Preparation of Layer-by-Layer Nanofiltration Membranes by Dynamic Deposition and Crosslinking
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
2.2. Preparation of Membranes
2.2.1. Support Membrane Modification
2.2.2. Multilayer Polyelectrolyte Membrane Preparation by Static Adsorption
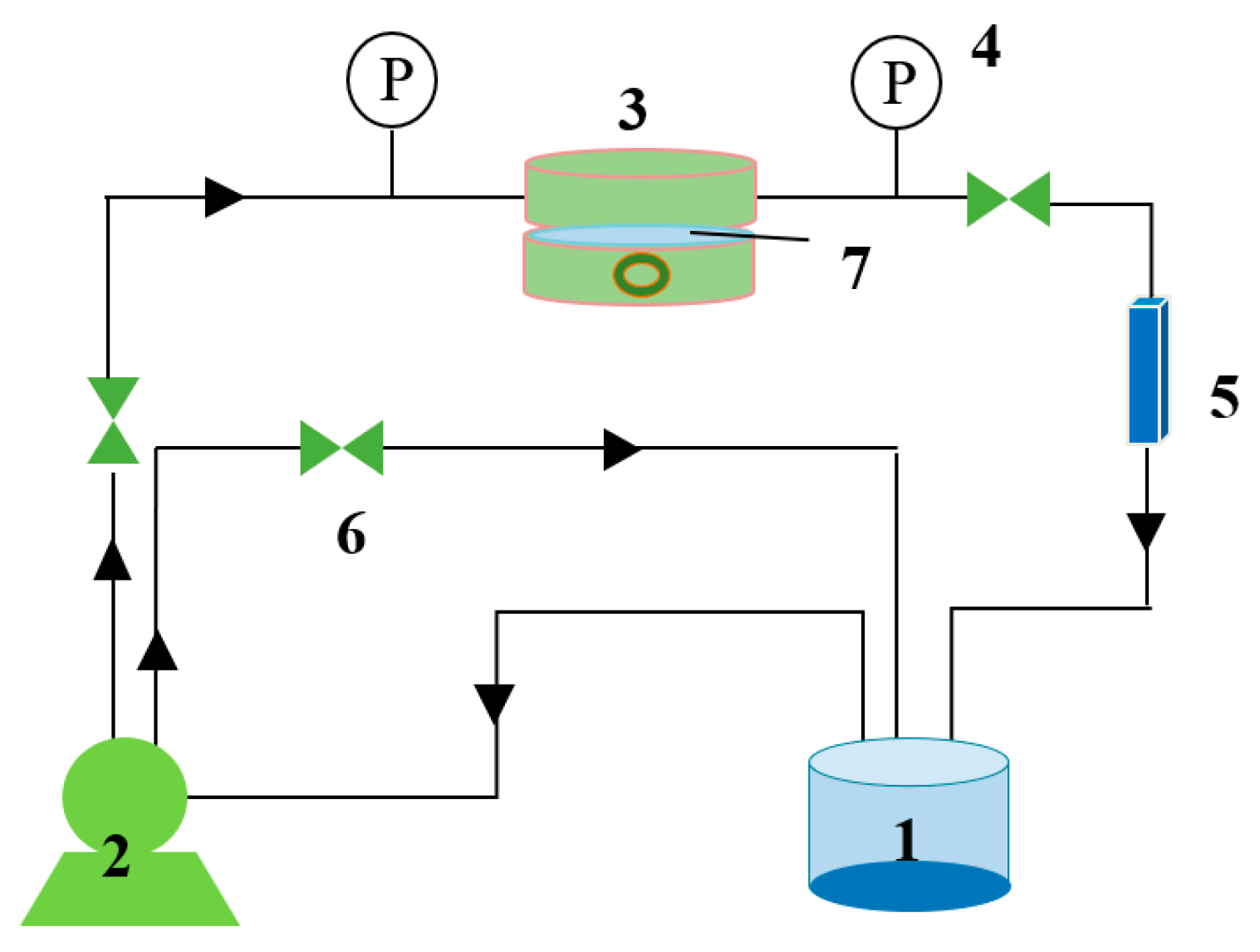
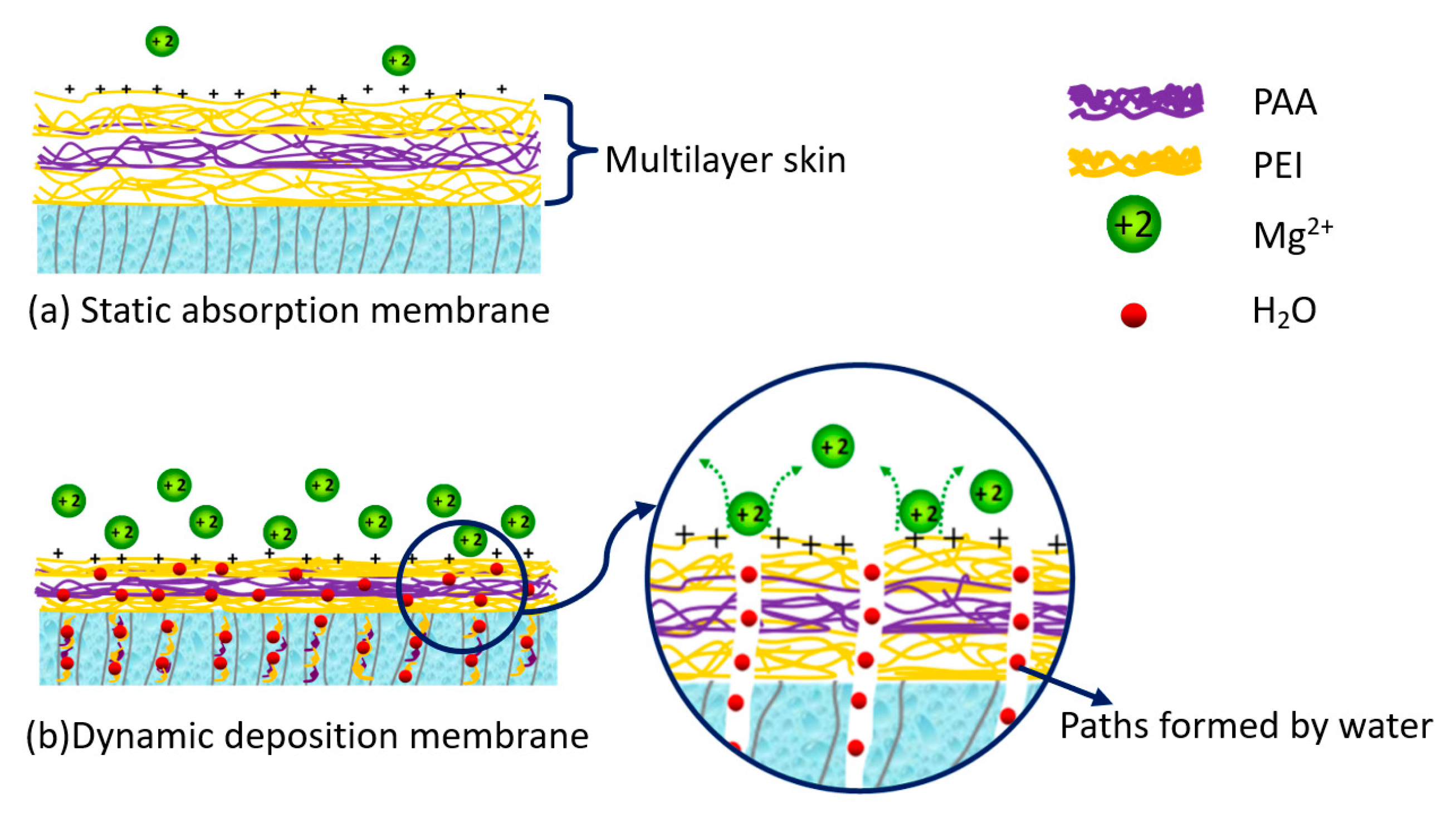
2.2.3. Multilayer Polyelectrolyte Membrane Preparation by Dynamic Deposition
2.2.4. Cross-Linking
2.3. Surface Characterization
2.4. Membrane Performance Test
3. Results and Discussion
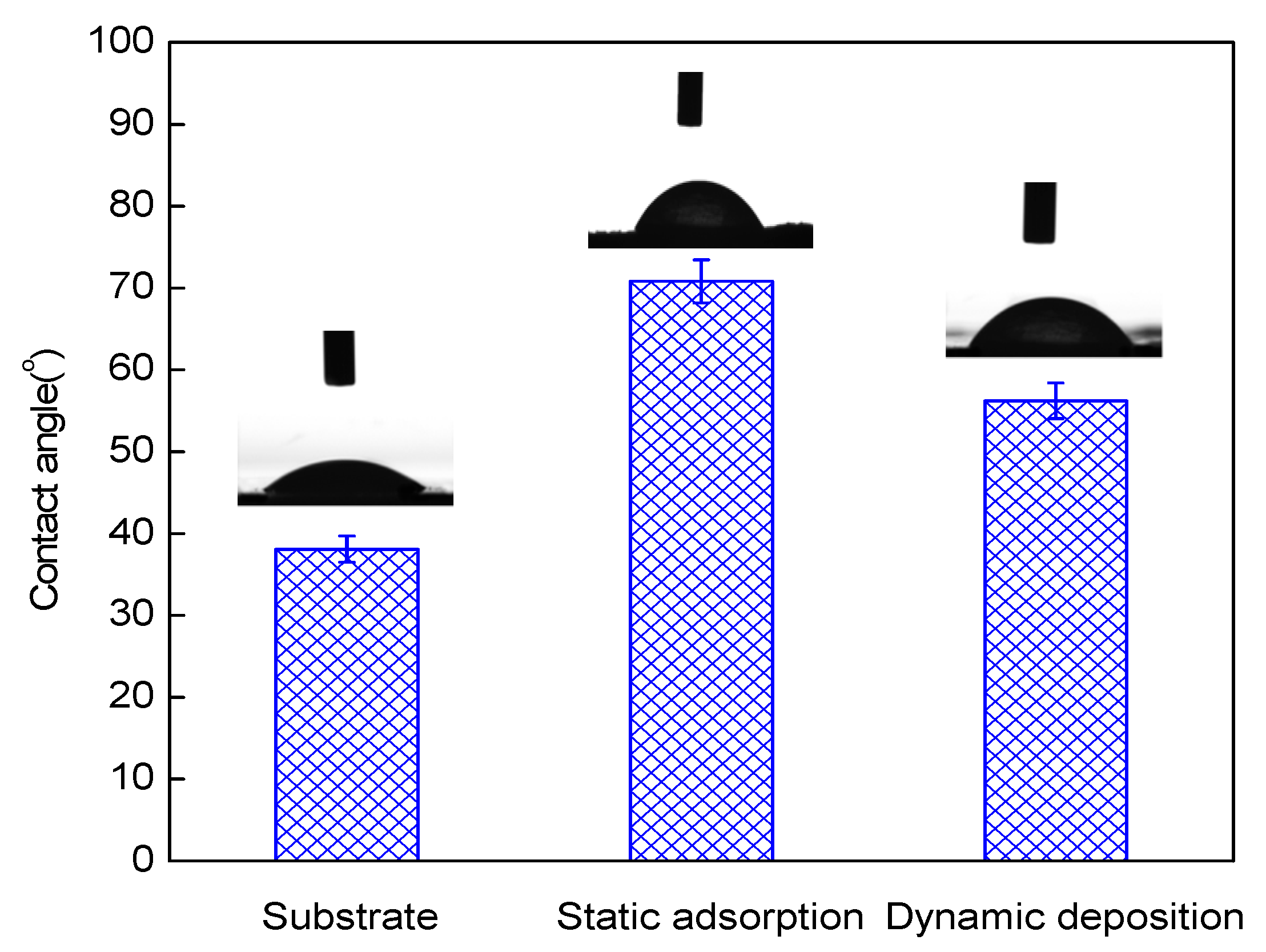
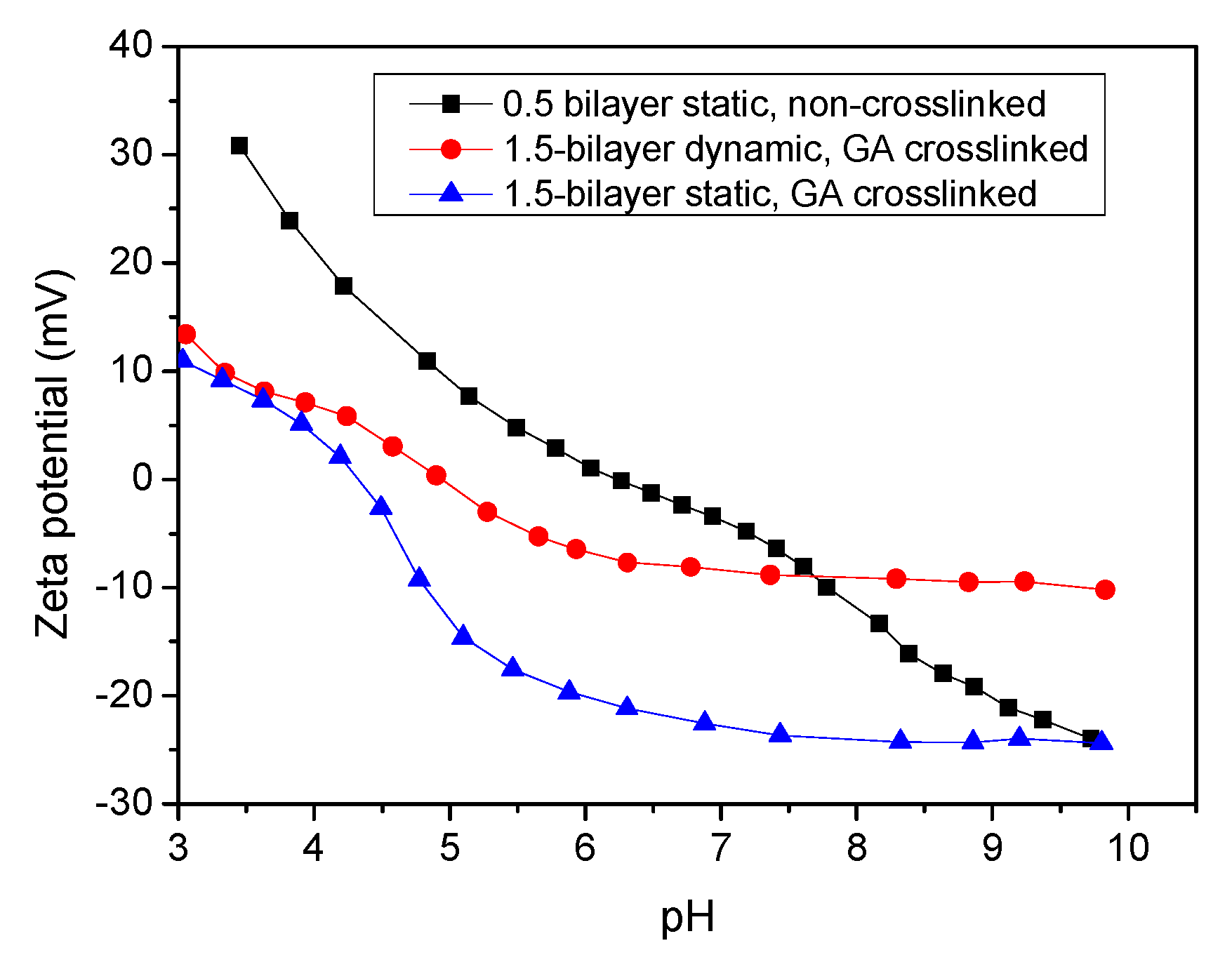
3.1. Characteristics of the Multilayer Membranes
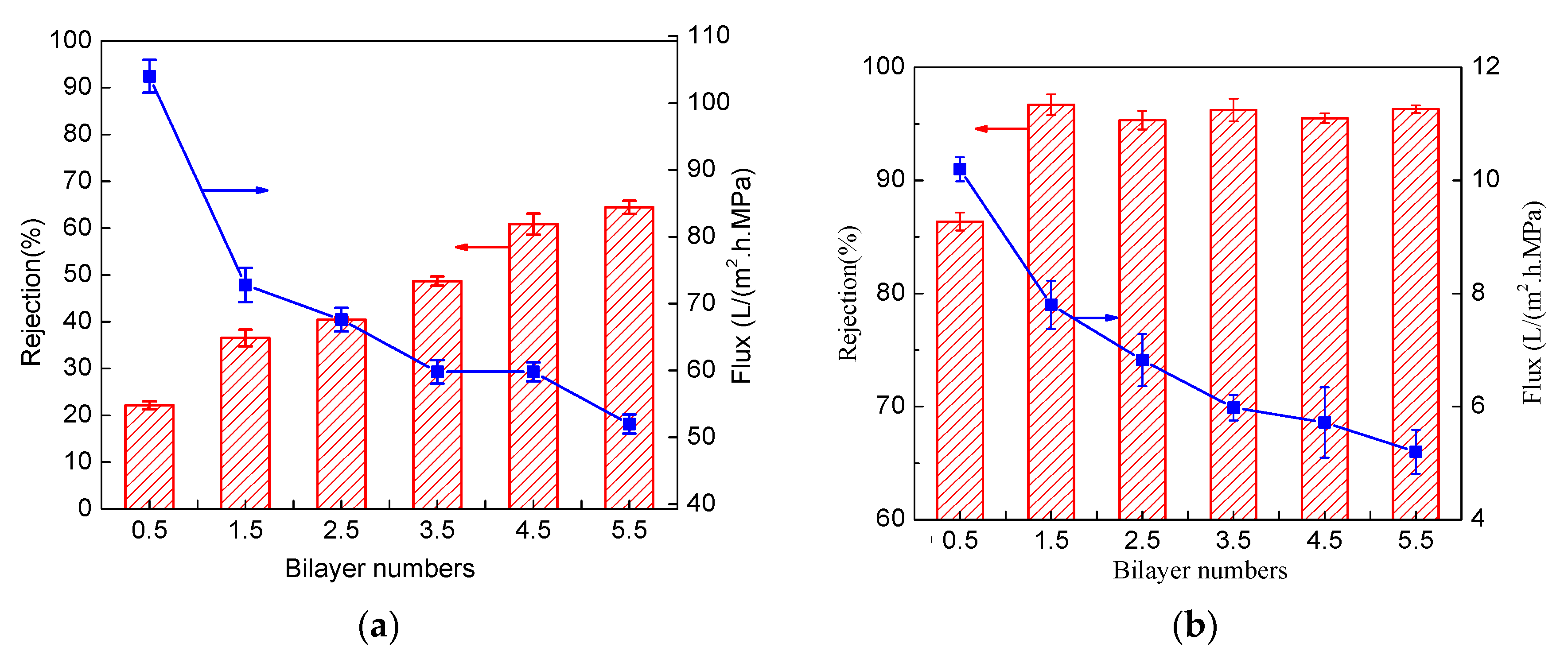
3.2. Effect of Cross-Linking on Separation Performance
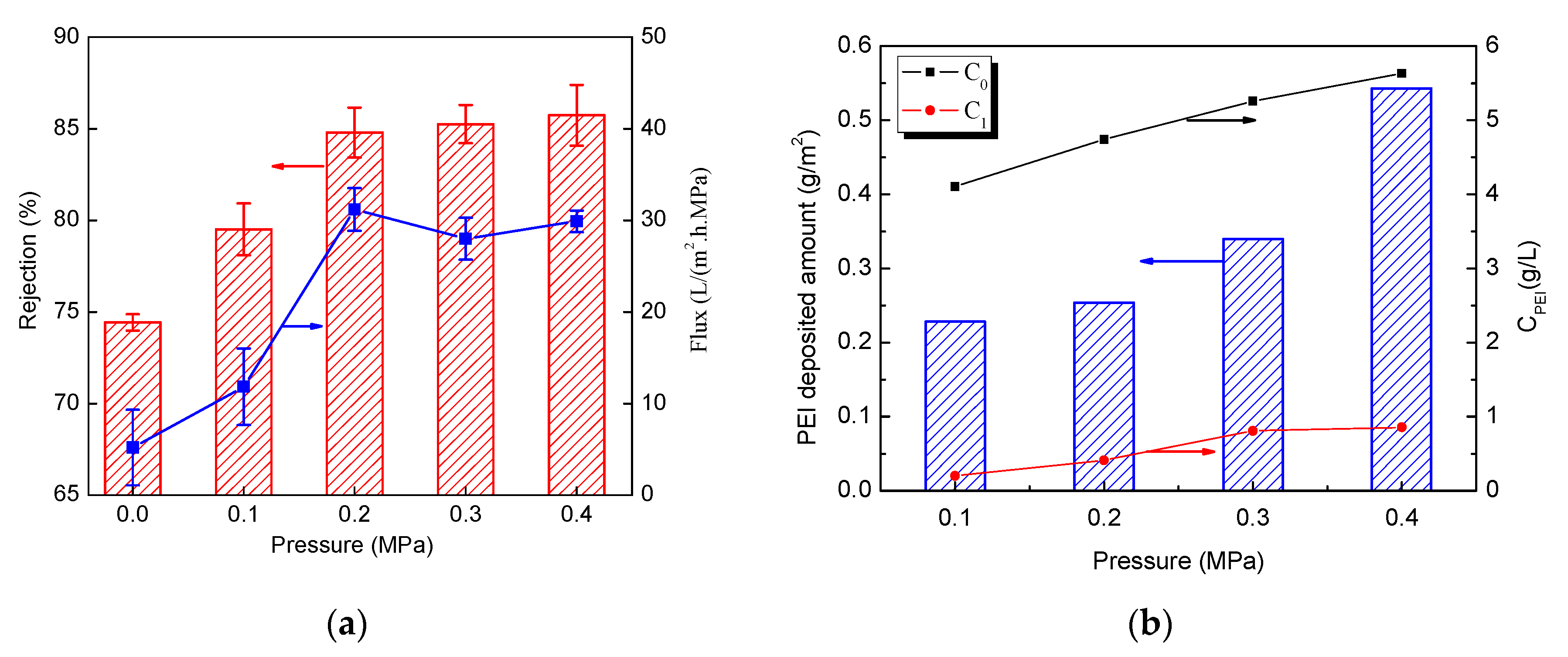
3.3. Effect of Pressure on Separation Performance of Membranes
3.4. Retention of Different Salts
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhou, D.; Zhu, L.; Fu, Y.; Zhu, M.; Xue, L. Development of lower cost seawater desalination processes using nanofiltration technologies—A review. Desalination 2015, 376, 109–116. [Google Scholar] [CrossRef]
- Agboola, O.; Maree, J.; Kolesnikov, A.; Mbaya, R.; Sadiku, R. Theoretical performance of nanofiltration membranes for wastewater treatment. Environ. Chem. Lett. 2015, 13, 37–47. [Google Scholar] [CrossRef]
