A Review on Genus Halichondria (Demospongiae, Porifera)
Abstract
1. Introduction
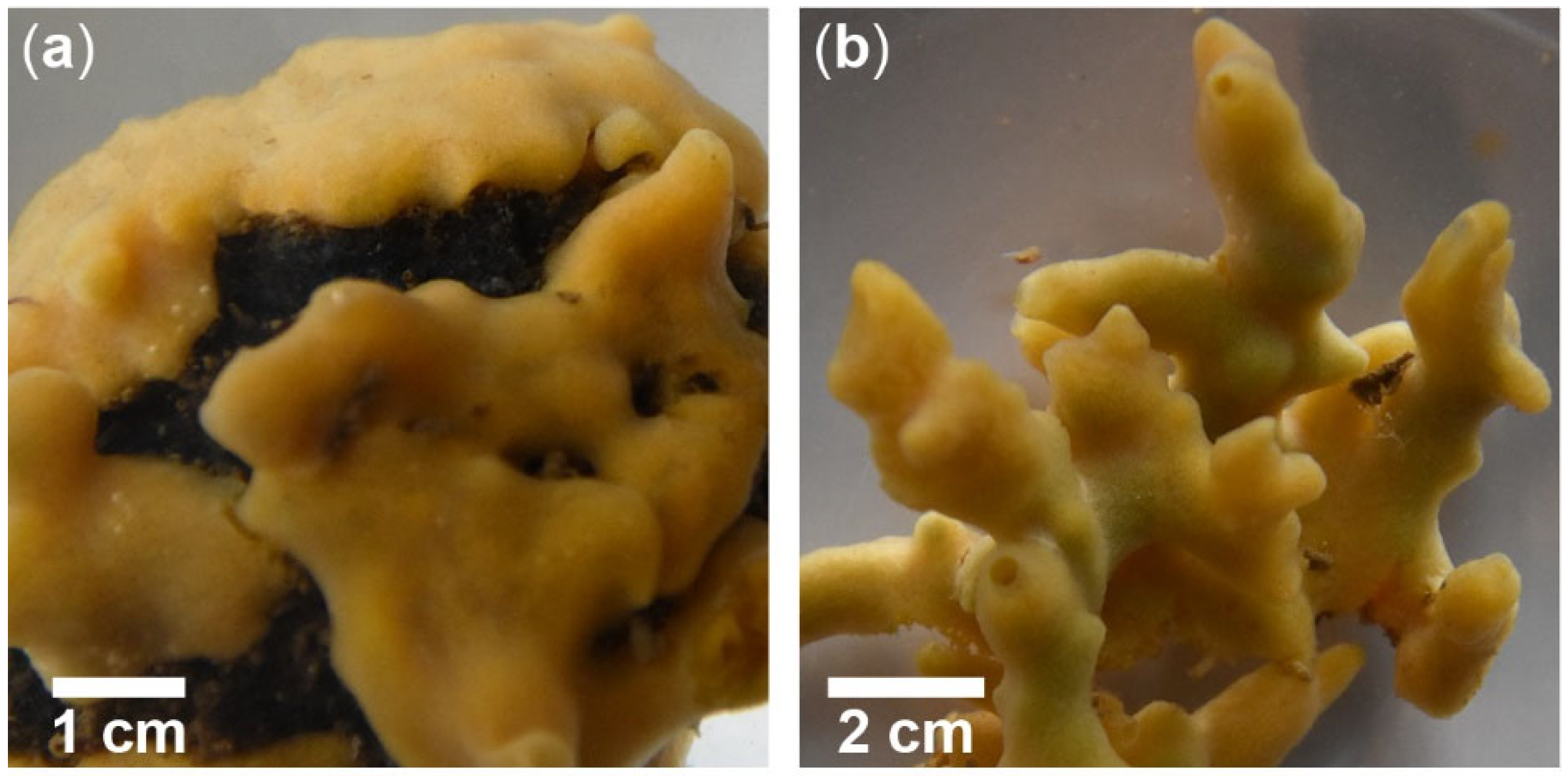
2. Morphology, Taxonomy, and Distribution
3. The Holobiont Halichondria
4. Life History
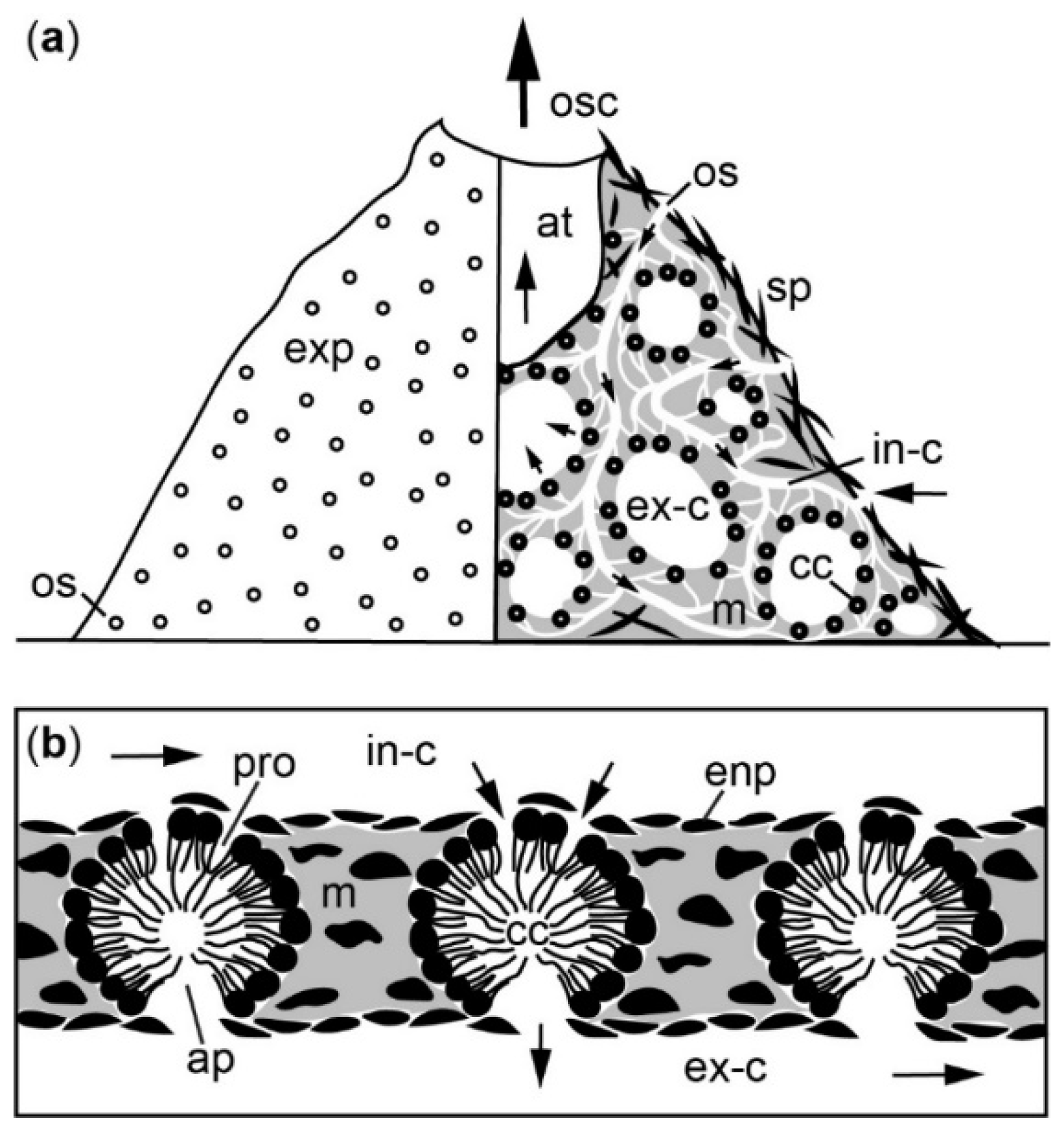
5. Hydrodynamics
6. Coordinated Behavior
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A



References
- Fleming, J. A History of British Animals: Exhibiting the Descriptive Characters and Systematical Arrangement of the Genera and Species of Quadrupeds, Birds, Reptiles, Fishes, Mollusca, and Radiata of the United Kingdom, Including the Indigenous, Extirpated, and Extinct Kinds, Together with Periodical and Occasional Visitors; Bell & Bradfute: London, UK, 1828; pp. 506–524. [Google Scholar]
- Erpenbeck, D.; Van Soest, R.W. Family Halichondriidae Gray, 1867. In Systema Porifera, 1st ed.; Hooper, J.N.A., Van Soest, R.W.M., Willenz, P., Eds.; Springer: Boston, MA, USA, 2002; pp. 787–815. [Google Scholar]
- Pallas, P.S. Elenchus Zoophytorum Sistens Generum Adumbrationes Generaliores et Specierum Cognitarum Succintas Descriptiones, Cum Selectis Auctorum Synonymis; Fransiscum Varrentrapp: The Hague, The Netherlands, 1766; p. 451. [Google Scholar]
- Burton, M. Additions to the sponge fauna at Plymouth. J. Mar. Biolog. Assoc. U. K. 1930, 16, 489–508. [Google Scholar] [CrossRef]
- Hiscock, K.; Jones, H. Halichondria (Halichondria) bowerbanki Bowerbank’s halichondria. In Marine Life Information Network: Biology and Sensitivity Key Information Reviews; Tyler-Walters, H., Hiscock, K., Eds.; Marine Biological Association of the United Kingdom: Plymouth, UK, 2007; pp. 1–14. [Google Scholar] [CrossRef]
- Hiscock, K. Halichondria (Halichondria) panicea breadcrumb sponge. In Marine Life Information Network: Biology and Sensitivity Key Information Reviews; Tyler-Walters, H., Hiscock, K., Eds.; Marine Biological Association of the United Kingdom: Plymouth, UK, 2008; pp. 1–16. [Google Scholar] [CrossRef]
- Forester, A.J. The association between the spongeHalichondriapanicea (Pallas) and scallop Chlamys varia (L.): A commensal-protective mutualism. J. Exp. Mar. Biol. Ecol. 1979, 36, 1–10. [Google Scholar] [CrossRef]
- Barthel, D. On the ecophysiology of the spongeHalichondriapanicea in Kiel Bight. 2. Biomass, production, energy budget and integration in environmental processes. Mar. Ecol. Prog. Ser. 1988, 43, 87–93. [Google Scholar] [CrossRef]
- Peattie, M.E.; Hoare, R. The sublittoral ecology of the Menai Strait: II. The spongeHalichondriapanicea (Pallas) and its associated fauna. Estuar. Coast. Shelf Sci. 1981, 13, 621–635. [Google Scholar] [CrossRef]
- Long, E.R. The associates of four species of marine sponges of Oregon and Washington. Pac. Sci. 1968, 22, 347–351. [Google Scholar]
