The Use of Hyperscanning to Investigate the Role of Social, Affective, and Informative Gestures in Non-Verbal Communication. Electrophysiological (EEG) and Inter-Brain Connectivity Evidence
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
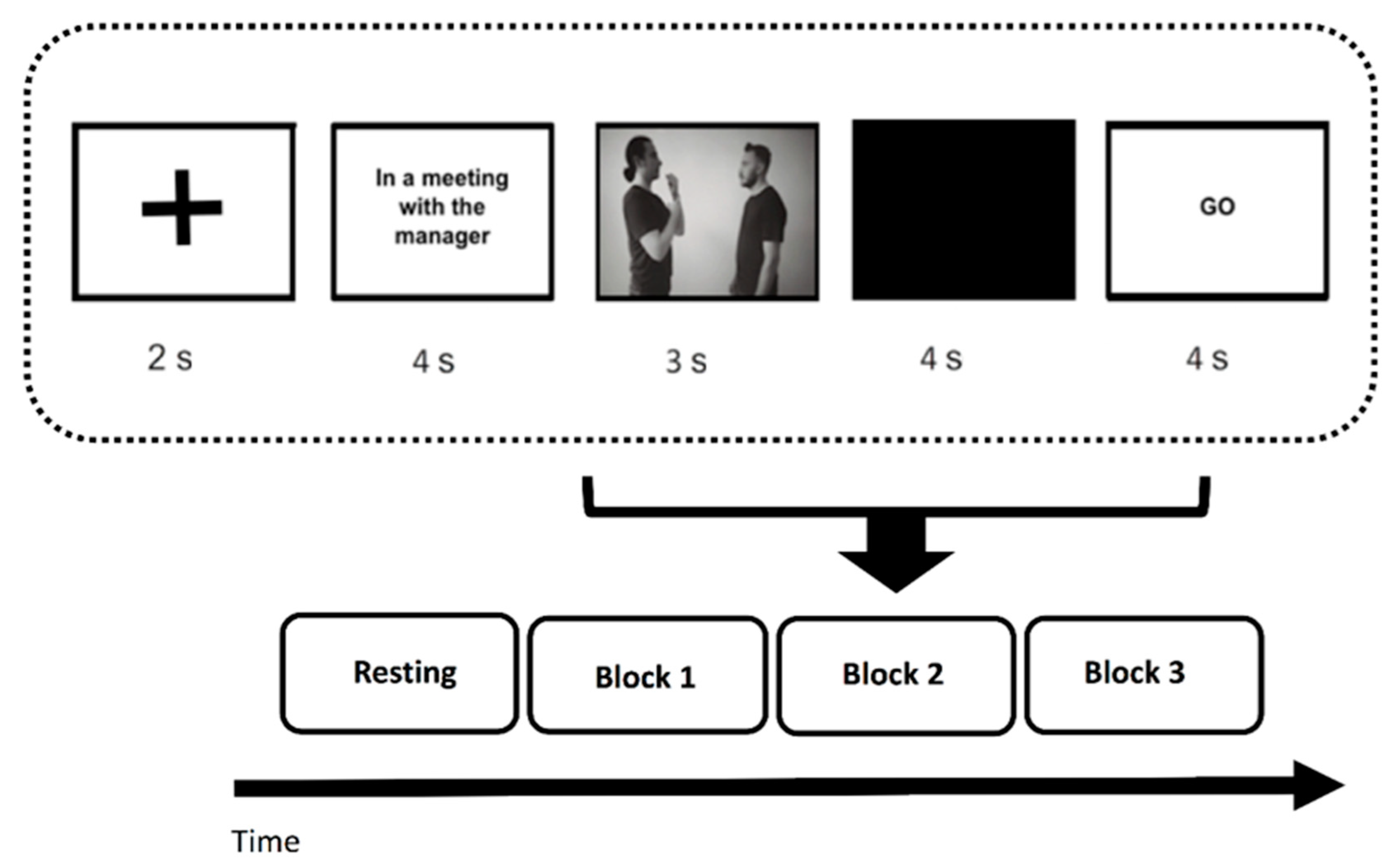
2.2. Procedure
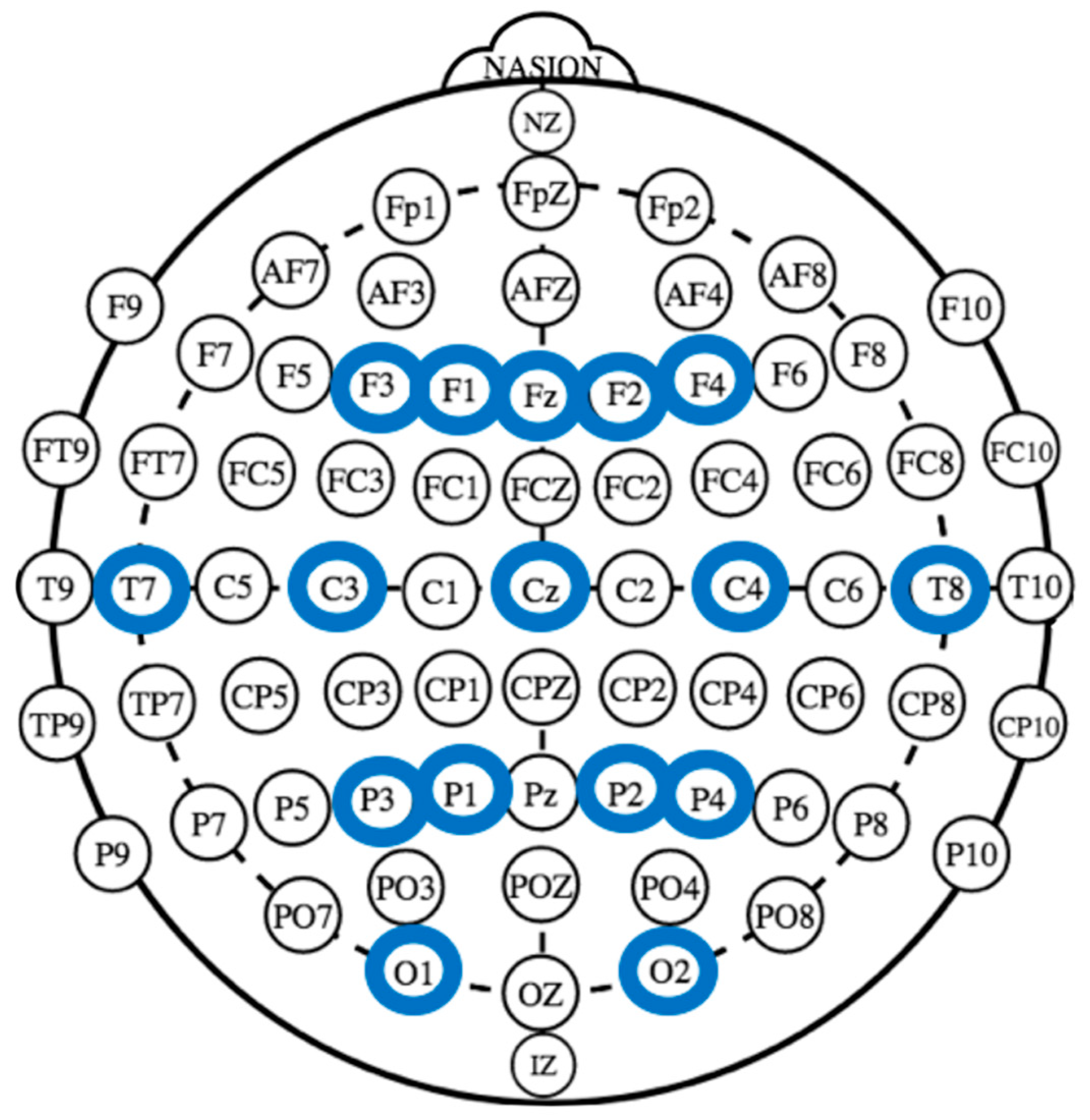
2.3. EEG Recording and Analysis
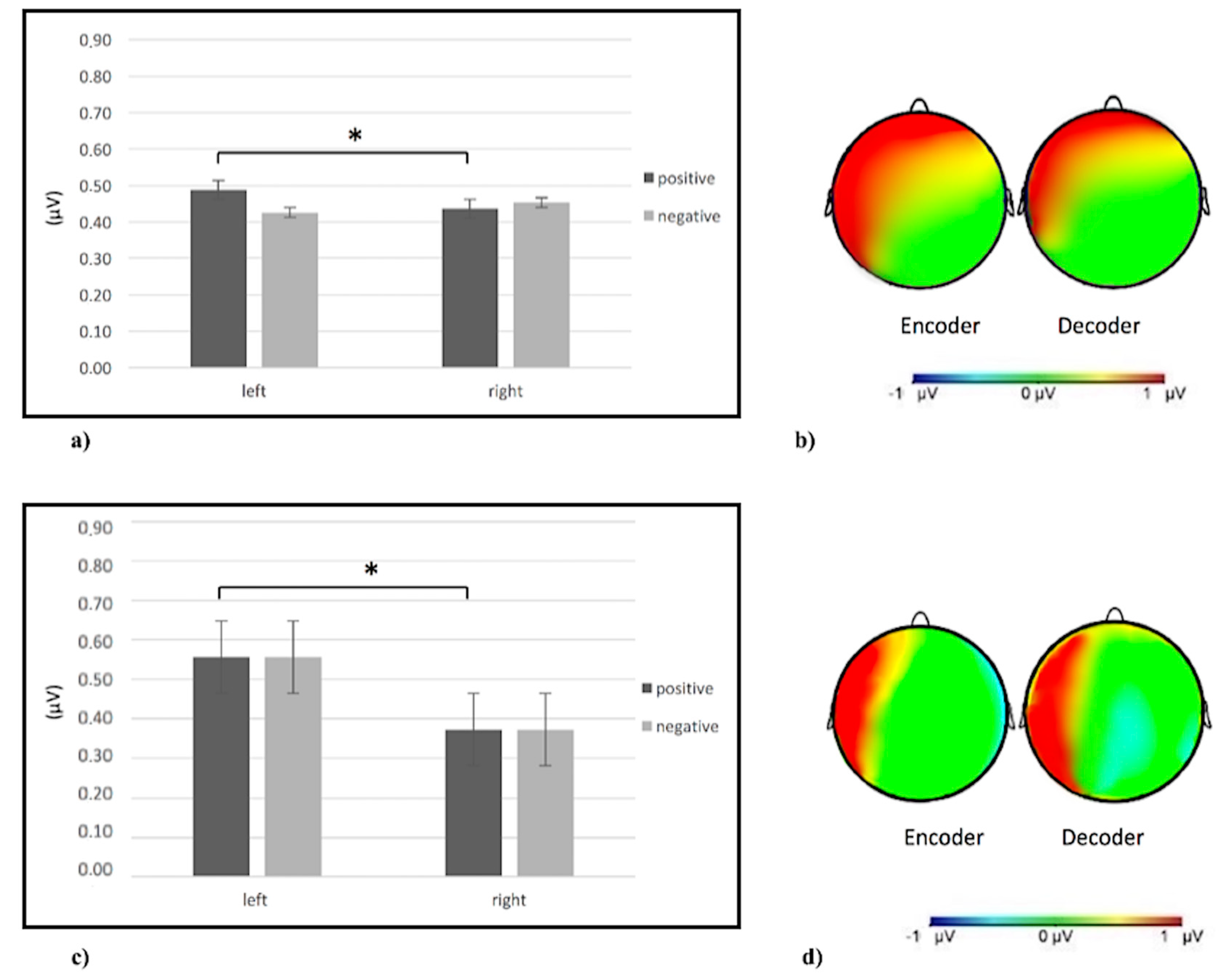
3. Results
3.1. Data Analysis
3.2. Single-Brain Analyses
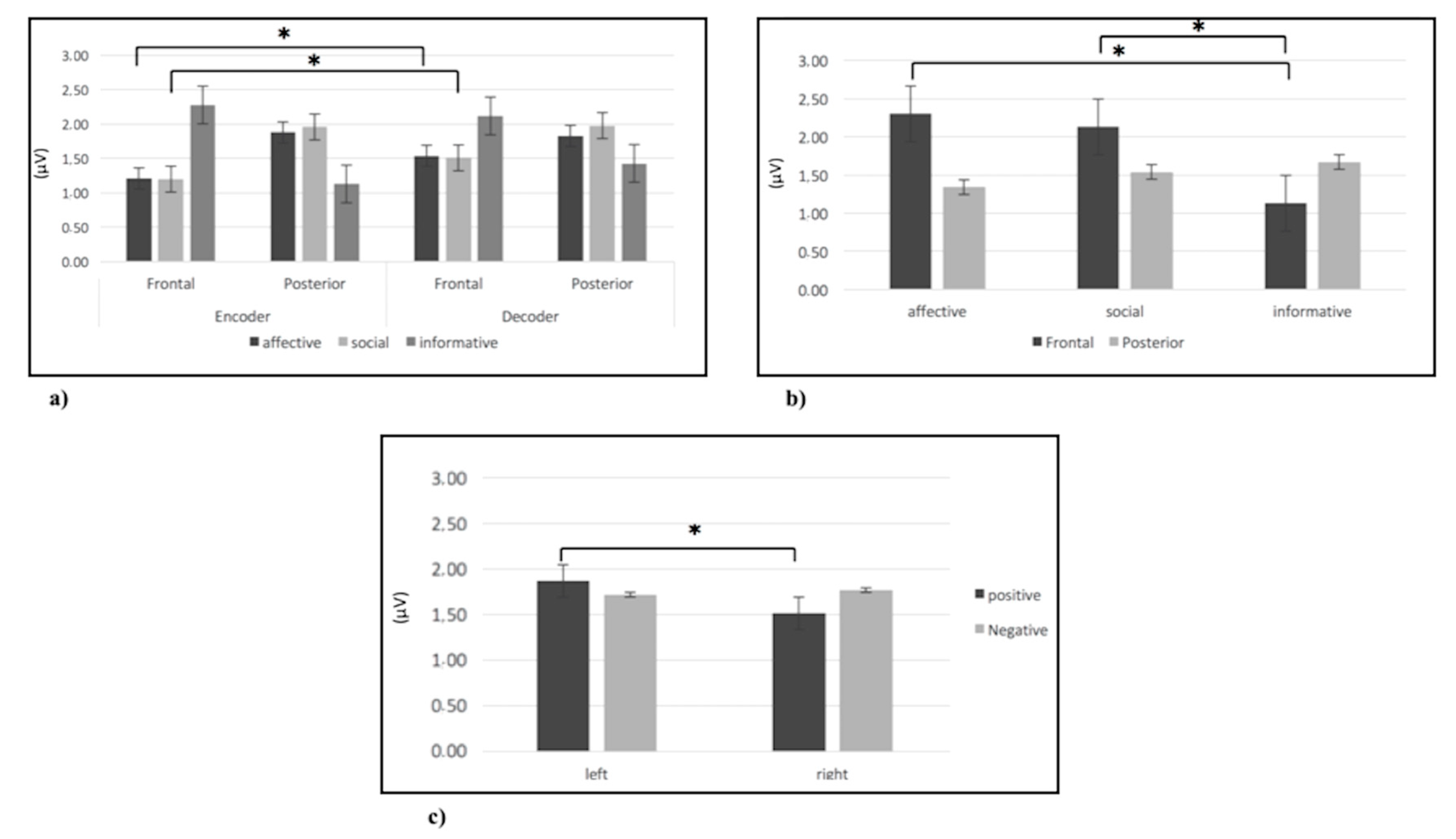
3.2.1. Alpha Band
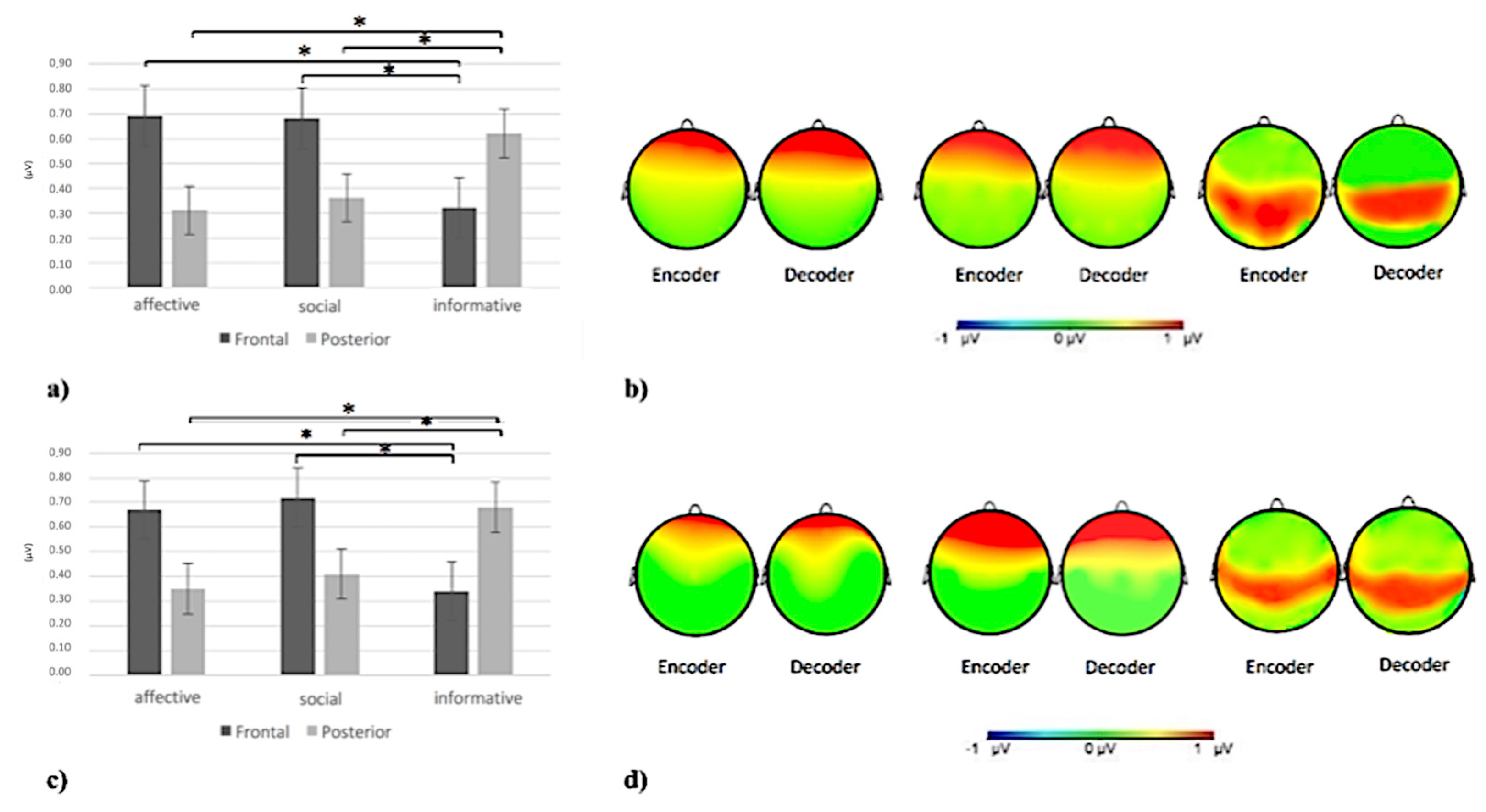
3.2.2. Delta Band
3.2.3. Theta Band
3.2.4. Beta Band
3.3. Inter-Brain Connectivity Analyses
3.3.1. Delta Band
3.3.2. Alpha Band
3.3.3. Theta Band
3.3.4. Beta Band
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Salzmann, Z.; Duranti, A.; Goodwin, C. Rethinking Context: Language as an Interactive Phenomenon; Cambridge University Press: New York, NY, USA, 1992. [Google Scholar]
- Streeck, J.; Hartge, U. Previews: Gestures at the transition place. In The Contextualization of Language; Auer, P., Luzio, A.D., Eds.; John Benjamins Publishing Company: Amsterdam, The Netherlands, 1992; pp. 135–157. [Google Scholar]
- Streeck, J.; Knapp, M.L. The Interaction of Visual and Verbal Features in Human Communication. In Advances in Nonverbal Communication; Poyatos, F., Ed.; John Benjamins Publishing Company: Amsterdam, The Netherlands, 1992; Volume 10, pp. 3–23. [Google Scholar]
- Kendon, A. Some Considerations for a Theory of Language Origins. Man 1991, 26, 199–221. [Google Scholar] [CrossRef]
- McNeill, D. Guide to Gesture Classification, Transcription and Distribution. In Hand Mind What Gestures Reveal about Thought; University of Chicago Press: Chicago, IL, USA, 1992. [Google Scholar]
- Goodwin, C. Gestures as a resource for the organization of mutual orientation. Semiotica 1986, 62, 29–50. [Google Scholar] [CrossRef]
- Goodwin, M.H.; Goodwin, C. Gesture and coparticipation in the activity of searching for a word. Semiotica 1986, 62, 51–76. [Google Scholar]
- Sherzer, J. Verbal and nonverbal deixis: The pointed lip gesture among the San Blas Cuna. Lang. Soc. 1973, 2, 117–131. [Google Scholar] [CrossRef]
- Heath, C.; Luff, P. Media Space and Communicative Asymmetries: Preliminary Observations of Video-Mediated Interaction. Hum.—Comput. Interact. 1992, 7, 315–346. [Google Scholar] [CrossRef]
