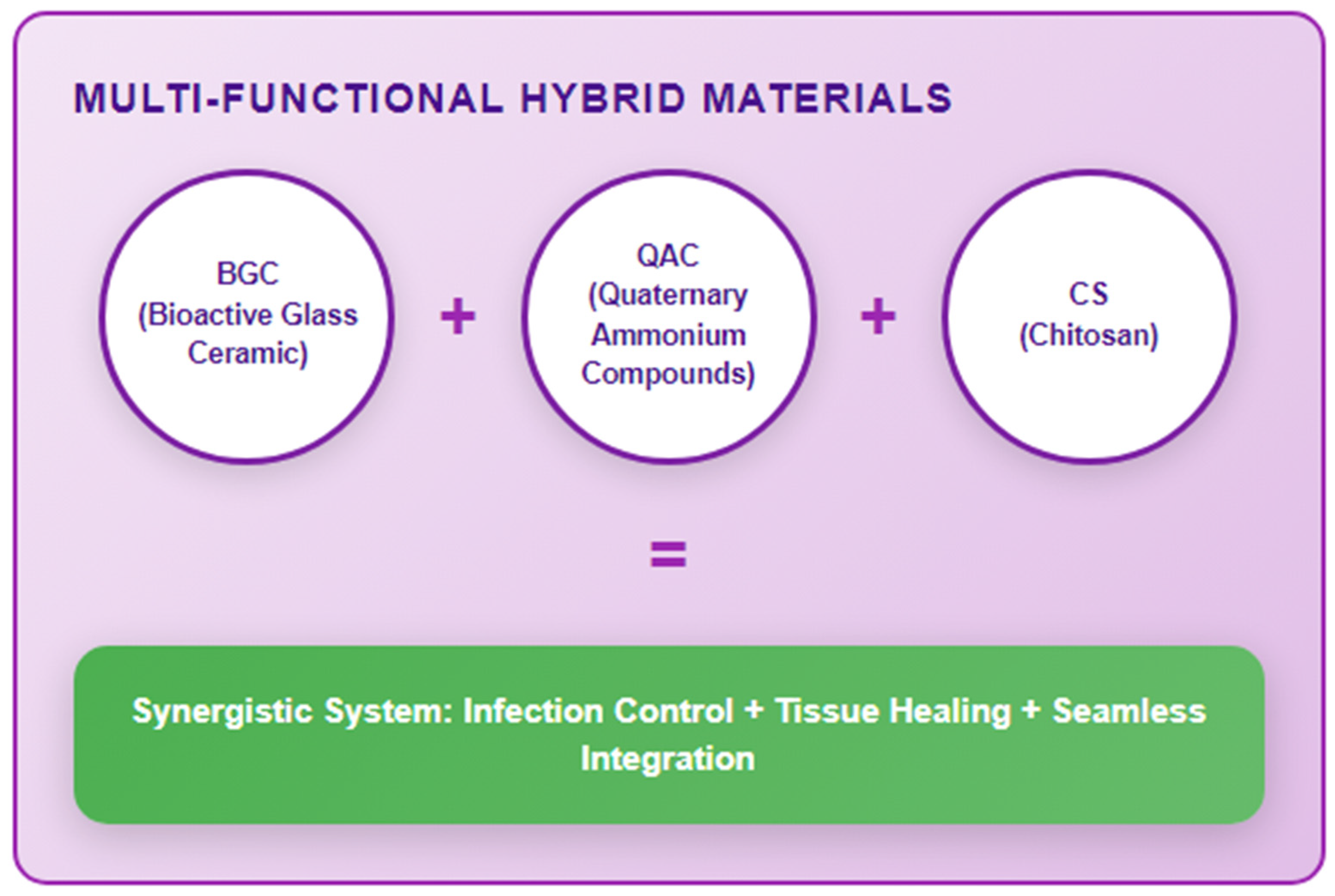
2.1. Bioactive Glass Ceramics (BGCs): Antimicrobial and Osteoconductive Dual Functionality
2.2. Silver Nanoparticle-Doped Polymers: Prolonged Antimicrobial Defense
2.3. Chitosan-Based pH-Responsive Coatings
3. Emerging Smart Materials in Dentistry: Quaternary Ammonium Compounds, Graphene Oxide, and Beyond
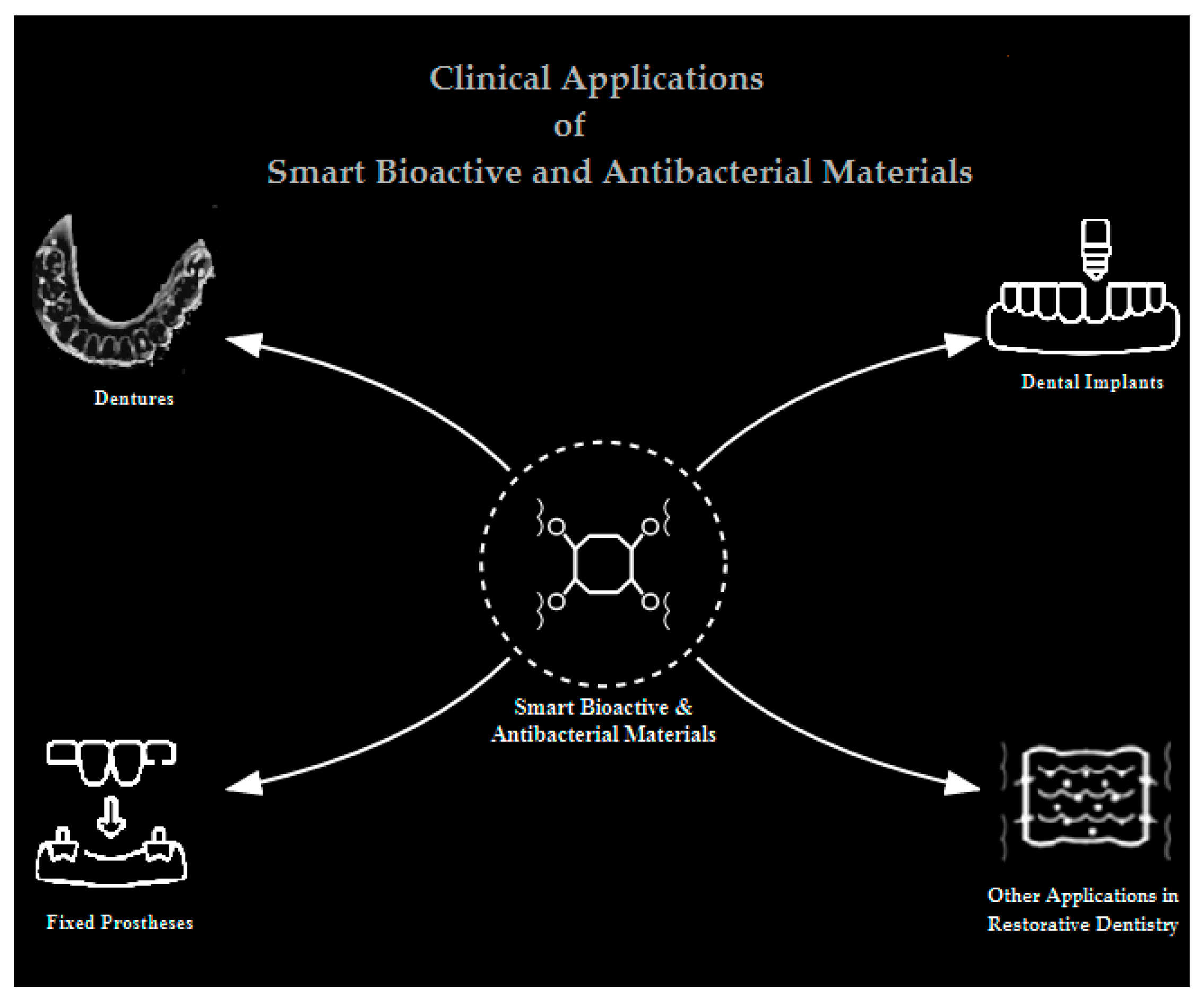
4. Clinical Applications in Prosthodontics
4.1. Dentures: Combating Candida Biofilms and Denture Stomatitis
4.1.1. Targeting Candida Biofilm Adhesion and Growth
4.1.2. Sustained Antimicrobial Protection in Tissue Conditioners
4.2. Dental Implants: Enhancing Osseointegration and Preventing Peri-Implantitis Through Advanced Surface Modifications
4.2.1. Surface Modifications for Antibacterial Efficacy
4.2.2. Enhancement of Osseointegration and Angiogenesis
4.2.3. Peri-Implantitis Prophylaxis and Treatment
4.3. Fixed Prostheses and Restorations
4.4. Other Restorative Applications: Expanding the Scope of Smart Materials
4.4.1. Bone Graft Substitutes and Enhancers
4.4.2. pH-Responsive Coatings for Targeted Therapy
- Denture liners: CS-citral-Zn2+ complexes release Zn2+ at low pH, reducing S. aureus and E. coli adhesion by 90% [123].
- Implant coatings:
- Sequential delivery systems: Pore-closed PLGA microparticles in CS hydrogels deliver vancomycin (burst) followed by rhBMP-2 (sustained), combining infection control and osseointegration [128].
4.4.3. Guided Bone Regeneration (GBR) Membranes
5. Discussion. Navigating Challenges and Charting Future Trajectories for Smart Bioactive Materials in Restorative Dentistry
5.1. Challenges Impeding Clinical Translation
- Cytotoxicity at high concentrations: Concentrations beyond 0.3 wt% in PMMA compromise fibroblast viability, necessitating diffusion-barrier strategies like silica-shell encapsulation or Zn2+ co-doping to suppress pro-apoptotic pathways.
- Nanoparticle aggregation: Uncontrolled agglomeration reduces antimicrobial efficacy and increases localized toxicity. Advanced stabilization methods, such as UiO-66 MOF confinement or PEG-based steric shielding, are critical to maintaining colloidal stability.
- Fungal resistance: C. albicans adapts via EPS production and efflux mechanisms, prompting the development of synergistic agents like chitosan-AgNP hybrids or curcumin-functionalized AgNPs to disrupt biofilm defenses (Figure 6).
5.2. Envisioning Future Directions: Innovation and Standardization
5.3. Future Research
5.4. Limitations and Strenghts of This Review
- Integrates pH/enzyme-responsive mechanisms with clinical outcomes (e.g., Section 4 on denture stomatitis and peri-implantitis).
- Critically evaluates translational challenges (e.g., Table 3 on cytotoxicity thresholds) and proposes standardized protocols.
- Highlights innovations like 4D-printed hydrogels and AI-driven design (Section 5.2), not previously reviewed in this context.
