Potent Neutralization Antibodies Induced by a Recombinant Trimeric Spike Protein Vaccine Candidate Containing PIKA Adjuvant for COVID-19
Abstract
1. Introduction
2. Methods
2.1. Expression and Purification of the Recombinant SARS-CoV-2 Spike Protein Trimer and Spike Protein Monomer
2.2. Vaccination in Mice
2.3. S-Protein-Specific Antibody Determined by ELISA
2.4. Protein/Peptide Array
2.5. SARS-CoV-2 Pseudo-Virus Production
2.6. Neutralization of Serum Antibody against Pseudo-Virus Infection
3. Results
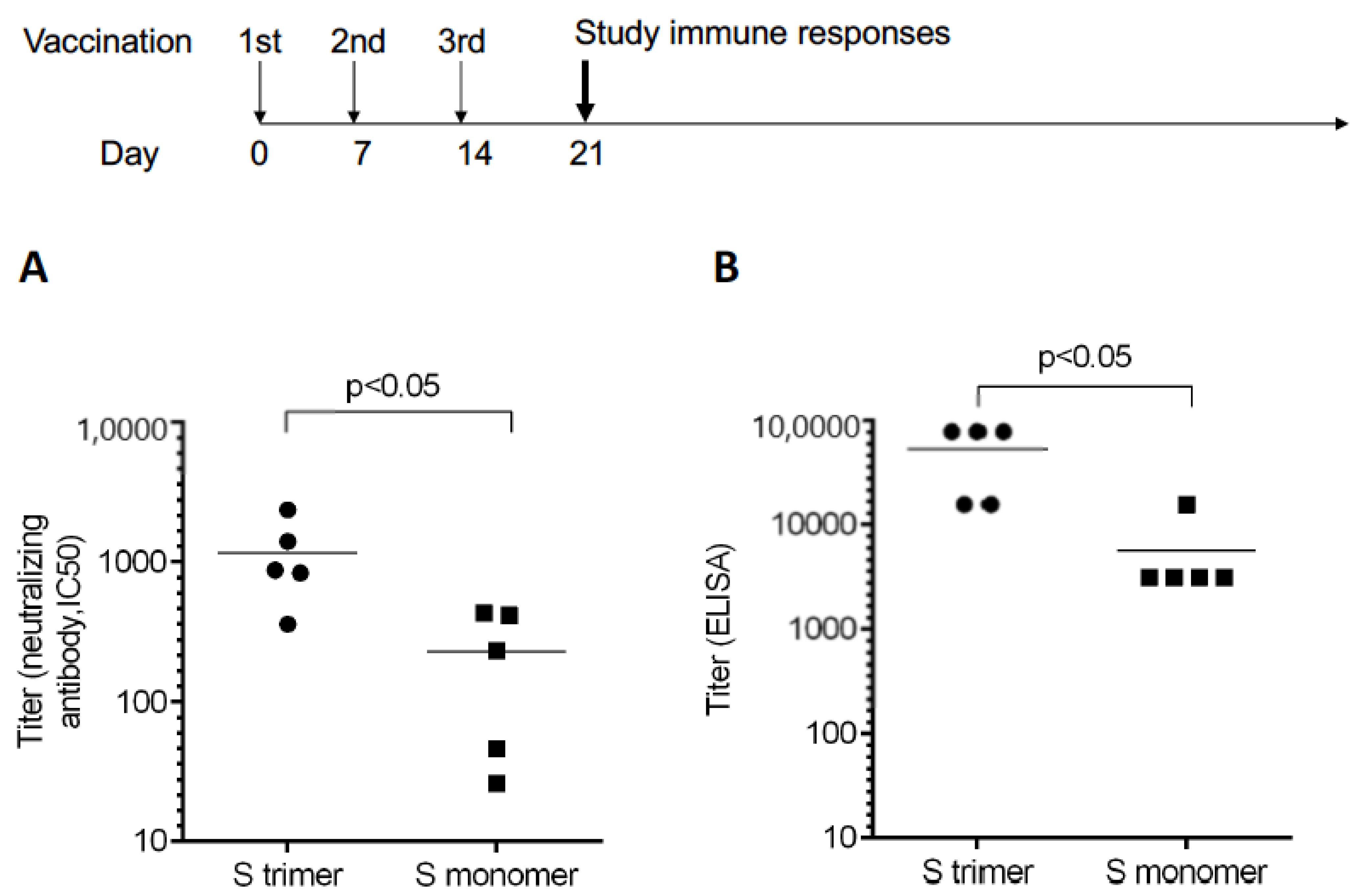
3.1. Trimeric S Protein Induced Higher Neutralizing Antibodies
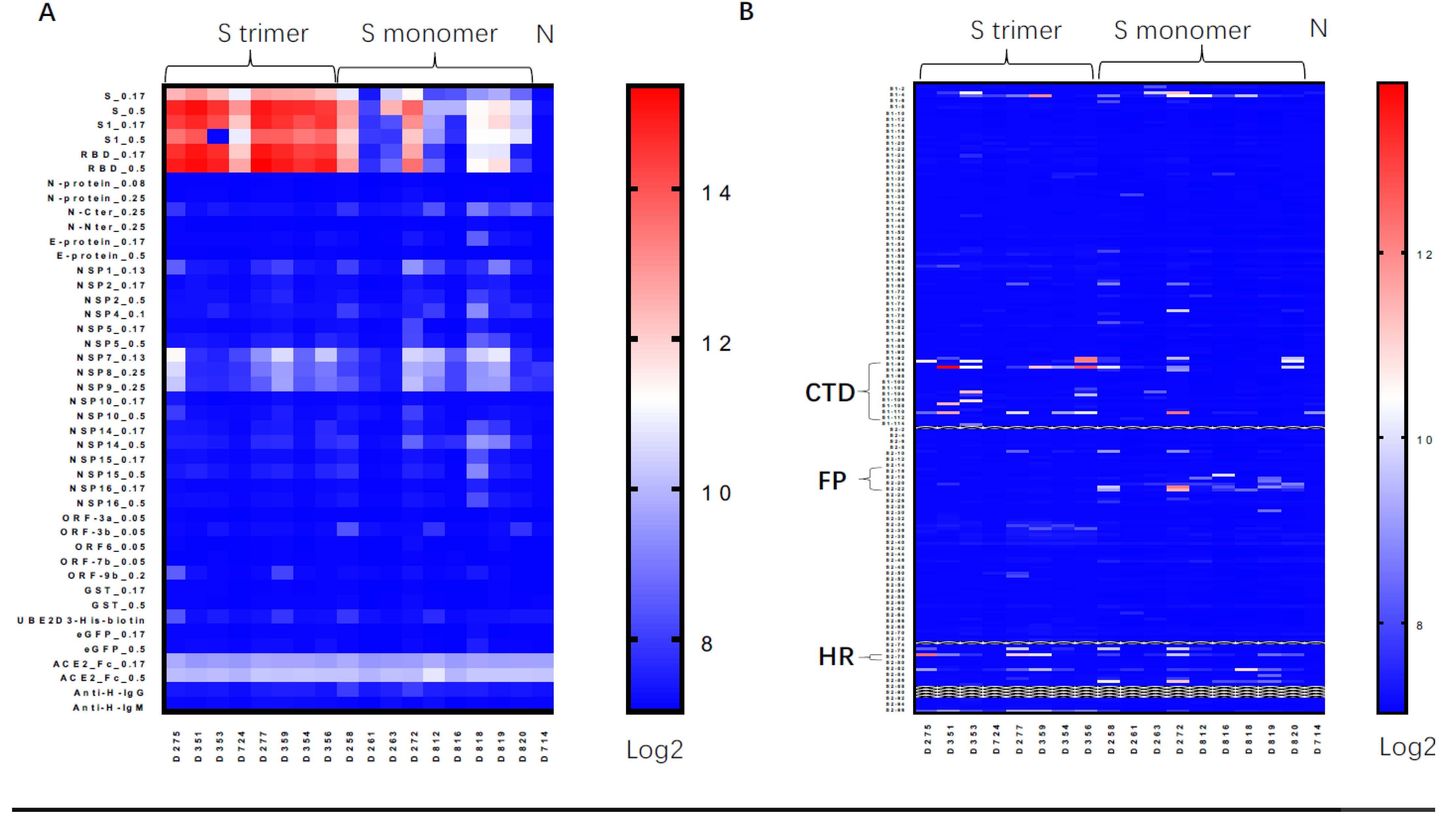
3.2. Trimeric S Protein Induced Similar Epitope Patterns as Natural Infection
4. Discussion
4.1. The Neutralizing Titer of Recombinant Trimeric Spike Protein Containing PIKA Adjuvant
4.2. The Neutralizing versus Binding Ratio of Vaccine-Induced Antibodies
4.3. The Epitope Map of Antibody Induced by Vaccines versus Natural Infection
4.4. The Efficacy of PIKA Adjuvant
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, Z.; Hu, Y.; Xu, M.; Chen, Z.; Yang, W.; Jiang, Z.; Li, M.; Jin, H.; Cui, G.; Chen, P.; et al. Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine (CoronaVac) in healthy adults aged 60 years and older: A randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet Infect Dis. 2021. [Google Scholar] [CrossRef]
- Zhu, F.C.; Li, Y.H.; Guan, X.H.; Hou, L.H.; Wang, W.J.; Li, J.X.; Wu, S.P.; Wang, B.S.; Wang, Z.; Wang, L.; et al. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: A dose-escalation, open-label, non-randomised, first-in-human trial. Lancet 2020, 395, 1845–1854. [Google Scholar] [CrossRef]
- Folegatti, P.M.; Ewer, K.J.; Aley, P.K.; Angus, B.; Becker, S.; Belij-Rammerstorfer, S.; Bellamy, D.; Bibi, S.; Bittaye, M.; Clutterbuck, E.A.; et al. Safety and immunogenicity of the ChAdOx1 nCoV-19 vaccine against SARS-CoV-2: A preliminary report of a phase 1/2, single-blind, randomised controlled trial. Lancet 2020, 396, 467–478. [Google Scholar] [CrossRef]
- Logunov, D.Y.; Dolzhikova, I.V.; Zubkova, O.V.; Tukhvatulin, A.I.; Shcheblyakov, D.V.; Dzharullaeva, A.S.; Grousova, D.M.; Erokhova, A.S.; Kovyrshina, A.V.; Botikov, A.G.; et al. Safety and immunogenicity of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine in two formulations: Two open, non-randomised phase 1/2 studies from Russia. Lancet 2020, 396, 887–897. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Jackson, L.A.; Anderson, E.J.; Rouphael, N.G.; Roberts, P.C.; Makhene, M.; Coler, R.N.; McCullough, M.P.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; et al. An mRNA Vaccine against SARS-CoV-2—Preliminary Report. N. Engl. J. Med. 2020, 383, 1920–1931. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.N.; Li, X.F.; Deng, Y.Q.; Zhao, H.; Huang, Y.J.; Yang, G.; Huang, W.J.; Gao, P.; Zhou, C.; Zhang, R.R.; et al. A Thermostable mRNA Vaccine against COVID-19. Cell 2020, 182, 1271–1283.e16. [Google Scholar] [CrossRef]
