Effect of Soybean Meal Substitution by Raw Chickpea Seeds on Thermal Properties and Fatty Acid Composition of Subcutaneous Fat Tissue of Broiler Chickens
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Animals and Experimental Design
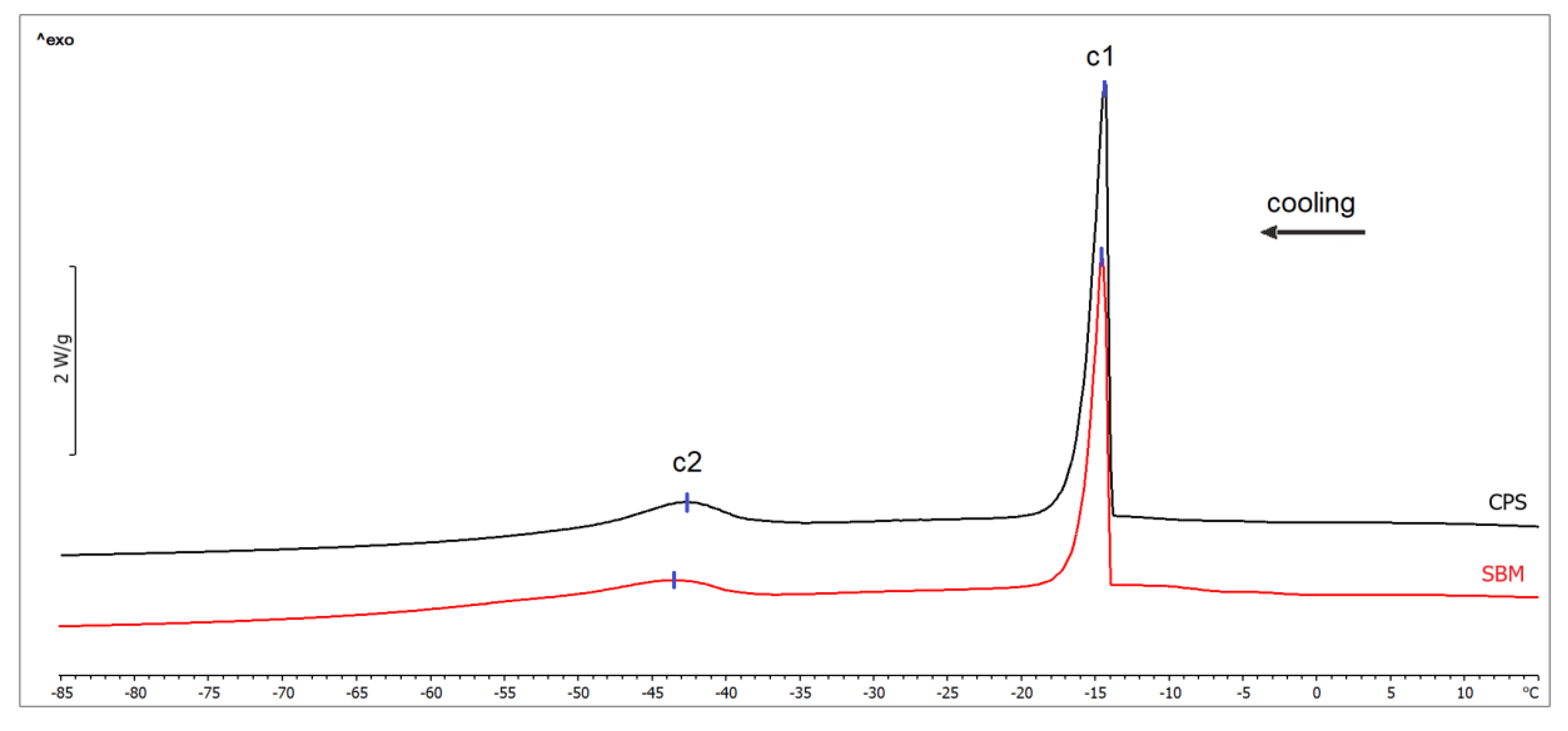
2.3. DSC Measurements
2.4. Determination of Fatty Acid Composition in Subcutaneous Fat
2.5. Statistical Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fouad, A.M.; El-Senousey, H.K. Nutritional factors affecting abdominal fat deposition in poultry: A review. Asian Australas. J. Anim. Sci. 2014, 27, 1057–1068. [Google Scholar] [CrossRef] [PubMed]
- Alleman, F.; Michel, J.; Chagneau, A.M.; Leclercq, B. The effects of dietary protein independent of essential amino acids on growth and body composition in genetically lean and fat chickens. Br. Poult. Sci. 2000, 41, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Adams, K.A.; Davis, A.J. Dietary protein concentration regulates the mRNA expression of chicken hepatic malic enzyme. J. Nutr. 2001, 131, 2269–2274. [Google Scholar] [CrossRef]
- Heydarpour, F.; Amini, B.; Kalantari, S.S.; Akbari, A.; Heydarpour, P. Mean percentage of skin and visible fat in 10 chicken carcass weight. Int. J. Poult. Sci. 2007, 6, 43–47. [Google Scholar]
- Marx, S.D.; Siares, J.M.; Rrestes, R.C.; Schinitzler, E.; Oliviera, C.S.; Demiate, I.M.; Backes, G.T.; Steffens, J. Influence of sex on the physical–chemical characteristics of abdominal chicken fat. Braz. J. Poult. Sci. 2016, 18, 269–276. [Google Scholar] [CrossRef]
- Wang, G.; Kin, W.K.; Cline, M.A.; Gilbert, E.R. Factors affecting adipose tissue development in chickens: A review. Poult. Sci. 2017, 96, 3687–3699. [Google Scholar] [CrossRef] [PubMed]
- Tomaszewska, E.; Muszyński, S.; Dobrowolski, P.; Kwiecień, M.; Klebaniuk, R.; Szymańczyk, S.; Tomczyk, A.; Kowalik, S.; Milczarek, A.; Świetlicka, I. The influence of dietary replacement of soybean meal with high–tannin faba beans on gut–bone axis and metabolic response in broiler chickens. Ann. Anim. Sci. 2018, 18, 801–824. [Google Scholar] [CrossRef]
- Tomaszewska, E.; Dobrowolski, P.; Klebaniuk, R.; Kwiecień, M.; Tomczyk-Warunek, A.; Szymańczyk, S.; Kowalik, S.; Milczarek, A.; Blicharski, T.; Muszyński, S. Gut–bone axis response to dietary replacement of soybean meal with raw low–tannin faba bean seeds in broiler chickens. PLoS ONE 2018, 13, e0194969. [Google Scholar] [CrossRef]
- Milczarek, A.; Osek, M. Effectiveness evaluation of use of various protein feeds for broiler chicken feeding. Ann. Anim. Sci. 2019, 19, 1063–1081. [Google Scholar] [CrossRef]
