Non-typhoidal Salmonella in the Pig Production Chain: A Comprehensive Analysis of Its Impact on Human Health
Abstract
1. Introduction
2. Non-Typhoidal Salmonella in Pigs and Pig Production Chain
3. Major Pig-Related Salmonella Serotypes Associated with Human Infections
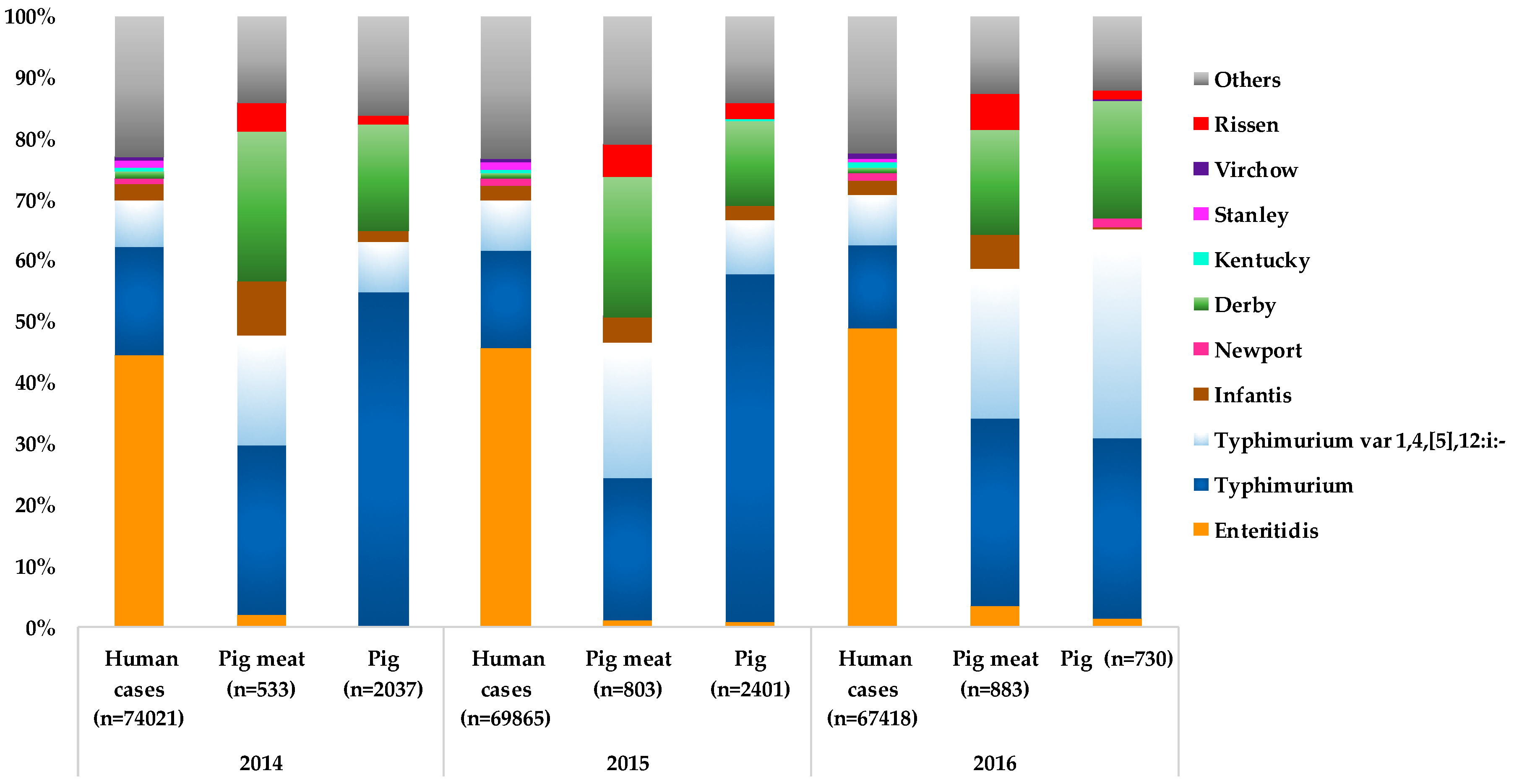
4. Dissemination of Pig-Associated Salmonella Serotypes and Clones
5. Antimicrobial Resistance in Salmonella and the Pork Linkage
5.1. Association between Antibiotic Use in Pig Production and Resistance in Salmonella
5.2. Correlation of Antimicrobial Resistance Rates between Salmonella from Pigs and Humans
5.3. Transmission of Antimicrobial Resistant Salmonella from Pork Meat to Humans
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Crump, J.A.; Sjölund-Karlsson, M.; Gordon, M.A.; Parry, C.M. Epidemiology, Clinical Presentation, Laboratory Diagnosis, Antimicrobial Resistance, and Antimicrobial Management of Invasive Salmonella Infections. Clin. Microbiol. Rev. 2015, 28, 901–937. [Google Scholar] [CrossRef] [PubMed]
- Arya, G.; Holtslander, R.; Robertson, J.; Yoshida, C.; Harris, J.; Parmley, J.; Nichani, A.; Johnson, R.; Poppe, C. Epidemiology, Pathogenesis, Genoserotyping, Antimicrobial Resistance, and Prevention and Control of Non-Typhoidal Salmonella Serovars. Curr. Clin. Microbiol. Rep. 2017, 4, 43–53. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Critically Important Antimicrobials for Human Medicine—5th Revision 2016—Ranking of Medically Important Antimicrobials for Risk Management of Antimicrobial Resistance Due to Non-Human Use; World Health Organization: Geneva, Switzerland, 2017; Available online: http://apps.who.int/iris/bitstream/handle/10665/255027/9789241512220-eng.pdf;jsessionid=72B265EF2D6C6CBC2136A8634744F0CB?sequence=1 (accessed on 13 March 2018).