- Zhu, Q.R.; Gu, X.H.; Duan, S.C.; Xu, H.F. Long-term immunogenicity and efficacy of recombinant yeast derived hepatitis B vaccine for interruption of mother-infant transmission of hepatitis B virus. Chin. Med. J. 1994, 107, 915–918. [Google Scholar] [PubMed]
- Tefit, J.N.; Crabé, S.; Orlandini, B.; Nell, H.; Bendelac, A.; Deng, S.; Savage, P.B.; Teyton, L.; Serra, V. Efficacy of ABX196, a new NKT agonist, in prophylactic human vaccination. Vaccine 2014, 32, 6138–6145. [Google Scholar] [CrossRef]
- Miskovsky, E.; Gershman, K.; Clements, M.L.; Cupps, T.; Calandra, G.; Hesley, T.; Ioli, V.; Ellis, R.; Kniskern, P.; Miller, W.; et al. Comparative safety and immunogenicity of yeast recombinant hepatitis B vaccines containing S and pre-S2 + S antigens. Vaccine 1991, 9, 346–350. [Google Scholar] [CrossRef]
- Leroux-Roels, G.; Desombere, I.; De Tollenaere, G.; Petit, M.A.; Desmons, P.; Hauser, P.; Delem, A.; De Grave, D.; Safary, A. Hepatitis B vaccine containing surface antigen and selected preS1 and preS2 sequences. 1. Safety and immunogenicity in young, healthy adults. Vaccine 1997, 15, 1724–1731. [Google Scholar] [CrossRef]
- Van Damme, P.; Bouillette-Marussig, M.; Hens, A.; De Coster, I.; Depuydt, C.; Goubier, A.; Van Tendeloo, V.; Cools, N.; Goossens, H.; Hercend, T.; et al. GTL001, A Therapeutic Vaccine for Women Infected with Human Papillomavirus 16 or 18 and Normal Cervical Cytology: Results of a Phase I Clinical Trial. Clin. Cancer Res. 2016, 22, 3238–3248. [Google Scholar] [CrossRef]
- Luxembourg, A.; Moreira, E.D., Jr.; Samakoses, R.; Kim, K.H.; Sun, X.; Maansson, R.; Moeller, E.; Christiano, S.; Chen, J. Phase III, randomized controlled trial in girls 9-15 years old to evaluate lot consistency of a novel nine-valent human papillomavirus L1 virus-like particle vaccine. Hum. Vaccin. Immunother. 2015, 11, 1306–1312. [Google Scholar] [CrossRef]
- Senders, S.; Bhuyan, P.; Jiang, Q.; Absalon, J.; Eiden, J.J.; Jones, T.R.; York, L.J.; Jansen, K.U.; O’Neill, R.E.; Harris, S.L.; et al. Immunogenicity, Tolerability and Safety in Adolescents of Bivalent rLP2086, a Meningococcal Serogroup B Vaccine, Coadministered with Quadrivalent Human Papilloma Virus Vaccine. Pediatr. Infect. Dis J. 2016, 35, 548–554. [Google Scholar] [CrossRef]
- Lehtinen, M.; Apter, D.; Baussano, I.; Eriksson, T.; Natunen, K.; Paavonen, J.; Vänskä, S.; Bi, D.; David, M.P.; Datta, S.; et al. Characteristics of a cluster-randomized phase IV human papillomavirus vaccination effectiveness trial. Vaccine 2015, 33, 1284–1290. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Ebihara, K.; Tanaka, Y.; Noda, K. Efficacy of quadrivalent human papillomavirus (types 6, 11, 16 and 18) vaccine(GARDASIL) in Japanese women aged 18–26 years. Cancer Sci. 2013, 104, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Ward, B.J.; Makarkov, A.; Séguin, A.; Pillet, S.; Trépanier, S.; Dhaliwall, J.; Libman, M.D.; Vesikari, T.; Landry, N. Efficacy, immunogenicity, and safety of a plant-derived, quadrivalent, virus-like particle influenza vaccine in adults (18–64 years) and older adults (65 years): Two multicentre, randomised phase 3 trials. Lancet 2020, 396, 1491–1503. [Google Scholar] [CrossRef]
