Effect of Mechanobiology of Cell Response on Titanium with Multilayered Aluminum Nitride/Tantalum Thin Film
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of the Investigated Specimens
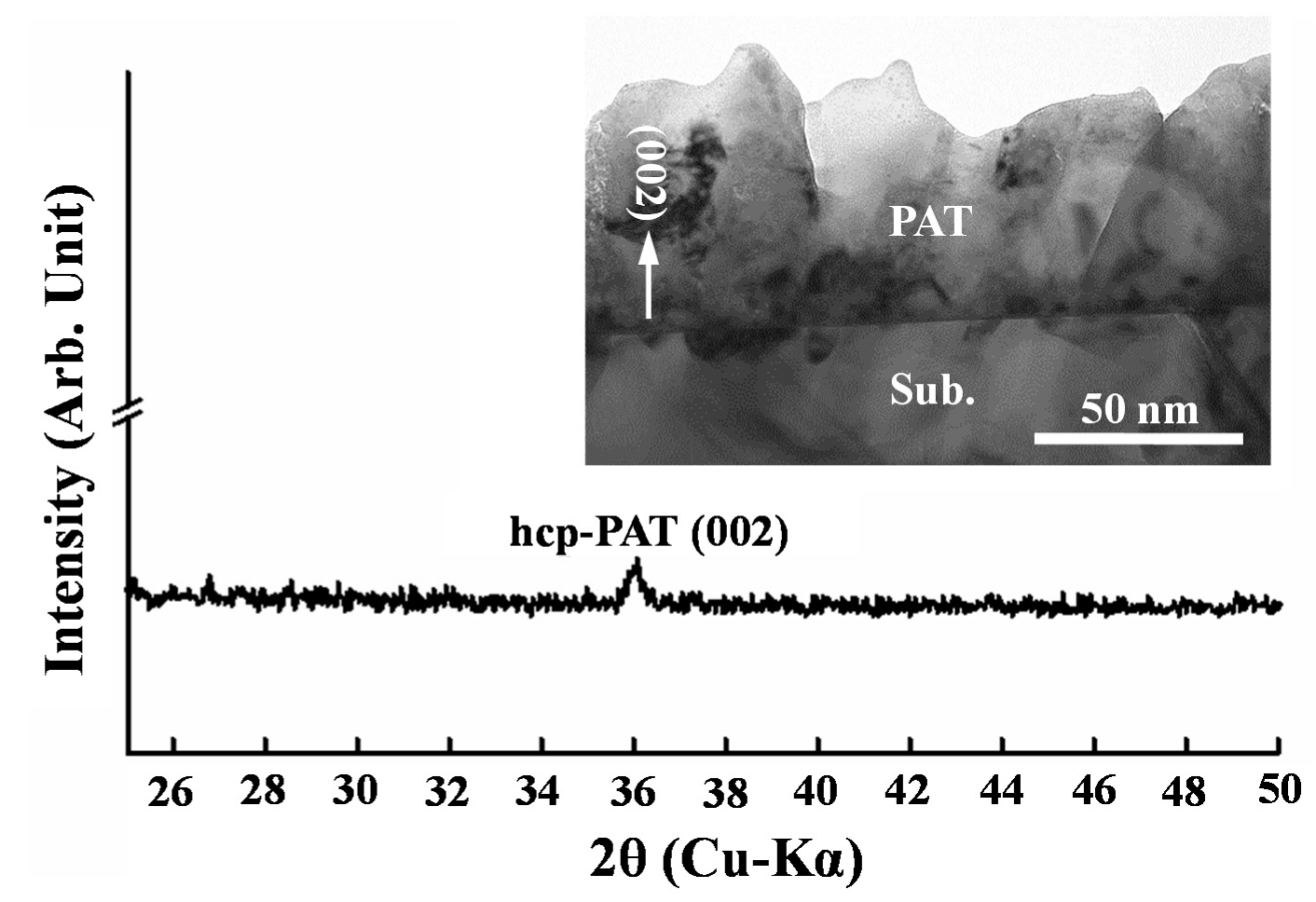
2.2. Microstructure Analysis
2.3. Cell Culturing
2.4. Biosensing Capability
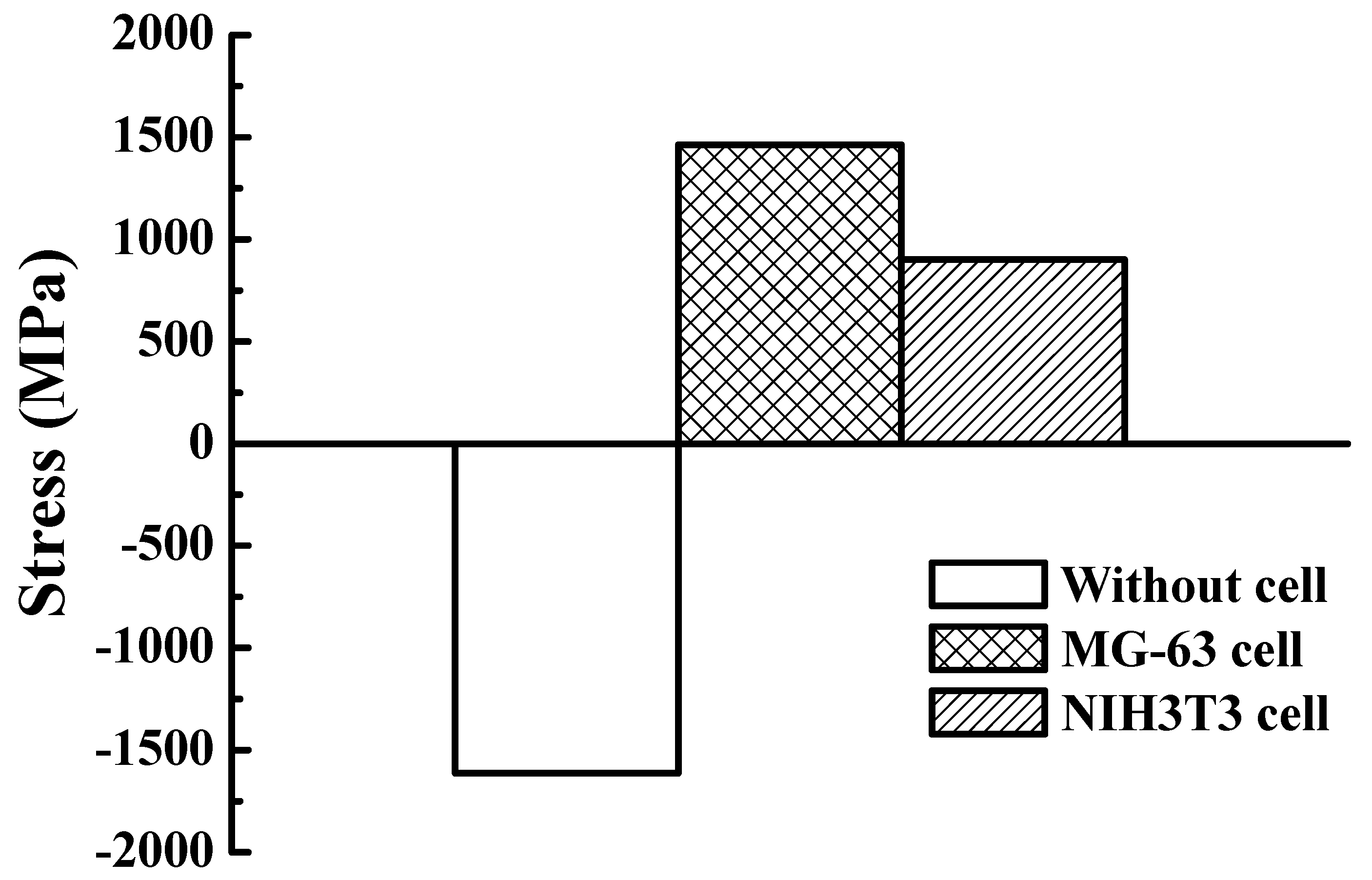
2.5. Stress Variation Measurement
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tsai, M.H.; Haung, C.F.; Shyu, S.S.; Chou, Y.R.; Lin, M.H.; Peng, P.W.; Ou, K.L.; Yu, C.H. Surface modification induced phase transformation and structure variation on the rapidly solidified recast layer of titanium. Mater. Charact. 2015, 106, 463–469. [Google Scholar] [CrossRef]
- Hou, P.J.; Ou, K.L.; Wang, C.C.; Huang, C.F.; Ruslin, M.; Sugiatno, E.; Yang, T.S.; Chou, H.H. Hybrid micro/nanostructural surface offering improved stress distribution and enhanced osseointegration properties of the biomedical titanium implant. J. Mech. Behav. Biomed. 2018, 79, 173–180. [Google Scholar] [CrossRef]
- Chiang, H.J.; Chou, H.H.; Ou, K.L.; Sugiatno, E.; Ruslin, M.; Waris, R.A.; Huang, C.F.; Liu, C.M.; Peng, P.W. Evaluation of Surface Characteristics and Hemocompatibility on the Oxygen Plasma-Modified Biomedical Titanium. Metals 2018, 8, 513. [Google Scholar] [CrossRef]
- Wu, C.M.; Liu, C.M.; Ou, K.L.; Chiang, H.J.; Sugiatno, E.; Wu, C.H.; Yen, H.J.; Chou, H.H. Nanostructured titanium dioxide layer combined with reactive functional groups as a promising biofunctional surface for biomedical applications. Ceram. Int. 2019, 45, 9712–9718. [Google Scholar] [CrossRef]
- Cheng, H.C.; Lee, S.Y.; Chen, C.C.; Shyng, Y.C.; Ou, K.L. Titanium nanostructural surface processing for improved biocompatibility. Appl. Phys. Lett. 2006, 89. [Google Scholar] [CrossRef]