- Centers for Disease Control and Prevention (CDC). Surveillance for Foodborne Disease Outbreaks, United States, 2015, Annual Report; US Department of Health and Human Services, CDC: Atlanta, Georgia, 2017. Available online: https://www.cdc.gov/foodsafety/pdfs/2015FoodBorneOutbreaks_508.pdf (accessed on 13 March 2018).
- European Food Safety Authority (EFSA) and European Centre for Disease Prevention and Control (ECDC). The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2017. EFSA J. 2018, 16, 5500. [Google Scholar]
- European Food Safety Authority (EFSA) and European Centre for Disease Prevention and Control (ECDC). The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2016. EFSA J. 2017, 15, 5077. [Google Scholar]
- European Food Safety Authority (EFSA) and European Centre for Disease Prevention and Control (ECDC). The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2015. EFSA J. 2016, 14, 4634. [Google Scholar]
- European Food Safety Authority (EFSA) and European Centre for Disease Prevention and Control (ECDC). The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2014. EFSA J. 2015, 13, 4329. [Google Scholar]
- Hugas, M.; Beloeil, P. Controlling Salmonella along the food chain in the European Union—Progress over the last ten years. Eurosurveillance 2014, 19, 20804. [Google Scholar] [CrossRef]
- Glass, K.; Fearnley, E.; Hocking, H.; Raupach, J.; Veitch, M.; Ford, L.; Kirk, M.D. Bayesian Source Attribution of Salmonellosis in South Australia. Risk Anal. 2016, 36, 561–570. [Google Scholar] [CrossRef]
- Bonardi, S. Salmonella in the pork production chain and its impact on human health in the European Union. Epidemiol. Infect. 2017, 145, 1513–1526. [Google Scholar] [CrossRef]
- Wong, D.M.A.L.F.; Hald, T.; van der Wolf, P.; Swanenburg, M. Epidemiology and control measures for Salmonella in pigs and pork. Livest. Prod. Sci. 2002, 76, 215–222. [Google Scholar] [CrossRef]
- Vidic, B.; Savic, S.; Prica, N.; Suvajdzic, L. Epizootiology and Control Measures for Salmonella in Pigs. Procedia Food Sci. 2015, 5, 312–315. [Google Scholar] [CrossRef]
- Communicable Diseases Branch. OzFoodNet—Enhancing Foodborne Disease Surveillance across Australia Third Quarter Summary, July—September, 2017 NSW; Health Protection NSW: Sydney, Australia, 2018. Available online: https://www.health.nsw.gov.au/Infectious/foodborne/Publications/NSW-3rd-quarterly-report-2017.pdf (accessed on 13 March 2018).
- Government of Canada. National Enteric Surveillance Program Annual Summary 2014; Public Health Agency of Canada: Guelph, ON, Canada, 2016. Available online: http://publications.gc.ca/collections/collection_2016/aspc-phac/HP37-15-2014-eng.pdf (accessed on 13 March 2018).
