Nature-Based Hydrogels Combined with Nanoparticles for Bone Regeneration
Abstract
1. Introduction
2. Healing Process of Bone Fractures
3. Bone Regeneration Strategies
4. Nature-Based Hydrogels Combined with Nanoparticles
4.1. Collagen
4.2. Silk Fibroin
4.3. Hyaluronic Acid
4.4. Fibrin
4.5. Alginate
4.6. Chitosan
4.7. Gelatin
4.8. Gellan Gum
5. Polymer Combination
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Ac-BP | Biphosphate nanoparticles |
| AgNPs | Silver nanoparticles |
| Alg | Alginate |
| ALP | Alkaline phosphatase |
| AuNPs | Gold nanoparticles |
| BGNPs | Bioglass nanoparticles |
| BMPs | Bone morphogenetic proteins |
| BTE | Bone tissue engineering |
| Chi | Chitosan |
| CNTs | Carbon nanotubes |
| Coll | Collagen |
| COOH-SWCNTs | Carboxylated single-walled carbon nanotubes |
| CTGF | Connective tissue growth factor |
| Cu-MsNs | Copper-doped mesoporous silica nanospheres |
| ECM | Extracellular matrix |
| FGF | Fibroblast growth factor |
| GG | Gellan gum |
| GNPs | Graphene nanoplatelets |
| GO | Graphene oxide nanoparticles |
| hMSCs | Human mesenchymal stem cells |
| MBG | Mesoporous bioglass nanoparticles |
| MC | Microcrystalline cellulose |
| MeHA | Methacrylated hyaluronic acid |
| MgONPs | Magnesium oxide nanoparticles |
| MSCs | Mesenchymal stem cells |
| nHA | Hydroxyapatite |
| NPs | Nanoparticles |
| PDGF | Platelet-derived growth factor |
| PEGDA | Poly(ethylene glycol)diacrylate |
| PVA | Polyvinyl alcohol |
| Se-BCP | Selenium biphasic calcium NPs |
| SF | Silk fibroin |
| SiNPs | Silica nanoparticles |
| TGF-beta | Transforming growth factor beta |
| TiO2NPs | Titanium dioxide NPs |
| VEGF | Vascular endothelial growth factor |
References
- Feng, X. Chemical and Biochemical Basis of Cell-Bone Matrix Interaction in Health and Disease. Curr. Chem. Biol. 2009, 3, 189–196. [Google Scholar]
- Šromová, V.; Sobola, D.; Kaspar, P. A Brief Review of Bone Cell Function and Importance. Cells 2023, 12, 2576. [Google Scholar] [CrossRef]
- Koushik, T.M.; Miller, C.M.; Antunes, E. Bone Tissue Engineering Scaffolds: Function of Multi-Material Hierarchically Structured Scaffolds. Adv. Healthc. Mater. 2023, 12, 2202766. [Google Scholar] [CrossRef]
- Guillén-Carvajal, K.; Valdez-Salas, B.; Beltrán-Partida, E.; Salomón-Carlos, J.; Cheng, N. Chitosan, Gelatin, and Collagen Hydrogels for Bone Regeneration. Polymers 2023, 15, 2762. [Google Scholar] [CrossRef]
- Liu, J.; Yang, L.; Liu, K.; Gao, F. Hydrogel scaffolds in bone regeneration: Their promising roles in angiogenesis. Front. Pharmacol. 2023, 14, 1050954. [Google Scholar] [CrossRef] [PubMed]
- Florencio-Silva, R.; Sasso, G.R.D.S.; Sasso-Cerri, E.; Simões, M.J.; Cerri, P.S. Biology of Bone Tissue: Structure, Function, and Factors That Influence Bone Cells. Biomed. Res. Int. 2015, 2015, 421746. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Gao, M.; Syed, S.; Zhuang, J.; Xu, X.; Zhang, X.Q. Bioactive hydrogels for bone regeneration. Bioact Mater. 2018, 3, 401–417. [Google Scholar] [CrossRef]
- Walmsley, G.G.; Ransom, R.C.; Zielins, E.R.; Leavitt, T.; Flacco, J.S.; Hu, M.S.; Lee, A.S.; Longaker, M.T.; Wan, D.C. Stem Cells in Bone Regeneration. Stem Cell Rev. Rep. 2016, 12, 524–529. [Google Scholar] [CrossRef] [PubMed]
- ElHawary, H.; Baradaran, A.; Abi-Rafeh, J.; Vorstenbosch, J.; Xu, L.; Efanov, J.I. Bone Healing and Inflammation: Principles of Fracture and Repair. Semin. Plast. Surg. 2021, 35, 198–203. [Google Scholar] [CrossRef]
- Steppe, L.; Megafu, M.; Tschaffon-Müller, M.E.A.; Ignatius, A.; Haffner-Luntzer, M. Fracture healing research: Recent insights. Bone Rep. 2023, 19, 101686. [Google Scholar] [CrossRef]
- Beederman, M.; Lamplot, J.D.; Nan, G.; Wang, J.; Liu, X.; Yin, L.; Li, R.; Shui, W.; Zhang, H.; Kim, S.H.; et al. BMP signaling in mesenchymal stem cell differentiation and bone formation. J. Biomed. Sci. Eng. 2013, 6, 32–52. [Google Scholar] [CrossRef]