- Corder, B.N.; Bullard, B.L.; Poland, G.A.; Weaver, E.A. A Decade in Review: A Systematic Review of Universal Influenza Vaccines in Clinical Trials during the 2010 Decade. Viruses 2020, 12, 1186. [Google Scholar] [CrossRef] [PubMed]
- Champion, C.R. Heplisav-B: A Hepatitis B Vaccine with a Novel Adjuvant. Ann Pharmacother. 2020. [Google Scholar] [CrossRef]
- Schillie, S.; Harris, A.; Link-Gelles, R.; Romero, J.; Ward, J.; Nelson, N. Recommendations of the Advisory Committee on Immunization Practices for Use of a Hepatitis B Vaccine with a Novel Adjuvant. MMWR Morb. Mortal Wkly. Rep. 2018, 67, 455–458. [Google Scholar] [CrossRef] [PubMed]
- Cooper, C.L.; Davis, H.L.; Morris, M.L.; Efler, S.M.; Adhami, M.A.; Krieg, A.M.; Cameron, D.W.; Heathcote, J. CPG 7909, an immunostimulatory TLR9 agonist oligodeoxynucleotide, as adjuvant to Engerix-B HBV vaccine in healthy adults: A double-blind phase I/II study. J. Clin. Immunol. 2004, 24, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Halperin, S.A.; Van Nest, G.; Smith, B.; Abtahi, S.; Whiley, H.; Eiden, J.J. A phase I study of the safety and immunogenicity of recombinant hepatitis B surface antigen co-administered with an immunostimulatory phosphorothioate oligonucleotide adjuvant. Vaccine 2003, 21, 2461–2467. [Google Scholar] [CrossRef]
- Garçon, N.; Morel, S.; Didierlaurent, A.; Descamps, D.; Wettendorff, M.; Van Mechelen, M. Development of an AS04-adjuvanted HPV vaccine with the adjuvant system approach. BioDrugs 2011, 25, 217–226. [Google Scholar] [CrossRef]
- Portnoff, A.D.; Patel, N.; Massare, M.J.; Zhou, H.; Tian, J.H.; Zhou, B.; Shinde, V.; Glenn, G.M.; Smith, G. Influenza Hemagglutinin Nanoparticle Vaccine Elicits Broadly Neutralizing Antibodies against Structurally Distinct Domains of H3N2 HA. Vaccines 2020, 8, 99. [Google Scholar] [CrossRef]
- Dunkle, L.M.; Izikson, R.; Patriarca, P.; Goldenthal, K.L.; Muse, D.; Callahan, J.; Cox, M.M.J.; PSC12 Study Team. Efficacy of Recombinant Influenza Vaccine in Adults 50 Years of Age or Older. N. Engl. J. Med. 2017, 376, 2427–2436. [Google Scholar] [CrossRef]
- Atsmon, J.; Caraco, Y.; Ziv-Sefer, S.; Shaikevich, D.; Abramov, E.; Volokhov, I.; Bruzil, S.; Haima, K.Y.; Gottlieb, T.; Ben-Yedidia, T. Priming by a novel universal influenza vaccine (Multimeric-001)-a gateway for improving immune response in the elderly population. Vaccine 2014, 32, 5816–5823. [Google Scholar] [CrossRef]
- Treanor, J.J.; Essink, B.; Hull, S.; Reed, S.; Izikson, R.; Patriarca, P.; Goldenthal, K.L.; Kohberger, R.; Dunkle, L.M. Evaluation of safety and immunogenicity of recombinant influenzahemagglutinin (H5/Indonesia/05/2005) formulated with and without a stable oil-in-water emulsion containing glucopyranosyl-lipid A (SE+GLA) adjuvant. Vaccine 2013, 31, 5760–5765. [Google Scholar] [CrossRef] [PubMed]