- Rutkowski, A.; Kaczmarek, S.A.; Hejdysz, M.; Nowaczewski, S.; Jamroz, D. Concentrates made from legume seeds (Lupinus Angustifolius, Lupinus Luteus and Pisum Sativum) and rapeseed meal as protein sources in laying hen diets. Ann. Anim. Sci. 2015, 15, 129–142. [Google Scholar] [CrossRef]
- Konieczka, P.; Czerwiński, J.; Jankowiak, J.; Ząbek, K.; Smulikowska, S. Effects of partial replacement of soybean meal with rapeseed meal, narrow–leaved lupin, DDGS, and probiotic supplementation, on performance and gut microbiota activity and diversity in broilers. Ann. Anim. Sci. 2019, 19, 1115–1131. [Google Scholar] [CrossRef]
- Muszyński, S.; Kwiecień, M.; Świetlicki, M.; Dobrowolski, P.; Tatarczak, J.; Gładyszewska, B. Effects of replacing soybean meal with chickpea seeds in the diet on mechanical and thermal properties of tendon tissue in broiler chicken. Poult. Sci. 2018, 97, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Muszyński, S.; Tomaszewska, E.; Dobrowolski, P.; Kwiecień, M.; Wiącek, D.; Świetlicka, I.; Skibińska, M.; Szymańska-Chargot, M.; Orzeł, J.; Świetlicki, M.; et al. Analysis of bone osteometry, mineralization, mechanical and histomorphometrical properties of tibiotarsus in broiler chickens demonstrates a influence of dietary chickpea seeds (Cicer arietinum L.) inclusion as a primary protein source. PLoS ONE 2018, 13, e0208921. [Google Scholar] [CrossRef] [PubMed]
- Józefiak, D.; Józefiak, A.; Kierończyk, B.; Rawski, M.; Świątkiewicz, S.; Długosz, J.; Engberg, R.M. Insects–a natural nutrient source for poultry–a review. Ann. Anim. Sci. 2016, 16, 297–313. [Google Scholar] [CrossRef]
- Józefiak, A.; Nogales-Mérida, S.; Mikołajczak, Z.; Rawski, M.; Kierończyk, B.; Mazurkiewicz, J. The utilization of full-fat insect meal in rainbow trout (Oncorhynchus mykiss) nutrition: The effects on growth performance, intestinal microbiota and gastro-intestinal tract histomorphology. Ann. Anim. Sci. 2019, 19, 747–765. [Google Scholar] [CrossRef]
- Lei, X.J.; Kim, T.H.; Park, J.H.; Kim, I.H. Evaluation of supplementation of defatted black soldier fly (Hermetia illucens) larvae meal in beagle dogs. Ann. Anim. Sci. 2019, 19, 767–777. [Google Scholar] [CrossRef]
- Bampidis, V.; Christodoulou, V. Chickpeas (Cicer arietinum L.) in animal nutrition: A review. Anim. Feed Sci. Technol. 2011, 168, 1–20. [Google Scholar] [CrossRef]
- Trębacz, H.; Szczęsna, A.; Arczewska, M. Thermal stability of collagen in naturally ageing and in vitro glycated rabbit tissues. J. Therm. Anal. Calorim. 2018, 134, 1903–1911. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, L.; Xiong, Y.; Liu, Y.; Yu, S.; Wu, C.; Guo, W. Effect of in vitro storage duration on measured mechanical properties of brain tissue. Sci. Rep. 2018, 8, 1247. [Google Scholar] [CrossRef]
