(Neuro) Peptides, Physical Activity, and Cognition
Abstract
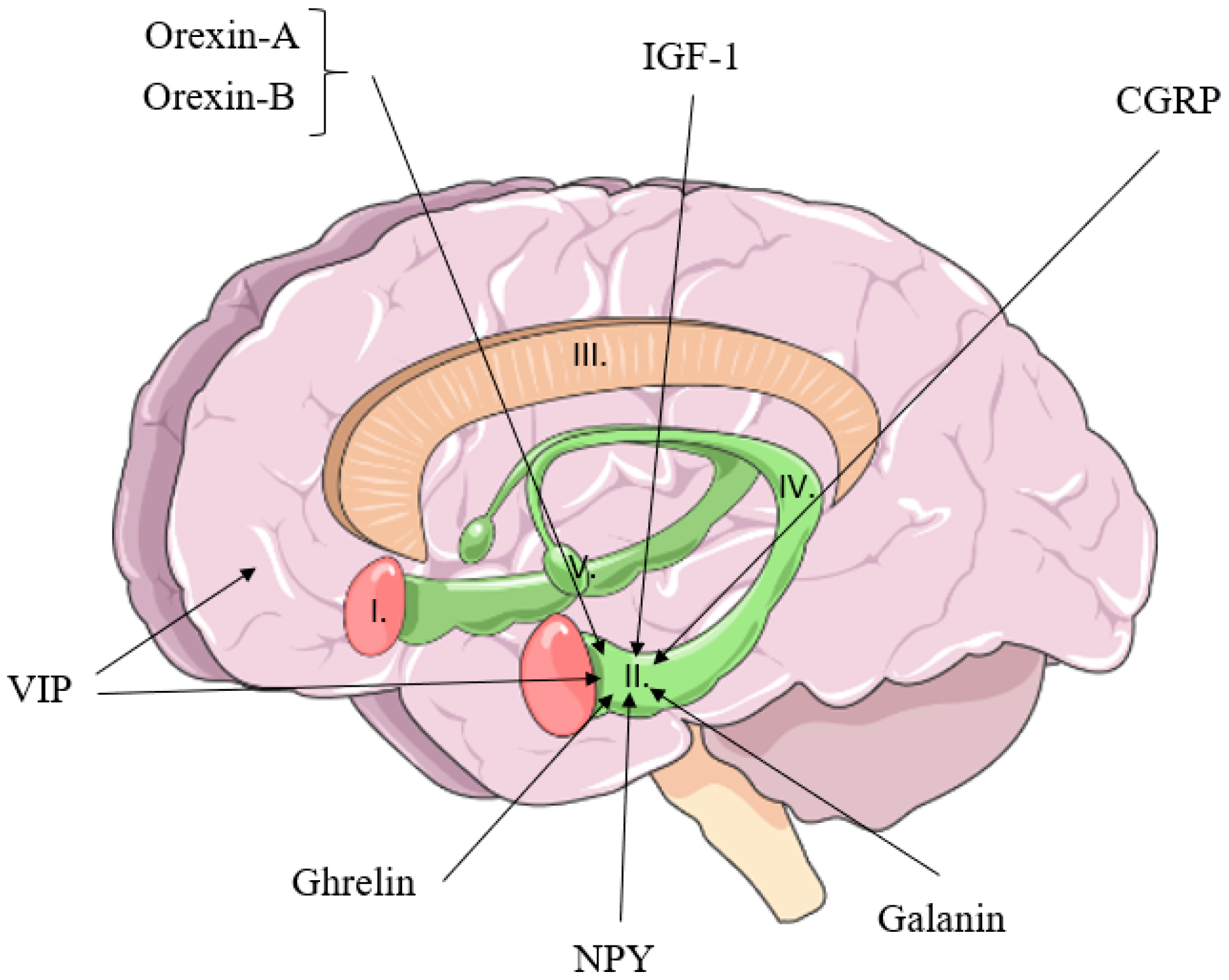
1. Introduction
2. Insulin Growth Factor 1 (IGF-1)
2.1. Centrally and Peripherally Released IGF-1
2.2. IGF-1 and Cognition
2.3. IGF-1 and Age
2.4. IGF-1 and Gender
2.5. IGF-1 and Fitness
2.6. IGF-1 and Physical Activity
2.7. Associations between Physical Activity, IGF-1, and Cognition
2.8. IGF-1 and Intervention Studies
2.9. IGF-1 and Prospective Trials
3. Orexins
4. Ghrelin
5. Neuropeptide Y (NPY)
6. Galanin
7. Vasoactive Intestinal Peptide (VIP)
8. Calcitonin Gene-Related Peptide (CGRP)
9. Other (Neuro-)Peptides
10. Conclusions and Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- Barch, D.M.; Burgess, G.C.; Harms, M.P.; Petersen, S.E.; Schlaggar, B.L.; Corbetta, M.; Glasser, M.F.; Curtiss, S.; Dixit, S.; Feldt, C.; et al. Function in the human connectome: Task-fMRI and individual differences in behavior. Neuroimage 2013, 80, 169–189. [Google Scholar] [CrossRef] [PubMed]
- Petersen, S.E.; Sporns, O. Brain Networks and Cognitive Architectures. Neuron 2015, 88, 207–219. [Google Scholar] [CrossRef] [PubMed]
- Bota, M.; Sporns, O.; Swanson, L.W. Architecture of the cerebral cortical association connectome underlying cognition. Proc. Natl. Acad. Sci. USA 2015, 112, E2093–E2101. [Google Scholar] [CrossRef] [PubMed]
- Owen, G.R.; Brenner, E.A. Mapping Molecular Memory: Navigating the Cellular Pathways of Learning. Cell. Mol. Neurobiol. 2012, 32, 919–941. [Google Scholar] [CrossRef] [PubMed]
- Ward, A.; Arrighi, H.M.; Michels, S.; Cedarbaum, J.M. Mild cognitive impairment: Disparity of incidence and prevalence estimates. Alzheimers Dement. 2012, 8, 14–21. [Google Scholar] [CrossRef]
- Fjell, A.M.; Walhovd, K.B. Structural Brain Changes in Aging: Courses, Causes and Cognitive Consequences. Rev. Neurosci. 2010, 21. [Google Scholar] [CrossRef]
- Blennow, K.; Zetterberg, H.; Fagan, A.M. Fluid Biomarkers in Alzheimer Disease. Cold Spring Harb. Perspect. Med. 2012, 2, a006221. [Google Scholar] [CrossRef]
- Alessi, C.; Anstey, K.; Ashby-Mitchell, K.; Barbui, C.; Comas-Herrera, A.; Dias, A.; Ferri, C.P.; Gouider, R.; Ishii, S.; Joanette, Y.; et al. Risk Reduction of Cognitive Decline and Dementia; WHO Guidelines; World Health Organization: Geneva, Switzerland, 2019; ISBN 978-92-4-155054-3. [Google Scholar]
- Pedersen, B.K.; Saltin, B. Exercise as medicine-evidence for prescribing exercise as therapy in 26 different chronic diseases. Scand. J. Med. Sci. Sports 2015, 25, 1–72. [Google Scholar] [CrossRef]
- Guthold, R.; Stevens, G.A.; Riley, L.M.; Bull, F.C. Worldwide trends in insufficient physical activity from 2001 to 2016: A pooled analysis of 358 population-based surveys with 1·9 million participants. Lancet Glob. Heal. 2018, 6, e1077–e1086. [Google Scholar] [CrossRef]
- Kozareva, D.A.; Cryan, J.F.; Nolan, Y.M. Born this way: Hippocampal neurogenesis across the lifespan. Aging Cell 2019, 18, e13007. [Google Scholar] [CrossRef]
- Bettio, L.E.B.; Thacker, J.S.; Rodgers, S.P.; Brocardo, P.S.; Christie, B.R.; Gil-Mohapel, J. Interplay between hormones and exercise on hippocampal plasticity across the lifespan. Biochim. Biophys. Acta Mol. Basis Dis. 2020, 1866, 165821. [Google Scholar] [CrossRef] [PubMed]
- Northey, J.M.; Cherbuin, N.; Pumpa, K.L.; Smee, D.J.; Rattray, B. Exercise interventions for cognitive function in adults older than 50: A systematic review with meta-analysis. Br. J. Sports Med. 2018, 52, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Cooper, C.; Moon, H.Y.; Van Praag, H. On the run for hippocampal plasticity. Cold Spring Harb. Perspect. Med. 2017, 8, a029736. [Google Scholar] [CrossRef] [PubMed]
- Hötting, K.; Röder, B. Beneficial effects of physical exercise on neuroplasticity and cognition. Neurosci. Biobehav. Rev. 2013, 37, 2243–2257. [Google Scholar] [CrossRef]
- Erickson, K.I.; Leckie, R.L.; Weinstein, A.M. Physical activity, fitness, and gray matter volume. Neurobiol. Aging 2014, 35, S20–S28. [Google Scholar] [CrossRef]
- Erickson, K.I.; Prakash, R.S.; Voss, M.W.; Chaddock, L.; Hu, L.; Morris, K.S.; White, S.M.; Wójcicki, T.R.; McAuley, E.; Kramer, A.F. Aerobic fitness is associated with hippocampal volume in elderly humans. Hippocampus 2009, 19, 1030–1039. [Google Scholar] [CrossRef]
- Maass, A.; Düzel, S.; Goerke, M.; Becke, A.; Sobieray, U.; Neumann, K.; Lövden, M.; Lindenberger, U.; Bäckman, L.; Braun-Dullaeus, R.; et al. Vascular hippocampal plasticity after aerobic exercise in older adults. Mol. Psychiatry 2015, 20, 585–593. [Google Scholar] [CrossRef]
- Duzel, E.; Van Praag, H.; Sendtner, M. Can physical exercise in old age improve memory and hippocampal function? Brain 2016, 139, 662–673. [Google Scholar] [CrossRef]
- Erickson, K.I.; Voss, M.W.; Prakash, R.S.; Basak, C.; Szabo, A.; Chaddock, L.; Kim, J.S.; Heo, S.; Alves, H.; White, S.M.; et al. Exercise training increases size of hippocampus and improves memory. Proc. Natl. Acad. Sci. USA 2011, 108, 3017–3022. [Google Scholar] [CrossRef]
- Bettio, L.; Thacker, J.S.; Hutton, C.; Christie, B.R. Modulation of synaptic plasticity by exercise. Int. Rev. Neurobiol. 2019, 147, 295–322. [Google Scholar]
- Van Praag, H.; Shubert, T.; Zhao, C.; Gage, F.H. Exercise enhances learning and hippocampal neurogenesis in aged mice. J. Neurosci. 2005, 25, 8680–8685. [Google Scholar] [CrossRef] [PubMed]
- Van Praag, H. Neurogenesis and exercise: Past and future directions. Neuromol. Med. 2008, 10, 128–140. [Google Scholar] [CrossRef] [PubMed]
- Triviño-Paredes, J.; Patten, A.R.; Gil-Mohapel, J.; Christie, B.R. The effects of hormones and physical exercise on hippocampal structural plasticity. Front. Neuroendocr. 2016, 41, 23–43. [Google Scholar] [CrossRef] [PubMed]
- Conlon, J.M.; Herzig, K.H. Towards establishing a higher acceptance rate for PEPTIDES – The peer review process and criteria for acceptance or rejection. Peptides 2019, 112, 32–33. [Google Scholar] [CrossRef]
- Huang, T.; Larsen, K.T.; Ried-Larsen, M.; Møller, N.C.; Andersen, L.B. The effects of physical activity and exercise on brain-derived neurotrophic factor in healthy humans: A review. Scand. J. Med. Sci. Sports 2014, 24, 1–10. [Google Scholar] [CrossRef]
- Vital, T.M.; Stein, A.M.; de Coelho, M.F.G.; Arantes, F.J.; Teodorov, E.; Santos-Galduróz, R.F. Physical exercise and vascular endothelial growth factor (VEGF) in elderly: A systematic review. Arch. Gerontol. Geriatr. 2014, 59, 234–239. [Google Scholar] [CrossRef]
- Kim, S.; Choi, J.Y.; Moon, S.; Park, D.H.; Kwak, H.B.; Kang, J.H. Roles of myokines in exercise-induced improvement of neuropsychiatric function. Pflug. Arch. Eur. J. Physiol. 2019, 471, 491–505. [Google Scholar] [CrossRef]
- Baker, L.D.; Frank, L.L.; Foster-Schubert, K.; Green, P.S.; Wilkinson, C.W.; McTiernan, A.; Plymate, S.R.; Fishel, M.A.; Watson, G.S.; Cholerton, B.A.; et al. Effects of aerobic exercise on mild cognitive impairment: A controlled trial. Arch. Neurol. 2010, 67, 71–79. [Google Scholar] [CrossRef]
- Cassilhas, R.C.; Viana, V.A.R.; Grassmann, V.; Santos, R.T.; Santos, R.F.; Tufik, S.; Mello, M.T. The impact of resistance exercise on the cognitive function of the elderly. Med. Sci. Sports Exerc. 2007, 39, 1401–1407. [Google Scholar] [CrossRef]
- Cho, S.Y.; Roh, H.T. Taekwondo enhances cognitive function as a result of increased neurotrophic growth factors in elderly women. Int. J. Environ. Res. Public Health 2019, 16, 962. [Google Scholar] [CrossRef]
- Ferro, D.A.; Deijen, J.B.; Koppes, L.L.; Van Mechelen, W.; Twisk, J.W.; Drent, M.L. The effects of physical activity and fitness in adolescence on cognition in adulthood and the role of insulin-like growth factor I. J. Phys. Act. Heal. 2016, 13, 392–402. [Google Scholar] [CrossRef] [PubMed]
