An Overview of Antibiotics as Emerging Contaminants: Occurrence in Bivalves as Biomonitoring Organisms
Abstract
:Simple Summary
Abstract
1. Introduction
2. Antibiotics’ Use and Their Presence in Aquatic Environment
3. Methods Used to Quantify Antibiotics in Bivalves
4. Antibiotics Occurrence in Bivalves Collected All around the World
4.1. Occurrence of Antibiotics in Bivalve Mollusks Harvested in Europe
4.2. Occurrence of Antibiotics in Bivalve Mollusks Harvested in Asia
4.3. Occurrence of Antibiotics in Bivalve Mollusks Harvested in America
5. Discussion and Perspectives
6. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ouda, M.; Kadadou, D.; Swaidan, B.; Al-Othman, A.; Al-Asheh, S.; Banat, F.; Hasan, S.W. Emerging contaminants in the water bodies of the Middle East and North Africa (MENA): A critical review. Sci. Total Environ. 2021, 754, 142177. [Google Scholar] [CrossRef]
- Kümmerer, K. Antibiotics in the aquatic environment—A review—Part I. Chemosphere 2009, 75, 417–434. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.-L.; Liu, Y.-S.; Liu, W.-R.; Jiang, Y.-X.; Su, H.-C.; Zhang, Q.-Q.; Chen, X.-W.; Yang, Y.-Y.; Chen, J.; Liu, S.-S.; et al. Tissue-specific bioaccumulation of human and veterinary antibiotics in bile, plasma, liver and muscle tissues of wild fish from a highly urbanized region. Environ. Pollut. 2015, 198, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Shi, Y.; Gao, L.; Liu, J.; Cai, Y. Investigation of antibiotics in mollusks from coastal waters in the Bohai Sea of China. Environ. Pollut. 2012, 162, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zhang, Y.; Zhou, C.; Guo, C.; Wang, D.; Du, P.; Luo, Y.; Wan, J.; Meng, W. Distribution, sources and composition of antibiotics in sediment, overlying water and pore water from Taihu Lake, China. Sci. Total Environ. 2014, 497–498, 267–273. [Google Scholar] [CrossRef]
- Chiesa, L.M.; Nobile, M.; Malandra, R.; Panseri, S.; Arioli, F. Occurrence of antibiotics in mussels and clams from various FAO areas. Food Chem. 2018, 240, 16–23. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Morcillo, S.; Rodríguez-Gil, J.L.; Fernández-Rubio, J.; Rodríguez-Mozaz, S.; Míguez-Santiyán, M.P.; Valdes, M.E.; Barceló, D.; Valcárcel, Y. Presence of pharmaceutical compounds, levels of biochemical biomarkers in seafood tissues and risk assessment for human health: Results from a case study in North-Western Spain. Int. J. Hyg. Environ. Health 2020, 223, 10–21. [Google Scholar] [CrossRef]
- Zouiten, A.; Beltifa, A.; Van Loco, J.; Ben Mansour, H.; Reyns, T. Ecotoxicological potential of antibiotic pollution–industrial wastewater: Bioavailability, biomarkers, and occurrence in Mytilus galloprovincialis. Environ. Sci. Pollut. Res. 2016, 23, 15343–15350. [Google Scholar] [CrossRef]
- Bayen, S.; Estrada, E.S.; Juhel, G.; Kelly, B.C. Direct injection of tissue extracts in liquid chromatography/tandem mass spectrometry for the determination of pharmaceuticals and other contaminants of emerging concern in mollusks. Anal. Bioanal. Chem. 2015, 407, 5553–5558. [Google Scholar] [CrossRef]
- Gaw, S.; Thomas, K.V.; Hutchinson, T. Sources, impacts and trends of pharmaceuticals in the marine and coastal environment. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130572. [Google Scholar] [CrossRef] [Green Version]
- McEneff, G.; Barron, L.; Kelleher, B.; Paull, B.; Quinn, B. A year-long study of the spatial occurrence and relative distribution of pharmaceutical residues in sewage effluent, receiving marine waters and marine bivalves. Sci. Total Environ. 2014, 476-477, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Lána, R.; Vávrová, M.; Navrátil, S.; Brabencová, E.; Večerek, V. Organochlorine Pollutants in Chub, Leuciscus cephalus, from the Svratka River, Czech Republic. Bull. Environ. Contam. Toxicol. 2010, 84, 726–730. [Google Scholar] [CrossRef]
- Varol, M.; Sünbül, M.R. Organochlorine pesticide, antibiotic and heavy metal residues in mussel, crayfish and fish species from a reservoir on the Euphrates River, Turkey. Environ. Pollut. 2017, 230, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Beyer, J.; Green, N.W.; Brooks, S.; Allan, I.J.; Ruus, A.; Gomes, T.; Bråte, I.L.N.; Schøyen, M. Blue mussels (Mytilus edulis spp.) as sentinel organisms in coastal pollution monitoring: A review. Mar. Environ. Res. 2017, 130, 338–365. [Google Scholar] [CrossRef] [PubMed]
- Poisonous Mussels from Impure Waters. Science 1886, 7, 175–176. [CrossRef]
- Goldberg, E.D. The mussel watch—A first step in global marine monitoring. Mar. Pollut. Bull. 1975, 6, 111. [Google Scholar] [CrossRef]
- Meador, J.P.; Stein, J.E.; Reichert, W.L.; Varanasi, U. Bioaccumulation of Polycyclic Aromatic Hydrocarbons by Marine Organisms. Rev. Environ. Contam. Toxicol. 1995, 143, 79–165. [Google Scholar] [CrossRef]
- Strand, J.; Jacobsen, J.A. Accumulation and trophic transfer of organotins in a marine food web from the Danish coastal waters. Sci. Total Environ. 2005, 350, 72–85. [Google Scholar] [CrossRef]
- Stara, A.; Pagano, M.; Capillo, G.; Fabrello, J.; Sandova, M.; Albano, M.; Zuskova, E.; Velisek, J.; Matozzo, V.; Faggio, C. Acute effects of neonicotinoid insecticides on Mytilus galloprovincialis: A case study with the active compound thiacloprid and the commercial formulation calypso 480 SC. Ecotoxicol. Environ. Saf. 2020, 203, 110980. [Google Scholar] [CrossRef]
- Stara, A.; Pagano, M.; Albano, M.; Savoca, S.; Di Bella, G.; Albergamo, A.; Koutkova, Z.; Sandova, M.; Velisek, J.; Fabrello, J.; et al. Effects of long-term exposure of Mytilus galloprovincialis to thiacloprid: A multibiomarker approach. Environ. Pollut. 2021, 289, 117892. [Google Scholar] [CrossRef]