- Lee, S.B.; Lee, H.J.; Park, J.B. Bone Morphogenetic Protein-9 Promotes Osteogenic Differentiation and Mineralization in Human Stem-Cell-Derived Spheroids. Medicina 2023, 59, 1315. [Google Scholar] [CrossRef]
- Yue, S.; He, H.; Li, B.; Hou, T. Hydrogel as a Biomaterial for Bone Tissue Engineering: A Review. Nanomaterials 2020, 10, 1511. [Google Scholar] [CrossRef]
- Ho-Shui-Ling, A.; Bolander, J.; Rustom, L.E.; Johnson, A.W.; Luyten, F.P.; Picart, C. Bone regeneration strategies: Engineered scaffolds, bioactive molecules and stem cells current stage and future perspectives. Biomaterials 2018, 180, 143–162. [Google Scholar] [CrossRef]
- Yousefi, A.M. A review of calcium phosphate cements and acrylic bone cements as injectable materials for bone repair and implant fixation. J. Appl. Biomater. Funct. Mater. 2019, 17, 2280800019872594. [Google Scholar] [CrossRef]
- Bai, L.; Tao, G.; Feng, M.; Xie, Y.; Cai, S.; Peng, S.; Xiao, J. Hydrogel Drug Delivery Systems for Bone Regeneration. Pharmaceutics 2023, 15, 1334. [Google Scholar] [CrossRef]
- De Pace, R.; Molinari, S.; Mazzoni, E.; Perale, G. Bone Regeneration: A Review of Current Treatment Strategies. J. Clin. Med. 2025, 14, 1838. [Google Scholar] [CrossRef] [PubMed]
- Ferraz, M.P. Bone Grafts in Dental Medicine: An Overview of Autografts, Allografts and Synthetic Materials. Materials 2023, 16, 4117. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Wang, H.; Zhong, L.; Wei, S.; Min, X.; Deng, H.; Zhang, X.; Zhong, M.; Huang, Y. Bioactivity and biomedical applications of pomegranate peel extract: A comprehensive review. Front. Pharmacol. 2025, 16, 1569141. [Google Scholar] [CrossRef] [PubMed]
- Correa, S.; Grosskopf, A.K.; Lopez Hernandez, H.; Chan, D.; Yu, A.C.; Stapleton, L.M.; Appel, E.A. Translational Applications of Hydrogels. Chem. Rev. 2021, 121, 11385–11457. [Google Scholar] [CrossRef]
- Deng, X.; Gould, M.; Ali, M.A. A review of current advancements for wound healing: Biomaterial applications and medical devices. J. Biomed. Mater. Res. B Appl. Biomater. 2022, 110, 2542–2573. [Google Scholar] [CrossRef]
- Cao, H.; Duan, L.; Zhang, Y.; Cao, J.; Zhang, K. Current hydrogel advances in physicochemical and biological response-driven biomedical application diversity. Signal Transduct. Target Ther. 2021, 6, 426. [Google Scholar] [CrossRef]
- Sun, Y.; Chen, L.G.; Fan, X.M.; Pang, J.L. Ultrasound Responsive Smart Implantable Hydrogels for Targeted Delivery of Drugs: Reviewing Current Practices. Int. J. Nanomed. 2022, 17, 5001–5026. [Google Scholar] [CrossRef]
- Jeong, D.; Jang, S.Y.; Roh, S.; Choi, J.H.; Seo, I.J.; Lee, J.H.; Kim, J.; Kwon, I.; Jung, Y.; Hwang, J.; et al. Sprayable hydrogel with optical mRNA nanosensors for Real-Time monitoring and healing of diabetic wounds. Chem. Eng. J. 2024, 493, 152711. [Google Scholar] [CrossRef]
- Alonso, J.M.; Andrade del Olmo, J.; Perez Gonzalez, R.; Saez-Martinez, V. Injectable Hydrogels: From Laboratory to Industrialization. Polymers 2021, 13, 650. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, L.M. Intelligent biobased hydrogels for diabetic wound healing: A review. Chem. Eng. J. 2024, 484, 149493. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, Y.; Yang, G.; Kong, H.; Guo, L.; Wei, G. Recent advances in protein hydrogels: From design, structural and functional regulations to healthcare applications. Chem. Eng. J. 2023, 451, 138494. [Google Scholar] [CrossRef]
- García-García, P.; Reyes, R.; Pérez-Herrero, E.; Arnau, M.R.; Évora, C.; Delgado, A. Alginate-hydrogel versus alginate-solid system. Efficacy in bone regeneration in osteoporosis. Mater. Sci. Eng. C 2020, 115, 111009. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Zhang, X.; Zhou, H. Biomimetic Hydrogel Applications and Challenges in Bone, Cartilage, and Nerve Repair. Pharmaceutics. 2023, 15, 2405. [Google Scholar] [CrossRef] [PubMed]
