Moving in the Dark—Evidence for an Influence of Artificial Light at Night on the Movement Behaviour of European Hedgehogs (Erinaceus europaeus)
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
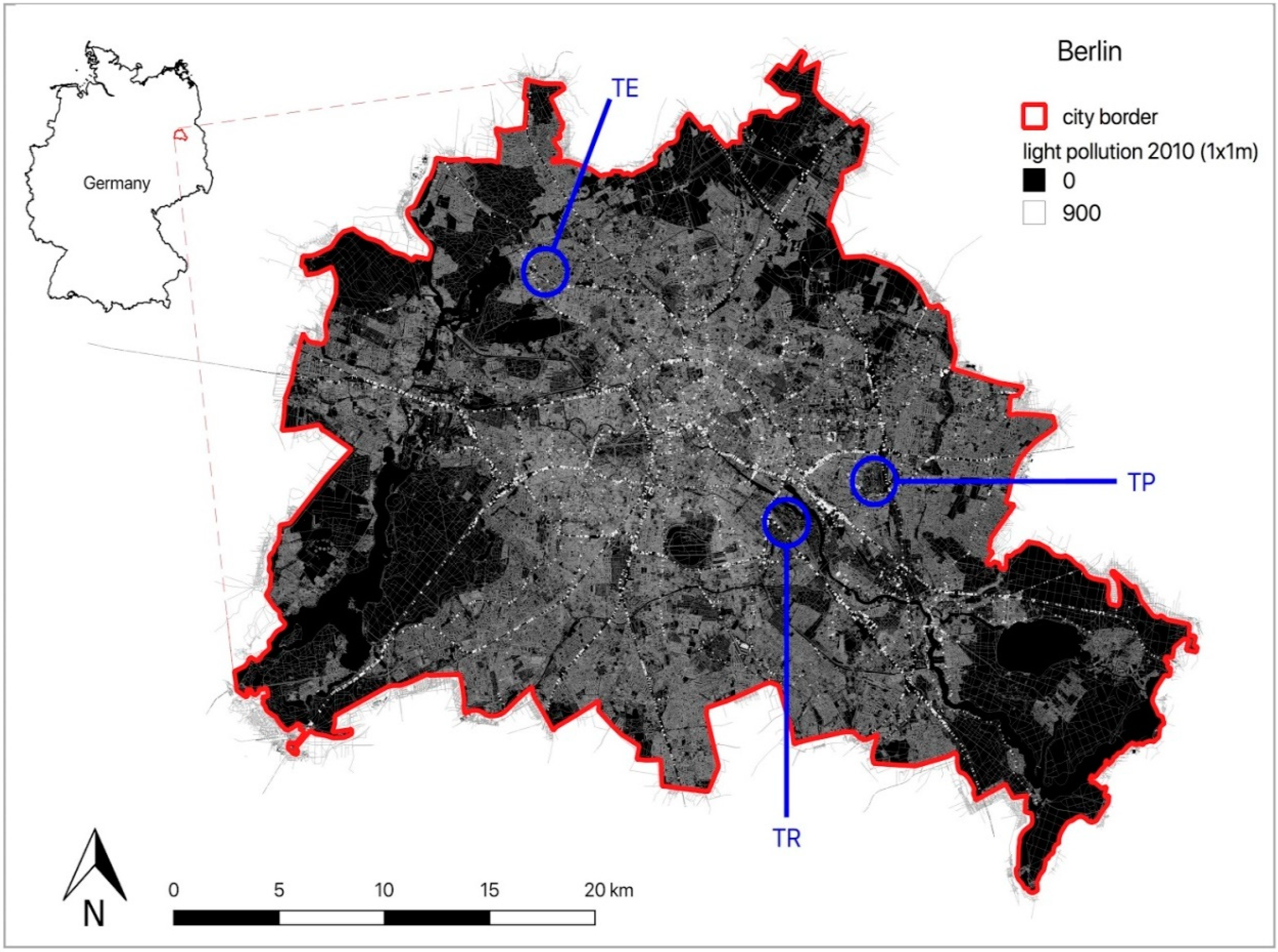
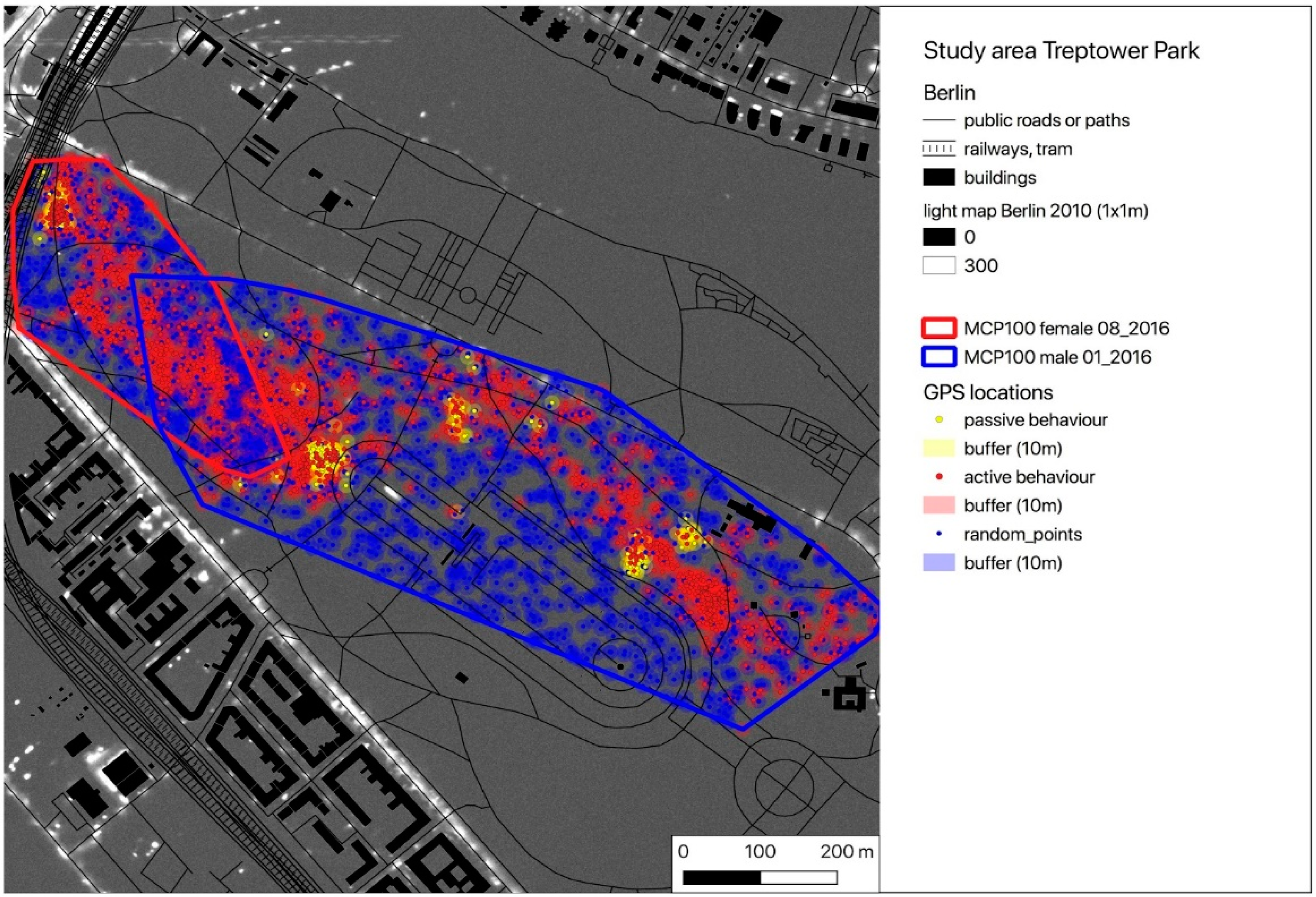
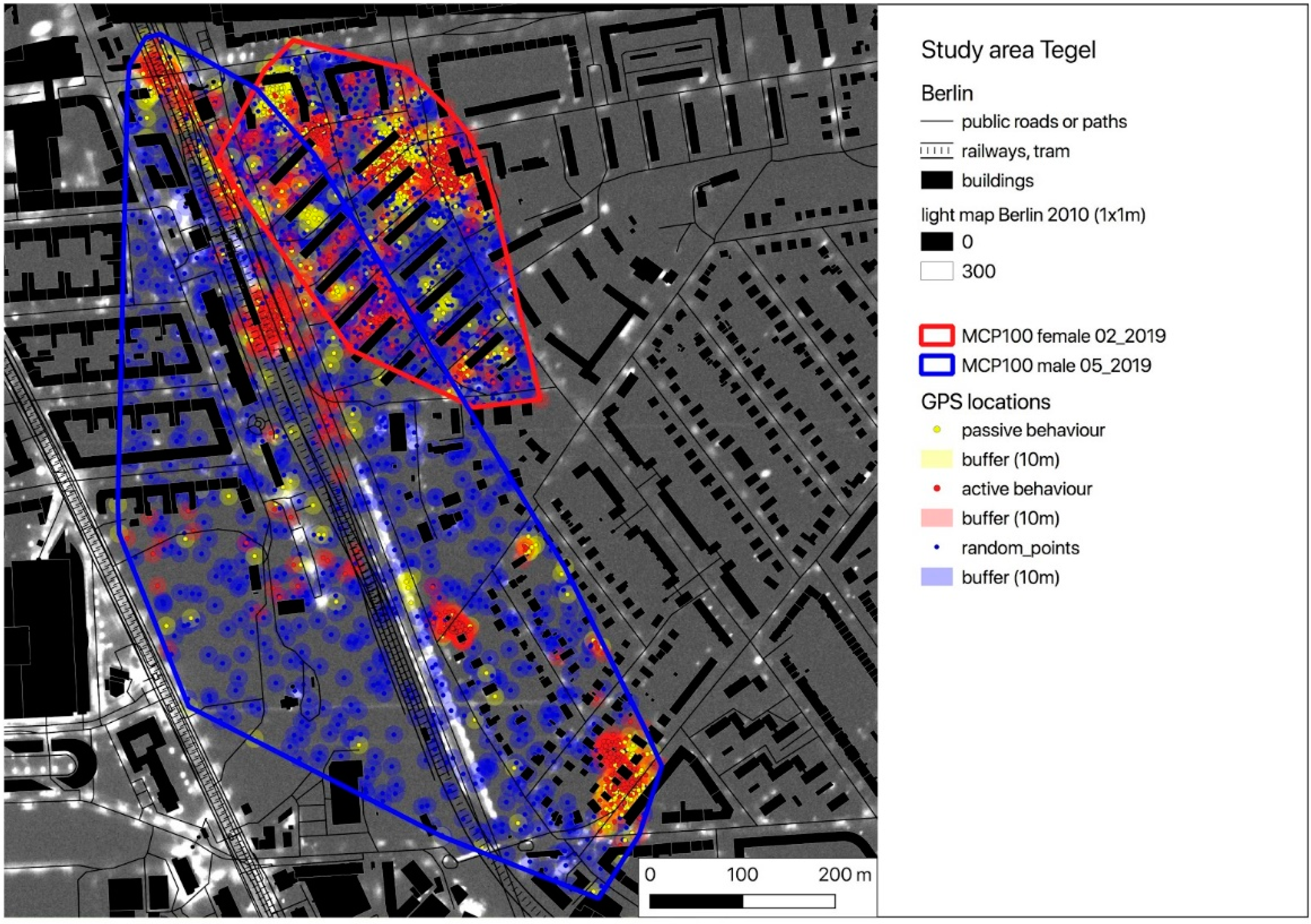
2.1. Study Area
2.2. Field Work
2.3. Data Logger Setup
2.4. Data Analysis and Statistics
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- van de Poel, J.L.; Dekker, J.; van Langevelde, F. Dutch hedgehogs Erinaceus europaeus are nowadays mainly found in urban areas, possibly due to the negative effects of badgers Meles meles. Wildl. Biol. 2015, 21, 51–55. [Google Scholar] [CrossRef]
- Pettett, C.E.; Moorhouse, T.P.; Johnson, P.J.; Macdonald, D.W. Factors affecting hedgehog (Erinaceus europaeus) attraction to rural villages in arable landscapes. Eur. J. Wildl. Res. 2017, 63, 54. [Google Scholar] [CrossRef]
- Hubert, P.; Julliard, R.; Biagianti, S.; Poulle, M.L. Ecological factors driving the higher hedgehog (Erinaceus europaeus) density in an urban area compared to the adjacent rural area. Landsc. Urban Plan. 2011, 103, 34–43. [Google Scholar] [CrossRef]
- Pettett, C.E.; Johnson, P.J.; Moorhouse, T.P.; Macdonald, D.W. National predictors of hedgehog Erinaceus europaeus distribution and decline in Britain. Mammal Rev. 2018, 48, 1–6. [Google Scholar] [CrossRef]
- Mathews, F.; Kubasiewicz, L.; Gurnell, J.; Harrower, C.; McDonald, R.; Shore, R. A Review of the Population and Conservation Status of British Mammals; A Report by the Mammal Society under Contract to Natural England, Natural Resources Wales and Scottish Natural Heritage; Natural England: Peterborough, UK, 2018. [Google Scholar]
