Antibiotic Treatment in Anopheles coluzzii Affects Carbon and Nitrogen Metabolism
Abstract
1. Introduction
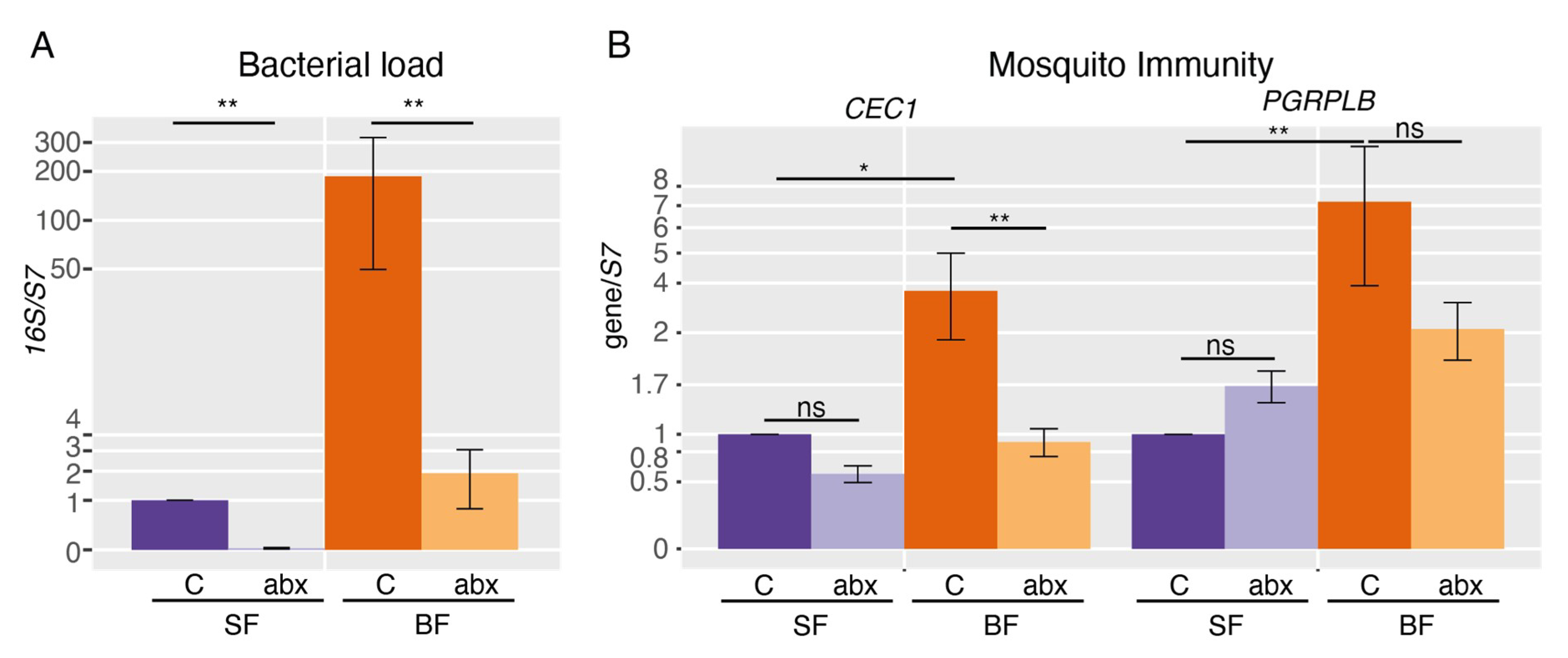
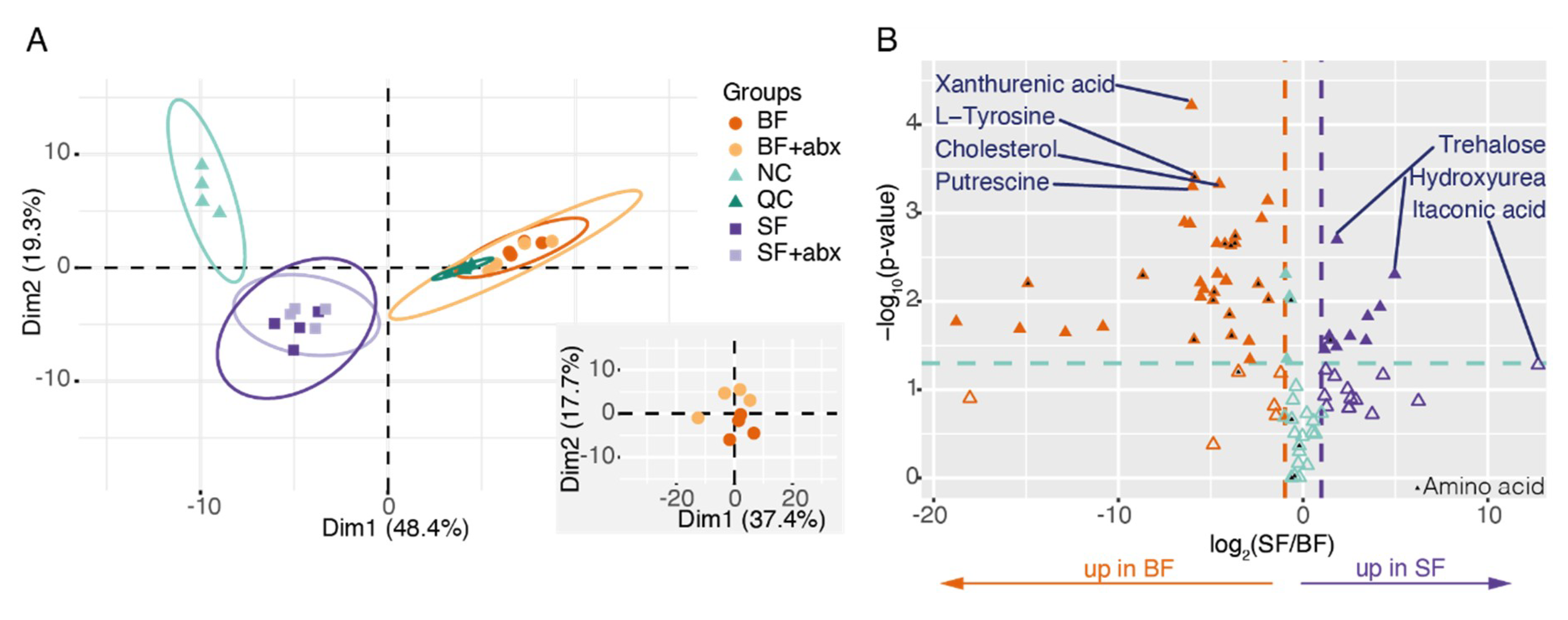
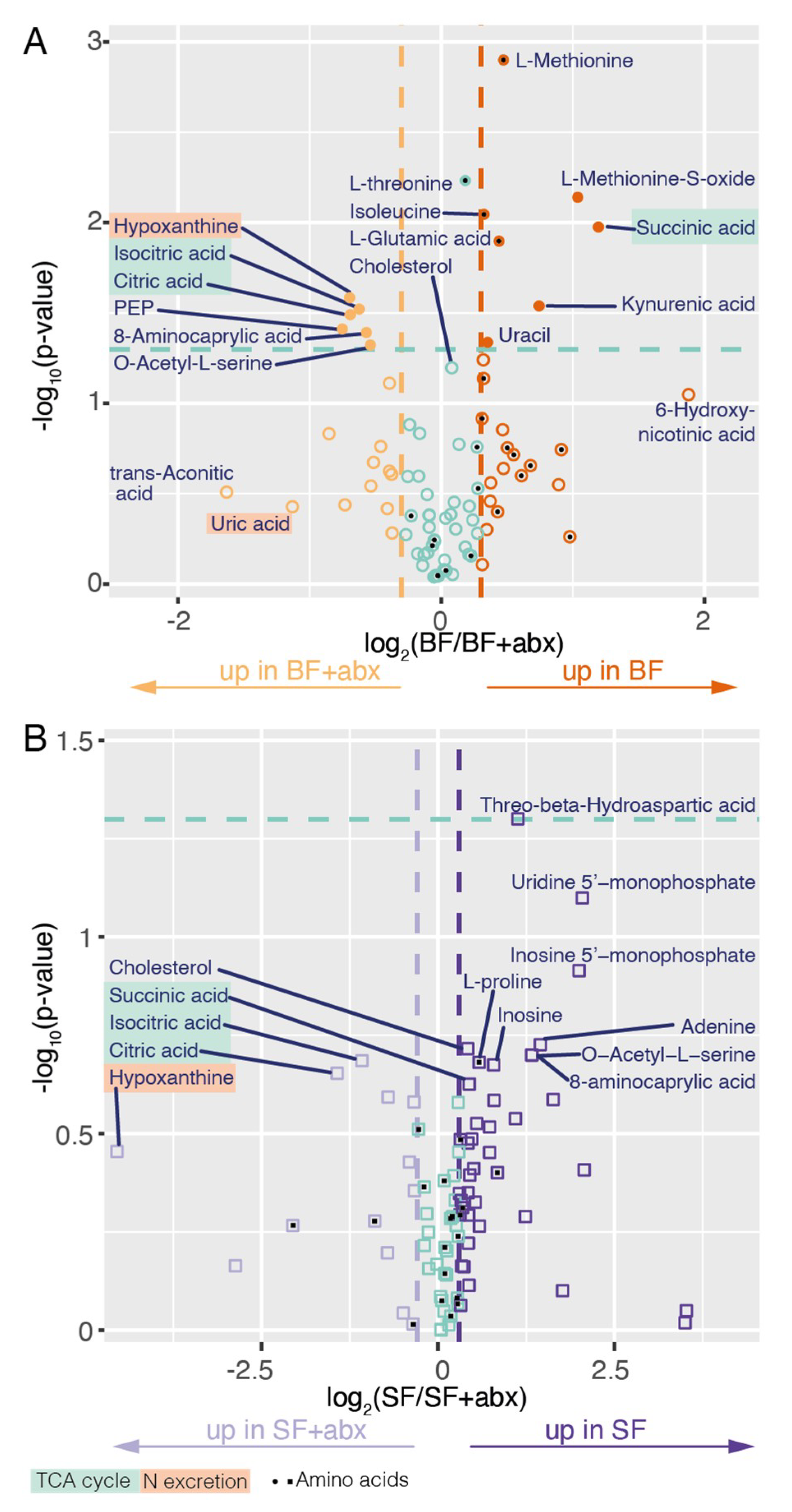
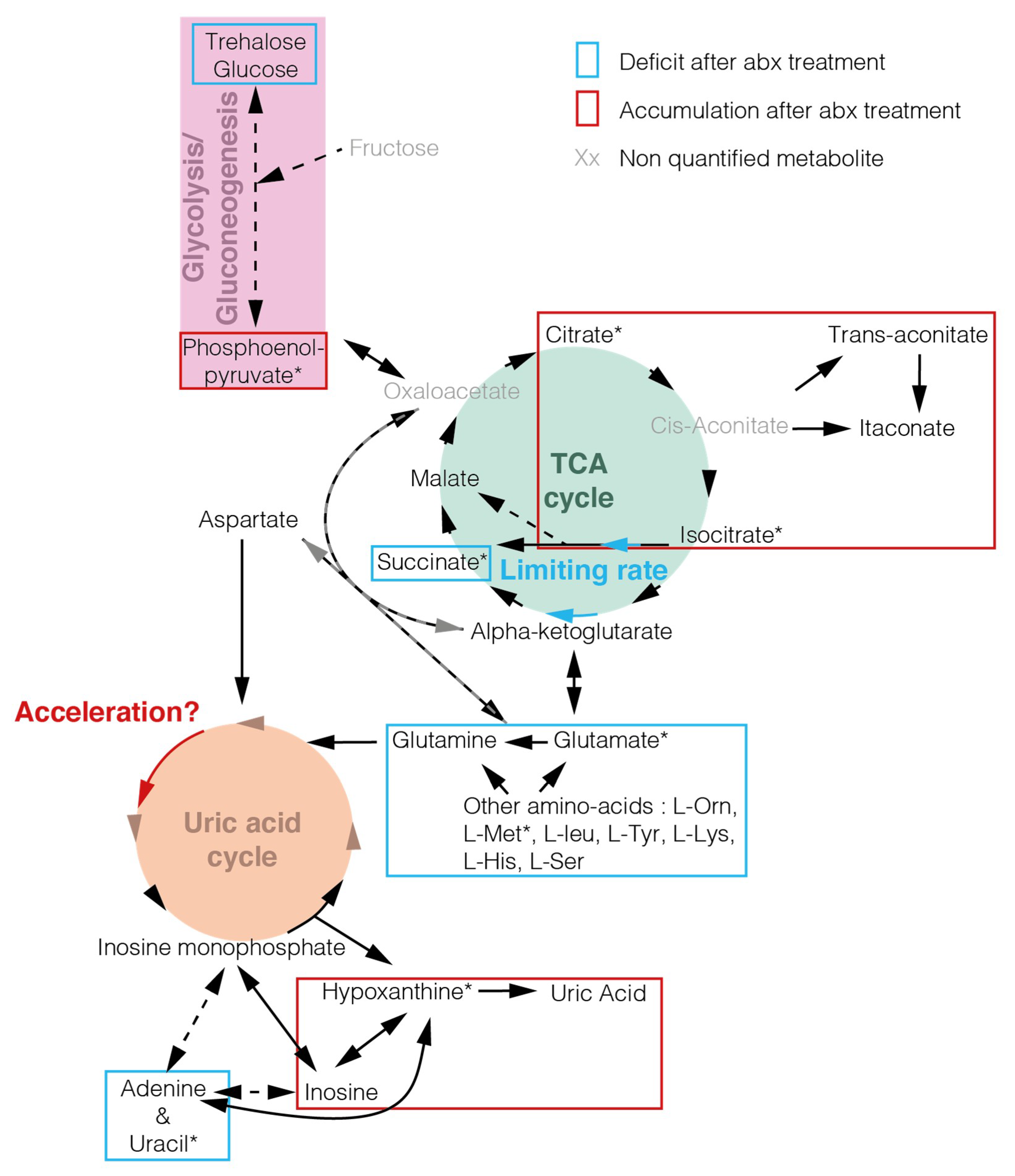
2. Results
3. Discussion
4. Materials and Methods
4.1. Mosquito Rearing
4.2. Blood Feeding
4.3. Antibiotic Treatment
4.4. Midgut Dissection
4.5. RNA Extraction and RT-qPCR
4.6. Metabolomics
4.6.1. Methanol Quenching
4.6.2. Dual Phase Extraction and Sample Preparation
4.6.3. Data Acquisition
4.7. Data Analysis
4.7.1. RT-qPCR Analysis
4.7.2. Metabolomic Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mancini, M.V.; Damiani, C.; Accoti, A.; Tallarita, M.; Nunzi, E.; Cappelli, A.; Bozic, J.; Catanzani, R.; Rossi, P.; Valzano, M.; et al. Estimating bacteria diversity in different organs of nine species of mosquito by next generation sequencing. BMC Microbiol. 2018, 18, 126. [Google Scholar] [CrossRef] [PubMed]
- Osei-Poku, J.; Mbogo, C.M.; Palmer, W.J.; Jiggins, F.M. Deep sequencing reveals extensive variation in the gut microbiota of wild mosquitoes from Kenya. Mol. Ecol. 2012, 21, 5138–5150. [Google Scholar] [CrossRef] [PubMed]
