Effects of Supplementing Grape Pomace to Broilers Fed Polyunsaturated Fatty Acids Enriched Diets on Meat Quality
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Broilers and Experimental Design
2.2. Diet Formulation
2.3. Broiler Slaughter
2.4. Sampling
2.5. Fatty Acids Evaluation
2.6. Resveratrol Content Determination
2.7. Total Polyphenols Content
2.8. Total Antioxidant Capacity
2.9. Instrumental Color Measurements
2.10. pH Assessment
2.11. Texture Profile Analysis
2.12. Evaluation of Lipid Oxidation Parameters
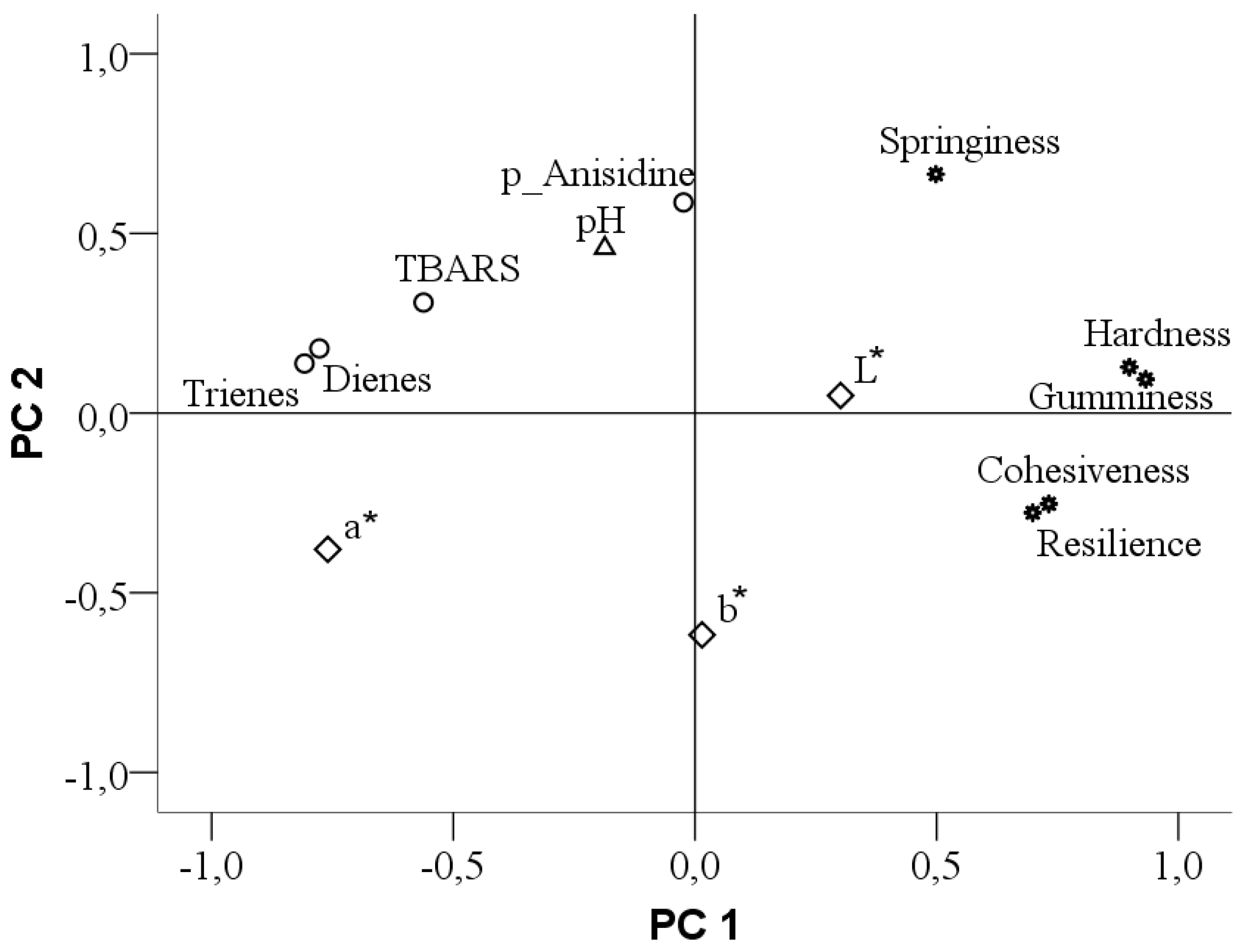
2.13. Principal Component Analysis
2.14. Statistical Analysis
3. Results
3.1. Chemical Composition
3.2. Effect of Grape Pomace on Production Parameters of Broiler
3.3. Meat Quality Parameters of Thigh and Breast Meat
3.3.1. Effect of Using Grape Pomace in Broiler Diets on Meat Color and pH
3.3.2. Effect of Using Grape Pomace in Broiler Diets on Meat Texture Parameters Determined by Double Cycle Compression
3.3.3. Effect of Using Grape Pomace in Broiler Diets on Lipid Oxidation
3.3.4. The Relationship between Meat Quality Parameters
4. Discussion
4.1. Production Parameters
4.2. Chemical Composition
4.3. Meat Colour
4.4. Meat pH
4.5. Texture Parameters
4.6. Oxidative Stability of the Meat
4.7. The Relationship between Meat Quality Parameters
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grunert, K.G. Food quality and safety: Consumer perception and demand. Eur. Rev. Agric. Econ. 2005, 32, 369–391. [Google Scholar] [CrossRef]
- Bourre, J. Dietary omega-3 Fatty acids and psychiatry: Mood, behaviour, stress, depression, dementia and aging. J. Nutr. Health Aging 2005, 9, 31–38. [Google Scholar] [PubMed]
- Lee, N.; Kwak, H.S.; Joo, J.Y.; Kang, J.; Lee, Y. Effects of partial replacement of pork meat with chicken or duck meat on the texture, flavor, and consumer acceptance of sausage. J. Food Qual. 2018, 2018, 1–9. [Google Scholar] [CrossRef]
- Pelicano, E.; De Souza, P.; De Souza, H.; Oba, A.; Norkus, E.; Kodawara, L.; De Lima, T. Effect of different probiotics on broiler carcass and meat quality. Revista Brasileira de Ciência Avícola 2003, 5, 207–214. [Google Scholar] [CrossRef]
- Grashorn, M.A. Functionality of poultry meat. J. Appl. Poult. Res. 2007, 16, 99–106. [Google Scholar] [CrossRef]
- Gaál, T.; Wagner, L.; Husveth, F.; Manilla, H.A.; Vajdovich, P.; Balogh, N.; Lóth, I.; Németh, K. Effects of saturated and unsaturated fats with vitamin e supplementation on the antioxidant status of broiler chicken tissues. Acta Vet. Hung. 2000, 48, 69–79. [Google Scholar] [CrossRef]
