A Systematic Review of Studies Published between 2016 and 2019 on the Effectiveness and Efficacy of Pneumococcal Vaccination on Pneumonia and Invasive Pneumococcal Disease in an Elderly Population
Abstract
1. Introduction
2. Materials and Methods
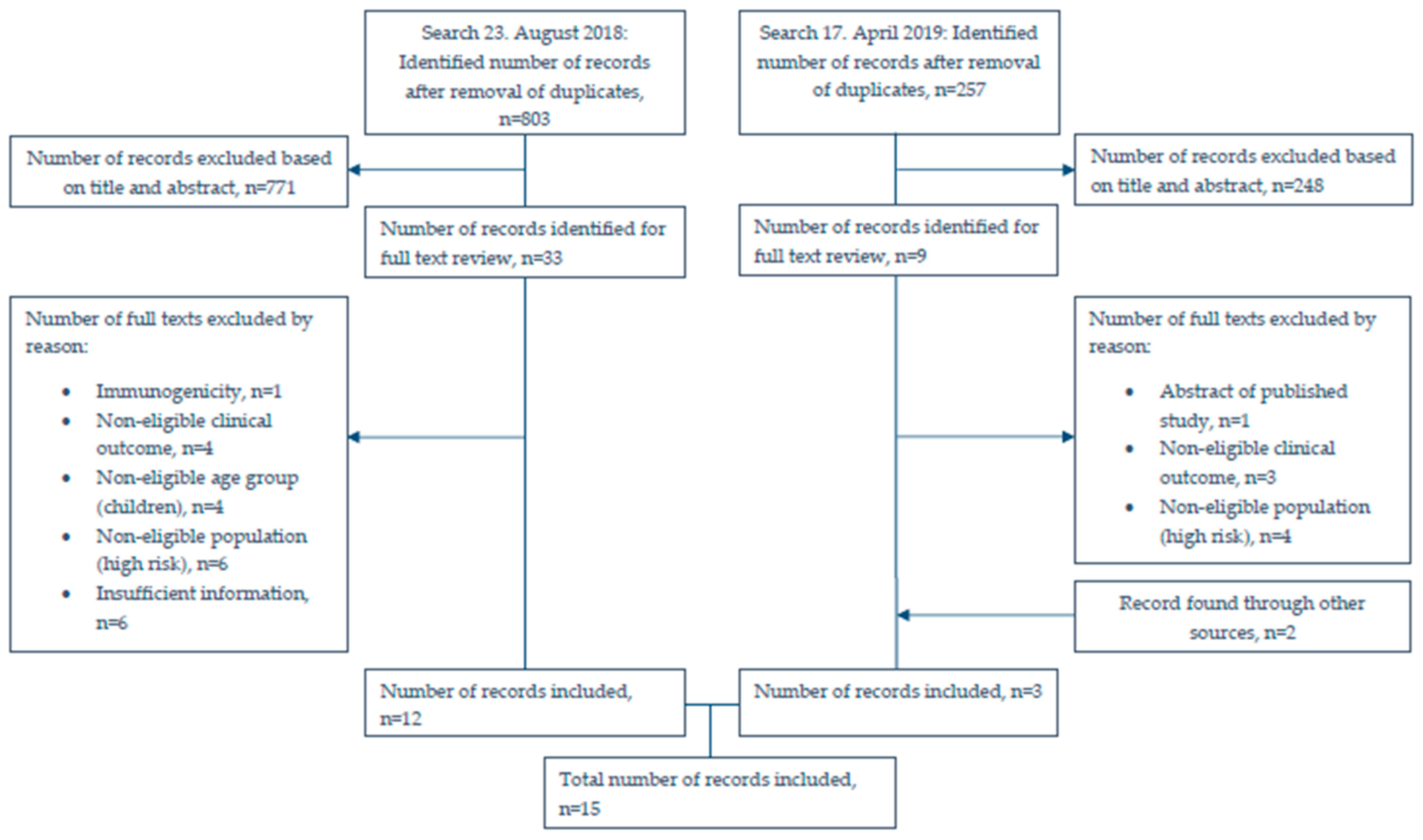
2.1. Literature Search
2.2. Data Extraction
2.3. Quality Assessment
2.4. Analysis
3. Results
3.1. Characteristics
3.1.1. Conjugate Vaccine
3.1.2. Polysaccharide Vaccine
3.2. Reported Outcomes
3.2.1. Conjugate Vaccine
3.2.2. Polysaccharide Vaccine
3.3. Risk of Bias
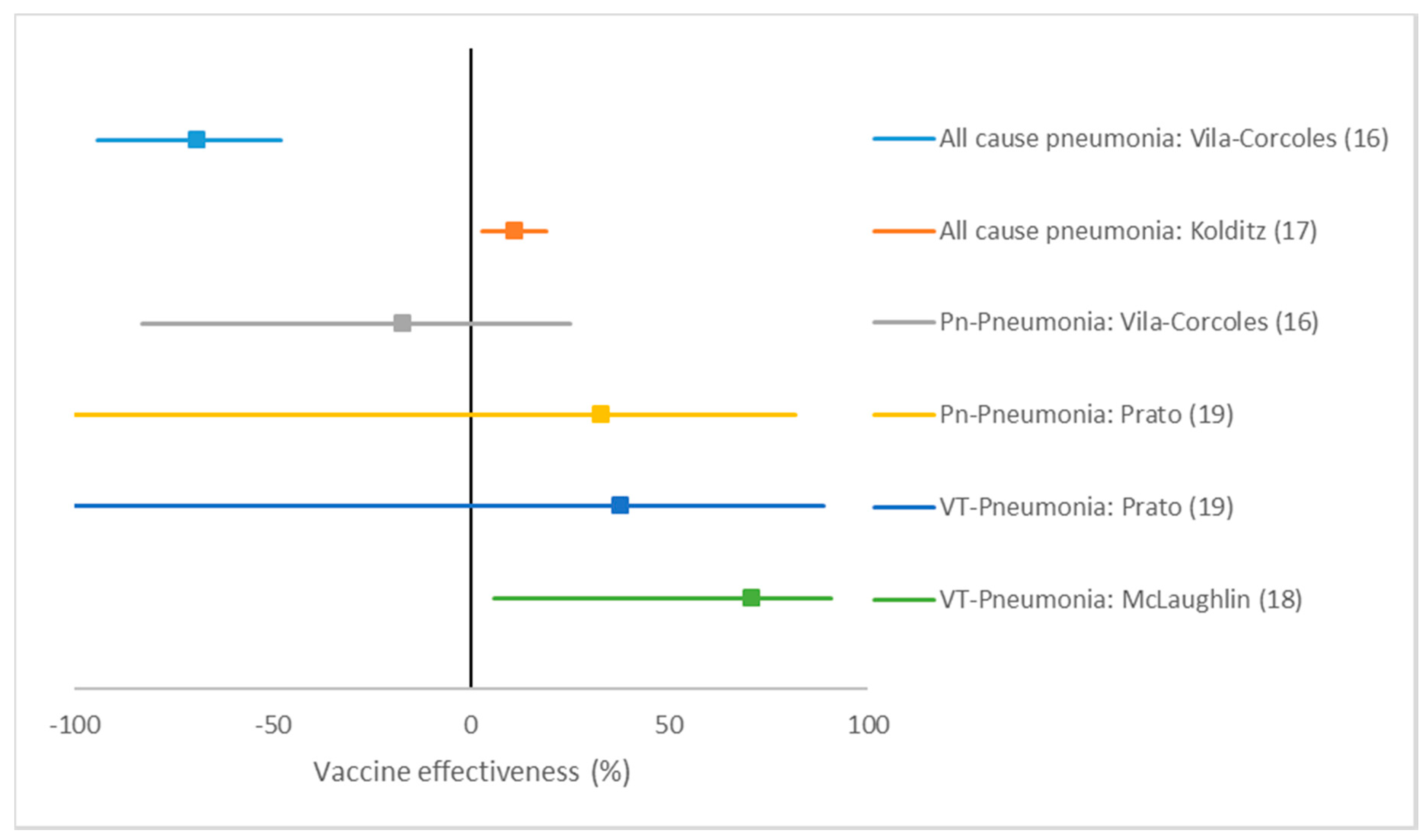
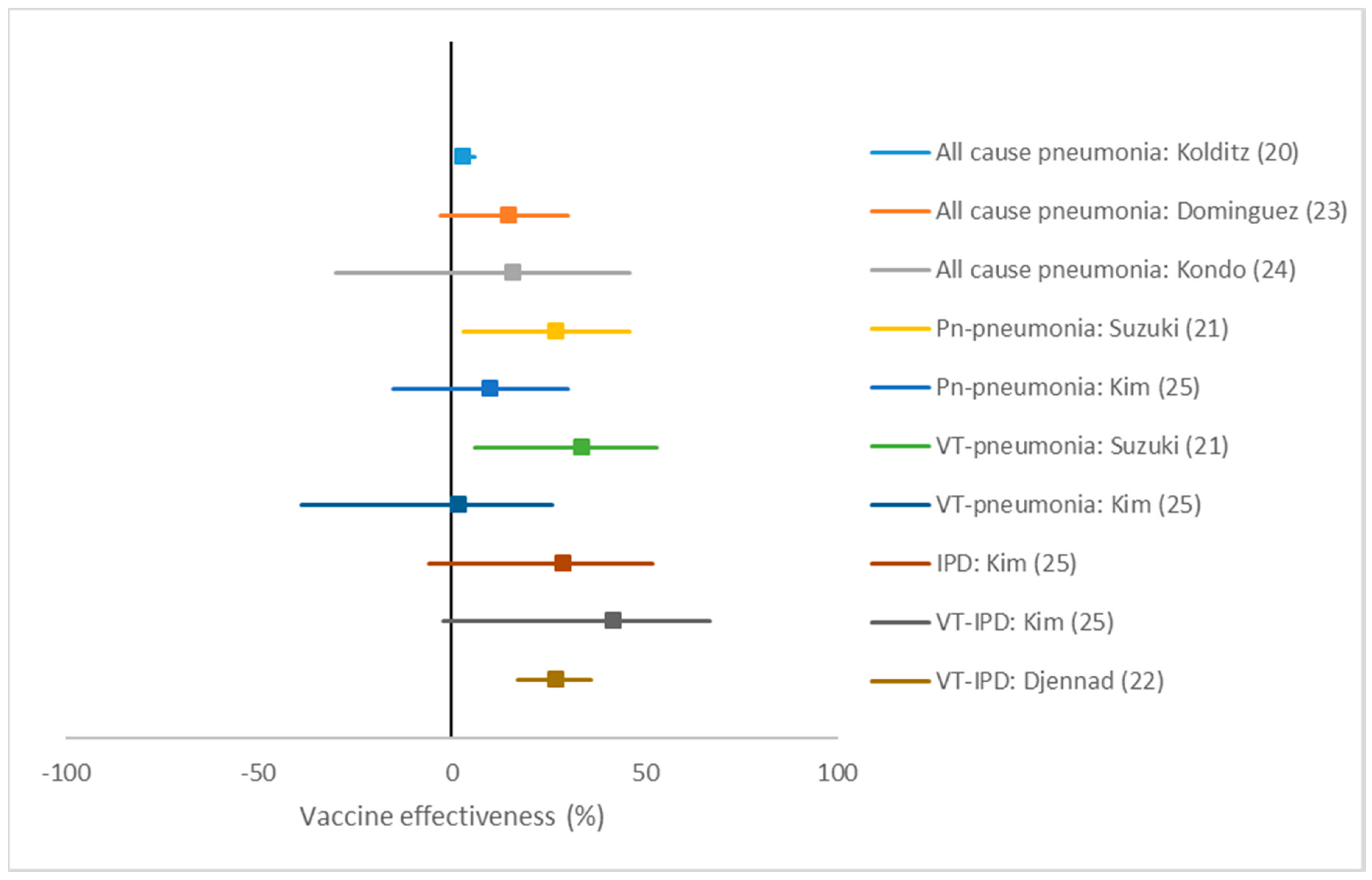
3.4. Vaccine efficacy/Effectiveness
3.4.1. Conjugate Vaccine
3.4.2. Polysaccharide Vaccine
3.5. Age
4. Discussion
4.1. General Comments
4.2. Evidence Base for PCV13
4.3. Evidence Base for PPV23
4.4. Future Perspectives