- Straif, S.C.; Mbogo, C.N.M.; Toure, A.M.; Walker, E.D.; Kaufman, M.; Toure, Y.T.; Beier, J.C. Midgut Bacteria in Anopheles gambiae and An. funestus (Diptera: Culicidae) from Kenya and Mali. J. Med. Entomol. 1998, 35, 222–226. [Google Scholar] [CrossRef] [PubMed]
- Boissière, A.; Tchioffo, M.T.; Bachar, D.; Abate, L.; Marie, A.; Nsango, S.E.; Shahbazkia, H.R.; Awono-Ambene, P.H.; Levashina, E.A.; Christen, R.; et al. Midgut Microbiota of the Malaria Mosquito Vector Anopheles gambiae and Interactions with Plasmodium falciparum Infection. PLoS Pathog. 2012, 8, e1002742. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Manfredini, F.; Dimopoulos, G. Implication of the Mosquito Midgut Microbiota in the Defense against Malaria Parasites. PLoS Pathog. 2009, 5, e1000423. [Google Scholar] [CrossRef]
- Akorli, J.; Gendrin, M.; Pels, N.A.P.; Yeboah-Manu, D.; Christophides, G.K.; Wilson, M.D. Seasonality and Locality Affect the Diversity of Anopheles gambiae and Anopheles coluzzii Midgut Microbiota from Ghana. PLoS ONE 2016, 11, e0157529. [Google Scholar] [CrossRef]