- Müller, F. Langzeit-Monitoring der Straßenverkehrsopfer beim Igel (Erinaceus europaeus L.) zur Indikation von Populationsdichteveränderungen entlang zweier Teststrecken in Landkreis Fulda. Beiträge Nat. Osthess. 2018, 54, 21–26, ISSN 0342-5452-54. [Google Scholar]
- Reichholf, J.H. Starker Rückgang der Häufigkeit überfahrener Igel Erinaceus europaeus in Südostbayern und seine Ursachen. MittI. Zool. Ges. Braunau 2015, 11, 309–314. [Google Scholar]
- Krange, M. Change in the Occurrence of the West European Hedgehog (Erinaceus europaeus) in Western Sweden during 1950–2010; Karlstad University: Karlstad, Sweden, 2015. [Google Scholar]
- Holsbeek, L.; Rodts, J.; Muyldermans, S. Hedgehog and other animal traffic victims in Belgium: Results of a countrywide survey. Lutra 1999, 42, 111–119. [Google Scholar]
- Hof, A.R.; Bright, P.W. The value of green-spaces in built-up areas for western hedgehogs. Lutra 2009, 52, 69–82. [Google Scholar]
- Williams, B.M.; Baker, P.J.; Thomas, E.; Wilson, G.; Judge, J.; Yarnell, R.W. Reduced occupancy of hedgehogs (Erinaceus europaeus) in rural England and Wales: The influence of habitat and an asymmetric intra-guild predator. Sci. Rep. 2018, 8, 12156. [Google Scholar] [CrossRef]
- Hof, A.R.; Allen, A.M.; Bright, P.W. Investigating the role of the eurasian badger (Meles meles) in the nationwide distribution of the western european hedgehog (Erinaceus europaeus) in England. Animals 2019, 9, 759. [Google Scholar] [CrossRef]
- Wright, P.G.; Coomber, F.G.; Bellamy, C.C.; Perkins, S.E.; Mathews, F. Predicting hedgehog mortality risks on British roads using habitat suitability modelling. PeerJ 2020, 7, e8154. [Google Scholar] [CrossRef] [PubMed]