- Chen, S.L.; Lin, M.H.; Chen, C.C.; Ou, K.L. Effect of electro-discharging on formation of biocompatible layer on implant surface. J. Alloys Compd. 2008, 456, 413–418. [Google Scholar] [CrossRef]
- Ou, K.L.; Lin, C.T.; Chen, S.L.; Huang, C.F.; Cheng, H.C.; Yeh, Y.M.; Lin, K.H. Effect of multi-nano-titania film on proliferation and differentiation of mouse fibroblast cell on titanium. J. Electrochem. Soc. 2008, 155, E79–E84. [Google Scholar] [CrossRef]
- Ou, K.L.; Chen, C.C.; Lin, C.T.; Chen, C.S.; Lin, C.C.; Lee, S.Y. Application of aluminum nitride thin film on biosensing of cell differentiation. J. Electrochem. Soc. 2007, 154, P11–P15. [Google Scholar] [CrossRef]
- Kaluderovic, M.R.; Krajnovic, T.; Maksimovic-Ivanic, D.; Graf, H.L.; Mijatovic, S. Ti-SLActive and TiZr-SLActive Dental Implant Surfaces Promote Fast Osteoblast Differentiation. Coatings 2017, 7, 102. [Google Scholar] [CrossRef]
- Pachauri, P.; Bathala, L.R.; Sangur, R. Techniques for dental implant nanosurface modifications. J. Adv. Prosthodont. 2014, 6, 498–504. [Google Scholar] [CrossRef]
- Ou, K.L.; Shih, Y.H.; Huang, C.F.; Chen, C.C.; Liu, C.M. Preparation of bioactive amorphous-like titanium oxide layer on titanium by plasma oxidation treatment. Appl. Surf. Sci. 2008, 255, 2046–2051. [Google Scholar] [CrossRef]
- Haimov, H.; Yosupov, N.; Pinchasov, G.; Juodzbalys, G. Bone Morphogenetic Protein Coating on Titanium Implant Surface: A Systematic Review. J. Oral Maxillofac. Res. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Solar, R.J.; Pollack, S.R.; Korostoff, E. In vitro corrosion testing of titanium surgical implant alloys: An approach to understanding titanium release from implants. J. Biomed. Mater. Res. 1979, 13, 217–250. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.C.; Lin, C.T.; Lee, S.Y.; Lin, L.H.; Huang, C.F.; Ou, K.L. Biosensing of biophysical characterization by metal-aluminum nitride-metal capacitor. Appl. Surf. Sci. 2007, 253, 5173–5178. [Google Scholar] [CrossRef]