- Bascuñán, P.; Niño-Garcia, J.P.; Galeano-Castañeda, Y.; Serre, D.; Correa, M.M. Factors shaping the gut bacterial community assembly in two main Colombian malaria vectors. Microbiome 2018, 6, 148. [Google Scholar] [CrossRef]
- Krajacich, B.J.; Huestis, D.L.; Dao, A.; Yaro, A.S.; Diallo, M.; Krishna, A.; Xu, J.; Lehmann, T. Investigation of the seasonal microbiome of Anopheles coluzzii mosquitoes in Mali. PLoS ONE 2018, 13, e0194899. [Google Scholar] [CrossRef]
- Gimonneau, G.; Tchioffo, M.T.; Abate, L.; Boissière, A.; Awono-Ambéné, P.H.; Nsango, S.E.; Christen, R.; Morlais, I. Composition of Anopheles coluzzii and Anopheles gambiae microbiota from larval to adult stages. Infect. Genet. Evol. 2014, 28, 715–724. [Google Scholar] [CrossRef]
- Pumpuni, C.B.; Demaio, J.; Kent, M.; Davis, J.R.; Beier, J.C. Bacterial Population Dynamics in Three Anopheline Species: The Impact on Plasmodium Sporogonic Development. Am. J. Trop. Med. Hyg. 1996, 54, 214–218. [Google Scholar] [CrossRef]
- Wang, Y.; Gilbreath, T.M., III; Kukutla, P.; Yan, G.; Xu, J. Dynamic Gut Microbiome across Life History of the Malaria Mosquito Anopheles gambiae in Kenya. PLoS ONE 2011, 6, e24767. [Google Scholar] [CrossRef] [PubMed]