- Mahlangu, T.O.; Msagati, T.A.M.; Hoek, E.M.V.; Verliefde, A.R.D.; Mamba, B.B. Rejection of pharmaceuticals by nanofiltration (NF) membranes: Effect of fouling on rejection behaviour. Phys. Chem. Earth 2014, 76–78, 28–34. [Google Scholar]
- Marchetti, P.; Solomon, M.F.J.; Szekely, G.; Livingston, A.G. Molecular Separation with Organic Solvent Nanofiltration: A Critical Review. Chem. Rev. 2014, 114, 10735–10806. [Google Scholar] [CrossRef] [PubMed]
- Salehi, F. Current and future applications for nanofiltration technology in the food processing. Food Bioprod. Process. 2014, 92, 161–177. [Google Scholar] [CrossRef]
- Feng, C.; Xu, J.; Li, M.; Tang, Y.; Gao, C. Studies on a novel nanofiltration membrane prepared by cross-linking of polyethyleneimine on polyacrylonitrile substrate. J. Membr. Sci. 2014, 451, 103–110. [Google Scholar] [CrossRef]
- Wu, X.; Hao, L.; Zhang, J.; Zhang, X.; Wang, J.; Liu, J. Polymer-Ti3C2Tx composite membranes to overcome the trade-off in solvent resistant nanofiltration for alcohol-based system. J. Membr. Sci. 2016, 515, 175–188. [Google Scholar] [CrossRef]
- Khoonsap, S.; Rugmai, S.; Hung, W.S.; Lee, K.R.; Klinsrisuk, S.; Amnuaypanich, S. Promoting Permeability-Selectivity Anti-Trade-Off Behavior in Polyvinyl alcohol (PVA) Nanocomposite Membranes. J. Membr. Sci. 2017, 544, 287–296. [Google Scholar] [CrossRef]