- Bayraktar, H.; Altan, O.; Açıkgöz, Z.; Baysal, Ş.H.; Şeremet, Ç. Effects of oxidised oil and vitamin E on performance and some blood traits of heat-stressed male broilers. S. Afr. J. Anim. Sci. 2011, 41, 288–296. [Google Scholar] [CrossRef]
- Salami, S.A.; Majoka, M.A.; Saha, S.; Garber, A.; Gabarrou, J.-F. Efficacy of dietary antioxidants on broiler oxidative stress, performance and meat quality: Science and market. Avian Boil. Res. 2015, 8, 65–78. [Google Scholar] [CrossRef]
- Brenes, A.; Viveros, A.; Chamorro, S.; Arija, I. Use of polyphenol-rich grape by-products in monogastric nutrition. A review. Anim. Feed. Sci. Technol. 2016, 211, 1–17. [Google Scholar] [CrossRef]
- Zhang, W.; Xiao, S.; Lee, J.E.; Ahn, D.U. Consumption of oxidised oil increases oxidative stress in broilers and affects the quality of breast meat. J. Agric. Food Chem. 2011, 59, 969–974. [Google Scholar] [CrossRef]
- Delles, R.M.; Xiong, Y.L.; Ao, T.; Dawson, K. Antioxidant Supplementation Minimizes the Negative Impact of Dietary Oxidised Oil on Chicken Broiler Breast and Thigh Meat Quality. In Proceedings of the 59th International Congress of Meat Science and Technology, Izmir, Turkey, 18–23 August 2013. [Google Scholar]
- Ryzner, M.; Takáčová, J.; Cobanova, K.; Venglovská, K.; Placha, I.; Faix, Š. Effect of dietary Salvia officinalis essential oil and sodium selenite supplementation on antioxidative status and blood phagocytic activity in broiler chickens. Acta Vet. Brno 2013, 82, 43–48. [Google Scholar] [CrossRef][Green Version]
- Iuga, M.; Ropciuc, S.; Mironeasa, S. Antioxidant activity and total phenolic content of grape seeds and peels from romanian varieties. Food Environ. Safety J. 2017, 16, 276–282. [Google Scholar]
- Costabile, G.; Vitale, M.; Luongo, D.; Naviglio, D.; Vetrani, C.; Ciciola, P.; Tura, A.; Castello, F.; Mena, P.; Del Rio, D.; et al. Grape pomace polyphenols improve insulin response to a standard meal in healthy individuals: A pilot study. Clin. Nutr. 2019, 38, 2727–2734. [Google Scholar] [CrossRef] [PubMed]
- Sebranek, J.; Bacus, J. Natural and Organic Cured Meat Products: Regulatory Manufacturing, Marketing, Quality and Safety Issues. AMSA. White Paper Series 1. 2007. Available online: http://www.meatscience.org/pubs/White%20Papers/wp_0012007_Natural_Organic_Cured_Meat.pdf (accessed on 24 March 2009).