- Matozzo, V.; Bertin, V.; Battistara, M.; Guidolin, A.; Masiero, L.; Marisa, I.; Orsetti, A. Does the antibiotic amoxicillin affect haemocyte parameters in non-target aquatic invertebrates? The clam Ruditapes philippinarum and the mussel Mytilus galloprovincialis as model organisms. Mar. Environ. Res. 2016, 119, 51–58. [Google Scholar] [CrossRef]
- Lacaze, E.; Pédelucq, J.; Fortier, M.; Brousseau, P.; Auffret, M.; Budzinski, H.; Fournier, M. Genotoxic and immunotoxic potential effects of selected psychotropic drugs and antibiotics on blue mussel (Mytilus edulis) hemocytes. Environ. Pollut. 2015, 202, 177–186. [Google Scholar] [CrossRef]
- McEneff, G.; Barron, L.; Kelleher, B.; Paull, B.; Quinn, B. The determination of pharmaceutical residues in cooked and uncooked marine bivalves using pressurised liquid extraction, solid-phase extraction and liquid chromatography–tandem mass spectrometry. Anal. Bioanal. Chem. 2013, 405, 9509–9521. [Google Scholar] [CrossRef]
- Puckowski, A.; Mioduszewska, K.; Łukaszewicz, P.; Borecka, M.; Caban, M.; Maszkowska, J.; Stepnowski, P. Bioaccumulation and analytics of pharmaceutical residues in the environment: A review. J. Pharm. Biomed. Anal. 2016, 127, 232–255. [Google Scholar] [CrossRef]
- Kovaláková, P.; Cizmas, L.; McDonald, T.J.; Marsalek, B.; Feng, M.; Sharma, V.K. Occurrence and toxicity of antibiotics in the aquatic environment: A review. Chemosphere 2020, 251, 126351. [Google Scholar] [CrossRef]
- Chang, X.; Meyer, M.; Liu, X.; Zhao, Q.; Chen, H.; Chen, J.-A.; Qiu, Z.; Yang, L.; Cao, J.; Shu, W. Determination of antibiotics in sewage from hospitals, nursery and slaughter house, wastewater treatment plant and source water in Chongqing region of Three Gorge Reservoir in China. Environ. Pollut. 2010, 158, 1444–1450. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Pei, J.; Zhang, R.; Wang, S.; Zeng, W.; Huang, D.; Wang, Y.; Zhang, Y.; Wang, Y.; Yu, K. Occurrence and distribution of antibiotics in mariculture farms, estuaries and the coast of the Beibu Gulf, China: Bioconcentration and diet safety of seafood. Ecotoxicol. Environ. Saf. 2018, 154, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Mello, F.V.; Cunha, S.C.; Fogaça, F.H.; Alonso, M.B.; Torres, J.P.M.; Fernandes, J.O. Occurrence of pharmaceuticals in seafood from two Brazilian coastal areas: Implication for human risk assessment. Sci. Total Environ. 2022, 803, 149744. [Google Scholar] [CrossRef]
- Felis, E.; Kalka, J.; Sochacki, A.; Kowalska, K.; Bajkacz, S.; Harnisz, M.; Korzeniewska, E. Antimicrobial pharmaceuticals in the aquatic environment—Occurrence and environmental implications. Eur. J. Pharmacol. 2020, 866, 172813. [Google Scholar] [CrossRef]
- Larsson, D.G.J. Antibiotics in the environment. Upsala J. Med. Sci. 2014, 119, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Massey, L.B.; Haggard, B.E.; Galloway, J.M.; Loftin, K.A.; Meyer, M.T.; Green, W.R. Antibiotic fate and transport in three effluent-dominated Ozark streams. Ecol. Eng. 2010, 36, 930–938. [Google Scholar] [CrossRef]
- Wang, J. Analysis of macrolide antibiotics, using liquid chromatography-mass spectrometry, in food, biological and environmental matrices. Mass Spectrom. Rev. 2008, 28, 50–92. [Google Scholar] [CrossRef]
- Chen, H.; Liu, S.; Xu, X.-R.; Liu, S.-S.; Zhou, G.; Sun, K.-F.; Zhao, J.-L.; Ying, G.-G. Antibiotics in typical marine aquaculture farms surrounding Hailing Island, South China: Occurrence, bioaccumulation and human dietary exposure. Mar. Pollut. Bull. 2015, 90, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Bin Zaman, S.; Hussain, M.A.; Nye, R.; Mehta, V.; Mamun, K.T.; Hossain, N. A Review on Antibiotic Resistance: Alarm Bells are Ringing. Cureus 2017, 9, e1403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klein, E.Y.; Van Boeckel, T.P.; Martinez, E.M.; Pant, S.; Gandra, S.; Levin, S.A.; Goossens, H.; Laxminarayan, R. Global increase and geographic convergence in antibiotic consumption between 2000 and 2015. Proc. Natl. Acad. Sci. USA 2018, 115, E3463–E3470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dadgostar, P. Antimicrobial Resistance: Implications and Costs. Infect. Drug Resist. 2019, 12, 3903–3910. [Google Scholar] [CrossRef] [Green Version]
- Sapkota, A.; Sapkota, A.R.; Kucharski, M.; Burke, J.; McKenzie, S.; Walker, P.; Lawrence, R. Aquaculture practices and potential human health risks: Current knowledge and future priorities. Environ. Int. 2008, 34, 1215–1226. [Google Scholar] [CrossRef] [PubMed]
- Bassetti, M.; Poulakou, G.; Ruppé, E.; Bouza, E.; Van Hal, S.J.; Brink, A.J. Antimicrobial resistance in the next 30 years, humankind, bugs and drugs: A visionary approach. Intensive Care Med. 2017, 43, 1464–1475. [Google Scholar] [CrossRef]
- European Union Council Regulation (EEC). No 2377/90, L 224. Off. J. Eur. Communities 2006, 33, 1–20. [Google Scholar]
- FAO, European Community Regulation (EC). No 1831/2003 of the European Parliament and of the Council of 22 September 2003 on Additives for Use in Animal Nutrition (Text with EEA Relevance). 2003. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32003R1831%20 (accessed on 9 November 2021).
- The European Commission Implementing Decision (EU) 2020/1161 of 4 August 2020 establishing a watch list of substances for Union-wide monitoring in the field of water policy pursuant to Directive 2008/105/EC of the European Parliament and of the Council. Off. J. Eur. Union 2020, L257/32, 32–35.