- Goekint, M.; De Pauw, K.; Roelands, B.; Njemini, R.; Bautmans, I.; Mets, T.; Meeusen, R. Strength training does not influence serum brain-derived neurotrophic factor. Eur. J. Appl. Physiol. 2010, 110, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.M.C.; Wong, M.L.; Lau, B.W.M.; Di Lee, J.C.; Yau, S.Y.; So, K.F. Aerobic exercise interacts with neurotrophic factors to predict cognitive functioning in adolescents. Psychoneuroendocrinology 2014, 39, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Rollero, A.; Murialdo, G.; Fonzi, S.; Garrone, S.; Gianelli, M.V.; Gazzerro, E.; Barreca, A.; Polleri, A. Relationship between cognitive function, growth hormone and insulin-like growth factor I plasma levels in aged subjects. Neuropsychobiology 1998, 38, 73–79. [Google Scholar] [CrossRef]
- Tsai, C.-L.; Wang, C.-H.; Pan, C.-Y.; Chen, F.-C.; Huang, T.-H.; Chou, F.-Y. Executive function and endocrinological responses to acute resistance exercise. Front. Behav. Neurosci. 2014, 8, 262. [Google Scholar] [CrossRef]
- Winker, R.; Lukas, I.; Perkmann, T.; Haslacher, H.; Ponocny, E.; Lehrner, J.; Tscholakoff, D.; Dal-Bianco, P. Cognitive function in elderly marathon runners: Cross-sectional data from the marathon trial (apsoem). Wien. Klin. Wochenschr. 2010, 122, 704–716. [Google Scholar] [CrossRef]
- Deighton, K.; Barry, R.; Connon, C.E.; Stensel, D.J. Appetite, gut hormone and energy intake responses to low volume sprint interval and traditional endurance exercise. Eur. J. Appl. Physiol. 2013, 113, 1147–1156. [Google Scholar] [CrossRef]
- Broom, D.R.; Stensel, D.J.; Bishop, N.C.; Burns, S.F.; Miyashita, M. Exercise-induced suppression of acylated ghrelin in humans. J. Appl. Physiol. 2007, 102, 2165–2171. [Google Scholar] [CrossRef]
- Broom, D.R.; Batterham, R.L.; King, J.A.; Stensel, D.J. Influence of resistance and aerobic exercise on hunger, circulating levels of acylated ghrelin, and peptide YY in healthy males. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 296, R29–R35. [Google Scholar] [CrossRef]
- Toshinai, K.; Kawagoe, T.; Shimbara, T.; Tobina, T.; Nishida, Y.; Mondal, M.S.; Yamaguchi, H.; Date, Y.; Tanaka, H.; Nakazato, M. Acute incremental exercise decreases plasma ghrelin level in healthy men. Horm. Metab. Res. 2007, 39, 849–851. [Google Scholar] [CrossRef]
- Larsen, P.S.; Donges, C.E.; Guelfi, K.J.; Smith, G.C.; Adams, D.R.; Duffield, R. Effects of aerobic, strength or combined exercise on perceived appetite and appetite-related hormones in inactive middle-aged men. Int. J. Sport Nutr. Exerc. Metab. 2017, 27, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Burns, S.F.; Broom, D.R.; Miyashita, M.; Mundy, C.; Stensel, D.J. A single session of treadmill running has no effect on plasma total ghrelin concentrations. J. Sports Sci. 2007, 25, 635–642. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, R.R.; Durand, R.J.; Hollander, D.B.; Tryniecki, J.L.; Hebert, E.P.; Castracane, V.D. Ghrelin and other glucoregulatory hormone responses to eccentric and concentric muscle contractions. Endocrine 2004, 24, 93–98. [Google Scholar] [CrossRef]
- Balaguera-Cortes, L.; Wallman, K.E.; Fairchild, T.J.; Guelfi, K.J. Energy intake and appetite-related hormones following acute aerobic and resistance exercise. Appl. Physiol. Nutr. Metab. 2011, 36, 958–966. [Google Scholar] [CrossRef] [PubMed]
- Martins, C.; Kulseng, B.; King, N.A.; Holst, J.J.; Blundell, J.E. The effects of exercise-induced weight loss on appetite-related peptides and motivation to eat. J. Clin. Endocrinol. Metab. 2010, 95, 1609–1616. [Google Scholar] [CrossRef] [PubMed]
- Guelfi, K.J.; Donges, C.E.; Duffield, R. Beneficial effects of 12 weeks of aerobic compared with resistance exercise training on perceived appetite in previously sedentary overweight and obese men. Metabolism 2013, 62, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Morishima, T.; Kurihara, T.; Hamaoka, T.; Goto, K. Whole body, regional fat accumulation, and appetite-related hormonal response after hypoxic training. Clin. Physiol. Funct. Imaging 2014, 34, 90–97. [Google Scholar] [CrossRef]
- Øktedalen, O.; Opstad, P.K.; Fahrenkrug, J.; Fonnum, F. Plasma Concentration of Vasoactive Intestinal Polypeptide during Prolonged Physical Exercise, Calorie Supply Deficiency, and Sleep Deprivation. Scand. J. Gastroenterol. 1983, 18, 1057–1062. [Google Scholar] [CrossRef]
- Galbo, H.; Hilsted, J.; Fahrenkrug, J.; Schaffalitzky de Muckadell, O.B. Fasting and prolonged exercise increase vasoactive intestinal polypeptide (VIP) in plasma. Acta Physiol. Scand. 1979, 105, 374–377. [Google Scholar] [CrossRef]
- Kaijser, L.; Pernow, J.; Berglund, B.; Lundberg, J.M. Neuropeptide Y is released together with noradrenaline from the human heart during exercise and hypoxia. Clin. Physiol. 1990, 10, 179–188. [Google Scholar] [CrossRef]
- Rämson, R.; Jürimäe, J.; Jürimäe, T.; Mäestu, J. The effect of 4-week training period on plasma neuropeptide Y, leptin and ghrelin responses in male rowers. Eur. J. Appl. Physiol. 2012, 112, 1873–1880. [Google Scholar] [CrossRef] [PubMed]
- Karamouzis, I.; Karamouzis, M.; Vrabas, I.S.; Christoulas, K.; Kyriazis, N.; Giannoulis, E.; Mandroukas, K. The Effects of Marathon Swimming on Serum Leptin and Plasma Neuropeptide Y Levels. Clin. Chem. Lab. Med. 2002, 40, 132–136. [Google Scholar] [CrossRef] [PubMed]
- Onuoha, G.N.; Nicholls, D.P.; Patterson, A.; Beringer, T. Neuropeptide secretion in exercise. Neuropeptides 1998, 32, 319–325. [Google Scholar] [CrossRef]
- Schifter, S.; Breum, L.; Niclasen, B.; Vollmer-Larsen, A.; Rasmussen, H.S.; Graff-Larsen, O. Calcitonin gene-related peptide during exercise and training. Horm. Metab. Res. 1995, 27, 473–475. [Google Scholar] [CrossRef] [PubMed]
- Nindl, B.C.; Pierce, J.R. Insulin-like growth factor i as a biomarker of health, fitness, and training status. Med. Sci. Sports Exerc. 2010, 42, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Gubbi, S.; Quipildor, G.F.; Barzilai, N.; Huffman, D.M.; Milman, S. 40 YEARS of IGF1: IGF1: The Jekyll and Hyde of the aging brain. J. Mol. Endocrinol. 2018, 61, T171–T185. [Google Scholar] [CrossRef]
- Adem, A.; Jossan, S.S.; d’Argy, R.; Gillberg, P.G.; Nordberg, A.; Winblad, B.; Sara, V. Insulin-like growth factor 1 (IGF-1) receptors in the human brain: Quantitative autoradiographic localization. Brain Res. 1989, 503, 299–303. [Google Scholar] [CrossRef]
- Bohannon, N.J.; Corp, E.S.; Wilcox, B.J.; Figlewicz, D.P.; Dorsa, D.M.; Baskin, D.G. Localization of binding sites for insulin-like growth factor-I (IGF-I) in the rat brain by quantitative autoradiography. Brain Res. 1988, 444, 205–213. [Google Scholar] [CrossRef]
- Araujo, D.M.; Lapchak, P.A.; Collier, B.; Chabot, J.G.; Quirion, R. Insulin-like growth factor-1 (somatomedin-C) receptors in the rat brain: Distribution and interaction with the hippocampal cholinergic system. Brain Res. 1989, 484, 130–138. [Google Scholar] [CrossRef]
- Bondy, C.; Werner, H.; Roberts, C.T.; LeRoith, D. Cellular pattern of type-I insulin-like growth factor receptor gene expression during maturation of the rat brain: Comparison with insulin-like growth factors I and II. Neuroscience 1992, 46, 909–923. [Google Scholar] [CrossRef]
- Anlar, B.; Sullivan, K.A.; Feldman, E.L. Insulin-like growth factor-I and central nervous system development. Horm. Metab. Res. 1999, 31, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Trejo, J.L.; Carro, E.; Torres-Alemán, I. Circulating insulin-like growth factor I mediates exercise-induced increases in the number of new neurons in the adult hippocampus. J. Neurosci. 2001, 21, 1628–1634. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Martín, M.; Cifuentes, M.; Grondona, J.M.; López-Ávalos, M.D.; Gómez-Pinedo, U.; García-Verdugo, J.M.; Fernández-Llebrez, P. IGF-I stimulates neurogenesis in the hypothalamus of adult rats. Eur. J. Neurosci. 2010, 31, 1533–1548. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, S.; Fernandez, A.M.; Lopez-Lopez, C.; Torres-Aleman, I. Emerging roles of insulin-like growth factor-I in the adult brain. Growth Horm. IGF Res. 2007, 17, 89–95. [Google Scholar] [CrossRef]
- Carro, E.; Nuñez, A.; Busiguina, S.; Torres-Aleman, I. Circulating insulin-like growth factor I mediates effects of exercise on the brain. J. Neurosci. 2000, 20, 2926–2933. [Google Scholar] [CrossRef]