- Huber, V.C.; McCullers, J.A. FluBlok, a recombinant influenza vaccine. Curr. Opin. Mol. Ther. 2008, 10, 75–85. [Google Scholar]
- Treanor, J.J.; Schiff, G.M.; Hayden, F.G.; Brady, R.C.; Hay, C.M.; Meyer, A.L.; Holden-Wiltse, J.; Liang, H.; Gilbert, A.; Cox, M. Safety and immunogenicity of a baculovirus-expressed hemagglutinin influenzavaccine: A randomized controlled trial. JAMA 2007, 297, 1577–1582. [Google Scholar] [CrossRef]
- Treanor, J.J.; Schiff, G.M.; Couch, R.B.; Cate, T.R.; Brady, R.C.; Hay, C.M.; Wolff, M.; She, D.; Cox, M.M. Dose-related safety and immunogenicity of a trivalent baculovirus-expressed influenza-virus hemagglutinin vaccine in elderly adults. J. Infect. Dis. 2006, 193, 1223–1228. [Google Scholar] [CrossRef] [PubMed]
- Hauser, M.I.; Muscatello, D.J.; Soh, A.C.Y.; Dwyer, D.E.; Turner, R.M. An indirect comparison meta-analysis of AS03 and MF59 adjuvants in pandemic influenza A(H1N1)pdm09 vaccines. Vaccine 2019, 37, 4246–4255. [Google Scholar] [CrossRef] [PubMed]
- Stassijns, J.; Bollaerts, K.; Baay, M.; Verstraeten, T. A systematic review and meta-analysis on the safety of newly adjuvanted vaccines among children. Vaccine 2016, 34, 714–722. [Google Scholar] [CrossRef]
- Chen, W.H.; Hotez, P.J.; Bottazzi, M.E. Potential for developing a SARS-CoV receptor-binding domain (RBD) recombinant protein as a heterologous human vaccine against coronavirus infectious disease (COVID)-19. Hum. Vaccin. Immunother. 2020, 16, 1239–1242. [Google Scholar] [CrossRef]
- Chen, W.H.; Tao, X.; Agrawal, A.S.; Algaissi, A.; Peng, B.H.; Pollet, J.; Strych, U.; Bottazzi, M.E.; Hotez, P.J.; Lustigman, S.; et al. Yeast-expressed SARS-CoV recombinant receptor-binding domain (RBD219-N1) formulated with aluminum hydroxide induces protective immunity and reduces immune enhancement. Vaccine 2020, 38, 7533–7541. [Google Scholar] [CrossRef]
- Su, Q.D.; Zou, Y.N.; Yi, Y.; Shen, L.P.; Ye, X.Z.; Zhang, Y.; Wang, H.; Ke, H.; Song, J.D.; Hu, K.P.; et al. Recombinant SARS-CoV-2 RBD with a built in T helper epitope induces strong neutralization antibody response. Vaccine 2021, 39, 1241–1247. [Google Scholar] [CrossRef]
- Dai, L.; Zheng, T.; Xu, K.; Han, Y.; Xu, L.; Huang, E.; An, Y.; Cheng, Y.; Li, S.; Liu, M.; et al. A Universal Design of Betacoronavirus Vaccines against COVID-19, MERS, and SARS. Cell 2020, 182, 722–733.e11. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Cao, H.; Liu, C. SARS-CoV-2 S1 is superior to the RBD as a COVID-19 subunit vaccine antigen. J. Med. Virol. 2021, 93, 892–898. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Ke, B.; Feng, Q.; Yang, D.; Lian, Q.; Li, Z.; Lu, L.; Ke, C.; Liu, Z.; Liao, G. Construction and immunogenic studies of a mFc fusion receptor binding domain (RBD) of spike protein as a subunit vaccine against SARS-CoV-2 infection. Chem. Commun. 2020, 56, 8683–8686. [Google Scholar] [CrossRef]