- Vethaak, A.D.; Cronie, R.J.A.; Van Soest, R.W.M. Ecology and distribution of two sympatric, closely related sponge species,Halichondriapanicea (Pallas, 1766) and H. bowerbanki Burton, 1930 (Porifera, Demospongiae), with remarks on their speciation. Bijdr. Dierkd. 1982, 52, 82–102. [Google Scholar] [CrossRef]
- Knobloch, S.; Jóhannsson, R.; Marteinsson, V. Bacterial diversity in the marine spongeHalichondriapanicea from Icelandic waters and host-specificity of its dominant symbiont “Candidatus Halichondribacter symbioticus”. FEMS Microbiol. Ecol. 2019, 95, fiy220. [Google Scholar] [CrossRef]
- Sacristán-Soriano, O.; Winkler, M.; Erwin, P.; Weisz, J.; Harriott, O.; Heussler, G.; Hill, M. Ontogeny of symbiont community structure in two carotenoid-rich, viviparous marine sponges: Comparison of microbiomes and analysis of culturable pigmented heterotrophic bacteria. Environ. Microbiol. Rep. 2019, 11, 249–261. [Google Scholar] [CrossRef]
- Tanaka, K. Growth dynamics and mortality of the intertidal encrusting spongeHalichondriaokadai (Demospongiae, Halichondrida). Mar. Biol. 2002, 140, 383–389. [Google Scholar]
- Fell, P.E.; Jacob, W.F. Reproduction and development ofHalichondriasp. in the Mystic Estuary, Connecticut. Biol. Bull. 1979, 156, 62–75. [Google Scholar] [CrossRef]
- Wapstra, M.; Van Soest, R.W.M. Sexual reproduction, larval morphology and behaviour in demosponges from the southwest of the Netherlands. In Taxonomy of Porifera, 1st ed.; Vacelet, J., Boury-Esnault, N., Eds.; Springer: Berlin/Heidelberg, Germany, 1987; pp. 281–307. [Google Scholar]
- Witte, U.; Barthel, D.; Tendal, O. The reproductive cycle of the spongeHalichondriapanicea Pallas (1766) and its relationship to temperature and salinity. J. Exp. Mar. Biol. Ecol. 1994, 183, 41–52. [Google Scholar] [CrossRef]
- Gerasimova, E.I.; Ereskovsky, A.V. Reproduction of two species ofHalichondria(Demospongiae: Halichondriidae) in the White Sea. In Porifera Research—Biodiversity, Innovation and Sustainability. Série Livros, 1st ed.; Custódio, M.R., Ed.; Museu Nacional: Rio de Janeiro, Brazil, 2007; Volume 28, pp. 327–333. [Google Scholar]
- Riisgård, H.U.; Kumala, L.; Charitonidou, K. Using the F/R-ratio for an evaluation of the ability of the demospongeHalichondriapanicea to nourish solely on phytoplankton versus free-living bacteria in the sea. Mar. Biol. Res. 2016, 12, 907–916. [Google Scholar] [CrossRef]
- Thomassen, S.; Riisgård, H.U. Growth and energetics of the spongeHalichondriapanicea. Mar. Ecol. Prog. Ser. 1995, 128, 239–246. [Google Scholar] [CrossRef]
- Vogel, S. Current-induced flow through the sponge, Halichondria. Biol. Bull. 1974, 147, 443–456. [Google Scholar] [CrossRef]
- Kealy, R.A.; Busk, T.; Goldstein, J.; Larsen, P.S.; Riisgård, H.U. Hydrodynamic characteristics of aquiferous modules in the demospongeHalichondriapanicea. Mar. Biol. Res. 2019, 15, 531–540. [Google Scholar] [CrossRef]
- Asadzadeh, S.S.; Larsen, P.S.; Riisgård, H.U.; Walther, J.H. Hydrodynamics of the leucon sponge pump. J. R. Soc. Interface 2019, 16, 20180630. [Google Scholar] [CrossRef]