- Wiegand, N.; Nőt, L.G.; Patczai, B.; Lőrinczy, D. The role of differential scanning calorimetry in the diagnostics of musculoskeletal disease. EC Orthop. 2017, 7, 164–177. [Google Scholar]
- Vega, S.; Garcia-Gonzalez, M.; Lanas, A.; Velazquez-Campoy, A.; Abian, O. Deconvolution analysis for classifying gastric adenocarcinoma patients based on differential scanning calorimetry serum thermograms. Sci. Rep. 2015, 5, 7988. [Google Scholar] [CrossRef] [PubMed]
- Ferencz, A.; Nedvig, K.; László, E.; Magyarlaki, T.; Lőrinczy, D. DSC examination of kidney tissue following warm ischemia and reperfusion injury. Thermochim. Acta 2011, 525, 161–166. [Google Scholar] [CrossRef]
- Szabó, I.; Bognár, G.; Kereskai, L.; Szász, K.; Lőrinczy, D. Differential scanning calorimetric and histological examinations of the long head of the biceps in cadavers. J. Therm. Anal. Calorim. 2007, 88, 343–349. [Google Scholar] [CrossRef]
- Chaudhury, S.; Holland, C.; Porter, D.; Tirlapur, U.K.; Vollrath, F.; Carr, A.J. Torn human rotator cuff tendons have reduced collagen thermal properties on differential scanning calorimetry. J. Orthop. Res. 2011, 29, 1938–1943. [Google Scholar] [CrossRef]
- Stangierski, J.; Tomaszewska-Gras, J.; Baranowska, H.M.; Krzywdzińska-Bartjowiak, M.; Konieczny, P. The effect of deep pectoral myopathy on the properties of broiler chicken muscles characterised by selected instrumental techniques. Eur. Food Res. Technol. 2019, 245, 459–467. [Google Scholar] [CrossRef]
- Wattanachant, S.; Benjakul, S.; Ledward, D.A. Microstructure and thermal characteristics of Thai indigenous and broiler chicken muscles. Poult. Sci. 2005, 84, 328–336. [Google Scholar] [CrossRef]
- Voutila, L.; Ruusunen, M.; Jouppila, K.; Puolanne, E. Thermal properties of connective tissue in breast and leg muscles of chickens and turkeys. J. Sci. Food Agric. 2009, 89, 890–896. [Google Scholar] [CrossRef]
- Saadi, S.; Ariffin, A.A.; Ghazali, H.M.; Miskandar, M.S.; Boo, H.C.; Abdulkarim, S.M. Application of differential scanning calorimetry (DSC), HPLC and pNMR for interpretation primary crystallization caused by combined low and high melting TAGs. Food Chem. 2012, 132, 603–612. [Google Scholar] [CrossRef]
- Yılmaz, M.T.; Karakaya, M.; Aktas, N. Composition and thermal properties of cattle fats. Eur. J. Lipid Sci. Technol. 2010, 112, 410–416. [Google Scholar] [CrossRef]
- Traffano-Schiffo, M.V.; Castro-Giraldez, M.; Colom, R.J.; Fito, P.J. Development of a spectrophotometric system to detect white striping physiopathy in whole chicken carcasses. Sensors 2017, 17, 1024. [Google Scholar] [CrossRef] [PubMed]