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PMMA | Polymethyl methacrylate |
| Ag+ | Silver ion |
| Zn2+ | Zinc ion |
| Sr2+ | Strontium ion |
| BG | Bioactive glass |
| BGCs | Bioactive glass ceramics |
| S53P4 | A specific bioactive glass composition (53% SiO2, 22% Na2O, 21% CaO, 4% P2O5) |
| F18 | A bioactive glass formulation |
| HCA | Hydroxycarbonate apatite |
| RUNX2 | Runt-related transcription factor 2 (osteogenic marker) |
| VEGF | Vascular endothelial growth factor |
| Zr-BGCs | Zirconium-modified bioactive glass ceramics |
| Nb2O5 | Niobium pentoxide |
| Cu-MBG | Copper-doped mesoporous bioactive glass |
| AgNPs | Silver nanoparticles |
| ROS | Reactive oxygen species |
| β-AgVO3 | Silver vanadate (antimicrobial agent) |
| UiO-66 | A zirconium-based metal–organic framework (MOF) |
| nHA | Nano-hydroxyapatite |
| Ag-Chi | Chitosan-conjugated silver nanoparticles |
| CS | Chitosan |
| GIC | Glass ionomer cement |
| CSNPs | Chitosan nanoparticles |
| PLGA | Poly(lactic-co-glycolic acid) |
| rhBMP-2 | Recombinant human bone morphogenetic protein-2 |
| QAM | Quaternary ammonium monomers |
| QACs | Quaternary ammonium compounds |
| QPEI | Quaternary ammonium-polyethyleneimine |
| GO | Graphene oxide |
| ZIF-8 | Zeolitic imidazolate framework-8 |
| TiO2 | Titanium dioxide |
| MoSe2 | Molybdenum diselenide |
| UDMA | Urethane dimethacrylate |
| Ag-QAS | Silver-quaternary ammonium silane |
| LBL | Layer-by-layer (coating technique) |
| CFU | Colony-forming unit |
| MBG | Mesoporous bioactive glass |
| Ag-HA | Silver-doped hydroxyapatite |
| LSTC | Lysostaphin-chitosan hydrogel |
| CBD | Cannabidiol (in experimental PMMA formulations) |
| IVIVC | In vitro-in vivo correlation |
References
- Banu Raza, F.; Vijayaraghavalu, S.; Kandasamy, R.; Krishnaswami, V.; Kumar, V.A. Microbiome and the Inflammatory Pathway in Peri-Implant Health and Disease with an Updated Review on Treatment Strategies. J. Oral Biol. Craniofacial Res. 2023, 13, 84–91. [Google Scholar] [CrossRef]
- Lasserre, J.F.; Brecx, M.C.; Toma, S. Oral Microbes, Biofilms and Their Role in Periodontal and Peri-Implant Diseases. Materials 2018, 11, 1802. [Google Scholar] [CrossRef]
- Kashyap, B.; Padala, S.R.; Kaur, G.; Kullaa, A. Candida Albicans Induces Oral Microbial Dysbiosis and Promotes Oral Diseases. Microorganisms 2024, 12, 2138. [Google Scholar] [CrossRef]
- Albani, R.; Habib, S.R.; AlQahtani, A.; AlHelal, A.A.; Alrabiah, M. Streptococcus-Mutans and Porphyromonas-Gingivalis Adhesion to Glazed/Polished Surfaces of CAD/CAM Restorations. Heliyon 2024, 10, e40276. [Google Scholar] [CrossRef]
- Du, Q.; Ren, B.; Zhou, X.; Zhang, L.; Xu, X. Cross-Kingdom Interaction between Candida Albicans and Oral Bacteria. Front. Microbiol. 2022, 13, 911623. [Google Scholar] [CrossRef]
- Metwalli, K.H.; Khan, S.A.; Krom, B.P.; Jabra-Rizk, M.A. Streptococcus Mutans, Candida Albicans, and the Human Mouth: A Sticky Situation. PLoS Pathog. 2013, 9, e1003616. [Google Scholar] [CrossRef]
- Wijesinghe, G.K.; Nobbs, A.H.; Bandara, H.M.H.N. Cross-Kingdom Microbial Interactions within the Oral Cavity and Their Implications for Oral Disease. Curr. Clin. Microbiol. Rep. 2023, 10, 29–35. [Google Scholar] [CrossRef]
- Ray, R.R. Dental Biofilm: Risks, Diagnostics and Management. Biocatal. Agric. Biotechnol. 2022, 43, 102381. [Google Scholar] [CrossRef]
- Sim, C.P.C.; Dashper, S.G.; Reynolds, E.C. Oral Microbial Biofilm Models and Their Application to the Testing of Anticariogenic Agents. J. Dent. 2016, 50, 1–11. [Google Scholar] [CrossRef]
- Dubey, S.; Dubey, S.; Gupta, A.; Sharma, V. Biofilm-Mediated Dental Diseases. In Biofilms in Human Diseases: Treatment and Control; Kumar, S., Chandra, N., Singh, L., Hashmi, M.Z., Varma, A., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2019; pp. 91–116. ISBN 9783030307578. [Google Scholar]
- Ma, W.; Huang, Z.; Zhang, Y.; Liu, K.; Li, D.; Liu, Q. Interaction between Inflammation and Biofilm Infection and Advances in Targeted Biofilm Therapy Strategies. Microbiol. Res. 2025, 298, 128199. [Google Scholar] [CrossRef]
- Kreve, S.; Cândido Dos Reis, A. Antibiofilm Capacity of PMMA Surfaces: A Review of Current Knowledge. Microb. Pathog. 2025, 202, 107426. [Google Scholar] [CrossRef]
- Zore, A.; Abram, A.; Učakar, A.; Godina, I.; Rojko, F.; Štukelj, R.; Škapin, A.S.; Vidrih, R.; Dolic, O.; Veselinovic, V.; et al. Antibacterial Effect of Polymethyl Methacrylate Resin Base Containing TiO2 Nanoparticles. Coatings 2022, 12, 1757. [Google Scholar] [CrossRef]
- Alrahlah, A.; Fouad, H.; Hashem, M.; Niazy, A.A.; AlBadah, A. Titanium Oxide (TiO2)/Polymethylmethacrylate (PMMA) Denture Base Nanocomposites: Mechanical, Viscoelastic and Antibacterial Behavior. Materials 2018, 11, 1096. [Google Scholar] [CrossRef] [PubMed]
- Ali Sabri, B.; Satgunam, M.; Abreeza, N.N.; Abed, A. A Review on Enhancements of PMMA Denture Base Material with Different Nano-Fillers. Cogent Eng. 2021, 8, 1875968. [Google Scholar] [CrossRef]
- Kresse-Walczak, K.; Meissner, H.; Mauer, R.; Trips, E.; Boening, K. Evaluation of a Protocol to Assess a Novel Artificial Biofilm Equivalent for Dentures—A Prospective Clinical Pilot Study. Gerodontology 2024, 41, 368–375. [Google Scholar] [CrossRef]
- Carvalho-Silva, J.M.; Gaspar, C.S.; Dos Reis, A.C.; Teixeira, A.B.V. Denture Stomatitis: Treatment with Antimicrobial Drugs or Antifungal Gels? A Systematic Review of Clinical Trials. J. Prosthet. Dent. 2024. [Google Scholar] [CrossRef]
- Bukhari, M.A.; Algahtani, M.A.; Alsuwailem, F.A.; Alogaiel, R.M.; Almubarak, S.H.; Alqahtani, S.S.; Alabdullatif, R.A.; Alghimlas, R.Y.; Alotaibi, N.F.; Qahtani, A.R.A.; et al. Epidemiology, Etiology, and Treatment of Denture Stomatitis. Int. J. Community Med. Public Health 2022, 9, 981–986. [Google Scholar] [CrossRef]
- Abuhajar, E.; Ali, K.; Zulfiqar, G.; Al Ansari, K.; Raja, H.Z.; Bishti, S.; Anweigi, L. Management of Chronic Atrophic Candidiasis (Denture Stomatitis)—A Narrative Review. Int. J. Environ. Res. Public Health 2023, 20, 3029. [Google Scholar] [CrossRef]
- Haque, M.; Sartelli, M.; Haque, S.Z. Dental Infection and Resistance—Global Health Consequences. Dent. J. 2019, 7, 22. [Google Scholar] [CrossRef]
- Khan, N.; Waleed, A.; Ali Shah, S.I.; Rashid, M.U.; Ali, H.; Waheed, K. Studying the Implications of Antibiotics Resistance in Dental Infection. Indus J. Biosci. Res. 2024, 2, 171–177. [Google Scholar] [CrossRef]
- Yu, K.; Zhang, Q.; Dai, Z.; Zhu, M.; Xiao, L.; Zhao, Z.; Bai, Y.; Zhang, K. Smart Dental Materials Intelligently Responding to Oral PH to Combat Caries: A Literature Review. Polymers 2023, 15, 2611. [Google Scholar] [CrossRef]
- Sabzevari, B.; Malekzade, M. The Science Behind Bioactive Dental Adhesive Systems: Benefits and Applications. Int. J. Med. Sci. Dent. Health 2024, 10, 112–130. [Google Scholar] [CrossRef]
- Ul Haq, I.; Krukiewicz, K. Antimicrobial Approaches for Medical Implants Coating to Prevent Implants Associated Infections: Insights to Develop Durable Antimicrobial Implants. Appl. Surf. Sci. Adv. 2023, 18, 100532. [Google Scholar] [CrossRef]
- Ahmed, W.; Zhai, Z.; Gao, C. Adaptive Antibacterial Biomaterial Surfaces and Their Applications. Mater. Today Bio 2019, 2, 100017. [Google Scholar] [CrossRef]
- Wu, Z.; Nie, R.; Wang, Y.; Wang, Q.; Li, X.; Liu, Y. Precise Antibacterial Therapeutics Based on Stimuli-Responsive Nanomaterials. Front. Bioeng. Biotechnol. 2023, 11, 1289323. [Google Scholar] [CrossRef]
- Morris, G.; Goodman, S.; Sorzabal Bellido, I.; Milanese, C.; Girella, A.; Pallavicini, P.; Taglietti, A.; Gaboardi, M.; Jäckel, F.; Diaz Fernandez, Y.A.; et al. Temperature and PH Stimuli-Responsive System Delivers Location-Specific Antimicrobial Activity with Natural Products. ACS Appl. Bio Mater. 2024, 7, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Jafari, N.; Habashi, M.S.; Hashemi, A.; Shirazi, R.; Tanideh, N.; Tamadon, A. Application of Bioactive Glasses in Various Dental Fields. Biomater. Res. 2022, 26, 31. [Google Scholar] [CrossRef] [PubMed]
- Kaou, M.H.; Furkó, M.; Balázsi, K.; Balázsi, C. Advanced Bioactive Glasses: The Newest Achievements and Breakthroughs in the Area. Nanomaterials 2023, 13, 2287. [Google Scholar] [CrossRef] [PubMed]