- Hendriksen, R.S.; Vieira, A.R.; Karlsmose, S.; Wong, D.M.A.L.F.; Jensen, A.B.; Wegener, H.C.; Aarestrup, F.M. Global Monitoring of Salmonella Serovar Distribution from the World Health Organization Global Foodborne Infections Network Country Data Bank: Results of Quality Assured Laboratories from 2001 to 2007. Foodborne Pathog. Dis. 2011, 8, 887–900. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Ke, B.; Deng, X.; Liang, J.; Ran, L.; Lu, L.; He, D.; Huang, Q.; Ke, C.; Li, Z.; et al. Serotypes, seasonal trends, and antibiotic resistance of non-typhoidal Salmonella from human patients in Guangdong Province, China, 2009–2012. BMC Infect. Dis. 2015, 15, 53. [Google Scholar] [CrossRef] [PubMed]
- Arai, N.; Sekizuka, T.; Tamamura, Y.; Tanaka, K.; Barco, L.; Izumiya, H.; Kusumoto, M.; Hinenoya, A.; Yamasaki, S.; Iwata, T.; et al. Phylogenetic Characterization of Salmonella enterica Serovar Typhimurium and Its Monophasic Variant Isolated from Food Animals in Japan Revealed Replacement of Major Epidemic Clones in the Last 4 Decades. J. Clin. Microbiol. 2018, 56, e01758-17. [Google Scholar] [CrossRef] [PubMed]
- Bonardi, S.; Alpigiani, I.; Bruini, I.; Barilli, E.; Brindani, F.; Morganti, M.; Cavallini, P.; Bolzoni, L.; Pongolini, S. Detection of Salmonella enterica in pigs at slaughter and comparison with human isolates in Italy. Int. J. Food Microbiol. 2016, 218, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Kerouanton, A.; Rose, V.; Weill, F.-X.; Granier, S.A.; Denis, M. Genetic Diversity and Antimicrobial Resistance Profiles of Salmonella enterica Serotype Derby Isolated from Pigs, Pork, and Humans in France. Foodborne Pathog. Dis. 2013, 10, 977–984. [Google Scholar] [CrossRef] [PubMed]
- García-Fierro, R.; Montero, I.; Bances, M.; González-Hevia, M.Á.; Rodicio, M.R. Antimicrobial Drug Resistance and Molecular Typing of Salmonella enterica Serovar Rissen from Different Sources. Microb. Drug Resist. 2016, 22, 211–217. [Google Scholar] [CrossRef]
- Piras, F.; Brown, D.J.; Meloni, D.; Mureddu, A.; Mazzette, R. Investigation of Salmonella enterica in Sardinian slaughter pigs: Prevalence, serotype and genotype characterization. Int. J. Food Microbiol. 2011, 151, 201–209. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Foodborne Diseases Active Surveillance Network (FoodNet): FoodNet 2015 Surveillance Report (Final Data); U.S. Department of Health and Human Services, CDC: Atlanta, Georgia, 2017. Available online: https://www.cdc.gov/foodnet/pdfs/FoodNet-Annual-Report-2015-508c.pdf (accessed on 13 March 2018).
- Mourão, J.; Machado, J.; Novais, C.; Antunes, P.; Peixe, L. Characterization of the emerging clinically-relevant multidrug-resistant Salmonella enterica serotype 4,[5],12:i:- (monophasic variant of S. Typhimurium) clones. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 2249–2257. [Google Scholar] [CrossRef]
- Mourão, J.; Marçal, S.; Ramos, P.; Campos, J.; Machado, J.; Peixe, L.; Novais, C.; Antunes, P. Tolerance to multiple metal stressors in emerging non-typhoidal MDR Salmonella serotypes: A relevant role for copper in anaerobic conditions. J. Antimicrob. Chemother. 2016, 71, 2147–2157. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA) and European Centre for Disease Prevention and Control (ECDC). The European Union summary report on antimicrobial resistance in zoonotic and indicator bacteria from humans, animals and food in 2015. EFSA J. 2017, 15, 4694. [Google Scholar]
- Hendriksen, R.S.; Bangtrakulnonth, A.; Pulsrikarn, C.; Pornreongwong, S.; Hasman, H.; Song, S.W.; Aarestrup, F.M. Antimicrobial Resistance and Molecular Epidemiology of Salmonella Rissen from Animals, Food Products, and Patients in Thailand and Denmark. Foodborne Pathog. Dis. 2008, 5, 605–619. [Google Scholar] [CrossRef] [PubMed]
- Arnott, A.; Wang, Q.; Bachmann, N.; Sadsad, R.; Biswas, C.; Sotomayor, C.; Howard, P.; Rockett, R.; Wiklendt, A.; Iredell, J.R.; et al. Multidrug-Resistant Salmonella enterica 4,[5],12:i:- Sequence Type 34, New South Wales, Australia, 2016–2017. Emerg. Infect. Dis. 2018, 24, 751–753. [Google Scholar] [CrossRef]
- Commission Regulation (EC) No 2160/2003 of the European Parliament and of the Council of 17 November 2003 on the Control of Salmonella and Other Specified Food-Borne Zoonotic Agents. Off. J. Eur. Union 2003, L 325, 1–15. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32003R2160 (accessed on 10 May 2018).
- Stiftung, H. Meat Atlas: Facts and Figures about the Animals We Eat; Heinrich-Böll-Stiftung: Berlin, Germany; Brussels, Belgium, 2014; Available online: https://www.boell.de/sites/default/files/meat_atlas2014_kommentierbar.pdf?dimension1=ds_fleischatlas_2014 (accessed on 15 May 2018).
- World’s Top Exports. Available online: www.worldstopexports.com/pork-exports-by-country (accessed on 15 May 2018).
- European Food Safety Authority (EFSA). Analysis of the baseline survey on the prevalence of Salmonella in holdings with breeding pigs in the EU, 2008—Part A: Salmonella prevalence estimates. EFSA J. 2009, 7, 1377. [Google Scholar]
- European Food Safety Authority (EFSA). Analysis of the baseline survey on the prevalence of Salmonella in holdings with breeding pigs, in the EU, 2008—Part B: Factors associated with Salmonella pen positivity. EFSA J. 2011, 9, 2329. [Google Scholar]
- Food Control Consultants Ltd Consortium (FCC Consortium). Analysis of the Costs and Benefits of Setting a Target for the Reduction of Salmonella in Breeding Pigs for European Commission Health and Consumers Directorate-General SANCO/2008/E2/056, Final Report. 2011. Available online: https://ec.europa.eu/food/sites/food/files/safety/docs/biosafety_food-borne-disease_salmonella_breeding-pigs_salm-cost-benefit.pdf (accessed on 10 May 2018).
