Pre-Caecal Disappearance of Starch and Volatile Fatty Acid (VFA) Content in Digesta of Caecum of Growing Pigs Fed with Ripe Hulled Shredded Acorns in Their Diet
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Diets
2.2. Sampling at Necropsy
2.3. Chemical Analyses
2.4. Calculations and Statistics
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Joffre, R.; Rambal, S.; Ratte, J.P. The dehesa system in southern Spain and Portugal as a natural ecosystem mimic. Agrofor. Syst. 1999, 45, 57–79. [Google Scholar] [CrossRef]
- Nieto, R.; Rivera, M.; Garca, M.A.; Aguilera, J.F. Amino acid availability and energy value of acorn in the Iberian pig. Livest. Sci. 2002, 77, 227–239. [Google Scholar] [CrossRef]
- Pinna, W.; Nieddu, G.; Moniello, G.; Cappai, M.G. Vegetable and animal food sorts found in the gastric content of sardinian wild boars (Sus scrofa meridionalis). J. Anim. Physiol. Anim. Nutr. 2007, 91, 252–255. [Google Scholar] [CrossRef] [PubMed]
- Cappai, M.G.; Wolf, P.; Liesner, V.G.; Kastner, A.; Nieddu, G.; Pinna, W.; Kamphues, J. Effect of whole acorns (Quercus pubescens) shred based diet on parotid gland in growing pigs in relation to tannins. Livest. Sci. 2010, 134, 183–186. [Google Scholar] [CrossRef]
- Lopez-Bote, C.; Fructuoso, G.; Mateos, G.G. El cerdo iberico. Sistemas de procion porcina y calidad de la carne. Cerdo Iber FEDNA 2000, 16, 77–111. [Google Scholar]
- Henkin, Z.; Gutman, M.; Aharon, H.; Perevolotsky, A.; Ungar, E.D.; Seligman, N.G. Suitability of Mediterranean oak woodland for beef herd husbandry. Agric. Ecosyst. Environ. 2005, 109, 155–161. [Google Scholar] [CrossRef]
- Chiofalo, B.; Liotta, L.; Zumbo, A.; Chiofalo, L. Seasonal variation of free fatty acids in plasma of Nero Siciliano pigs living in extensive conditions. Vet. Res. Commun. 2013, 27, 253–255. [Google Scholar] [CrossRef]
- Papanastasis, V.P.; Yiakoulaki, M.D.; Decandia, M.; Dini-Papanastasi, O. Integrating woody species into livestock feeding in the Mediterranean areas of Europe. Anim. Feed Sci. Technol. 2008, 140, 1–17. [Google Scholar] [CrossRef]
- Rey, A.I.; Daza, A.; Lopez-Carrasco, C.; Lòpez-Bote, C.J. Feeding Iberian pigs with acorns and grass in either free-range of confinement affects the carcass characteristics ad fatty acids and tocopherols accumulation in Longissimus dorsi muscle and backfat. Meat Sci. 2005, 73, 66–74. [Google Scholar] [CrossRef]
- Dasa, A.; Mateos, A.; Rey, A.I.; Ovejero, I.; Lòpez-Bote, C.J. Effect of duration of feeding under free-range condition on production results and carcass and fat quality in Iberian pigs. Meat Sci. 2007, 6, 411–461. [Google Scholar]
- Tejerina, D.; García-Torres, S.; Cabeza de Vaca, M.; Vázquez, F.M.; Cava, R. Acorns (Quercus rotundifolia Lam.) and grass as natural sources of antioxidants and fatty acids in the “montanera” feeding of Iberian pig: Intra- and inter-annual variations. Food Chem. 2011, 124, 997–1004. [Google Scholar] [CrossRef]
- Holman, R.M.; Robbins, W.W. A Textbook of General Botany; Wiley and Sons, Inc.: New York, NY, USA, 1944. [Google Scholar]