- Streeck, J. The significance of gesture: How it is established. IPrA Pap. Pragmat. 1988, 2, 60–83. [Google Scholar] [CrossRef]
- Grèzes, J.; Decety, J. Functional anatomy of execution, mental simulation, observation, and verb generation of actions: A meta-analysis. Hum. Brain Mapp. 2000, 12, 1–19. [Google Scholar] [CrossRef]
- Van Overwalle, F. Social cognition and the brain: A meta-analysis. Hum. Brain Mapp. 2009, 30, 829–858. [Google Scholar] [CrossRef] [PubMed]
- Van Overwalle, F.; Baetens, K. Understanding others’ actions and goals by mirror and mentalizing systems: A meta-analysis. Neuroimage 2009, 48, 564–584. [Google Scholar] [CrossRef]
- Gazzola, V.; Keysers, C. The observation and execution of actions share motor and somatosensory voxels in all tested subjects: Single-subject analyses of unsmoothed fMRI data. Cereb. Cortex 2009, 19, 1239–1255. [Google Scholar] [CrossRef]
- Amodio, D.M.; Frith, C.D. Meeting of minds: The medial frontal cortex and social cognition. Nat. Rev. Neurosci. 2006, 7, 268. [Google Scholar] [CrossRef] [PubMed]
- Kendon, A. Reflections on the “gesture-first” hypothesis of language origins. Psychon. Bull. Rev. 2017, 24, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Tomasello, M.; Carpenter, M.; Call, J.; Behne, T.; Moll, H. Understanding and sharing intentions: The origins of cultural cognition. Behav. Brain Sci. 2005, 28, 675–691. [Google Scholar] [CrossRef] [PubMed]
- Enfield, N.J. ‘Lip-pointing’: A discussion of form and function with reference to data from Laos. Gesture 2007, 1, 185–211. [Google Scholar] [CrossRef]
- Kita, S. Cross-cultural variation of speech-accompanying gesture: A review. Lang. Cogn. Process. 2009, 24, 145–167. [Google Scholar] [CrossRef]
- Balconi, M.; Grippa, E.; Vanutelli, M.E. What hemodynamic (fNIRS), electrophysiological (EEG) and autonomic integrated measures can tell us about emotional processing. Brain Cogn. 2015, 95, 67–76. [Google Scholar] [CrossRef]
- Davidson, R.J. Emotion and Affective Style: Hemispheric Substrates. Psychol. Sci. 1992, 3, 39–43. [Google Scholar] [CrossRef]
- Holle, H.; Gunter, T.C.; Rüschemeyer, S.A.; Hennenlotter, A.; Iacoboni, M. Neural correlates of the processing of co-speech gestures. Neuroimage 2008, 39, 2010–2024. [Google Scholar] [CrossRef]