- Scheinpflug, J.; Pfeiffenberger, M.; Damerau, A.; Schwarz, F.; Textor, M.; Lang, A.; Schulze, F. Journey into Bone Models: A Review. Genes 2018, 9, 247. [Google Scholar] [CrossRef]
- Li, B.; Li, C.; Yan, Z.; Yang, X.; Xiao, W.; Zhang, D.; Liu, Z.; Liao, X. A review of self-healing hydrogels for bone repair and regeneration: Materials, mechanisms, and applications. Int. J. Biol. Macromol. 2025, 287, 138323. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wu, Y.; Zhang, X.; Wu, T.; Huang, K.; Wang, B.; Liao, J. Self-healing hydrogels for bone defect repair. RSC Adv. 2023, 13, 16773–16788. [Google Scholar] [CrossRef]
- Choi, H.; Choi, W.S.; Jeong, J.O. A Review of Advanced Hydrogel Applications for Tissue Engineering and Drug Delivery Systems as Biomaterials. Gels 2024, 10, 693. [Google Scholar] [CrossRef]
- Ho, T.C.; Chang, C.C.; Chan, H.P.; Chung, T.W.; Shu, C.W.; Chuang, K.P.; Duh, T.-H.; Yang, M.-H.; Tyan, Y.-C. Hydrogels: Properties and Applications in Biomedicine. Molecules 2022, 27, 2902. [Google Scholar] [CrossRef]
- Wen, J.; Cai, D.; Gao, W.; He, R.; Li, Y.; Zhou, Y.; Klein, T.; Xiao, L.; Xiao, Y. Osteoimmunomodulatory Nanoparticles for Bone Regeneration. Nanomaterials 2023, 13, 692. [Google Scholar] [CrossRef]
- Ahmad, N.; Bukhari, S.N.A.; Hussain, M.A.; Ejaz, H.; Munir, M.U.; Amjad, M.W. Nanoparticles incorporated hydrogels for delivery of antimicrobial agents: Developments and trends. RSC Adv. 2024, 14, 13535–13564. [Google Scholar] [CrossRef] [PubMed]
- Lyons, J.G.; Plantz, M.A.; Hsu, W.K.; Hsu, E.L.; Minardi, S. Nanostructured Biomaterials for Bone Regeneration. Front. Bioeng. Biotechnol. 2020, 8, 922. [Google Scholar] [CrossRef]
- Kupikowska-Stobba, B.; Kasprzak, M. Fabrication of nanoparticles for bone regeneration: New insight into applications of nanoemulsion technology. J. Mater. Chem. B 2021, 9, 5221–5244. [Google Scholar] [CrossRef]
- Hajiali, H.; Ouyang, L.; Llopis-Hernandez, V.; Dobre, O.; Rose, F.R.A.J. Review of emerging nanotechnology in bone regeneration: Progress, challenges, and perspectives. Nanoscale 2021, 13, 10266–10280. [Google Scholar] [CrossRef]
- Saiz, E.; Zimmermann, E.A.; Lee, J.S.; Wegst, U.G.K.; Tomsia, A.P. Perspectives on the role of nanotechnology in bone tissue engineering. Dental Mater. 2013, 29, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Sun, S.; Wang, N.; Kang, R.; Xie, L.; Liu, X. Therapeutic application of hydrogels for bone-related diseases. Front. Bioeng. Biotechnol. 2022, 10, 998988. [Google Scholar] [CrossRef]
- Hwang, H.S.; Lee, C.S. Nanoclay-Composite Hydrogels for Bone Tissue Engineering. Gels 2024, 10, 513. [Google Scholar] [CrossRef]
- Omidian, H.; Chowdhury, S.D. Advancements and Applications of Injectable Hydrogel Composites in Biomedical Research and Therapy. Gels 2023, 9, 533. [Google Scholar] [CrossRef]
- Kuang, L.; Ma, X.; Ma, Y.; Yao, Y.; Tariq, M.; Yuan, Y.; Changsheng, L. Self-Assembled Injectable Nanocomposite Hydrogels Coordinated by in Situ Generated CaP Nanoparticles for Bone Regeneration. ACS Appl. Mater. Interfaces 2019, 11, 17234–17246. [Google Scholar] [CrossRef] [PubMed]
- Du, C.; Huang, W. Progress and prospects of nanocomposite hydrogels in bone tissue engineering. Nanocomposites 2022, 8, 102–124. [Google Scholar] [CrossRef]
- Pablos, J.L.; Lozano, D.; Manzano, M.; Vallet-Regí, M. Regenerative medicine: Hydrogels and mesoporous silica nanoparticles. Mater. Today Bio. 2024, 29, 101342. [Google Scholar] [CrossRef]
- Alshangiti, D.M.; El-damhougy, T.K.; Zaher, A.; Madani, M.; Mohamady ghobashy, M. Revolutionizing biomedicine: Advancements, applications, and prospects of nanocomposite macromolecular carbohydrate-based hydrogel biomaterials: A review. RSC Adv. 2023, 13, 35251–35291. [Google Scholar] [CrossRef] [PubMed]
