1. Introduction
Fluorescence correlation spectroscopy (FCS) is a technique that provides molecular diffusion dynamics and the number of molecules of fluorescently labeled target molecules [
1,
2,
3,
4]. The FCS system comprise a confocal fluorescence detection system. The number of target molecules inside the confocal volume (measurement volume) fluctuates owing to the translational diffusion in the sample solution. As a result, the detected fluorescence intensity signal fluctuated. This means that the fluctuation analysis of the fluorescence intensity signal can provide information about translational diffusion, which is a concept of FCS. Because FCS measurements can be performed even in living cells with low damage, FCS is a useful tool, especially in the field of biology [
5]. For example, Sigaut et al. analyzed the mobility of a focal adhesion protein zyxin at focal adhesions of living HC11 cells by FCS. They found that the stiffness of the substrate for cell culturing did not affected to the mobility of zyxin [
6]. A polarization dependent FCS (Pol-FCS), an advanced method of FCS, was used to evaluate the macromolecular crowding environment in cells [
7]. Reentry, FCS is used also in the field of soft matter to evaluate the structure of materials [
7,
8].
In FCS, the speed of fluctuation of fluorescence intensity is analyzed by transforming a fluorescence intensity signal into an auto-correlation function (ACF). The characteristic decay time of the ACF, which is called the diffusion time in FCS, is inversely proportional to the diffusion coefficient of the target molecules. The amplitude of the ACF is inversely proportional to the number of molecules in the measurement volume. If a sample includes two species with different diameters, the ACF is a superposition of two ACFs with different diffusion times as described by Equation (A9) in
Appendix A. If the two ACFs are separated by fitting analysis of the resulting ACF, the diffusion coefficients and the number of molecules can be obtained for each species. Therefore, FCS can be used to evaluate the molecular interactions. For example, Bhunia et al. analyzed the complex formation of enhanced green fluorescent protein (EGFP) and anti-EGFP antibodies [
9]. Lagerkvist et al. established a phage display based on FCS. They successfully detected specific interactions between fluorescently labeled antibodies and their antigens using FCS [
10]. Thus, FCS is useful for evaluating molecular interactions.
However, if the sample contains two species with different brightness levels, interpreting the result is difficult. The estimated number of each species becomes incorrect because their amplitude in the ACF is weighted, corresponding to the brightness of each species [
5,
11]. We can correctly obtain the number of each species only if their brightness was measured in advance. Tiwari et al. successfully measured the dimerization of green fluorescent protein-fused human glucocorticoid receptor alpha (GFP-GRα) using FCS. They assumed that there were only monomers and dimers of GFP-GRα, and the brightness of dimers was two times larger than that of the monomer. In addition, the brightness of the GFP-GRα monomer was measured as a negative control before the sample measurement [
12]. There are similar approaches for the dimer and oligomer formation analysis [
13,
14,
15,
16]. Katoozi et al. proposed the high-order image correlation spectroscopy (HICS) which is based on high-order ACF analysis [
17]. They successfully evaluate the numbers of each species and the brightness ratio between multiple species without any assumption. This approach may be useful for FCS too. However, most of the available FCS instruments and hardware correlators do not provide high-order autocorrelation function so far. Introducing high-order ACF is difficult for current FCS users.
The brightness of a target molecule is characterized by the photon count rate per molecule, which is called the count per molecule (CPM), because the fluorescence intensity is measured by a photon counting method in FCS. The CPM of the second species is often unknown in applications, such as multimer, aggregation, antigen, and antibody response of one-to-many association. Methods to analyze the ACF of FCS correctly in the case of samples containing multiple brightness species are strongly desired in FCS.
In this report, we introduced a calculation procedure for FCS analysis for samples containing multiple brightness species. This calculation can be applied if the diffusion time of each species is obtained separately, and the brightness of M-1 species in M species is determined before measurement. Furthermore, the calculation requires only the first order ACF, which is provided by general FCS instruments, and its fitting results are based on the common model equation. We describe the simulation results of the calculation and its experimental demonstration.
2. Materials and Methods
2.1. Monte Carlo Simulation of Fluorescence Intensity Signal in FCS Measurement
To validate our correction method for a number of molecules and CPM in the FCS, a Monte Carlo simulation was performed. Fluorescence intensity signals from a sample solution containing two species with different CPMs were generated. The details of this simulation are provided in
Appendix B.