- Andres, V.M.; Davies, R.H. Biosecurity Measures to Control Salmonella and Other Infectious Agents in Pig Farms: A Review. Compr. Rev. Food Sci. Food Saf. 2015, 14, 317–335. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Opinion of the Scientific Panel on biological hazards (BIOHAZ) related to “Risk assessment and mitigation options of Salmonella in pig production”. EFSA J. 2006, 4, 341. [Google Scholar] [CrossRef]
- Department for Environment, Food and Rural Affairs (DEFRA) of Public Health England. Zoonoses Report UK 2013. 2015. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/447771/pb13987-zoonoses-report-2013.pdf (accessed on 10 May 2018).
- Commission Regulation (EC) No 853/2004 of the European Parliament and of the Council of 29 April 2004 Laying down Specific Hygiene Rules for Food of Animal Origin. Off. J. Eur. Union 2004, L 139, 55–205. Available online: https://eur-lex.europa.eu/legal-content/en/ALL/?uri=CELEX%3A32004R0853 (accessed on 10 May 2018).
- Gomes-Neves, E.; Antunes, P.; Tavares, A.; Themudo, P.; Cardoso, M.F.; Gärtner, F.; Costa, J.M.; Peixe, L. Salmonella cross-contamination in swine abattoirs in Portugal: Carcasses, meat and meat handlers. Int. J. Food Microbiol. 2012, 157, 82–87. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Scientific Opinion on a Quantitative Microbiological Risk Assessment of Salmonella in slaughter and breeder pigs. EFSA J. 2010, 8, 1547. [Google Scholar] [CrossRef]
- Zhang, J.; Li, Z.; Cao, Z.; Wang, L.; Li, X.; Li, S.; Xu, Y. Bacteriophages as antimicrobial agents against major pathogens in swine: A review. J. Anim. Sci. Biotechnol. 2015, 6, 39. [Google Scholar] [CrossRef]
- Commission Regulation (EC) No 2073/2005 of 15 November 2005 on microbiological criteria for foodstuffs. Off. J. Eur. Union 2005, 50, 1–26.
- Commission Regulation (EC) No 217/2014 of 7 March 2014 amending Regulation (EC) No 2073/2005 as regards Salmonella in pig carcases. Off. J. Eur. Union 2014, 69, 93–94. Available online: https://publications.europa.eu/en/publication-detail/-/publication/2a5f9172-a699-11e3-8438-01aa75ed71a1/language-en (accessed on 10 May 2018).
- U.S. Food and Drug Administration (FDA). Available online: www.fda.gov/AnimalVeterinary/SafetyHealth/AntimicrobialResistance/NationalAntimicrobialResistanceMonitoringSystem/ucm059103.htm (accessed on 2 June 2018).
- Kich, J.D.; Coldebella, A.; Morés, N.; Nogueira, M.G.; Cardoso, M.; Fratamico, P.M.; Call, J.E.; Fedorka-Cray, P.; Luchansky, J.B. Prevalence, distribution, and molecular characterization of Salmonella recovered from swine finishing herds and a slaughter facility in Santa Catarina, Brazil. Int. J. Food Microbiol. 2011, 151, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Montes de Oca, S.; Talavera-Rojas, M.; Soriano-Vargas, E.; Barba-León, J.; Vázquez-Navarrete, J.; Acosta-Dibarrat, J.; Salgado-Miranda, C. Phenotypic and genotypic profile of clinical and animal multidrug-resistant Salmonella enterica isolates from Mexico. J. Appl. Microbiol. 2018, 124, 67–74. [Google Scholar] [CrossRef]
- Kikuvi, G.M.; Ombui, J.N.; Mitema, E.S. Serotypes and antimicrobial resistance profiles of Salmonella isolates from pigs at slaughter in Kenya. J. Infect. Dev. Ctries. 2010, 4, 243–248. [Google Scholar] [CrossRef]
- Akoachere, J.-F.T.K.; Tanih, N.F.; Ndip, L.M.; Ndip, R.N. Phenotypic characterization of Salmonella Typhimurium isolates from food-animals and abattoir drains in Buea, Cameroon. J. Health Popul. Nutr. 2009, 27, 612–618. [Google Scholar]