- Huxham, C.; Dick, B.; Stringer, E. Theory in action research. Action Res. 2009, 7, 5–12. [Google Scholar]
- Rizzolatti, G.; Sinigaglia, C. The functional role of the parieto-frontal mirror circuit: Interpretations and misinterpretations. Nat. Rev. Neurosci. 2010, 11, 264. [Google Scholar] [CrossRef]
- Shepherd, S.V.; Klein, J.T.; Deaner, R.O.; Platt, M.L. Mirroring of attention by neurons in macaque parietal cortex. Proc. Natl. Acad. Sci. USA 2009, 106, 9489–9494. [Google Scholar] [CrossRef]
- Ishida, H.; Murata, A. Self and other’s body part representation in parietal visuo-tactile bimodal areas. Neurosci. Res. 2007, 58, S115. [Google Scholar] [CrossRef]
- Dumas, G.; Nadel, J.; Soussignan, R.; Martinerie, J.; Garnero, L. Inter-brain synchronization during social interaction. PLoS ONE 2010, 5, e12166. [Google Scholar] [CrossRef]
- Konvalinka, I.; Bauer, M.; Stahlhut, C.; Hansen, L.K.; Roepstorff, A.; Frith, C.D. Frontal alpha oscillations distinguish leaders from followers: Multivariate decoding of mutually interacting brains. Neuroimage 2014, 94, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Lindenberger, U.; Li, S.C.; Gruber, W.; Müller, V. Brains swinging in concert: Cortical phase synchronization while playing guitar. BMC Neurosci. 2009, 10, 22. [Google Scholar] [CrossRef]
- Balconi, M.; Gatti, L.; Vanutelli, M.E. When cooperation goes wrong: Brain and behavioural correlates of ineffective joint strategies in dyads. Int. J. Neurosci. 2018, 128, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Montague, P.R.; Berns, G.S.; Cohen, J.D.; McClure, S.M.; Pagnoni, G.; Dhamala, M.; Wiest, M.C.; Karpov, I.; King, R.D.; Apple, N.; et al. Hyperscanning: Simultaneous fMRI during linked social interactions. Neuroimage 2002, 1159–1164. [Google Scholar] [CrossRef] [PubMed]
- Hari, R.; Kujala, M.V. Brain Basis of Human Social Interaction: From Concepts to Brain Imaging. Physiol. Rev. 2009, 89, 453–479. [Google Scholar] [CrossRef]
- Koike, T.; Tanabe, H.C.; Sadato, N. Hyperscanning neuroimaging technique to reveal the “two-in-one” system in social interactions. Neurosci. Res. 2015, 90, 25–32. [Google Scholar] [CrossRef]
- Konvalinka, I.; Roepstorff, A. The two-brain approach: How can mutually interacting brains teach us something about social interaction? Front. Hum. Neurosci. 2012, 6, 215. [Google Scholar] [CrossRef]