- Liu, X.; Drelich, A.; Li, W.; Chen, C.; Sun, Z.; Shi, M.; Adams, C.; Mellors, J.W.; Tseng, C.T.; Dimitrov, D.S. Enhanced elicitation of potent neutralizing antibodies by the SARS-CoV-2 spike receptor binding domain Fc fusion protein in mice. Vaccine 2020, 38, 7205–7212. [Google Scholar] [CrossRef] [PubMed]
- Haun, B.K.; Lai, C.Y.; Williams, C.A.; Wong, T.A.S.; Lieberman, M.M.; Pessaint, L.; Andersen, H.; Lehrer, A.T. CoVaccine HT Adjuvant Potentiates Robust Immune Responses to Recombinant SARS-CoV-2 Spike S1 Immunization. Front. Immunol. 2020, 11, 599587. [Google Scholar] [CrossRef]
- Kim, E.; Erdos, G.; Huang, S.; Kenniston, T.W.; Balmert, S.C.; Carey, C.D.; Raj, V.S.; Epperly, M.W.; Klimstra, W.B.; Haagmans, B.L.; et al. Microneedle array delivered recombinant coronavirus vaccines: Immunogenicity and rapid translational development. EBioMedicine 2020, 55, 102743. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Zheng, Q.; Yu, H.; Wu, D.; Xue, W.; Xiong, H.; Huang, X.; Nie, M.; Yue, M.; Rong, R.; et al. SARS-CoV-2 spike produced in insect cells elicits high neutralization titres in non-human primates. Emerg. Microbes Infect. 2020, 9, 2076–2090. [Google Scholar] [CrossRef]
- Guebre-Xabier, M.; Patel, N.; Tian, J.H.; Zhou, B.; Maciejewski, S.; Lam, K.; Portnoff, A.D.; Massare, M.J.; Frieman, M.B.; Piedra, P.A.; et al. NVX-CoV2373 vaccine protects cynomolgus macaque upper and lower airways against SARS-CoV-2 challenge. Vaccine 2020, 38, 7892–7896. [Google Scholar] [CrossRef]
- Tian, J.H.; Patel, N.; Haupt, R.; Zhou, H.; Weston, S.; Hammond, H.; Logue, J.; Portnoff, A.D.; Norton, J.; Guebre-Xabier, M.; et al. SARS-CoV-2 spike glycoprotein vaccine candidate NVX-CoV2373immunogenicity in baboons and protection in mice. Nat. Commun. 2021, 12, 372. [Google Scholar] [CrossRef]
- Alter, G.; Gorman, M.; Patel, N.; Guebre-Xabier, M.; Zhu, A.; Atyeo, C.; Pullen, K.; Loos, C.; Goez-Gazi, Y.; Carrion, R.; et al. Collaboration between the Fab and Fc contribute to maximal protection against SARS-CoV-2 following NVX-CoV2373 subunit vaccine with Matrix-M vaccination. Res. Sq. 2021. [Google Scholar] [CrossRef]
- Keech, C.; Albert, G.; Cho, I.; Robertson, A.; Reed, P.; Neal, S.; Plested, J.S.; Zhu, M.; Cloney-Clark, S.; Zhou, H.; et al. Phase 1-2 Trial of a SARS-CoV-2 Recombinant Spike Protein Nanoparticle Vaccine. N. Engl. J. Med. 2020, 383, 2320–2332. [Google Scholar] [CrossRef] [PubMed]
- Voss, W.N.; Hou, Y.J.; Johnson, N.V.; Kim, J.E.; Delidakis, G.; Horton, A.P.; Bartzoka, F.; Paresi, C.J.; Tanno, Y.; Abbasi, S.A.; et al. Prevalent, protective, and convergent IgG recognition of SARS-CoV-2 non-RBDspike epitopes in COVID-19 convalescent plasma. bioRxiv 2020. [Google Scholar] [CrossRef]
- Weissman, D.; Alameh, M.G.; de Silva, T.; Collini, P.; Hornsby, H.; Brown, R.; LaBranche, C.C.; Edwards, R.J.; Sutherland, L.; Santra, S.; et al. D614G Spike Mutation Increases SARS CoV-2 Susceptibility to Neutralization. Cell Host Microb. 2021, 29, 23–31.e4. [Google Scholar] [CrossRef]