- Khodayari, F.; Shahria, H.A. The effect of red grape pomace on performance, lipid peroxidation (MDA) and some serum biochemical parameters in broiler. Adv. Biores 2014, 5, 82–87. [Google Scholar] [CrossRef]
- Olteanu, M.; Criste, R.D.; Panaite, T.D.; Ropota, M.; Vlaicu, P.A.; Turcu, R.P. Bioproductive parameters and fatty acids profile of the meat from broilers treated with flax meal and grape seeds meal. Sci. Pap. Anim. Sci. Biotechno. 2017, 50, 15–21. [Google Scholar]
- Aditya, S.; Ohh, S.J.; Ahammed, M.; Lohakare, J. Supplementation of grape pomace (Vitis vinifera) in broiler diets and its effect on growth performance, apparent total tract digestibility of nutrients, blood profile, and meat quality. Anim. Nutr. 2018, 4, 210–214. [Google Scholar] [CrossRef]
- Aguirre, M.E.; Owens, C.M.; Miller, R.; Alvarado, C. Descriptive sensory and instrumental texture profile analysis of woody breast in marinated chicken. Poult. Sci. 2018, 97, 1456–1461. [Google Scholar] [CrossRef]
- Cavitt, L.C.; Meullenet, J.F.; Gandhapuneni, R.K.; Youm, G.W.; Owens, C.M. Rigor development and meat quality of large and small broilers and the use of Allo-Kramer shear, needle puncture, and razor blade shear to measure texture. Poult. Sci. 2005, 84, 113–118. [Google Scholar] [CrossRef]
- Fletcher, D.L. Poultry meat quality. World’s Poult. Sci. J. 2002, 58, 131–145. [Google Scholar] [CrossRef]
- Petracci, M.; Betti, M.; Bianchi, M.; Cavani, C. Color variation and characterization of broiler breast meat during processing in Italy. Poult. Sci. 2004, 83, 2086–2092. [Google Scholar] [CrossRef]
- Qiao, M.; Fletcher, D.L.; Smith, D.P.; Northcutt, J.K. The Effect of Broiler Breast Meat Color on pH, Moisture, Water-Holding Capacity, and Emulsification Capacity. Poult. Sci. 2001, 80, 676–680. [Google Scholar] [CrossRef] [PubMed]
- Panaite, T.D.; Criste, R.D.; Ropota, M.; Criste, V.; Vasile, G.; Olteanu, M.; Vlaicu, P.A.; Socoliuc, R.P. Determination of the feeding value of food industry by-products. Sci. Pap. Anim. Sci. Ser. Lucrări Ştiinţifice—Seria Zootehnie 2016, 66, 106–111. [Google Scholar]
- Careri, M.; Corradini, C.; Elviri, L.; Nicoletti, I.; Zagnoni, I. Direct HPLC Analysis of Quercetin andtrans-Resveratrol in Red Wine, Grape, and Winemaking Byproducts. J. Agric. Food Chem. 2003, 51, 5226–5231. [Google Scholar] [CrossRef] [PubMed]
- Untea, A.E.; Varzaru, I.; Panaite, T.D.; Gavris, T.; Lupu, A.; Ropotă, M. The Effects of Dietary Inclusion of Bilberry and Walnut Leaves in Laying Hens’ Diets on the Antioxidant Properties of Eggs. Animal 2020, 10, 191. [Google Scholar] [CrossRef]
- Panaite, T.D.; Mironeasa, M.I.S.; Vlaicu, P.A.; Iuga, M. Liquid egg products characterization during storage as a response of novel phyto-additives added in hens diet. Emir. J. Food Agric. 2019, 31, 304–314. [Google Scholar] [CrossRef]
- Jeacocke, R.E. Continuous measurements of the pH of beef muscle in intact beef carcases. Int. J. Food Sci. Technol. 2007, 12, 375–386. [Google Scholar] [CrossRef]
- Korkeala, H.; Mäki-Petäys, O.; Alanko, T.; Sorvettula, O. Determination of pH in meat. Meat Sci. 1986, 18, 121–132. [Google Scholar] [CrossRef]
- Ebrahimzadeh, S.; Navidshad, B.; Farhoomand, P.; Aghjehgheshlagh, F.M. Effects of grape pomace and vitamin E on performance, antioxidant status, immune response, gut morphology and histopathological responses in broiler chickens. S. Afr. J. Anim. Sci. 2018, 48, 324. [Google Scholar] [CrossRef]
- Kumanda, C.; Mlambo, V.; Mnisi, C. From Landfills to the Dinner Table: Red Grape Pomace Waste as a Nutraceutical for Broiler Chickens. Sustainability 2019, 11, 1931. [Google Scholar] [CrossRef]
- Oomah, B. Flaxseed as a functional food source. J. Sci. Food Agric. 2001, 81, 889–894. [Google Scholar] [CrossRef]
- Ganorkar, P.M.; Jain, R.K. Flaxseed—A nutritional punch. Int. Food Res. J. 2013, 20, 519–525. [Google Scholar]