- WHO. WHO Report on Surveillance of Antibiotic Consumption 2016–2018 Early Implementation; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- World Organization for Animal Health. OIE Annual Report on Antimicrobial Agents Intended for Use in Animals; Annual Report; World Organization for Animal Health: Paris, France, 2021. [Google Scholar]
- Zhu, Y.-G.; Johnson, T.A.; Su, J.-Q.; Qiao, M.; Guo, G.-X.; Stedtfeld, R.D.; Hashsham, S.; Tiedje, J.M. Diverse and abundant antibiotic resistance genes in Chinese swine farms. Proc. Natl. Acad. Sci. USA 2013, 110, 3435–3440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.; Steele, J.C.; Meng, X.-Z. Usage, residue, and human health risk of antibiotics in Chinese aquaculture: A review. Environ. Pollut. 2017, 223, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Tang, Y.; Du, X.; Han, Y.; Shi, W.; Sun, S.; Zhang, W.; Zheng, H.; Liu, G. Fine polystyrene microplastics render immune responses more vulnerable to two veterinary antibiotics in a bivalve species. Mar. Pollut. Bull. 2021, 164, 111995. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Chen, W. Use of veterinary Medicines in Chinese Aquaculture: Current Status; FAO: Roma, Italy, 2012; ISBN 2070-7010. [Google Scholar]
- Bayen, S.; Estrada, E.S.; Juhel, G.; Kit, L.W.; Kelly, B.C. Pharmaceutically active compounds and endocrine disrupting chemicals in water, sediments and mollusks in mangrove ecosystems from Singapore. Mar. Pollut. Bull. 2016, 109, 716–722. [Google Scholar] [CrossRef]
- Baralla, E.; Pasciu, V.; Varoni, M.V.; Nieddu, M.; Demuro, R.; Demontis, M.P. Bisphenols’ occurrence in bivalves as sentinel of environmental contamination. Sci. Total Environ. 2021, 785, 147263. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Torres, R.; López, M.A.B.; Consentino, M.O.; Mochon, M.C.; Payán, M.R. Enzymatic-microwave assisted extraction and high-performance liquid chromatography–mass spectrometry for the determination of selected veterinary antibiotics in fish and mussel samples. J. Pharm. Biomed. Anal. 2011, 54, 1146–1156. [Google Scholar] [CrossRef]
- Alvarez-Muñoz, D.; Rodríguez-Mozaz, S.; Maulvault, A.L.; Tediosi, A.; Fernández-Tejedor, M.; Heuvel, F.V.D.; Kotterman, M.; Marques, A.; Barceló, D. Occurrence of pharmaceuticals and endocrine disrupting compounds in macroalgaes, bivalves, and fish from coastal areas in Europe. Environ. Res. 2015, 143, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Muñoz, D.; Huerta, B.; Fernández-Tejedor, M.; Rodríguez-Mozaz, S.; Barceló, D. Multi-residue method for the analysis of pharmaceuticals and some of their metabolites in bivalves. Talanta 2015, 136, 174–182. [Google Scholar] [CrossRef]
- Xie, H.; Hao, H.; Xu, N.; Liang, X.; Gao, D.; Xu, Y.; Gao, Y.; Tao, H.; Wong, M. Pharmaceuticals and personal care products in water, sediments, aquatic organisms, and fish feeds in the Pearl River Delta: Occurrence, distribution, potential sources, and health risk assessment. Sci. Total Environ. 2019, 659, 230–239. [Google Scholar] [CrossRef]