- Nishimura, I. Genetic networks in osseointegration. J. Dent. Res. 2013, 92, 109S–118S. [Google Scholar] [CrossRef]
- Smeets, R.; Stadlinger, B.; Schwarz, F.; Beck-Broichsitter, B.; Jung, O.; Precht, C.; Kloss, F.; Grobe, A.; Heiland, M.; Ebker, T. Impact of Dental Implant Surface Modifications on Osseointegration. Biomed. Res. Int. 2016. [Google Scholar] [CrossRef]
- Agarwal, R.; Garcia, A.J. Biomaterial strategies for engineering implants for enhanced osseointegration and bone repair. Adv. Drug Deliv. Rev. 2015, 94, 53–62. [Google Scholar] [CrossRef]
- Gao, X.; Fraulob, M.; Haiat, G. Biomechanical behaviours of the bone-implant interface: A review. J. R. Soc. Interface 2019, 16. [Google Scholar] [CrossRef]
- Duan, Y.; Ma, W.; Li, D.; Wang, T.; Liu, B. Enhanced osseointegration of titanium implants in a rat model of osteoporosis using multilayer bone mesenchymal stem cell sheets. Exp. Ther. Med. 2017, 14, 5717–5726. [Google Scholar] [CrossRef]
- Fraioli, R.; Rechenmacher, F.; Neubauer, S.; Manero, J.M.; Gil, J.; Kessler, H.; Mas-Moruno, C. Mimicking bone extracellular matrix: Integrin-binding peptidomimetics enhance osteoblast-like cells adhesion, proliferation, and differentiation on titanium. Colloids Surf. 2015, 128, 191–200. [Google Scholar] [CrossRef]
- Kasemo, B. Biocompatibility of titanium implants: Surface science aspects. J. Prosthet. Dent. 1983, 49, 832–837. [Google Scholar] [CrossRef]
- Park, J.Y.; Gemmell, C.H.; Davies, J.E. Platelet interactions with titanium: Modulation of platelet activity by surface topography. Biomaterials 2001, 22, 2671–2682. [Google Scholar] [CrossRef]