- Muszyński, S.; Tomaszewska, E.; Kwiecień, M.; Dobrowolski, P.; Świetlicka, I.; Tanaś, W.; Sołowiej, B.; Ejtel, M.; Szcześniak, E.; Tomczyk-Warunek, A. The dietary inclusion of chickpea seeds (Cicer arietinum L.) influences thermal properties of muscle proteins but not the texture of drumstick muscle in broiler chickens. Braz. J. Poult. Sci. 2019, 21, eRBCA-2018-0806. [Google Scholar] [CrossRef]
- Muszyński, S.; Tomaszewska, E.; Kwiecień, M.; Dobrowolski, P. The effect of raw chickpea seeds in broiler chickens feed on the thermal properties of collagen in the skin tissue. Pasze Przem. 2019, 28, 43–46. [Google Scholar]
- Blicharski, T.; Tomaszewska, E.; Dobrowolski, P.; Hułas-Stasiak, M.; Muszyński, S. A metabolite of leucine (β–hydroxy–β–methylbutyrate) given to sows during pregnancy alters bone development of their newborn offspring by hormonal modulation. PLoS ONE 2017, 12, e0179693. [Google Scholar] [CrossRef] [PubMed]
- Dahimi, O.; Rahim, A.A.; Abdulkarim, S.M.; Hassan, M.S.; Hashari, S.B.T.; Mashitoh, A.S.; Saadi, S. Multivariate statistical analysis treatment of DSC thermal properties for animal fat adulteration. Food Chem. 2014, 158, 132–138. [Google Scholar] [CrossRef]
- Marikkar, J.M.N.; Ghazali, H.M.; Che Man, Y.B.; Lai, O.M. The use of cooling and heating thermograms to monitoring of tallow, lard and chicken fat adulterations in canola oil. Food Res. Int. 2002, 35, 1007–1014. [Google Scholar] [CrossRef]
- Folch, J.M.; Lees, M.; Stanley, G.H.S. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar]
- Ciurescu, G.; Vasilachi, A.; Grosu, H. Efficacy of microbial phytase on growth performance, carcass traits, bone mineralization, and blood biochemistry parameters in broiler turkeys fed raw chickpea (Cicer arietinum L., cv. Burnas) diets. J. App. Poult. Res. 2020, 29, 171–184. [Google Scholar] [CrossRef]
- Knothe, G.; Dunn, R.O. A comprehensive evaluation of the melting points of fatty acids and esters determined by differential scanning calorimetry. J. Am. Oil Chem. Soc. 2009, 86, 843–856. [Google Scholar] [CrossRef]
- Lee, J.W.; Kil, D.Y.; Keever, B.D.; Killefer, J.; McKeith, F.K.; Sulabo, R.C.; Stein, H.H. Carcass fat quality of pigs is not improved by adding corn germ, beef tallow, palm kernel oil, or glycerol to finishing diets containing distillers dried grains with solubles. J. Anim. Sci. 2013, 91, 2426–2437. [Google Scholar] [CrossRef]
- Jukanti, A.K.; Gaur, P.M.; Gowda, C.L.; Chibbar, R.N. Nutritional quality and health benefits of chickpea (Cicer arietinum L.): A review. Br. J. Nutr. 2012, 108 (Suppl. 1), S11–S26. [Google Scholar] [CrossRef]