- Cappai, M.G.; Wolf, P.; Pinna, W.; Kamphues, J. Pigs use endogenous proline to cope with acorn (Quercus pubescens Willd.) combined diet high in hydrolysable tannins. Livest. Sci. 2013, 155, 316–322. [Google Scholar] [CrossRef]
- Cappai, M.G.; Wolf, P.; Pinna, W.; Kamphues, J. The bilateral parotidomegaly (hypertrophy) induced by acorn consumption in pigs is dependent on individual’s age but not on intake duration. Livest. Sci 2014, 167, 263–268. [Google Scholar] [CrossRef]
- Cappai, M.G.; Wolf, P.; Rust, P.; Pinna, W.; Kamphues, J. Digestibility coefficient of crude nutrients in raw hulled acorns (Quercus pubescens Willd.) fed to growing pigs. Anim. Feed Sci. Technol. 2014, 197, 148–154. [Google Scholar] [CrossRef]
- Cappai, M.G.; Alesso, G.A.; Nieddu, G.; Sanna, M.; Pinna, W. Electron microscopy and composition of raw acorn starch in relation to in vivo acorn starch digestibility. Food Funct. 2013, 4, 917–922. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Estevez, V.; Garcia Martinez, C.; Perea Munoz, J.M.; Gomez Castro, A.G. Measures and nutritional characteristics of Quercus acorns from the dehesa. Arch. Zootec. 2008, 57, 1–12. [Google Scholar]
- Rezar, V.; Salobir, J.; Levart, A.; Tomažin, U.; Škrlep, M.; Batorek Lukač, N.; Čandek-Potokar, M. Supplementing entire male pig diet with hydrolyzable tannins: Effect on carcass traits, meat quality and oxidative stability. Meat Sci. 2017, 133, 95–102. [Google Scholar] [CrossRef]
- Galassi, G.; Mason, F.; Rapetti, L.; Crovetto, G.M.; Spanghero, M. Digestibility and metabolic utilisation of diets containing chestnut tannins and their effects on growth and slaughter traits of heavy pigs. Ital. J. Anim. Sci. 2019, 18, 746–753. [Google Scholar] [CrossRef]
- Aldritt, I.; Whitham-Agut, B.; Sipin, M.; Studholme, J.; Trentacoste, A.; Tripp, J.A.; Cappai, M.G.; Ditchfield, P.; Deviése, T.; Hedges, R.E.M.; et al. Metabolomics reveals diet-derived polyphenols accumulate in physiological bone. Sci. Rep. 2019, 9, 8047. [Google Scholar] [CrossRef]
- Morales, J.; Pérez, J.F.; Martín-Orúe, S.M.; Fondevila, M.; Gasa, J. Large bowel fermentation of maize or sorghum acorn diets fed as different source of carbohydrates to Landrace and Iberian pigs. Br. J. Nutr. 2002, 88, 489–497. [Google Scholar] [CrossRef]
- Fondevila, M.; Morales, J.; Pérez, J.F.; Barrios-Urdaneta, A.; Baucells, M.D. Microbial caecal fermentation Iberic or Landrace pigs given acorn/sorghum or maize diets estimated in vitro by the gas production technique. Anim. Feed Sci. Technol. 2002, 102, 93–107. [Google Scholar] [CrossRef]
- Mößeler, A.; Vagt, S.; Beyerbach, M.; Kamphues, J. Starch origin and thermal processing affect starch digestion in a minipig model of pancreatic exocrine insufficiency. Gastroenterol. Res. Pract. 2015, 872872. [Google Scholar] [CrossRef] [PubMed]
- Directive 2010/63/EU of the European Parliament of and of the Council of 22 September 2010 on the Protection of Animals Used for Scientific Purposes. 2010. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2010:276:0033:0079:en:PDF (accessed on 26 June 2020).