- Harun-Ur-Rashid, M.; Foyez, T.; Krishna, S.B.N.; Poda, S.; Imran, A.B. Recent Advances of Silver Nanoparticle-Based Polymer Nanocomposites for Biomedical Applications. RSC Adv. 2025, 15, 8480–8505. [Google Scholar] [CrossRef]
- Guo, Y.; Hou, X.; Fan, S.; Jin, C. Research Status of Silver Nanoparticles for Dental Applications. Inorganics 2025, 13, 168. [Google Scholar] [CrossRef]
- Paradowska-Stolarz, A.; Mikulewicz, M.; Laskowska, J.; Karolewicz, B.; Owczarek, A. The Importance of Chitosan Coatings in Dentistry. Mar. Drugs 2023, 21, 613. [Google Scholar] [CrossRef]
- Mahmood, A.; Maher, N.; Amin, F.; Alqutaibi, A.Y.; Kumar, N.; Zafar, M.S. Chitosan-Based Materials for Dental Implantology: A Comprehensive Review. Int. J. Biol. Macromol. 2024, 268, 131823. [Google Scholar] [CrossRef]
- Obermeier, F.; Mutschlechner, M.; Haller, S.; Schöbel, H.; Strube, O.I. Sustainable Antimicrobial and Antibiofilm Strategies: Monoterpene-Based Compounds as Potential Substitute for Low-Molecular-Weight Biocides in the Coating Industry. Eur. Polym. J. 2025, 225, 113734. [Google Scholar] [CrossRef]
- Mitwalli, H.; Alsahafi, R.; Balhaddad, A.A.; Weir, M.D.; Xu, H.H.K.; Melo, M.A.S. Emerging Contact-Killing Antibacterial Strategies for Developing Anti-Biofilm Dental Polymeric Restorative Materials. Bioengineering 2020, 7, 83. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.; Song, W.; Song, H.; Kang, D.K.; Park, S.; Seong, M.; Jeong, H.E. Sustainable Biofilm Inhibition Using Chitosan-Mesoporous Nanoparticle-Based Hybrid Slippery Composites. ACS Appl. Mater. Interfaces 2024, 16, 27728–27740. [Google Scholar] [CrossRef] [PubMed]
- Thirumalaivasan, N.; Nangan, S.; Verma, D.; Shellaiah, M.; Ali, S.; Rajendran, S.; Kanagaraj, K.; Pothu, R.; Boddula, R.; Radwan, A.B.; et al. Exploring the Diverse Nanomaterials Employed in Dental Prosthesis and Implant Techniques: An Overview. Nanotechnol. Rev. 2025, 14. [Google Scholar] [CrossRef]
- Qi, J.; Si, C.; Liu, H.; Li, H.; Kong, C.; Wang, Y.; Chang, B. Advances of Metal-Based Nanomaterials in the Prevention and Treatment of Oral Infections. Adv. Healthc. Mater. 2025, 14, 2500416. [Google Scholar] [CrossRef]
- Maher, N.; Mahmood, A.; Fareed, M.A.; Kumar, N.; Rokaya, D.; Zafar, M.S. An Updated Review and Recent Advancements in Carbon-Based Bioactive Coatings for Dental Implant Applications. J. Adv. Res. 2025, 72, 265–286. [Google Scholar] [CrossRef]
- Dini, C.; Borges, M.H.R.; Malheiros, S.S.; Piazza, R.D.; Van Den Beucken, J.J.J.P.; De Avila, E.D.; Souza, J.G.S.; Barão, V.A.R. Progress in Designing Therapeutic Antimicrobial Hydrogels Targeting Implant-associated Infections: Paving the Way for a Sustainable Platform Applied to Biomedical Devices. Adv. Healthc. Mater. 2025, 14, 2402926. [Google Scholar] [CrossRef]
- Piccoli, C.; Soliani, G.; Piccoli, P.; Zupi, A. Long-Term Success in Dental Implant Revisions: A 31-Year Case Study of Alveolar Atrophy Management in an Elderly Woman. Am. J. Case Rep. 2024, 25. [Google Scholar] [CrossRef]
- Simão, B.S.; Costa, D.D.; Cangussu, M.C.T.; Sotto-Maior, B.S.; Devita, R.L.; De Carvalho, J.J.; Da Silva Brum, I. Observational Study on the Success Rate of Osseointegration: A Prospective Analysis of 15,483 Implants in a Public Health Setting. BioMed 2022, 2, 422–430. [Google Scholar] [CrossRef]
- Alves, M.A.L.; Both, J.; Mourão, C.F.; Ghiraldini, B.; Bezerra, F.; Granjeiro, J.M.; Sartoretto, S.C.; Calasans-Maia, M.D. Long-Term Success of Dental Implants in Atrophic Maxillae: A 3-Year Case Series Using Hydroxyapatite and L-PRF. Bioengineering 2024, 11, 1207. [Google Scholar] [CrossRef]
- Zhang, K.; Le, Q.V. Bioactive Glass Coated Zirconia for Dental Implants: A Review. J. Compos. Compd. 2020, 2, 10–17. [Google Scholar] [CrossRef]
- Drevet, R.; Fauré, J.; Benhayoune, H. Electrophoretic Deposition of Bioactive Glass Coatings for Bone Implant Applications: A Review. Coatings 2024, 14, 1084. [Google Scholar] [CrossRef]
- Skallevold, H.E.; Rokaya, D.; Khurshid, Z.; Zafar, M.S. Bioactive Glass Applications in Dentistry. Int. J. Mol. Sci. 2019, 20, 5960. [Google Scholar] [CrossRef]
- Fernando, D.; Attik, N.; Pradelle-Plasse, N.; Jackson, P.; Grosgogeat, B.; Colon, P. Bioactive Glass for Dentin Remineralization: A Systematic Review. Mater. Sci. Eng. C 2017, 76, 1369–1377. [Google Scholar] [CrossRef] [PubMed]
- Park, I.-S.; Kim, H.-J.; Kwon, J.; Kim, D.-S. Comparative In Vitro Study of Sol–Gel-Derived Bioactive Glasses Incorporated into Dentin Adhesives: Effects on Remineralization and Mechanical Properties of Dentin. J. Funct. Biomater. 2025, 16, 29. [Google Scholar] [CrossRef] [PubMed]
- Seifi, M.; Eskandarloo, F.; Amdjadi, P.; Farmany, A. Investigation of Mechanical Properties, Remineralization, Antibacterial Effect, and Cellular Toxicity of Composite Orthodontic Adhesive Combined with Silver-Containing Nanostructured Bioactive Glass. BMC Oral Heal. 2024, 24, 650. [Google Scholar] [CrossRef] [PubMed]
- Gad, M.M.; Abu-Rashid, K.; Alkhaldi, A.; Alshehri, O.; Khan, S.Q. Evaluation of the Effectiveness of Bioactive Glass Fillers against Candida Albicans Adhesion to PMMA Denture Base Materials: An in Vitro Study. Saudi Dent. J. 2022, 34, 730–737. [Google Scholar] [CrossRef]
- Villar, C.; Lin, A.; Cao, Z.; Zhao, X.; Wu, L.; Chen, S.; Sun, Y.; Yeh, C. Anticandidal Activity and Biocompatibility of a Rechargeable Antifungal Denture Material. Oral Dis. 2013, 19, 287–295. [Google Scholar] [CrossRef]
- Lindfors, N.C.; Hyvönen, P.; Nyyssönen, M.; Kirjavainen, M.; Kankare, J.; Gullichsen, E.; Salo, J. Bioactive Glass S53P4 as Bone Graft Substitute in Treatment of Osteomyelitis. Bone 2010, 47, 212–218. [Google Scholar] [CrossRef]
- Barrak, F.N.; Li, S.; Mohammed, A.A.; Myant, C.; Jones, J.R. Anti-Inflammatory Properties of S53P4 Bioactive Glass Implant Material. J. Dent. 2022, 127, 104296. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, D.M.B.; Rosso, M.P.D.O.; Santos, P.S.D.S.; Sousa-Neto, M.D.; Silva-Sousa, A.C.; Soares, C.T.; Reis, C.H.B.; Rossi, J.D.O.; Bueno, C.R.D.S.; Buchaim, D.V.; et al. Biological Behavior of Bioactive Glasses SinGlass (45S5) and SinGlass High (F18) in the Repair of Critical Bone Defects. Biomolecules 2025, 15, 112. [Google Scholar] [CrossRef] [PubMed]
- Augusto, T.A.; Crovace, M.C.; Pinto, L.A.; Costa, L.C. Polycaprolactone/F18 Bioactive Glass Scaffolds Obtained via Fused Filament Fabrication. ACS Appl. Polym. Mater. 2025, 7, 2359–2370. [Google Scholar] [CrossRef]
- Marin, C.P.; Crovace, M.C.; Zanotto, E.D. Competent F18 Bioglass-Biosilicate® Bone Graft Scaffold Substitutes. J. Eur. Ceram. Soc. 2021, 41, 7910–7920. [Google Scholar] [CrossRef]
- De Paula, K.D.S.; Dos Reis-Prado, A.H.; De Jesus, W.P.; Goto, J.; De Arantes, L.C.; Verçosa, M.; Cintra, L.T.A.; Ervolino, E.; Szawka, R.E.; Crovace, M.C.; et al. Final Irrigation with Bioglass Solution in Regenerative Endodontic Procedure Induces Tissue Formation inside the Root Canals, Collagen Maturation, Proliferation Cell and Presence of Osteocalcin. Int. Endod. J. 2024, 57, 586–600. [Google Scholar] [CrossRef]
- Fuchs, M.; Gentleman, E.; Shahid, S.; Hill, R.G.; Brauer, D.S. Therapeutic Ion-Releasing Bioactive Glass Ionomer Cements with Improved Mechanical Strength and Radiopacity. Front. Mater. 2015, 2. [Google Scholar] [CrossRef]
- Rabiee, S.M.; Nazparvar, N.; Azizian, M.; Vashaee, D.; Tayebi, L. Effect of Ion Substitution on Properties of Bioactive Glasses: A Review. Ceram. Int. 2015, 41, 7241–7251. [Google Scholar] [CrossRef]
- Sidhu, V.P.S.; Borges, R.; Yusuf, M.; Mahmoudi, S.; Ghorbani, S.F.; Hosseinikia, M.; Salahshour, P.; Sadeghi, F.; Arefian, M. A Comprehensive Review of Bioactive Glass: Synthesis, Ion Substitution, Application, Challenges, and Future Perspectives. J. Compos. Compd. 2021, 3, 247–261. [Google Scholar] [CrossRef]
- Han, J.; Hassani Besheli, N.; Deng, D.; Van Oirschot, B.A.J.A.; Leeuwenburgh, S.C.G.; Yang, F. Tailoring Copper-Doped Bioactive Glass/Chitosan Coatings with Angiogenic and Antibacterial Properties. Tissue Eng. Part C Methods 2022, 28, 314–324. [Google Scholar] [CrossRef]