- Suryadevara, N.; Shrihari, S.; Gilchuk, P.; VanBlargan, L.A.; Binshtein, E.; Zost, S.J.; Nargi, R.S.; Sutton, R.E.; Winkler, E.S.; Chen, E.C.; et al. Neutralizing and protective human monoclonal antibodies recognizing the N-terminal domain of the SARS-CoV-2 spike protein. bioRxiv 2021. [Google Scholar] [CrossRef]
- Graham, C.; Seow, J.; Huettner, I.; Khan, H.; Kouphou, N.; Acors, S.; Winstone, H.; Pickering, S.; Pedro Galao, R.; Jose Lista, M.; et al. Impact of the B.1.1.7 variant on neutralizing monoclonal antibodies recognizing diverse epitopes on SARS-CoV-2 Spike. bioRxiv 2021. [Google Scholar] [CrossRef]
- Ho, D.; Wang, P.; Liu, L.; Iketani, S.; Luo, Y.; Guo, Y.; Wang, M.; Yu, J.; Zhang, B.; Kwong, P.; et al. Increased Resistance of SARS-CoV-2 Variants B.1.351 and B.1.1.7 to Antibody Neutralization. Res. Sq. 2021. [Google Scholar] [CrossRef]
- Shen, X.; Tang, H.; McDanal, C.; Wagh, K.; Fischer, W.; Theiler, J.; Yoon, H.; Li, D.; Haynes, B.F.; Sanders, K.O.; et al. SARS-CoV-2 variant B.1.1.7 is susceptible to neutralizing antibodies elicited by ancestral Spike vaccines. bioRxiv 2021. [Google Scholar] [CrossRef]
- Hsieh, C.L.; Goldsmith, J.A.; Schaub, J.M.; DiVenere, A.M.; Kuo, H.C.; Javanmardi, K.; Le, K.C.; Wrapp, D.; Lee, A.G.; Liu, Y.; et al. Structure-based design of prefusion-stabilized SARS-CoV-2 spikes. Science 2020, 369, 1501–1505. [Google Scholar] [CrossRef] [PubMed]
- Kalimuddin, S.; Wijaya, L.; Chan, Y.F.Z.; Wong, A.W.L.; Oh, H.M.L.; Wang, L.F.; Kassim, J.A.; Zhao, J.; Shi, Z.; Low, J.G. A phase II randomized study to determine the safety and immunogenicity of the novel PIKA rabies vaccine containing the PIKA adjuvant using an accelerated regimen. Vaccine 2017, 35, 7127–7132. [Google Scholar] [CrossRef]
- Zhou, D.; Tian, X.; Qi, R.; Peng, C.; Zhang, W. Identification of 22 N-glycosites on spike glycoprotein of SARS-CoV-2 and accessible surface glycopeptide motifs: Implications for vaccination and antibody therapeutics. Glycobiology 2021, 31, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lai, D.Y.; Zhang, H.N.; Jiang, H.W.; Tian, X.; Ma, M.L.; Qi, H.; Meng, Q.F.; Guo, S.J.; Wu, Y.; et al. Linear epitopes of SARS-CoV-2 spike protein elicit neutralizing antibodies in COVID-19 patients. Cell Mol. Immunol. 2020, 17, 1095–1097. [Google Scholar] [CrossRef]
- Poh, C.M.; Carissimo, G.; Wang, B.; Amrun, S.N.; Lee, C.Y.; Chee, R.S.; Fong, S.W.; Yeo, N.K.; Lee, W.H.; Torres-Ruesta, A.; et al. Two linear epitopes on the SARS-CoV-2 spike protein that elicit neutralising antibodies in COVID-19 patients. Nat. Commun. 2020, 11, 2806. [Google Scholar] [CrossRef] [PubMed]
- Farrera-Soler, L.; Daguer, J.P.; Barluenga, S.; Vadas, O.; Cohen, P.; Pagano, S.; Yerly, S.; Kaiser, L.; Vuilleumier, N.; Winssinger, N. Identification of immunodominant linear epitopes from SARS-CoV-2 patient plasma. PLoS ONE 2020, 15, e0238089. [Google Scholar] [CrossRef]
- Walsh, E.E.; Frenck, R.W., Jr.; Falsey, A.R.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Mulligan, M.J.; Bailey, R.; et al. Safety and Immunogenicity of Two RNA-Based Covid-19 Vaccine Candidates. N. Engl. J. Med. 2020, 383, 2439–2450. [Google Scholar] [CrossRef]