- Willing, B.P.; Van Kassel, A.G. Enterocyte proliferation and apoptosis in the caudal small intestine is influenced by the composition of colonizing commensal bacteria in the neonatal gnotobiotic pig. J. Anim. Sci. 2007, 85, 3256–3266. [Google Scholar] [CrossRef]
- Naumann, C.; Bassler, R. Die Chemische Untersuchung von Futtermitteln (Chemical Analyses of Animal Feed); VDLUFA-Verlag: Darmstadt, Germany, 2012. [Google Scholar]
- Commission Directive 1999/79/EC of 27 July 1999 Amending the Third Commission Directive 72/199/EEC of 27 April 1972 Establishing Community Methods of Analysis for the Official Control of Feeding Stuffs. 1999. Available online: https://eur-lex.europa.eu/eli/dir/1999/79/oj (accessed on 26 June 2020).
- Folin, O.; Ciocalteau, V. On tyrosine and tryptophane determination in proteins. J. Biol. Chem. 1927, 73, 424–427. [Google Scholar]
- Waterman, P.G.; Mole, S. Analysis of plant metabolites. In Methods in Ecology Series; Waterman, P.G., Mole, S., Eds.; Blackwell Scientific Publications: Oxford, UK, 1994. [Google Scholar]
- Ramana, D.B.V.; Vijay Kumar, A.; Sudheer, D.; Maini, S. Improvement in nutrient utilization in Deccani rams fed with coarse cereal crop residue as basal diet by polyherbal supplements. J. Vet. Sci. 2014, 115, 338–343. [Google Scholar]
- Martin, L.J.M.; Dumon, H.J.W.; Champ, M.M.J. Production of short-chain fatty acids from resistant starch in a pig model. J. Sci. Food Agric. 1998, 77, 71–80. [Google Scholar] [CrossRef]
- Canibe, N.; Bach Knudsen, K.E. Degradation and physicochemical changes of barley and pea fibre along the gastrointestinal tract of pigs. J. Sci. Food Agric. 2001, 82, 27–39. [Google Scholar] [CrossRef]
- Bird, A.R.; Vuaran, M.; Brown, I.; Topping, D.L. Two high amylosemaize starches with different amounts of resistant starch vary in their effects on fermentation, tissue and digesta mass accretion, and bacterial populations in the large bowel of pigs. Br. J. Nutr. 2007, 97, 134–144. [Google Scholar] [CrossRef]