Ten fluorescence intensity signals with a duration of 10 s and a time interval of 1 ms were generated. The lateral radius of the confocal volume was nm, and the axial radius was nm. The measurement volume of the FCS, which is called the effective measurement volume, was m3 (0.435 fL). The diameters of the first and second species were 10 and 100 nm, respectively. The medium was water with a viscosity of 0.89 cP, and the absolute temperature was 300 K. In this case, the diffusion coefficient of the first species was 49.4 μm2/s, and the typical step length in Equations (A22)–(A24) (the square root of the mean square displacement) was nm. This was shorter than the lateral diameter of the confocal volume, however it was not enough short to correctly analyze the diffusion time. However, the amplitudes of the ACF, which is focused on in this work, can be correctly obtained because that is given by only the variance and the average of fluorescence intensity signal as obtained by substituting for Equation (A5) in the case that the shot noise can be negligible.
2.2. Number of Molecules Calculation
It is assumed that there are two species: fluorescently labeled small molecules and larger particles containing multiple fluorescent dyes, such as aggregates. If the CPM of the first species
can be determined by FCS measurement on a pure solution in advance, the number of the first species
can be estimated by applying the proposed calculation. The CPM of the second species
and the number
were also estimated by further correction. The details of the correction method are provided in
Appendix A.
2.3. FCS Measurement
Model experiments were performed to experimentally demonstrate the proposed correction method. The FCS measurements were performed on mixtures of two fluorescent latex beads, a fluorescent bead with a diameter of 30 nm (PSYF030NM, MAGSPHERE, Pasadena, CA, USA), and a brighter bead with a diameter of 51 nm (PSYF050NM, MAGSPHERE, Pasadena, CA, USA). A compact FCS system (307-15471, Wako, Japan) was used for the measurements. The lateral diameter of the confocal volume was 200.3 nm, and the axial diameter was 1.14 μm. The effective measurement volume was determined to be 0.255 fL. The excitation wavelength was 473 nm. The excitation laser power was 0.5 μW at the focal point. The 3 s measurement was repeated 20 times, and one mean ACF was obtained. The non-linear least squares analysis was performed on the mean ACF for one experiment.
3. Results
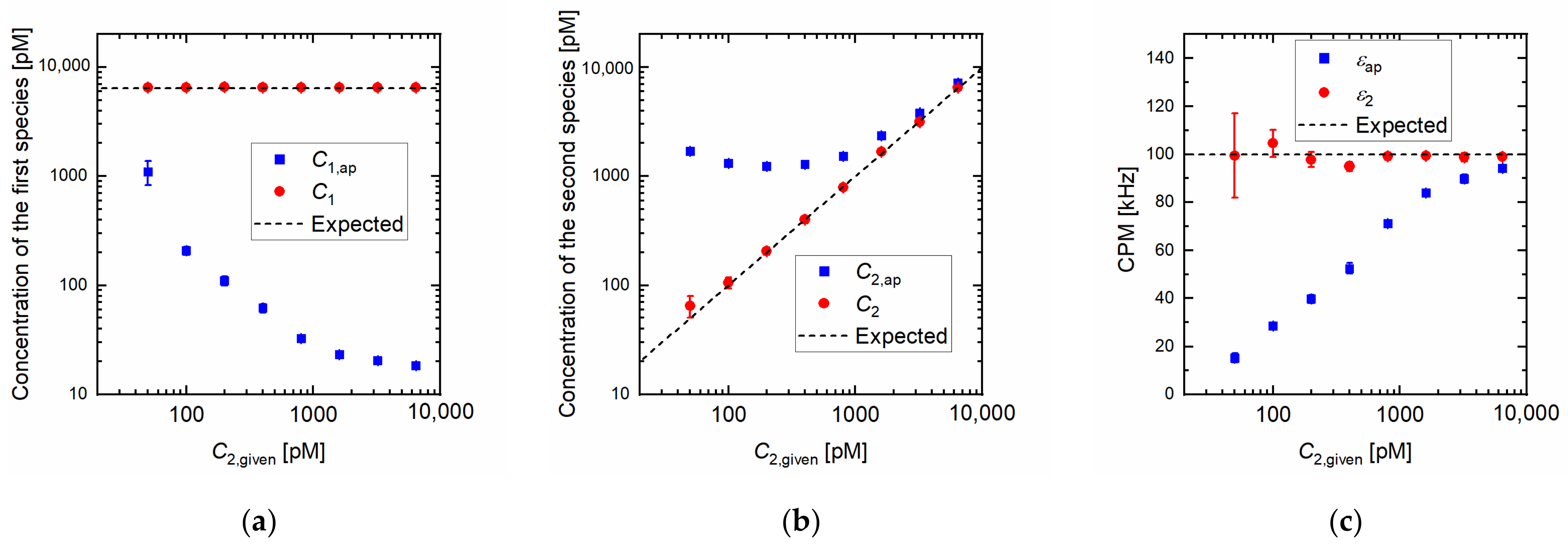
Figure 1 shows the dependency of the estimated parameters on the concentration of the second species in the Monte Carlo simulation. The CPMs of the first and second species were 5 and 100 kHz, respectively. We assumed that the CPM and the diffusion time of the first species were determined by measuring the pure solution of the first species, and we can correctly separate the fractions of the first and second species in the obtained ACF. The number and CPM of the second species were unknown. The concentration of the first species was 6400 pM, and the second species was the dilution series in the generation of the fluorescence signal by simulation described in
Appendix B.