- Ma, S.; Lei, C.; Kong, L.; Jiang, W.; Liu, B.; Men, S.; Yang, Y.; Cheng, G.; Chen, Y.; Wang, H. Prevalence, Antimicrobial Resistance, and Relatedness of Salmonella Isolated from Chickens and Pigs on Farms, Abattoirs, and Markets in Sichuan Province, China. Foodborne Pathog. Dis. 2017, 14, 667–677. [Google Scholar] [CrossRef]
- Trongjit, S.; Angkititrakul, S.; Tuttle, R.E.; Poungseree, J.; Padungtod, P.; Chuanchuen, R. Prevalence and antimicrobial resistance in Salmonella enterica isolated from broiler chickens, pigs and meat products in Thailand-Cambodia border provinces. Microbiol. Immunol. 2017, 61, 23–33. [Google Scholar] [CrossRef]
- Rahmat, S.; Cheong, C.B.; Hamid, M.S.R.B.A. Challenges of Developing Countries in Complying Quality and Enhancing Standards in Food Industries. Procedia Soc. Behav. Sci. 2016, 224, 445–451. [Google Scholar] [CrossRef]
- Marder, E.P.; Griffin, P.M.; Cieslak, P.R.; Dunn, J.; Hurd, S.; Jervis, R.; Lathrop, S.; Muse, A.; Ryan, P.; Smith, K.; et al. Preliminary Incidence and Trends of Infections with Pathogens Transmitted Commonly Through Food—Foodborne Diseases Active Surveillance Network, 10 U.S. Sites, 2006–2017. MMWR. Morb. Mortal. Wkly. Rep. 2018, 67, 324–328. [Google Scholar] [CrossRef] [PubMed]
- Lan, N.P.H.; Phuong, T.L.T.; Huu, H.N.; Thuy, L.; Mather, A.E.; Park, S.E.; Marks, F.; Thwaites, G.E.; Van Vinh Chau, N.; Thompson, C.N.; et al. Invasive Non-typhoidal Salmonella Infections in Asia: Clinical Observations, Disease Outcome and Dominant Serovars from an Infectious Disease Hospital in Vietnam. PLoS Negl. Trop. Dis. 2016, 10, e0004857. [Google Scholar]
- Flockhart, L.; Pintar, K.; Cook, A.; McEwen, S.; Friendship, R.; Kelton, D.; Pollari, F. Distribution of Salmonella in Humans, Production Animal Operations and a Watershed in a FoodNet Canada Sentinel Site. Zoonoses Public Health 2017, 64, 41–52. [Google Scholar] [CrossRef]
- Silveira, L.; Marques, A.; Machado, J. Infecҫões por Salmonella Enterica no Período Entre 2000–2012; Boletim Epidemiológico; Instituto Nacional de Saúde Dr Ricardo Jorge (INSA): Lisbon, Portugal, 2013; Available online: http://repositorio.insa.pt/bitstream/10400.18/1680/1/observações%20Nº%20Especial%201%202013_artigo6.pdf (accessed on 10 May 2018).
- Silveira, L.; Pista, Â.; Machado, J. Caracterização Fenotípica e Genotípica de Isolados de Salmonella Enterica Recebidos no INSA Entre 2014 e 2016; Boletim Epidemiológico; Instituto Nacional de Saúde Dr Ricardo Jorge (INSA): Linbon, Portugal, 2017; Available online: http://repositorio.insa.pt/bitstream/10400.18/5592/1/Boletim_Epidemiologico_Observacoes_N22_2018_artigo11.pdf (accessed on 10 May 2018).
- Tamang, M.D.; Gurung, M.; Nam, H.-M.; Moon, D.C.; Kim, S.-R.; Jang, G.-C.; Jung, D.-Y.; Jung, S.-C.; Park, Y.-H.; Lim, S.-K. Prevalence and characterization of Salmonella in pigs from conventional and organic farms and first report of S. serovar 1,4,[5],12:i:- from Korea. Vet. Microbiol. 2015, 178, 119–124. [Google Scholar] [CrossRef]
- Zhao, X.; Yang, J.; Zhang, B.; Sun, S.; Chang, W. Characterization of Integrons and Resistance Genes in Salmonella Isolates from Farm Animals in Shandong Province, China. Front. Microbiol. 2017, 8, 1300. [Google Scholar] [CrossRef] [PubMed]
- Dahshan, H.; Chuma, T.; Shahada, F.; Akiba, M.; Fujimoto, H.; Akasaka, K.; Kamimura, Y.; Okamoto, K. Characterization of antibiotic resistance and the emergence of AmpC-producing Salmonella Infantis from pigs. J. Vet. Med. Sci. 2010, 72, 1437–1442. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Available online: http://www.cdc.gov/salmonella/outbreaks.html (accessed on 5 April 2018).
- Australian Government Department of Health (AGDH). Available online: www.health.gov.au/ (accessed on 5 April 2018).