- Hof, A.R.; Bright, P.W. The value of agri-environment schemes for macro-invertebrate feeders: Hedgehogs on arable farms in Britain. Anim. Conserv. 2010, 13, 467–473. [Google Scholar] [CrossRef]
- Becher, S.A.; Griffiths, R. Genetic differentiation among local populations of the European hedgehog (Erinaceus europaeus) in mosaic habitats. Mol. Ecol. 1998, 7, 1599–1604. [Google Scholar] [CrossRef] [PubMed]
- Rondinini, C.; Doncaster, C. Roads as barriers to movement for hedgehogs. Funct. Ecol. 2002, 16, 504–509. [Google Scholar] [CrossRef]
- Braaker, S.; Kormann, U.; Bontadina, F.; Obrist, M.K. Prediction of genetic connectivity in urban ecosystems by combining detailed movement data, genetic data and multi-path modelling. Landsc. Urban Plan. 2017, 160, 107–114. [Google Scholar] [CrossRef]
- Rasmussen, S.L.; Yashiro, E.; Sverrisdóttir, E.; Nielsen, K.L.; Lukassen, M.B.; Nielsen, J.L.; Asp, T.; Pertoldi, C. Applying the GBS technique for the genomic characterization of a Danish population of European hedgehogs (Erinaceus europaeus). Gen. Biodiv. J. 2019, 3, 78–86. [Google Scholar]
- Hölker, F.; Wolter, C.; Perkin, E.K.; Tockner, K. Light pollution as a biodiversity threat. Trends Ecol. Evol. 2010, 25, 681–682. [Google Scholar] [CrossRef]
- Evans, W.R.; Akashi, Y.; Altman, N.S.; Manville, A.M. Response of night-migrating songbirds in cloud to colored and flashing light. N. Am. Birds 2007, 60, 476–488. [Google Scholar]
- Robert, K.A.; Lesku, J.A.; Partecke, J.; Chambers, B. Artificial light at night desynchronizes strictly seasonal reproduction in a wild mammal. Proc. R. Soc. B Biol. Sci. 2015, 282, 20151745. [Google Scholar] [CrossRef]
- Dominoni, D.; Quetting, M.; Partecke, J. Artificial light at night advances avian reproductive physiology. Proc. R. Soc. B 2013, 280, 20123017. [Google Scholar] [CrossRef]