Figure 1a shows a comparison of the estimated concentration of the first species
and the given concentration of the second species
. The apparent concentration
, which was obtained using Equation (A11), of the first species decreased with an increase in the
despite the concentration of the first species being independent of the concentration of the second species. Note that the apparent concentration obtained by Equation (A11) is not correct value and should not be used in the case of the samples containing multiple brightness species because the apparent value is theoretically incorrect and the discrepancy between the apparent values and the expected value can be large as shown in the
Figure 1. Conversely, the concentration of the first species obtained by Equations (A14) and (A21)
was independent of the
, and the values agreed well with the expected value.
Figure 2a shows a comparison between the estimated
and the given
. The error of the apparent concentration of the second species
was large, especially at low concentrations of brighter species; however, the concentration obtained by our calculation
was in good agreement with the expected concentration.
Figure 1c shows the results of the CPM. The apparent CPM is given by Equation (A12) in
Appendix A. The apparent CPM increased with the increase in the number of brighter species and approached the given CPM of the second species. In contrast, the corrected CPM was in good agreement with the given CPM of the second species.
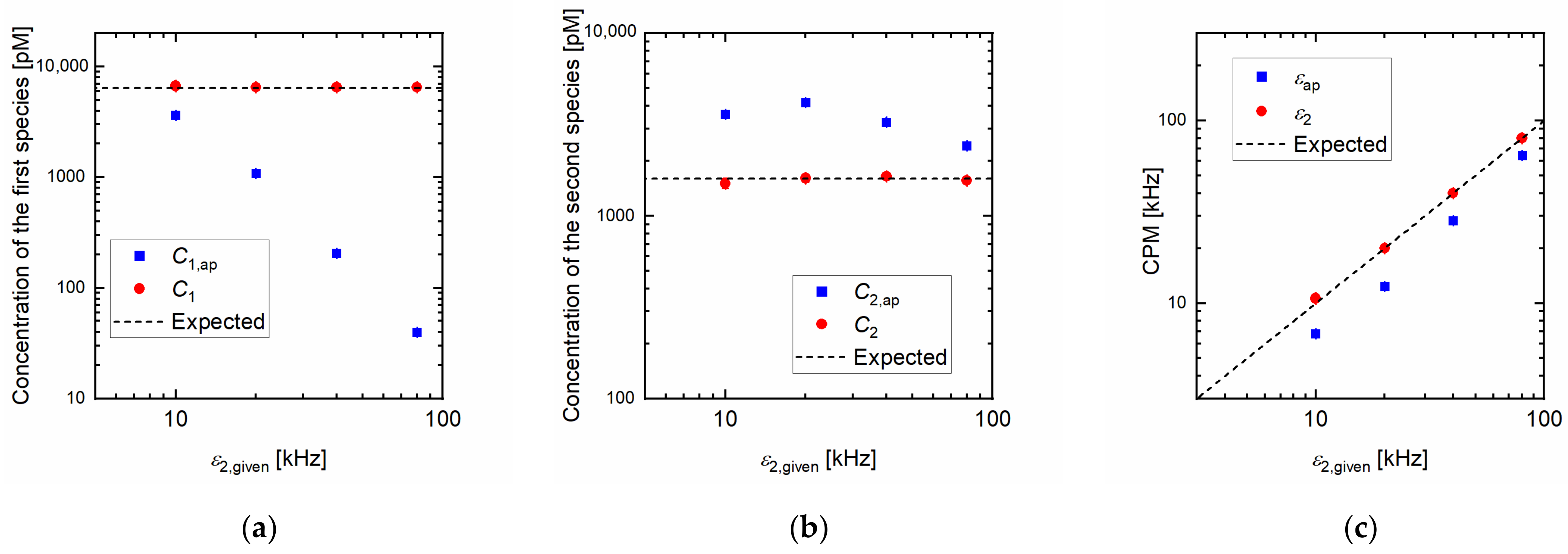
Figure 2 shows the dependency of the estimated parameters on the CPM of the second species
in the Monte Carlo simulation. The given
and
were fixed at 6400 and 1600 pM, respectively. The given CPM of the first species
was fixed at 5 kHz. The error of
became larger with the increase in the given
, and the error of
became smaller, as shown in
Figure 2a,b. The apparent CPM
was always underestimated, as shown in
Figure 2c.