- Woollacott, R.M. Structure and swimming behavior of the larva ofHalichondriamelanadocia (Porifera: Demospongiae). J. Morphol. 1990, 205, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Burton, M. Observations on littoral sponges, including the supposed swarming of larvae, movement and coalescence in mature individuals, longevity and death. Proc. Zool. Soc. Lond. 1949, 118, 893–915. [Google Scholar] [CrossRef]
- Kumala, L.; Riisgård, H.U.; Canfield, D.E. Osculum dynamics and filtration activity studied in small single-osculum explants of the demospongeHalichondriapanicea. Mar. Ecol. Prog. Ser. 2017, 572, 117–128. [Google Scholar] [CrossRef]
- Goldstein, J.; Riisgård, H.U.; Larsen, P.S. Exhalant jet speed of single-osculum explants of the demospongeHalichondriapanicea and basic properties of the sponge-pump. J. Exp. Mar. Biol. Ecol. 2019, 511, 82–90. [Google Scholar] [CrossRef]
- Goldstein, J.; Bisbo, N.; Funch, P.; Riisgård, H.U. Contraction-expansion and the effects on the aquiferous system in the demospongeHalichondriapanicea. Front. Mar. Sci. 2020, 7, 113. [Google Scholar] [CrossRef]
- Abe, T.; Sahin, F.P.; Akiyama, K.; Naito, T.; Kishigami, M.; Miyamoto, K.; Sakakibara, Y.; Uemura, D. Construction of a metagenomic library for the marine spongeHalichondriaokadai. Biosc. Biotechnol. Biochem. 2012, 76, 633–639. [Google Scholar] [CrossRef]
- Laubenfels, M.W. A discussion of the sponge fauna of the Dry Tortugas in particular and the West Indies in general, with material for a revision of the families and orders of the Porifera. Publ. Carnegie Instit. Wash. 1936, 467, 1–225. [Google Scholar]
- Bergquist, P.R. A collection of Porifera from Northern New Zealand, with descriptions of seventeen new species. Pac. Sci. 1961, 15, 33–48. [Google Scholar]
- Lamarck, J.B. Sur les polypiers empâtés. Ann. Mus. Natl. Hist. Nat. 1814, 20, 294–312. [Google Scholar]
- Van Soest, R.W.; Boury-Esnault, N.; Hooper, J.N.; Rützler, K.; de Voogd, N.J.; de Glasby, B.A.; Hajdu, E.; Pisera, A.B.; Manconi, R.; Schoenberg, C.; et al. Halichondria Fleming, 1828. World Porifera Database. World Register of Marine Species 2020. Available online: https://www.marinespecies.org/aphia.php?p=taxdetails&id=131807 (accessed on 6 July 2022).
- Kadota, J. Observations of two new species of the genus Reniera of monaxonid sponges. Zool. Mag. 1922, 34, 700–711. [Google Scholar]
- Erpenbeck, D.; Hall, K.; Alvarez, B.; Büttner, G.; Sacher, K.; Schätzle, S.; Schuster, A.; Vargas, S.; Hooper, J.N.A.; Wörheide, G. The phylogeny of halichondrid demosponges: Past and present re-visited with DNA-barcoding data. Org. Divers. Evol. 2012, 12, 57–70. [Google Scholar] [CrossRef]
- Tanita, S.; Hoshino, T. The Demospongiae of Sagami Bay; Biological Laboratory, Imperial Household: Tokyo, Japan, 1989; p. 197.