- Antunes, P.; Mourão, J.; Pestana, N.; Peixe, L. Leakage of emerging clinically relevant multidrug-resistant Salmonella clones from pig farms. J. Antimicrob. Chemother. 2011, 66, 2028–2032. [Google Scholar] [CrossRef] [PubMed]
- Antunes, P.; Machado, J.; Peixe, L. Characterization of antimicrobial resistance and class 1 and 2 integrons in Salmonella enterica isolates from different sources in Portugal. J. Antimicrob. Chemother. 2006, 58, 297–304. [Google Scholar] [CrossRef]
- Butaye, P.; Michael, G.B.; Schwarz, S.; Barrett, T.J.; Brisabois, A.; White, D.G. The clonal spread of multidrug-resistant non-typhi Salmonella serotypes. Microbes Infect. 2006, 8, 1891–1897. [Google Scholar] [CrossRef]
- Gomes-Neves, E.; Antunes, P.; Manageiro, V.; Gärtner, F.; Caniça, M.; da Costa, J.M.C.; Peixe, L. Clinically relevant multidrug resistant Salmonella enterica in swine and meat handlers at the abattoir. Vet. Microbiol. 2014, 168, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Threlfall, E.J. Epidemic Salmonella Typhimurium DT 104—A truly international multiresistant clone. J. Antimicrob. Chemother. 2000, 46, 7–10. [Google Scholar] [CrossRef]
- Antunes, P.; Coque, T.M.; Peixe, L. Emergence of an IncIγ plasmid encoding CMY-2 ß-lactamase associated with the international ST19 OXA-30-producing ß-lactamase Salmonella Typhimurium multidrug-resistant clone. J. Antimicrob. Chemother. 2010, 65, 2097–2100. [Google Scholar] [CrossRef] [PubMed]
- Antunes, P.; Machado, J.; Sousa, J.C.; Peixe, L. Dissemination amongst humans and food products of animal origin of a Salmonella Typhimurium clone expressing an integron-borne OXA-30 beta-lactamase. J. Antimicrob. Chemother. 2004, 54, 429–434. [Google Scholar] [CrossRef] [PubMed]
- García, V.; Montero, I.; Bances, M.; Rodicio, R.; Rodicio, M.R. Incidence and Genetic Bases of Nitrofurantoin Resistance in Clinical Isolates of Two Successful Multidrug-Resistant Clones of Salmonella enterica Serovar Typhimurium: Pandemic “DT 104” and pUO-StVR2. Microb. Drug Resist. 2017, 23, 405–412. [Google Scholar] [CrossRef]
- Lucarelli, C.; Dionisi, A.M.; Torpdahl, M.; Villa, L.; Graziani, C.; Hopkins, K.; Threlfall, J.; Caprioli, A.; Luzzi, I. Evidence for a Second Genomic Island Conferring Multidrug Resistance in a Clonal Group of Strains of Salmonella enterica Serovar Typhimurium and its Monophasic Variant Circulating in Italy, Denmark, and the United Kingdom. J. Clin. Microbiol. 2010, 48, 2103–2109. [Google Scholar] [CrossRef]
- Hopkins, K.L.; Kirchner, M.; Guerra, B.; Granier, S.A.; Lucarelli, C.; Porrero, M.C.; Jakubczak, A.; Threlfall, E.J.; Mevius, D.J. Multiresistant Salmonella enterica serovar 4,[5],12:i:- in Europe: A new pandemic strain? Euro Surveill. 2010, 15, 19580. [Google Scholar]
- Hauser, E.; Tietze, E.; Helmuth, R.; Junker, E.; Blank, K.; Prager, R.; Rabsch, W.; Appel, B.; Fruth, A.; Malorny, B. Pork Contaminated with Salmonella enterica Serovar 4,[5],12:i:-, an Emerging Health Risk for Humans. Appl. Environ. Microbiol. 2010, 76, 4601–4610. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Scientific Opinion on monitoring and assessment of the public health risk of “Salmonella Typhimurium-like” strains. EFSA J. 2010, 8, 1826. [Google Scholar] [CrossRef]
- Pesciaroli, M.; Cucco, L.; De Luca, S.; Massacci, F.R.; Maresca, C.; Medici, L.; Paniccià, M.; Scoccia, E.; Staffolani, M.; Pezzotti, G.; et al. Association between pigs with high caecal Salmonella loads and carcass contamination. Int. J. Food Microbiol. 2017, 242, 82–86. [Google Scholar] [CrossRef]
- Carnevali, C.; Morganti, M.; Scaltriti, E.; Bolzoni, L.; Pongolini, S.; Casadei, G. Occurance of MCR-1 colistin-resitant Salmonella isolates recovered from human and animals in Italy, 2012–2015. Antimicrob. Agents Chemother. 2016, 60, 7532–7534. [Google Scholar]
- Lucarelli, C.; Dionisi, A.M.; Filetici, E.; Owczarek, S.; Luzzi, I.; Villa, L. Nucleotide sequence of the chromosomal region conferring multidrug resistance (R-type ASSuT) in Salmonella Typhimurium and monophasic Salmonella Typhimurium strains. J. Antimicrob. Chemother. 2012, 67, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Mandilara, G.; Lambiri, M.; Polemis, M.; Passiotou, M.; Vatopoulos, A. Phenotypic and molecular characterisation of multiresistant monophasic Salmonella Typhimurium (1,4,[5],12:i:-) in Greece, 2006 to 2011. Eurosurveillance 2013, 18, 20496. [Google Scholar] [PubMed]