- de Jong, M.; Ouyang, J.Q.; Da Silva, A.; van Grunsven, R.H.A.; Kempenaers, B.; Visser, M.E.; Spoelstra, K. Effects of nocturnal illumination on life-history decisions and fitness in two wild songbird species. Phil. Trans. R. Soc. B 2015, 370, 20140128. [Google Scholar] [CrossRef] [PubMed]
- Bennie, J.; Davies, T.W.; Cruse, D.; Inger, R.; Gaston, K.J. Cascading effects of artificial light at night: Resource-mediated control of herbivores in a grassland ecosystem. Phil. Trans. R. Soc. B 2015, 370, 20140131. [Google Scholar] [CrossRef] [PubMed]
- Titulaer, M.; Spoelstra, K.; Lange, C.Y.M.J.G.; Visser, M.E. Activity patterns during food provisioning are affected by artificial light in free living great tits (Parus major). PLoS ONE 2012, 7, e37377. [Google Scholar] [CrossRef] [PubMed]
- De Silva, A.; Valcu, M.; Kempenaers, B. Light pollution alters the phenology of dawn and dusk singing in common European songbirds. Phil. Trans. R. Soc. B 2015, 370, 20140126. [Google Scholar] [CrossRef]
- Stone, E.L.; Wakefield, A.; Harris, S.; Jones, G. The impacts of new street light technologies: Experimentally testing the effects on bats of changing from low-pressure sodium to white metal halide. Phil. Trans. R. Soc. B 2015, 370, 20140127. [Google Scholar] [CrossRef]
- Straka, T.M.; Wolf, M.; Gras, P.; Buchholz, S.; Voigt, C.C. Tree Cover Mediates the Effect of Artificial Light on Urban Bats. Front. Ecol. Evol. 2019, 7, 201900091. [Google Scholar] [CrossRef]
- Finch, D.; Smith, B.R.; Marshall, C.Â.; Coomber, F.G.; Kubasiewicz, L.M.; Anderson, M.; Wright, P.G.R.; Mathews, F. Effects of Artificial Light at Night (ALAN) on European Hedgehog Activity at Supplementary Feeding Stations. Animals 2020, 10, 768. [Google Scholar] [CrossRef]