- Labban, O.; Liu, C.; Chong, T.H.; John, H.L.V. Relating Transport Modeling to Nanofiltration Membrane Fabrication: Navigating the Permeability-Selectivity Trade-off in Desalination Pretreatment. J. Membr. Sci. 2018, 554, 26–38. [Google Scholar] [CrossRef]
- Liu, Z.; Kuang, W.; Kang, G.; Yu, H.; Jin, Y.; Zhou, M.; Liu, D.; Cao, Y. Preparation and characterization of a composite nanofiltration membrane interfacially polymerized from cis,cis-1,3,5-triaminocyclohexane and trimesoyl chloride. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Yang, M.; Zhao, C.; Zhang, S.; Li, P.; Hou, D. Preparation of graphene oxide modified poly(m-phenylene isophthalamide) nanofiltration membrane with improved water flux and antifouling property. Appl. Surf. Sci. 2016, 394, 149–159. [Google Scholar] [CrossRef]
- Mustafa, G.; Wyns, K.; Buekenhoudt, A.; Meynen, V. New insights into the fouling mechanism of dissolved organic matter applying nanofiltration membranes with a variety of surface chemistries. Water. Res. 2016, 93, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Zhang, Q.; Zheng, J.; Hou, S.; Mao, H.; Zhang, S. Green fabrication of a positively charged nanofiltration membrane by grafting poly(ethylene imine) onto a poly (arylene ether sulfone) membrane containing tertiary amine groups. J. Membr. Sci. 2016, 517, 39–46. [Google Scholar] [CrossRef]