- Hedemann, M.S.; Bach Knudsen, K.E. Resistant starch for weaning pigs—effect on concentration of short chain fatty acids in digesta and intestinal morphology. Livest. Sci. 2007, 108, 175–177. [Google Scholar] [CrossRef]
- Cherbut, C.; Aubé, A.C.; Blottiére, H.M.; Galmiche, J.P. Effects of short chain fatty acids on gastrointestinal motility. Scan. J. Gastroenterol. Suppl. 1997, 222, 58–61. [Google Scholar] [CrossRef]
- Bravo, L.; Abia, R.; Eastwood, M.A.; Saura-Calixtol, F. Degradation of polyphenols (catechin and tannic acid) in rat intestinal tract. Effect on colonic fermentation and faecal output. Br. J. Nutr. 1994, 71, 933–946. [Google Scholar] [CrossRef] [PubMed]
| Ingredients (g/kg Diet, as Fed) | Control Diet | Acorn Combined Diet |
|---|---|---|
| Hulled shredded acorns | 0 | 700 |
| Wheat | 400 | 120 |
| Barley | 340 | 102 |
| Soybean meal | 200 | 60 |
| Soy oil | 11 | 3 |
| Mineral premix | 22 | 66 |
| Lysine/Methionine | 7 | |
| Analyzed chemical composition | ||
| Dry matter (g/kg, as fed) | 890 | 695 |
| Ash (g/kg DM) | 684 | 586 |
| Crude protein (g/kg DM) | 175 | 113 |
| Crude fibre (g/kg DM) | 45.0 | 112 |
| Crude fat (g/kg DM) | 22.8 | 45.9 |
| Starch (g/kg DM) | 435 | 447 |
| OM (g/kg DM) | 931 | 941 |
| TAE (g/kg DM) | n. d. | 36.1 |
| Diet | CD | ABD | ||
|---|---|---|---|---|
| Animals | 8 | 8 | SE | p-value |
| Parameters of digesta of caudal ileum | ||||
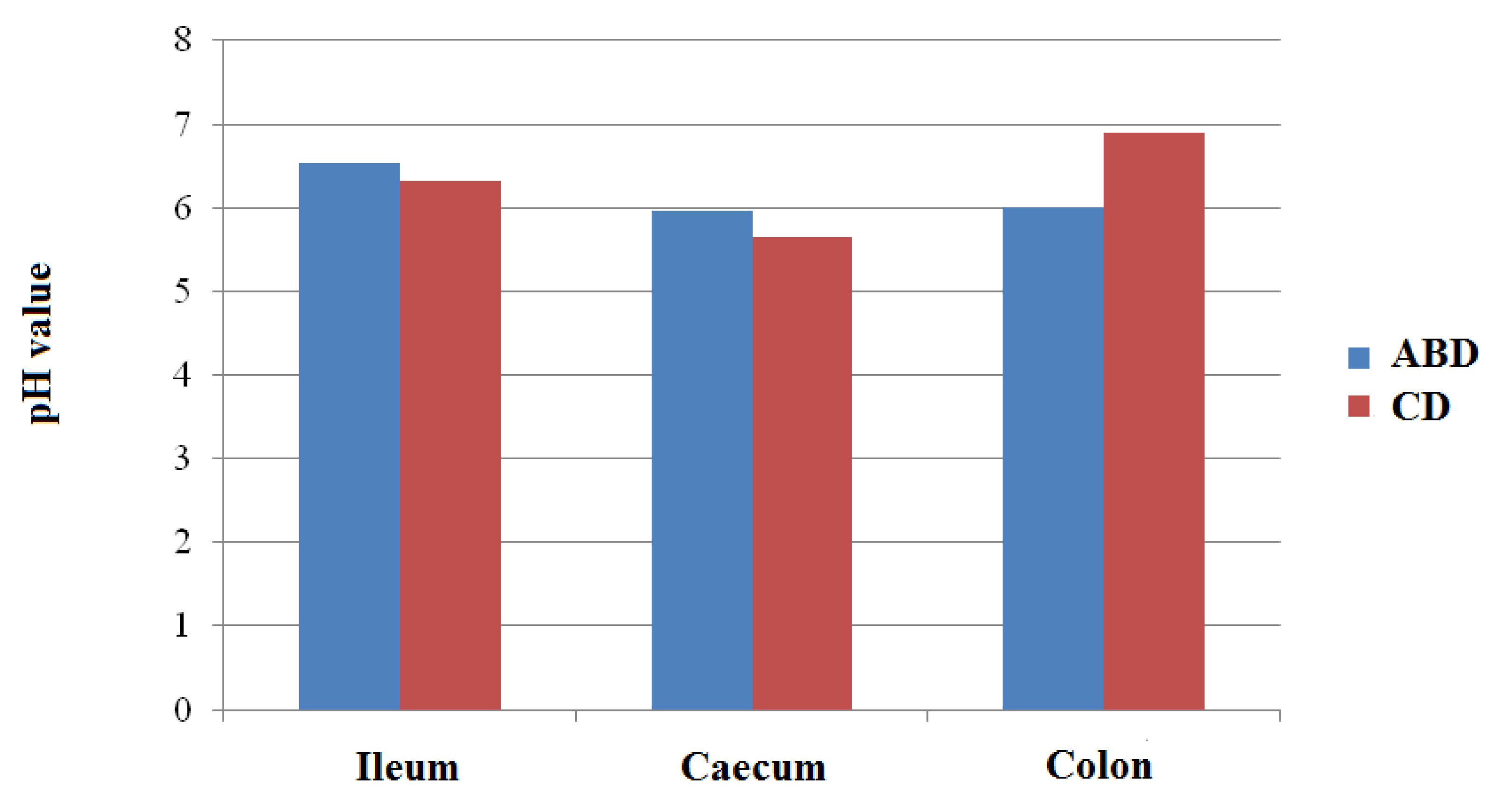
| pH | 6.33 | 6.54 | 0.51 | 0.638 |
| DM g/kg | 153 | 143 | 87.1 | 0.245 |
| Starch g/kg DM | 136 | 131 | 21.1 | 0.749 |
| Parameters of digesta of caecum | ||||
| pH | 5.65 | 5.97 | 0.13 | 0.134 |
| DM g/kg | 149.5 | 264 | 85 | 0.107 |
| Starch g/kg DM | 103 | 154 | 19.4 | 0.010 |
| VFA (g/kg OM) | 8.17 | 5.92 | 0.50 | 0.047 |
| acetic acid (g/molar weight) | 0.070 | 0.059 | 0.006 | 0.165 |
| propionic acid (g/molar weight) | 0.037 | 0.018 | 0.005 | 0.075 |
| n-butyric acid (g/molar weight) | 0.018 | 0.009 | 0.006 | 0.277 |
| n-valeric acid (g/molar weight) | 0.001 | 0.002 | 0.001 | 0.359 |