- Watkins, B.A. Importance of essential fatty acids and their derivatives in poultry. J. Nutr. 1991, 121, 1475–1485. [Google Scholar] [CrossRef]
- Bavelaar, F.J.; Beynen, A.C. Relationships between dietary fatty acid composition and either melting point or fatty acid profile of adipose tissue in broilers. Meat Sci. 2003, 64, 133–149. [Google Scholar] [CrossRef]
- Gallardo, M.A.; Perez, D.D.; Leighton, F.M. Modification of fatty acid composition in broiler chickens fed canola oil. Biol. Res. 2012, 45, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Diaw, M.T.; Dieng, A.; Mergeai, G.; Dotreppe, O.; Youssouf, I.; Hornick, J.L. Effect of groundnut cake substitution by glandless cottonseed kernels on broilers production: Animal performance, nutrient digestibility, carcass characteristics and fatty acid composition of muscle and fat. Int. J. Poult. Sci. 2010, 9, 473–481. [Google Scholar]
- Smulikowska, S.; Rutkowski, A. Recommended Allowances and Nutritive Value of Feedstuffs. In Poultry Feeding Standards, 5th ed.; The Kielanowski Institute of Animal Physiology and Nutrition, PAS: Jabłonna, Poland, 2018; p. 148. [Google Scholar]
- Niu, J.L.; Zhang, J.; Wei, L.Q.; Zhang, W.J.; Nie, C.X. Effect of fermented cottonseed meal on the lipid-related indices and serum metabolic profiles in broiler chickens. Animas 2019, 9, 930. [Google Scholar] [CrossRef] [PubMed]
- Moran, E.T. Response of broiler strains differing in body fat to inadequate methionine: Live performance and processing yields. Poult. Sci. 1994, 73, 1116–1126. [Google Scholar] [CrossRef] [PubMed]
- Murawska, D.; Kubińska, M.; Gesek, M.; Zduńczyk, Z.; Brzostowska, U.; Jankowski, J. The effect of different dietary levels and sources of methionine on the growth performance of turkeys, carcass and meat quality. Ann. Anim. Sci. 2018, 18, 525–540. [Google Scholar] [CrossRef]
- Grisoni, M.L.; Uzu, G.; Larbier, M.; Geraert, P.A. Effect of dietary lysine on lipogenesis in broilers. Reprod. Nutr. Dev. 1991, 31, 683–690. [Google Scholar] [CrossRef]
- Nasr, J.; Kheiri, F. Effects of lysine levels of diets formulated based on total or digestible amino acids on broiler carcass composition. Rev. Bras. Cienc. Avıcola 2012, 14, 249–258. [Google Scholar] [CrossRef]
- Orgaard, A.; Jensen, L. The effects of soy isoflavones on obesity. Exp. Biol. Med. 2008, 233, 1066–1080. [Google Scholar] [CrossRef]