- Llorens-Martín, M.; Torres-Alemán, I.; Trejo, J.L. Growth factors as mediators of exercise actions on the brain. Neuromol. Med. 2008, 10, 99–107. [Google Scholar] [CrossRef]
- Reinhardt, R.R.; Bondy, C.A. Insulin-like growth factors cross the blood-brain barrier. Endocrinology 1994, 135, 1753–1761. [Google Scholar] [CrossRef]
- Wrigley, S.; Arafa, D.; Tropea, D. Insulin-like growth factor 1: At the crossroads of brain development and aging. Front. Cell. Neurosci. 2017, 11, 14. [Google Scholar] [CrossRef]
- Sjögren, K.; Liu, J.L.; Blad, K.; Skrtic, S.; Vidal, O.; Wallenius, V.; Leroith, D.; Törnell, J.; Isaksson, O.G.P.; Jansson, J.O.; et al. Liver-derived insulin-like growth factor I (IGF-I) is the principal source of IGF-I in blood but is not required for postnatal body growth in mice. Proc. Natl. Acad. Sci. USA 1999, 96, 7088–7092. [Google Scholar] [CrossRef]
- Yakar, S.; Liu, J.L.I.; Stannard, B.; Butler, A.; Accili, D.; Sauer, B.; Leroith, D. Normal growth and development in the absence of hepatic insulin-like growth factor I. Proc. Natl. Acad. Sci. USA 1999, 96, 7324–7329. [Google Scholar] [CrossRef]
- Yamamoto, H.; Sohmiya, M.; Oka, N.; Kato, Y. Effects of aging and sex on plasma insulin-like growth factor I (IGF-I) levels in normal adults. Acta Endocrinol. 1991, 124, 497–500. [Google Scholar] [CrossRef] [PubMed]
- Trejo, J.I.; Piriz, J.; Llorens-Martin, M.V.; Fernandez, A.M.; Bolós, M.; LeRoith, D.; Nuñez, A.; Torres-Aleman, I. Central actions of liver-derived insulin-like growth factor I underlying its pro-cognitive effects. Mol. Psychiatry 2007, 12, 1118–1128. [Google Scholar] [CrossRef] [PubMed]
- Svensson, J.; Diez, M.; Engel, J.; Wass, C.; Tivesten, Å.; Jansson, J.O.; Isaksson, O.; Archer, T.; Hökfelt, T.; Ohlsson, C. Endocrine, liver-derived IGF-I is of importance for spatial learning and memory in old mice. J. Endocrinol. 2006, 189, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Carro, E.; Trejo, J.L.; Busiguina, S.; Torres-Aleman, I. Circulating insulin-like growth factor I mediates the protective effects of physical exercise against brain insults of different etiology and anatomy. J. Neurosci. 2001, 21, 5678–5684. [Google Scholar] [CrossRef]
- Ashpole, N.M.; Sanders, J.E.; Hodges, E.L.; Yan, H.; Sonntag, W.E. Growth hormone, insulin-like growth factor-1 and the aging brain. Exp. Gerontol. 2015, 68, 76–81. [Google Scholar] [CrossRef]
- Muller, A.P.; Fernandez, A.M.; Haas, C.; Zimmer, E.; Portela, L.V.; Torres-Aleman, I. Reduced brain insulin-like growth factor I function during aging. Mol. Cell. Neurosci. 2012, 49, 9–12. [Google Scholar] [CrossRef]
- Arwert, L.I.; Deijen, J.B.; Drent, M.L. The relation between insulin-like growth factor I levels and cognition in healthy elderly: A meta-analysis. Growth Horm. IGF Res. 2005, 15, 416–422. [Google Scholar] [CrossRef]
- Arwert, L.I.; Veltman, D.J.; Deijen, J.B.; Lammertsma, A.A.; Jonker, C.; Drent, M.L. Memory performance and the growth hormone/insulin-like growth factor axis in elderly: A positron emission tomography study. Neuroendocrinology 2005, 81, 31–40. [Google Scholar] [CrossRef]
- Kalmijn, S.; Janssen, J.A.M.J.L.; Pols, H.A.P.; Lamberts, S.W.J.; Breteler, M.M.B. A prospective study on circulating insulin-like growth factor I (IGF-I), IGF-binding proteins, and cognitive function in the elderly. J. Clin. Endocrinol. Metab. 2000, 85, 4551–4555. [Google Scholar] [CrossRef]
- Okereke, O.; Kang, J.H.; Ma, J.; Hankinson, S.E.; Pollak, M.N.; Grodstein, F. Plasma IGF-I levels and cognitive performance in older women. Neurobiol. Aging 2007, 28, 135–142. [Google Scholar] [CrossRef]
- Deak, F.; Sonntag, W.E. Aging, synaptic dysfunction, and insulin-like growth factor (IGF)-1. J. Gerontol. Ser. A Biol. Sci. Med Sci. 2012, 67A, 611–625. [Google Scholar] [CrossRef] [PubMed]
- Okereke, O.I.; Kang, J.H.; Ma, J.; Gaziano, J.M.; Grodstein, F. Midlife plasma insulin-like growth factor I and cognitive function in older men. J. Clin. Endocrinol. Metab. 2006, 91, 4306–4312. [Google Scholar] [CrossRef] [PubMed]
- Aleman, A.; Verhaar, H.J.J.; De Haan, E.H.F.; De Vries, W.R.; Samson, M.M.; Drent, M.L.; Van Der Veen, E.A.; Koppeschaar, H.P.F. Insulin-like growth factor-I and cognitive function in healthy older men. J. Clin. Endocrinol. Metab. 1999, 84, 471–475. [Google Scholar] [CrossRef]
- Aleman, A.; De Vries, W.R.; Koppeschaar, H.P.F.; Osman-Dualeh, M.; Verhaar, H.J.J.; Samson, M.M.; Bol, E.; De Haan, E.H.F. Relationship between circulating levels of sex hormones and insulin-like growth factor-1 and fluid intelligence in older men. Exp. Aging Res. 2001, 27, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Dik, M.G.; Pluijm, S.M.F.; Jonker, C.; Deeg, D.J.H.; Lomecky, M.Z.; Lips, P. Insulin-like growth factor I (IGF-I) and cognitive decline in older persons. Neurobiol. Aging 2003, 24, 573–581. [Google Scholar] [CrossRef]
- Howden, E.J.; Perhonen, M.; Peshock, R.M.; Zhang, R.; Arbab-Zadeh, A.; Adams-Huet, B.; Levine, B.D. Females have a blunted cardiovascular response to one year of intensive supervised endurance training. J. Appl. Physiol. 2015, 119, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Morley, J.E.; Kaiser, F.; Raum, W.J.; Perry, H.M.; Flood, J.F.; Jensen, J.; Silver, A.J.; Roberts, E. Potentially predictive and manipulable blood serum correlates of aging in the healthy human male: Progressive decreases in bioavailable testosterone, dehydroepiandrosterone sulfate, and the ratio of insulin-like growth factor 1 to growth hormone. Proc. Natl. Acad. Sci. USA 1997, 94, 7537–7542. [Google Scholar] [CrossRef]
- Haydar, Z.R.; Blackman, M.R.; Tobin, J.D.; Wright, J.G.; Fleg, J.L. The relationship between aerobic exercise capacity and circulating IGF-1 levels in healthy men and women. J. Am. Geriatr. Soc. 2000, 48, 139–145. [Google Scholar] [CrossRef]
- Kraemer, W.J.; Häkkinen, K.; Newton, R.U.; Nindl, B.C.; Volek, J.S.; Mccormick, M.; Gotshalk, L.A.; Gordon, S.E.; Fleck, S.J.; Campbell, W.W.; et al. Effects of heavy-resistance training on hormonal response patterns in younger vs. older men. J. Appl. Physiol. 1999, 87, 982–992. [Google Scholar] [CrossRef]
- Tsai, C.L.; Wang, C.H.; Pan, C.Y.; Chen, F.C. The effects of long-term resistance exercise on the relationship between neurocognitive performance and GH, IGF-1, and homocysteine levels in the elderly. Front. Behav. Neurosci. 2015, 9, 23. [Google Scholar] [CrossRef]
- Shibuya-Tayoshi, S.; Sumitani, S.; Kikuchi, K.; Tanaka, T.; Tayoshi, S.; Ueno, S.I.; Ohmori, T. Activation of the prefrontal cortex during the Trail-Making Test detected with multichannel near-infrared spectroscopy. Psychiatry Clin. Neurosci. 2007, 61, 616–621. [Google Scholar] [CrossRef] [PubMed]
- Häkkinen, K.; Pakarinen, A.; Kraemer, W.J.; Häkkinen, A.; Valkeinen, H.; Alen, M. Selective muscle hypertrophy, changes in EMG and force, and serum hormones during strength training in older women. J. Appl. Physiol. 2001, 91, 569–580. [Google Scholar] [CrossRef] [PubMed]
- Pareja-Galeano, H.; Brioche, T.; Sanchis-Gomar, F.; Montal, A.; Jovaní, C.; Martínez-Costa, C.; Gomez-Cabrera, M.C.; Viña, J. Impact of exercise training on neuroplasticity-related growth factors in adolescents. J. Musculoskelet. Neuronal Interact. 2013, 13, 368–371. [Google Scholar] [PubMed]
- Nindl, B.C.; Santtila, M.; Vaara, J.; Hakkinen, K.; Kyrolainen, H. Circulating IGF-I is associated with fitness and health outcomes in a population of 846 young healthy men. Growth Horm. IGF Res. 2011, 21, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, A.J.; Brasel, J.A.; Hintz, R.L.; Mohan, S.; Cooper, D.M. Acute effect of brief low- and high-intensity exercise on circulating insulin-like growth factor (IGF) I, II, and IGF-binding protein-3 and its proteolysis in young healthy men. J. Clin. Endocrinol. Metab. 1996, 81, 3492–3497. [Google Scholar]
- Borges Bastos, C.L.; Miranda, H.; De Souza Vale, R.G.; Portal, M.D.N.; Thiago Gomes, M.; Da Silva Novaes, J.; Winchester, J.B. Chronic effect of static stretching on strength performance and basal serum IGF-1 levels. J. Strength Cond. Res. 2013, 27, 2465–2472. [Google Scholar] [CrossRef]