- Serra-Compte, A.; Pikkemaat, M.G.; Elferink, A.; Almeida, D.; Diogène, J.; Campillo, J.A.; Llorca, M.; Álvarez-Muñoz, D.; Barceló, D.; Rodríguez-Mozaz, S. Combining an effect-based methodology with chemical analysis for antibiotics determination in wastewater and receiving freshwater and marine environment. Environ. Pollut. 2021, 271, 116313. [Google Scholar] [CrossRef]
- Wu, C.-F.; Chen, C.-H.; Wu, C.-Y.; Lin, C.-S.; Su, Y.-C.; Wu, C.-F.; Tsai, H.-P.; Fan, P.-S.; Yeh, C.-H.; Yang, W.-C.; et al. Quinolone and Organophosphorus Insecticide Residues in Bivalves and Their Associated Risks in Taiwan. Molecules 2020, 25, 3636. [Google Scholar] [CrossRef] [PubMed]
- Klosterhaus, S.L.; Grace, R.; Hamilton, M.C.; Yee, D. Method validation and reconnaissance of pharmaceuticals, personal care products, and alkylphenols in surface waters, sediments, and mussels in an urban estuary. Environ. Int. 2013, 54, 92–99. [Google Scholar] [CrossRef]
- Dodder, N.G.; Maruya, K.A.; Ferguson, P.L.; Grace, R.; Klosterhaus, S.; La Guardia, M.; Lauenstein, G.G.; Ramirez, J. Occurrence of contaminants of emerging concern in mussels (Mytilus spp.) along the California coast and the influence of land use, storm water discharge, and treated wastewater effluent. Mar. Pollut. Bull. 2014, 81, 340–346. [Google Scholar] [CrossRef]
- Maruya, K.A.; Dodder, N.G.; Weisberg, S.B.; Gregorio, D.; Bishop, J.S.; Klosterhaus, S.; Alvarez, D.A.; Furlong, E.T.; Bricker, S.; Kimbrough, K.L.; et al. The Mussel Watch California pilot study on contaminants of emerging concern (CECs): Synthesis and next steps. Mar. Pollut. Bull. 2014, 81, 355–363. [Google Scholar] [CrossRef]
- Ramón, M.; Cano, J.; Peña, J.B.; Campos, M.J. Current Status and Perspectives of Mollusc (Bivalves and Gastropods) Culture in the Spanish Mediterranean. Bol. Inst. Esp. De Oceanogr. 2005, 21, 361. [Google Scholar]
- Zhou, W.; Han, Y.; Tang, Y.; Shi, W.; Du, X.; Sun, S.; Liu, G. Microplastics Aggravate the Bioaccumulation of Two Waterborne Veterinary Antibiotics in an Edible Bivalve Species: Potential Mechanisms and Implications for Human Health. Environ. Sci. Technol. 2020, 54, 8115–8122. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Zhou, W.; Tang, Y.; Shi, W.; Shao, Y.; Ren, P.; Zhang, J.; Xiao, G.; Sun, H.; Liu, G. Microplastics aggravate the bioaccumulation of three veterinary antibiotics in the thick shell mussel Mytilus coruscus and induce synergistic immunotoxic effects. Sci. Total Environ. 2021, 770, 145273. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, R.; Jiang, G. Investigation of organochlorine pesticides (OCPs) in mollusks collected from coastal sites along the Chinese Bohai Sea from 2002 to 2004. Environ. Pollut. 2007, 146, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.-N.; Shi, J.-B.; He, B.; Jiang, G.-B.; Yuan, C.-G. Investigation of Methylmercury and Total Mercury Contamination in Mollusk Samples Collected from Coastal Sites along the Chinese Bohai Sea. J. Agric. Food Chem. 2003, 51, 7373–7378. [Google Scholar] [CrossRef] [PubMed]