- Takahashi, Y.; Ide, T. Effects of soy protein and isoflavone on hepatic fatty acid synthesis and oxidation and mRNA expression of uncoupling proteins and peroxisome proliferator–activated receptor γ in adipose tissues of rats. J. Nutr. Biochem. 2008, 19, 682–693. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Mu, Y.; Li, H.; Ding, N.; Wang, Q.; Wang, Y.; Wang, S.; Wang, N. Peroxisome proliferator–activated receptor gamma gene: A key regulator of adipocyte differentiation in chickens. Poult. Sci. 2008, 87, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Payne, R.; Bidner, T.; Southern, L.; McMillin, K. Dietary effects of soy isoflavones on growth and carcass traits of commercial broilers. Poult. Sci. 2001, 80, 1201–1207. [Google Scholar] [CrossRef]
- Chang, W.H.; Zheng, A.J.; Chen, Z.M.; Zhang, S.; Cai, H.Y.; Liu, G.H. β-Casomorphin increases fat deposition in broiler chickens by modulating expression of lipid metabolism genes. Animal 2019, 13, 777–783. [Google Scholar] [CrossRef]
- Bai, S.; Wang, G.; Zhang, W.; Zhang, S.; Rice, B.B.; Cline, M.A.; Gilbert, E.R. Broiler chicken adipose tissue dynamics during the first two weeks post-hatch. Comp. Biochem. Physiol. A 2015, 189, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.Y.; Tang, S.Q. Insulin-induced gene: A new regulator in lipid metabolism. Peptides 2010, 31, 2145–2150. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Sun, B.; Shang, Z.; Leng, L.; Wang, Y.; Wang, N.; Li, H. Comparison of adipose tissue cellularity in chicken lines divergently selected for fatness. Poult. Sci. 2011, 90, 2024–2034. [Google Scholar] [CrossRef]
- Husbands, D.R. The effect of dietary copper on the composition of adipose tissue triglycerides in the broiler chicken. Br. Poult. Sci. 1972, 13, 201–205. [Google Scholar] [CrossRef]
- Hrdinka, C.; Zollitsch, W.; Knaus, W.; Lettner, F. Effects of dietary fatty acid pattern on melting point and composition of adipose tissues and intramuscular fat of broiler carcasses. Poult. Sci. 1996, 75, 208–215. [Google Scholar] [CrossRef]
- Wang, G.; McConn, B.R.; Liu, D.; Cline, M.A.; Gilbert, E.R. The effects of dietary macronutrient composition on lipid metabolism associated factor gene expression in the adipose tissue of chickens are influenced by fasting and refeeding. BMC Obes. 2017, 4, 14. [Google Scholar] [CrossRef]
- Resnyk, C.W.; Carré, W.; Wang, X.; Porter, T.E.; Simon, J.; Le Bihan-Duval, E.; Duclos, M.J.; Aggrey, S.E.; Cogburn, L.A. Transcriptional analysis of abdominal fat in chickens divergently selected on bodyweight at two ages reveals novel mechanisms controlling adiposity: Validating visceral adipose tissue as a dynamic endocrine and metabolic organ. BMC Genom. 2017, 18, 626. [Google Scholar] [CrossRef] [PubMed]
- De Almeida Mallmann, B.; Martin, E.M.; Soo Kim, K.; Calderon-Apodaca, N.L.; Baxter, M.F.A.; Latorre, J.D.; Hernandez-Velasco, X.; Paasch-Martinez, L.; Owens, C.M.; Tellez-Isaias, G.; et al. Evaluation of bone marrow adipose tissue and bone mineralization on broiler chickens affected by wooden breast myopathy. Front. Physiol. 2019, 10, 674. [Google Scholar] [CrossRef] [PubMed]
