Effects of Transpulmonary Administration of Caffeine on Brain Activity in Healthy Men
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Stimuli
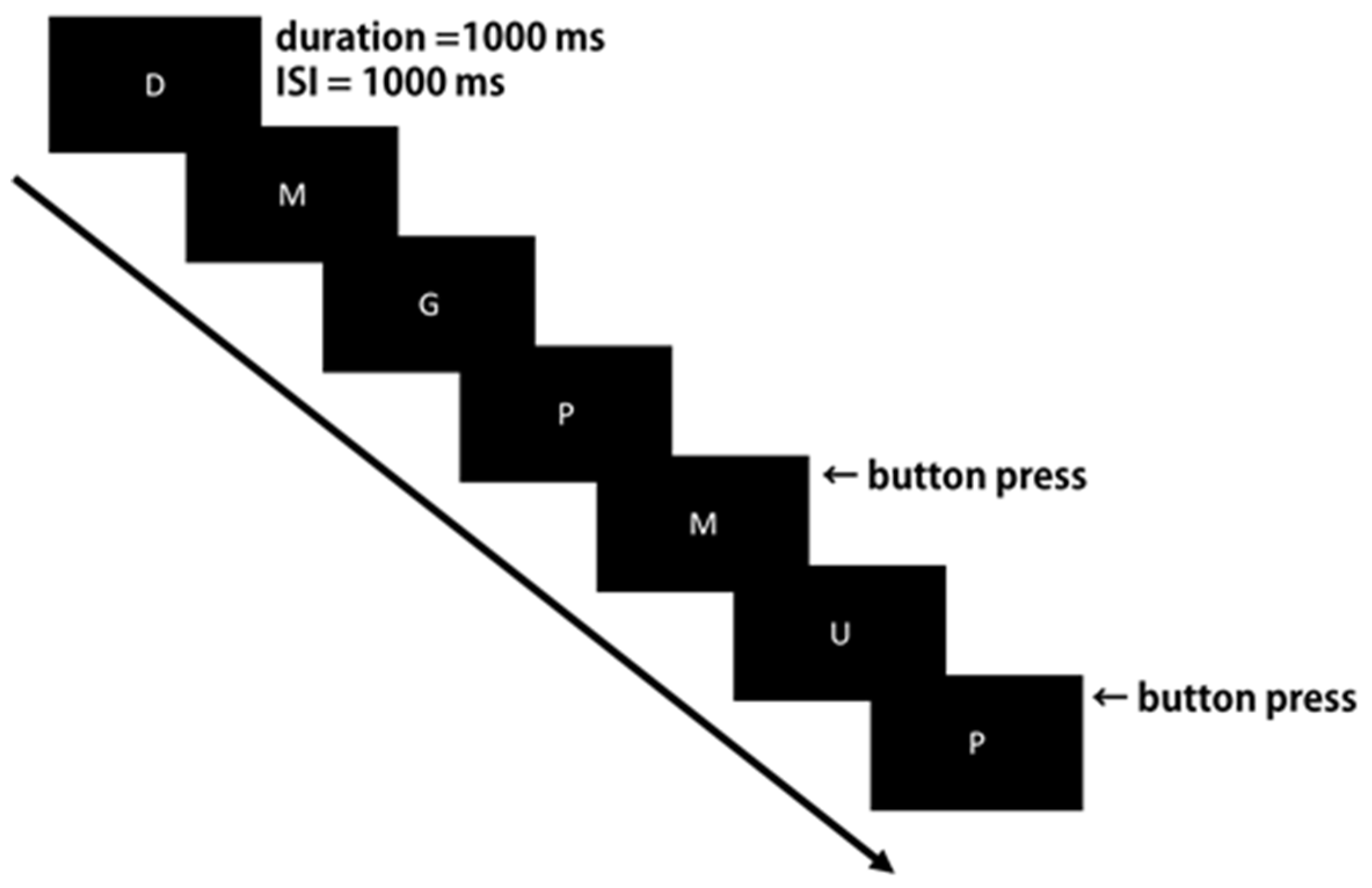
2.3. Experimental Task
2.4. Procedures
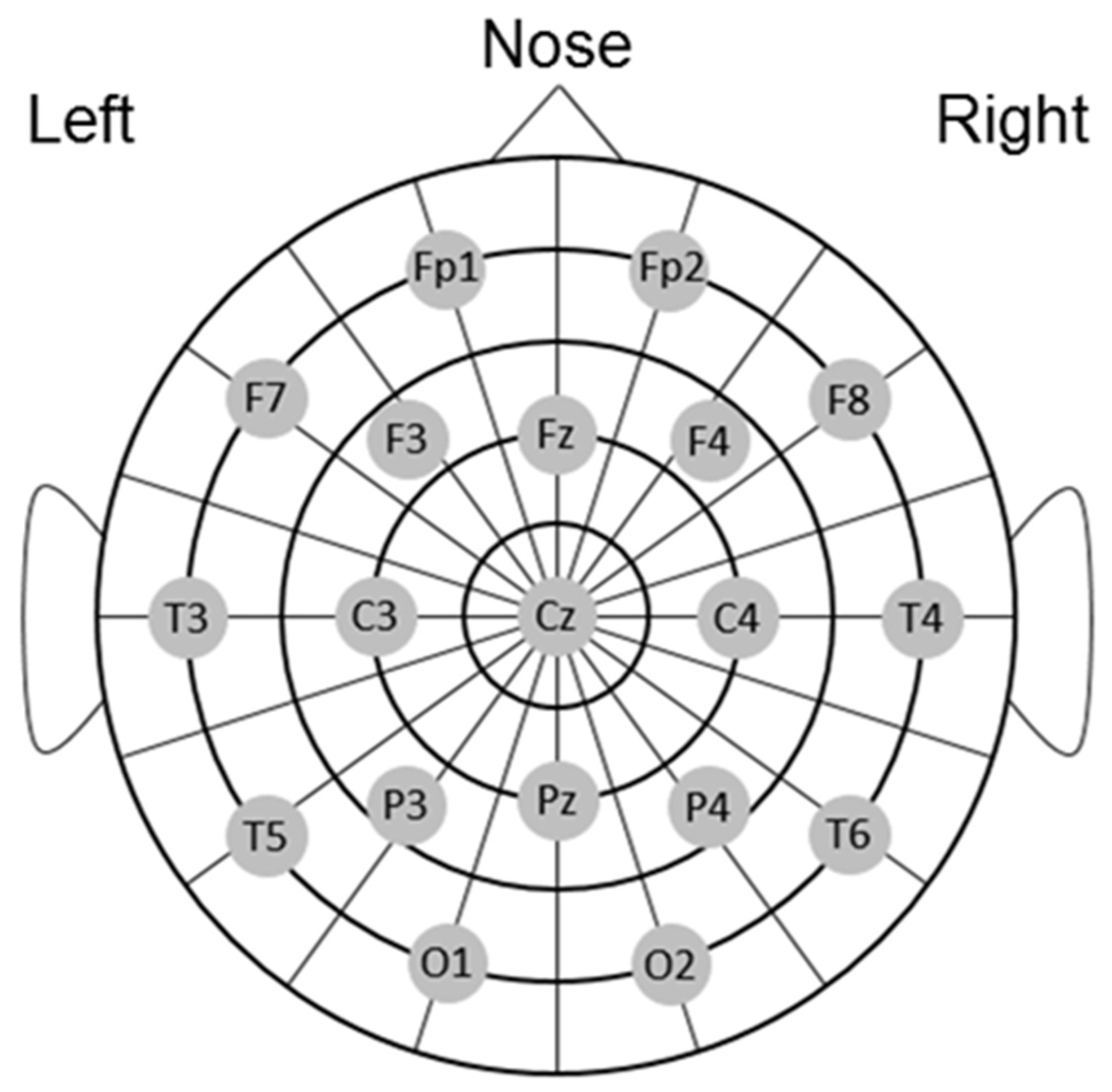
2.5. EEG Recording and Analysis
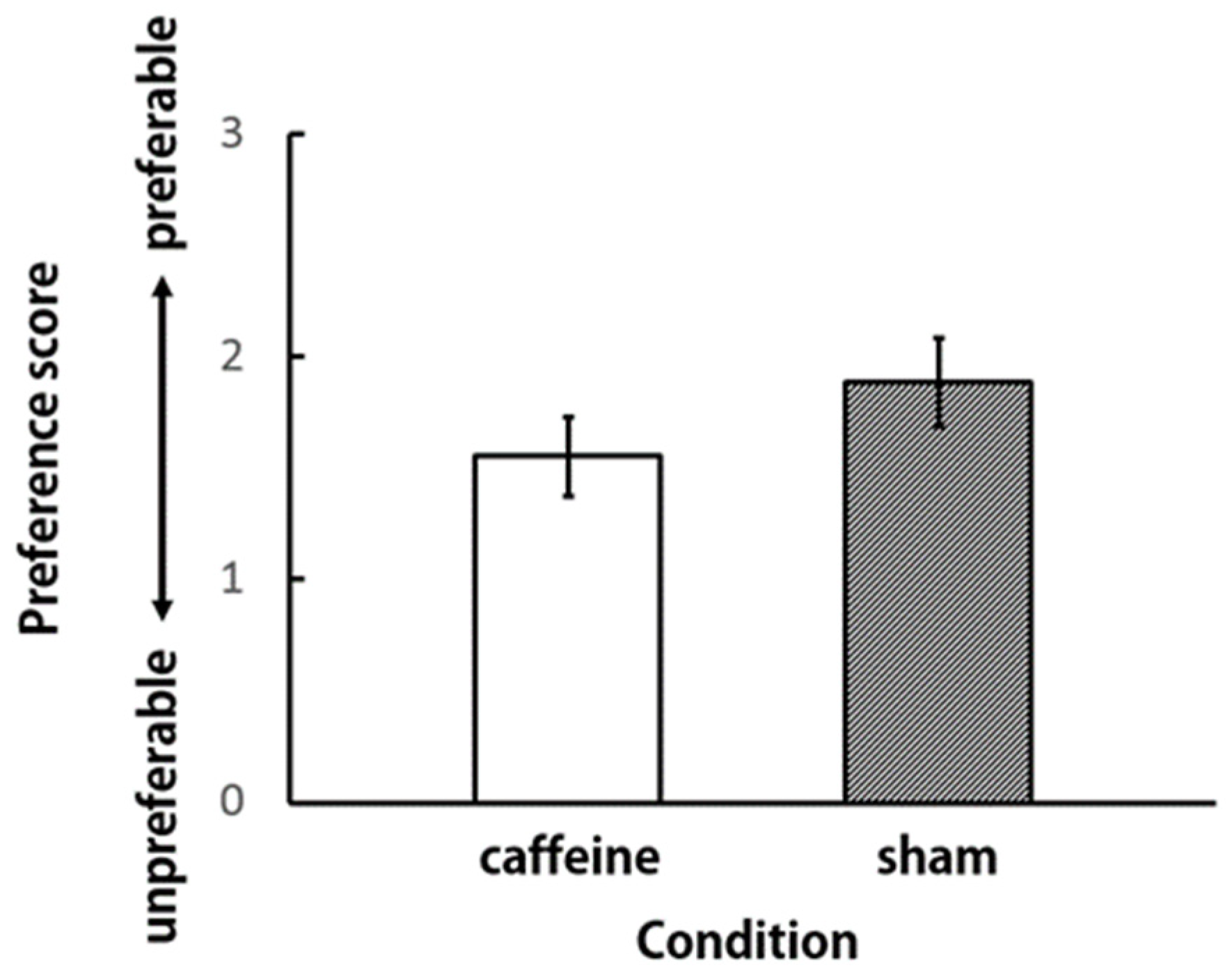
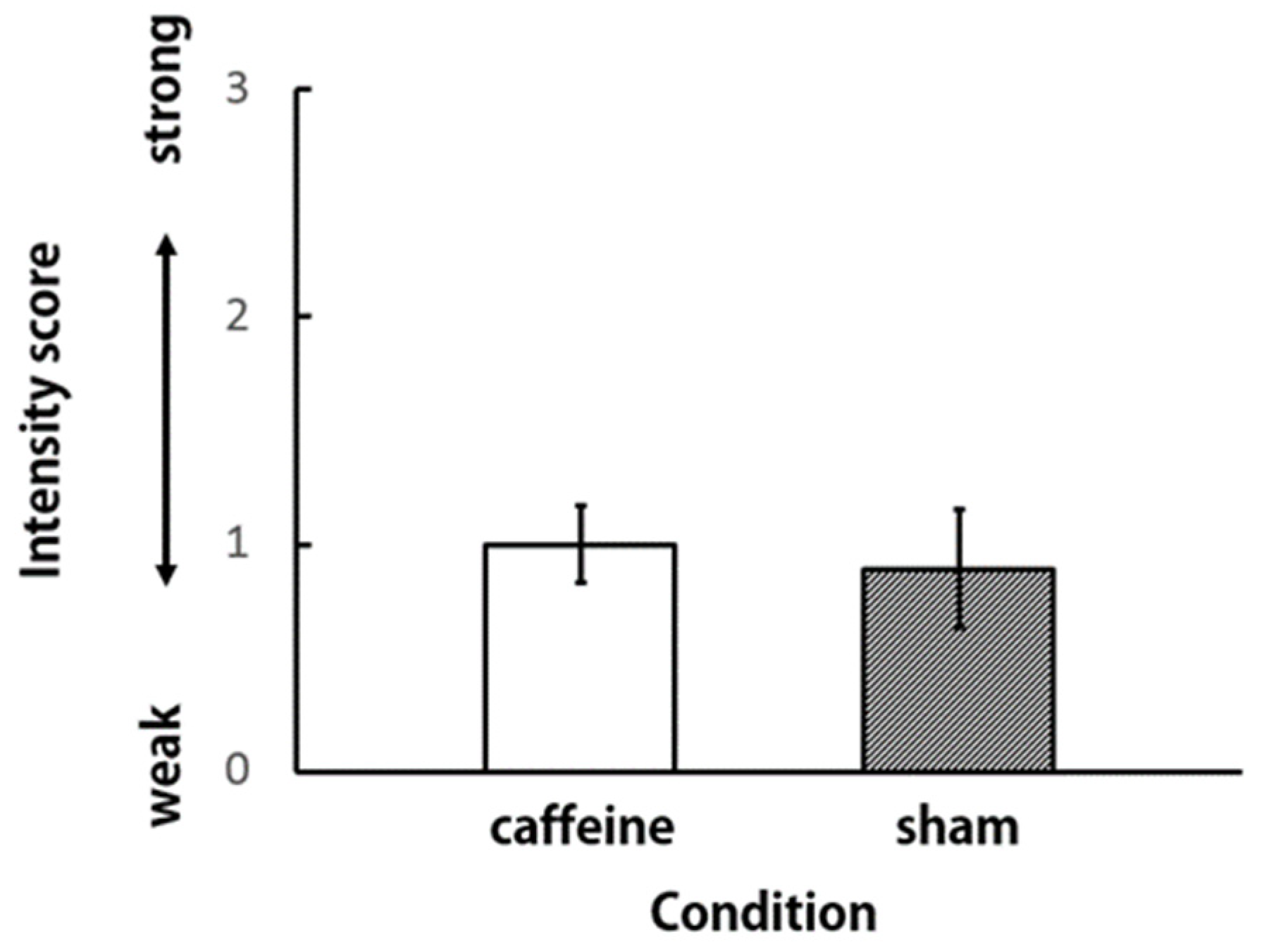
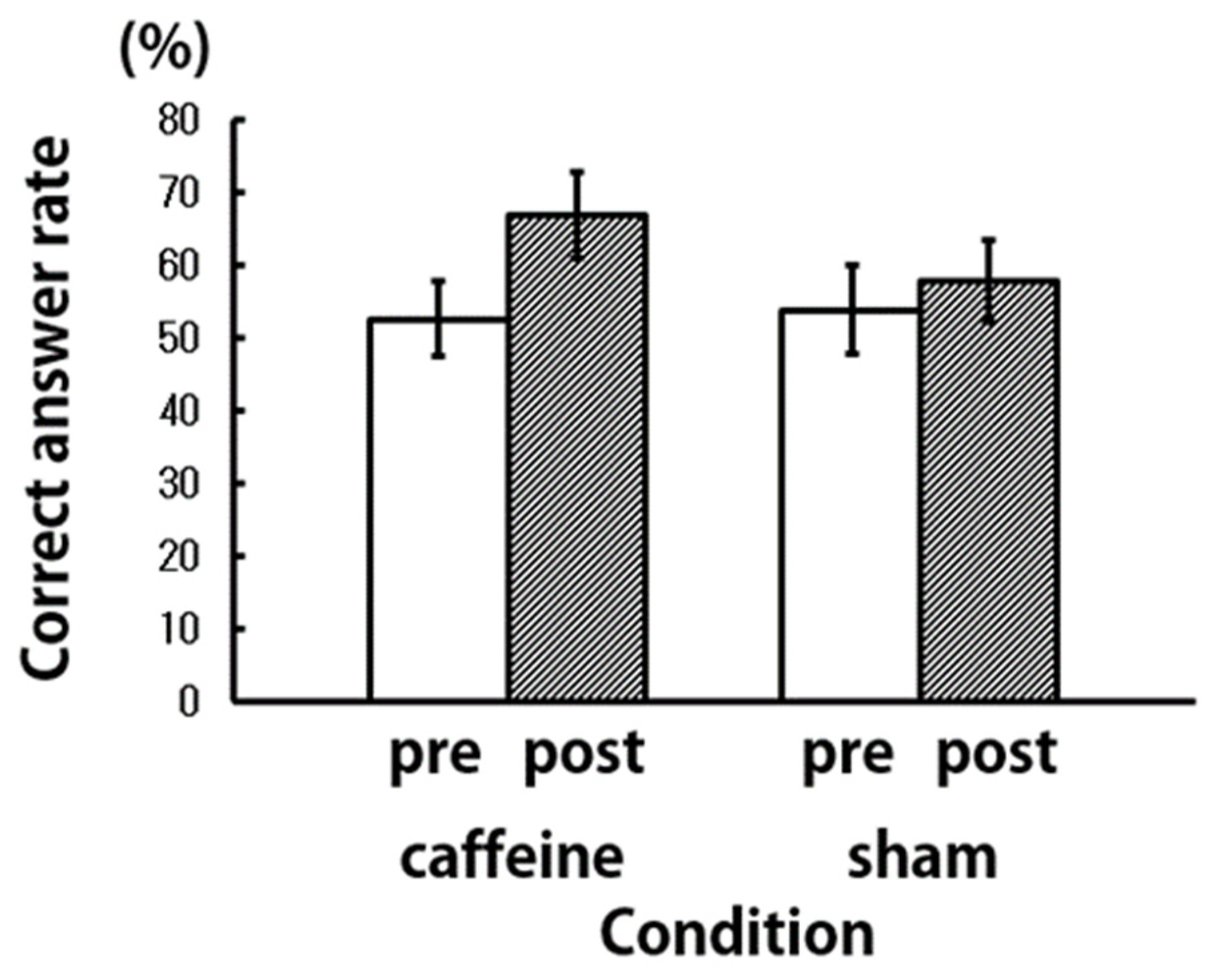
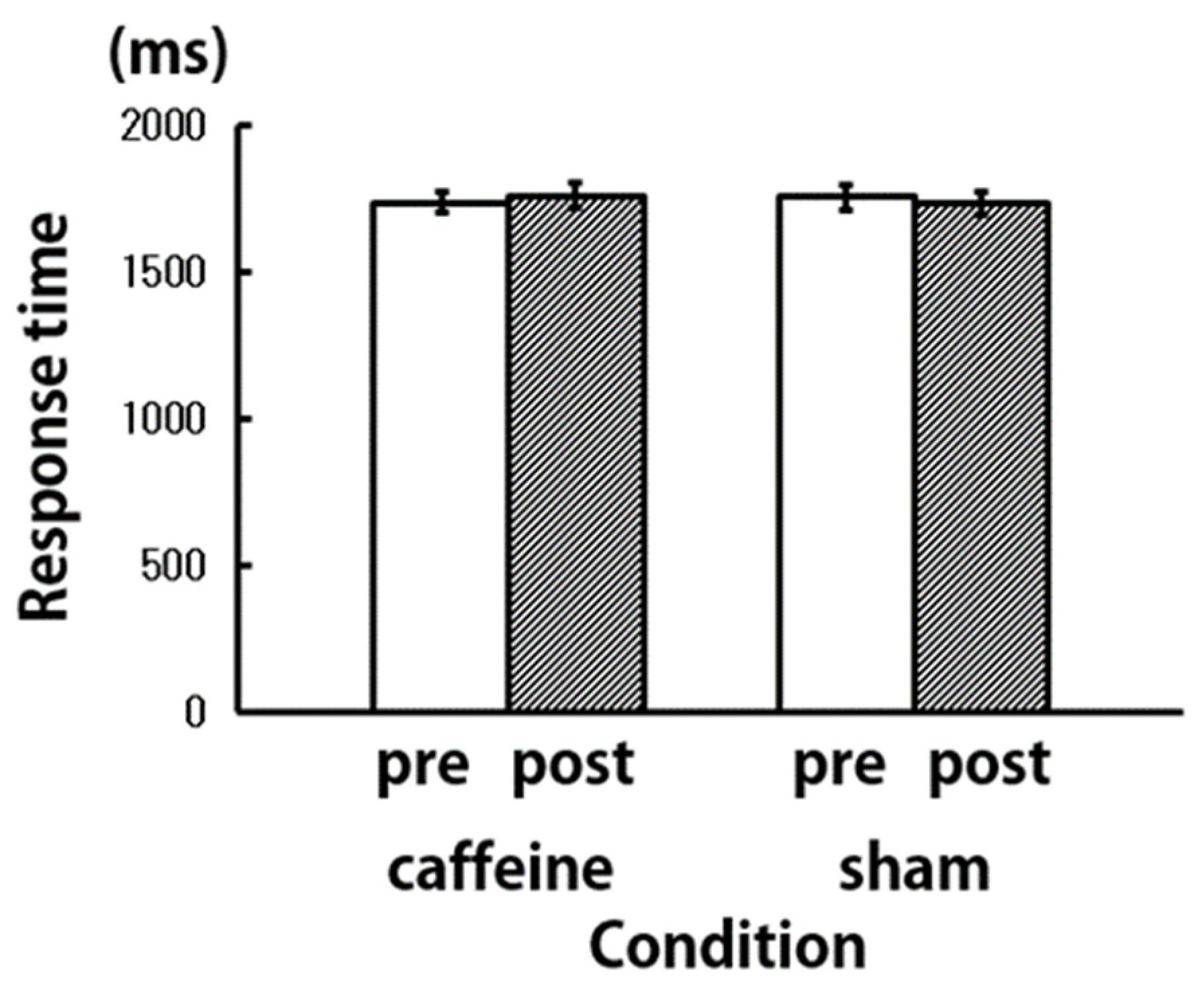
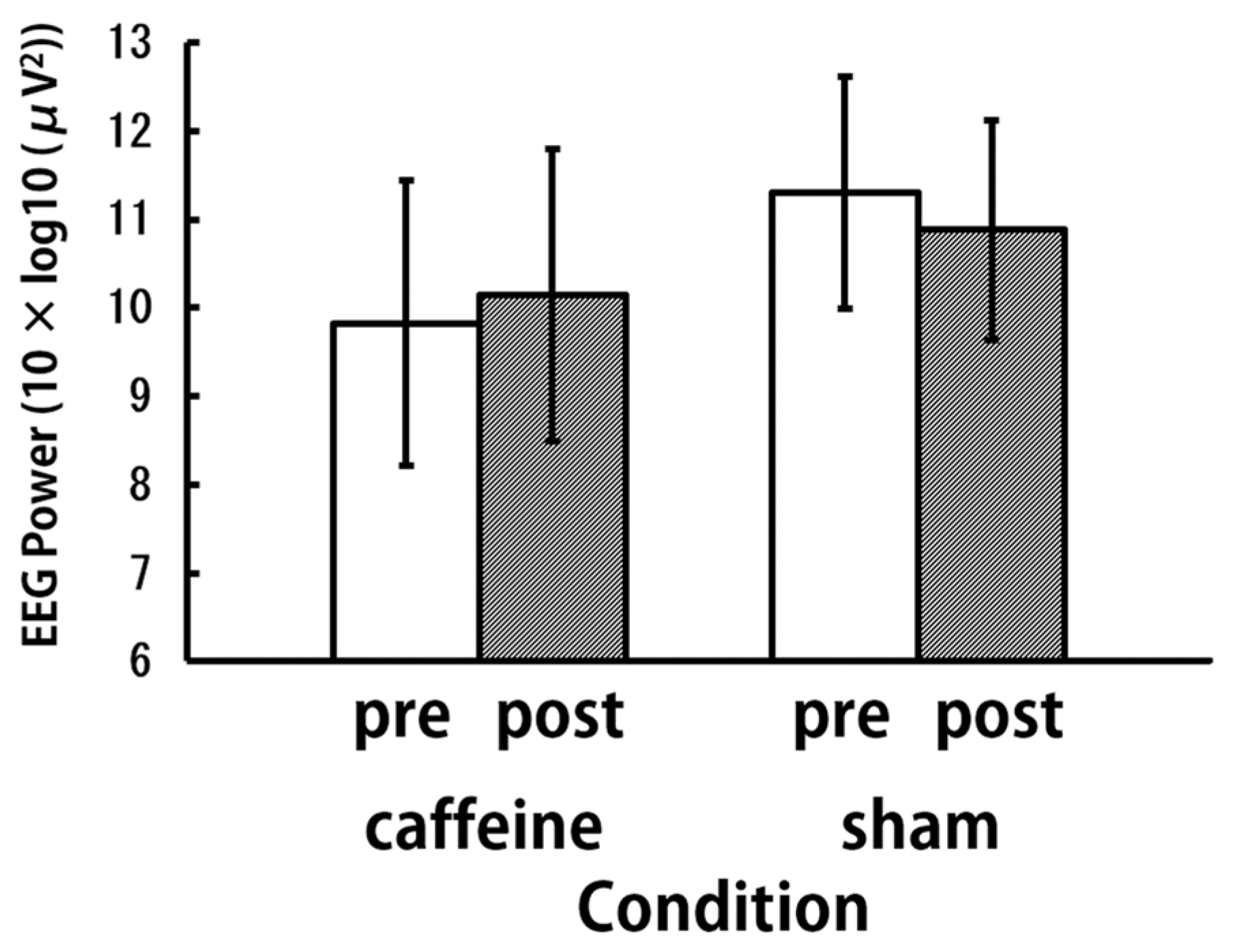
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Van Dongen, H.P.A.; Price, N.J.; Mullington, J.M.; Szuba, M.P.; Kapoor, S.C.; Dinges, D.F. Caffeine eliminates psychomotor vigilance deficits from sleep inertia. Sleep 2001, 24, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, S.; Laxminarayan, S.; Wesensten, N.J.; Kamimori, G.H.; Balkin, T.J.; Reitman, J. Dose-dependent model of caffeine effects on human vigilance during total sleep deprivation. J. Theor. Biol. 2014, 358, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Einother, S.J.L.; Giesbrecht, T. Caffeine as an attention enhancer: Reviewing existing assumptions. Psychopharmacology 2013, 225, 251–274. [Google Scholar] [CrossRef] [PubMed]
- Brunyé, T.T.; Mahoney, C.R.; Lieberman, H.R.; Taylor, H.A. Caffeine modulates attention network function. Brain Cogn. 2010, 72, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Nehlig, A. Is caffeine a cognitive enhancer? J. Alzheimers Dis. 2010, 20, S85–S94. [Google Scholar] [CrossRef] [PubMed]
- Haskell, C.F.; Kennedy, D.O.; Wesnes, K.A.; Scholey, A.B. Cognitive and mood improvements of caffeine in habitual consumers and habitual non-consumers of caffeine. Psychopharmacology 2005, 179, 813–825. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, J.; Sawers, S.J.A. The Absolute Bioavailability of Caffeine in Man. Eur. J. Clin. Pharmacol. 1983, 24, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Zandvliet, A.S.; Huitema, A.D.R.; de Jonge, M.E.; den Hoed, R.; Sparidans, R.W.; Hendriks, V.M.; van den Brink, W.; van Ree, J.M.; Beijnen, J.H. Population pharmacokinetics of caffeine and its metabolites theobromine, paraxanthine and theophylline after inhalation in combination with diacetylmorphine. Basic Clin. Pharmacol. 