- Borst, S.E.; De Hoyos, D.V.; Garzarella, L.; Vincent, K.; Pollock, B.H.; Lowenthal, D.T.; Pollock, M.L. Effects of resistance training on insulin-like growth factor-I and IGF binding proteins. Med. Sci. Sports Exerc. 2001, 33, 648–653. [Google Scholar] [CrossRef]
- Rubin, M.R.; Kraemer, W.J.; Maresh, C.M.; Volek, J.S.; Ratamess, N.A.; Vanheest, J.L.; Silvestre, R.; French, D.N.; Sharman, M.J.; Judelson, D.A.; et al. High-affinity growth hormone binding protein and acute heavy resistance exercise. Med. Sci. Sports Exerc. 2005, 37, 395–403. [Google Scholar] [CrossRef]
- Ehrnborg, C.; Lange, K.H.W.; Dall, R.; Christiansen, J.S.; Lundberg, P.-A.; Baxter, R.C.; Boroujerdi, M.A.; Bengtsson, B.-A.; Healey, M.-L.; Pentecost, C.; et al. The Growth Hormone/Insulin-Like Growth Factor-I Axis Hormones and Bone Markers in Elite Athletes in Response to a Maximum Exercise Test. J. Clin. Endocrinol. Metab. 2003, 88, 394–401. [Google Scholar] [CrossRef]
- Gregory, S.M.; Spiering, B.A.; Alemany, J.A.; Tuckow, A.P.; Rarick, K.R.; Staab, J.S.; Hatfield, D.L.; Kraemer, W.J.; Maresh, C.M.; Nindl, B.C. Exercise-induced insulin-like growth factor I system concentrations after training in women. Med. Sci. Sports Exerc. 2013, 45, 420–428. [Google Scholar] [CrossRef]
- Nemet, D.; Connolly, P.H.; Pontello-Pescatello, A.M.; Rose-Gottron, C.; Larson, J.K.; Galassetti, P.; Cooper, D.M. Negative energy balance plays a major role in the IGF-I response to exercise training. J. Appl. Physiol. 2004, 96, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Buckley, J.D.; Abbott, M.J.; Brinkworth, G.D.; Whyte, P.B.D. Bovine colostrum supplementation during endurance running training improves recovery, but not performance. J. Sci. Med. Sport 2002, 5, 65–79. [Google Scholar] [CrossRef]
- Davison, G.; Jones, A.W.; Marchbank, T.; Playford, R.J. Oral bovine colostrum supplementation does not increase circulating insulin-like growth factor-1 concentration in healthy adults: Results from short- and long-term administration studies. Eur. J. Nutr. 2020, 59, 1473–1479. [Google Scholar] [CrossRef] [PubMed]
- Schiffer, T.; Schulte, S.; Hollmann, W.; Bloch, W.; Strüder, H.K. Effects of strength and endurance training on brain-derived neurotrophic factor and insulin-like growth factor 1 in humans. Horm. Metab. Res. 2009, 41, 250–254. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo, M.; Ibañez, J.; González-Badillo, J.J.; Häkkinen, K.; Ratamess, N.A.; Kraemer, W.J.; French, D.N.; Eslava, J.; Altadill, A.; Asiain, X.; et al. Differential effects of strength training leading to failure versus not to failure on hormonal responses, strength, and muscle power gains. J. Appl. Physiol. 2006, 100, 1647–1656. [Google Scholar] [CrossRef] [PubMed]
- Scheett, T.P.; Nemet, D.; Stoppani, J.; Maresh, C.M.; Newcomb, R.; Cooper, D.M. The effect of endurance-type exercise training on growth mediators and inflammatory cytokines in pre-pubertal and early pubertal males. Pediatr. Res. 2002, 52, 491–497. [Google Scholar] [CrossRef]
- Seo, D.; Jun, T.W.; Park, K.S.; Chang, H.; So, W.Y.; Song, W. 12 Weeks of combined exercise is better than aerobic exercise for increasing growth hormone in middle-aged women. Int. J. Sport Nutr. Exerc. Metab. 2010, 20, 21–26. [Google Scholar] [CrossRef]
- Cotman, C.W.; Berchtold, N.C.; Christie, L.A. Exercise builds brain health: Key roles of growth factor cascades and inflammation. Trends Neurosci. 2007, 30, 464–472. [Google Scholar] [CrossRef]
- Trejo, J.L.; Carro, E.; Garcia-Galloway, E.; Torres-Aleman, I. Role of insulin-like growth factor I signaling in neurodegenerative diseases. J. Mol. Med. 2004, 82, 156–162. [Google Scholar] [CrossRef]
- Trejo, J.L.; LLorens-Martín, M.V.; Torres-Alemán, I. The effects of exercise on spatial learning and anxiety-like behavior are mediated by an IGF-I-dependent mechanism related to hippocampal neurogenesis. Mol. Cell. Neurosci. 2008, 37, 402–411. [Google Scholar] [CrossRef]
- Yamada, M.; Ohnishi, H.; Sano, S.; Nakatani, A.; Ikeuchi, T.; Hatanaka, H. Insulin receptor substrate (IRS)-1 and IRS-2 are tyrosine-phosphorylated and associated with phosphatidylinositol 3-kinase in response to brain-derived neurotrophic factor in cultured cerebral cortical neurons. J. Biol. Chem. 1997, 272, 30334–30339. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.; Vaynman, S.; Akhavan, M.; Ying, Z.; Gomez-Pinilla, F. Insulin-like growth factor I interfaces with brain-derived neurotrophic factor-mediated synaptic plasticity to modulate aspects of exercise-induced cognitive function. Neuroscience 2006, 140, 823–833. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Pinilla, F.; Vaynman, S.; Ying, Z. Brain-derived neurotrophic factor functions as a metabotrophin to mediate the effects of exercise on cognition. Eur. J. Neurosci. 2008, 28, 2278–2287. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Quan, M.; Su, L.; Zhang, H.; Zhang, J.; Zhang, J.; Fang, H.; Cao, Z.B.; Zhu, Z.; Niu, Z.; et al. Effect of Physical Activity on Cognitive Development: Protocol for a 15-Year Longitudinal Follow-Up Study. BioMed Res. Int. 2017, 2017, 8568459. [Google Scholar] [CrossRef] [PubMed]
- De Lecea, L.; Kilduff, T.S.; Peyron, C.; Gao, X.B.; Foye, P.E.; Danielson, P.E.; Fukuhara, C.; Battenberg, E.L.F.; Gautvik, V.T.; Bartlett, F.S.; et al. The hypocretins: Hypothalamus-specific peptides with neuroexcitatory activity. Proc. Natl. Acad. Sci. USA 1998, 95, 322–327. [Google Scholar] [CrossRef]
- Sakurai, T.; Amemiya, A.; Ishii, M.; Matsuzaki, I.; Chemelli, R.M.; Tanaka, H.; Williams, S.C.; Richardson, J.A.; Kozlowski, G.P.; Wilson, S.; et al. Orexins and orexin receptors: A family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell 1998, 92, 573–585. [Google Scholar] [CrossRef]
- Zink, A.N.; Perez-Leighton, C.E.; Kotz, C.M. The orexin neuropeptide system: Physical activity and hypothalamic function throughout the aging process. Front. Syst. Neurosci. 2014, 8, 211. [Google Scholar] [CrossRef]
- Heinonen, M.V.; Purhonen, A.K.; Mäkelä, K.A.; Herzig, K.H. Functions of orexins in peripheral tissues. Acta Physiol. 2008, 192, 471–485. [Google Scholar] [CrossRef]
- Mäkelä, K.A.; Wigren, H.-K.; Zant, J.C.; Sakurai, T.; Alhonen, L.; Kostin, A.; Porkka-Heiskanen, T.; Herzig, K.-H. Characterization of sleep-wake patterns in a novel transgenic mouse line overexpressing human prepro-orexin/hypocretin. Acta Physiol. 2010, 198, 237–249. [Google Scholar] [CrossRef]
- Messina, G.; Di Bernardo, G.; Viggiano, A.; De Luca, V.; Monda, V.; Messina, A.; Chieffi, S.; Galderisi, U.; Monda, M. Exercise increases the level of plasma orexin A in humans. J. Basic Clin. Physiol. Pharmacol. 2016, 27, 611–616. [Google Scholar] [CrossRef]
- Mäkelä, K.A.; Karhu, T.; Jurado Acosta, A.; Vakkuri, O.; Leppäluoto, J.; Herzig, K.-H. Plasma Orexin-A Levels Do Not Undergo Circadian Rhythm in Young Healthy Male Subjects. Front. Endocrinol. (Lausanne) 2018, 9, 710. [Google Scholar] [CrossRef]
- Kastin, A.J.; Akerstrom, V. Orexin A but not orexin B rapidly enters brain from blood by simple diffusion. J. Pharmacol. Exp. Ther. 1999, 289, 219–223. [Google Scholar] [PubMed]
- Zhao, X.; Zhang, R.; Tang, S.; Ren, Y.Y.; Yang, W.X.; Liu, X.M.; Tang, J.Y. Orexin-A-induced ERK1/2 activation reverses impaired spatial learning and memory in pentylenetetrazol-kindled rats via OX1R-mediated hippocampal neurogenesis. Peptides 2014, 54, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Brownell, S.E.; Conti, B. Age- and gender-specific changes of hypocretin immunopositive neurons in C57Bl/6 mice. Neurosci. Lett. 2010, 472, 29–32. [Google Scholar] [CrossRef]
- Sawai, N.; Ueta, Y.; Nakazato, M.; Ozawa, H. Developmental and aging change of orexin-A and -B immunoreactive neurons in the male rat hypothalamus. Neurosci. Lett. 2010, 468, 51–55. [Google Scholar] [CrossRef]
- Kessler, B.A.; Stanley, E.M.; Frederick-Duus, D.; Fadel, J. Age-related loss of orexin/hypocretin neurons. Neuroscience 2011, 178, 82–88. [Google Scholar] [CrossRef]