- Reeve, N.J. A Field Study of the Hedgehog (Erinaceus europaeus) with Particular Reference to Movements and Behaviour. Ph.D. Thesis, University of London, London, UK, 1981. [Google Scholar]
- Morris, P. Hedgehog; William Collins: Sydney, Australia, 2018; ISBN 978-0-00-823573-4. [Google Scholar]
- Yalden, D. The food of the hedgehog in England. Acta Theriol. 1976, 21, 401–424. [Google Scholar] [CrossRef]
- Wroot, A.J. Feeding Ecology of the European Hedgehog Erinaceus europaeus; Royal Holloway; University of London: London, UK, 1984. [Google Scholar]
- Dickman, C. Age-related dietary change in the European hedgehog, Erinaceus europaeus. J. Zool. 1988, 215, 1–14. [Google Scholar] [CrossRef]
- Davies, T.W.; Bennie, J.; Gaston, K.J. Street lighting changes the composition of invertebrate communities. Biol. Lett. 2012, 8, 764–767. [Google Scholar] [CrossRef]
- Mori, E.; Menchetti, M.; Bertolino, S.; Mazza, G.; Ancillotto, L. Reappraisal of an old cheap method for marking the European hedgehog. Mam. Res. 2015, 60, 189–193. [Google Scholar] [CrossRef]
- Reeve, N.J.; Bowen, C.; Gurnell, J. An Improved Identification Marking Method for Hedgehogs; The Mammal Society: Southampton, UK, 2019. [Google Scholar]
- Barthel, L.M.F.; Hofer, H.; Berger, A. An easy, flexible solution to attach devices to hedgehogs (Erinaceus europaeus) enables long-term high-resolution studies. Ecol. Evol. 2019, 9, 672–679. [Google Scholar] [CrossRef] [PubMed]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- RStudio Team. RStudio: Integrated Development Environment for R; RStudio, Inc.: Boston, MA, USA, 2016. [Google Scholar]
- QGIS. Free Software Foundation, Inc. Available online: http://www.gnu.org (accessed on 22 August 2019).