- Vlaicu, P.A.; Panaite, T.D.; Dragotoiu, D.; Ropota, M.; Bobe, E.; Olteanu, M.; Criste, R.D. Feeding quality of the meat from broilers fed with dietary food industry by-products (flaxseed, rapeseeds and buckthorn meal, grape pomace). Sci. Pap. Ser. D Anim. Sci. 2017, 60, 123–130. [Google Scholar]
- López, M.; Martínez, F.; Del Valle, C.; Orte, C.; Miró, M. Analysis of phenolic constituents of biological interest in red wines by high-performance liquid chromatography. J. Chromatogr. A 2001, 922, 359–363. [Google Scholar] [CrossRef]
- Leonard, S.S.; Xia, C.; Jiang, B.-H.; Stinefelt, B.; Klandorf, H.; Harris, G.K.; Shi, X. Resveratrol scavenges reactive oxygen species and effects radical-induced cellular responses. Biochem. Biophys. Res. Commun. 2003, 309, 1017–1026. [Google Scholar] [CrossRef] [PubMed]
- Vitrac, X.; Desmoulière, A.; Brouillaud, B.; Krisa, S.; Deffieux, G.; Barthe, N.; Rosenbaum, J.; Mérillon, J.M. Distribution of [14C]-trans-resveratrol, a cancer chemopreventive polyphenol, in mouse tissues after oral administration. Life Sci. 2003, 72, 2219–2233. [Google Scholar] [CrossRef]
- Klinge, C.M.; Blankenship, K.A.; Risinger, K.E.; Bhatnagar, S.; Noisin, E.L.; Sumanasekera, W.K.; Zhao, L.; Brey, D.M.; Keynton, R.S. Resveratrol and Estradiol Rapidly Activate MAPK Signaling through Estrogen Receptors α and β in Endothelial Cells. J. Boil. Chem. 2004, 280, 7460–7468. [Google Scholar] [CrossRef]
- Wang, Y.; Catana, F.; Yang, Y.; Roderick, R.; Van Breemen, R.B. An LC-MS Method for analyzing total resveratrol in grape juice, cranberry juice, and in wine. J. Agric. Food Chem. 2002, 50, 431–435. [Google Scholar] [CrossRef]
- Frum, A.; Georgescu, C.; Gligor, F.G.; Lengyel, E.; Stegarus, D.I.; Dobrea, C.M.; Tita, O. Identification and quantification of phenolic compounds from red grape pomace. Sci. Study Res. Chem. Chem. Eng. Biotechnol. Food Ind. 2018, 19, 45–52, 1582-540X. [Google Scholar]
- Casas, L.; Mantell, C.; Rodríguez, M.; De La Ossa, E.M.; Roldan, A.; De Ory, I.; Caro, I.; Blandino, A.; Cardoso, L.C. Extraction of resveratrol from the pomace of Palomino fino grapes by supercritical carbon dioxide. J. Food Eng. 2010, 96, 304–308. [Google Scholar] [CrossRef]
- Fabris, S.; Momo, F.; Ravagnan, G.; Stevanato, R. Antioxidant properties of resveratrol and piceid on lipid peroxidation in micelles and monolamellar liposomes. Biophys. Chem. 2008, 135, 76–83. [Google Scholar] [CrossRef]
- Shahidi, F.; Wanasundara, U.N. Methods for Evaluation of the Oxidative Stability of Lipid-Containing Foods. Food Sci. Technol. Int. Tokyo 1996, 2, 73–81. [Google Scholar] [CrossRef]
- Buckley, D.J.; Morrissey, P.A.; Gray, J.I. Influence of dietary vitamin E on the oxidative stability and quality of pig meat. J. Anim. Sci. 1995, 73, 3122. [Google Scholar] [CrossRef] [PubMed]
- Gonnet, J.-F. Colour effects of co-pigmentation of anthocyanins revisited—1. A colorimetric definition using the CIELAB scale. Food Chem. 1998, 63, 409–415. [Google Scholar] [CrossRef]
- Gimeno, O.; Ansorena, D.; Astiasarán, I.; Bello, J. Characterization of chorizo de Pamplona. Food Chem. 2000, 69, 195–200. [Google Scholar] [CrossRef]
- Bozkurt, H.; Bayram, M. Colour and textural attributes of sucuk during ripening. Meat Sci. 2006, 73, 344–350. [Google Scholar] [CrossRef]
- Sánchez-Alonso, I.; Borderias, A.J. Technological effect of red grape antioxidant dietary fibre added to minced fish muscle. Int. J. Food Sci. Technol. 2008, 43, 1009–1018. [Google Scholar] [CrossRef]
- Sáyago-Ayerdi, S.; Brenes, A.; Goñi, I. Effect of grape antioxidant dietary fiber on the lipid oxidation of raw and cooked chicken hamburgers. LWT 2009, 42, 971–976. [Google Scholar] [CrossRef]