- Kojima, M.; Hosoda, H.; Date, Y.; Nakazato, M.; Matsuo, H.; Kangawa, K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 1999, 402, 656–660. [Google Scholar] [CrossRef]
- Karhunen, L.J.; Juvonen, K.R.; Huotari, A.; Purhonen, A.K.; Herzig, K.H. Effect of protein, fat, carbohydrate and fibre on gastrointestinal peptide release in humans. Regul. Pept. 2008, 149, 70–78. [Google Scholar] [CrossRef]
- Heinonen, M.V.; Karhunen, L.J.; Chabot, E.D.; Toppinen, L.K.; Juntunen, K.S.; Laaksonen, D.E.; Siloaho, M.; Liukkonen, K.-H.; Herzig, K.-H.; Niskanen, L.K.; et al. Plasma ghrelin levels after two high-carbohydrate meals producing different insulin responses in patients with metabolic syndrome. Regul. Pept. 2007, 138, 118–125. [Google Scholar] [CrossRef]
- Witte, A.V.; Fobker, M.; Gellner, R.; Knecht, S.; Floel, A. Caloric restriction improves memory in elderly humans. Proc. Natl. Acad. Sci. USA 2009, 106, 1255–1260. [Google Scholar] [CrossRef]
- Banks, W.A.; Tschöp, M.; Robinson, S.M.; Heiman, M.L. Extent and direction of ghrelin transport across the blood-brain barrier is determined by its unique primary structure. J. Pharmacol. Exp. Ther. 2002, 302, 822–827. [Google Scholar] [CrossRef] [PubMed]
- Banks, W.A. The blood-brain barrier: Connecting the gut and the brain. Regul. Pept. 2008, 149, 11–14. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.; Kim, S.; Park, S. Neurogenic effects of ghrelin on the hippocampus. Int. J. Mol. Sci. 2017, 18, 588. [Google Scholar] [CrossRef] [PubMed]
- Kent, B.A.; Oomen, C.A.; Bekinschtein, P.; Bussey, T.J.; Saksida, L.M. Cognitive enhancing effects of voluntary exercise, caloric restriction and environmental enrichment: A role for adult hippocampal neurogenesis and pattern separation? Curr. Opin. Behav. Sci. 2015, 4, 179–185. [Google Scholar] [CrossRef]
- Carlini, V.P.; Monzón, M.E.; Varas, M.M.; Cragnolini, A.B.; Schiöth, H.B.; Scimonelli, T.N.; De Barioglio, S.R. Ghrelin increases anxiety-like behavior and memory retention in rats. Biochem. Biophys. Res. Commun. 2002, 299, 739–743. [Google Scholar] [CrossRef]
- Carlini, V.P.; Varas, M.M.; Cragnolini, A.B.; Schiöth, H.B.; Scimonelli, T.N.; De Barioglio, S.R. Differential role of the hippocampus, amygdala, and dorsal raphe nucleus in regulating feeding, memory, and anxiety-like behavioral responses to ghrelin. Biochem. Biophys. Res. Commun. 2004, 313, 635–641. [Google Scholar] [CrossRef]
- Tóth, K.; László, K.; Lukács, E.; Lénárd, L. Intraamygdaloid microinjection of acylated-ghrelin influences passive avoidance learning. Behav. Brain Res. 2009, 202, 308–311. [Google Scholar] [CrossRef]
- Davis, J.F.; Choi, D.L.; Clegg, D.J.; Benoit, S.C. Signaling through the ghrelin receptor modulates hippocampal function and meal anticipation in mice. Physiol. Behav. 2011, 103, 39–43. [Google Scholar] [CrossRef]
- Kent, B.A.; Beynon, A.L.; Hornsby, A.K.E.; Bekinschtein, P.; Bussey, T.J.; Davies, J.S.; Saksida, L.M. The orexigenic hormone acyl-ghrelin increases adult hippocampal neurogenesis and enhances pattern separation. Psychoneuroendocrinology 2015, 51, 431–439. [Google Scholar] [CrossRef]
- Zhao, Z.; Liu, H.; Xiao, K.; Yu, M.; Cui, L.; Zhu, Q.; Zhao, R.; Li, G.D.; Zhou, Y. Ghrelin administration enhances neurogenesis but impairs spatial learning and memory in adult mice. Neuroscience 2014, 257, 175–185. [Google Scholar] [CrossRef]
- Dorling, J.; Broom, D.R.; Burns, S.F.; Clayton, D.J.; Deighton, K.; James, L.J.; King, J.A.; Miyashita, M.; Thackray, A.E.; Batterham, R.L.; et al. Acute and chronic effects of exercise on appetite, energy intake, and appetite-related hormones: The modulating effect of adiposity, sex, and habitual physical activity. Nutrients 2018, 10, 1140. [Google Scholar] [CrossRef] [PubMed]
- Tatemoto, K.; Carlquist, M.; Mutt, V. Neuropeptide Y-A novel brain peptide with structural similarities to peptide YY and pancreatic polypeptide. Nature 1982, 296, 659–660. [Google Scholar] [CrossRef] [PubMed]
- Tatemoto, K. Neuropeptide Y: Complete amino acid sequence of the brain peptide. Proc. Natl. Acad. Sci. USA 1982, 79, 5485–5489. [Google Scholar] [CrossRef] [PubMed]
- Redrobe, J.P.; Dumont, Y.; St-Pierre, J.A.; Quirion, R. Multiple receptors for neuropeptide Y in the hippocampus: Putative roles in seizures and cognition. Brain Res. 1999, 848, 153–166. [Google Scholar] [CrossRef]
- Flood, J.F.; Hernandez, E.N.; Morley, J.E. Modulation of memory processing by neuropeptide Y. Brain Res. 1987, 421, 280–290. [Google Scholar] [CrossRef]
- Kastin, A.J.; Akerstrom, V. Nonsaturable entry of neuropeptide Y into brain. Am. J. Physiol. Endocrinol. Metab. 1999, 276, E479–E482. [Google Scholar] [CrossRef]
- Bjørnebekk, A.; Mathé, A.A.; Brené, S. Running has differential effects on NPY, opiates, and cell proliferation in an animal model of depression and controls. Neuropsychopharmacology 2006, 31, 256–264. [Google Scholar] [CrossRef][Green Version]
- Chen, J.X.; Zhao, X.; Yue, G.X.; Wang, Z.F. Influence of acute and chronic treadmill exercise on rat plasma lactate and brain NPY, L-ENK, DYN A1-13. Cell. Mol. Neurobiol. 2007, 27, 1–10. [Google Scholar] [CrossRef]
- Lewis, D.E.; Shellard, L.; Koeslag, D.G.; Boer, D.E.; McCarthy, H.D.; McKibbin, P.E.; Russell, J.C.; Williams, G. Intense exercise and food restriction cause similar hypothalamic neuropeptide Y increases in rats. Am. J. Physiol. Metab. 1993, 264, E279–E284. [Google Scholar] [CrossRef]
- Lundberg, J.M.; Martinsson, A.; Hemsén, A.; Theodorsson-Norheim, E.; Svedenhag, J.; Ekblom, B.; Hjemdahl, P. Co-release of neuropeptide Y and catecholamines during physical exercise in man. Biochem. Biophys. Res. Commun. 1985, 133, 30–36. [Google Scholar] [CrossRef]
- Coiro, V.; Volpi, R.; Cataldo, S.; Araldi, A.; Stella, A.; Saccani-Jotti, G.; Maffei, M.L.; Volta, E.; Chiodera, P. Somatostatin Reduces Neuropeptide Y Rise Induced by Physical Exercise. Horm. Metab. Res. 2011, 43, 361–363. [Google Scholar] [CrossRef] [PubMed]
- Beck, B.; Pourié, G. Ghrelin, neuropeptide Y, and other feeding-regulatory peptides active in the Hippocampus: Role in learning and memory. Nutr. Rev. 2013, 71, 541–561. [Google Scholar] [CrossRef] [PubMed]
- Flood, J.F.; Baker, M.L.; Hernandez, E.N.; Morley, J.E. Modulation of memory processing by neuropeptide Y varies with brain injection site. Brain Res. 1989, 503, 73–82. [Google Scholar] [CrossRef]
- Ishida, H.; Shirayama, Y.; Iwata, M.; Katayama, S.; Yamamoto, A.; Kawahara, R.; Nakagome, K. Infusion of neuropeptide Y into CA3 region of hippocampus produces antidepressant-like effect via Y1 receptor. Hippocampus 2007, 17, 271–280. [Google Scholar] [CrossRef]
- Bouchard, P.; Maurice, T.; St-Pierre, S.; Privat, A.; Quirion, R. Neuropeptide Y and the calcitonin gene-related peptide attenuate learning impairments induced by MK-801 via a sigma receptor-related mechanism. Eur. J. Neurosci. 1997, 9, 2142–2151. [Google Scholar] [CrossRef] [PubMed]
- Karl, T.; Duffy, L.; Herzog, H. Behavioural profile of a new mouse model for NPY deficiency. Eur. J. Neurosci. 2008, 28, 173–180. [Google Scholar] [CrossRef]
- Bannon, A.W.; Seda, J.; Carmouche, M.; Francis, J.M.; Norman, M.H.; Karbon, B.; McCaleb, M.L. Behavioral characterization of neuropeptide Y knockout mice. Brain Res. 2000, 868, 79–87. [Google Scholar] [CrossRef]
- Nakajima, M.; Inui, A.; Teranishi, A.; Miura, M.; Hirosue, Y.; Okita, M.; Himori, N.; Baba, S.; Kasuga, M. Effects of pancreatic polypeptide family peptides on feeding and learning behavior in mice. J. Pharmacol. Exp. Ther. 1994, 268, 1010–1014. [Google Scholar]
- Sørensen, A.T.; Kanter-Schlifke, I.; Carli, M.; Balducci, C.; Noe, F.; During, M.J.; Vezzani, A.; Kokaia, M. NPY gene transfer in the hippocampus attenuates synaptic plasticity and learning. Hippocampus 2008, 18, 564–574. [Google Scholar] [CrossRef]
- Thorsell, A.; Michalkiewicz, M.; Dumont, Y.; Quirion, R.; Caberlotto, L.; Rimondini, R.; Mathé, A.A.; Heilig, M. Behavioral insensitivity to restraint stress, absent fear suppression of behavior and impaired spatial learning in transgenic rats with hippocampal neuropeptide Y overexpression. Proc. Natl. Acad. Sci. USA 2000, 97, 12852–12857. [Google Scholar] [CrossRef]