- Calenge, C. The package “adehabitat” for the R software: A tool for the analysis of space and habitat use by animals. Ecol. Model. 2006, 197, 516–519. [Google Scholar] [CrossRef]
- Munger, J.C. Home ranges of horned lizards (Phrynosoma): Circumscribed and exclusive? Oecologia 1984, 62, 351–360. [Google Scholar] [CrossRef]
- Spencer, S.R.; Cameron, G.N.; Swihart, R.K. Operationally defining home range: Temporal dependence exhibited by hispid cotton rats. Ecology 1990, 71, 1817–1822. [Google Scholar] [CrossRef]
- Ruby, N.F.; Joshi, N.; Heller, H.C. Constant darkness restores entrainment to phase-delayed Siberian hamsters. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2002, 283, R1314–R1320. [Google Scholar] [CrossRef]
- Kuechly, H.U.; Kyba, C.C.M.; Ruhtz, T.; Lindemann, C.; Wolter, C.; Fischer, J.; Hölker, F. Aerial survey and spatial analysis of sources of light pollution in Berlin, Germany. Remote Sens. Environ. 2012, 126, 39–50. [Google Scholar] [CrossRef]
- Dewey, M.; Metap: Meta-Analysis of Significance Values. R package version 1.0. 2018. Available online: http://www.dewey.myzen.co.uk/meta/meta.html (accessed on 30 July 2020).
- Rast, W.; Barthel, L.M.F.; Berger, A. Music festival makes Hedgehogs move: How individuals cope behaviorally in response to human-induced stressors. Animals 2019, 9, 455. [Google Scholar] [CrossRef]
- Frank, K.D. Effects of artificial night lighting on moths. In Ecological Consequences of Artificial Night Lighting; Rich, C., Longcore, T., Eds.; Island Press: Washington, DC, USA, 2006; pp. 305–344. [Google Scholar]
- Molenaar, J.D.; Jonkers, D.A.; Henkens, R.J.H. Wegverlichting en Natuur; DWW Ontsnipperingsreeks: Wageningen, The Netherlands, 1997; pp. 1–98. [Google Scholar]
- Dowding, C.V.; Harris, S.; Poulton, S.; Baker, P.J. Nocturnal ranging behaviour of urban hedgehogs, Erinaceus europaeus, in relation to risk and reward. Anim. Behav. 2010, 80, 13–21. [Google Scholar] [CrossRef]
- Bird, B.L.; Branch, L.C.; Miller, D.L. Effects of coastal lighting on foraging behavior of beach mice. Conserv. Biol. 2004, 18, 1435–1439. [Google Scholar] [CrossRef]
- Molenaar, J.D.; Henkens, R.J.H.; ter Braak, C.; van Duyne, C.; Hoefsloot, G.; Jonkers, D.A. Effects of Road Lights on the Spatial Behaviour of Mammals; Alterra, Green World Research: Wageningen, The Netherlands, 2003; pp. 1–72. [Google Scholar]
- Reeve, N.J. Hedgehogs; T & AD Poyser: London, UK, 1994. [Google Scholar]
- Doncaster, C.P.; Krebs, J.R. The wider countryside-principles underlying the responses of mammals to heterogenous environments. Mamm. Rev. 1993, 23, 113–120. [Google Scholar] [CrossRef]
- WTimpact–Citizen Science as a Tool for Knowledge Transfer. Available online: http://www.izw-berlin.de/en/wtimpact-citizen-science-as-a-tool-for-knowledge-transfer.html (accessed on 22 July 2020).