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Wroe, P.C.; Finkelstein, J.A.; Ray, G.T.; Linder, J.A.; Johnson, K.M.; Rifas-Shiman, S.; Moore, M.R.; Huang, S.S. Aging population and future burden of pneumococcal pneumonia in the United States. J. Infect. Dis. 2012, 205, 1589–1592. [Google Scholar] [CrossRef] [PubMed]
- Bonten, M.J.; Huijts, S.M.; Bolkenbaas, M.; Webber, C.; Patterson, S.; Gault, S.; van Werkhoven, C.H.; van Deursen, A.M.; Sanders, E.A.; Verheij, T.J.; et al. Polysaccharide conjugate vaccine against pneumococcal pneumonia in adults. N. Engl. J. Med. 2015, 372, 1114–1125. [Google Scholar] [CrossRef] [PubMed]
- Diao, W.Q.; Shen, N.; Yu, P.X.; Liu, B.B.; He, B. Efficacy of 23-valent pneumococcal polysaccharide vaccine in preventing community-acquired pneumonia among immunocompetent adults: A systematic review and meta-analysis of randomized trials. Vaccine 2016, 34, 1496–1503. [Google Scholar] [CrossRef] [PubMed]
- Falkenhorst, G.; Remschmidt, C.; Harder, T.; Hummers-Pradier, E.; Wichmann, O.; Bogdan, C. Effectiveness of the 23-Valent Pneumococcal Polysaccharide Vaccine (PPV23) against Pneumococcal Disease in the Elderly: Systematic Review and Meta-Analysis. PLoS ONE 2017, 12, e0169368. [Google Scholar] [CrossRef] [PubMed]
- Kraicer-Melamed, H.; O’Donnell, S.; Quach, C. The effectiveness of pneumococcal polysaccharide vaccine 23 (PPV23) in the general population of 50 years of age and older: A systematic review and meta-analysis. Vaccine 2016, 34, 1540–1550, Corrigendum in 2016, 34, 4083–4084, doi:10.1016/j.vaccine.2016.06.045. [Google Scholar] [CrossRef] [PubMed]
- Schiffner-Rohe, J.; Witt, A.; Hemmerling, J.; von Eiff, C.; Leverkus, F.W. Efficacy of PPV23 in Preventing Pneumococcal Pneumonia in Adults at Increased Risk—A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0146338. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tin Tin Htar, M.; Stuurman, A.L.; Ferreira, G.; Alicino, C.; Bollaerts, K.; Paganino, C.; Reinert, R.R.; Schmitt, H.J.; Trucchi, C.; Vestraeten, T.; et al. Effectiveness of pneumococcal vaccines in preventing pneumonia in adults, a systematic review and meta-analyses of observational studies. PLoS ONE 2017, 12, e0177985. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savović, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; Higgins, J.P.; Sterne, J.; Tugwell, P.; Reeves, B.C. Checklists of methodological issues for review authors to consider when including non-randomized studies in systematic reviews. Res. Synth. Methods 2013, 4, 63–77. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 11 November 2019).