- Bangaru, S.; Ozorowski, G.; Turner, H.L.; Antanasijevic, A.; Huang, D.; Wang, X.; Torres, J.L.; Diedrich, J.K.; Tian, J.H.; Portnoff, A.D.; et al. Structural analysis of full-length SARS-CoV-2 spike protein from an advanced vaccine candidate. Science 2020, 370, 1089–1094. [Google Scholar] [CrossRef]
- Laczkó, D.; Hogan, M.J.; Toulmin, S.A.; Hicks, P.; Lederer, K.; Gaudette, B.T.; Castaño, D.; Amanat, F.; Muramatsu, H.; Oguin Ojha, A.; et al. A Single Immunization with Nucleoside-Modified mRNA Vaccines Elicits Strong Cellular and Humoral Immune Responses against SARS-CoV-2 in Mice. Immunity 2020, 53, 724–732.e7. [Google Scholar] [CrossRef] [PubMed]
- Corbett, K.S.; Edwards, D.K.; Leist, S.R.; Abiona, O.M.; Boyoglu-Barnum, S.; Gillespie, R.A.; Himansu, S.; Schäfer, A.; Ziwawo, C.T.; DiPiazza, A.T.; et al. SARS-CoV-2 mRNA vaccine design enabled by prototype pathogen preparedness. Nature 2020, 586, 567–571. [Google Scholar] [CrossRef]
- Chi, X.; Yan, R.; Zhang, J.; Zhang, G.; Zhang, Y.; Hao, M.; Zhang, Z.; Fan, P.; Dong, Y.; Yang, Y.; et al. A neutralizing human antibody binds to the N-terminal domain of the Spike protein of SARS-CoV-2. Science 2020, 369, 650–655. [Google Scholar] [CrossRef]
- Xu, C.; Wang, Y.; Liu, C.; Zhang, C.; Han, W.; Hong, X.; Wang, Y.; Hong, Q.; Wang, S.; Zhao, Q.; et al. Conformational dynamics of SARS-CoV-2 trimeric spike glycoprotein in complex with receptor ACE2 revealed by cryo-EM. Sci. Adv. 2021, 7, eabe5575. [Google Scholar] [CrossRef]
- Du, S.; Cao, Y.; Zhu, Q.; Yu, P.; Qi, F.; Wang, G.; Du, X.; Bao, L.; Deng, W.; Zhu, H.; et al. Structurally Resolved SARS-CoV-2 Antibody Shows High Efficacy in Severely Infected Hamsters and Provides a Potent Cocktail Pairing Strategy. Cell 2020, 183, 1013–1023.e13. [Google Scholar] [CrossRef]
- Lau, Y.F.; Tang, L.H.; McCall, A.W.; Ooi, E.E.; Subbarao, K. An adjuvant for the induction of potent, protective humoral responses to an H5N1 influenza virus vaccine with antigen-sparing effect in mice. J. Virol. 2010, 84, 8639–8649. [Google Scholar] [CrossRef]
- Lau, Y.F.; Tang, L.H.; Ooi, E.E.; Subbarao, K. Activation of the innate immune system provides broad-spectrum protection against influenza A viruses with pandemic potential in mice. Virology 2010, 406, 80–87. [Google Scholar] [CrossRef]
- Shen, E.; Li, L.; Li, L.; Feng, L.; Lu, L.; Yao, Z.; Lin, H.; Wu, C. PIKA as an adjuvant enhances specific humoral and cellular immune responses following the vaccination of mice with HBsAg plus PIKA. Cell Mol. Immunol. 2007, 4, 113–120. [Google Scholar] [PubMed]
- Lin, H.X.; Gontier, C.; Saron, M.F.; Perrin, P. A new immunostimulatory complex (PICKCa) in experimental rabies: Antiviral and adjuvant effects. Arch. Virol. 1993, 131, 307–319. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, S.; Li, W.; Hu, Y.; Zhao, J.; Liu, F.; Lin, H.; Liu, Y.; Wang, L.; Xu, S.; et al. A novel rabies vaccine based-on toll-like receptor 3 (TLR3) agonist PIKA adjuvant exhibiting excellent safety and efficacy in animal studies. Virology 2016, 489, 165–172. [Google Scholar] [CrossRef]