- Ao, Y.; Zhang, E.; Liu, Y.; Yang, L.; Li, J.; Wang, F. Advanced Hydrogels With Nanoparticle Inclusion for Cartilage Tissue Engineering. Front. Bioeng. Biotechnol. 2022, 10, 951513. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Gurav, N.; Elsharkawy, S.; Deb, S. Hydrogel Composite Magnetic Scaffolds: Toward Cell-Free In Situ Bone Tissue Engineering. ACS Appl. Bio. Mater. 2024, 7, 168–181. [Google Scholar] [CrossRef]
- Suba Sri, M.; Usha, R. An insightful overview on osteogenic potential of nano hydroxyapatite for bone regeneration. Cell Tissue Bank. 2025, 26, 13. [Google Scholar] [CrossRef]
- Zhang, G.; Zhen, C.; Yang, J.; Wang, J.; Wang, S.; Fang, Y.; Shang, P. Recent advances of nanoparticles on bone tissue engineering and bone cells. Nanoscale Adv. 2024, 6, 1957–1973. [Google Scholar] [CrossRef]
- Gong, T.; Xie, J.; Liao, J.; Zhang, T.; Lin, S.; Lin, Y. Nanomaterials and bone regeneration. Bone Res. 2015, 3, 15029. [Google Scholar] [CrossRef]
- Filippi, M.; Born, G.; Chaaban, M.; Scherberich, A. Natural Polymeric Scaffolds in Bone Regeneration. Front. Bioeng. Biotechnol. 2020, 8, 474. [Google Scholar] [CrossRef] [PubMed]
- Pourhajrezaei, S.; Abbas, Z.; Khalili, M.A.; Madineh, H.; Jooya, H.; Babaeizad, A.; Gross, J.D.; Samadi, A. Bioactive polymers: A comprehensive review on bone grafting biomaterials. Int. J. Biol. Macromol. 2024, 278, 134615. [Google Scholar] [CrossRef]
- Montalbano, G.; Borciani, G.; Pontremoli, C.; Ciapetti, G.; Mattioli-Belmonte, M.; Fiorilli, S.; Vitale-Brovarone, C. Development and Biocompatibility of Collagen-Based Composites Enriched with Nanoparticles of Strontium Containing Mesoporous Glass. Materials 2019, 12, 3719. [Google Scholar] [CrossRef] [PubMed]
- Borciani, G.; Montalbano, G.; Melo, P.; Baldini, N.; Ciapetti, G.; Vitale-Brovarone, C. Assessment of Collagen-Based Nanostructured Biomimetic Systems with a Co-Culture of Human Bone-Derived Cells. Cells 2021, 11, 26. [Google Scholar] [CrossRef]
- Naruphontjirakul, P.; Panpisut, P.; Patntirapong, S. Zinc and Strontium-Substituted Bioactive Glass Nanoparticle/Alginate Composites Scaffold for Bone Regeneration. Int. J. Mol. Sci. 2023, 24, 6150. [Google Scholar] [CrossRef]
- Deshmukh, K.; Kovářík, T.; Křenek, T.; Docheva, D.; Stich, T.; Pola, J. Recent advances and future perspectives of sol–gel derived porous bioactive glasses: A review. RSC Adv. 2020, 10, 33782–33835. [Google Scholar] [CrossRef]
- Lee, K.Z.; Jeon, J.; Jiang, B.; Subramani, S.V.; Li, J.; Zhang, F. Protein-Based Hydrogels and Their Biomedical Applications. Molecules 2023, 28, 4988. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, Z.; Chen, X.; Jiang, X.; Fu, G. Silk fibroin hydrogel membranes prepared by a sequential cross-linking strategy for guided bone regeneration. J. Mech. Behav. Biomed. Mater. 2023, 147, 106133. [Google Scholar] [CrossRef] [PubMed]
- Amorim, S.; Dudik, O.; Soares da Costa, D.; Reis, R.L.; Silva, T.H.; Pires, R.A. Fucoidan-Coated Silica Nanoparticles Promote the Differentiation of Human Mesenchymal Stem Cells into the Osteogenic Lineage. ACS Biomater. Sci. Eng. 2023, 9, 4907–4915. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, M.; de Moraes, M.A.; Beppu, M.M.; Garcia, M.P.; Fernandes, M.H.; Monteiro, F.J.; Ferraz, M.P. Development of silk fibroin/nanohydroxyapatite composite hydrogels for bone tissue engineering. Eur. Polym. J. 2015, 67, 66–77. [Google Scholar] [CrossRef]
- Ma, W.; Chen, H.; Cheng, S.; Wu, C.; Wang, L.; Du, M. Gelatin hydrogel reinforced with mussel-inspired polydopamine-functionalized nanohydroxyapatite for bone regeneration. Int. J. Biol. Macromol. 2023, 240, 124287. [Google Scholar] [CrossRef]
- Barros, J.; Ferraz, M.P.; Azeredo, J.; Fernandes, M.H.; Gomes, P.S.; Monteiro, F.J. Alginate-nanohydroxyapatite hydrogel system: Optimizing the formulation for enhanced bone regeneration. Mater. Sci. Eng. C 2019, 105, 109985. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, M.; Ferraz, M.P.; Monteiro, F.J.; Fernandes, M.H.; Beppu, M.M.; Mantione, D.; Sardon, H. Antibacterial silk fibroin/nanohydroxyapatite hydrogels with silver and gold nanoparticles for bone regeneration. Nanomedicine 2017, 13, 231–239. [Google Scholar] [CrossRef]