- Yan, F.; Chen, H.; Yang, L.; Lü, Z.; Yu, S.; Liu, M.; Gao, C. Improving the water permeability and antifouling property of thin-film composite polyamide nanofiltration membrane by modifying the active layer with triethanolamine. J. Membr. Sci. 2016, 513, 108–116. [Google Scholar] [CrossRef]
- Decher, G. Toward Layered Polymeric Multicomposites. Science 1997, 227, 1232–1237. [Google Scholar] [CrossRef]
- Richardson, J.J.; Björnmalm, M.; Caruso, F. Multilayer assembly. Technology-driven layer-by-layer assembly of nanofilms. Science 2015, 348, aaa2491. [Google Scholar]
- El-Hashani, A.; Toutianoush, A.; Tieke, B. Use of layer-by-layer assembled ultrathin membranes of dicopper-[18]azacrown-N 6 complex and polyvinylsulfate for water desalination under nanofiltration conditions. J. Membr. Sci. 2008, 318, 65–70. [Google Scholar] [CrossRef]
- Hong, S.U.; Malaisamy, R.; Bruening, M.L. Optimization of flux and selectivity in Cl−/SO42− separations with multilayer polyelectrolyte membranes. J. Membr. Sci. 2006, 283, 366–372. [Google Scholar] [CrossRef]
- Lu, O.; Malaisamy, R.; Bruening, M.L. Multilayer polyelectrolyte films as nanofiltration membranes for separating monovalent and divalent cations. J. Membr. Sci. 2008, 310, 76–84. [Google Scholar]
- Hong, S.U.; Bruening, M.L. Separation of amino acid mixtures using multilayer polyelectrolyte nanofiltration membranes. J. Membr. Sci. 2006, 280, 1–5. [Google Scholar]