- Coon, K.L.; Vogel, K.J.; Brown, M.R.; Strand, M.R. Mosquitoes rely on their gut microbiota for development. Mol. Ecol. 2014, 23, 2727–2739. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Dhayal, D.; Singh, O.P.; Adak, T.; Bhatnagar, R.K. Gut microbes influence fitness and malaria transmission potential of Asian malaria vector Anopheles stephensi. Acta Trop. 2013, 128, 41–47. [Google Scholar] [CrossRef]
- Gendrin, M.; Rodgers, F.H.; Yerbanga, R.S.; Ouédraogo, J.B.; Basáñez, M.-G.; Cohuet, A.; Christophides, G.K. Antibiotics in ingested human blood affect the mosquito microbiota and capacity to transmit malaria. Nat. Commun. 2015, 6, 5921. [Google Scholar] [CrossRef]
- Dada, N.; Sheth, M.; Liebman, K.; Pinto, J.; Lenhart, A. Whole metagenome sequencing reveals links between mosquito microbiota and insecticide resistance in malaria vectors. Sci. Rep. 2018, 8, 2084. [Google Scholar] [CrossRef] [PubMed]
- Graumans, W.; Jacobs, E.; Bousema, T.; Sinnis, P. When Is a Plasmodium-Infected Mosquito an Infectious Mosquito? Trends Parasitol. 2020, 36, 705–716. [Google Scholar] [CrossRef] [PubMed]