- Wijaya, L.; Tham, C.Y.L.; Chan, Y.F.Z.; Wong, A.W.L.; Li, L.T.; Wang, L.F.; Bertoletti, A.; Low, J.G. An accelerated rabies vaccine schedule based on toll-like receptor 3 (TLR3) agonist PIKA adjuvant augments rabies virus specific antibody and T cell response in healthy adult volunteers. Vaccine 2017, 35, 1175–1183. [Google Scholar] [CrossRef] [PubMed]
| Name of Vaccine | Developer | Protein or Subunit | Production | Adjuvant | Reference |
|---|---|---|---|---|---|
| RBD219-N1 | Baylor College of Medicine | RBD | Yeast | Alum | [33,34] |
| SERS-S1 | University of Pittsburgh School of Medicine | S1 subunit | HEK293K cells | MPLA | [41] |
| NVX-CoV2373 | Novavax, Inc | Full-length trimeric Spike, protease resistant along with 2 proline substitutions at residues K986 and V987 | Sf9 insect cells | Matrix-M (Quillaja saponins formulated with cholesterol and phospholipids into nanoparticles) | [43] |
| RBD-mFc | Guangzhou University of Chinese Medicine | RBD | Expi293 cells | Alum/Freund’s complete adjuvant | [38] |
| S-2P | Xiamen University | Secreted, prefusion stabilized trimeric Spike | Sf9 insect cells | Alum | [42] |
| RBD-Fc | University of Pittsburgh Medical School | RBD | Expi293 | MF59 (squalene) | [39] |
| Spike S1 | University of Hawaii | S1 subunit | HEK293T cells | CoVaccine HT™ (synthetic sucrose fatty acid sulphate esters (SFASES) immobilized inside the oily droplets of the submicron squalane in water emulsion) | [40] |
| S1-4 | Chinese Center For Disease Control and Prevention | RBD | E.coli. BL21(DE3) | Alum | [35] |
| RBD-sc-dimer | University of Chinese Academy of Sciences | RBD-dimer protein | HEK293T cells | AddaVax (MF59) | [36] |
| S1, RBD | Chinese Academy of Medical Sciences and Peking Union Medical College | S1 subunit and RBD | HEK293T cells | Alum | [37] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tong, J.; Zhu, C.; Lai, H.; Feng, C.; Zhou, D. Potent Neutralization Antibodies Induced by a Recombinant Trimeric Spike Protein Vaccine Candidate Containing PIKA Adjuvant for COVID-19. Vaccines 2021, 9, 296. https://doi.org/10.3390/vaccines9030296
Tong J, Zhu C, Lai H, Feng C, Zhou D. Potent Neutralization Antibodies Induced by a Recombinant Trimeric Spike Protein Vaccine Candidate Containing PIKA Adjuvant for COVID-19. Vaccines. 2021; 9(3):296. https://doi.org/10.3390/vaccines9030296
Chicago/Turabian StyleTong, Jiao, Chenxi Zhu, Hanyu Lai, Chunchao Feng, and Dapeng Zhou. 2021. "Potent Neutralization Antibodies Induced by a Recombinant Trimeric Spike Protein Vaccine Candidate Containing PIKA Adjuvant for COVID-19" Vaccines 9, no. 3: 296. https://doi.org/10.3390/vaccines9030296
APA StyleTong, J., Zhu, C., Lai, H., Feng, C., & Zhou, D. (2021). Potent Neutralization Antibodies Induced by a Recombinant Trimeric Spike Protein Vaccine Candidate Containing PIKA Adjuvant for COVID-19. Vaccines, 9(3), 296. https://doi.org/10.3390/vaccines9030296