- Reichert, G.; Hilgert, S.; Fuchs, S.; Azevedo, J.C.R. Emerging contaminants and antibiotic resistance in the different environmental matrices of Latin America. Environ. Pollut. 2019, 255, 113140. [Google Scholar] [CrossRef]
- Das, N.; Madhavan, J.; Selvi, A.; Das, D. An overview of cephalosporin antibiotics as emerging contaminants: A serious environmental concern. 3 Biotech 2019, 9, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Farrington, J.W.; Tripp, B.W.; Tanabe, S.; Subramanian, A.; Sericano, J.L.; Wade, T.L.; Knap, A.H.; Edward, D. Goldberg’s proposal of “the Mussel Watch”: Reflections after 40 years. Mar. Pollut. Bull. 2016, 110, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Muñoz, D.; Rodríguez-Mozaz, S.; Jacobs, S.; Serra-Compte, A.; Cáceres, N.; Sioen, I.; Verbeke, W.; Barbosa, V.L.; Ferrari, F.; Fernández-Tejedor, M.; et al. Pharmaceuticals and endocrine disruptors in raw and cooked seafood from European market: Concentrations and human exposure levels. Environ. Int. 2018, 119, 570–581. [Google Scholar] [CrossRef]
- Alvarez-Muñoz, D.; Alegre, M.R.; Carrasco, N.; de Alda, M.L.; Barceló, D. Fast analysis of relevant contaminants mixture in commercial shellfish. Talanta 2019, 205, 119884. [Google Scholar] [CrossRef] [PubMed]
- The European Commission Regulation (EU) No 37/2010 of 22 December 2009 on pharmacologically active substances and their classification regarding maximum residue limits in foodstuffs of animal origin. Off. J. Eur. Union 2010, L15/1-72. Available online: https://ec.europa.eu/health/sites/default/files/files/eudralex/vol-5/reg_2010_37/reg_2010_37_en.pdf (accessed on 9 November 2021).
- FAO/WHO; Codex Alimentarius Commission. Maximum Residue Limits (MRLs) and Risk Management Recommendations (RMRs) for Residues of Veterinary Drugs in Foods- CX/MRL 2-, Codex Aliment; Chicago, IL, USA, 2018; Available online: https://www.fao.org/fao-who-codexalimentarius/sh-proxy/es/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FStandards%252FCXM%2B2%252FMRL2e.pdf (accessed on 9 November 2021).
- FAO. FAO Global Aquaculture Production 1950–2015; FAO: Sofia, Bulgaria, 2016. [Google Scholar]
- Notice No. 235, Ministry of Agriculture, P.R. of C. Maximum Residue Limits of Veterinary Drugs in Animal Foods. Minist. Agric. Beijing (in Chinese) 2002. Available online: www.msybeijing.com (accessed on 9 November 2021).
- Gatidou, G.; Vassalou, E.; Thomaidis, N. Bioconcentration of selected endocrine disrupting compounds in the Mediterranean mussel, Mytilus galloprovincialis. Mar. Pollut. Bull. 2010, 60, 2111–2116. [Google Scholar] [CrossRef]