- Cirimotich, C.M.; Dong, Y.; Clayton, A.M.; Sandiford, S.L.; Souza-Neto, J.A.; Mulenga, M.; Dimopoulos, G. Natural Microbe-Mediated Refractoriness to Plasmodium Infection in Anopheles gambiae. Science 2011, 332, 855. [Google Scholar] [CrossRef]
- Gendrin, M.; Turlure, F.; Rodgers, F.H.; Cohuet, A.; Morlais, I.; Christophides, G.K. The Peptidoglycan Recognition Proteins PGRPLA and PGRPLB Regulate Anopheles Immunity to Bacteria and Affect Infection by Plasmodium. J. Innate Immun. 2017, 9, 333–342. [Google Scholar] [CrossRef]
- Meister, S.; Agianian, B.; Turlure, F.; Relógio, A.; Morlais, I.; Kafatos, F.C.; Christophides, G.K. Anopheles gambiae PGRPLC-Mediated Defense against Bacteria Modulates Infections with Malaria Parasites. PLoS Pathog. 2009, 5, e1000542. [Google Scholar] [CrossRef]
- Stecher, B.; Hardt, W.-D. Mechanisms controlling pathogen colonization of the gut. Curr. Opin. Microbiol. 2011, 14, 82–91. [Google Scholar] [CrossRef]