- Yang, D.H.; Nah, H.; Lee, D.; Min, S.J.; Park, S.; An, S.H.; Wang, J.; He, H.; Choi, K.-S.; Ko, W.-K.; et al. A review on gold nanoparticles as an innovative therapeutic cue in bone tissue engineering: Prospects and future clinical applications. Mater. Today Bio. 2024, 26, 101016. [Google Scholar] [CrossRef]
- Cheng, Y.; Cheng, G.; Xie, C.; Yin, C.; Dong, X.; Li, Z.; Zhou, X.; Wang, Q.; Deng, H.; Li, Z. Biomimetic Silk Fibroin Hydrogels Strengthened by Silica Nanoparticles Distributed Nanofibers Facilitate Bone Repair. Adv. Healthc. Mater. 2021, 10, 2001646. [Google Scholar] [CrossRef]
- Daneshvar, A.; Farokhi, M.; Bonakdar, S.; Vossoughi, M. Synthesis and characterization of injectable thermosensitive hydrogel based on Pluronic-grafted silk fibroin copolymer containing hydroxyapatite nanoparticles as potential for bone tissue engineering. Int. J. Biol. Macromol. 2024, 277, 134412. [Google Scholar] [CrossRef]
- Hwang, H.S.; Lee, C.S. Recent Progress in Hyaluronic-Acid-Based Hydrogels for Bone Tissue Engineering. Gels 2023, 9, 588. [Google Scholar] [CrossRef]
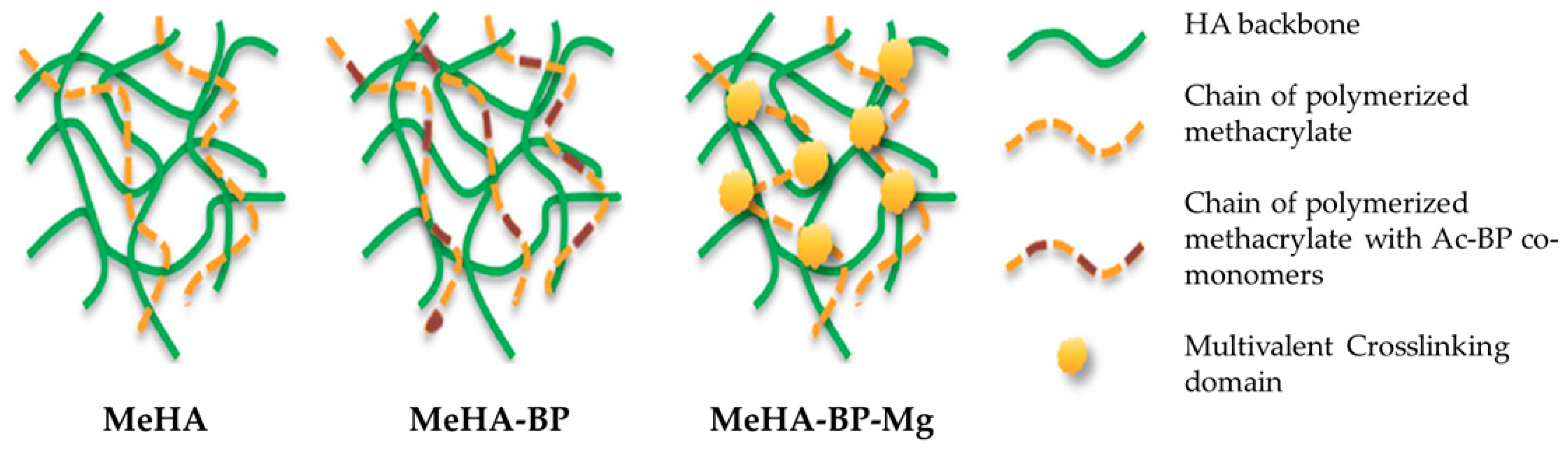
- Zhang, K.; Lin, S.; Feng, Q.; Dong, C.; Yang, Y.; Li, G.; Bian, L. Nanocomposite hydrogels stabilized by self-assembled multivalent bisphosphonate-magnesium nanoparticles mediate sustained release of magnesium ion and promote in-situ bone regeneration. Acta Biomater. 2017, 64, 389–400. [Google Scholar] [CrossRef]
- Pathmanapan, S.; Periyathambi, P.; Anandasadagopan, S.K. Fibrin hydrogel incorporated with graphene oxide functionalized nanocomposite scaffolds for bone repair—In vitro and in vivo study. Nanomedicine 2020, 29, 102251. [Google Scholar] [CrossRef]
- Brinkmann, J.; Malyaran, H.; Enezy-Ulbrich MAAl Jung, S.; Radermacher, C.; Buhl, E.M.; Pich, A.; Neuss, S. Assessment of Fibrin-Based Hydrogels Containing a Fibrin-Binding Peptide to Tune Mechanical Properties and Cell Responses. Macromol. Mater. Eng. 2023, 308, 2200678. [Google Scholar] [CrossRef]
- Govindarajan, D.; Saravanan, S.; Sudhakar, S.; Vimalraj, S. Graphene: A Multifaceted Carbon-Based Material for Bone Tissue Engineering Applications. ACS Omega 2024, 9, 67–80. [Google Scholar] [CrossRef]
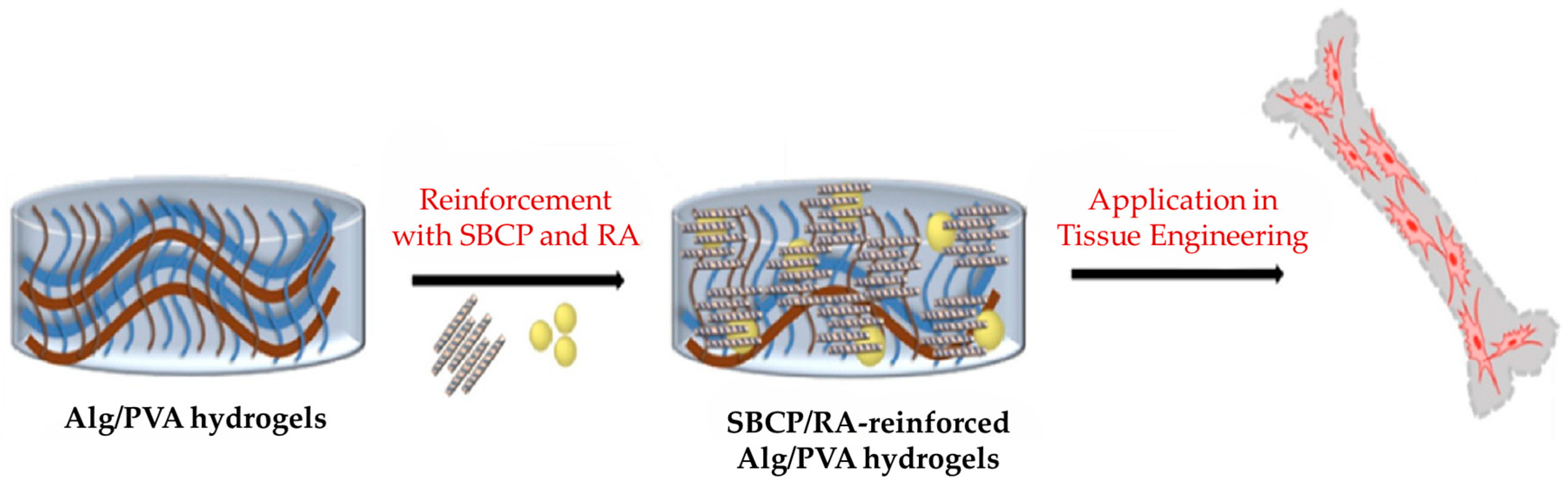
- Singhmar, R.; Son, Y.; Jo, Y.J.; Zo, S.; Min, B.K.; Sood, A.; Han, S.S. Fabrication of alginate composite hydrogel encapsulated retinoic acid and nano Se doped biphasic CaP to augment in situ mineralization and osteoimmunomodulation for bone regeneration. Int. J. Biol. Macromol. 2024, 275, 133597. [Google Scholar] [CrossRef]
- Chen, Y.; Sheng, W.; Lin, J.; Fang, C.; Deng, J.; Zhang, P.; Zhou, M.; Liu, P.; Weng, J.; Yu, F.; et al. Magnesium Oxide Nanoparticle Coordinated Phosphate-Functionalized Chitosan Injectable Hydrogel for Osteogenesis and Angiogenesis in Bone Regeneration. ACS Appl. Mater. Interfaces 2022, 14, 7592–7608. [Google Scholar] [CrossRef] [PubMed]
- Malaiappan, S.; Harris, J. Osteogenic Potential of Magnesium Oxide Nanoparticles in Bone Regeneration: A Systematic Review. Cureus 2024, 16, e55502. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, S.F.; Galefi, A.; Hosseini, S.; Shaabani, A.; Farrokhi, N.; Jahanfar, M.; Nourany, M.; Homaeigohar, S.; Alipour, A.; Shahsavarani, H. Magnesium oxide nanoparticle reinforced pumpkin-derived nanostructured cellulose scaffold for enhanced bone regeneration. Int. J. Biol. Macromol. 2024, 281, 136303. [Google Scholar] [CrossRef]
- Chen, Y.; Li, C.; Wang, Z.; Long, J.; Wang, R.; Zhao, J.; Tang, W.; Zhao, Y.; Qin, L.; Peng, S.; et al. Self-assembled nanocomposite hydrogels enhanced by nanoparticles phosphonate-magnesium coordination for bone regeneration. Appl. Mater. Today 2021, 25, 101182. [Google Scholar] [CrossRef]