- Hölker, F.; Moss, T.; Griefahn, B.; Kloas, W.; Voigt, C.C.; Henckel, D.; Hänel, A.; Kappeler, P.M.; Völker, S.; Schwope, A.; et al. The dark side of light: A transdisciplinary research agenda for light pollution policy. Ecol. Soc. 2010, 15. Available online: https://www.jstor.org/stable/26268230 (accessed on 30 July 2020). [CrossRef]
- Bontadina, F. Strassenüberquerungen von Igeln (Erinaceus europaeus). Ph.D. Thesis, University of Zurich, Zurich, Swiss, 1991. [Google Scholar]

| Animal_ID | Sex | Start Date [YYYY-MM-DD] | End Date [YYYY-MM-DD] | Number GPS_Total | Number GPS_Act | Number GPS_Pas | ACC_Thres | GPS Interval [Min] |
|---|---|---|---|---|---|---|---|---|
| TR_01_2016 | m | 2016-08-10 | 2016-09-19 | 1313 | 1140 | 173 | 1.87 | 10 |
| TR_02_2016 | f | 2016-08-10 | 2016-09-19 | 1141 | 936 | 205 | 2.01 | 10 |
| TR_08_2016 | f | 2016-08-10 | 2016-08-27 | 576 | 514 | 62 | 0.97 | 10 |
| TR_09_2016 | m | 2016-08-10 | 2016-08-24 | 528 | 459 | 69 | 0.60 | 10 |
| TR_13_2016 | f | 2016-08-10 | 2016-09-19 | 1357 | 1237 | 120 | 0.82 | 10 |
| TR_17_2016 | f | 2016-08-10 | 2016-09-18 | 1227 | 1098 | 129 | 0.74 | 10 |
| TR_19_2016 | m | 2016-08-10 | 2016-09-19 | 1275 | 1090 | 185 | 0.89 | 10 |
| TR_21_2016 | m | 2016-08-10 | 2016-09-19 | 2566 | 2450 | 116 | 0.84 | 10 |
| TP_13_2018 | m | 2018-05-22 | 2018-06-30 | 1127 | 1071 | 56 | 0.86 | 5 |
| TP_18_2018 | m | 2018-05-22 | 2018-06-30 | 1110 | 994 | 116 | 0.99 | 5 |
| TP_20_2017 | f | 2017-08-14 | 2017-09-04 | 489 | 449 | 40 | 0.84 | 10 |
| TP_21_2018 | m | 2018-05-22 | 2018-06-30 | 1226 | 1151 | 75 | 0.84 | 5 |
| TP_26_2017 | f | 2017-08-16 | 2017-09-12 | 487 | 400 | 87 | 0.88 | 10 |
| TP_28_2017 | f | 2017-08-14 | 2017-09-04 | 684 | 606 | 78 | 0.92 | 10 |
| TP_29_2018 | m | 2018-05-28 | 2018-06-23 | 941 | 828 | 113 | 0.94 | 5 |
| TP_30_2018 | m | 2018-05-23 | 2018-06-09 | 991 | 838 | 153 | 0.94 | 5 |
| TE_02_2019 | f | 2019-07-20 | 2019-09-30 | 973 | 446 | 527 | 1.15 | 5 |
| TE_03_2019 | m | 2019-07-10 | 2019-08-01 | 870 | 633 | 237 | 1.00 | 5 |
| TE_04_2019 | m | 2019-08-30 | 2019-10-08 | 1769 | 1100 | 669 | 0.86 | 5 |
| TE_05_2019 | m | 2019-07-24 | 2019-09-16 | 1318 | 745 | 573 | 1.03 | 5 |
| TE_07_2019 | m | 2019-07-24 | 2019-09-27 | 1906 | 1320 | 586 | 1.06 | 5 |
| TE_10_2019 | f | 2019-08-24 | 2019-10-08 | 2469 | 1595 | 874 | 0.69 | 5 |
| Animal_ID | Animal_Mean | Animal_SD | Animal_Min | Animal_Max | Random_Mean | Random_SD | Random_Min | Random_Max | p-Values | Significance Level |
|---|---|---|---|---|---|---|---|---|---|---|
| TR_01_2016 | 102.0 | 4.2 | 99.3 | 140.9 | 118.9 | 25.1 | 101.4 | 226.8 | 0.001 | *** |
| TR_02_2016 | 102.8 | 6.2 | 99.6 | 177.0 | 125.2 | 150.5 | 98.6 | 10,339.3 | 0.001 | *** |
| TR_08_2016 | 106.5 | 20.2 | 100.6 | 290.2 | 125.6 | 159.2 | 99.0 | 8220.8 | 0.03 | * |
| TR_09_2016 | 102.3 | 5.4 | 100.2 | 205.6 | 126.4 | 165.2 | 99.0 | 9845.4 | 0.001 | *** |
| TR_13_2016 | 115.2 | 39.6 | 100.2 | 578.1 | 125.8 | 160.3 | 98.7 | 11,076.4 | 0.993 | n.s. |