- Han, J.; Andrée, L.; Deng, D.; Van Oirschot, B.A.J.A.; Plachokova, A.S.; Leeuwenburgh, S.C.G.; Yang, F. Biofunctionalization of Dental Abutments by a Zinc/Chitosan/Gelatin Coating to Optimize Fibroblast Behavior and Antibacterial Properties. J Biomed. Mater. Res 2024, 112, 1873–1892. [Google Scholar] [CrossRef]
- Hammami, I.; Gavinho, S.R.; Pádua, A.S.; Sá-Nogueira, I.; Silva, J.C.; Borges, J.P.; Valente, M.A.; Graça, M.P.F. Bioactive Glass Modified with Zirconium Incorporation for Dental Implant Applications: Fabrication, Structural, Electrical, and Biological Analysis. Int. J. Mol. Sci. 2023, 24, 10571. [Google Scholar] [CrossRef]
- Khvostenko, D.; Hilton, T.J.; Ferracane, J.L.; Mitchell, J.C.; Kruzic, J.J. Bioactive Glass Fillers Reduce Bacterial Penetration into Marginal Gaps for Composite Restorations. Dent. Mater. 2016, 32, 73–81. [Google Scholar] [CrossRef]
- Sergi, R.; Bellucci, D.; Salvatori, R.; Anesi, A.; Cannillo, V. A Novel Bioactive Glass Containing Therapeutic Ions with Enhanced Biocompatibility. Materials 2020, 13, 4600. [Google Scholar] [CrossRef]
- Al-Harbi, N.; Mohammed, H.; Al-Hadeethi, Y.; Bakry, A.S.; Umar, A.; Hussein, M.A.; Abbassy, M.A.; Vaidya, K.G.; Al Berakdar, G.; Mkawi, E.M.; et al. Silica-Based Bioactive Glasses and Their Applications in Hard Tissue Regeneration: A Review. Pharmaceuticals 2021, 14, 75. [Google Scholar] [CrossRef]
- Taye, M.B.; Ningsih, H.S.; Shih, S.-J. Exploring the Advancements in Surface-Modified Bioactive Glass: Enhancing Antibacterial Activity, Promoting Angiogenesis, and Modulating Bioactivity. J. Nanoparticle Res. 2024, 26, 28. [Google Scholar] [CrossRef]
- Krishnan, L.; Chakrabarty, P.; Govarthanan, K.; Rao, S.; Santra, T.S. Bioglass and Nano Bioglass: A next-Generation Biomaterial for Therapeutic and Regenerative Medicine Applications. Int. J. Biol. Macromol. 2024, 277, 133073. [Google Scholar] [CrossRef]
- Sarin, S.; Rekhi, A. Bioactive Glass: A Potential next Generation Biomaterial. SRM J. Res. Dent. Sci. 2016, 7, 27. [Google Scholar] [CrossRef]
- Meskher, H.; Sharifianjazi, F.; Tavamaishvili, K.; Irandoost, M.; Nejadkoorki, D.; Makvandi, P. Limitations, Challenges and Prospective Solutions for Bioactive Glasses-Based Nanocomposites for Dental Applications: A Critical Review. J. Dent. 2024, 150, 105331. [Google Scholar] [CrossRef] [PubMed]
- Piatti, E.; Miola, M.; Verné, E. Tailoring of Bioactive Glass and Glass-Ceramics Properties for in Vitro and in Vivo Response Optimization: A Review. Biomater. Sci. 2024, 12, 4546–4589. [Google Scholar] [CrossRef]
- Yoshida, Y.G.; Yan, S.; Xu, H.; Yang, J. Novel Metal Nanomaterials to Promote Angiogenesis in Tissue Regeneration. Eng. Regen. 2023, 4, 265–276. [Google Scholar] [CrossRef]
- Shearer, A.; Montazerian, M.; Mauro, J.C. Modern Definition of Bioactive Glasses and Glass-Ceramics. J. Non-Cryst. Solids 2023, 608, 122228. [Google Scholar] [CrossRef]
- Kumar, A.; Mariappan, C.R. A Review on Embedding Therapeutic Nanoparticles in Mesoporous Bioactive Glass-Ceramics for Biomedical Applications. Inorg. Chem. Commun. 2025, 179, 114855. [Google Scholar] [CrossRef]
- Hallmann, L.; Gerngross, M.-D. Antibacterial Ceramics for Dental Applications. Appl. Sci. 2025, 15, 4553. [Google Scholar] [CrossRef]
- Pant, G.; Krishna, B.V.; Abed, N.K.; Nagpal, A.; Meheta, A. Advances in Dental Materials: Bioactive Glass and Ceramic Composites: A Review. E3S Web Conf. 2024, 505, 01026. [Google Scholar] [CrossRef]
- Montazerian, M.; Baino, F.; Fiume, E.; Migneco, C.; Alaghmandfard, A.; Sedighi, O.; DeCeanne, A.V.; Wilkinson, C.J.; Mauro, J.C. Glass-Ceramics in Dentistry: Fundamentals, Technologies, Experimental Techniques, Applications, and Open Issues. Prog. Mater. Sci. 2023, 132, 101023. [Google Scholar] [CrossRef]
- Vargas-Blanco, D.; Lynn, A.; Rosch, J.; Noreldin, R.; Salerni, A.; Lambert, C.; Rao, R.P. A Pre-Therapeutic Coating for Medical Devices That Prevents the Attachment of Candida Albicans. Ann. Clin. Microbiol. Antimicrob. 2017, 16, 41. [Google Scholar] [CrossRef]
- Farmani, M.; Mirahmadi-Zare, S.Z.; Masaeli, E.; Tabatabaei, F.; Houreh, A.B. Macroporous Coating of Silver-Doped Hydroxyapatite/Silica Nanocomposite on Dental Implants by EDTA Intermediate to Improve Osteogenesis, Antibacterial, and Corrosion Behavior. Biomed. Mater. 2025, 20, 025010. [Google Scholar] [CrossRef]
- Miola, M.; Vitale-Brovarone, C.; Mattu, C.; Verné, E. Antibiotic Loading on Bioactive Glasses and Glass-Ceramics: An Approach to Surface Modification. J. Biomater. Appl. 2013, 28, 308–319. [Google Scholar] [CrossRef]
- Gerhardt, L.-C.; Boccaccini, A.R. Bioactive Glass and Glass-Ceramic Scaffolds for Bone Tissue Engineering. Materials 2010, 3, 3867–3910. [Google Scholar] [CrossRef]
- Sharifianjazi, F.; Sharifianjazi, M.; Irandoost, M.; Tavamaishvili, K.; Mohabatkhah, M.; Montazerian, M. Advances in Zinc-Containing Bioactive Glasses: A Comprehensive Review. J. Funct. Biomater. 2024, 15, 258. [Google Scholar] [CrossRef]
- Tulyaganov, D.U.; Agathopoulos, S.; Dimitriadis, K.; Fernandes, H.R.; Gabrieli, R.; Baino, F. The Story, Properties and Applications of Bioactive Glass “1d”: From Concept to Early Clinical Trials. Inorganics 2024, 12, 224. [Google Scholar] [CrossRef]
- Kaur, G.; Kumar, V.; Baino, F.; Mauro, J.C.; Pickrell, G.; Evans, I.; Bretcanu, O. Mechanical Properties of Bioactive Glasses, Ceramics, Glass-Ceramics and Composites: State-of-the-Art Review and Future Challenges. Mater. Sci. Eng. C 2019, 104, 109895. [Google Scholar] [CrossRef] [PubMed]
- Abushahba, F.; Söderling, E.; Aalto-Setälä, L.; Hupa, L.; Närhi, T.O. Air Abrasion with Bioactive Glass Eradicates Streptococcus Mutans Biofilm from a Sandblasted and Acid-Etched Titanium Surface. J. Oral Implantol. 2019, 45, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Cuenca, M.; Sánchez, M.C.; Diz, P.; Martínez-Lamas, L.; Álvarez, M.; Limeres, J.; Sanz, M.; Herrera, D. In Vitro Anti-Biofilm and Antibacterial Properties of Streptococcus Downii Sp. Nov. Microorganisms 2021, 9, 450. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-C.; Lin, S.-H.; Chien, C.-S.; Kung, J.-C.; Shih, C.-J. In Vitro Bioactivity and Antibacterial Effects of a Silver-Containing Mesoporous Bioactive Glass Film on the Surface of Titanium Implants. IJMS 2022, 23, 9291. [Google Scholar] [CrossRef]
- Atif, M.; Dilawaiz; Akhtar, H.; Imran, M.; Ullah, M.Z.; Andaleeb, H.; Hussain, M.A. In Vitro Hydroxyapatite Nucleation in Cationically Cured Epoxy Composites with Pulverized Date Seed. Polymers 2024, 16, 3463. [Google Scholar] [CrossRef]
- Butler, J.; Handy, R.D.; Upton, M.; Besinis, A. Review of Antimicrobial Nanocoatings in Medicine and Dentistry: Mechanisms of Action, Biocompatibility Performance, Safety, and Benefits Compared to Antibiotics. ACS Nano 2023, 17, 7064–7092. [Google Scholar] [CrossRef]
- Kii, S.; Miyamoto, H.; Ueno, M.; Noda, I.; Hashimoto, A.; Nakashima, T.; Shobuike, T.; Kawano, S.; Sonohata, M.; Mawatari, M. Long-Term Antibacterial Activity of Silver-Containing Hydroxyapatite Coatings against Staphylococcus Aureus in Vitro and in Vivo. J. Orthop. Sci. 2024, 29, 1503–1512. [Google Scholar] [CrossRef]
- Chen, X.; Liu, Y.; Zhao, Y.; Ouyang, Z.; Zhou, H.; Li, L.; Li, L.; Li, F.; Xie, X.; Hill, R.G.; et al. Halide-Containing Bioactive Glasses Enhance Osteogenesis in Vitro and in Vivo. Biomater. Adv. 2022, 143, 213173. [Google Scholar] [CrossRef]
- Rajendran, A.K.; Anthraper, M.S.J.; Hwang, N.S.; Rangasamy, J. Osteogenesis and Angiogenesis Promoting Bioactive Ceramics. Mater. Sci. Eng. R Rep. 2024, 159, 100801. [Google Scholar] [CrossRef]
- Abbasi, M.; Hashemi, B. Fabrication and Characterization of Bioactive Glass-Ceramic Using Soda–Lime–Silica Waste Glass. Mater. Sci. Eng. C 2014, 37, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Burghardt, I.; Lüthen, F.; Prinz, C.; Kreikemeyer, B.; Zietz, C.; Neumann, H.-G.; Rychly, J. A Dual Function of Copper in Designing Regenerative Implants. Biomaterials 2015, 44, 36–44. [Google Scholar] [CrossRef] [PubMed]