- Deng, H.Y.; Xu, Y.Y.; Zhu, B.K.; Wei, X.Z.; Liu, F.; Cui, Z.Y. Polyelectrolyte membranes prepared by dynamic self-assembly of poly (4-styrenesulfonic acid- co -maleic acid) sodium salt (PSSMA) for nanofiltration (I). J. Membr. Sci. 2008, 323, 125–133. [Google Scholar] [CrossRef]
- Zhang, G.; Gu, W.; Ji, S.; Liu, Z.; Peng, Y.; Wang, Z. Preparation of polyelectrolyte multilayer membranes by dynamic layer-by-layer process for pervaporation separation of alcohol/water mixtures. J. Membr. Sci. 2006, 280, 727–733. [Google Scholar] [CrossRef]
- Baowei, S.U.; Wang, T.; Wang, Z.; Gao, X.; Gao, C. Preparation and performance of dynamic layer-by-layer PDADMAC/PSS nanofiltration membrane. J. Membr. Sci. 2012, 423, 324–331. [Google Scholar]
- Liu, C.; Shi, L.; Wang, R. Crosslinked layer-by-layer polyelectrolyte nanofiltration hollow fiber membrane for low-pressure water softening with the presence of SO42− in feed water. J. Membr. Sci. 2015, 486, 169–176. [Google Scholar] [CrossRef]
- Qiu, C.; Qi, S.; Tang, C.Y. Synthesis of high flux forward osmosis membranes by chemically crosslinked layer-by-layer polyelectrolytes. J. Membr. Sci. 2011, 381, 74–80. [Google Scholar] [CrossRef]
- Zheng, Y.; Yao, G.; Cheng, Q.; Yu, S.; Liu, M.; Gao, C. Positively charged thin-film composite hollow fiber nanofiltration membrane for the removal of cationic dyes through submerged filtration. Desalination 2013, 328, 42–50. [Google Scholar] [CrossRef]