- Tanita, S. Two sponges obtained by the Training Ship ‘Oshoro Maru’ in the Eastern Behring Sea. Bull. Fish. Sci. Hokkaido Univ. 1961, 11, 183–187. [Google Scholar]
- Keller, C. Die Spongienfauna des Rothen Meeres (II. Hälfte). Z. Wiss. Zool. 1891, 52, 294–368. [Google Scholar]
- Bergquist, P.R. Additions to the sponge fauna of the Hawaiian Islands. Micronesica 1967, 3, 159–174. [Google Scholar]
- Olesen, T.M.E.; Weeks, J.M. Accumulation of Cd by the marine spongeHalichondriapanicea Pallas: Effects upon filtration rate and its relevance for biomonitoring. Bull. Environ. Contam. Toxicol. 1994, 52, 722–728. [Google Scholar] [CrossRef] [PubMed]
- Musser, J.M.; Schippers, K.J.; Nickel, M.; Mizzon, G.; Kohn, A.B.; Pape, C.; Ronchi, P.; Papadopoulos, N.; Tarashansky, A.J.; Hammel, J.U.; et al. Profiling cellular diversity in sponges informs animal cell type and nervous system evolution. Science 2021, 374, 717–723. [Google Scholar] [CrossRef]
- Kang, D.W.; Sim, C.J. Two new sponges of the genusHalichondria(Halichondrida: Halichondriidae) from Korea. Anim. Cells Syst. 2008, 12, 65–68. [Google Scholar] [CrossRef]
- Barthel, D. Influence of different current regimes on the growth form ofHalichondriapanicea Pallas. In Fossil and Recent Sponges, 1st ed.; Reitner, J., Keupp, H., Eds.; Springer: Berlin/Heidelberg, Germany, 1991; pp. 387–394. [Google Scholar]
- Bell, J.J.; Barnes, D.K. The influences of bathymetry and flow regime upon the morphology of sublittoral sponge communities. J. Mar. Biolog. Assoc. U. K. 2000, 80, 707–718. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, Y.; Huang, D. The complete mitochondrial genome of spongeHalichondria(Halichondria) sp. (Demospongiae, Suberitida, Halichondriidae). Mitochondrial DNA B Resour. 2016, 1, 512–514. [Google Scholar] [CrossRef][Green Version]
- Kim, H.; Kim, H.J.; Jung, Y.H.; Yu, C.; An, Y.R.; Han, D.; Kang, D.W. The complete mitochondrial genome of spongeHalichondriaokadai (Demospongiae, Suberitida, Halichondriidae) from Korea water. Mitochondrial DNA B Resour. 2017, 2, 873–874. [Google Scholar] [CrossRef]
- Kim, H.; Kang, D.W.; Yu, C.; Jung, Y.H.; Yoon, M.; Kim, H.J. The complete mitochondrial genome of spongeHalichondriasp. (Demospongiae, Suberitida, Halichondriidae) from Dok-do. Mitochondrial DNA B Resour. 2019, 4, 1729–1730. [Google Scholar] [CrossRef]
- Erpenbeck, D.; Breeuwer, J.; van der Velde, H.; van Soest, R. Unravelling host and symbiont phylogenies of halichondrid sponges (Demospongiae, Porifera) using a mitochondrial marker. Mar. Biol. 2002, 141, 377–386. [Google Scholar]
- Erpenbeck, D.; Duran, S.; Rützler, K.; Paul, V.J.; Hooper, J.N.; Wörheide, G. Towards a DNA taxonomy of Caribbean demosponges: A gene tree reconstructed from partial mitochondrial CO1 gene sequences supports previous rDNA phylogenies and provides a new perspective on the systematics of Demospongiae. J. Mar. Biolog. Assoc. U. K. 2007, 87, 1563–1570. [Google Scholar] [CrossRef]
- Morrow, C.C.; Picton, B.E.; Erpenbeck, D.; Boury-Esnault, N.; Maggs, C.A.; Allcock, A.L. Congruence between nuclear and mitochondrial genes in Demospongiae: A new hypothesis for relationships within the G4 clade (Porifera: Demospongiae). Mol. Phylogenetics Evol. 2012, 62, 174–190. [Google Scholar] [CrossRef]
- Alvarez, B.; Crisp, M.D.; Driver, F.; Hooper, J.N.; Van Soest, R.W. Phylogenetic relationships of the family Axinellidae (Porifera: Demospongiae) using morphological and molecular data. Zool. Scr. 2000, 29, 169–198. [Google Scholar] [CrossRef]
- Castellanos, L.; Zea, S.; Osorno, O.; Duque, C. Phylogenetic analysis of the order Halichondrida (Porifera, Demospongiae), using 3β-hydroxysterols as chemical characters. Biochem. Syst. Ecol. 2003, 31, 1163–1183. [Google Scholar] [CrossRef]
- Erpenbeck, D. A molecular comparison of Alaskan and North East AtlanticHalichondriapanicea (Pallas 1766) (Porifera: Demospongiae) populations. Boll. Mus. Ist. Biol. Univ. Genova 2004, 68, 319–325. [Google Scholar]
- Erpenbeck, D.; Breeuwer, J.A.; Van Soest, R.W. Identification, characterization and phylogenetic signal of an elongation factor-1 alpha fragment in demosponges (Metazoa, Porifera, Demospongiae). Zool. Scr. 2005, 34, 437–445. [Google Scholar] [CrossRef]
- Hooper, J.N.A.; van Soest, R.W.M. Systema Porifera. A Guide to the Classification of Sponges; Kluwer Academic/Plenum Publishers: New York, NY, USA, 2002; Volume 1, pp. 1–1101. [Google Scholar]
- Alvarez, B.; Hooper, J.N. Taxonomic revision of the order Halichondrida (Porifera: Demospongiae) of northern Australia. Family Halichondriidae. Beagle Rec. Mus. Art Galleries North. Territ. 2011, 27, 55–84. [Google Scholar] [CrossRef]
- Fell, P.E.; Lewandrowski, K.B. Population dynamics of the estuarine sponge,Halichondriasp., within a New England eelgrass community. J. Exp. Mar. Biol. Ecol. 1981, 55, 49–63. [Google Scholar] [CrossRef]
- Picton, B.E.; Goodwin, C.E. Sponge biodiversity of Rathlin Island, Northern Ireland. J. Mar. Biolog. Assoc. U. K. 2007, 87, 1441–1458. [Google Scholar] [CrossRef]
- Carvalho, M.D.S.; Hajdu, E. Comments on brazilianHalichondriaFleming (Halichondriidae, Halichondrida, Demospongiae), with the description of four new species from the São Sebastião Channel and its environs (Tropical Southwestern Atlantic). Rev. Bras. Zool. 2001, 18, 161–180. [Google Scholar] [CrossRef]