- You, J.; Zhang, Y.; Zhou, Y. Strontium Functionalized in Biomaterials for Bone Tissue Engineering: A Prominent Role in Osteoimmunomodulation. Front. Bioeng. Biotechnol. 2022, 10, 928799. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Lin, Z.; Qiao, W.; Chen, B.; Shen, J. Cross-Talk between Biometal Ions and Immune Cells for Bone Repair. Eng. Regen. 2024, 5, 375–408. [Google Scholar] [CrossRef]
- Shearer, A.; Montazerian, M.; Deng, B.; Sly, J.J.; Mauro, J.C. Zirconia-containing Glass-ceramics: From Nucleating Agent to Primary Crystalline Phase. Int. J. Ceram. Eng. Sci. 2024, 6, e10200. [Google Scholar] [CrossRef]
- Yang, Y.; Lu, C.; Yang, M.; Wang, D.; Peng, S.; Tian, Z.; Shuai, C. Copper-Doped Mesoporous Bioactive Glass Endows Magnesium-Based Scaffold with Antibacterial Activity and Corrosion Resistance. Mater. Chem. Front. 2021, 5, 7228–7240. [Google Scholar] [CrossRef]
- Li, Z.; Sun, J.; Lan, J.; Qi, Q. Effect of a Denture Base Acrylic Resin Containing Silver Nanoparticles on Candida Albicans Adhesion and Biofilm Formation. Gerodontology 2016, 33, 209–216. [Google Scholar] [CrossRef]
- Almeida, N.L.M.; Peralta, L.C.F.; Pontes, F.M.L.; Rinaldo, D.; Porto, V.C.; Lara, V.S. Anti-Candida Activity and Biocompatibility of Silver Nanoparticles Associated with Denture Glaze: A New Approach to the Management of Denture Stomatitis. Folia Microbiol. 2024, 69, 1229–1246. [Google Scholar] [CrossRef]
- De Matteis, V.; Cascione, M.; Toma, C.C.; Albanese, G.; De Giorgi, M.L.; Corsalini, M.; Rinaldi, R. Silver Nanoparticles Addition in Poly(Methyl Methacrylate) Dental Matrix: Topographic and Antimycotic Studies. Int. J. Mol. Sci. 2019, 20, 4691. [Google Scholar] [CrossRef]
- Bendjama, A.; Bakar, M.F.A.; Sabran, S.F.; Iya, S.G.D. Preparation and Characterization of PMMA-AgNPs Polymer Composite as a Dental Prosthesis. Asian J. Chem. 2020, 32, 1451–1455. [Google Scholar] [CrossRef]
- Galant, K.; Turosz, N.; Chęcińska, K.; Chęciński, M.; Cholewa-Kowalska, K.; Karwan, S.; Chlubek, D.; Sikora, M. Silver Nanoparticles (AgNPs) Incorporation into Polymethyl Methacrylate (PMMA) for Dental Appliance Fabrication: A Systematic Review and Meta-Analysis of Mechanical Properties. Int. J. Mol. Sci. 2024, 25, 12645. [Google Scholar] [CrossRef]
- Takamiya, A.S.; Monteiro, D.R.; Gorup, L.F.; Silva, E.A.; De Camargo, E.R.; Gomes-Filho, J.E.; De Oliveira, S.H.P.; Barbosa, D.B. Biocompatible Silver Nanoparticles Incorporated in Acrylic Resin for Dental Application Inhibit Candida Albicans Biofilm. Mater. Sci. Eng. C 2021, 118, 111341. [Google Scholar] [CrossRef]
- More, P.R.; Pandit, S.; Filippis, A.D.; Franci, G.; Mijakovic, I.; Galdiero, M. Silver Nanoparticles: Bactericidal and Mechanistic Approach against Drug Resistant Pathogens. Microorganisms 2023, 11, 369. [Google Scholar] [CrossRef] [PubMed]
- Fahim, M.; Shahzaib, A.; Nishat, N.; Jahan, A.; Bhat, T.A.; Inam, A. Green Synthesis of Silver Nanoparticles: A Comprehensive Review of Methods, Influencing Factors, and Applications. JCIS Open 2024, 16, 100125. [Google Scholar] [CrossRef]
- Asadi, A.; Rezaee, M.; Ghahramani, Y. Sliver Nanoparticles: A Promising Strategy in Preventive Dentistry. J. Oral Dent. Health Nexus 2025, 2, 24–37. [Google Scholar] [CrossRef]
- Chandra, D.K.; Kumar, A.; Mahapatra, C. Smart Nano-Hybrid Metal-Organic Frameworks: Revolutionizing Advancements, Applications, and Challenges in Biomedical Therapeutics and Diagnostics. Hybrid Adv. 2025, 9, 100406. [Google Scholar] [CrossRef]
- Yu, C.; Yu, Y.; Lu, Y.; Quan, K.; Mao, Z.; Zheng, Y.; Qin, L.; Xia, D. UiO-66/AgNPs Coating for Dental Implants in Preventing Bacterial Infections. J. Dent. Res. 2024, 103, 516–525. [Google Scholar] [CrossRef]
- De Castro, D.T.; Ferreira, I.; Oliveira, V.C.; Schiavon, M.A.; Dos Reis, A.C. Denture Adhesives Associated with Silver Vanadate: Antimicrobial Approach Against Multi- Species Biofilms on Acrylic Resin Surfaces. Eur. J. Prosthodont. Restor. Dent. 2024, 32, 203. [Google Scholar] [CrossRef]
- Gunputh, U.F.; Le, H.; Lawton, K.; Besinis, A.; Tredwin, C.; Handy, R.D. Antibacterial Properties of Silver Nanoparticles Grown in Situ and Anchored to Titanium Dioxide Nanotubes on Titanium Implant against Staphylococcus aureus. Nanotoxicology 2020, 14, 97–110. [Google Scholar] [CrossRef]
- Divakar, D.D.; Jastaniyah, N.T.; Altamimi, H.G.; Alnakhli, Y.O.; Muzaheed; Alkheraif, A.A.; Haleem, S. Enhanced Antimicrobial Activity of Naturally Derived Bioactive Molecule Chitosan Conjugated Silver Nanoparticle against Dental Implant Pathogens. Int. J. Biol. Macromol. 2018, 108, 790–797. [Google Scholar] [CrossRef]
- Langguth, K.J.; Maccagnano-Zachera, S.; Heinemann, J. Microfluidic Production of Silver Nanoparticles Demonstrates Ability for on Demand Synthesis of a Wide Size Distribution of Particles. J. Nanoparticle Res. 2024, 26, 40. [Google Scholar] [CrossRef]
- Abdelwahab, M.A.; Nabil, A.; El-Hosainy, H.; Tahway, R.; Taha, M.S. Green Synthesis of Silver Nanoparticles Using Curcumin: A Comparative Study of Antimicrobial and Antibiofilm Effects on Acinetobacter Baumannii against Chemical Conventional Methods. Results Chem. 2024, 7, 101274. [Google Scholar] [CrossRef]
- Chen, R.; Han, Z.; Huang, Z.; Karki, J.; Wang, C.; Zhu, B.; Zhang, X. Antibacterial Activity, Cytotoxicity and Mechanical Behavior of Nano-Enhanced Denture Base Resin with Different Kinds of Inorganic Antibacterial Agents. Dent. Mater. J. 2017, 36, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, A.B.V.; Carvalho-Silva, J.M.; Ferreira, I.; Schiavon, M.A.; Cândido Dos Reis, A. Silver Vanadate Nanomaterial Incorporated into Heat-Cured Resin and Coating in Printed Resin—Antimicrobial Activity in Two Multi-Species Biofilms and Wettability. J. Dent. 2024, 145, 104984. [Google Scholar] [CrossRef] [PubMed]
- Lampé, I.; Beke, D.; Biri, S.; Csarnovics, I.; Csik, A.; Dombrádi, Z.; Hajdu, P.; Hegedűs, V.; Rácz, R.; Varga, I.; et al. Investigation of Silver Nanoparticles on Titanium Surface Created by Ion Implantation Technology. Int. J. Nanomed. 2019, 14, 4709–4721. [Google Scholar] [CrossRef]
- Curylofo, P.C.; Raile, P.N.; Oliveira, V.; Macedo, A.P.; Guedes, D.F.C.; Paranhos, H.O.; Pagnano, V.O. Antimicrobial Activity of Experimental Chitosan Solutions on Acrylic Resin and Cobalt-Chromium Surfaces. Int. J. Prosthodont. 2023, 36. [Google Scholar] [CrossRef]
- Araujo, H.C.; Ramírez Carmona, W.; Sato, C.; Dos Santos Oliveira, M.; Alves, G.D.S.G.; Morato, D.N.; Pessan, J.P.; Monteiro, D.R. In Vitro Antimicrobial Effects of Chitosan on Microcosm Biofilms of Oral Candidiasis. J. Dent. 2022, 125, 104246. [Google Scholar] [CrossRef]
- Mascarenhas, R.; Hegde, S.; Manaktala, N. Chitosan Nanoparticle Applications in Dentistry: A Sustainable Biopolymer. Front. Chem. 2024, 12, 1362482. [Google Scholar] [CrossRef]
- Ibrahim, M.A.; Neo, J.; Esguerra, R.J.; Fawzy, A.S. Characterization of Antibacterial and Adhesion Properties of Chitosan-Modified Glass Ionomer Cement. J. Biomater. Appl. 2015, 30, 409–419. [Google Scholar] [CrossRef]
- Altınışık, H.; Erten Can, H.; Mutlu Ağardan, N.B.; Berkkan, A.; Güney, M. Prevention of Secondary Caries Using Fluoride-Loaded Chitosan Nanoparticle-Modified Glass-Ionomer Cement. Clin. Oral Investig. 2024, 28, 504. [Google Scholar] [CrossRef]
- Bonifacio, M.A.; Cometa, S.; Dicarlo, M.; Baruzzi, F.; De Candia, S.; Gloria, A.; Giangregorio, M.M.; Mattioli-Belmonte, M.; De Giglio, E. Gallium-Modified Chitosan/Poly(Acrylic Acid) Bilayer Coatings for Improved Titanium Implant Performances. Carbohydr. Polym. 2017, 166, 348–357. [Google Scholar] [CrossRef]
- França, R.C.S.; Dias, R.T.A.; Reis, R.M.; Sousa, F.B.D.; Carlo, H.L.; Santos, R.L.D.; Carvalho, F.G.D. Chitosan Nanoparticles Suspension Can Minimize Enamel Loss after in Vitro Erosive Challenge. J. Appl. Oral Sci. 2025, 33, e20240445. [Google Scholar] [CrossRef]
- Tao, R.; Wang, Y.; Zhang, N.; Zhang, L.; Khan, M.S.; Xu, H.; Zhao, J.; Qi, Z.; Chen, Y.; Lu, Y.; et al. Bioactive Chitosan-Citral Schiff Base Zinc Complex: A PH-Responsive Platform for Potential Therapeutic Applications. Int. J. Biol. Macromol. 2024, 261, 129857. [Google Scholar] [CrossRef]
- Zeza, B.; Wohlfahrt, C.; Pilloni, A. Chitosan Brush for Professional Removal of Plaque in Mild Peri-Implantitis. Minerva Dent. Oral Sci. 2017, 66. [Google Scholar] [CrossRef]