- Liu, T.; Saito, H.; Oi, M. Role of the right inferior frontal gyrus in turn-based cooperation and competition: A near-infrared spectroscopy study. Brain Cogn. 2015, 99, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Ruby, P.; Decety, J. How would you feel versus how do you think She would feel? A neuroimaging study of perspective-taking with social emotions. J. Cogn. Neurosci. 2004, 16, 988–999. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Bryant, D.M.; Reiss, A.L. NIRS-based hyperscanning reveals increased interpersonal coherence in superior frontal cortex during cooperation. Neuroimage 2012, 59, 2430–2437. [Google Scholar] [CrossRef] [PubMed]
- Balconi, M.; Pezard, L.; Nandrino, J.L.; Vanutelli, M.E. Two is better than one: The effects of strategic cooperation on intra- and inter-brain connectivity by fNIRS. PLoS ONE 2017, 12, e0187652. [Google Scholar] [CrossRef] [PubMed]
- Nozawa, T.; Sasaki, Y.; Sakaki, K.; Yokoyama, R.; Kawashima, R. Interpersonal frontopolar neural synchronization in group communication: An exploration toward fNIRS hyperscanning of natural interactions. Neuroimage 2016, 133, 484–497. [Google Scholar] [CrossRef]
- Balconi, M.; Canavesio, Y. High-frequency rTMS on DLPFC increases prosocial attitude in case of decision to support people. Soc. Neurosci. 2014, 9, 82–93. [Google Scholar] [CrossRef]
- Balconi, M.; Fronda, G.; Vanutelli, M.E. Donate or receive? Social hyperscanning application with fNIRS. Curr. Psychol. 2019, 38, 1–12. [Google Scholar] [CrossRef]
- Crivelli, D.; Balconi, M. Agentività e competenze sociali: Riflessioni teoriche e implicazioni per il management. Ricerche di Psicologia 2017, 349–363. [Google Scholar] [CrossRef]
- Balconi, M.; Gatti, L.; Vanutelli, M.E. Cooperate or not cooperate EEG, autonomic, and behavioral correlates of ineffective joint strategies. Brain Behav. 2018, 8, e00902. [Google Scholar] [CrossRef]
- Balconi, M.; Bortolotti, A. Emotional face recognition, empathic trait (BEES), and cortical contribution in response to positive and negative cues.The effect of rTMS on dorsal medial prefrontal cortex. Cogn. Neurodynamics 2013, 7, 13–21. [Google Scholar] [CrossRef]