- Paltanea, G.; Manescu (Paltanea), V.; Antoniac, I.; Antoniac, A.; Nemoianu, I.V.; Robu, A.; Dura, H. A Review of Biomimetic and Biodegradable Magnetic Scaffolds for Bone Tissue Engineering and Oncology. Int. J. Mol. Sci. 2023, 24, 4312. [Google Scholar] [CrossRef]
- Li, J.; Wang, W.; Li, M.; Song, P.; Lei, H.; Gui, X.; Zhou, C.; Liu, L. Biomimetic Methacrylated Gelatin Hydrogel Loaded With Bone Marrow Mesenchymal Stem Cells for Bone Tissue Regeneration. Front. Bioeng. Biotechnol. 2021, 9, 770049. [Google Scholar] [CrossRef]
- Heo, D.N.; Ko, W.K.; Bae, M.S.; Lee, J.B.; Lee, D.W.; Byun, W.; Zhou, C.; Liu, L. Enhanced bone regeneration with a gold nanoparticle–hydrogel complex. J. Mater. Chem. B 2014, 2, 1584–1593. [Google Scholar] [CrossRef]
- Wu, S.; Xiao, R.; Wu, Y.; Xu, L. Advances in tissue engineering of gellan gum-based hydrogels. Carbohydr. Polym. 2024, 324, 121484. [Google Scholar] [CrossRef]
- Thangavelu, M.; Kim, P.Y.; Cho, H.; Song, J.E.; Park, S.; Bucciarelli, A.; Khang, G. A Gellan Gum, Polyethylene Glycol, Hydroxyapatite Composite Scaffold with the Addition of Ginseng Derived Compound K with Possible Applications in Bone Regeneration. Gels 2024, 10, 257. [Google Scholar] [CrossRef]
- Liu, H.; Li, K.; Guo, B.; Yuan, Y.; Ruan, Z.; Long, H.; Zhu, J.; Zhu, Y.; Chen, C. Engineering an injectable gellan gum-based hydrogel with osteogenesis and angiogenesis for bone regeneration. Tissue Cell 2024, 86, 102279. [Google Scholar] [CrossRef]
- Douglas, T.E.L.; Piwowarczyk, W.; Pamula, E.; Liskova, J.; Schaubroeck, D.; Leeuwenburgh, S.C.G.; Brackman, G.; Balcaen, L.; Detsch, R.; Declercq, H.; et al. Injectable self-gelling composites for bone tissue engineering based on gellan gum hydrogel enriched with different bioglasses. Biomed. Mater. 2014, 9, 045014. [Google Scholar] [CrossRef]
- Shahrebabaki, K.E.; Labbaf, S.; Karimzadeh, F.; Goli, M.; Mirhaj, M. Alginate-gelatin based nanocomposite hydrogel scaffold incorporated with bioactive glass nanoparticles and fragmented nanofibers promote osteogenesis: From design to in vitro studies. Int. J. Biol. Macromol. 2024, 282, 137104. [Google Scholar] [CrossRef] [PubMed]
- Chavez-Granados, P.A.; Garcia-Contreras, R.; Reyes-Lopez, C.A.S.; Correa-Basurto, J.; Hernandez-Rojas, I.E.; Hernandez-Gomez, G.; Jurado, C.A.; Alhotan, A. Green Synthesis of Silver Nanoparticles with Roasted Green Tea: Applications in Alginate–Gelatin Hydrogels for Bone Regeneration. Gels 2024, 10, 706. [Google Scholar] [CrossRef]
- Alajmi, K.; Hartford, M.; Roy, N.S.; Bhattacharya, A.; Kaity, S.; Cavanagh, B.L.; Roy, S.; Kaur, K. Selenium nanoparticle-functionalized injectable chitosan/collagen hydrogels as a novel therapeutic strategy to enhance stem cell osteoblastic differentiation for bone regeneration. J. Mater. Chem. B 2024, 12, 9268–9282. [Google Scholar] [CrossRef] [PubMed]
- Karthik, K.K.; Cheriyan, B.V.; Rajeshkumar, S.; Gopalakrishnan, M. A review on selenium nanoparticles and their biomedical applications. Biomed. Technol. 2024, 6, 61–74. [Google Scholar] [CrossRef]
- Kaur, K.; Paiva, S.S.; Caffrey, D.; Cavanagh, B.L.; Murphy, C.M. Injectable chitosan/collagen hydrogels nano-engineered with functionalized single wall carbon nanotubes for minimally invasive applications in bone. Mater. Sci. Eng. C 2021, 128, 112340. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, X. The utilization of carbon-based nanomaterials in bone tissue regeneration and engineering: Respective featured applications and future prospects. Med. Nov. Technol. Devices 2022, 16, 100168. [Google Scholar] [CrossRef]
- Pei, B.; Wang, W.; Dunne, N.; Li, X. Applications of Carbon Nanotubes in Bone Tissue Regeneration and Engineering: Superiority, Concerns, Current Advancements, and Prospects. Nanomaterials 2019, 9, 1501. [Google Scholar] [CrossRef]