- Jongberg, S.; Skov, S.H.; Tørngren, M.A.; Skibsted, L.H.; Lund, M.N. Effect of white grape extract and modified atmosphere packaging on lipid and protein oxidation in chill stored beef patties. Food Chem. 2011, 128, 276–283. [Google Scholar] [CrossRef]
- Barbut, S.; Zhang, L.; Marcone, M. Effects of pale, normal, and dark chicken breast meat on microstructure, extractable proteins, and cooking of marinated fillets. Poult. Sci. 2005, 84, 797–802. [Google Scholar] [CrossRef]
- Haščík, P.; Trembecká, L.; Bobko, M.; Kačániová, M.; Bučko, O.; Tkačová, J.; Kunová, S. Effect of different feed supplements on selected quality indicators of chicken meat. Potravinarstvo Slovak J. Food Sci. 2015, 9, 427–434. [Google Scholar] [CrossRef][Green Version]
- Jiang, S.-Q.; Jiang, Z.-Y.; Zhou, G.-L.; Lin, Y.-C.; Zheng, C. Effects of Dietary Isoflavone Supplementation on Meat Quality and Oxidative Stability During Storage in Lingnan Yellow Broilers. J. Integr. Agric. 2014, 13, 387–393. [Google Scholar] [CrossRef]
- Mainente, F.; Menin, A.; Alberton, A.; Zoccatelli, G.; Rizzi, C. Evaluation of the sensory and physical properties of meat and fish derivatives containing grape pomace powders. Int. J. Food Sci. Technol. 2018, 54, 952–958. [Google Scholar] [CrossRef]
- Rojas, M.; Brewer, M. Effect of Natural Antioxidants on Oxidative Stability of Cooked, Refrigerated Beef and Pork. J. Food Sci. 2007, 72, S282–S288. [Google Scholar] [CrossRef] [PubMed]
- Balș, C. Quality management and factors on poultry pathway. Lucrări Ştiinţifice Seria Zootehnie 2010, 53, 434–440. [Google Scholar]
- Brannan, R. Effect of grape seed extract on descriptive sensory analysis of ground chicken during refrigerated storage. Meat Sci. 2009, 81, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Rababah, T.M.; Ereifej, K.I.; Al-Mahasneh, M.A.; Al-Rababah, M.A. Effect of Plant Extracts on Physicochemical Properties of Chicken Breast Meat Cooked Using Conventional Electric Oven or Microwave. Poult. Sci. 2006, 85, 148–154. [Google Scholar] [CrossRef]
- Kumanda, C.; Mlambo, V.; Mnisi, C. Valorization of Red Grape Pomace Waste Using Polyethylene Glycol and Fibrolytic Enzymes: Physiological and Meat Quality Responses in Broilers. Animals 2019, 9, 779. [Google Scholar] [CrossRef]
- Zhang, S.; Farouk, M.; Young, O.; Wieliczko, K.; Podmore, C. Functional stability of frozen normal and high pH beef. Meat Sci. 2005, 69, 765–772. [Google Scholar] [CrossRef]
- Mir, N.A.; Rafiq, A.; Kumar, F.; Singh, V.; Shukla, V. Determinants of broiler chicken meat quality and factors affecting them: A review. J. Food Sci. Technol. 2017, 54, 2997–3009. [Google Scholar] [CrossRef]
- Anadon, H.L.S. Biological, Nutritional, and Processing Factors Affecting Breast Meat Quality of Broilers. Ph.D. Thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA, USA, 2002. [Google Scholar]
- Purriños, L.; Bermúdez, R.; Temperán, S.; Franco, D.; Carballo, J.; Lorenzo, J.M. Influence of salt content and processing time on sensory characteristics of cooked “lacón”. Meat Sci. 2011, 87, 436–442. [Google Scholar] [CrossRef]
- Amaral, A.B.; Da Silva, M.V.; Lannes, S.C.D.S. Lipid oxidation in meat: Mechanisms and protective factors—A review. Food Sci. Technol. 2018, 38, 1–15. [Google Scholar] [CrossRef]
- Ruiz-Capillas, C.; Nardoia, M.; Herrero, A.M.; Jimnez-Colmenero, F.; Chamorro, S.; Brenes, A. Effect of Added Grape Seed and Skin on Chicken Thigh Patties during Chilled Storage. Int. J. Food Nutr. Sci. 2017, 4, 67–73. [Google Scholar] [CrossRef]
- Domínguez, R.; Pateiro, M.; Gagaoua, M.; Barba, F.J.; Zhang, W.; Lorenzo, J. A Comprehensive Review on Lipid Oxidation in Meat and Meat Products. Antioxidants 2019, 8, 429. [Google Scholar] [CrossRef] [PubMed]