- Balconi, M.; Bortolotti, A. Detection of the facial expression of emotion and self-report measures in empathic situations are influenced by sensorimotor circuit inhibition by low-frequency rTMS. Brain Stimul. 2012, 5, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Balconi, M.; Caldiroli, C. Semantic violation effect on object-related action comprehension. N400-like event-related potentials for unusual and incorrect use. Neuroscience 2011, 197, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Balconi, M.; Bortolotti, A.; Gonzaga, L. Emotional face recognition, EMG response, and medial prefrontal activity in empathic behaviour. Neurosci. Res. 2011, 71, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Balconi, M.; Falbo, L.; Conte, V.A. BIS and BAS correlates with psychophysiological and cortical response systems during aversive and appetitive emotional stimuli processing. Motiv. Emot. 2012, 36, 218–231. [Google Scholar] [CrossRef]
- Rameson, L.T.; Lieberman, M.D. Empathy: A Social Cognitive Neuroscience Approach. Soc. Personal. Psychol. Compass 2009, 3, 94–110. [Google Scholar] [CrossRef]
- Rosso, I.M.; Young, A.D.; Femia, L.A.; Yurgelun-Todd, D.A. Cognitive and emotional components of frontal lobe functioning in childhood and adolescence. Ann. N. Y. Acad. Sci. 2004, 1021, 355–362. [Google Scholar] [CrossRef]
- Perry, A.; Stein, L.; Bentin, S. Motor and attentional mechanisms involved in social interaction-Evidence from mu and alpha EEG suppression. Neuroimage 2011, 58, 895–904. [Google Scholar] [CrossRef]
- Rushworth, M.F.S.; Krams, M.; Passingham, R.E. The attentional role of the left parietal cortex: The distinct lateralization and localization of motor attention in the human brain. J. Cogn. Neurosci. 2001, 13, 698–710. [Google Scholar] [CrossRef]
- Ahern, G.L.; Schwartz, G.E. Differential lateralization for positive and negative emotion in the human brain: EEG spectral analysis. Neuropsychologia 1985, 23, 745–755. [Google Scholar] [CrossRef]
- Lindsley, D.B.; Wicke, J.D. The Electroencephalogram: Autonomous Electrical Activity in Man and Animals. In Bioelectric Recording Techniques; Thompson, R.F., Patterson, M.M., Eds.; Academic Press: New York, NY, USA, 1974; pp. 3–83. [Google Scholar]
- Marshall, P.J.; Bouquet, C.A.; Shipley, T.F.; Young, T. Effects of brief imitative experience on EEG desynchronization during action observation. Neuropsychologia 2009, 47, 2100–2106. [Google Scholar] [CrossRef]