- Rodgers, F.H.; Gendrin, M.; Wyer, C.A.S.; Christophides, G.K. Microbiota-induced peritrophic matrix regulates midgut homeostasis and prevents systemic infection of malaria vector mosquitoes. PLoS Pathog. 2017, 13, e1006391. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, S.; Behrends, V.; Bundy, J.G.; Crisanti, A.; Nolan, T. Phenylalanine Metabolism Regulates Reproduction and Parasite Melanization in the Malaria Mosquito. PLoS ONE 2014, 9, e84865. [Google Scholar] [CrossRef] [PubMed]
- Champion, C.J.; Kukutla, P.; Glennon, E.K.K.; Wang, B.; Luckhart, S.; Xu, J. Anopheles gambiae: Metabolomic Profiles in Sugar-Fed, Blood-Fed, and Plasmodium falciparum-Infected Midgut. Dataset Pap. Sci. 2017, 2017. [Google Scholar] [CrossRef]
- Benzylpenicillin (DrugBank). Available online: https://www.drugbank.ca/drugs/DB01053 (accessed on 14 July 2020).
- Streptomycin (DrugBank). Available online: https://www.drugbank.ca/drugs/DB01082 (accessed on 14 July 2020).
- Gentamicin (DrugBank). Available online: https://www.drugbank.ca/drugs/DB00798 (accessed on 14 July 2020).
- Amphotericin B (DrugBank). Available online: https://www.drugbank.ca/drugs/DB00681 (accessed on 14 July 2020).
- Nephrotoxic agents (DrugBank). Available online: https://www.drugbank.ca/categories/DBCAT003959 (accessed on 14 July 2020).