- Carvajal, C.C.; Vercauteren, F.; Dumont, Y.; Michalkiewicz, M.; Quirion, R. Aged neuropeptide Y transgenic rats are resistant to acute stress but maintain spatial and non-spatial learning. Behav. Brain Res. 2004, 153, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Gøtzsche, C.R.; Woldbye, D.P.D. The role of NPY in learning and memory. Neuropeptides 2016, 55, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Tatemoto, K.; Rökaeus, Å.; Jörnvall, H.; McDonald, T.J.; Mutt, V. Galanin-a novel biologically active peptide from porcine intestine. FEBS Lett. 1983, 164, 124–128. [Google Scholar] [CrossRef]
- Bulaj, G.; Green, B.R.; Lee, H.K.; Robertson, C.R.; White, K.; Zhang, L.; Sochanska, M.; Flynn, S.P.; Scholl, E.A.; Pruess, T.H.; et al. Design, synthesis, and characterization of high-affinity, systemically-active galanin analogues with potent anticonvulsant activities. J. Med. Chem. 2008, 51, 8038–8047. [Google Scholar] [PubMed]
- Murray, P.S.; Groves, J.L.; Pettett, B.J.; Britton, S.L.; Koch, L.G.; Dishman, R.K.; Holmes, P.V. Locus coeruleus galanin expression is enhanced after exercise in rats selectively bred for high capacity for aerobic activity. Peptides 2010, 31, 2264–2268. [Google Scholar] [CrossRef]
- Branchek, T.A.; Smith, K.E.; Gerald, C.; Walker, M.W. Galanin receptor subtypes. Trends Pharmacol. Sci. 2000, 21, 109–117. [Google Scholar] [CrossRef]
- Hökfelt, T.; Barde, S.; Xu, Z.-Q.D.; Kuteeva, E.; Rüegg, J.; Le Maitre, E.; Risling, M.; Kehr, J.; Ihnatko, R.; Theodorsson, E.; et al. Neuropeptide and Small Transmitter Coexistence: Fundamental Studies and Relevance to Mental Illness. Front. Neural Circuits 2018, 12, 106. [Google Scholar] [CrossRef]
- Fuxe, K.; Borroto-Escuela, D.O.; Romero-Fernandez, W.; Tarakanov, A.O.; Calvo, F.; Garriga, P.; Tena, M.; Narvaez, M.; Millón, C.; Parrado, C.; et al. On the existence and function of galanin receptor heteromers in the central nervous system. Front. Endocrinol. (Lausanne) 2012, 3, 127. [Google Scholar] [CrossRef]
- Dutar, P.; Lamour, Y.; Nicoll, R.A. Galanin blocks the slow cholinergic EPSP in CA1 pyramidal neurons from ventral hippocampus. Eur. J. Pharmacol. 1989, 164, 355–360. [Google Scholar] [CrossRef]
- Zini, S.; Roisin, M.P.; Langel, U.; Bartfai, T.; Ben-Ari, Y. Galanin reduces release of endogeneous excitatory amino acids in the rat hippocampus. Eur. J. Pharmacol. Mol. Pharmacol. 1993, 245, 1–7. [Google Scholar] [CrossRef]
- Ögren, S.O.; Kehr, J.; Schött, P.A. Effects of ventral hippocampal galanin on spatial learning and on in vivo acetylcholine release in the rat. Neuroscience 1996, 75, 1127–1140. [Google Scholar] [CrossRef]
- Schött, P.A.; Hökfelt, T.; Ögren, S.O. Galanin and spatial learning in the rat. Evidence for a differential role for galanin in subregions of the hippocampal formation. Neuropharmacology 2000, 39, 1386–1403. [Google Scholar] [CrossRef]
- Ögren, S.O.; Kuteeva, E.; Elvander-Tottie, E.; Hökfelt, T. Neuropeptides in learning and memory processes with focus on galanin. Eur. J. Pharmacol. 2010, 626, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Borbély, É.; Scheich, B.; Helyes, Z. Neuropeptides in learning and memory. Neuropeptides 2013, 47, 439–450. [Google Scholar] [CrossRef] [PubMed]
- McDonald, M.P.; Gleason, T.C.; Robinson, J.K.; Crawley, J.N. Galanin inhibits performance on rodent memory tasks. Ann. N. Y. Acad. Sci. 1998, 863, 305–322. [Google Scholar] [CrossRef] [PubMed]
- Melander, T.; Staines, W.A.; Hökfelt, T.; Rökaeus, Å.; Eckenstein, F.; Salvaterra, P.M.; Wainer, B.H. Galanin-like immunoreactivity in cholinergic neurons of the septum-basal forebrain complex projecting to the hippocampus of the rat. Brain Res. 1985, 360, 130–138. [Google Scholar] [CrossRef]
- Kordower, J.H.; Mufson, E.J. Galanin-like immunoreactivity within the primate basal forebrain: Differential staining patterns between humans and monkeys. J. Comp. Neurol. 1990, 294, 281–292. [Google Scholar] [CrossRef]
- Sundström, E.; Archer, T.; Melander, T.; Hökfelt, T. Galanin impairs acquisition but not retrieval of spatial memory in rats studied in the Morris swim maze. Neurosci. Lett. 1988, 88, 331–335. [Google Scholar] [CrossRef]
- Kinney, J.W.; Starosta, G.; Crawley, J.N. Central galanin administration blocks consolidation of spatial learning. Neurobiol. Learn. Mem. 2003, 80, 42–54. [Google Scholar] [CrossRef]
- Schött, P.A.; Bjelke, B.; Ögren, S.O. Distribution and kinetics of galanin infused into the ventral hippocampus of the rat: Relationship to spatial learning. Neuroscience 1998, 83, 123–136. [Google Scholar] [CrossRef]
- Schött, P.A.; Bjelke, B.; Ögren, S.O. Time-dependent effects of intrahippocampal galanin on spatial learning. Ann. N. Y. Acad. Sci. 1998, 863, 454–456. [Google Scholar] [CrossRef] [PubMed]
- Steiner, R.A.; Hohmann, J.G.; Holmes, A.; Wrenn, C.C.; Cadd, G.; Juréus, A.; Clifton, D.K.; Luo, M.; Gutshall, M.; Ma, S.Y.; et al. Galanin transgenic mice display cognitive and neurochemical deficits characteristic of Alzheimer’s disease. Proc. Natl. Acad. Sci. USA 2001, 98, 4184–4189. [Google Scholar] [CrossRef] [PubMed]
- Kuteeva, E.; Hökfelt, T.; Ögren, S.O. Behavioural characterisation of young adult transgenic mice overexpressing galanin under the PDGF-B promoter. Regul. Pept. 2005, 125, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Wrenn, C.C.; Marriott, L.K.; Kinney, J.W.; Holmes, A.; Wenk, G.L.; Crawley, J.N. Galanin peptide levels in hippocampus and cortex of galanin-overexpressing transgenic mice evaluated for cognitive performance. Neuropeptides 2002, 36, 413–426. [Google Scholar] [CrossRef]
- O’Meara, G.; Coumis, U.; Ma, S.Y.; Kehr, J.; Mahoney, S.; Bacon, A.; Allen, S.J.; Holmes, F.; Kahl, U.; Wang, F.H.; et al. Galanin regulates the postnatal survival of a subset of basal forebrain cholinergic neurons. Proc. Natl. Acad. Sci. USA 2000, 97, 11569–11574. [Google Scholar] [CrossRef]
- Pirondi, S.; D’Intino, G.; Gusciglio, M.; Massella, A.; Giardino, L.; Kuteeva, E.; Ögren, S.O.; Hökfelt, T.; Calzà, L. Changes in brain cholinergic markers and spatial learning in old galanin-overexpressing mice. Brain Res. 2007, 1138, 10–20. [Google Scholar] [CrossRef]
- Wrenn, C.C.; Kinney, J.W.; Marriott, L.K.; Holmes, A.; Harris, A.P.; Saavedra, M.C.; Starosta, G.; Innerfield, C.E.; Jacoby, A.S.; Shine, J.; et al. Learning and memory performance in mice lacking the GAL-R1 subtype of galanin receptor. Eur. J. Neurosci. 2004, 19, 1384–1396. [Google Scholar] [CrossRef]
- Barde, S.; Rüegg, J.; Prud’homme, J.; Ekström, T.J.; Palkovits, M.; Turecki, G.; Bagdy, G.; Ihnatko, R.; Theodorsson, E.; Juhasz, G.; et al. Alterations in the neuropeptide galanin system in major depressive disorder involve levels of transcripts, methylation, and peptide. Proc. Natl. Acad. Sci. USA 2016, 113, E8472–E8481. [Google Scholar] [CrossRef]
- Counts, S.E.; He, B.; Che, S.; Ginsberg, S.D.; Mufson, E.J. Galanin Fiber Hyperinnervation Preserves Neuroprotective Gene Expression in Cholinergic Basal Forebrain Neurons in Alzheimer’s Disease. J. Alzheimer’s Dis. 2009, 18, 885–896. [Google Scholar] [CrossRef]
- Henning, R.J.; Sawmiller, D.R. Vasoactive intestinal peptide: Cardiovascular effects. Cardiovasc. Res. 2001, 49, 27–37. [Google Scholar] [CrossRef]
- Said, S.I. The discovery of VIP: Initially looked for in the lung, isolated from intestine, and identified as a neuropeptide. Peptides 2007, 28, 1620–1621. [Google Scholar] [CrossRef] [PubMed]
- Said, S.I.; Mutt, V. Polypeptide with Broad Biological Activity: Isolation from Small Intestine. Science 1970, 169, 1217–1218. [Google Scholar] [CrossRef] [PubMed]
- Lorén, I.; Emson, P.C.; Fahrenkrug, J.; Björklund, A.; Alumets, J.; Håkanson, R.; Sundler, F. Distribution of vasoactive intestinal polypeptide in the rat and mouse brain. Neuroscience 1979, 4, 1953–1976. [Google Scholar] [CrossRef]