- Quandt, L.C.; Marshall, P.J.; Bouquet, C.A.; Young, T.; Shipley, T.F. Experience with novel actions modulates frontal alpha EEG desynchronization. Neurosci. Lett. 2011, 499, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Quandt, L.C.; Marshall, P.J.; Shipley, T.F.; Beilock, S.L.; Goldin-Meadow, S. Sensitivity of alpha and beta oscillations to sensorimotor characteristics of action: An EEG study of action production and gesture observation. Neuropsychologia 2012, 50, 2745–2751. [Google Scholar] [CrossRef]
- Balconi, M.; Vanutelli, M.E. Interbrains cooperation: Hyperscanning and self-perception in joint actions. J. Clin. Exp. Neuropsychol. 2017, 39, 607–620. [Google Scholar] [CrossRef] [PubMed]
- Vanutelli, M.E.; Nandrino, J.L.; Balconi, M. The boundaries of cooperation: Sharing and coupling from ethology to neuroscience. Neuropsychol. Trends 2016, 19, 83–104. [Google Scholar] [CrossRef]
- Caplan, B. Putting things in contexts. Philos. Rev. 2003, 112, 191–214. [Google Scholar] [CrossRef]
- Ekstrom, A.D.; Caplan, J.B.; Ho, E.; Shattuck, K.; Fried, I.; Kahana, M.J. Human hippocampal theta activity during virtual navigation. Hippocampus 2005, 15, 881–889. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.W.; Wilson, M.A. Phase precession of medial prefrontal cortical activity relative to the hippocampal theta rhythm. Hippocampus 2005, 15, 867–873. [Google Scholar] [CrossRef]
- Sirota, A.; Montgomery, S.; Fujisawa, S.; Isomura, Y.; Zugaro, M.; Buzsáki, G. Entrainment of Neocortical Neurons and Gamma Oscillations by the Hippocampal Theta Rhythm. Neuron 2008, 60, 683–697. [Google Scholar] [CrossRef]
- Ludwig, K.A.; Miriani, R.M.; Langhals, N.B.; Joseph, M.D.; Anderson, D.J.; Kipke, D.R. Using a Common Average Reference to Improve Cortical Neuron Recordings from Microelectrode Arrays. J. Neurophysiol. 2009, 101, 1679–1689. [Google Scholar] [CrossRef] [PubMed]
- Keil, A.; Stolarova, M.; Heim, S.; Gruber, T.; Müller, M.M. Temporal Stability of High-Frequency Brain Oscillations in the Human EEG. Brain Topogr. 2003, 16, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Mathewson, K.E.; Lleras, A.; Beck, D.M.; Fabiani, M.; Ro, T.; Gratton, G. Pulsed out of awareness: EEG alpha oscillations represent a pulsed-inhibition of ongoing cortical processing. Front. Psychol. 2011, 2, 99. [Google Scholar] [CrossRef]