- Seifi, S.; Shamloo, A.; Barzoki, A.K.; Bakhtiari, M.A.; Zare, S.; Cheraghi, F.; Peyrovan, A. Engineering biomimetic scaffolds for bone regeneration: Chitosan/alginate/polyvinyl alcohol-based double-network hydrogels with carbon nanomaterials. Carbohydr. Polym. 2024, 339, 122232. [Google Scholar] [CrossRef]
- Raslan, A.; Saenz del Burgo, L.; Ciriza, J.; Pedraz, J.L. Graphene oxide and reduced graphene oxide-based scaffolds in regenerative medicine. Int. J. Pharm. 2020, 580, 119226. [Google Scholar] [CrossRef]
- Wang, S.; Lei, H.; Mi, Y.; Ma, P.; Fan, D. Chitosan and hyaluronic acid based injectable dual network hydrogels—Mediating antimicrobial and inflammatory modulation to promote healing of infected bone defects. Int. J. Biol. Macromol. 2024, 274, 133124. [Google Scholar] [CrossRef] [PubMed]
- Hia, E.M.; Jang, S.R.; Maharjan, B.; Park, J.; Park, C.H.; Kim, C.S. Construction of a PEGDA/chitosan hydrogel incorporating mineralized copper-doped mesoporous silica nanospheres for accelerated bone regeneration. Int. J. Biol. Macromol. 2024, 262, 130218. [Google Scholar] [CrossRef] [PubMed]
- Kasi, P.B.; Azar, M.G.; Dodda, J.M.; Bělský, P.; Kovářík, T.; Šlouf, M.; Dobrá, J.K.; Babuška, V. Chitosan and cellulose-based composite hydrogels with embedded titanium dioxide nanoparticles as candidates for biomedical applications. Int. J. Biol. Macromol. 2023, 243, 125334. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.H.; Qi, C.; Ma, M.G.; Wan, P. Multifunctional cellulose-based hydrogels for biomedical applications. J. Mater. Chem. B 2019, 7, 1541–1562. [Google Scholar] [CrossRef]
- Kundu, R.; Mahada, P.; Chhirang, B.; Das, B. Cellulose hydrogels: Green and sustainable soft biomaterials. Curr. Res. Green Sustain. Chem. 2022, 5, 100252. [Google Scholar] [CrossRef]
- Saber-Samandari, S.; Yekta, H.; Ahmadi, S.; Alamara, K. The role of titanium dioxide on the morphology, microstructure, and bioactivity of grafted cellulose/hydroxyapatite nanocomposites for a potential application in bone repair. Int. J. Biol. Macromol. 2018, 106, 481–488. [Google Scholar] [CrossRef]
- Rubina, A.; Sceglovs, A.; Ramata-Stunda, A.; Pugajeva, I.; Skadins, I.; Boyd, A.R.; Tumilovica, A.; Stipniece, L.; Salma-Ancane, K. Injectable mineralized Sr-hydroxyapatite nanoparticles-loaded ɛ-polylysine-hyaluronic acid composite hydrogels for bone regeneration. Int. J. Biol. Macromol. 2024, 280, 135703. [Google Scholar] [CrossRef] [PubMed]


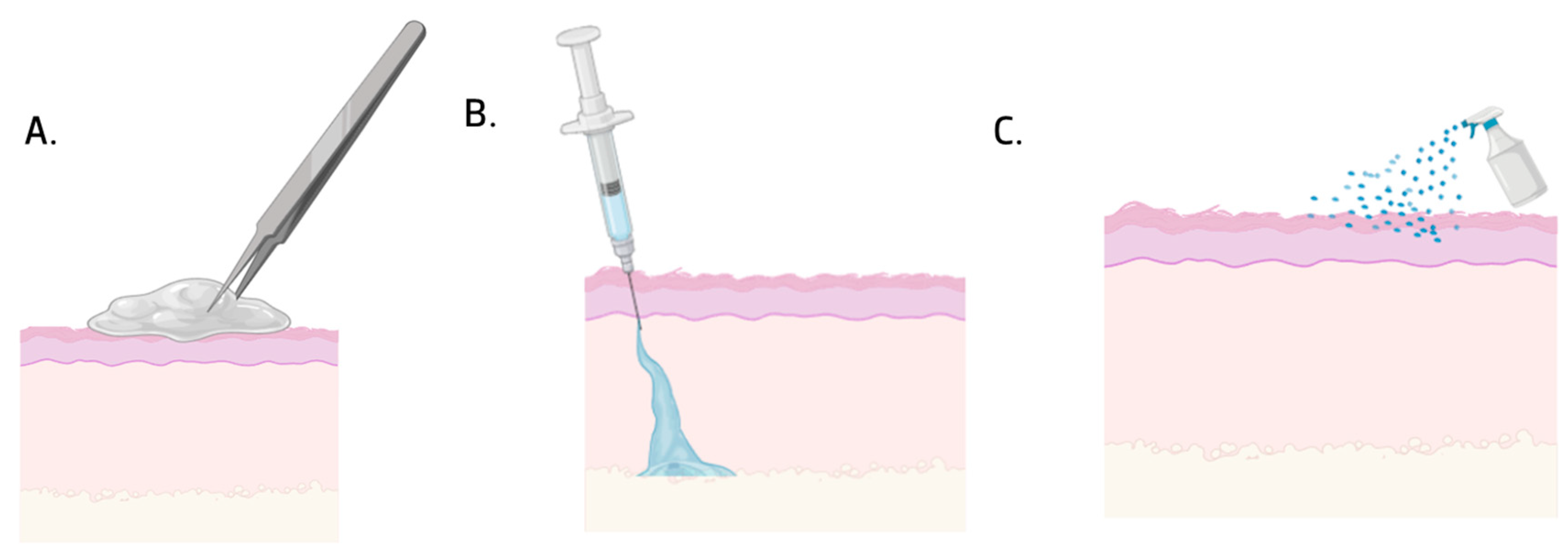


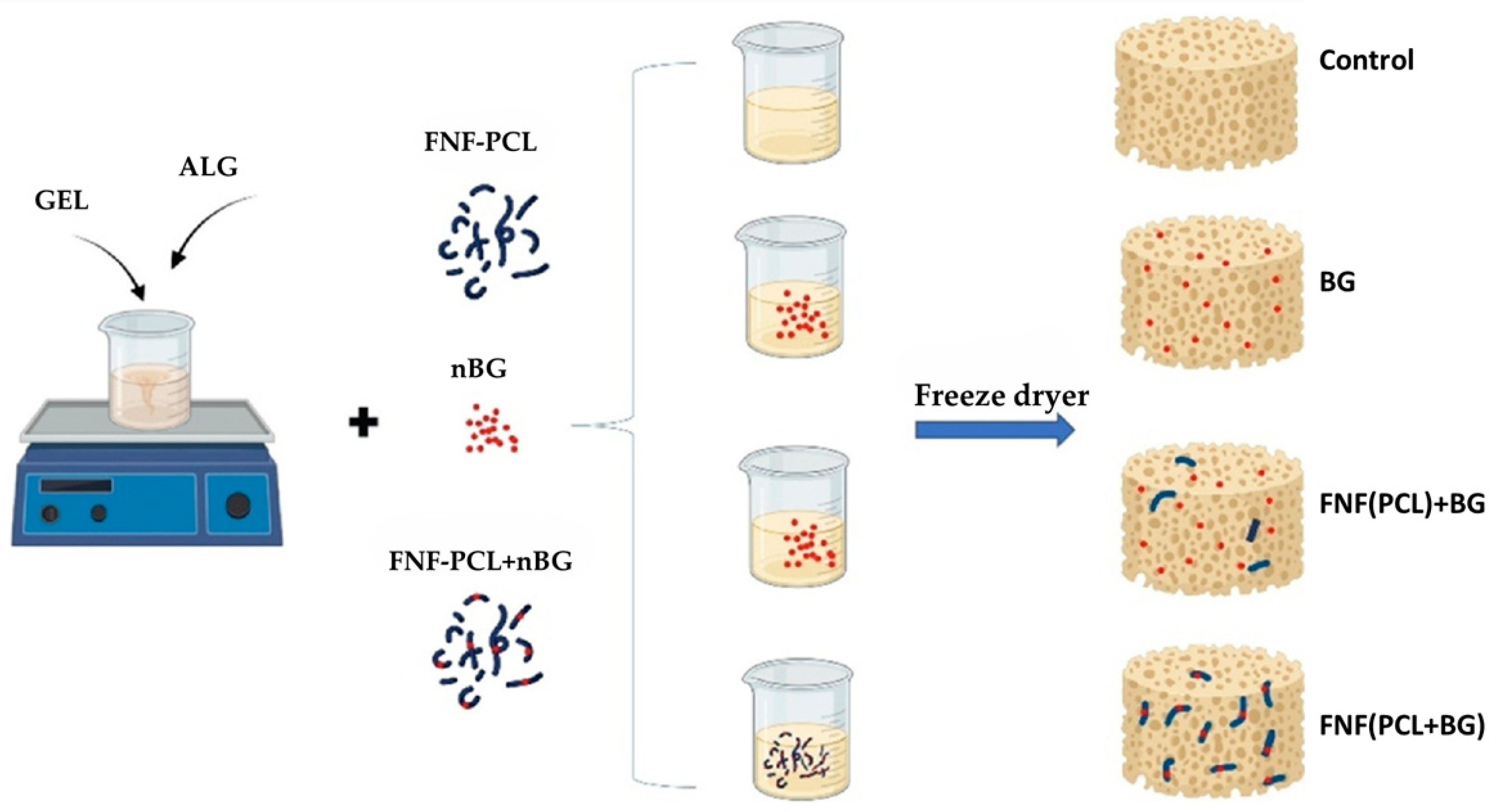


| Composite | Features | Reference |
|---|---|---|
| Collagen–chitosan + selenium NPs | Increase in cell proliferation and osteogenic differentiation of MSCs | [88] |
| Gelatin + gold NPs | Osteogenic differentiation ADSC mineralization In vivo new bone formation | [81] |
| Chitosan/collagen + carboxylated single-walled carbon nanotubes | Increase in MC3T3-E1 cell proliferation Augmentation of osteogenic markers expression, calcium, and ALP | [90] |
| Alginate + selenium doped with biphasic calcium phosphate NPs + retinoic acid | Bioactive hydrogel with formation of HAp crystals Increase MC3T3-E1 cell viability and proliferation | [74] |
| PVA + chitosan + sodium alginate + carbon nanotubes or graphene nanoplatelets | Augmentation of MG-63 cell viability Swelling and degradation are favorable for bone tissue engineering | [93] |
| Gellan gum + nanohydroxyapatite + magnesium sulfate | Increase cell viability and proliferation of BMSCs Good cell adhesion Increase in the expression of osteogenic genes Osteogenic differentiation In vivo stimulated vascularization and new bone formation | [84] |
| Pluronic grafted silk fibroin hydrogel + hydroxyapatite NPs | Osteogenic promotion Cytocompatibility and cell proliferation of MG-63 | [67] |
| Gelatin (catechol) + hydroxyapatite NPs | Cytocompatibility to MC3T3-E1 cells In vivo bone formation | [63] |