- Dogrukol-Ak, D.; Banks, W.A.; Tuncel, N.; Tuncel, M. Passage of vasoactive intestinal peptide across the blood-brain barrier. Peptides 2003, 24, 437–444. [Google Scholar] [CrossRef]
- Maslov, A.Y.; Barone, T.A.; Plunkett, R.J.; Pruitt, S.C. Neural Stem Cell Detection, Characterization, and Age-Related Changes in the Subventricular Zone of Mice. J. Neurosci. 2004, 24, 1726–1733. [Google Scholar] [CrossRef]
- Cha, C.I.; Lee, Y.I.; Lee, E.; Park, K.H.; Baik, S.H. Age-related changes of VIP, NPY and somatostatin-immunoreactive neurons in the cerebral cortex of aged rats. Brain Res. 1997, 753, 235–244. [Google Scholar] [CrossRef]
- Chaudhury, D.; Loh, D.H.; Dragich, J.M.; Hagopian, A.; Colwell, C.S. Select cognitive deficits in Vasoactive Intestinal Peptide deficient mice. Bmc Neurosci. 2008, 9, 63. [Google Scholar] [CrossRef]
- Takashima, A.; Maeda, Y.; Itoh, S. Influence of chronic intracerebroventricular infusion of vasoactive intestinal peptide (VIP) on memory processes in Morris water pool test in the rat. Peptides 1993, 14, 1073–1078. [Google Scholar] [CrossRef]
- Ivanova, M.; Ternianov, A.; Tashev, R.; Belcheva, S.; Belcheva, I. Lateralized learning and memory effects of vasoactive intestinal peptide infused into the rat hippocampal CA1 area. Regul. Pept. 2009, 156, 42–46. [Google Scholar] [CrossRef]
- Flood, J.F.; Garland, J.S.; Morley, J.E. Vasoactive intestinal peptide (VIP): An amnestic neuropeptide. Peptides 1990, 11, 933–938. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Kobayashi, H. Effects of vasoactive intestinal peptide (VIP) on scopolamine-induced amnesia in the rat. Neuropeptides 1994, 26, 153–158. [Google Scholar] [CrossRef]
- Glowa, J.R.; Panlilio, L.V.; Brenneman, D.E.; Gozes, I.; Fridkin, M.; Hill, J.M. Learning impairment following intracerebral administration of the HIV envelope protein gp120 or a VIP antagonist. Brain Res. 1992, 570, 49–53. [Google Scholar] [CrossRef][Green Version]
- Gressens, P.; Paindaveine, B.; Hill, J.M.; Brenneman, D.E.; Evrard, P. Growth factor properties of VIP during early brain development. Whole embryo culture and in vivo studies. Ann. N. Y. Acad. Sci. 1997, 814, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.M.; Hauser, J.M.; Sheppard, L.M.; Abebe, D.; Spivak-Pohis, I.; Kushnir, M.; Deitch, I.; Gozes, I. Blockage of VIP during mouse embryogenesis modifies adult behavior and results in permanent changes in brain chemistry. J. Mol. Neurosci. 2007, 31, 183–200. [Google Scholar]
- Hill, J.M.; Cuasay, K.; Abebe, D.T. Vasoactive intestinal peptide antagonist treatment during mouse embryogenesis impairs social behavior and cognitive function of adult male offspring. Exp. Neurol. 2007, 206, 101–113. [Google Scholar] [CrossRef]
- Wu, J.Y.; Henins, K.A.; Gressens, P.; Gozes, I.; Fridkin, M.; Brenneman, D.E.; Hill, J.M. Neurobehavioral development of neonatal mice following blockade of VIP during the early embryonic period. Peptides 1997, 18, 1131–1137. [Google Scholar] [CrossRef]
- Gozes, I.; Glowa, J.; Brenneman, D.E.; McCune, S.K.; Lee, E.; Westphal, H. Learning and sexual deficiencies in transgenic mice carrying a chimeric vasoactive intestinal peptide gene. J. Mol. Neurosci. 1993, 4, 185–193. [Google Scholar] [CrossRef]
- MacLaren, D.P.; Raine, N.M.; O’Connor, A.M.; Buchanan, K.D. Human gastrin and vasoactive intestinal polypeptide responses to endurance running in relation to training status and fluid ingested. Clin. Sci. 1995, 89, 137–143. [Google Scholar] [CrossRef]
- Rolandi, E.; Reggiani, E.; Franceschini, R.; Arras, G.B.; Cataldi, A.; De Lucia, F.; Barreca, T. Prolactin release induced by physical exercise is independent from peripheral vasoactive intestinal polypeptide secretion. Ann. Clin. Res. 1988, 20, 428–430. [Google Scholar]
- Woie, L.; Kaada, B.; Opstad, P.K. Increase in plasma vasoactive intestinal polypeptide (VIP) in muscular exercise in humans. Gen. Pharmacol. Vasc. Syst. 1986, 17, 321–326. [Google Scholar] [CrossRef]
- Oektedalen, O.; Opstad, P.K.; Schaffalitzky de Muckadell, O.B.; Fausa, O.; Flaten, O. Basal hyperchlorhydria and its relation to the plasma concentrations of secretin, vasoactive intestinal polypeptide (VIP) and gastrin during prolonged strain. Regul. Pept. 1983, 5, 235–244. [Google Scholar] [CrossRef]
- Amara, S.G.; Jonas, V.; Rosenfeld, M.G.; Ong, E.S.; Evans, R.M. Alternative RNA processing in calcitonin gene expression generates mRNAs encoding different polypeptide products. Nature 1982, 298, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Kraenzlin, M.E.; Ch’ng, J.L.C.; Mulderry, P.K.; Ghatei, M.A.; Bloom, S.R. Infusion of a novel peptide, calcitonin gene-related peptide (CGRP) in man. Pharmacokinetics and effects on gastric acid secretion and on gastrointestinal hormones. Regul. Pept. 1985, 10, 189–197. [Google Scholar] [CrossRef]
- Russell, F.A.; King, R.; Smillie, S.-J.; Kodji, X.; Brain, S.D. Calcitonin Gene-Related Peptide: Physiology and Pathophysiology. Physiol. Rev. 2014, 94, 1099–1142. [Google Scholar] [CrossRef] [PubMed]
- Harada, N.; Narimatsu, N.; Kurihara, H.; Nakagata, N.; Okajima, K. Stimulation of sensory neurons improves cognitive function by promoting the hippocampal production of insulin-like growth factor-I in mice. Transl. Res. 2009, 154, 90–102. [Google Scholar] [CrossRef]
- Messlinger, K. The big CGRP flood-sources, sinks and signalling sites in the trigeminovascular system. J. Headache Pain 2018, 19, 22. [Google Scholar] [CrossRef]
- Hu, D.-E.; Easton, A.S.; Fraser, P.A. TRPV1 activation results in disruption of the blood-brain barrier in the rat. Br. J. Pharmacol. 2005, 146, 576–584. [Google Scholar] [CrossRef]
- Chen, R. Ghrelin improves cognitive function in mice by increasing the production of insulin-like growth factor-I in the hippocampus. Nagoya Med. J. 2012, 52, 117–134. [Google Scholar]
- Pickett, J.; Brayne, C. The scale and profile of global dementia research funding. Lancet 2019, 394, 1888–1889. [Google Scholar] [CrossRef]
| Reference and Study Design | Subjects | Target Area | Type of PA | (Neuro) Peptide | Main Results |
|---|---|---|---|---|---|
| RCT [29] | 33 M and F Sedentary with mild cognitive impairment. 55–85 years old (mean age: 70 years) | Executive function and short-term memory | Acute aerobic exercise | IGF-1 | For women, aerobic exercise improved cognitive performance. For men, aerobic exercise increased plasma IGF-1 levels and improved performance on trails B test. |
| RCT [30] | 62 M Sedentary 65–75 years old | Executive function, short-term memory, attention and long-term episodic memory | Acute, moderate- and high-intensity resistance exercise | IGF-1 | Moderate and high-intensity resistance training have equally beneficial effects on cognition. IGF-1 levels were higher in subjects compared to controls. |
| RCT [31] | 37 F Sedentary 65+ years old | Selective attention, cognitive flexibility, and processing speed | Acute aerobic exercise | IGF-1 | Taekwondo training may improve physical fitness and cognitive functioning in elderly females. IGF-1, VEGF and BDNF may mediate the latter effect. |
| Longitudinal study [32] | 303 M and F Followed from age 13 to 42 years old | Executive functioning and visual-spatial memory | Physical activity and fitness were assessed annually between ages 13 to 16. At mean age 36, physical activity and fitness were assessed. | IGF-1 | For males, there is a significant association between physical activity in adolescence and cognitive capabilities in adulthood. Such association was not found for females. IGF-1 was not found to have an intermediate role for either sex. |
| Controlled trial [33] | 23 M and F Untrained 18–30 years old | Short- and mid-term memory | Regular resistance exercise | IGF-1 | 10 Week strength training period did not influence serum BDNF, IGF-1 or IGFBP-3 levels. There was no difference in short-term memory between experimental and control group. Mid-term memory was not improved after 10-week training program. |