- Liao, C.; Kannan, K. Species-specific accumulation and temporal trends of bisphenols and benzophenones in mollusks from the Chinese Bohai Sea during 2006–2015. Sci. Total Environ. 2019, 653, 168–175. [Google Scholar] [CrossRef]
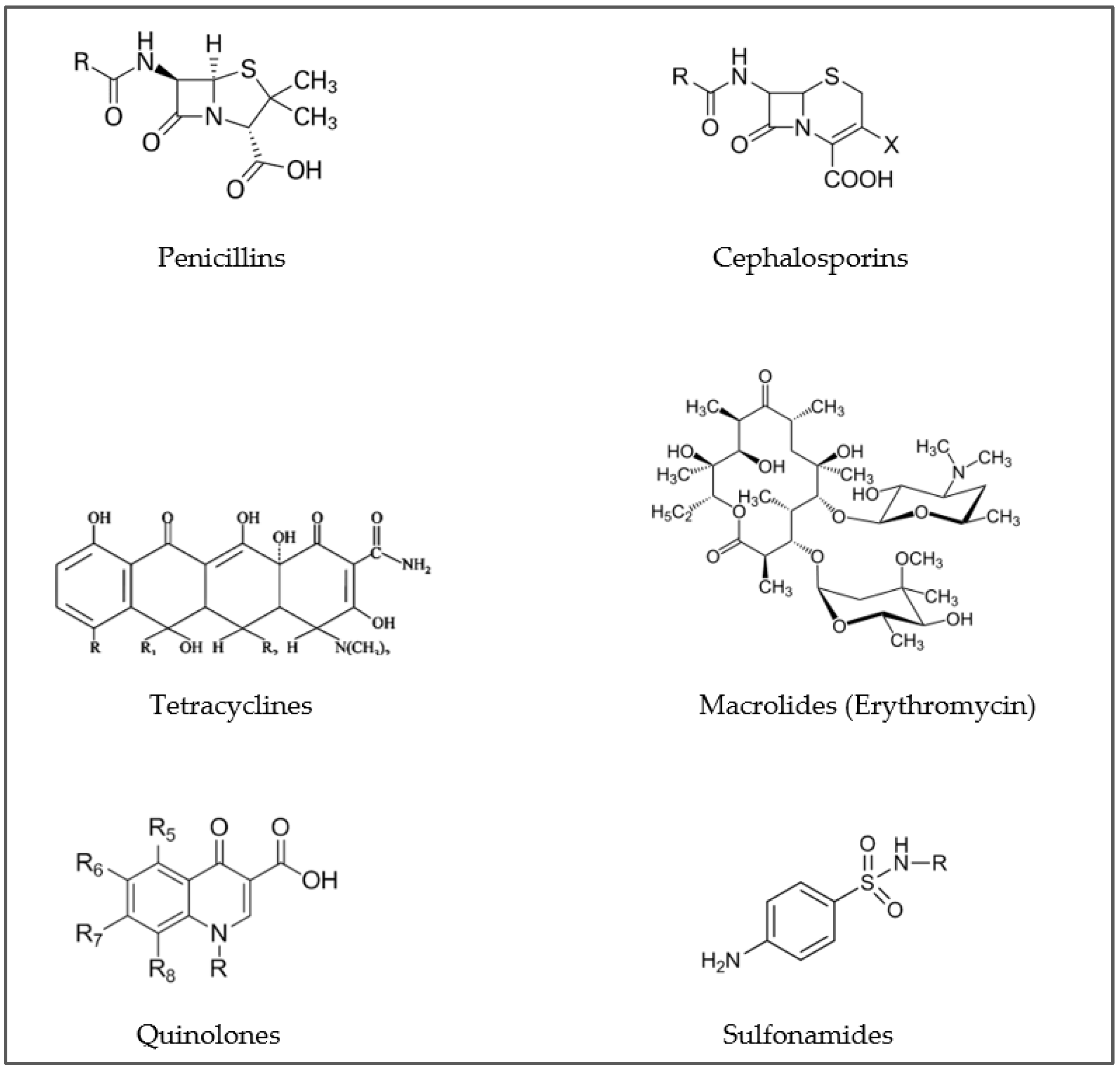

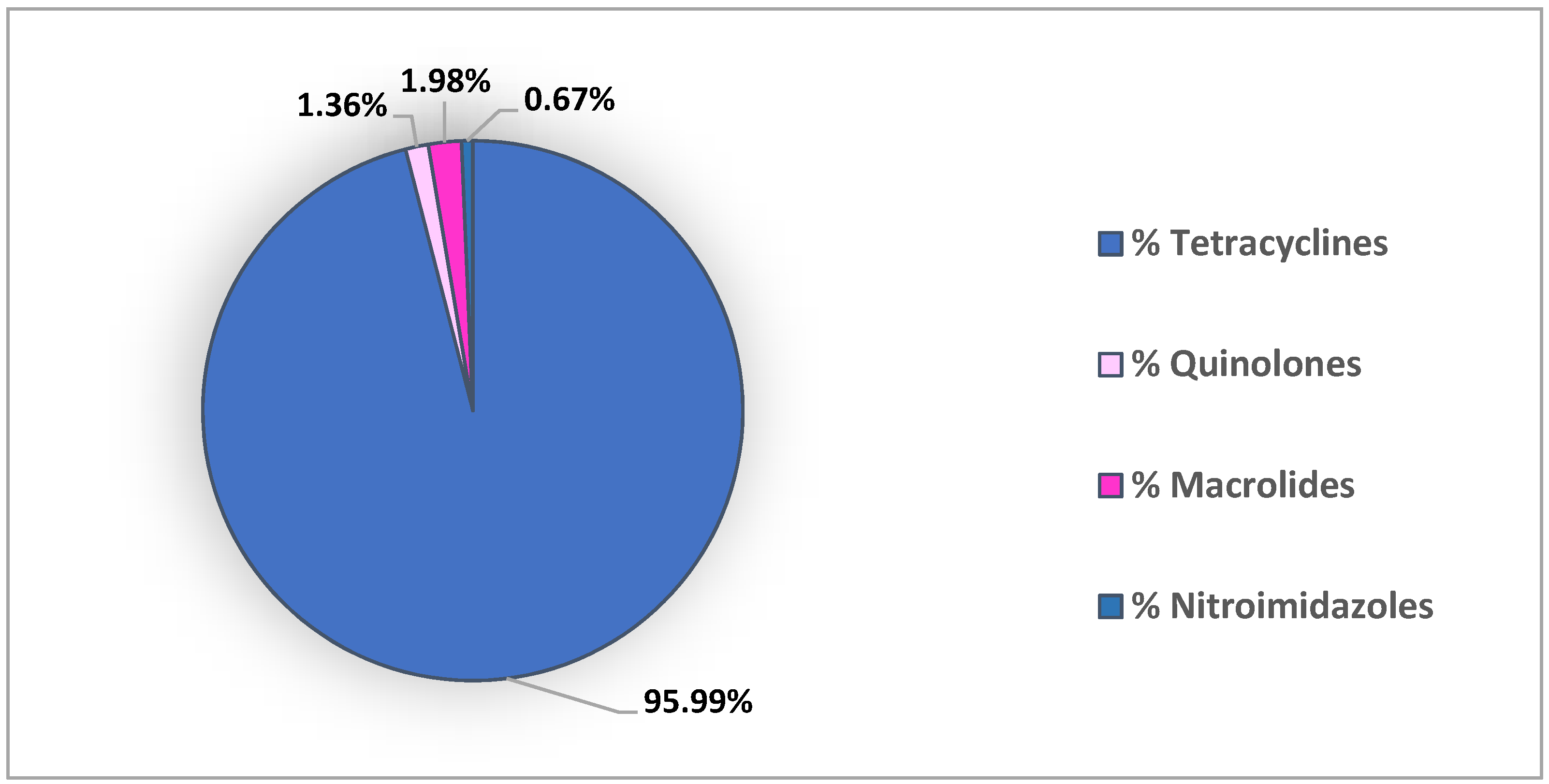
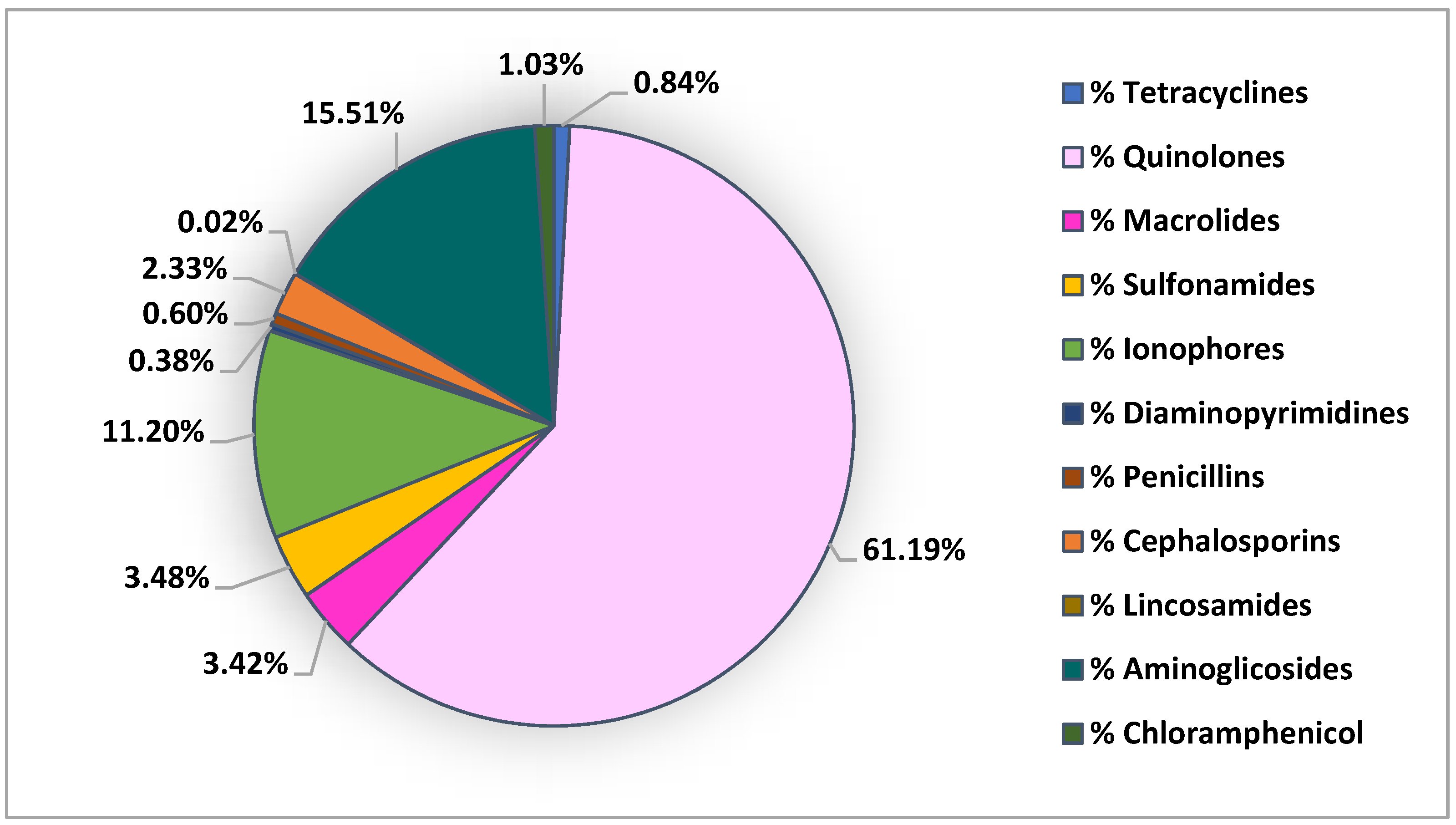
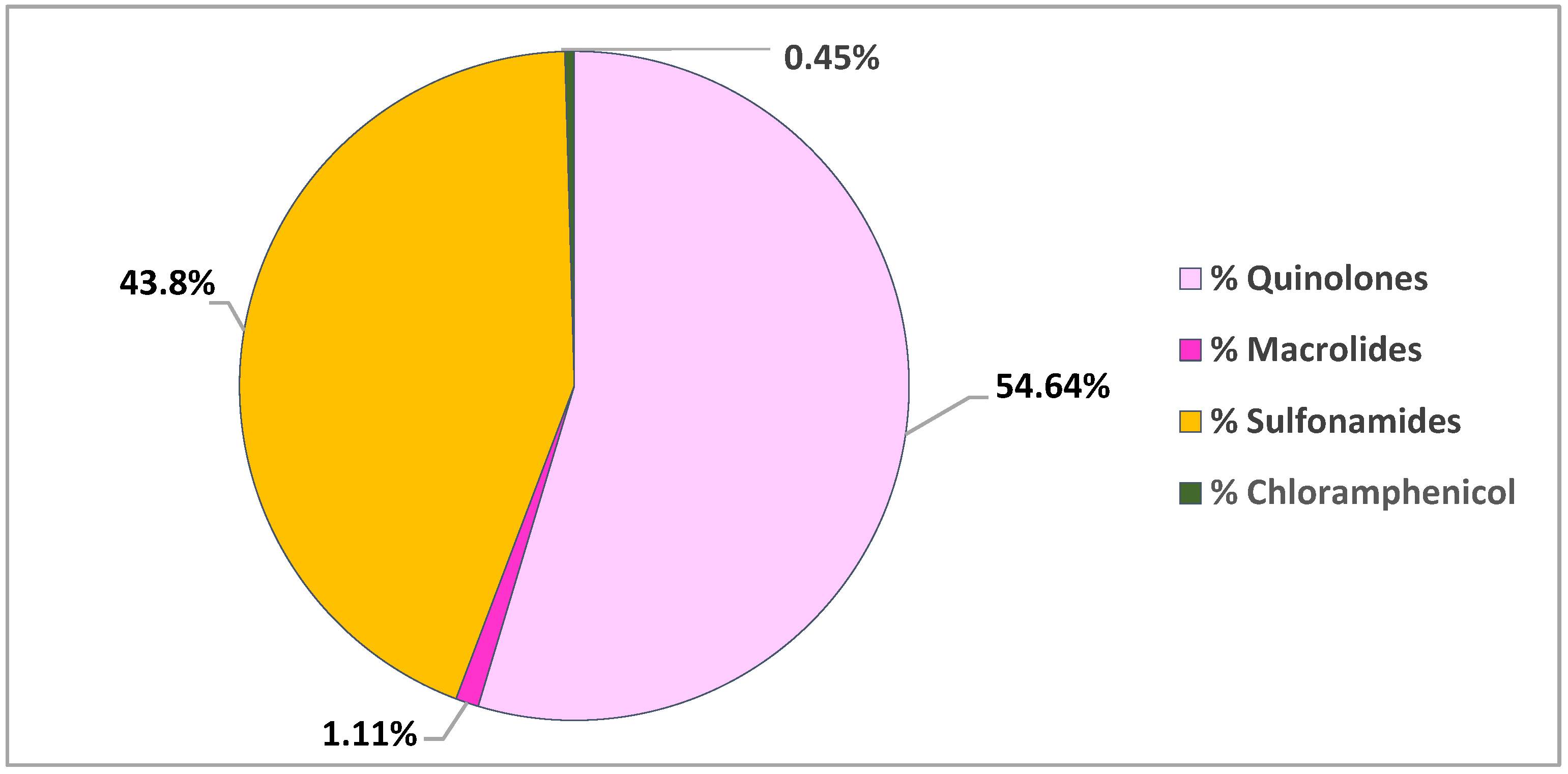
| Antibiotic Class | Extraction Method | Detection | Recovery % | LOD (ng/g dw) | LOQ (ng/g dw) | Reference |
|---|---|---|---|---|---|---|
| Quinolones Sulfonamides Macrolides | PLE+SPE | LC-MS/MS | n.d. | 0.1–0.3 0.02–0.05 0.04–0.6 | 0.3–0.9 0.06–0.6 0.1–1 | [4] |
| Tetracyclines Quinolones | LE+SPE | LC-MS/MS | 92–113 89–91 | 0.51–1.26 * 0.54 * | 0.65–1.48 * 0.83 * | [6] |
| Sulfonamides Quinolones Macrolides | SPE | UPLC-MS-MS | 67–85 70–93 63–80 | 0.02–0.03 * 0.06–0.16 * 0.01–0.25 * | 0.06–0.10 * 0.21–0.54 * 0.02–0.85 * | [27] |
| Macrolides Ionophores | LE-SPE | RRLC-MS/MS | 76.5–379.2 53.42–366.5 | 0.23–0.63 * | 0.76–2.11 * | [33] |
| Nitroimidazole | PLE+SPE | UPLC–QqLIT | n.d. | 0.2 | 0.7 | [7] |
| Sulfonamides Lincosamides | LE | LC-MS/MS | 61–131 51 | 0.01–0.03 0.01 | n.d. | [48] |
| Macrolides Nitroimidazoles Sulfonamides | PLE+SPE | UPLC-MS/MS | 45–54.4 65.8–66 34.7–41.3 | 0.01 0.01–0.10 0.01–0.03 | 0.02–0.03 0.03–0.33 0.02–0.09 | [51,52] |
| Sulfonamides Diaminopyrimidines Quinolones Tetracyclines Penicillins Cephalosporins Macrolides Chloramphenicol | LE+SPE | UHPLC-MS/MS | 43–127 | 0.01–0.05 * 0.01 * 0.01–1.9 * 0.03–0.3 * 0.01 * 0.09 * 0.03–0.3 * 0.2 * | 0.03–0.2 * 0.03 * 0.03–5.6 * 0.1–0.9 * 0.03 * 0.3 * 0.1–1 * 0.6 * | [53] |
| Continent | State-Country | Antibiotic | Location | Mollusk | Analytical Method Used | Antibiotic Concentration Found (ng/g dw) | Reference | |
|---|---|---|---|---|---|---|---|---|
| Mean | Range | |||||||
| Europe | Italy | Tetracycline | North Adriatic Sea | Meretrix lyrata | LC-MS/MS | 49.45 * | n.d. | [6] |
| Oxytetracycline | Venerupis | 125.03 * | n.d. | |||||
| Doxycycline | decussata | 60.45 * | n.d. | |||||
| Chlortetracycline | Venerupis philippinarum | 77.48 * | n.d | |||||
| Flumequine | 0.84 * | n.d. | ||||||
| Meretrix meretrix | ||||||||
| Paphia textile | ||||||||
| Venus gallina | ||||||||
| Flumequine | Mytilus | 3.59 * | n.d. | |||||
| galloprovincialis | ||||||||
| Mytilus edulis | ||||||||
| Mytilus chilensis | ||||||||
| Azithromycin | Po Delta | Mytilus galloprovincialis | UPLC-MS/MS | 13.3 | 13–13.6 | [51] | ||
| Spain | Tetracycline | Atlantic Spain | Mytilus galloprovincialis Mytilus edulis Mytilus chilensis | LC-MS/MS | 0.55 * | n.d. | [6] | |
| Ronidazole | Ebro Delta | Chamelea gallina | UPLC-MS/MS | 1 | n.d. | [51] | ||
| Crassostrea gigas | 1.8 | n.d. | ||||||
| Sulfamethoxazole | Chamelea gallina | <0.02 | n.d. | |||||