- Barthel, D. Population dynamics of the spongeHalichondriapanicea (Pallas) in Kiel Bight. In Marine Eutrophication and Population Dynamics: 25th European Marine Biology Symposium; Colombo, G., Ed.; Olsen & Olsen: Fredensborg, Denmark, 1992; pp. 203–209. [Google Scholar]
- Khalaman, V.V.; Komendantov, A.Y. Experimental study of the ability of the spongeHalichondriapanicea (Porifera: Demospongiae) to compete for a substrate in shallow-water fouling communities of the White Sea. Biol. Bull. Russ. Acad. Sci. 2016, 43, 69–74. [Google Scholar] [CrossRef]
- Gaino, E.; Lepore, E.; Rebora, M.; Mercurio, M.; Sciscioli, M. Some steps of spermatogenesis inHalichondriasemitubulosa (Demospongiae, Halichondriidae). Ital. J. Zool. 2007, 74, 117–122. [Google Scholar] [CrossRef]
- Knowlton, A.L.; Highsmith, R.C. Convergence in the time-space continuum: A predator-prey interaction. Mar. Ecol. Prog. Ser. 2000, 197, 285–291. [Google Scholar] [CrossRef]
- Wulff, J. Ecological interactions and the distribution, abundance, and diversity of sponges. Adv. Mar. Biol. 2012, 61, 273–344. [Google Scholar]
- Hoshino, S. Systematic status ofHalichondriajaponica (Kadota) (Demospongiae, Halichondrida) from Japan. Boll. Mus. Ist. Biol. Univ. Genova 2004, 68, 373–379. [Google Scholar]
- Jeon, Y.J.; Sim, C.J. A new record of genusHalichondria(Demospongiae: Halichondrida: Halichondriidae) from Korea. Anim. Syst. Evol. Diversity 2009, 25, 137–139. [Google Scholar] [CrossRef]
- Li, Z.; Hu, Y.; Liu, Y.; Huang, Y.; He, L.; Miao, X. 16S rDNA clone library-based bacterial phylogenetic diversity associated with three South China Sea sponges. World J. Microbiol. Biotechnol. 2007, 23, 1265–1272. [Google Scholar] [CrossRef]
- Hechtel, G.J. A systematic study of the Demospongiae of Port Royal, Jamaica. Bull. Peabody Mus. Nat. Hist. 1965, 20, 1–103. [Google Scholar]
- Santos, G.G.; Nascimento, E.; Pinheiro, U. Halichondriidae Gray, 1867 from the Northeastern Brazil with description of a new species. Zootaxa 2018, 4379, 556–566. [Google Scholar] [CrossRef]
- Kim, H.S.; Parkc, B.J.; Sim, C.J. Marine sponges in South Korea (I). Korean J. Syst. Zool. 1986, 11, 37–47. [Google Scholar]
- Barthel, D. On the ecophysiology of the spongeHalichondriapanicea in Kiel Bight. I. Substrate specificity, growth and reproduction. Mar. Ecol. Prog. Ser. 1986, 32, 291–298. [Google Scholar] [CrossRef]
- Kobayashi, J.; Ishibashi, M. Bioactive metabolites of symbiotic marine microorganisms. Chem. Rev. 1993, 93, 1753–1769. [Google Scholar] [CrossRef]
- Lee, Y.K.; Lee, J.H.; Lee, H.K. Microbial symbiosis in marine sponges. J. Microbiol. 2001, 39, 254–264. [Google Scholar]
- Hummel, H.; Sepers, A.B.; De Wolf, L.; Melissen, F.W. Bacterial growth on the marine spongeHalichondriapanicea induced by reduced waterflow rate. Mar. Ecol. Prog. Ser. 1988, 42, 195–198. [Google Scholar] [CrossRef]
- Hentschel, U.; Hopke, J.; Horn, M.; Friedrich, A.B.; Wagner, M.; Hacker, J.; Moore, B.S. Molecular evidence for a uniform microbial community in sponges from different oceans. Appl. Environ. Microbiol. 2002, 68, 4431–4440. [Google Scholar] [CrossRef]
- Rusanova, A.; Fedorchuk, V.; Toshchakov, S.; Dubiley, S.; Sutormin, D. An interplay between viruses and bacteria associated with the White Sea sponges revealed by metagenomics. Life 2021, 12, 25. [Google Scholar] [CrossRef]
- Jeong, J.B.; Kim, K.H.; Park, J.S. Sponge-specific unknown bacterial groups detected in marine sponges collected from Korea through barcoded pyrosequencing. J. Microbiol. Biotechnol. 2015, 25, 1–10. [Google Scholar] [CrossRef]
- Schmidt, O. Grundzüge einer Spongien-Fauna des atlantischen Gebietes; Wilhelm Engelmann: Leipzig, Germany, 1870; pp. 1–88. [Google Scholar]
- Wichels, A.; Würtz, S.; Döpke, H.; Schütt, C.; Gerdts, G. Bacterial diversity in the breadcrumb spongeHalichondriapanicea (Pallas). FEMS Microbiol. Ecol. 2006, 56, 102–118. [Google Scholar] [CrossRef]
- Steinert, G.; Rohde, S.; Janussen, D.; Blaurock, C.; Schupp, P.J. Host-specific assembly of sponge-associated prokaryotes at high taxonomic ranks. Sci. Rep. 2017, 7, 2542. [Google Scholar] [CrossRef]
- Naim, M.A.; Morillo, J.A.; Sørensen, S.J.; Waleed, A.A.S.; Smidt, H.; Sipkema, D. Host-specific microbial communities in three sympatric North Sea sponges. FEMS Microbiol. Ecol. 2014, 90, 390–403. [Google Scholar] [CrossRef]
- Strehlow, B.W.; Schuster, A.; Francis, W.R.; Canfield, D.E. Metagenomic data forHalichondriapanicea from Illumina and nanopore sequencing and preliminary genome assemblies for the sponge and two microbial symbionts. BMC Res. Notes 2022, 15, 135. [Google Scholar] [CrossRef]
- Sun, J.F.; Wu, Y.; Yang, B.; Liuc, Y. Chemical constituents of marine sponge Halichondria sp. from south China sea. Chem. Nat. Compd. 2015, 51, 975–977. [Google Scholar] [CrossRef]
- Hirata, Y.; Uemura, D. Halichondrins-antitumor polyether macrolides from a marine sponge. Pure Appl. Chem. 1986, 58, 701–710. [Google Scholar] [CrossRef]
- Tachibana, K.; Scheuer, P.J.; Tsukitani, Y.; Kikuchi, H.; Van Engen, D.; Clardy, J.; Gopichand, Y.; Schmitz, F.J. Okadaic acid, a cytotoxic polyether from two marine sponges of the genus Halichondria. J. Am. Chem. Soc. 1981, 103, 2469–2471. [Google Scholar] [CrossRef]
- Amagata, T.; Tanaka, M.; Yamada, T.; Minoura, K.; Numata, A. Gymnastatins and dankastatins, growth inhibitory metabolites of a Gymnascella species from aHalichondriasponge. J. Nat. Prod. 2008, 71, 340–345. [Google Scholar] [CrossRef]