| Between-group design [34] | 91 M and F Healthy, both sedentary and physically active 14–18 years old | Selective attention, cognitive flexibility, processing speed, spatial memory, spatial memory and primary visual cortex function | Regular aerobic exercise | IGF-1 | Regular aerobic exercise improves cognitive function in teens and these beneficial effects are associated with serum levels of BDNF and VEGF. No such association was found for IGF-1. |
| Cross-sectional study [35] | 22 M and F 65–85 years old (median age: 77 years) | Temporo-spatial orientation, memory, attention, calculation capacity, language, and pragmatic praxia | Questionnaire about physical activity and performance | IGF-1 | No association between physical activity and GH or IGF-1 levels were demonstrated. There is a direct relationship between cognitive function and IGF-1 plasma levels in aged subjects with cognitive impairment. |
| RCT [36] | 60 M Exercise status was not mentioned 20–29 years old | Executive function, inhibitory control and attention | Acute resistance exercise | IGF-1 | The beneficial effects of resistance exercise on cognitive function might be explained by changes in arousal state. The change could possibly be modulated by serum cortisol levels. |
| Cross-sectional study [37] | 114 M and F Physically active 60+ years Old (mean age: 66 years) | Executive function, visuo- construction, concentration/attention, language, and memory | Regular aerobic exercise | IGF-1 | Extensive aerobic training might be beneficial in maintaining cognitive function in older age. BDNF and IGF-1 was not found to be associated with duration of daily exercise and no differences in the basal levels of BDNF and IGF-1 between exercise and control group was found. |
| Reference | Subjects | Target Area | Type of PA | (Neuro) Peptide | Main Results |
|---|---|---|---|---|---|
| Controlled trial [38] | 12 M Healthy Exercise status was not mentioned 19–29 years old No fasted state | Levels of plasma acylated ghrelin | Acute sprint interval and endurance exercise | Ghrelin | Sprint interval exercises lead to greater suppression in plasma acylated ghrelin levels. |
| Randomized crossover [39] | 9 M Healthy Physically active 19–25 years old Fasted state | Levels of plasma acylated ghrelin | Acute aerobic exercises | Ghrelin | Single sessions of aerobic exercises lead to lower plasma levels of acylated ghrelin. |
| Randomized crossover [40] | 11 M Healthy Physically active 19–23 years old Fasted state | Levels of plasma acylated ghrelin | Acute aerobic and resistance exercises | Ghrelin | Aerobic and resistance exercises lead to lower plasma levels of acylated ghrelin. |
| PA intervention, no control group [41] | 5 M Healthy Sedentary 26 ± 1 years old Fasted state | Levels of plasma ghrelin | Acute incremental exercise until exhaustion | Ghrelin | Acute incremental exercise lads to decrease in plasma ghrelin levels. |
| Randomized crossover [42] | 12 M overweight Sedentary 48 ± 5 years old Fasted state | Levels of plasma acylated ghrelin | Acute aerobic, strength and combined (aerobic and strength) exercises | Ghrelin | Single sessions of aerobic exercises lead to lower plasma levels of acylated ghrelin. Strength and combined exercises did not alter acylated ghrelin levels. |
| Randomized crossover [43] | 18 M and F Healthy Physically active 19–32 years old Fasted state | Levels of total plasma ghrelin | Acute aerobic exercises | Ghrelin | Aerobic exercise did not alter total ghrelin levels. |
| Counter-balanced [44] | 9 M Healthy Physically active 25 ± 1 years old Fasted state | Levels of plasma ghrelin | Acute resistance exercises | Ghrelin | Eccentric resistance exercises did not alter ghrelin levels and concentric resistance exercises suppressed ghrelin levels. |
| Randomized counter-balanced [45] | 10 M Physically active 21 ± 1 years old Fasted state | Levels of plasma active ghrelin | Acute aerobic and resistance exercises | Ghrelin | Resistance exercises suppress plasma active ghrelin levels. Aerobic exercise did not alter plasma active ghrelin levels. |
| Longitudinal study [46] | 22 M and F Overweight Sedentary 37 ± 8 years old Fasted state | Levels of plasma acylated ghrelin | Regular aerobic exercises | Ghrelin | Long-term aerobic training program results in slight increase in plasma acylated ghrelin levels. |
| RCT [47] | 33 M overweight Sedentary 49 ± 7 years old Fasted and postprandial state | Levels of plasma active ghrelin | Regular aerobic and regular resistance exercises | Ghrelin | Aerobic and resistance exercises result in no change in acylated ghrelin levels in fasted and postprandial state. |
| Randomized trial [48] | 20 M Healthy Sedentary 33 ± 2 years old Fasted state | Levels of plasma active ghrelin | Regular aerobic exercises in normoxic and normobaric hypoxic conditions | Ghrelin | 4-week program of aerobic exercises in normoxic and normobaric hypoxic conditions result in no change in active ghrelin levels. |
| Reference | Subjects | Target Area | Type of PA | (Neuro)Peptide | Main Results |
|---|---|---|---|---|---|
| Randomized PA intervention, no control group [49] | 24 M Physically active 22–24 years old military cadets | Levels of plasma VIP | 5-day period of strenuous exercise (35% of O2 uptake) | VIP | 2 to 5-fold increase in plasma VIP, peak at day 2. Calorie-compensation and glucose infusion lowered the plasma VIP levels during the exercise period within 30-60 min of ingestion. |
| PA intervention, no control group [50] | 6 M Exercise status was not mentioned 17–31 years old | Levels of plasma VIP | 3 h of mild PA, short term-submaximal and maximal, fasting of 59 h | VIP | Increase of plasma VIP after 3 h of mild exercise and after fasting period. No elevation with short-term submaximal or maximal PA. |
| PA intervention, no control group [51] | 18 M Age and exercise status was not mentioned Healthy | NPY release from the myocardial tissue during exercise | Supine position ergometer exercise | NPY | Small increase of NPY and NA release was detected during normoxic exercise, in hypoxic conditions 4 (NPY) and 2 (NA)–fold increases were detected. |
| PA intervention, no control group [52] | 12 M Physically active 22 ± 3 years old elite rowers | Levels of plasma NPY | High-volume low intensity resistance training combined with endurance training for 2 weeks. | NPY | The post-exercise plasma NPY levels increased significantly with the 2-week training period but the levels normalized after 1-week of recovery. |
| PA intervention, no control group [53] | 16 M Physically active 18–45 years old long distance swimmers | Levels of plasma NPY | 25 km sea swimming competition (mean completion time 8,5 h) | NPY | Plasma NPY significantly increased. The level of plasma NPY app. doubled with swimming. |
| Between-group design [54] | 10 F 66–81 years old And 6 M and F 21–46 years old Exercise status was not mentioned | Levels of plasma CGRP | Acute aerobic exercises | CGRP | Aerobic exercise increases the plasma CGRP levels. |
| PA intervention, no control group [55] | 9 M Physically active 41–50 years old runners | Levels of plasma CGRP | Long-term aerobic exercises | CGRP | Aerobic exercise increases the plasma CGRP levels. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Autio, J.; Stenbäck, V.; Gagnon, D.D.; Leppäluoto, J.; Herzig, K.-H. (Neuro) Peptides, Physical Activity, and Cognition. J. Clin. Med. 2020, 9, 2592. https://doi.org/10.3390/jcm9082592
Autio J, Stenbäck V, Gagnon DD, Leppäluoto J, Herzig K-H. (Neuro) Peptides, Physical Activity, and Cognition. Journal of Clinical Medicine. 2020; 9(8):2592. https://doi.org/10.3390/jcm9082592
Chicago/Turabian StyleAutio, Juho, Ville Stenbäck, Dominique D. Gagnon, Juhani Leppäluoto, and Karl-Heinz Herzig. 2020. "(Neuro) Peptides, Physical Activity, and Cognition" Journal of Clinical Medicine 9, no. 8: 2592. https://doi.org/10.3390/jcm9082592
APA StyleAutio, J., Stenbäck, V., Gagnon, D. D., Leppäluoto, J., & Herzig, K.-H. (2020). (Neuro) Peptides, Physical Activity, and Cognition. Journal of Clinical Medicine, 9(8), 2592. https://doi.org/10.3390/jcm9082592