- Saeki, D.; Imanishi, M.; Ohmukai, Y.; Maruyama, T.; Matsuyama, H. Stabilization of layer-by-layer assembled nanofiltration membranes by crosslinking via amide bond formation and siloxane bond formation. J. Membr. Sci. 2013, 447, 128–133. [Google Scholar]
- Xu, J.; Xu, L.; Xu, H.; Sun, F.; Gao, X.; Gao, C. Stability and permeation behavior of a porous membrane modified by polyelectrolyte networks enabled by electro-deposition and cross-linking for water purification. J. Membr. Sci. 2015, 496, 21–30. [Google Scholar]
- Nan, Q.; Li, P.; Cao, B. Fabrication of positively charged nanofiltration membrane via the layer-by-layer assembly of graphene oxide and polyethylenimine for desalination. Appl. Surf. Sci. 2016, 387, 521–528. [Google Scholar] [CrossRef]
- Huang, F.; Wei, Q.; Cai, Y.; Wu, N. Surface Structures and Contact Angles of Electrospun Poly(vinylidene fluoride) Nanofiber Membranes. Int. J. Polym. Anal. Charact. 2008, 13, 292–301. [Google Scholar] [CrossRef]
- Zhang, Y.; Guo, M.; Pan, G.; Yan, H.; Xu, J.; Shi, Y.; Shi, H.; Liu, Y. Preparation and properties of novel pH-stable TFC membrane based on organic–inorganic hybrid composite materials for nanofiltration. J. Membr. Sci. 2015, 476, 500–507. [Google Scholar]