| Crassostrea gigas | <0.02 | n.d. | ||||||
| Azithromycin | Mytilus galloprovincialis | 2.9 | n.d. | |||||
| galloprovincialis | ||||||||
| Chamelea gallina | 1.3 | n.d. | ||||||
| Crassostrea gigas | 3 | n.d | ||||||
| Ronidazole | Ría de Muros y Noia | Ensis siliqua | UPLC–QqLIT | 2.26 | n.d. | [7] | ||
| Enrofloxacin | Alfacs Bay Mediterranean Sea | Mytilus galloprovincialis | UHPLC-QqLIT | 0.230 ▲ | 0.198–0.208 ▲ | [54] | ||
| Marbofloxacin | 0.435 ▲ | 0.424–0.446 ▲ | ||||||
| Portugal | Dimetridazole | Tagus estuary | Mytilus spp. | UPLC-MS/MS | 7.7 | 10.2–5.2 | [51] | |
| Azithromycin | 11.8 | 12–11.6 | ||||||
| Asia | China | Norfloxacin | Coastal cities along the Bohai Sea: Dalian, Yingkou, Huludao, Beidaihe, Tianjin, Shouguang, Penglai, Yantai and Weihai | Crassosteratalienwhanensis Chlamys farreri, Amussium, Scapharca subcrenata, Meretrix merehjgntrix Linnaeus, Mactra veneriformis, Mactra chinesis, Mya arenaria, Neverita didyma, Rapana venosa, Mytilus edulis | LC-MS/MS | 18.82 | 0–370 | [4] |
| Ciprofloxacin | 14.54 | 0–208 | ||||||
| Difloxacin | 3.14 | 0–57.1 | ||||||
| Enrofloxacin | 5.43 | 0–147 | ||||||
| Fleroxacin | 13.57 | 0–250 | ||||||
| Ofloxacin | 14.65 | 0–242 | ||||||
| Lomefloxacin | 5.67 | 0–141 | ||||||
| Sarafloxacin | 10.94 | 0–160 | ||||||
| Sulfathiazole | 1.33 | 0–35.2 | ||||||
| Sulfamethoxazole | 1.01 | 0–20.1 | ||||||
| Sulfisoxazole | 0.91 | 0–71.6 | ||||||
| Sulfadiazine | 0.16 | 0–2.72 | ||||||
| Sulfapyridine | 0.26 | 0–3.65 | ||||||
| Sulfadimethoxine | 0.08 | 0–1.75 | ||||||
| Sulfamethazine | 0.2 | 0–29.8 | ||||||
| Sulfamerazine | 0.42 | 0–5.98 | ||||||
| Sulfamonomethoxine | 1.60 | 0–15.4 | ||||||
| Spiramycin | 0.78 | 0–23.2 | ||||||
| Josamycin | 0.03 | 0–1.14 | ||||||
| Tylosin | 0.01 | 0–1.81 | ||||||
| Erythromycin | 1.65 | 0–31.3 | ||||||
| Roxithromycin | 0.11 | 0–1.92 | ||||||
| Erythromycin-H2O | Hailing Island of South China | Meretrix lusoria | RRLC-MS/MS | n.d. | n.d.–0.9 * | [33] | ||
| Salinomycin | 13 * | n.d.–14.5 * | ||||||
| Narasin | n.d | n.d.–7.5 * | ||||||
| Sulfadiazine | Dalang, Jingu, Dafeng and Nanliu River estuaries–Beibu Gulf | Crassostrea rivularis Gould | UPLC-ESI-MS/MS | 0.12 * | n.d. | [27] | ||
| Sulfapyridine | 0.45 * | n.d. | ||||||
| Sulfamethazine | 0.03 * | n.d | ||||||
| Sulfamethoxazole | 0.03 * | n.d. | ||||||
| Trimethoprim | 0.14 *. | n.d. | ||||||
| Norfloxacin | 0.37 * | n.d. | ||||||
| Ciprofloxacin | 0.54 *. | n.d. | ||||||
| Enrofloxacin | 0.17 * | n.d. | ||||||
| Ofloxacin | 0.14 * | n.d. | ||||||
| Enoxacin | 0.27 * | n.d. | ||||||
| Clarithromycin | 0.06 * | n.d. | ||||||
| Roxithromycin | 0.11 * | n.d. | ||||||
| Erythromycin | 1.08 * | n.d. | ||||||
| Sulfamerazine | Pearl River Delta | Ostrea gigas Mimachlamys nobilis Mytilus edulis | UHPLC-MS/MS | 0.03 * | n.d.–0.9 * | [53] # | ||