- Prego-Faraldo, M.V.; Valdiglesias, V.; Méndez, J.; Eirín-López, J.M. Okadaic acid meet and greet: An insight into detection methods, response strategies and genotoxic effects in marine invertebrates. Mar. Drugs 2013, 11, 2829–2845. [Google Scholar] [CrossRef]
- Fu, L.L.; Zhao, X.Y.; Ji, L.D.; Xu, J. Okadaic acid (OA): Toxicity, detection and detoxification. Toxicon 2019, 160, 1–7. [Google Scholar] [CrossRef]
- Corriere, M.; Soliño, L.; Costa, P.R. Effects of the marine biotoxins okadaic acid and dinophysistoxins on fish. J. Mar. Sci. Eng. 2021, 9, 293. [Google Scholar] [CrossRef]
- Khalaman, V.V.; Sharov, A.N.; Kholodkevich, S.V.; Komendantov, A.Y.; Kuznetsova, T.V. Influence of the White Sea spongeHalichondriapanicea (Pallas, 1766) on physiological state of the blue mussel Mytilus edulis (Linnaeus, 1758), as evaluated by heart rate characteristics. J. Evol. Biochem. Physiol. 2017, 53, 225–232. [Google Scholar] [CrossRef]
- Pita, L.; Fraune, S.; Hentschel, U. Emerging sponge models of animal-microbe symbioses. Front. Microbiol. 2016, 7, 2102. [Google Scholar] [CrossRef]
- Lemoine, N.; Buell, N.; Hill, A.; Hill, M. Assessing the utility of sponge microbial symbiont communities as models to study global climate change: A case study withHalichondriabowerbanki. In Porifera Research: Biodiversity, Innovation and Sustainability; Série Livros, 1st ed.; Custódio, M.R., Ed.; Museu Nacional: Rio de Janeiro, Brazil, 2007; Volume 28, pp. 419–425. [Google Scholar]
- Frøhlich, H.; Barthel, D. Silica uptake of the marine spongeHalichondriapanicea in Kiel Bight. Mar. Biol. 1997, 128, 115–125. [Google Scholar] [CrossRef]
- Barthel, D.; Detmer, A. The spermatogenesis ofHalichondriapanicea (Porifera, Demospongiae). Zoomorphology 1991, 110, 9–15. [Google Scholar] [CrossRef]
- Sokolova, A.M.; Pozdnyakov, I.R.; Ereskovsky, A.V.; Karpov, S.A. Kinetid structure in larval and adult stages of the demosponges Haliclona aquaeductus (Haplosclerida) andHalichondriapanicea (Suberitida). Zoomorphology 2019, 138, 171–184. [Google Scholar] [CrossRef]
- Amano, S. Larval release in response to a light signal by the intertidal spongeHalichondriapanicea. Biol. Bull. 1986, 171, 371–378. [Google Scholar] [CrossRef]
- Lüskow, F.; Riisgård, H.U.; Solovyeva, V.; Brewer, J.R. Seasonal changes in bacteria and phytoplankton biomass control the condition index of the demospongeHalichondriapanicea in temperate Danish waters. Mar. Ecol. Prog. Ser. 2019, 608, 119–132. [Google Scholar] [CrossRef]
- Riisgård, H.U.; Thomassen, S.; Jakobsen, H.; Weeks, J.M.; Larsen, P.S. Suspension feeding in marine spongesHalichondriapanicea and Haliclona urceolus: Effects of temperature on filtration rate and energy cost of pumping. Mar. Ecol. Prog. Ser. 1993, 96, 177–188. [Google Scholar] [CrossRef]
- Koopmans, M.; Martens, D.; Wijffels, R.H. Growth efficiency and carbon balance for the sponge Haliclona oculata. Mar. Biotechnol. 2010, 12, 340–349. [Google Scholar] [CrossRef][Green Version]
- Riisgård, H.U.; Larsen, P.S. Actual and model-predicted growth of sponges—with a bioenergetic comparison to other filter-feeders. J. Mar. Sci. Eng. 2022, 10, 607. [Google Scholar] [CrossRef]
- Fell, P.E.; Parry, E.H.; Balsamo, A.M. The life histories of sponges in the Mystic and Thames estuaries (Connecticut), with emphasis on larval settlement and postlarval reproduction. J. Exp. Mar. Biol. Ecol. 1984, 78, 127–141. [Google Scholar] [CrossRef]
- Hartman, W.D. Natural history of the marine sponges of southern New England. Bull. Peabody Mus. Yale 1958, 12, 1–155. [Google Scholar]
- Knowlton, A.L.; Highsmith, R.C. Nudibranch-sponge feeding dynamics: Benefits of symbiont-containing sponge to Archidoris montereyensis (Cooper, 1862) and recovery of nudibranch feeding scars byHalichondriapanicea (Pallas, 1766). J. Exp. Mar. Biol. Ecol. 2005, 327, 36–46. [Google Scholar] [CrossRef]
- Wulff, J. Regeneration of sponges in ecological context: Is regeneration an integral part of life history and morphological strategies? Integr. Comp. Biol. 2010, 50, 494–505. [Google Scholar] [CrossRef]
- Alcolado, P.M. Nuevas especies de esponjas encontradas en Cuba. Poeyana 1984, 271, 1–22. [Google Scholar]
- Wulff, J.L. Parrotfish predation on cryptic sponges of Caribbean coral reefs. Mar. Biol. 1997, 129, 41–52. [Google Scholar] [CrossRef]
- Wulff, J.L. Sponge predators may determine differences in sponge fauna between two sets of mangrove cays, Belize barrier reef. Atoll Res. Bull. 2000, 477, 251–263. [Google Scholar] [CrossRef]
- De Goeij, J.M.; Van Oevelen, D.; Vermeij, M.J.; Osinga, R.; Middelburg, J.J.; De Goeij, A.F.; Admiraal, W. Surviving in a marine desert: The sponge loop retains resources within coral reefs. Science 2013, 342, 108–110. [Google Scholar] [CrossRef]
- Barthel, D.; Wolfrath, B. Tissue sloughing in the spongeHalichondriapanicea: A fouling organism prevents being fouled. Oecologia 1989, 78, 357–360. [Google Scholar] [CrossRef]
- Hansen, I.V.; Weeks, J.M.; Depledge, M.H. Accumulation of copper, zinc, cadmium and chromium by the marine spongeHalichondriapanicea Pallas and the implications for biomonitoring. Mar. Pollut. Bull. 1995, 31, 133–138. [Google Scholar] [CrossRef]
- Langenbruch, P.F.; Scalera-Liaci, L. Structure of Choanocyte Chambers in Haplosclerid Sponges; Smithsonian Institution Press: Washington, DC, USA; Woods Hole, MA, USA, 1985; pp. 245–251. [Google Scholar]