- Gessner, B.D.; Jiang, Q.; Van Werkhoven, C.H.; Sings, H.L.; Webber, C.; Scott, D.; Neuzil, K.M.; O’Brien, K.L.; Wunderink, R.G.; Grobbee, D.E.; et al. A public health evaluation of 13-valent pneumococcal conjugate vaccine impact on adult disease outcomes from a randomized clinical trial in the Netherlands. Vaccine 2018. [Google Scholar] [CrossRef]
- Huijts, S.M.; van Werkhoven, C.H.; Bolkenbaas, M.; Grobbee, D.E.; Bonten, M.J. Post-hoc analysis of a randomized controlled trial: Diabetes mellitus modifies the efficacy of the 13-valent pneumococcal conjugate vaccine in elderly. Vaccine 2017, 35, 4444–4449. [Google Scholar] [CrossRef] [PubMed]
- Suaya, J.A.; Jiang, Q.; Scott, D.A.; Gruber, W.C.; Webber, C.; Schmoele-Thoma, B.; Hall-Murray, C.K.; Jodar, L.; Isturiz, R.E. Post hoc analysis of the efficacy of the 13-valent pneumococcal conjugate vaccine against vaccine-type community-acquired pneumonia in at-risk older adults. Vaccine 2018, 36, 1477–1483. [Google Scholar] [CrossRef] [PubMed]
- Webber, C.; Patton, M.; Patterson, S.; Schmoele-Thoma, B.; Huijts, S.M.; Bonten, M.J.; CAPiTA Study Group. Exploratory efficacy endpoints in the Community-Acquired Pneumonia Immunization Trial in Adults (CAPiTA). Vaccine 2017, 35, 1266–1272. [Google Scholar] [CrossRef] [PubMed]
- Patterson, S.; Webber, C.; Patton, M.; Drews, W.; Huijts, S.M.; Bolkenbaas, M.; Gruber, W.C.; Scott, D.A.; Bonten, M.J.M. A post hoc assessment of duration of protection in CAPiTA (Community Acquired Pneumonia immunization Trial in Adults). Trials Vaccinol. 2016, 5, 92–96. [Google Scholar] [CrossRef]
- Vila-Corcoles, A.; Ochoa-Gondar, O.; de Diego, C.; Satue, E.; Aragón, M.; Vila-Rovira, A.; Gomez-Bertomeu, F.; Magarolas, R.; Figuerola-Massana, E.; Raga, X.; et al. Evaluating clinical effectiveness of 13-valent pneumococcal conjugate vaccination against pneumonia among middle-aged and older adults in Catalonia: Results from the EPIVAC cohort study. BMC Infect. Dis. 2018, 18, 196. [Google Scholar] [CrossRef] [PubMed]
- Kolditz, M.; Schmitt, J.; Pletz, M.W.; Tesch, F. Impact of the 13-valent pneumococcal conjugate vaccine on incidence of all-cause pneumonia in adults aged >= 60 years-a population-based retrospective cohort study. Clin. Infect. Dis. 2018, 21, 21. [Google Scholar] [CrossRef]
- McLaughlin, J.M.; Jiang, Q.; Isturiz, R.E.; Sings, H.L.; Swerdlow, D.L.; Gessner, B.D.; Carrico, R.M.; Peyrani, P.; Wiemken, T.L.; Mattingly, W.A.; et al. Effectiveness of 13-Valent Pneumococcal Conjugate Vaccine Against Hospitalization for Community-Acquired Pneumonia in Older US Adults: A Test-Negative Design. Clin. Infect. Dis. 2018. [Google Scholar] [CrossRef]
- Prato, R.; Fortunato, F.; Cappelli, M.G.; Chironna, M.; Martinelli, D. Effectiveness of the 13-valent pneumococcal conjugate vaccine against adult pneumonia in Italy: A case-control study in a 2-year prospective cohort. BMJ Open 2018, 8, e019034. [Google Scholar] [CrossRef]
- Kolditz, M.; Schmitt, J.; Pletz, M.W.; Tesch, F. Impact of pneumococcal polysaccharide vaccine on incidence and mortality after pneumonia in adults aged >/=60 years-a population-based retrospective cohort study. Clin. Microbiol. Infect. 2018, 24, 500–504. [Google Scholar] [CrossRef]