- Van Ede, F.; De Lange, F.; Jensen, O.; Maris, E. Orienting attention to an upcoming tactile event involves a spatially and temporally specific modulation of sensorimotor alpha- and beta-band oscillations. J. Neurosci. 2011, 31, 2016–2024. [Google Scholar] [CrossRef] [PubMed]
- Posner, M.I.; Walker, J.A.; Friedrich, F.J.; Rafal, R.D. Effects of parietal injury on covert orienting of attention. J. Neurosci. 1984, 4, 1863–1874. [Google Scholar] [CrossRef] [PubMed]
- Mu, Y.; Fan, Y.; Mao, L.; Han, S. Event-related theta and alpha oscillations mediate empathy for pain. Brain Res. 2008, 1234, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Balconi, M.; Vanutelli, M.E. Competition in the brain. The contribution of EEG and fNIRS modulation and personality effects in social ranking. Front. Psychol. 2016, 7, 1587. [Google Scholar] [CrossRef] [PubMed]
- De Stefani, E.; Innocenti, A.; Secchi, C.; Papa, V.; Gentilucci, M. Type of gesture, valence, and gaze modulate the influence of gestures on observer’s behaviors. Front. Hum. Neurosci. 2013, 7, 542. [Google Scholar] [CrossRef] [PubMed]
- Di Pellegrino, G.; Fadiga, L.; Fogassi, L.; Gallese, V.; Rizzolatti, G. Understanding motor events: A neurophysiological study. Exp. Brain Res. 1992, 91, 176–180. [Google Scholar] [CrossRef]
- Gallese, V.; Fadiga, L.; Fogassi, L.; Rizzolatti, G. Action recognition in the premotor cortex. Brain 1996, 132, 1685–1689. [Google Scholar] [CrossRef]
- Rizzolatti, G.; Fadiga, L.; Gallese, V.; Fogassi, L. Premotor cortex and the recognition of motor actions. Cogn. Brain Res. 1996, 3, 131–141. [Google Scholar] [CrossRef]
- Calvo-Merino, B.; Glaser, D.E.; Grèzes, J.; Passingham, R.E.; Haggard, P. Action observation and acquired motor skills: An fMRI study with expert dancers. Cereb. Cortex 2005, 15, 1243–1249. [Google Scholar] [CrossRef]
- Hari, R.; Henriksson, L.; Malinen, S.; Parkkonen, L. Centrality of Social Interaction in Human Brain Function. Neuron 2015, 88, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Balconi, M.; Canavesio, Y. Emotional contagion and trait empathy in prosocial behavior in young people: The contribution of autonomic (facial feedback) and balanced emotional empathy scale (BEES) measures. J. Clin. Exp. Neuropsychol. 2013, 35, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Knoblich, G.; Butterfill, S.; Sebanz, N. Psychological Research on Joint Action: Theory and Data. In Psychology of Learning and Motivation; Ross, B.H., Ed.; Academic Press: Burlington, MA, USA, 2011; ISBN 9780123855275. [Google Scholar]