- Frayn, K. Adipose tissue as a buffer for daily lipid flux. Diabetologia 2002, 45, 1201–1210. [Google Scholar] [CrossRef] [PubMed]

| Starter (d. 1–21) | Grower (d. 22–35) | Finisher (d. 36–42) | ||||
|---|---|---|---|---|---|---|
| SBM | CPS | SBM | CPS | SBM | CPS | |
| Ingredient (%) | ||||||
| Maize | 10.00 | 10.00 | 10.00 | 10.00 | 15.00 | 10.00 |
| Wheat | 53.75 | 21.40 | 44.91 | 19.41 | 35.25 | 19.95 |
| Soybean meal 1 | 28.65 | - | 21.50 | - | 19.40 | - |
| Chickpea seeds 2 | - | 45.00 | - | 45.00 | - | 45.00 |
| Triticale | - | 10.00 | 10.00 | 10.00 | 15.00 | 10.00 |
| Rapeseed meal | - | 2.00 | 4.00 | - | 5.00 | - |
| Soybean oil | 2.40 | 2.40 | 4.40 | 4.40 | 5.20 | 5.20 |
| Monocalcium phosphate | 0.88 | 0.88 | 0.83 | 0.83 | 0.80 | 0.80 |
| Limestone | 1.35 | 1.35 | 1.31 | 1.31 | 1.30 | 1.30 |
| Sodium bicarbonate | 0.08 | 0.08 | 0.08 | 0.08 | 0.08 | 0.08 |
| Sodium chloride | 0.30 | 0.30 | 0.27 | 0.27 | 0.27 | 0.27 |
| Fat-protein concentrate 3 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| Premix vita-min | 0.50 I | 0.50 I | 0.50 II | 0.50 II | 0.50 III | 0.50 III |
| Choline chloride | - | 4.00 | - | 6.00 | - | 4.70 |
| DL-methionine 99% | 0.09 | 0.09 | 0.10 | 0.10 | 0.10 | 0.10 |
| L-lysine HCl 78% | 0.30 | 0.30 | 0.30 | 0.30 | 0.30 | 0.30 |
| L-threonine 99% | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 | 0.50 |
| Carbovet 4 | 0.20 | 0.20 | 0.30 | 0.30 | 0.30 | 0.30 |
| Analyzed composition 5 | ||||||
| Crude protein, % | 21.1 | 21.2 | 19.0 | 19.1 | 18.0 | 18.1 |
| Crude fat, % | 4.28 | 5.21 | 6.23 | 8.23 | 7.09 | 9.0 |
| Crude fiber, % | 3.12 | 1.32 | 3.34 | 1.23 | 3.37 | 1.24 |
| Lysine, % | 1.34 | 0.98 | 1.21 | 0.86 | 1.14 | 0.78 |
| Methionine + Cysteine, % | 0.97 | 0.82 | 0.88 | 0.65 | 0.90 | 0.61 |
| Total Ca, % | 0.93 | 0.83 | 0.91 | 0.82 | 0.82 | 0.81 |
| Total P, % | 0.69 | 0.51 | 0.69 | 0.45 | 0.68 | 0.44 |
| Calculated composition | ||||||
| ME, MJ/kg | 12.4 | 12.5 | 12.9 | 13.0 | 13.1 | 13.1 |
| Bioavailable P, % | 0.44 | 0.35 | 0.42 | 0.34 | 0.41 | 0.33 |
| Total Ca / bioavailable P | 2.12 | 2.32 | 2.14 | 2.40 | 2.17 | 2.41 |
| Transition | Temperature T (°C) | p-value | Enthalpy ΔH (J/g) | p-value | ||
|---|---|---|---|---|---|---|
| SBM | CPS | SBM | CPS | |||
| Water freezing (c1) | −14.21 ± 1.36 | −13.44 ± 1.86 | 0.361 | 22.91 ± 5.50 | 29.38 ± 5.10 | 0.029 |
| Lipids crystallization (c2) | −43.25 ± 0.99 | −42.98 ± 2.16 | 0.753 | 5.58 ± 0.95 | 6.15 ± 1.78 | 0.434 |