- Montali, R.J.; Bush, M.; Smeller, J.M. The Pathology of Nephrotoxicity of Gentamicin in Snakes. Vet. Pathol 1979, 16, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Balakumar, P.; WitnessKoe, W.E.; Gan, Y.S.; JemayPuah, S.M.; Kuganesswari, S.; Prajapati, S.K.; Varatharajan, R.; Jayachristy, S.A.; Sundram, K.; Bahari, M.B. Effects of pre and post-treatments with dipyridamole in gentamicin-induced acute nephrotoxicity in the rat. Regul. Toxicol. Pharmacol. 2017, 84, 35–44. [Google Scholar] [CrossRef]
- Liberopoulos, E.N.; Kei, A.A.; Elisaf, M.S. Lysis syndrome during therapy of visceral leishmaniasis. Infection 2012, 40, 121–123. [Google Scholar] [CrossRef]
- Rodgers, F.H.; Gendrin, M.; Wyer, C.A.S.; Christophides, G.K. RNA-Seq Experiment Antibiotic Treatment of Midgut Microbiota during Blood Feeding (VectorBase). 2017. Available online: https://www.vectorbase.org/expression-browser/experiment/SRP106793 (accessed on 14 July 2020).
- Kondrashov, F.A.; Koonin, E.V.; Morgunov, I.G.; Finogenova, T.V.; Kondrashova, M.N. Evolution of glyoxylate cycle enzymes in Metazoa: Evidence of multiple horizontal transfer events and pseudogene formation. Biol. Direct 2006, 1, 31. [Google Scholar] [CrossRef]
- Lavazec, C.; Boudin, C.; Lacroix, R.; Bonnet, S.; Diop, A.; Thiberge, S.; Boisson, B.; Tahar, R.; Bourgouin, C. Carboxypeptidases B of Anopheles gambiae as Targets for a Plasmodium falciparum Transmission-Blocking Vaccine. Infect. Immun. 2007, 75, 1635. [Google Scholar] [CrossRef]
- de O Gaio, A.; Gusmão, D.S.; Santos, A.V.; Berbert-Molina, M.A.; Pimenta, P.F.; Lemos, F.J. Contribution of midgut bacteria to blood digestion and egg production in Aedes aegypti (diptera: Culicidae) (L.). Parasites Vectors 2011, 4, 105. [Google Scholar] [CrossRef]
- Hughes, G.L.; Dodson, B.L.; Johnson, R.M.; Murdock, C.C.; Tsujimoto, H.; Suzuki, Y.; Patt, A.A.; Cui, L.; Nossa, C.W.; Barry, R.M.; et al. Native microbiome impedes vertical transmission of Wolbachia in Anopheles mosquitoes. Proc. Natl. Acad. Sci. USA 2014, 111, 12498. [Google Scholar] [CrossRef]
- Consuegra, J.; Grenier, T.; Baa-Puyoulet, P.; Rahioui, I.; Akherraz, H.; Gervais, H.; Parisot, N.; da Silva, P.; Charles, H.; Calevro, F.; et al. Drosophila-associated bacteria differentially shape the nutritional requirements of their host during juvenile growth. PLoS Biol. 2020, 18, e3000681. [Google Scholar] [CrossRef] [PubMed]
- Cirimotich, C.M.; Ramirez, J.L.; Dimopoulos, G. Native Microbiota Shape Insect Vector Competence for Human Pathogens. Cell Host Microbe 2011, 10, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Beier, M.S.; Pumpuni, C.B.; Beier, J.C.; Davis, J.R. Effects of Para-Aminobenzoic Acid, Insulin, and Gentamicin on Plasmodium falciparum Development in Anopheline Mosquitoes (Diptera: Culicidae). J. Med. Entomol. 1994, 31, 561–565. [Google Scholar] [CrossRef] [PubMed]