- Falowo, A.B.; Fayemi, P.O.; Muchenje, V. Natural antioxidants against lipid–protein oxidative deterioration in meat and meat products: A review. Food Res. Int. 2014, 64, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Wąsowicz, E.; Gramza, A.; Hęś, M.; Jeleń, H.H.; Korczak, J.; Małecka, M.; Mildner-Szkudlarz, S.; Rudzińska, M.; Samotyja, U.; Zawirska-Wojtasiak, R. Oxidation of lipids in food. Pol. J. Food Nutr. Sci. 2004, 13, 87–100. [Google Scholar]
- Gatellier, P.; Gomez, S.; Gigaud, V.; Berri, C.; Le Bihan-Duval, E.; Santé-Lhoutellier, V. Use of a fluorescence front face technique for measurement of lipid oxidation during refrigerated storage of chicken meat. Meat Sci. 2007, 76, 543–547. [Google Scholar] [CrossRef]
- Antolovich, M.; Prenzler, P.D.; Patsalides, E.; McDonald, S.; Robards, K. Methods for testing antioxidant activity. Analyst 2002, 127, 183–198. [Google Scholar] [CrossRef]
- Shahidi, F.; Wanasundara, U. Methods for measuring oxidative rancidity in fats and oils. In Food Lipids; Informa UK Limited: New York, NY, USA, 2002; Volume 117, pp. 484–507. [Google Scholar]
- Grotto, D.; Maria, L.S.; Valentini, J.; Paniz, C.; Schmitt, G.; Garcia, S.C.; Pomblum, V.J.; Rocha, J.B.T.; Farina, M. Importance of the lipid peroxidation biomarkers and methodological aspects FOR malondialdehyde quantification. Química Nova 2009, 32, 169–174. [Google Scholar] [CrossRef]
- Pereira, A.L.F.; Abreu, V.K.G. Lipid peroxidation in meat and meat products. In Lipid Peroxidation Research; IntechOpen: London, UK, 2020. [Google Scholar]
- Campo, M.M.; Nute, G.; Hughes, S.; Enser, M.; Wood, J.; Richardson, I. Flavour perception of oxidation in beef. Meat Sci. 2006, 72, 303–311. [Google Scholar] [CrossRef]
- Zhang, Y.; Holman, B.W.B.; Ponnampalam, E.N.; Kerr, M.G.; Bailes, K.L.; Kilgannon, A.K.; Collins, D.; Hopkins, D.L. Understanding beef flavour and overall liking traits using two different methods for determination of thiobarbituric acid reactive substance (TBARS). Meat Sci. 2019, 149, 114–119. [Google Scholar] [CrossRef]
- Goni, I.; Brenes, A.; Centeno, C.; Viveros, A.; Saura-Calixto, F.; Rebole, A.; Arija, I.; Estevez, R. Effect of Dietary Grape Pomace and Vitamin E on Growth Performance, Nutrient Digestibility, and Susceptibility to Meat Lipid Oxidation in Chickens. Poult. Sci. 2007, 86, 508–516. [Google Scholar] [CrossRef] [PubMed]
- Kasapidou, E.; Sossidou, E.N.; Zdragas, A.; Papadaki, C.; Vafeas, G.; Mitlianga, P. Effect of grape pomace supplementation on broiler meat quality characteristics. Eur. Poult. Sci. 2016, 80, 135–142. [Google Scholar] [CrossRef]
- Jayaprakasha, G.K.; Selvi, T.; Sakariah, K. Antibacterial and antioxidant activities of grape (Vitis vinifera) seed extracts. Food Res. Int. 2003, 36, 117–122. [Google Scholar] [CrossRef]
- Hatzidimitriou, E.; Nenadis, N.; Tsimidou, M.Z. Changes in the catechin and epicatechin content of grape seeds on storage under different water activity (aw) conditions. Food Chem. 2007, 105, 1504–1511. [Google Scholar] [CrossRef]
- Lachman, J.; Šulc, M.; Schilla, M. Comparison of the total antioxidant status of Bohemian wines during the wine-making process. Food Chem. 2007, 103, 802–807. [Google Scholar] [CrossRef]
- Iacopini, P.; Baldi, M.; Storchi, P.; Sebastiani, L. Catechin, epicatechin, quercetin, rutin and resveratrol in red grape: Content, in vitro antioxidant activity and interactions. J. Food Compos. Anal. 2008, 21, 589–598. [Google Scholar] [CrossRef]
- Rababah, T.M.; Ereifej, K.I.; Al-Mahasneh, M.A.; Ismaeal, K.; Hidar, A.-G.; Yang, W. Total Phenolics, Antioxidant Activities, and Anthocyanins of Different Grape Seed Cultivars Grown in Jordan. Int. J. Food Prop. 2008, 11, 472–479. [Google Scholar] [CrossRef]
- Xu, C.; Zhang, Y.; Cao, L.; Lu, J. Phenolic compounds and antioxidant properties of different grape cultivars grown in China. Food Chem. 2010, 119, 1557–1565. [Google Scholar] [CrossRef]
- Du, J.; Zeng, Y.; Wang, H.; Qian, Y.; Li, H.; Chen, Q.; Chen, W.; Cui, J. CuZnSOD gene expression and its relationship with anti-oxidative capacity and pork quality. S. Afr. J. Anim. Sci. 2010, 40, 265–272. [Google Scholar] [CrossRef]