- Mulvey, M.R.; Finley, R.; Allen, V.; Ang, L.; Bekal, S.; El Bailey, S.; Haldane, D.; Hoang, L.; Horsman, G.; Louie, M.; et al. Emergence of multidrug-resistant Salmonella enterica serotype 4,[5],12:i:- involving human cases in Canada: Results from the Canadian Integrated Program on Antimicrobial Resistance Surveillance (CIPARS), 2003–2010. J. Antimicrob. Chemother. 2013, 68, 1982–1986. [Google Scholar] [CrossRef] [PubMed]
- Moreno Switt, A.I.; Soyer, Y.; Warnick, L.D.; Wiedmann, M. Emergence, Distribution, and Molecular and Phenotypic Characteristics of Salmonella enterica Serotype 4,5,12:i:-. Foodborne Pathog. Dis. 2009, 6, 407–415. [Google Scholar] [CrossRef] [PubMed]
- García, P.; Malorny, B.; Hauser, E.; Mendoza, M.C.; Rodicio, M.R. Genetic Types, Gene Repertoire, and Evolution of Isolates of the Salmonella enterica Serovar 4,5,12:i:- Spanish Clone Assigned to Different Phage Types. J. Clin. Microbiol. 2013, 51, 973–978. [Google Scholar] [CrossRef] [PubMed]
- García, P.; Guerra, B.; Bances, M.; Mendoza, M.C.; Rodicio, M.R. IncA/C plasmids mediate antimicrobial resistance linked to virulence genes in the Spanish clone of the emerging Salmonella enterica serotype 4,[5],12:i:-. J. Antimicrob. Chemother. 2011, 66, 543–549. [Google Scholar] [CrossRef]
- Antunes, P.; Machado, J.; Peixe, L. Dissemination of sul3-containing elements linked to class 1 integrons with an unusual 3′ conserved sequence region among Salmonella isolates. Antimicrob. Agents Chemother. 2007, 51, 1545–1548. [Google Scholar] [CrossRef]
- García, P.; Hopkins, K.L.; García, V.; Beutlich, J.; Mendoza, M.C.; Threlfall, J.; Mevius, D.; Helmuth, R.; Rodicio, M.R.; Guerra, B. Diversity of Plasmids Encoding Virulence and Resistance Functions in Salmonella enterica subsp. enterica Serovar Typhimurium Monophasic Variant 4,[5],12:i:- Strains Circulating in Europe. PLoS ONE 2014, 9, e89635. [Google Scholar]
- U.S. Food and Drug Administration (FDA). Available online: https://www.fda.gov/downloads/AnimalVeterinary/SafetyHealth/AntimicrobialResistance/NationalAntimicrobialResistanceMonitoringSystem/UCM498134.pdf (accessed on 2 June 2018).
- Kérouanton, A.; Hirchaud, E.; Rose, V.; Esnault, E.; Naquin, D.; Denis, M. First Complete Genome Sequence of a Salmonella enterica subsp. enterica Serovar Derby Strain Associated with Pork in France. Genome Announc. 2015, 3, e00853-15. [Google Scholar]
- Vo, A.T.T.; van Duijkeren, E.; Fluit, A.C.; Wannet, W.J.B.; Verbruggen, A.J.; Maas, H.M.E.; Gaastra, W. Antibiotic resistance, integrons and Salmonella genomic island 1 among non-typhoidal Salmonella serovars in The Netherlands. Int. J. Antimicrob. Agents 2006, 28, 172–179. [Google Scholar] [CrossRef]
- Valdezate, S.; Vidal, A.; Herrera-León, S.; Pozo, J.; Rubio, P.; Usera, M.A.; Carvajal, A.; Echeita, M.A. Salmonella Derby Clonal Spread from Pork. Emerg. Infect. Dis. 2005, 11, 694–698. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Ren, X.; Feng, Z.; Fu, Y.; Hong, Y.; Shen, Z.; Zhang, L.; Liao, M.; Xu, X.; Zhang, J. Phenotypic Characteristics and Genetic Diversity of Salmonella enterica Serotype Derby Isolated from Human Patients and Foods of Animal Origin. Foodborne Pathog. Dis. 2017, 14, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Pornsukarom, S.; Patchanee, P.; Erdman, M.; Cray, P.F.; Wittum, T.; Lee, J.; Gebreyes, W.A. Comparative phenotypic and genotypic analyses of Salmonella Rissen that originated from food animals in Thailand and United States. Zoonoses Public Health 2015, 62, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Van Hoek, A.H.A.M.; de Jonge, R.; van Overbeek, W.M.; Bouw, E.; Pielaat, A.; Smid, J.H.; Malorny, B.; Junker, E.; Löfström, C.; Pedersen, K.; et al. A quantitative approach towards a better understanding of the dynamics of Salmonella spp. in a pork slaughter-line. Int. J. Food Microbiol. 2012, 153, 45–52. [Google Scholar] [CrossRef]
- Luzzi, I.; Galetta, P.; Massari, M.; Rizzo, C.; Dionisi, A.M.; Filetici, E.; Cawthorne, A.; Tozzi, A.; Argentieri, M.; Bilei, S.; et al. An Easter outbreak of Salmonella Typhimurium DT 104A associated with traditional pork salami in Italy. Eurosurveillance 2007, 12, 11–12. [Google Scholar] [CrossRef]