- Suzuki, M.; Dhoubhadel, B.G.; Ishifuji, T.; Yasunami, M.; Yaegashi, M.; Asoh, N.; Ishida, M.; Hamaguchi, S.; Aoshima, M.; Ariyoshi, K.; et al. Adult Pneumonia Study, Group-Japan. Serotype-specific effectiveness of 23-valent pneumococcal polysaccharide vaccine against pneumococcal pneumonia in adults aged 65 years or older: A multicentre, prospective, test-negative design study. Lancet Infect. Dis. 2017, 17, 313–321. [Google Scholar] [CrossRef]
- Djennad, A.; Ramsay, M.E.; Pebody, R.; Fry, N.K.; Sheppard, C.; Ladhani, S.N.; Andrews, N.J. Effectiveness of 23-Valent Polysaccharide Pneumococcal Vaccine and Changes in Invasive Pneumococcal Disease Incidence from 2000 to 2017 in Those Aged 65 and Over in England and Wales. EClinicalMedicine 2018, 6, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Domínguez, À.; Soldevila, N.; Toledo, D.; Torner, N.; Force, L.; Pérez, M.J.; Martin, V.; Rodriguez-Rojas, L.; Astray, J.; Egurrola, M.; et al. Effectiveness of 23-valent pneumococcal polysaccharide vaccination in preventing community-acquired pneumonia hospitalization and severe outcomes in the elderly in Spain. PLoS ONE 2017, 12, e0171943. [Google Scholar] [CrossRef] [PubMed]
- Kondo, K.; Suzuki, K.; Washio, M.; Ohfuji, S.; Fukushima, W.; Maeda, A.; Hirota, Y. Effectiveness of 23-valent pneumococcal polysaccharide vaccine and seasonal influenza vaccine for pneumonia among the elderly-Selection of controls in a case-control study. Vaccine 2017, 35, 4806–4810. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Chun, B.C.; Song, J.Y.; Kim, H.Y.; Bae, I.-G.; Kim, D.-M.; Choi, Y.H.; Jun, Y.H.; Choi, W.S.; Kang, S.H.; et al. Direct effectiveness of pneumococcal polysaccharide vaccine against invasive pneumococcal disease and non-bacteremic pneumococcal pneumonia in elderly population in the era of pneumococcal conjugate vaccine: A case-control study. Vaccine 2019, 37, 2797–2804. [Google Scholar] [CrossRef] [PubMed]
- Said, M.A.; Johnson, H.L.; Nonyane, B.A.; Deloria-Knoll, M.; O’Brien, K.L.; Andreo, F.; Beovic, B.; Blanco, S.; Boersma, W.G.; Boulware, D.R.; et al. Estimating the burden of pneumococcal pneumonia among adults: A systematic review and meta-analysis of diagnostic techniques. PLoS ONE 2013, 8, e60273. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, W.; Hirota, Y. Basic principles of test-negative design in evaluating influenza vaccine effectiveness. Vaccine 2017, 35, 4796–4800. [Google Scholar] [CrossRef]
- D’Ancona, F.; Caporali, M.G.; Del Manso, M.; Giambi, C.; Camilli, R.; D’Ambrosio, F.; Del Grosso, M.; Iannazzo, S.; Rizzuto, E.; Pantosti, A. Invasive pneumococcal disease in children and adults in seven Italian regions after the introduction of the conjugate vaccine, 2008–2014. Epidemiol. Prev. 2015, 39, 134–138. [Google Scholar]
- Hanquet, G.; Krizova, P.; Valentiner-Branth, P.; Ladhani, S.N.; Nuorti, J.P.; Lepoutre, A.; Mereckiene, J.; Knol, M.; Winje, B.A.; Ciruela, P.; et al. Effect of childhood pneumococcal conjugate vaccination on invasive disease in older adults of 10 European countries: Implications for adult vaccination. Thorax 2019, 74, 473–482. [Google Scholar] [CrossRef]