2005, 96, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Arnaud, M.J. Metabolism of caffeine and other components of coffee. In Caffeine, Coffee, and Health; Garattini, S., Ed.; Raven Press: New York, NY, USA, 1993; pp. 43–95. [Google Scholar]
- Sauseng, P.; Griesmayr, B.; Freunberger, R.; Klimesch, W. Control mechanisms in working memory: A possible function of EEG theta oscillations. Neurosci. Biobehav. Rev. 2010, 34, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Okubo, M.; Suzuki, H.; Nicholls, M.E. A Japanese version of the FLANDERS handedness questionnaire. Shinrigaku Kenkyu 2014, 85, 474–481. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Owen, A.M.; McMillan, K.M.; Laird, A.R.; Bullmore, E.T. N-back working memory paradigm: A meta-analysis of normative functional neuroimaging. Hum. Brain Mapp. 2005, 25, 46–59. [Google Scholar] [CrossRef] [PubMed]
- Klem, G.H.; Luders, H.O.; Jasper, H.H.; Elger, C. The ten-twenty electrode system of the International Federation. The International Federation of Clinical Neurophysiology. Electroencephalogr. Clin. Neurophysiol. Suppl. 1999, 52, 3–6. [Google Scholar] [PubMed]
- Delorme, A.; Makeig, S. EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Mullen, T.; Kothe, C.; Chi, Y.M.; Ojeda, A.; Kerth, T.; Makeig, S.; Cauwenberghs, G.; Jung, T.P. Real-Time Modeling and 3D Visualization of Source Dynamics and Connectivity Using Wearable EEG. In Proceedings of the 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; pp. 2184–2187. [Google Scholar]