- Wu, T.; Zhou, Q.; Hong, G.; Bai, Z.; Bian, J.; Xie, H.; Chen, C. A Chlorogenic Acid-Chitosan Complex Bifunctional Coating for Improving Osteogenesis Differentiation and Bactericidal Properties of Zirconia Implants. Colloids Surf. B Biointerfaces 2023, 230, 113484. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Xiao, Y. Sequential Drug Delivery of Vancomycin and RhBMP-2 via Pore-Closed PLGA Microparticles Embedded Photo-Crosslinked Chitosan Hydrogel for Enhanced Osteointegration. Int. J. Biol. Macromol. 2021, 182, 612–625. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Xia, Y.; Gong, W.; Zhou, J.; Yu, D.-G.; Xie, Y. Electrospun Chitosan//Ethylcellulose-Vitamin E//Ethylcellulose-Curcumin Tri-Chamber Eccentric Janus Nanofibers for a Joint Antibacterial and Antioxidant Performance. Int. J. Biol. Macromol. 2024, 281, 135753. [Google Scholar] [CrossRef]
- Gondim, B.L.C.; Castellano, L.R.C.; De Castro, R.D.; Machado, G.; Carlo, H.L.; Valença, A.M.G.; De Carvalho, F.G. Effect of Chitosan Nanoparticles on the Inhibition of Candida Spp. Biofilm on Denture Base Surface. Arch. Oral Biol. 2018, 94, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Srimaneepong, V.; Thanamee, T.; Wattanasirmkit, K.; Muangsawat, S.; Matangkasombut, O. Efficacy of Low-molecular Weight Chitosan against Candida Albicans Biofilm on Polymethyl Methacrylate Resin. Aust. Dent. J. 2021, 66, 262–269. [Google Scholar] [CrossRef]
- Namangkalakul, W.; Benjavongkulchai, S.; Pochana, T.; Promchai, A.; Satitviboon, W.; Howattanapanich, S.; Phuprasong, R.; Ungvijanpunya, N.; Supakanjanakanti, D.; Chaitrakoonthong, T.; et al. Activity of Chitosan Antifungal Denture Adhesive against Common Candida Species and Candida Albicans Adherence on Denture Base Acrylic Resin. J. Prosthet. Dent. 2020, 123, 181.e1–181.e7. [Google Scholar] [CrossRef]
- Jung, J.; Li, L.; Yeh, C.-K.; Ren, X.; Sun, Y. Amphiphilic Quaternary Ammonium Chitosan/Sodium Alginate Multilayer Coatings Kill Fungal Cells and Inhibit Fungal Biofilm on Dental Biomaterials. Mater. Sci. Eng. C 2019, 104, 109961. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Lai, Y.; Guo, Y.; Cai, Z.; Mao, C.; Lu, M.; Ren, C.; Ong, J.L.; Chen, W. Aspirin/Amoxicillin Loaded Chitosan Microparticles and Polydopamine Modified Titanium Implants to Combat Infections and Promote Osteogenesis. Sci. Rep. 2024, 14, 7624. [Google Scholar] [CrossRef] [PubMed]
- Nithya, S.; Nimal, T.R.; Baranwal, G.; Suresh, M.K.; CP, A.; Anil Kumar, V.; Gopi Mohan, C.; Jayakumar, R.; Biswas, R. Preparation, Characterization and Efficacy of Lysostaphin-Chitosan Gel against Staphylococcus Aureus. Int. J. Biol. Macromol. 2018, 110, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Waßmann, M.; Winkel, A.; Haak, K.; Dempwolf, W.; Stiesch, M.; Menzel, H. Influence of Quaternization of Ammonium on Antibacterial Activity and Cytocompatibility of Thin Copolymer Layers on Titanium. J. Biomater. Sci. Polym. Ed. 2016, 27, 1507–1519. [Google Scholar] [CrossRef]
- Sun, Y.; Zhao, Y.-Q.; Zeng, Q.; Wu, Y.-W.; Hu, Y.; Duan, S.; Tang, Z.; Xu, F.-J. Dual-Functional Implants with Antibacterial and Osteointegration-Promoting Performances. ACS Appl. Mater. Interfaces 2019, 11, 36449–36457. [Google Scholar] [CrossRef]
- Radhi, A.; Mohamad, D.; Abdul Rahman, F.S.; Abdullah, A.M.; Hasan, H. Mechanism and Factors Influence of Graphene-Based Nanomaterials Antimicrobial Activities and Application in Dentistry. J. Mater. Res. Technol. 2021, 11, 1290–1307. [Google Scholar] [CrossRef]
- Wang, X.; Su, K.; Tan, L.; Liu, X.; Cui, Z.; Jing, D.; Yang, X.; Liang, Y.; Li, Z.; Zhu, S.; et al. Rapid and Highly Effective Noninvasive Disinfection by Hybrid Ag/CS@MnO2 Nanosheets Using Near-Infrared Light. ACS Appl. Mater. Interfaces 2019, 11, 15014–15027. [Google Scholar] [CrossRef]
- Alshammari, H.; Neilands, J.; Jeppesen, C.S.; Almtoft, K.P.; Andersen, O.Z.; Stavropoulos, A. Antimicrobial Potential of Strontium-Functionalized Titanium Against Bacteria Associated with Peri-Implantitis. Clin. Exp. Dent. Res. 2024, 10, e903. [Google Scholar] [CrossRef]
- Chen, J.; Xing, X.; Liu, D.; Gao, L.; Liu, Y.; Wang, Y.; Cheng, H. Copper Nanoparticles Incorporated Visible Light-Curing Chitosan-Based Hydrogel Membrane for Enhancement of Bone Repair. J. Mech. Behav. Biomed. Mater. 2024, 158, 106674. [Google Scholar] [CrossRef]
- Yu, Y.; Guo, X.; Chen, J.; Zhao, Y.; Song, J.; Alshawwa, H.; Zou, X.; Zhao, H.; Zhang, Z. Biodegradation of Urethane Dimethacrylate-Based Materials (CAD/CAM Resin-Ceramic Composites) and Its Effect on the Adhesion and Proliferation of Streptococcus Mutans. J. Mech. Behav. Biomed. Mater. 2024, 150, 106280. [Google Scholar] [CrossRef]
- Ma, X.; Zhou, S.; Xu, X.; Du, Q. Copper-Containing Nanoparticles: Mechanism of Antimicrobial Effect and Application in Dentistry-a Narrative Review. Front. Surg. 2022, 9, 905892. [Google Scholar] [CrossRef]
- Yin, I.X.; Udduttulla, A.; Xu, V.W.; Chen, K.J.; Zhang, M.Y.; Chu, C.H. Use of Antimicrobial Nanoparticles for the Management of Dental Diseases. Nanomaterials 2025, 15, 209. [Google Scholar] [CrossRef]
- Offermanns, V.; Andersen, O.Z.; Riede, G.; Sillassen, M.; Jeppesen, C.S.; Almtoft, K.P.; Talasz, H.; Öhman-Mägi, C.; Lethaus, B.; Tolba, R.; et al. Effect of Strontium Surface-Functionalized Implants on Early and Late Osseointegration: A Histological, Spectrometric and Tomographic Evaluation. Acta Biomater. 2018, 69, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Alshammari, H.; Bakitian, F.; Neilands, J.; Andersen, O.Z.; Stavropoulos, A. Antimicrobial Properties of Strontium Functionalized Titanium Surfaces for Oral Applications, A Systematic Review. Coatings 2021, 11, 810. [Google Scholar] [CrossRef]
- Perić, M.; Miličić, B.; Kuzmanović Pfićer, J.; Živković, R.; Arsić Arsenijević, V. A Systematic Review of Denture Stomatitis: Predisposing Factors, Clinical Features, Etiology, and Global Candida Spp. Distribution. J. Fungi 2024, 10, 328. [Google Scholar] [CrossRef] [PubMed]
- McReynolds, D.E.; Moorthy, A.; Moneley, J.O.; Jabra-Rizk, M.A.; Sultan, A.S. Denture Stomatitis—An Interdisciplinary Clinical Review. J. Prosthodont. 2023, 32, 560–570. [Google Scholar] [CrossRef]
- Wady, A.F.; Machado, A.L.; Zucolotto, V.; Zamperini, C.A.; Berni, E.; Vergani, C.E. Evaluation of Candida Albicans Adhesion and Biofilm Formation on a Denture Base Acrylic Resin Containing Silver Nanoparticles: Antifungal Activity of Silver Nanoparticles. J. Appl. Microbiol. 2012, 112, 1163–1172. [Google Scholar] [CrossRef]
- Alvim, G.C.; Oliveira, V.D.C.; Dos Reis, A.C.; Schiavon, M.A.; Pinto, M.R.; Da Silva, M.V.; Lepri, C.P.; De Castro, D.T. Effect of Silver Vanadate on the Antibiofilm, Adhesion and Biocompatibility Properties of Denture Adhesive. Future Microbiol. 2024, 19, 655–665. [Google Scholar] [CrossRef]
- Abar, E.S.; Vandghanooni, S.; Memar, M.Y.; Eskandani, M.; Torab, A. Enhancing Antifungal and Antibacterial Properties of Denture Resins with Nystatin-Coated Silver Nanoparticles. Sci. Rep. 2024, 14, 23770. [Google Scholar] [CrossRef]
- Dodou Lima, H.V.; De Paula Cavalcante, C.S.; Rádis-Baptista, G. Antifungal In Vitro Activity of Pilosulin- and Ponericin-Like Peptides from the Giant Ant Dinoponera Quadriceps and Synergistic Effects with Antimycotic Drugs. Antibiotics 2020, 9, 354. [Google Scholar] [CrossRef]
- Costa, E.; Silva, S.; Tavaria, F.; Pintado, M. Antimicrobial and Antibiofilm Activity of Chitosan on the Oral Pathogen Candida Albicans. Pathogens 2014, 3, 908–919. [Google Scholar] [CrossRef]
- Ashrafi, B.; Rashidipour, M.; Marzban, A.; Soroush, S.; Azadpour, M.; Delfani, S.; Ramak, P. Mentha Piperita Essential Oils Loaded in a Chitosan Nanogel with Inhibitory Effect on Biofilm Formation against S. Mutans on the Dental Surface. Carbohydr. Polym. 2019, 212, 142–149. [Google Scholar] [CrossRef]
- Hemmingsen, L.M.; Škalko-Basnet, N.; Jøraholmen, M.W. The Expanded Role of Chitosan in Localized Antimicrobial Therapy. Mar. Drugs 2021, 19, 697. [Google Scholar] [CrossRef]
- Ashraf, H.; Gul, H.; Jamil, B.; Saeed, A.; Pasha, M.; Kaleem, M.; Khan, A.S. Synthesis, Characterization, and Evaluation of the Antifungal Properties of Tissue Conditioner Incorporated with Essential Oils-Loaded Chitosan Nanoparticles. PLoS ONE 2022, 17, e0273079. [Google Scholar] [CrossRef]