| Sulfamethazine | 0.07 * | n.d.–2.1 * | ||||||
| Sulfamethoxazole | 1.6 * | n.d.–26 * | ||||||
| Trimethoprim | 0.3 * | 0.04–1 * | ||||||
| Ciprofloxacin | 9.1 * | n.d.–42 * | ||||||
| Norfloxacin | 31 * | n.d.–256 * | ||||||
| Ofloxacin | 3 * | n.d.–72 * | ||||||
| Flumequine | 8.8 * | n.d.–118 * | ||||||
| Tetracycline | 0.5 * | 0.03–2.4 * | ||||||
| Oxytetracycline | 0.4 * | n.d.–1.8 * | ||||||
| Isochlortetracycline | 0.08 * | n.d.–0.6 * | ||||||
| PenicillinG-Na | 0.7 * | n.d.–3.4 * | ||||||
| Cefotaxime-Na | 2.7 * | 0.4–13 * | ||||||
| Spectinomycin | 18 * | n.d.–366 * | ||||||
| Roxithromycin | 0.1 * | n.d.–1.8 * | ||||||
| Erythromycin-H2O | 0.9 * | n.d.–5.5 * | ||||||
| Clarithromycin | 1.2 * | n.d.–23 * | ||||||
| Chloramphenicol | 1.3 * | n.d.–37 * | ||||||
| Enrofloxacin Flumequine | Taiwan: Changhua, Yunlin, Chiayi, Tainan, Kaohsiumg, Hwalien | Corbicula fluminea Meretrix lusoria | LC-MS/MS | 0.5 * 0.8 * | n.d. n.d. | [55] | ||
| Singapore | Sulfadiazine Lincomycin | Mangrove sites: Sungei Mandai Sungei Buloh Wetland Reserve Pandan Mangrove Sungei Changi Pulau Ubin Pasir Ris | Polymesoda expansa | LC-MS/MS | 0.51 * 0.02 * | 0.15–1.4 * 0.01–0.03 * | [48] | |
| America | California | Erythromycin-H2O Sulfamethizole | San Francisco Bay: | Geukensia demissa | LC-MS/MS | 0.1 * <0.25 * | <0.06–0.1 * <0.25–0.2 * | [56] |
| Richmond | ||||||||
| San Leandro Bay | ||||||||
| Eden Landing | ||||||||
| Foster City | ||||||||
| Cooley Landing | ||||||||
| Lomefloxacin | Coast of California | Mytilus spp. | LC-MS/MS | 29 | 0–170 | [57,58] | ||
| Sulfamethazine | 24 | 0–430 | ||||||
| Enrofloxacin | 1.3 | 0–12 | ||||||
| Erytromycin-H2O | 0.14 | 0–2 | ||||||
| Ofloxacin | 1.2 | 0–18 | ||||||
| Brazil | Chloramphenicol | Sepetiba Bay and Parnaiba Delta River | Anomalocardia | LC-MS/MS | <0.26 * | n.d.–0.5 * | [28] | |
| Brasiliana | ||||||||
| Mytilus edulis | ||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baralla, E.; Demontis, M.P.; Dessì, F.; Varoni, M.V. An Overview of Antibiotics as Emerging Contaminants: Occurrence in Bivalves as Biomonitoring Organisms. Animals 2021, 11, 3239. https://doi.org/10.3390/ani11113239
Baralla E, Demontis MP, Dessì F, Varoni MV. An Overview of Antibiotics as Emerging Contaminants: Occurrence in Bivalves as Biomonitoring Organisms. Animals. 2021; 11(11):3239. https://doi.org/10.3390/ani11113239
Chicago/Turabian StyleBaralla, Elena, Maria P. Demontis, Filomena Dessì, and Maria V. Varoni. 2021. "An Overview of Antibiotics as Emerging Contaminants: Occurrence in Bivalves as Biomonitoring Organisms" Animals 11, no. 11: 3239. https://doi.org/10.3390/ani11113239
APA StyleBaralla, E., Demontis, M. P., Dessì, F., & Varoni, M. V. (2021). An Overview of Antibiotics as Emerging Contaminants: Occurrence in Bivalves as Biomonitoring Organisms. Animals, 11(11), 3239. https://doi.org/10.3390/ani11113239