| Diet Composition % | Starter Stage (1–13 Days) | Grower Stage (14–28 Days) | Finisher Stage (29–42 Days) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | E1 | E2 | E3 | E4 | Control | E1 | E2 | E3 | E4 | ||
| Corn | 32.57 | 35.50 | 31.37 | 37.16 | 31.60 | 37.36 | 39.37 | 34.23 | 31.12 | 34.46 | 31.97 |
| Wheat | 20.00 | 20.00 | 20.00 | 10.00 | 20.00 | 10.00 | 20.00 | 20.00 | 20.00 | 20.00 | 19.58 |
| Corn gluten | 2.00 | 4.00 | 6.00 | 9.25 | 6.00 | 8.73 | 6.00 | 6.00 | 10.00 | 6.00 | 10.00 |
| Soybean meal | 36.17 | 27.02 | 25.02 | 22.50 | 24.86 | 22.83 | 20.81 | 21.32 | 16.81 | 21.16 | 16.53 |
| Flax meal | 0.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 | 4.00 |
| White grape pomace | 0.00 | 0.00 | 3.00 | 6.00 | 0.00 | 0.00 | 0.00 | 3.00 | 6.00 | 0.00 | 0.00 |
| Red grape pomace | 0.00 | 0.00 | 0.00 | 0.00 | 3.00 | 6.00 | 0.00 | 0.00 | 0.00 | 3.00 | 6.00 |
| Vegetable oil | 3.85 | 4.21 | 5.21 | 5.50 | 5.13 | 5.50 | 4.67 | 6.29 | 6.68 | 6.21 | 6.50 |
| Monocalcium phosphate | 1.68 | 1.54 | 1.56 | 1.65 | 1.56 | 1.65 | 1.45 | 1.45 | 1.49 | 1.45 | 1.49 |
| Calcium carbonate | 1.51 | 1.40 | 1.41 | 1.40 | 1.41 | 1.40 | 1.33 | 1.32 | 1.34 | 1.32 | 1.35 |
| Salt | 0.39 | 0.36 | 0.36 | 0.37 | 0.36 | 0.37 | 0.33 | 0.34 | 0.34 | 0.34 | 0.34 |
| Methionine | 0.33 | 0.29 | 0.28 | 0.24 | 0.28 | 0.25 | 0.26 | 0.27 | 0.22 | 0.27 | 0.23 |
| Lysine | 0.30 | 0.41 | 0.47 | 0.55 | 0.48 | 0.54 | 0.48 | 0.47 | 0.60 | 0.48 | 0.61 |
| Threonine | 0.15 | 0.22 | 0.27 | 0.33 | 0.27 | 0.32 | 0.25 | 0.26 | 0.35 | 0.26 | 0.35 |
| Choline | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 |
| Vitamin-mineral premix 1 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| Calculated analysis % | |||||||||||
| Metabolizable energy, kcal/kg | 3039.79 | 3100.00 | 3100.00 | 3100.00 | 3100.00 | 3100.00 | 3200.00 | 3200.00 | 3200.00 | 3200.00 | 3200.00 |
| Crude protein | 23.00 | 21.50 | 21.50 | 21.50 | 21.50 | 21.50 | 20.00 | 20.00 | 20.00 | 20.00 | 20.00 |
| Ether extractives | 5.48 | 6.34 | 7.33 | 7.80 | 7.26 | 7.80 | 6.86 | 8.42 | 8.81 | 8.34 | 8.69 |
| Crude fiber | 3.77 | 3.36 | 3.45 | 3.56 | 3.54 | 3.78 | 3.14 | 3.34 | 3.33 | 3.44 | 3.52 |
| Lysine | 1.44 | 1.29 | 1.29 | 1.29 | 1.29 | 1.29 | 1.19 | 1.19 | 1.19 | 1.19 | 1.19 |
| Methionine | 0.69 | 0.63 | 0.63 | 0.62 | 0.63 | 0.62 | 0.59 | 0.60 | 0.58 | 0.60 | 0.59 |
| Threonine | 0.97 | 0.88 | 0.88 | 0.88 | 0.88 | 0.88 | 0.81 | 0.81 | 0.81 | 0.81 | 0.81 |
| Tryptophan | 0.25 | 0.20 | 0.19 | 0.16 | 0.19 | 0.17 | 0.17 | 0.17 | 0.14 | 0.17 | 0.14 |
| Chemical analysis (g/100 g total FAME) | |||||||||||
| Alpha-linolenic acid (C18: 3n3) | 8.98 | 9.26 | 8.79 | 9.29 | 9.14 | 8.78 | 8.58 | 8.84 | 8.79 | 8.90 | |
| Linoleic acid (C18: 2n6) | - | 49.46 | 51.00 | 51.28 | 50.97 | 51.06 | 50.50 | 51.53 | 51.85 | 50.55 | 52.68 |
| PUFA | - | 59.23 | 61.23 | 61.15 | 61.12 | 61.11 | 60.07 | 60.87 | 61.61 | 60.38 | 62.14 |
| n-3 | - | 9.30 | 9.58 | 9.18 | 9.63 | 9.51 | 9.11 | 8.91 | 9.14 | 9.10 | 9.08 |
| n-6 | - | 49.93 | 51.65 | 51.97 | 51.49 | 51.60 | 50.96 | 51.96 | 52.47 | 51.28 | 53.06 |
| Groups | Initial Weight (g) | Final Weight (g) | Average Daily Weight Gain (g/Broiler/Day) | Average Daily Feed Intake (g Feed/Broiler/Day) | Feed Conversion Ratio (g Feed/g Gain) |
|---|---|---|---|---|---|
| C | 506.54 | 3136.67 | 94.04 | 149.36 | 1.57 |
| E1 | 506.51 | 2966.11 | 91.26 | 148.61 | 1.61 |
| E2 | 506.91 | 2999.81 | 91.63 | 146.44 | 1.58 |
| E3 | 506.85 | 2937.04 | 90.09 | 145.85 | 1.60 |
| E4 | 506.33 | 2995.74 | 89.25 | 140.62 | 1.56 |
| SEM | 3.192 | 38.353 | 1.237 | 4.693 | 0.033 |
| p value | >0.9999 | 0.7559 | 0.8139 | 0.9826 | 0.9904 |
| Parameters | L* | a* | b* | h* | C* | ΔΕ | pH |
|---|---|---|---|---|---|---|---|
| Thigh meat | |||||||