- Orenstein, E.W.; De Serres, G.; Haber, M.J.; Shay, D.K.; Bridges, C.B.; Gargiullo, P.; Orenstein, W.A. Methodologic issues regarding the use of three observational study designs to assess influenza vaccine effectiveness. Int. J. Epidemiol. 2007, 36, 623–631. [Google Scholar] [CrossRef]
| Author & Publication Year (Ref) | Type of Study | Country | Study Period | Time between Outcome and Vaccination | Outcome | Patient Group | Age (y) | Quality |
|---|---|---|---|---|---|---|---|---|
| PCV13 | ||||||||
| Gessner 2018 (11) | Post hoc of RCT | Netherlands | 2008-2013 | Up to 5 years | Pneumonia, pn-pneumonia, VT-pneumonia, IPD and VT-IPD | Hospitalized and out-patient | ≥65 | NA |
| Huijts 2017 (12) | ||||||||
| Suaya 2018 (13) | ||||||||
| Webber 2017 (14) | ||||||||
| Patterson 2016 (15) | ||||||||
| Vila-Corcoles 2018 (16) | Cohort | Spain | 2015 | Not stated | Pneumonia and pn-pneumonia | Hospitalized | ≥50 | High |
| Kolditz 2018 (17) | Cohort | Germany | 2014-2016 | Up to 5 years | Pneumonia | Hospitalized and out-patient | ≥60 | High |
| McLaughlin 2018 (18) | TND | US | 2015-2016 | Up to 5 years | VT-pneumonia | Hospitalized | ≥65 | High |
| Prato 2018 (19) | TND | Italy | 2013-2015 | Not stated | Pn-pneumonia and VT-pneumonia | Hospitalized and out-patient | ≥65 | Low |
| PPV23 | ||||||||
| Kolditz 2018 (20) | Cohort | Germany | 2010-2011 | Up to 5 years | Pneumonia | Hospitalized and out-patient | ≥60 | Low |
| Suzuki 2017 (21) | TND | Japan | 2011-2014 | Up to 5 years | Pn-pneumonia and VT-pneumonia | Hospitalized and out-patient | ≥65 | High |
| Djennad 2018 (22) | Indirect cohort | UK | 2000-2016 | Vaccine given at any time | VT-IPD | Hospitalized | ≥65 | High |
| Dominguez 2017 (23) | Case-control | Spain | 2013-2015 | Up to 5 years | Pneumonia | Hospitalized | ≥65 | Low |
| Kondo 2018 (24) | Case-control | Japan | 2009-2014 | Up to 5 years | Pneumonia | Outpatients | ≥65 | Low |
| Kim 2019 (25) | Case-control | South Korea | 2013-2015 | Up to 5 years | Pn-pneumonia a, VT-pneumonia a, IPD and VT-IPD | Hospitalized | ≥65 | High |
| Author (Ref) | Episodes or Cases Vaccinated | Individuals Vaccinated or Cases not Vaccinated | Episodes or Controls Vaccinated | Individuals not Vaccinated or Controls not Vaccinated | VE % Pneumonia | VE % Pn-Pneumonia | VE % VT-Pneumonia | VE % IPD | VE % VT-IPD |
|---|---|---|---|---|---|---|---|---|---|
| PCV 13 | |||||||||
| Vila-Corcoles (16) | 228 | 6912 (PY) | 12471 | 1983789 (PY) | −69 (−94 to −48) | ||||
| 20 | 6912 (PY) | 1628 | 1983789 (PY) | −17 (−83 to 25) | |||||
| Kolditz (17) | 532 | 11395 | 1812 | 34185 | 11 (3 to 19) | ||||
| McLaughlin (18) | 3 | 65 | 285 | 1681 | 71 (6 to 91) | ||||
| Prato (19) | 5 | 54 | 15 | 108 | 33 (−107 to 82) | ||||
| 3 | 36 | 17 | 126 | 38 (−132 to 89) | |||||
| PPV 23 | |||||||||
| Kolditz (20) | 7501 | 213431 | 23243 | 640293 | 3 (1 to 6) | ||||
| Suzuki (21) | 95 | 214 | 427 | 745 | 27 (3 to 46) | ||||
| 58 | 146 | 427 | 745 | 34 (6 to 53) | |||||
| Djennad (22) | 2741 | 1682 | 1288 | 534 | 27 (17 to 35) | ||||
| Dominguez (23) | 259 | 1636 | 272 | 1623 | 15 (−3 to 30) | ||||