- Saeed, A.; Haider, A.; Zahid, S.; Khan, S.A.; Faryal, R.; Kaleem, M. In-Vitro Antifungal Efficacy of Tissue Conditioner-Chitosan Composites as Potential Treatment Therapy for Denture Stomatitis. Int. J. Biol. Macromol. 2019, 125, 761–766. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, S.A.; Ghotaslou, R.; Kordi, S.; Khoramdel, A.; Aeenfar, A.; Kahjough, S.T.; Akbarzadeh, A. Antibacterial and Antifungal Effects of Chitosan Nanoparticles on Tissue Conditioners of Complete Dentures. Int. J. Biol. Macromol. 2018, 118, 881–885. [Google Scholar] [CrossRef] [PubMed]
- AlQurashi, D.M.; AlQurashi, T.F.; Alam, R.I.; Shaikh, S.; Tarkistani, M.A.M. Advanced Nanoparticles in Combating Antibiotic Resistance: Current Innovations and Future Directions. J. Nanotheranostics 2025, 6, 9. [Google Scholar] [CrossRef]
- Munir, A.; Marovic, D.; Nogueira, L.P.; Simm, R.; Naemi, A.-O.; Landrø, S.M.; Helgerud, M.; Zheng, K.; Par, M.; Tauböck, T.T.; et al. Using Copper-Doped Mesoporous Bioactive Glass Nanospheres to Impart Anti-Bacterial Properties to Dental Composites. Pharmaceutics 2022, 14, 2241. [Google Scholar] [CrossRef]
- Silva, M.P.D.; Silva, V.D.O.; Pasetto, S.; Ando-Suguimoto, E.S.; Kawamoto, D.; Mata, G.M.S.C.; Murata, R.M.; Mayer, M.P.A.; Chen, C. Aggregatibacter Actinomycetemcomitans Outer Membrane Proteins 29 and 29 Paralogue Induce Evasion of Immune Response. Front. Oral. Health 2022, 3, 835902. [Google Scholar] [CrossRef]
- Kung, J.-C.; Yang, T.-Y.; Hung, C.-C.; Shih, C.-J. Silica-Based Silver Nanocomposite 80S/Ag as Aggregatibacter Actinomycetemcomitans Inhibitor and Its in Vitro Bioactivity. J. Dent. Sci. 2024, 19, 568–579. [Google Scholar] [CrossRef]
- Vasluianu, R.-I.; Dima, A.M.; Bobu, L.; Murariu, A.; Stamatin, O.; Baciu, E.-R.; Luca, E.-O. Dentistry Insights: Single-Walled and Multi-Walled Carbon Nanotubes, Carbon Dots, and the Rise of Hybrid Materials. J. Funct. Biomater. 2025, 16, 110. [Google Scholar] [CrossRef]
- Turu, I.C.; Bayraktar, S.; Akgul, B.; Ilhan-Sungur, E.; Abamor, E.S.; Cansever, N. Formation of TiO2 Nanotubes and Deposition of Silver Nanoparticle and Reduced Graphene Oxide: Antibacterial and Biocompatibility Behavior. Surf. Coat. Technol. 2023, 470, 129866. [Google Scholar] [CrossRef]
- Besinis, A.; Hadi, S.D.; Le, H.R.; Tredwin, C.; Handy, R.D. Antibacterial Activity and Biofilm Inhibition by Surface Modified Titanium Alloy Medical Implants Following Application of Silver, Titanium Dioxide and Hydroxyapatite Nanocoatings. Nanotoxicology 2017, 11, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Ke, D.; Vu, A.A.; Bandyopadhyay, A.; Bose, S. Compositionally Graded Doped Hydroxyapatite Coating on Titanium Using Laser and Plasma Spray Deposition for Bone Implants. Acta Biomater. 2019, 84, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Kheirmand-Parizi, M.; Doll-Nikutta, K.; Gaikwad, A.; Denis, H.; Stiesch, M. Effectiveness of Strontium/Silver-Based Titanium Surface Coatings in Improving Antibacterial and Osteogenic Implant Characteristics: A Systematic Review of in-Vitro Studies. Front. Bioeng. Biotechnol. 2024, 12, 1346426. [Google Scholar] [CrossRef]
- Han, J.; Sanders, J.G.F.; Andrée, L.; Van Oirschot, B.A.J.A.; Plachokova, A.S.; Van Den Beucken, J.J.J.P.; Leeuwenburgh, S.C.G.; Yang, F. Development of Zinc-Containing Chitosan/Gelatin Coatings with Immunomodulatory Effect for Soft Tissue Sealing around Dental Implants. Tissue Eng. Regen. Med. 2025, 22, 57–75. [Google Scholar] [CrossRef]
- Kadirvelu, L.; Sivaramalingam, S.S.; Jothivel, D.; Chithiraiselvan, D.D.; Karaiyagowder Govindarajan, D.; Kandaswamy, K. A Review on Antimicrobial Strategies in Mitigating Biofilm-Associated Infections on Medical Implants. Curr. Res. Microb. Sci. 2024, 6, 100231. [Google Scholar] [CrossRef]
- Hosseini Hooshiar, M.; Mozaffari, A.; Hamed Ahmed, M.; Abdul Kareem, R.; Jaber Zrzo, A.; Salah Mansoor, A.H.; Athab, Z.; Parhizgar, Z.; Amini, P. Potential Role of Metal Nanoparticles in Treatment of Peri-Implant Mucositis and Peri-Implantitis. BioMed. Eng. OnLine 2024, 23, 101. [Google Scholar] [CrossRef]
- Liu, X.; Liu, L.; Zeng, Z. Effectiveness of a Local Drug Delivery System Based on Antimicrobial Peptides in Early Treatment of Peri-Implantitis. Int. Dent. J. 2025, 75, 1400–1408. [Google Scholar] [CrossRef]
- Sun, Z.; Ma, L.; Sun, X.; Sloan, A.J.; O’Brien-Simpson, N.M.; Li, W. The Overview of Antimicrobial Peptide-coated Implants against Oral Bacterial Infections. Aggregate 2023, 4, e309. [Google Scholar] [CrossRef]
- Yao, Y.; Ye, Z.; Zhang, Y.; Wang, Y.; Yu, C. Quaternary Ammonium Compounds and Their Composites in Antimicrobial Applications. Adv. Mater. Interfaces 2024, 11, 2300946. [Google Scholar] [CrossRef]
- Khan, S.N.; Koldsland, O.C.; Roos-Jansåker, A.; Wohlfahrt, J.C.; Verket, A.; Mdala, I.; Magnusson, A.; Salvesen, E.; Hjortsjö, C. Non-surgical Treatment of Mild to Moderate Peri-implantitis Using an Oscillating Chitosan Brush or a Titanium Curette—A Randomized Multicentre Controlled Clinical Trial. Clin. Oral Implant. Res. 2022, 33, 1254–1264. [Google Scholar] [CrossRef] [PubMed]
- Shiba, T.; Komatsu, K.; Takeuchi, Y.; Koyanagi, T.; Taniguchi, Y.; Takagi, T.; Maekawa, S.; Nagai, T.; Kobayashi, R.; Matsumura, S.; et al. Novel Flowchart Guiding the Non-Surgical and Surgical Management of Peri-Implant Complications: A Narrative Review. Bioengineering 2024, 11, 118. [Google Scholar] [CrossRef]
- Chai, M.; An, M.; Zhang, X. Construction of a TiO2/MoSe2/CHI Coating on Dental Implants for Combating Streptococcus Mutans Infection. Mater. Sci. Eng. C 2021, 129, 112416. [Google Scholar] [CrossRef]
- Liu, D.; Chen, J.; Gao, L.; Chen, X.; Lin, L.; Liu, Y.; Wei, X.; Pan, Y.; Wang, Y.; Cheng, H. Nano Sim@ZIF8@PDA Modified Injectable Temperature Sensitive Nanocomposite Hydrogel for Photothermal/Drug Therapy for Peri-Implantitis. Carbohydr. Polym. 2025, 354, 123327. [Google Scholar] [CrossRef]
- Elshenawy, E.A.; El-Ebiary, M.A.; Kenawy, E.-R.; El-Olimy, G.A. Modification of Glass-Ionomer Cement Properties by Quaternized Chitosan-Coated Nanoparticles. Odontology 2023, 111, 328–341. [Google Scholar] [CrossRef]
- Fang, L.; Zhou, H.; Cheng, L.; Wang, Y.; Liu, F.; Wang, S. The Application of Mesoporous Silica Nanoparticles as a Drug Delivery Vehicle in Oral Disease Treatment. Front. Cell. Infect. Microbiol. 2023, 13, 1124411. [Google Scholar] [CrossRef]
- Lu, M.-M.; Ge, Y.; Qiu, J.; Shao, D.; Zhang, Y.; Bai, J.; Zheng, X.; Chang, Z.-M.; Wang, Z.; Dong, W.-F.; et al. Redox/PH Dual-Controlled Release of Chlorhexidine and Silver Ions from Biodegradable Mesoporous Silica Nanoparticles against Oral Biofilms. Int. J. Nanomed. 2018, 13, 7697–7709. [Google Scholar] [CrossRef]
- Sasany, R.; Jamjoom, F.Z.; Yilmaz, B. Mechanical and Optical Properties of Additively Manufactured Denture Base Resin in Different Colors Modified with Antimicrobial Substances: An in Vitro Study. J. Prosthet. Dent. 2025, 133, 890.e1–890.e8. [Google Scholar] [CrossRef]
- Kong, S.; Li, J.; Fan, O.; Lin, F.; Xie, J.; Lin, J. Controllable Fabrication of ZnO Nanorod Arrays on the Surface of Titanium Material and Their Antibacterial and Anti-Adhesion Properties. Materials 2025, 18, 1645. [Google Scholar] [CrossRef]
- Krishnaswami, V.; Muruganantham, S.; Raja, J.; Janakiraman, K.; Sethuraman, V.; Kanakaraj, L. Biofunctional Applications of Chitosan in Dentistry—An Overview. Biomater. Connect. 2024, 1, 1. [Google Scholar] [CrossRef]
- Ben-Arfa, B.A.E.; Palamá, I.E.; Miranda Salvado, I.M.; Ferreira, J.M.F.; Pullar, R.C. Cytotoxicity and Bioactivity Assessments for Cu2+ and La3+ Doped High-silica Sol-gel Derived Bioglasses: The Complex Interplay between Additive Ions Revealed. J. Biomed. Mater. Res. Part A 2019, 107, 2680–2693. [Google Scholar] [CrossRef]
- Pokrowiecki, R.; Zaręba, T.; Szaraniec, B.; Pałka, K.; Mielczarek, A.; Menaszek, E.; Tyski, S. In Vitro Studies of Nanosilver-Doped Titanium Implants for Oral and Maxillofacial Surgery. Int. J. Nanomed. 2017, 12, 4285–4297. [Google Scholar] [CrossRef]