| Control | 54.41 a | 3.42 b | 11.62 b | 0.79 | 12.16 ab | 0.00 | 6.25 a |
| E1 | 55.44 a | 6.74 a | 12.80 ab | 1.33 | 13.16 a | 3.86 | 5.97 b |
| E2 | 53.04 ab | 5.31 ab | 13.80 ab | 1.34 | 12.36 ab | 3.19 | 6.07 ab |
| E3 | 50.66 b | 2.17 b | 12.18 b | 1.32 | 12.04 ab | 3.99 | 5.96 b |
| E4 | 52.91 ab | 7.63 a | 17.20 a | 1.41 | 11.01 b | 7.16 | 6.04 b |
| SEM | 0.625 | 0.577 | 0.756 | 0.100 | 0.295 | - | 0.033 |
| p value | 0.0170 | 0.0061 | 0.0200 | 0.2786 | 0.0254 | - | 0.0284 |
| Breast meat | |||||||
| Control | 53.24 | 1.10 | 13.89 b | 0.89 | 13.81 ab | 0.00 | 5.92 |
| E1 | 55.35 | 1.75 | 15.90 ab | 0.89 | 12.68 ab | 2.98 | 5.90 |
| E2 | 54.28 | 0.98 | 16.80 a | 0.81 | 14.34 a | 3.09 | 6.02 |
| E3 | 53.11 | 1.56 | 18.21 a | 0.87 | 13.70 ab | 4.34 | 5.90 |
| E4 | 55.37 | 1.35 | 17.10 a | −0.50 | 11.84 b | 3.86 | 6.04 |
| SEM | 0.483 | 0.331 | 0.440 | 0.225 | 0.358 | - | 0.030 |
| p value | 0.4143 | 0.9550 | 0.0167 | 0.6456 | 0.1802 | - | 0.3758 |
| Parameters | Hardness (N) | Springiness (%) | Resilience (adm) | Cohesiveness (adm) | Gumminess (N) |
|---|---|---|---|---|---|
| Thigh meat | |||||
| Control | 32.76 b | 99.63 b | 2.96 ab | 0.44 | 15.50 |
| E1 | 35.87 ab | 99.73 ab | 3.26 b | 0.48 | 17.79 |
| E2 | 39.56 ab | 99.64 ab | 3.37 b | 0.48 | 18.98 |
| E3 | 44.75 a | 99.86 a | 2.80 a | 0.44 | 20.05 |
| E4 | 37.65 ab | 99.55 b | 3.27 b | 0.45 | 17.40 |
| SEM | 1.765 | 0.035 | 0.072 | 0.008 | 0.957 |
| p value | 0.0253 | 0.0320 | 0.0352 | 0.4112 | 0.6110 |
| Breast meat | |||||
| Control | 8.88c | 99.76 | 1.29c | 0.24 c | 2.17 b |
| E1 | 13.83 bc | 99.70 | 2.06 b | 0.35 ab | 5.01 a |
| E2 | 14.10 bc | 99.74 | 1.89 b | 0.29 bc | 4.38 a |
| E3 | 11.91 bc | 99.65 | 2.49 a | 0.40 a | 4.83 a |
| E4 | 16.01 a | 99.71 | 1.97 b | 0.28 c | 4.36 a |
| SEM | 0.603 | 0.024 | 0.081 | 0.012 | 0.257 |
| p value | 0.0017 | 0.6530 | <0.0001 | <0.0001 | 0.0004 |
| Parameters | Primary Parameters | Secondary Parameters | ||
|---|---|---|---|---|
| Conjugated Dienes (µmoli/g) | Conjugated Trienes (µmoli/g) | p-Anisidine Value | TBARS (mg/kg) | |
| Thigh meat | ||||
| Control | 7.52 | 3.49 | 17.95 | 0.23 a |
| E1 | 6.15 | 2.96 | 15.55 | 0.15 b |
| E2 | 7.23 | 3.22 | 15.27 | 0.18 bc |
| E3 | 6.86 | 3.30 | 15.29 | 0.17 bc |
| E4 | 6.83 | 3.45 | 18.47 | 0.19 c |
| SEM | 0.232 | 0.082 | 0.536 | 0.007 |
| p Value | 0.4415 | 0.2706 | 0.1281 | 0.0001 |
| Breast meat | ||||
| Control | 5.16 | 2.50 | 18.49a | 0.17 a |
| E1 | 4.31 | 2.03 | 14.92ab | 0.14 b |
| E2 | 4.43 | 2.20 | 10.29b | 0.16 ab |
| E3 | 4.78 | 2.28 | 15.58ab | 0.15 ab |
| E4 | 4.50 | 2.39 | 17.61ab | 0.14 b |
| SEM | 0.274 | 0.099 | 1.038 | 0.019 |
| p Value | 0.8973 | 0.6621 | 0.0119 | 0.0218 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turcu, R.P.; Panaite, T.D.; Untea, A.E.; Șoica, C.; Iuga, M.; Mironeasa, S. Effects of Supplementing Grape Pomace to Broilers Fed Polyunsaturated Fatty Acids Enriched Diets on Meat Quality. Animals 2020, 10, 947. https://doi.org/10.3390/ani10060947
Turcu RP, Panaite TD, Untea AE, Șoica C, Iuga M, Mironeasa S. Effects of Supplementing Grape Pomace to Broilers Fed Polyunsaturated Fatty Acids Enriched Diets on Meat Quality. Animals. 2020; 10(6):947. https://doi.org/10.3390/ani10060947
Chicago/Turabian StyleTurcu, Raluca Paula, Tatiana Dumitra Panaite, Arabela Elena Untea, Cristina Șoica, Mădălina Iuga, and Silvia Mironeasa. 2020. "Effects of Supplementing Grape Pomace to Broilers Fed Polyunsaturated Fatty Acids Enriched Diets on Meat Quality" Animals 10, no. 6: 947. https://doi.org/10.3390/ani10060947
APA StyleTurcu, R. P., Panaite, T. D., Untea, A. E., Șoica, C., Iuga, M., & Mironeasa, S. (2020). Effects of Supplementing Grape Pomace to Broilers Fed Polyunsaturated Fatty Acids Enriched Diets on Meat Quality. Animals, 10(6), 947. https://doi.org/10.3390/ani10060947