| Transition | Temperature T (°C) | p-value | Enthalpy ΔH (J/g) | p-value | ||
|---|---|---|---|---|---|---|
| SBM | CPS | SBM | CPS | |||
| Low-melting fraction of monounsaturated TAGs (h1) | −6.76 ± 0.55 | −15.81 ± 4.28 | <0.001 | 84.64 ± 7.48 | 106.76 ± 18.42 | 0.007 |
| High-melting fraction of polyunsaturated TAGs (h2) | 0.34 ± 0.54 | 0.52 ± 0.35 | 0.458 | |||
| Melting of saturated TAGs (h3) | 30.31 ± 1.03 | 29.43 ± 0.67 | 0.064 | 2.88 ± 0.34 | 1.80 ± 0.46 | <0.001 |
| Fatty Acid | SBM | CPS | p-Value |
|---|---|---|---|
| Miristic (14:0) | 0.33 ± 0.03 | 0.41 ± 0.16 | 0.175 |
| Palmitic (16:0) | 22.97 ± 0.25 | 20.37 ± 0.47 | <0.001 |
| Heptadecanoic (17:0) | 0.11 ± 0.04 | 0.10 ± 0.03 | 0.611 |
| Estearic (18:0) | 4.86 ± 0.20 | 4.90 ± 0.50 | 0.835 |
| Palmitoleic (16:1) | 1.99 ± 0.17 | 1.72 ± 0.06 | <0.001 |
| Oleic (18:1) | 35.99 ± 2.59 | 42.81 ± 1.35 | <0.001 |
| Linoleic (18:2) | 20.82 ± 3.42 | 21.56 ± 4.01 | 0.803 |
| Linolenic (18:3) | 1.06 ± 0.04 | 1.38 ± 0.17 | <0.001 |
| Saturated (SFA) | 28.27 ± 1.50 | 25.78 ± 1.21 | 0.003 |
| Monounsaturated (MUFA) | 37.98 ± 2.37 | 44.54 ± 1.72 | <0.001 |
| Polyunsaturated (PUFA) | 21.88 ± 2.65 | 22.94 ± 3.11 | 0.475 |
| SFA: MUFA:PUFA | 1:1.3:0.7 | 1:1.7:0.9 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paszkiewicz, W.; Muszyński, S.; Kwiecień, M.; Zhyla, M.; Świątkiewicz, S.; Arczewska-Włosek, A.; Tomaszewska, E. Effect of Soybean Meal Substitution by Raw Chickpea Seeds on Thermal Properties and Fatty Acid Composition of Subcutaneous Fat Tissue of Broiler Chickens. Animals 2020, 10, 533. https://doi.org/10.3390/ani10030533
Paszkiewicz W, Muszyński S, Kwiecień M, Zhyla M, Świątkiewicz S, Arczewska-Włosek A, Tomaszewska E. Effect of Soybean Meal Substitution by Raw Chickpea Seeds on Thermal Properties and Fatty Acid Composition of Subcutaneous Fat Tissue of Broiler Chickens. Animals. 2020; 10(3):533. https://doi.org/10.3390/ani10030533
Chicago/Turabian StylePaszkiewicz, Waldemar, Siemowit Muszyński, Małgorzata Kwiecień, Mykola Zhyla, Sylwester Świątkiewicz, Anna Arczewska-Włosek, and Ewa Tomaszewska. 2020. "Effect of Soybean Meal Substitution by Raw Chickpea Seeds on Thermal Properties and Fatty Acid Composition of Subcutaneous Fat Tissue of Broiler Chickens" Animals 10, no. 3: 533. https://doi.org/10.3390/ani10030533
APA StylePaszkiewicz, W., Muszyński, S., Kwiecień, M., Zhyla, M., Świątkiewicz, S., Arczewska-Włosek, A., & Tomaszewska, E. (2020). Effect of Soybean Meal Substitution by Raw Chickpea Seeds on Thermal Properties and Fatty Acid Composition of Subcutaneous Fat Tissue of Broiler Chickens. Animals, 10(3), 533. https://doi.org/10.3390/ani10030533