- Kamarajan, C.; Rangaswamy, M.; Chorlian, D.B.; Manz, N.; Tang, Y.; Pandey, A.K.; Roopesh, B.N.; Stimus, A.T.; Porjesz, B. Theta oscillations during the processing of monetary loss and gain: A perspective on gender and impulsivity. Brain Res. 2008, 1235, 45–62. [Google Scholar] [CrossRef] [PubMed]
- Knyazev, G.G.; Bocharov, A.V.; Levin, E.A.; Savostyanov, A.N.; Slobodskoj-Plusnin, J.Y. Anxiety and oscillatory responses to emotional facial expressions. Brain Res. 2008, 1227, 174–188. [Google Scholar] [CrossRef] [PubMed]
- Knyazev, G.G.; Slobodskoj-Plusnin, J.Y. Behavioural approach system as a moderator of emotional arousal elicited by reward and punishment cues. Personal. Individ. Differ. 2007, 42, 49–59. [Google Scholar] [CrossRef]
- Khalighinejad, N.; Schurger, A.; Desantis, A.; Zmigrod, L.; Haggard, P. Precursor processes of human self-initiated action. Neuroimage 2018, 165, 35–47. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balconi, M.; Fronda, G. The Use of Hyperscanning to Investigate the Role of Social, Affective, and Informative Gestures in Non-Verbal Communication. Electrophysiological (EEG) and Inter-Brain Connectivity Evidence. Brain Sci. 2020, 10, 29. https://doi.org/10.3390/brainsci10010029
Balconi M, Fronda G. The Use of Hyperscanning to Investigate the Role of Social, Affective, and Informative Gestures in Non-Verbal Communication. Electrophysiological (EEG) and Inter-Brain Connectivity Evidence. Brain Sciences. 2020; 10(1):29. https://doi.org/10.3390/brainsci10010029
Chicago/Turabian StyleBalconi, Michela, and Giulia Fronda. 2020. "The Use of Hyperscanning to Investigate the Role of Social, Affective, and Informative Gestures in Non-Verbal Communication. Electrophysiological (EEG) and Inter-Brain Connectivity Evidence" Brain Sciences 10, no. 1: 29. https://doi.org/10.3390/brainsci10010029
APA StyleBalconi, M., & Fronda, G. (2020). The Use of Hyperscanning to Investigate the Role of Social, Affective, and Informative Gestures in Non-Verbal Communication. Electrophysiological (EEG) and Inter-Brain Connectivity Evidence. Brain Sciences, 10(1), 29. https://doi.org/10.3390/brainsci10010029