| TR_17_2016 | 103.4 | 5.1 | 100.1 | 179.0 | 125.8 | 155.6 | 98.7 | 10,103.6 | 0.001 | *** |
| TR_19_2016 | 102.0 | 2.4 | 100.0 | 123.6 | 125.9 | 170.3 | 98.8 | 11,093.1 | 0.001 | *** |
| TR_21_2016 | 102.9 | 7.4 | 99.8 | 212.8 | 125.2 | 165.3 | 98.6 | 11,295.8 | 0.001 | *** |
| mean_TR | 104.6 | 11.3 | 100.0 | 238.4 | 124.8 | 143.9 | 99.1 | 9025.1 | - | - |
| TP_13_2018 | 105.2 | 26.9 | 100.5 | 544.2 | 120.6 | 83.9 | 43.6 | 3386.4 | 0.74 | n.s. |
| TP_18_2018 | 102.7 | 1.1 | 100.7 | 116.3 | 120.5 | 81.8 | 47.8 | 3404.3 | 0.001 | *** |
| TP_20_2017 | 102.7 | 0.8 | 101.1 | 106.2 | 119.8 | 70.8 | 50.0 | 3269.8 | 0.001 | *** |
| TP_21_2018 | 103.9 | 9.9 | 100.6 | 235.7 | 120.2 | 80.4 | 44.8 | 3371.5 | 0.004 | ** |
| TP_26_2017 | 103.3 | 6.1 | 100.7 | 166.2 | 118.3 | 57.0 | 49.2 | 2835.1 | 0.001 | *** |
| TP_28_2017 | 103.0 | 1.2 | 100.1 | 106.6 | 120.5 | 86.4 | 43.9 | 3360.7 | 0.001 | *** |
| TP_29_2018 | 116.8 | 25.0 | 102.1 | 366.0 | 120.7 | 84.4 | 47.0 | 3317.2 | 0.987 | n.s. |
| TP_30_2018 | 105.0 | 7.6 | 101.3 | 197.5 | 120.3 | 78.7 | 45.9 | 3337.0 | 0.005 | ** |
| mean_TP | 105.3 | 9.8 | 100.9 | 229.8 | 120.1 | 77.9 | 46.5 | 3285.2 | - | - |
| TE_02_2019 | 118.9 | 25.1 | 101.4 | 226.8 | 122.3 | 83.7 | 10.4 | 3348.0 | 0.001 | *** |
| TE_03_2019 | 159.8 | 351.7 | 101.5 | 3274.0 | 122.3 | 81.1 | 7.3 | 3332.2 | 1.0 | n.s. |
| TE_04_2019 | 120.8 | 47.1 | 100.8 | 445.0 | 122.2 | 84.8 | 6.0 | 3346.3 | 0.02 | * |
| TE_05_2019 | 160.9 | 86.3 | 101.9 | 443.3 | 122.2 | 82.6 | 5.1 | 3294.6 | 1.0 | n.s. |
| TE_07_2019 | 127.0 | 44.4 | 101.2 | 623.6 | 122.3 | 83.2 | 6.1 | 3402.3 | 0.02 | * |
| TE_10_2019 | 112.7 | 25.3 | 101.4 | 399.7 | 122.1 | 81.5 | 5.4 | 3396.6 | 0.001 | *** |
| mean_TE | 133.4 | 96.6 | 101.4 | 902.1 | 122.2 | 82.8 | 6.7 | 3353.3 | - | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berger, A.; Lozano, B.; Barthel, L.M.F.; Schubert, N. Moving in the Dark—Evidence for an Influence of Artificial Light at Night on the Movement Behaviour of European Hedgehogs (Erinaceus europaeus). Animals 2020, 10, 1306. https://doi.org/10.3390/ani10081306
Berger A, Lozano B, Barthel LMF, Schubert N. Moving in the Dark—Evidence for an Influence of Artificial Light at Night on the Movement Behaviour of European Hedgehogs (Erinaceus europaeus). Animals. 2020; 10(8):1306. https://doi.org/10.3390/ani10081306
Chicago/Turabian StyleBerger, Anne, Briseida Lozano, Leon M. F. Barthel, and Nadine Schubert. 2020. "Moving in the Dark—Evidence for an Influence of Artificial Light at Night on the Movement Behaviour of European Hedgehogs (Erinaceus europaeus)" Animals 10, no. 8: 1306. https://doi.org/10.3390/ani10081306
APA StyleBerger, A., Lozano, B., Barthel, L. M. F., & Schubert, N. (2020). Moving in the Dark—Evidence for an Influence of Artificial Light at Night on the Movement Behaviour of European Hedgehogs (Erinaceus europaeus). Animals, 10(8), 1306. https://doi.org/10.3390/ani10081306