- Zhu, Z.; Jiang, J.; Wang, X.; Huo, X.; Xu, Y.; Li, Q.; Wang, L. Improving the hydrophilic and antifouling properties of polyvinylidene fluoride membrane by incorporation of novel nanohybrid GO@SiO2 particles. Chem. Eng. J. 2017, 314, 266–276. [Google Scholar] [CrossRef]
- Böhme, F.; Klinger, C.; Bellmann, C. Surface properties of polyamidines. Colloid. Surf. A 2001, 189, 21–27. [Google Scholar] [CrossRef]
- Ahmadiannamini, P.; Bruening, M.L.; Tarabara, V.V. Sacrificial polyelectrolyte multilayer coatings as an approach to membrane fouling control: Disassembly and regeneration mechanisms. J. Membr. Sci. 2015, 491, 149–158. [Google Scholar] [CrossRef]
- Menne, D.; Üzüm, C.; Koppelmann, A.; Wong, J.E.; Foeken, C.V.; Borre, F.; Dähne, L.; Laakso, T.; Pihlajamäki, A.; Wessling, M. Regenerable polymer/ceramic hybrid nanofiltration membrane based on polyelectrolyte assembly by layer-by-layer technique. J. Membr. Sci. 2016, 520, 924–932. [Google Scholar]
- Weng, X.D.; Ji, Y.L.; Ma, R.; Zhao, F.Y.; An, Q.F.; Gao, C.J. Superhydrophilic and antibacterial zwitterionic polyamide nanofiltration membranes for antibiotics separation. J. Membr. Sci. 2016, 510, 122–130. [Google Scholar] [CrossRef]
- Jin, W.; Toutianoush, A.; Tieke, B. Use of polyelectrolyte layer-by-layer assemblies as nanofiltration and reverse osmosis membranes. Langmuir 2003, 19, 2550–2553. [Google Scholar] [CrossRef]