- Haeckel, E. XXVII. On the Calcispongiae, their position in the animal kingdom, and their relation to the theory of descendence. J. Nat. Hist. 1873, 11, 241–262. [Google Scholar]
- Reiswig, H.M. The aquiferous systems of three marine Demospongiae. J. Morphol. 1975, 145, 493–502. [Google Scholar] [CrossRef]
- Fry, W.G. The sponge as a population: A biometric approach. Symp. Zool. Soc. Lond. 1970, 25, 135–162. [Google Scholar]
- Fry, W.G. Taxonomy, the individual and the sponge. In Biology and Systematics of Colonial Organisms: Proceedings of an International Symposium Held at the University of Durham; Published for the Systematics Association; Academic Press: London, UK, 1979; Volume 11, pp. 39–80. [Google Scholar]
- Riisgård, H.U.; Larsen, P.S. Filtration rates and scaling in demosponges. J. Mar. Sci. Eng. 2022, 10, 643. [Google Scholar] [CrossRef]
- Larsen, P.S.; Riisgård, H.U. The sponge pump. J. Theor. Biol. 1994, 168, 53–63. [Google Scholar] [CrossRef]
- Weissenfels, N. The filtration apparatus for food collection in freshwater sponges (Porifera, Spongillidae). Zoomorphology 1992, 112, 51–55. [Google Scholar] [CrossRef]
- Mah, J.L.; Christensen-Dalsgaard, K.K.; Leys, S.P. Choanoflagellate and choanocyte collar-flagellar systems and the assumption of homology. Evol. Dev. 2014, 16, 25–37. [Google Scholar] [CrossRef]
- Suarez, P.A.; Leys, S.P. The sponge pump as a morphological character in the fossil record. Paleobiology 2022, 48, 446–461. [Google Scholar] [CrossRef]
- Evans, C.W. The ultrastructure of larvae from the marine spongeHalichondriamoorei Bergquist (Porifera, Demospongiae). Cah. Biol. Mar. 1997, 18, 427–433. [Google Scholar]
- Harris, A.K. Cell motility and the problem of anatomical homeostasis. J. Cell. Sci. 1987, 8, 121–140. [Google Scholar] [CrossRef]
- Gaino, E.; Burlando, B. Sponge cell motility: A model system for the study of morphogenetic processes. Ital. J. Zool. 1990, 57, 109–118. [Google Scholar] [CrossRef]
- Bond, C. Continuous cell movements rearrange anatomical structures in intact sponges. J. Exp. Zool. 1992, 263, 284–302. [Google Scholar] [CrossRef]
- Lavrov, A.I.; Kosevich, I.A. Sponge cell reaggregation: Cellular structure and morphogenetic potencies of multicellular aggregates. J. Exp. Zool. A. Ecol. Genet. Physiol. 2016, 325, 158–177. [Google Scholar] [CrossRef]
- Saito, Y. Self and nonself recognition in a marine sponge,Halichondriajaponica (Demospongiae). Zool. Sci. 2013, 30, 651–657. [Google Scholar] [CrossRef]
- Storr, J.F. Field observations of sponge reactions as related to their ecology. In Aspects of Sponge Biology; Harrison, F.W., Cowden, R.R., Eds.; Academic Press Inc.: New York, NY, USA, 1976; pp. 277–282. [Google Scholar]
- Reiswig, H.M. In situ pumping activities of tropical Demospongiae. Mar. Biol. 1971, 9, 38–50. [Google Scholar] [CrossRef]
- Nickel, M. Kinetics and rhythm of body contractions in the sponge Tethya wilhelma (Porifera: Demospongiae). J. Exp. Biol. 2004, 207, 4515–4524. [Google Scholar] [CrossRef]
- Nickel, M.; Scheer, C.; Hammel, J.U.; Herzen, J.; Beckmann, F. The contractile sponge epithelium sensu lato—Body contraction of the demosponge Tethya wilhelma is mediated by the pinacoderm. J. Exp. Biol. 2011, 214, 1692–1698. [Google Scholar] [CrossRef]
- Elliott, G.R.; Leys, S.P. Coordinated contractions effectively expel water from the aquiferous system of a freshwater sponge. J. Exp. Biol. 2007, 210, 3736–3748. [Google Scholar] [CrossRef]
- Kumala, L.; Canfield, D.E. Contraction dynamics and respiration of small single-osculum explants of the demospongeHalichondriapanicea. Front. Mar. Sci. 2018, 5, 410. [Google Scholar] [CrossRef]
- Kumala, L.; Larsen, M.; Glud, R.N.; Canfield, D.E. Spatial and temporal anoxia in single-osculumHalichondriapanicea demosponge explants studied with planar optodes. Mar. Biol. 2021, 168, 173. [Google Scholar] [CrossRef]
- Leys, S.P. Elements of a ‘nervous system’ in sponges. J. Exp. Biol. 2015, 218, 581–591. [Google Scholar] [CrossRef]
- Gerrodette, T.; Flechsig, A.O. Sediment-induced reduction in the pumping rate of the tropical sponge Verongia lacunosa. Mar. Biol. 1979, 55, 103–110. [Google Scholar] [CrossRef]
- Bell, J.J.; McGrath, E.; Biggerstaff, A.; Bates, T.; Bennett, H.; Marlow, J.; Shaffer, M. Sediment impacts on marine sponges. Mar. Pollut. Bull. 2015, 94, 5–13. [Google Scholar] [CrossRef]
- Nickel, M. Evolutionary emergence of synaptic nervous systems: What can we learn from the non-synaptic, nerveless Porifera? Invertebr. Biol. 2010, 129, 1–16. [Google Scholar] [CrossRef]
- Jones, W.C. Is there a nervous system in sponges? Biol. Rev. 1962, 37, 1–47. [Google Scholar] [CrossRef]
- Ludeman, D.A.; Farrar, N.; Riesgo, A.; Paps, J.; Leys, S.P. Evolutionary origins of sensation in metazoans: Functional evidence for a new sensory organ in sponges. BMC Evol. Biol. 2014, 14, 3. [Google Scholar] [CrossRef]
- Leys, S.P.; Nichols, S.A.; Adams, E.D. Epithelia and integration in sponges. Integr. Comp. Biol. 2009, 49, 167–177. [Google Scholar] [CrossRef]
- Bagby, R.M. The fine structure of myocytes in the sponges Microciona prolifera (Ellis and Solander) and Tedania ignis (Duchassaing and Michelotti). J. Morphol. 1966, 118, 167–181. [Google Scholar] [CrossRef]
- Hammel, J.U.; Nickel, M. A new flow-regulating cell type in the demosponge Tethya wilhelma—Functional cellular anatomy of a leuconoid canal system. PLoS ONE 2014, 9, e113153. [Google Scholar] [CrossRef]