| Diet | CD | ABD | ||
|---|---|---|---|---|
| Animals | 8 | 8 | ||
| SE | p-value | |||
| Feed intake (g as fed/kg BW/8 h) | 46.1 | 36.9 | ||
| DM intake (g/kg BW/8 h) | 41.0 | 25.7 | 23.4 | 0.631 |
| pCD of Starch (%) | 99.7 | 99.5 | 20.1 | 0.365 |
| VFA | Acetic a. | Propionic a. | n-Butyrric a. | n-Valeric a. | |
|---|---|---|---|---|---|
| Acetic a. | 0.873 | ||||
| 0.127 | |||||
| Propionic a. | 0.953 | 0.687 | |||
| 0.047 | 0.313 | ||||
| n-butyrric a. | 0.792 | 0.406 | 0.919 | ||
| 0.208 | 0.594 | 0.081 | |||
| n-valeric a. | −0.379 | −0.366 | −0.395 | −0.101 | |
| 0.621 | 0.634 | 0.605 | 0.899 | ||
| pH | −0.977 | −0.843 | −0.924 | −0.822 | 0.173 |
| 0.023 | 0.157 | 0.076 | 0.178 | 0.827 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cappai, M.G.; Wolf, P.; Pinna, W.; Rust, P.; Kamphues, J. Pre-Caecal Disappearance of Starch and Volatile Fatty Acid (VFA) Content in Digesta of Caecum of Growing Pigs Fed with Ripe Hulled Shredded Acorns in Their Diet. Agriculture 2020, 10, 508. https://doi.org/10.3390/agriculture10110508
Cappai MG, Wolf P, Pinna W, Rust P, Kamphues J. Pre-Caecal Disappearance of Starch and Volatile Fatty Acid (VFA) Content in Digesta of Caecum of Growing Pigs Fed with Ripe Hulled Shredded Acorns in Their Diet. Agriculture. 2020; 10(11):508. https://doi.org/10.3390/agriculture10110508
Chicago/Turabian StyleCappai, Maria Grazia, Petra Wolf, Walter Pinna, Peter Rust, and Josef Kamphues. 2020. "Pre-Caecal Disappearance of Starch and Volatile Fatty Acid (VFA) Content in Digesta of Caecum of Growing Pigs Fed with Ripe Hulled Shredded Acorns in Their Diet" Agriculture 10, no. 11: 508. https://doi.org/10.3390/agriculture10110508
APA StyleCappai, M. G., Wolf, P., Pinna, W., Rust, P., & Kamphues, J. (2020). Pre-Caecal Disappearance of Starch and Volatile Fatty Acid (VFA) Content in Digesta of Caecum of Growing Pigs Fed with Ripe Hulled Shredded Acorns in Their Diet. Agriculture, 10(11), 508. https://doi.org/10.3390/agriculture10110508