- Sun, Z.L.; Wataha, J.C.; Hanks, C.T. Effects of Metal Ions on Osteoblast-like Cell Metabolism and Differentiation. J. Biomed. Mater. Res. 1997, 34, 29–37. [Google Scholar] [CrossRef]
- Jiang, Z.L.; Fletcher, N.M.; Diamond, M.P.; Abu-Soud, H.M.; Saed, G.M. S-nitrosylation of Caspase-3 Is the Mechanism by Which Adhesion Fibroblasts Manifest Lower Apoptosis. Wound Repair Regen. 2009, 17, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Selli, N.T.; Başaran, N.; Kesmez, Ö.; Yavaş, A.; Demir, F.; Ertürk, A.T. Bioactivity and Cytotoxicity of LiF-Modified Bioactive Glass Ceramics for Bone Regeneration. Ceram. Int. 2025, 51, 26014–26024. [Google Scholar] [CrossRef]
- Tsamesidis, I.; Kazeli, K.; Lymperaki, E.; Pouroutzidou, G.K.; Oikonomou, I.M.; Komninou, P.; Zachariadis, G.; Reybier, K.; Pantaleo, A.; Kontonasaki, E. Effect of Sintering Temperature of Bioactive Glass Nanoceramics on the Hemolytic Activity and Oxidative Stress Biomarkers in Erythrocytes. Cel. Mol. Bioeng. 2020, 13, 201–218. [Google Scholar] [CrossRef]
- Peng, S.; Fu, H.; Li, R.; Li, H.; Wang, S.; Li, B.; Sun, J. A New Direction in Periodontitis Treatment: Biomaterial-Mediated Macrophage Immunotherapy. J. Nanobiotechnology 2024, 22, 359. [Google Scholar] [CrossRef]
- Husain, S.; Al-Samadani, K.H.; Najeeb, S.; Zafar, M.S.; Khurshid, Z.; Zohaib, S.; Qasim, S.B. Chitosan Biomaterials for Current and Potential Dental Applications. Materials 2017, 10, 602. [Google Scholar] [CrossRef]
- Wareham-Mathiassen, S.; Nateqi, M.; Badrinarayanan, S.A.; Glenting, V.P.; Dragheim, M.B.; Agner, A.R.; Rasmussen, T.S.; Bay, L.; Jelsbak, L.; Bengtsson, H.; et al. Evaluating Antimicrobial Efficacy in Medical Devices: The Critical Role of Simulating in Use Test Conditions. Biomater. Adv. 2025, 172, 214241. [Google Scholar] [CrossRef]
- Bento De Carvalho, T.; Barbosa, J.B.; Teixeira, P. Assessing Antimicrobial Efficacy on Plastics and Other Non-Porous Surfaces: A Closer Look at Studies Using the ISO 22196:2011 Standard. Biology 2024, 13, 59. [Google Scholar] [CrossRef]
- Tahsin, K.; Xu, W.; Watson, D.; Rizkalla, A.; Charpentier, P. Antimicrobial Denture Material Synthesized from Poly(Methyl Methacrylate) Enriched with Cannabidiol Isolates. Molecules 2025, 30, 943. [Google Scholar] [CrossRef]
- Palaskar, J.N.; Hindocha, A.D.; Mishra, A.; Gandagule, R.; Korde, S. Evaluating the Antifungal Effectiveness, Leaching Characteristics, Flexural Strength, and Impact Strength of Polymethyl Methacrylate Added with Small-Scale Silver Nanoparticles—An in Vitro Study. J. Indian Prosthodont. Soc. 2024, 24, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Ashammakhi, N.; Ahadian, S.; Zengjie, F.; Suthiwanich, K.; Lorestani, F.; Orive, G.; Ostrovidov, S.; Khademhosseini, A. Advances and Future Perspectives in 4D Bioprinting. Biotechnol. J. 2018, 13, 1800148. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yang, Y.; Yang, Z.; Ma, R.; Aimaijiang, M.; Xu, J.; Zhang, Y.; Zhou, Y. Four-Dimensional Printing and Shape Memory Materials in Bone Tissue Engineering. Int. J. Mol. Sci. 2023, 24, 814. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, J.; Arias Moliz, T.; Bettencourt, A.; Costa, J.; Martins, F.; Rabadijeva, D.; Rodriguez, D.; Visai, L.; Combes, C.; Farrugia, C.; et al. Standardization of Antimicrobial Testing of Dental Devices. Dent. Mater. 2020, 36, e59–e73. [Google Scholar] [CrossRef]
- Thieme, L.; Hartung, A.; Tramm, K.; Graf, J.; Spott, R.; Makarewicz, O.; Pletz, M.W. Adaptation of the Start-Growth-Time Method for High-Throughput Biofilm Quantification. Front. Microbiol. 2021, 12, 631248. [Google Scholar] [CrossRef]
- Hu, J.; Hu, Q.; He, X.; Liu, C.; Kong, Y.; Cheng, Y.; Zhang, Y. Stimuli-Responsive Hydrogels with Antibacterial Activity Assembled from Guanosine, Aminoglycoside, and a Bifunctional Anchor. Adv. Healthc. Mater. 2020, 9, 1901329. [Google Scholar] [CrossRef]
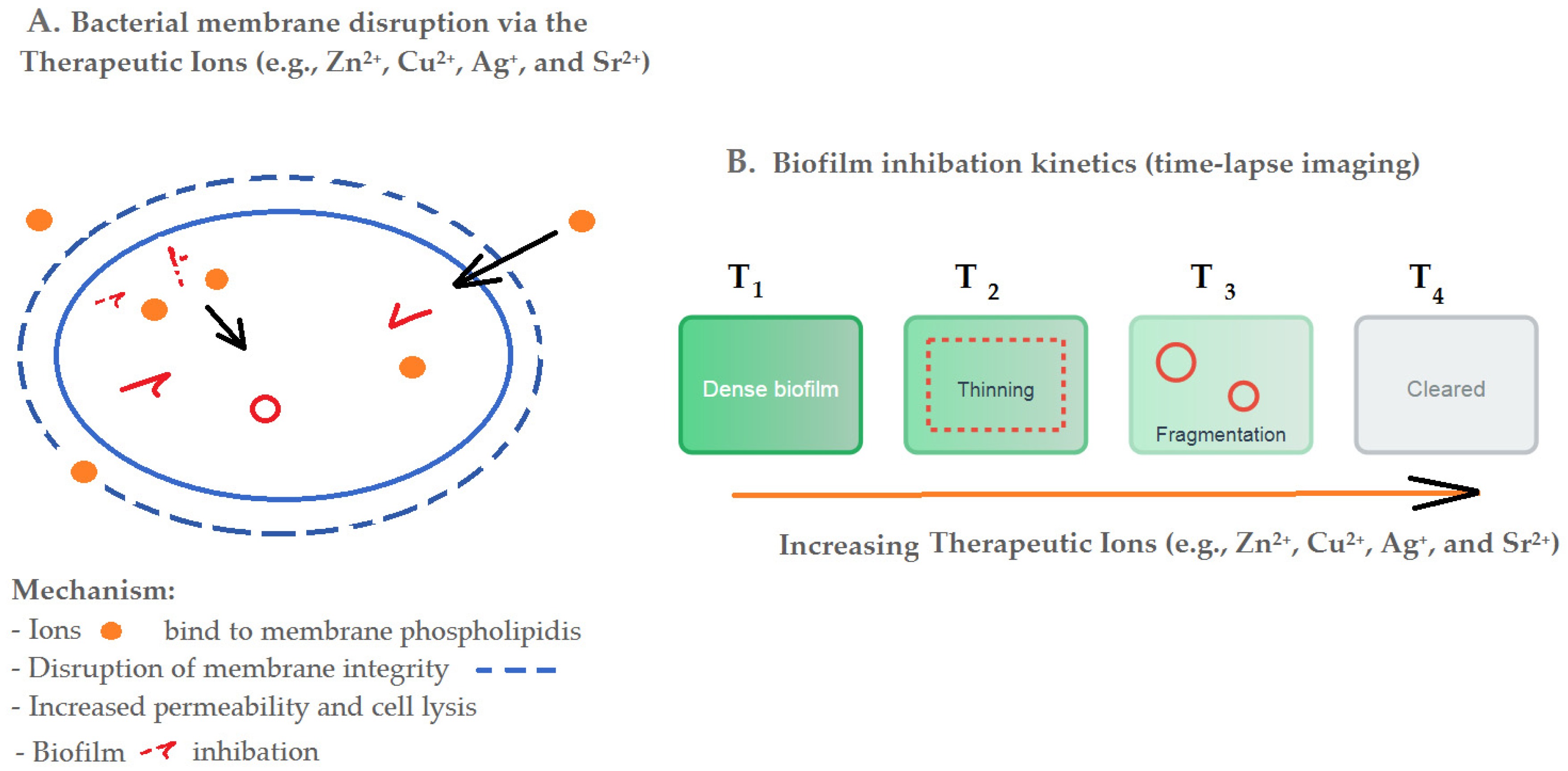
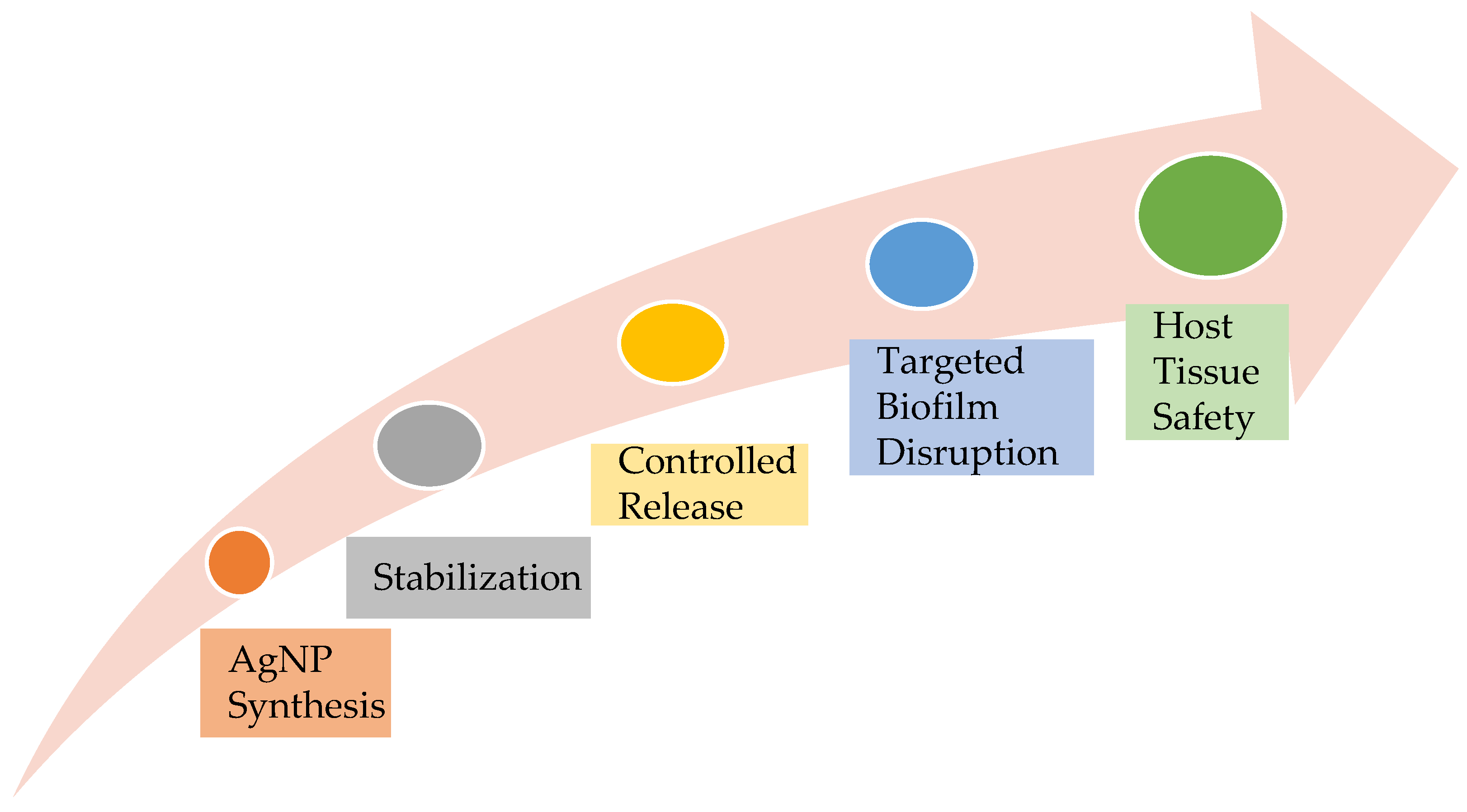
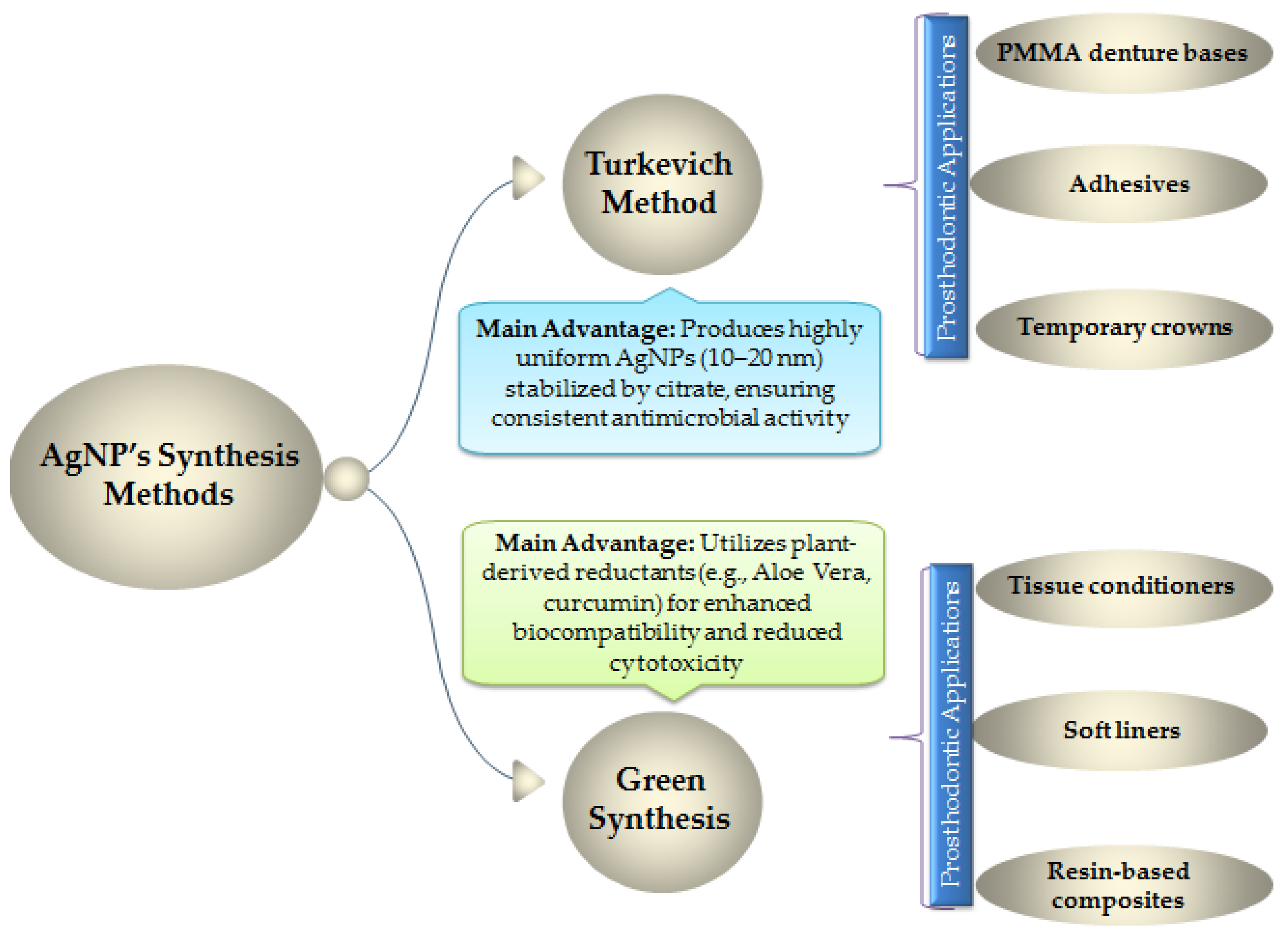
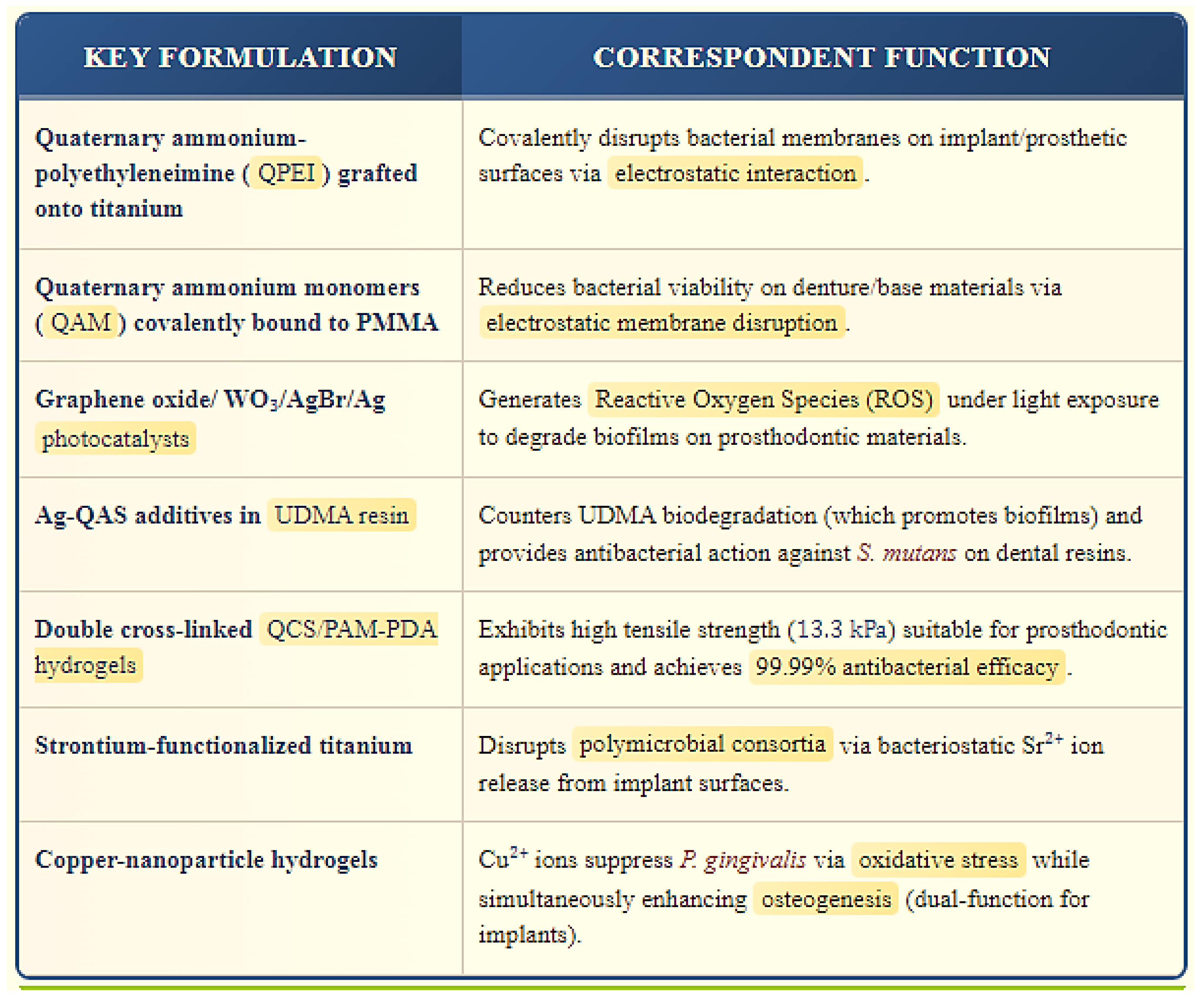


| Ion | Primary Biological Target | Mechanism of Action | Clinical Impact |
|---|---|---|---|
| Cu2+ | Angiogenesis & Osteogenesis |
| Accelerates vascularization and osseointegration of implants; improves bone-implant healing. |
| Zn2+ | Antimicrobial & Osteo-support |
| Reduces peri-implantitis risk; supports bone metabolism and mineralization. |
| Sr2+ | Osteoblast/Osteoclast Balance |
| Enhances bone density around implants; useful in osteoporotic patients. |
| Ag+ | Broad-Spectrum Antimicrobial |
| Prevents bacterial/fungal infections; reduces biofilm formation on implants. |
| Key Formulation | Correspondent Function |
|---|---|
| Chitosan-Citral Schiff Base (Zn2+ loaded) | Releases Zn2+ in acidic biofilm microenvironments; Antibacterial effects against S. aureus, E. coli, oral pathogens |
| Fluoride-loaded Chitosan Nanoparticles in GIC | Reduces secondary caries depth by 9%; Inhibits S. mutans; Enhances remineralization |
| Chitosan-based pH-responsive Coating (General) | Exhibits cationic disruption of microbial membranes; Controlled drug release under acidic biofilm conditions |
| Aspirin/Amoxicillin co-loaded Microparticles | pH-dependent erosion enables sustained dual-drug elution; Achieves 99% metabolic biofilm reduction; Enhances rBMSC adhesion |
| Zn2+-chelated Chitosan/Gelatin | Disrupts quorum sensing and bacterial adhesion (90% reduction); Accelerates human gingival fibroblast migration |
| Chitosan Nanoparticles (30.1 μg/mL) | Cationic disruption of fungal membranes; Reduces Candida biomass by 51.5% on acrylic resin |
| Low-MW Chitosan Solutions (3–6 mg/mL) | Disrupts C. albicans biofilms on PMMA resin through electrostatic membrane destabilization |
| Chitosan/Ethylcellulose Nanocomposite | Enables redox/pH-triggered drug release |
| Protonated Chitosan Amines (Acidic pH) | Enhances biofilm penetration and drug release (e.g., gallium, D-arginine) |
| High-MW Chitosan Gel | Inhibits 100% C. albicans adherence to acrylic resin (p < 0.001) |
| Lysostaphin-Chitosan Hydrogel | Reduces S. aureus viability by ∼3 Log10 CFU/mL ex vivo |
| Quaternary Ammonium Chitosan/Alginate LBL Coatings | Reduces Candida biofilms > 90% post-mechanical stress |
| Pore-closed PLGA MPs in Chitosan Hydrogel | Enables sequential vancomycin (burst) and rhBMP-2 (sustained) release |
| ZIF-8 (Naringin loaded) on TiO2 Nanotubes | Enables pH-responsive Zn2+ release; Synergizes antibacterial and osteogenic effects |
| Material | Primary Toxicity Mechanism | Critical Threshold | Key Mitigation Strategy |
|---|---|---|---|
| BGCs | Ag+/Cu2+-induced ROS, pH imbalance | ≥10 wt% Cu2+ inhibits fibroblasts | Sr2+ substitution, polymer hybridization |
| AgNP-Polymers | Ag+ leaching, nanoparticle aggregation | >0.3 wt% in PMMA | MOF confinement, silica-shell encapsulation |
| Chitosan Coatings | pH-dependent membrane disruption | High cationic density at low pH | Crosslinking, buffering agents |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasluianu, R.I.; Bobu, L.; Lupu, I.-C.; Antohe, M.; Bulancea, B.P.; Moldovanu, A.; Stamatin, O.; Holban, C.C.; Dima, A.M. Innovative Smart Materials in Restorative Dentistry. J. Funct. Biomater. 2025, 16, 318. https://doi.org/10.3390/jfb16090318
Vasluianu RI, Bobu L, Lupu I-C, Antohe M, Bulancea BP, Moldovanu A, Stamatin O, Holban CC, Dima AM. Innovative Smart Materials in Restorative Dentistry. Journal of Functional Biomaterials. 2025; 16(9):318. https://doi.org/10.3390/jfb16090318
Chicago/Turabian StyleVasluianu, Roxana Ionela, Livia Bobu, Iulian-Costin Lupu, Magda Antohe, Bogdan Petru Bulancea, Antonia Moldovanu, Ovidiu Stamatin, Catalina Cioloca Holban, and Ana Maria Dima. 2025. "Innovative Smart Materials in Restorative Dentistry" Journal of Functional Biomaterials 16, no. 9: 318. https://doi.org/10.3390/jfb16090318
APA StyleVasluianu, R. I., Bobu, L., Lupu, I.-C., Antohe, M., Bulancea, B. P., Moldovanu, A., Stamatin, O., Holban, C. C., & Dima, A. M. (2025). Innovative Smart Materials in Restorative Dentistry. Journal of Functional Biomaterials, 16(9), 318. https://doi.org/10.3390/jfb16090318