In contrast, the values obtained by the calculation we proposed are in good agreement with the expected values.
Finally, we applied the correction method to the experimental results.
Figure 3 shows the results of FCS measurement on the mixture of two fluorescent beads: Beads1 with a diameter of 30 nm and Beads2 with a diameter of 51 nm. The CPM of Beads1 and Beads2 were measured as
and
kHz, respectively, in pure solutions. The number of Beads1 was fixed at 57.9 (final concentration), and the dilution series of Beads2 was mixed into the Beads1 solution. In the fitting analysis of the obtained ACFs, the model Equation (A9) was used for two species. The diffusion time of the first species was fixed to the value measured in pure solutions to improve the accuracy of the fitting because we assumed that the first species was known. The measurements were performed four times on the same samples, but at different positions in the solution (
). The numbers of the first species
obtained by Equation (A14) agreed well with the expected values, as shown in
Figure 3a. The numbers of the second species
obtained by Equation (A17), also agreed with the expected values in the range of dilution ratio of Beads2 was higher than 0.1 as shown in
Figure 3b. In the range of dilution ratio of Beads2 lower than 0.1, the error of
was large. This was likely caused by too low concentration of the Beads2.
Figure 3c shows the apparent CPM
and the corrected CPM
2 . In the high concentration range of Beads2, some data points took the values around the expected value. There were also the data points with
higher than the expected value. This was likely indicating that there was some small aggregation of fluorescent beads. The data points of
were decreased from
n=8 because the corrected
was 0 at some data points, and we could not calculate
because of division by zero in that case.
4. Discussion
We investigated the dependency of the estimated parameters on the concentration of the second species (
Figure 1). The CPMs of the first and second species,
and
, were 5 and 100 kHz, respectively. It was assumed that the first species is known and had been measured by FCS in pure solution in advance, and the second species is its aggregation or molecules labeled by multiple first species. Therefore, the concentration of the second species
was lower than that of the first species
, and
in the simulations. The results demonstrate the effectiveness of the proposed correction method.
Figure 1a shows that the apparent
can be underestimated by one order even if the
is two orders lower than
in the simulation condition.
Figure 1b,c show that the concentration and CPM of brighter species are likely to be measured without our calculation if the concentration of the brighter species is sufficiently high because, in that case, the contribution of the brighter species in fluorescence intensity becomes much larger than that of the first species. The deviation of the
is relatively large. This is likely because the error in the estimation of
is enhanced as the
was estimated based on the estimated value of
.
The dependency of the estimated parameters on the given CPM of the second species
is also shown in
Figure 2. It was shown that the error of
became larger with the increase in
. Thus, the FCS results can be easily distorted by contamination of the brighter species in the samples.
In the experiment on the mix of two fluorescent bead solutions with different brightness, the calculation of
worked well; however, the accuracies of the calculation on
and
were lower than those on the
. This was likely caused by the estimation of
and
based on the estimated value of
as shown in
Appendix A. To obtain accurate values of
and
, the measurement should be performed carefully and accurately. Small differences in particle brightness owing to hardware conditions, such as laser power fluctuation and/or changing fluorescence collection efficiency by changing the light path during the measurement can deviate the corrected values.
5. Conclusions
In this work, we proposed a calculation procedure for the concentration (number of molecules/effective measurement volume) and brightness (CPM) in FCS measurements on samples containing multiple species with different CPMs. From the simulations, it was shown that the presence of the second species with a higher CPM than the first species can distort the results of FCS.
If all CPMs of every species in a sample are known, we can correctly analyze the number of molecules in each species [
11]. On the other hand, the proposed calculation procedure can be applied if the CPM of only the first species is known, and the fraction of each species can be accurately separated by fitting the analysis of FCS. It was shown that the proposed calculation worked well by the simulations and the experiments.
Theoretically, this calculation can be expanded to include more species. However, it may not be applicable practically because an increase in the species should degrade the accuracy.
We expect that accurate measurement of the mixtures, for example, the sample containing the monomer and its aggregation or its oligomer, and the sample containing the free fluorescent antibody and the particles associated with multiple antibodies, will be available using FCS and our calculation procedure.