| Chitosan (sulfated) + oxidized hyaluronic acid + CuSrBG mesoporous bioactive glass dopes | Promotes osteogenesis and osteogenic differentiation of hBMSCs Increase in hBMSC viability In vivo bone regeneration and antibacterial activity against S.aureus | [95] |
| Silk fibroin/nanohydroxyapatite + silver NPs + gold NPs | Increase in MG-63 cell viability Allowed osteoblast adhesion and spreading | [65] |
| Chitosan (modified) + MgO NPs | In vitro mineralization Increase in osteogenic differentiation of MC3T3-E1 In vivo bone formation | [78] |
| Hyaluronic acid + biphosphate–magnesium NPs | Increase in cell adhesion and spreading of hMSCs Promoted mineralization and osteogenic differentiation In situ bone regeneration In vivo bone formation | [70] |
| Alginate + nanohydroxyapatite | In vitro osteoblast proliferation of hMSCs Ex vivo studies showed collagenase deposition and trabecular bone formation | [64] |
| Alginate–gelatin + fragmented nanofiber of polycaprolactone + bioactive glass NPs | Supported cell growth and proliferation of MG-63 Allowed cell adhesion | [86] |
| Chitosan phosphocreatine (functionalized) + MgO NPs | Increased cell proliferation of MC3T3-E1 Osteogenic differentiation Elevated expression of osteogenic genes In vivo bone regeneration | [75] |
| Poly (ethylene glycol) diacrylate (PEGDA) + chitosan + copper-doped mesoporous silica nanospheres | Elevated mechanical stability Increase MC3TE-E1 cell viability Osteogenic differentiation | [96] |
| Alginate–gelatin + silver NPs | Increase the cell viability of SHEDs Promotes osteogenic differentiation, with an increase in ALP activity | [87] |
| Fibrin + graphene oxide NPs | Enhanced cell viability at concentrations of 40 mg/mL Increased ALP activity and osteogenic differentiation In vivo bone healing | [71] |
| Silk fibroin + hydroxyapatite NPs | Increase in the ALP activity | [62] |
| Silk fibroin + silica NPs | Enhanced cell adhesion and proliferation of MC3T3-E1 In vitro osteogenic differentiation In vivo bone formation in cranial fractures | [67] |
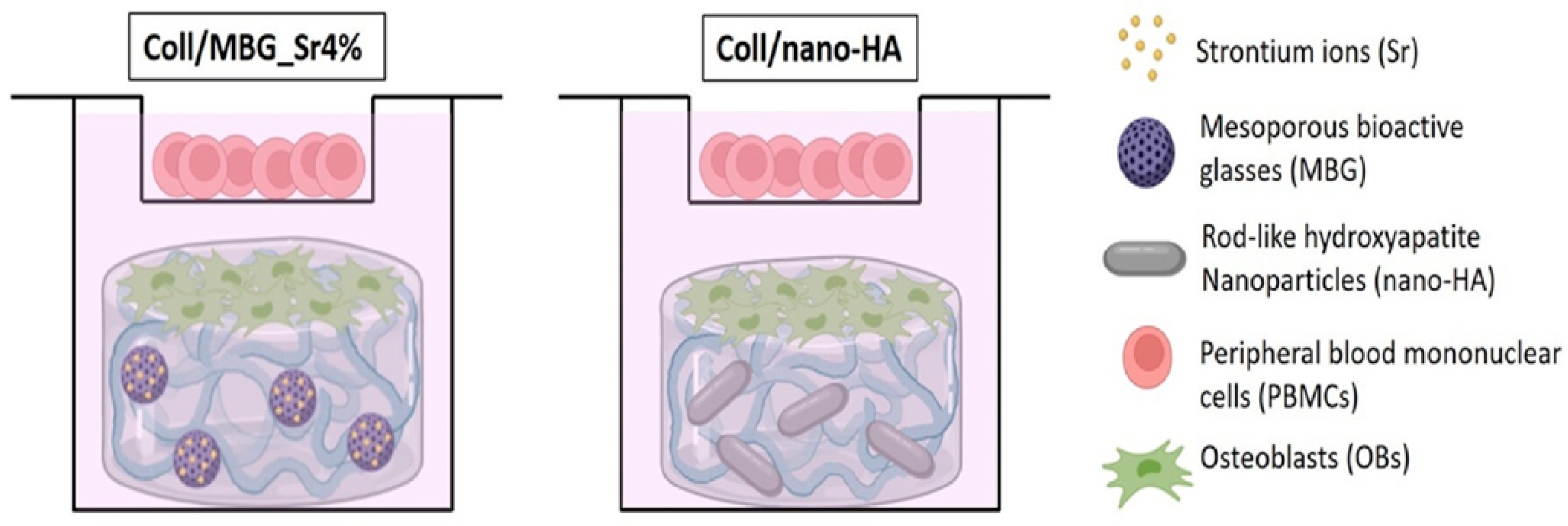
| Collagen + strontium-containing nanosized mesoporous bioglass NPs | Osteogenic differentiation Viable to MG-63 cells Allow cell adhesion to the hydrogel structure | [55] |
| Collagen + strontium-enriched mesoporous bioglass or rod-like hydroxyapatite | Increase in PBMC viability Increased values of ALP activity | [56] |
| Chitosan + cellulose hydrogels + titanium dioxide NPs | Allowed cell adhesion, spreading, and MG-63 osteoblast differentiation | [97] |
| ε-polylysine + hyaluronic acid + Sr-substituted hydroxyapatite NPs | Osteogenic differentiation Increase the cell proliferation of MC3T3-E1 and MG-63 | [101] |
| Gellan gum + bioglass nanoparticles | Bioactive hydrogels with the formation of HAp crystals Antibacterial activity against MRSA Cytocompatibility and supported MG-63 cell growth | [85] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernandes, M.; Vieira, M.; Peixoto, D.; Alves, N.M. Nature-Based Hydrogels Combined with Nanoparticles for Bone Regeneration. J. Funct. Biomater. 2025, 16, 317. https://doi.org/10.3390/jfb16090317
Fernandes M, Vieira M, Peixoto D, Alves NM. Nature-Based Hydrogels Combined with Nanoparticles for Bone Regeneration. Journal of Functional Biomaterials. 2025; 16(9):317. https://doi.org/10.3390/jfb16090317
Chicago/Turabian StyleFernandes, Margarida, Mónica Vieira, Daniela Peixoto, and Natália M. Alves. 2025. "Nature-Based Hydrogels Combined with Nanoparticles for Bone Regeneration" Journal of Functional Biomaterials 16, no. 9: 317. https://doi.org/10.3390/jfb16090317
APA StyleFernandes, M., Vieira, M., Peixoto, D., & Alves, N. M. (2025). Nature-Based Hydrogels Combined with Nanoparticles for Bone Regeneration. Journal of Functional Biomaterials, 16(9), 317. https://doi.org/10.3390/jfb16090317