| Kondo (24) | 64 | 170 | 131 | 307 | 16 (−30 to 46) | ||||
| Kim (25) | 231 | 326 | 247 | 310 | 10 (−15 to 30) | ||||
| 106 | 137 | 247 | 310 | 2 (−39 to 26) | |||||
| 54 | 94 | 130 | 165 | 29 (−6 to 52) | |||||
| 21 | 43 | 130 | 165 | 42 (−2 to 67) | |||||
| Author (Ref) | Outcome | Number of Cases § | All | 50–59 | 60–64 | 65–69 | 70–74 | 75–79 | 80–84 | ≥85 |
|---|---|---|---|---|---|---|---|---|---|---|
| PCV 13 | ||||||||||
| Vila-Corcoles (16) | Pneumonia | 12699 | −69 (−94 to −48) | −21 (−72 to 15) | −76 (−104 to −52) A | |||||
| Pn-Pneumonia | 1648 | −17 (−83 to 25) | 42 (−67 to 80) | −32 (−118 to 19) | ||||||
| Kolditz (17) | Pneumonia | 2344 | 11 (3 to 19) | − | 8 (−6 to 19) | 10 (−3 to 28) B | ||||
| PPV 23 | ||||||||||
| Kolditz (20) | Pneumonia | 30744 | 3 (1 to 6) | - | 2 (−6 to 2) | 0 (−4 to 3) C | ||||
| Suzuki (21) | Pn-Pneumonia | 419 | 27 (3 to 46) | - | 32 (−21 to 62) | 24 (−6 to 46) D | ||||
| VT-Pneumonia | 272 | 34 (6 to 53) | - | 40 (−6 to 69) | 28 (−10 to 53) D | |||||
| Djennad (22) | VT-IPD | 4423 | 27 (17 to 35) | - | 31 (16 to 44) E | 17 (−3 to 32) F | 34 (17 to 47) G | |||
| Dominguez (23) | Pneumonia | 1895 | 15 (−3 to 30) | - | 24 (−3 to 43) H | 12 (−22 to 36) I | 0 (−58 to 37) J | |||
| Kim (25) | Pn-Pneumonia | 557 | 10 (−15 to 30) | - | 35 (2 to 57) | −13 (−56 to 18) K | ||||
| VT-Pneumonia | 243 | −2 (−40 to 26) | - | 21 (−31 to 52) | −35 (−107 to 12) | |||||
| IPD | 148 | 29 (−6 to 52) | - | 57 (19 to 78) | 7 (−74 to 50) | |||||
| VT-IPD | 64 | 42 (−2 to 67) | - | 70 (25 to 88) | −20 (−184 to 60) | |||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berild, J.D.; Winje, B.A.; Vestrheim, D.F.; Slotved, H.-C.; Valentiner-Branth, P.; Roth, A.; Storsäter, J. A Systematic Review of Studies Published between 2016 and 2019 on the Effectiveness and Efficacy of Pneumococcal Vaccination on Pneumonia and Invasive Pneumococcal Disease in an Elderly Population. Pathogens 2020, 9, 259. https://doi.org/10.3390/pathogens9040259
Berild JD, Winje BA, Vestrheim DF, Slotved H-C, Valentiner-Branth P, Roth A, Storsäter J. A Systematic Review of Studies Published between 2016 and 2019 on the Effectiveness and Efficacy of Pneumococcal Vaccination on Pneumonia and Invasive Pneumococcal Disease in an Elderly Population. Pathogens. 2020; 9(4):259. https://doi.org/10.3390/pathogens9040259
Chicago/Turabian StyleBerild, Jacob Dag, Brita Askeland Winje, Didrik Frimann Vestrheim, Hans-Christian Slotved, Palle Valentiner-Branth, Adam Roth, and Jann Storsäter. 2020. "A Systematic Review of Studies Published between 2016 and 2019 on the Effectiveness and Efficacy of Pneumococcal Vaccination on Pneumonia and Invasive Pneumococcal Disease in an Elderly Population" Pathogens 9, no. 4: 259. https://doi.org/10.3390/pathogens9040259
APA StyleBerild, J. D., Winje, B. A., Vestrheim, D. F., Slotved, H.-C., Valentiner-Branth, P., Roth, A., & Storsäter, J. (2020). A Systematic Review of Studies Published between 2016 and 2019 on the Effectiveness and Efficacy of Pneumococcal Vaccination on Pneumonia and Invasive Pneumococcal Disease in an Elderly Population. Pathogens, 9(4), 259. https://doi.org/10.3390/pathogens9040259