- Kanzawa, N.; Takano-Ohmuro, H.; Maruyama, K. Isolation and characterization of sea sponge myosin. Zool. Sci. 1995, 12, 765–769. [Google Scholar] [CrossRef]
- De Ceccatty, M.P. Coordination in sponges. The foundations of integration. Am. Zool. 1974, 14, 895–903. [Google Scholar] [CrossRef]
- Perovic, S.; Krasko, A.; Prokic, I.; Müller, I.M.; Müller, W.E. Origin of neuronal-like receptors in Metazoa: Cloning of a metabotropic glutamate/GABA-like receptor from the marine sponge Geodia cydonium. Cell Tissue Res. 1999, 296, 395–404. [Google Scholar] [CrossRef]
- Lieberkühn, N. Über Bewegungserscheinungen der Zellen; Band IX, N.G., Ed.; Elwert’sche Universitätsbuchhandlung: Marburg/Leipzig, Germany, 1870; pp. 9–22. [Google Scholar]
- Galtsoff, P.S. The amoeboid movement of dissociated sponge cells. Biol. Bull. 1923, 45, 153–161. [Google Scholar] [CrossRef]
- Loewenstein, W.R. On the genesis of cellular communication. Devel. Biol. 1967, 15, 503–520. [Google Scholar] [CrossRef]
| Species | No. Articles | (%) | Web of Science Categories (%) |
|---|---|---|---|
| Halichondria panicea | 4040 (229) | 36.4 | Marine Freshwater Biology (43.5), Ecology (19.2), Oceanography (16.2) |
| Halichondria okadai | 2980 (96) | 26.8 | Organic Chemistry (39.6), Pharmacology Pharmacy (16.7), Biochemistry Molecular Biology (14.5) |
| Halichondria sp./spp. | 1390 (73) | 12.5 | Organic Chemistry (34.3), Medicinal Chemistry (20.6), Pharmacology Pharmacy (20.6) |
| Genus Halichondria | 723 (14) | 6.5 | Organic Chemistry (21.4), Pharmacology Pharmacy (21.4), Biochemistry Molecular Biology (14.3) |
| Halichondria bowerbanki | 447 (10) | 4.0 | Ecology (50.0), Marine Freshwater Biology (40.0), Zoology (30.0) |
| Halichondria japonica | 260 (20) | 2.3 | Biochemistry Molecular Biology (30.0), Organic Chemistry (20.0), Fisheries (15.0) |
| Halichondria cylindrata | 173 (10) | 1.6 | Organic Chemistry (70.0), Medicinal Chemistry (30.0), Biochemistry Molecular Biology (10.0) |
| Halichondria melanadocia | 169 (17) | 1.5 | Marine Freshwater Biology (52.9), Ecology (29.4), Anatomy Morphology (50.0) |
| Halichondria moorei | 108 (2) | 1.0 | Marine and Freshwater Biology (50.0), Multidisciplinary Sciences (50.0) |
| Halichondria sitiens | 89 (5) | 0.8 | Biodiversity Conservation (20.0), Biology (20.0), Ecology (20.0) |
| Halichondria oshoro | 82 (2) | 0.7 | Microbiology (100.0) |
| Halichondria magniconulosa | 67 (2) | 0.6 | Applied Chemistry (50.0), Medicinal Chemistry (50.0), Ecology (50.0) |
| Halichondria semitubulosa | 25 (1) | 0.2 | Zoology (100.0) |
| Halichondria cartilaginea | 19 (0) | 0.2 | - |
| Halichondria genitrix | 19 (0) | 0.2 | - |
| Halichondria albescens | 18 (0) | 0.2 | - |
| Halichondria lutea | 18 (3) | 0.2 | Biochemistry Molecular Biology (66.7), Ecology (66.7), Evolutionary Biology (66.7) |
| Halichondria coerulea | 14 (1) | 0.1 | Ecology (100.0), Marine Freshwater Biology (100.0), Oceanography (100.0) |
| Halichondria glabrata | 14 (2) | 0.1 | Anatomy and Morphology (50.0), Biology (50.0), Food Science Technology (50.0) |
| Halichondria diazae | 13 (0) | 0.1 | - |
| Halichondria cebimarensis | 12 (1) | 0.1 | Ecology (100.0), Marine Freshwater Biology (100.0) |
| Halichondria phakellioides | 12 (1) | 0.1 | Fisheries (100.0), Limnology (100.0), Marine Freshwater Biology (100.0) |
| Halichondria attenuata | 11 (2) | 0.1 | Marine Freshwater Biology (50.0), Zoology (50.0) |
| Halichondria contorta | 10 (1) | 0.1 | Zoology (100.0) |
| Halichondria topsenti | 10 (0) | 0.1 | - |
| Halichondria oblonga | 9 (0) | 0.1 | - |
| Halichondria aspera | 8 (0) | 0.1 | - |
| Halichondria cristata | 7 (0) | 0.1 | - |
| Halichondria agglomerans | 5 (0) | 0.0 | - |
| Halichondria flava | 5 (0) | 0.0 | - |
| Halichondria kelleri | 5 (0) | 0.0 | - |
| Halichondria migottea | 5 (0) | 0.0 | - |
| Halichondria osculum | 5 (1) | 0.0 | Medicinal Chemistry (100.0), Pharmacology Pharmacy (100.0) |
| Halichondria colossea | 4 (0) | 0.0 | - |
| Halichondria marianae | 4 (2) | 0.0 | Marine Freshwater Biology (50.0), Zoology (50.0) |
| Halichondria prostrata | 4 (0) | 0.0 | - |
| Halichondria tenebrica | 4 (0) | 0.0 | - |
| Halichondria capensis | 3 (0) | 0.0 | - |
| Halichondria convolvens | 3 (0) | 0.0 | - |
| Halichondria elenae | 3 (1) | 0.0 | Ecology (100.0), Marine Freshwater Biology (100.0) |
| Other species | 316 (36) | 2.1 | Cell biology (100.0), Zoology (100.0) |
| Total | 11,100 (532) | 100.0 | Marine and Freshwater Biology (27.3), Organic Chemistry (17.5), Ecology (12.8) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goldstein, J.; Funch, P. A Review on Genus Halichondria (Demospongiae, Porifera). J. Mar. Sci. Eng. 2022, 10, 1312. https://doi.org/10.3390/jmse10091312
Goldstein J, Funch P. A Review on Genus Halichondria (Demospongiae, Porifera). Journal of Marine Science and Engineering. 2022; 10(9):1312. https://doi.org/10.3390/jmse10091312
Chicago/Turabian StyleGoldstein, Josephine, and Peter Funch. 2022. "A Review on Genus Halichondria (Demospongiae, Porifera)" Journal of Marine Science and Engineering 10, no. 9: 1312. https://doi.org/10.3390/jmse10091312
APA StyleGoldstein, J., & Funch, P. (2022). A Review on Genus Halichondria (Demospongiae, Porifera). Journal of Marine Science and Engineering, 10(9), 1312. https://doi.org/10.3390/jmse10091312