- Koppelstaetter, F.; Poeppel, T.D.; Siedentopf, C.M.; Ischebeck, A.; Verius, M.; Haala, I.; Mottaghy, F.M.; Rhomberg, P.; Golaszewski, S.; Gotwald, T. Does caffeine modulate verbal working memory processes? An fMRI study. NeuroImage 2008, 39, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Klaassen, E.B.; de Groot, R.H.M.; Evers, E.A.T.; Snel, J.; Veerman, E.C.I.; Ligtenberg, A.J.M.; Jolles, J.; Veltman, D.J. The effect of caffeine on working memory load-related brain activation in middle-aged males. Neuropharmacology 2013, 64, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Gevins, A.; Smith, M.E.; McEvoy, L.; Yu, D. High-resolution EEG mapping of cortical activation related to working memory: Effects of task difficulty, type of processing, and practice. Cereb. Cortex 1997, 7, 374–385. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, D.; Halligan, P. Opinion—The relevance of behavioural measures for functional-imaging studies of cognition. Nat. Rev. Neurosci. 2004, 5, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Hershey, T.; Black, K.J.; Hartlein, J.; Braver, T.S.; Barch, D.A.; Carl, J.L.; Perlmutter, J.S. Dopaminergic modulation of response inhibition: An fMRI study. Cogn. Brain Res. 2004, 20, 438–448. [Google Scholar] [CrossRef] [PubMed]
| Treatment Condition | ||||
|---|---|---|---|---|
| Caffeine | Sham | |||
| ΔPower (Post–Pre) | ΔPower (Post–Pre) | |||
| EEG Location | Mean | SE | Mean | SE |
| F8 | 0.314 | 0.225 | −0.420 | 0.231 |
| F4 | 0.164 | 0.168 | −0.416 | 0.248 |
| C4 | 0.173 | 0.171 | −0.335 | 0.189 |
| T4 | 0.290 | 0.199 | −0.318 | 0.239 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ueda, K.; Nakao, M. Effects of Transpulmonary Administration of Caffeine on Brain Activity in Healthy Men. Brain Sci. 2019, 9, 222. https://doi.org/10.3390/brainsci9090222
Ueda K, Nakao M. Effects of Transpulmonary Administration of Caffeine on Brain Activity in Healthy Men. Brain Sciences. 2019; 9(9):222. https://doi.org/10.3390/brainsci9090222
Chicago/Turabian StyleUeda, Kazutaka, and Masayuki Nakao. 2019. "Effects of Transpulmonary Administration of Caffeine on Brain Activity in Healthy Men" Brain Sciences 9, no. 9: 222. https://doi.org/10.3390/brainsci9090222
APA StyleUeda, K., & Nakao, M. (2019). Effects of Transpulmonary Administration of Caffeine on Brain Activity in Healthy Men. Brain Sciences, 9(9), 222. https://doi.org/10.3390/brainsci9090222