- Maitz, M.F.; Pham, M.T.; Wieser, E. Blood compatibility of titanium oxides with various crystal structure and element doping. J. Biomater. Appl. 2003, 17, 303–319. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.C.; Cheng, H.C.; Huang, C.F.; Lin, C.T.; Lee, S.Y.; Chen, C.S.; Ou, K.L. Enhancement of biocompatibility on bioactive titanium surface by low-temperature plasma treatment. Jpn. J. Appl. Phys. 2005, 44, 8590–8598. [Google Scholar] [CrossRef]
- Stanford, C.M.; Brand, R.A. Toward an understanding of implant occlusion and strain adaptive bone modeling and remodeling. J. Prosthet. Dent. 1999, 81, 553–561. [Google Scholar] [CrossRef]



© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, M.-S.; Yu, S.-Y.; Chiang, P.-C.; Huang, B.-H.; Saito, T.; Huang, C.-C.; Pai, F.-T.; Wu, C.-Y.; Lan, W.-C. Effect of Mechanobiology of Cell Response on Titanium with Multilayered Aluminum Nitride/Tantalum Thin Film. Appl. Sci. 2020, 10, 645. https://doi.org/10.3390/app10020645
Huang M-S, Yu S-Y, Chiang P-C, Huang B-H, Saito T, Huang C-C, Pai F-T, Wu C-Y, Lan W-C. Effect of Mechanobiology of Cell Response on Titanium with Multilayered Aluminum Nitride/Tantalum Thin Film. Applied Sciences. 2020; 10(2):645. https://doi.org/10.3390/app10020645
Chicago/Turabian StyleHuang, Mao-Suan, Shang-Yang Yu, Pao-Chang Chiang, Bai-Hung Huang, Takashi Saito, Chien-Chia Huang, Fang-Tzu Pai, Chia-Yu Wu, and Wen-Chien Lan. 2020. "Effect of Mechanobiology of Cell Response on Titanium with Multilayered Aluminum Nitride/Tantalum Thin Film" Applied Sciences 10, no. 2: 645. https://doi.org/10.3390/app10020645
APA StyleHuang, M.-S., Yu, S.-Y., Chiang, P.-C., Huang, B.-H., Saito, T., Huang, C.-C., Pai, F.-T., Wu, C.-Y., & Lan, W.-C. (2020). Effect of Mechanobiology of Cell Response on Titanium with Multilayered Aluminum Nitride/Tantalum Thin Film. Applied Sciences, 10(2), 645. https://doi.org/10.3390/app10020645