- Chen, Q.; Yu, P.; Huang, W.; Yu, S.; Liu, M.; Gao, C. High-flux composite hollow fiber nanofiltration membranes fabricated through layer-by-layer deposition of oppositely charged crosslinked polyelectrolytes for dye removal. J. Membr. Sci. 2015, 492, 312–321. [Google Scholar] [CrossRef]


| Static-PW | Static-R | Dynamic-PW | Dynamic-R | |
|---|---|---|---|---|
| Flux (L/(m2·h·MPa)) | 31.2 | 16.8 | 20.9 | 22.7 |
| Rejection (%) | 83.2 | 74.0 | 96.5 | 95.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Chen, G.Q.; Yang, X.; Deng, H. Preparation of Layer-by-Layer Nanofiltration Membranes by Dynamic Deposition and Crosslinking. Membranes 2019, 9, 20. https://doi.org/10.3390/membranes9020020
Liu Y, Chen GQ, Yang X, Deng H. Preparation of Layer-by-Layer Nanofiltration Membranes by Dynamic Deposition and Crosslinking. Membranes. 2019; 9(2):20. https://doi.org/10.3390/membranes9020020
Chicago/Turabian StyleLiu, Yan, George Q. Chen, Xiuli Yang, and Huining Deng. 2019. "Preparation of Layer-by-Layer Nanofiltration Membranes by Dynamic Deposition and Crosslinking" Membranes 9, no. 2: 20. https://doi.org/10.3390/membranes9020020
APA StyleLiu, Y., Chen, G. Q., Yang, X., & Deng, H. (2019). Preparation of Layer-by-Layer Nanofiltration Membranes by Dynamic Deposition and Crosslinking. Membranes, 9(2), 20. https://doi.org/10.3390/membranes9020020