- Mancio-Silva, L.; Slavic, K.; Grilo Ruivo, M.T.; Grosso, A.R.; Modrzynska, K.K.; Vera, I.M.; Sales-Dias, J.; Gomes, A.R.; MacPherson, C.R.; Crozet, P.; et al. Nutrient sensing modulates malaria parasite virulence. Nature 2017, 547, 213–216. [Google Scholar] [CrossRef]
- Wang, M.; An, Y.; Dong, S.; Feng, Y.; Gao, L.; Wang, P.; Dimopoulus, G.; Tang, H.; Wang, J. Glucose-mediated expansion of a gut commensal bacterium promotes Plasmodium infection through alkalizing mosquito midgut. bioRxiv 2020. [Google Scholar] [CrossRef]
- Hayward, R.E. Plasmodium falciparum phosphoenolpyruvate carboxykinase is developmentally regulated in gametocytes. Mol. Biochem. Parasitol. 2000, 107, 227–240. [Google Scholar] [CrossRef]
- Downie, M.J.; Kirk, K.; Mamoun, C.B. Purine Salvage Pathways in the Intraerythrocytic Malaria Parasite Plasmodium falciparum. Eukaryot Cell 2008, 7, 1231. [Google Scholar] [CrossRef]
- El Bissati, K.; Zufferey, R.; Witola, W.H.; Carter, N.S.; Ullman, B.; Ben Mamoun, C. The plasma membrane permease PfNT1 is essential for purine salvage in the human malaria parasite Plasmodium falciparum. Proc. Natl. Acad. Sci. USA 2006, 103, 9286. [Google Scholar] [CrossRef]
- Sinden, R.; Talman, A.; Marques, S.; Wass, M.; Sternberg, M. The flagellum in malarial parasites. Curr. Opin. Microbiol. 2010, 13, 491–500. [Google Scholar] [CrossRef]
- Lee, K.-A.; Kim, S.-H.; Kim, E.-K.; Ha, E.-M.; You, H.; Kim, B.; Kim, M.-J.; Kwon, Y.; Ryu, J.-H.; Lee, W.-J. Bacterial-Derived Uracil as a Modulator of Mucosal Immunity and Gut-Microbe Homeostasis in Drosophila. Cell 2013, 153, 797–811. [Google Scholar] [CrossRef]
- Kind, T.; Wohlgemuth, G.; Lee, D.Y.; Lu, Y.; Palazoglu, M.; Shahbaz, S.; Fiehn, O. FiehnLib: Mass Spectral and Retention Index Libraries for Metabolomics Based on Quadrupole and Time-of-Flight Gas Chromatography/Mass Spectrometry. Anal. Chem. 2009, 81, 10038–10048. [Google Scholar] [CrossRef] [PubMed]
- Behrends, V.; Tredwell, G.D.; Bundy, J.G. A software complement to AMDIS for processing GC-MS metabolomic data. Anal. Biochem. 2011, 415, 206–208. [Google Scholar] [CrossRef] [PubMed]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chabanol, E.; Behrends, V.; Prévot, G.; Christophides, G.K.; Gendrin, M. Antibiotic Treatment in Anopheles coluzzii Affects Carbon and Nitrogen Metabolism. Pathogens 2020, 9, 679. https://doi.org/10.3390/pathogens9090679
Chabanol E, Behrends V, Prévot G, Christophides GK, Gendrin M. Antibiotic Treatment in Anopheles coluzzii Affects Carbon and Nitrogen Metabolism. Pathogens. 2020; 9(9):679. https://doi.org/10.3390/pathogens9090679
Chicago/Turabian StyleChabanol, Estelle, Volker Behrends, Ghislaine Prévot, George K. Christophides, and Mathilde Gendrin. 2020. "Antibiotic Treatment in Anopheles coluzzii Affects Carbon and Nitrogen Metabolism" Pathogens 9, no. 9: 679. https://doi.org/10.3390/pathogens9090679
APA StyleChabanol, E., Behrends, V., Prévot, G., Christophides, G. K., & Gendrin, M. (2020). Antibiotic Treatment in Anopheles coluzzii Affects Carbon and Nitrogen Metabolism. Pathogens, 9(9), 679. https://doi.org/10.3390/pathogens9090679