- Torpdahl, M.; Sorensen, G.; Ethelberg, S.; Sandø, G.; Gammelgård, K.; Porsbo, L.J. A regional outbreak of S. Typhimurium in Denmark and identification of the source using MLVA typing. Eurosurveillance 2006, 11, 134–136. [Google Scholar] [CrossRef]
- Bruun, T.; Sørensen, G.; Forshell, L.P.; Jensen, T.; Nygard, K.; Kapperud, G.; Lindstedt, B.A.; Berglund, T.; Wingstrand, A.; Petersen, R.F.; et al. An outbreak of Salmonella Typhimurium infections in Denmark, Norway and Sweden, 2008. Eurosurveillance 2009, 14, 19147. [Google Scholar]
- Kuhn, K.; Torpdahl, M.; Frank, C.; Sigsgaard, K.; Ethelberg, S. An outbreak of Salmonella Typhimurium traced back to salami, Denmark, April to June 2010. Eurosurveillance 2011, 16, 19863. [Google Scholar]
- Wójcik, O.P.; Kjelsø, C.; Kuhn, K.G.; Müller, L.; Jensen, T.; Kjeldsen, M.K.; Ethelberg, S. Salmonella Typhimurium outbreak associated with smoked pork tenderloin in Denmark, January to March 2011. Scand. J. Infect. Dis. 2012, 44, 903–908. [Google Scholar] [CrossRef]
- Arnedo-Pena, A.; Sabater-Vidal, S.; Herrera-León, S.; Bellido-Blasco, J.B.; Silvestre-Silvestre, E.; Meseguer-Ferrer, N.; Yague-Muñoz, A.; Gil-Fortuño, M.; Romeu-García, A.; Moreno-Muñoz, R. An outbreak of monophasic and biphasic Salmonella Typhimurium, and Salmonella Derby associated with the consumption of dried pork sausage in Castellon (Spain). Enferm. Infecc. Microbiol. Clin. 2016, 34, 544–550. [Google Scholar] [CrossRef]
- Australian Government Department of Health (AGDH). OzFoodNet: Foodborne Disease in Australia Annual Reports of the OzFoodNet Network. Available online: http://www.health.gov.au (accessed on 1 June 2018).
- RASFF (Rapid Alert System for Food and Feed). Available online: https://ec.europa.eu/food/safety/rasff_en (accessed on 1 June 2018).
- Mossong, J.; Marques, P.; Ragimbeau, C.; Huberty-Krau, P.; Losch, S.; Meyer, G.; Moris, G.; Strottner, C.; Rabsch, W.; Schneider, F. Outbreaks of monophasic Salmonella enterica serovar 4,[5],12:i:- in Luxembourg, 2006. Eurosurveillance 2007, 12, E11–E12. [Google Scholar] [CrossRef]
- Bone, A.; Noel, H.; Le Hello, S.; Pihier, N.; Danan, C.; Raguenaud, M.E.; Salah, S.; Bellali, H.; Vaillant, V.; Weill, F.X.; et al. Nationwide outbreak of Salmonella enterica serotype 4,12:i:- infections in France, linked to dried pork sausage, March–May 2010. Eurosurveillance 2010, 15, 1–3. [Google Scholar]
- Gossner, C.M.; van Cauteren, D.; Le Hello, S.; Weill, F.X.; Terrien, E.; Tessier, S.; Janin, C.; Brisabois, A.; Dusch, V.; Vaillant, V.; et al. Nationwide outbreak of Salmonella enterica serotype 4,[5],12:i:- infection associated with consumption of dried pork sausage, France, November to December 2011. Eurosurveillance 2012, 17, 20071. [Google Scholar] [CrossRef]
- Morganti, M.; Bolzoni, L.; Scaltriti, E.; Casadei, G.; Carra, E.; Rossi, L.; Gherardi, P.; Faccini, F.; Arrigoni, N.; Sacchi, A.R.; et al. Rise and fall of outbreak-specific clone inside endemic pulsotype of Salmonella 4,[5],12:i:-; insights from high-resolution molecular surveillance in Emilia-Romagna, Italy, 2012 to 2015. Eurosurveillance 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Hernández Arricibita, E.; Santamaria Zuazua, R.; Ramos López, G.; Herrera-León, S.; Kárkamo Zuñeda, J.A.; Muniozguren Agirre, N. Brote de infecciones por Salmonella enterica serovar Typhimurium asociado al consumo de chorizo en Bizkaia. Enferm. Infecc. Microbiol. Clin. 2016, 34, 577–578. [Google Scholar] [CrossRef] [PubMed]
- Simon, S.; Trost, E.; Bender, J.; Fuchs, S.; Malorny, B.; Rabsch, W.; Prager, R.; Tietze, E.; Flieger, A. Evaluation of WGS based approaches for investigating a food-borne outbreak caused by Salmonella enterica serovar Derby in Germany. Food Microbiol. 2018, 71, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Gilsdorf, A.; Jansen, A.; Alpers, K.; Dieckmann, H.; van Treeck, U.; Hauri, A.M.; Fell, G.; Littmann, M.; Rautenberg, P.; Prager, R.; et al. A nationwide outbreak of Salmonella Bovismorbificans PT24, Germany, December 2004–March 2005. Eurosurveillance 2005, 10, E050324.1. [Google Scholar] [CrossRef]
- Brandwagt, D.; van den Wijngaard, C.; Tulen, A.D.; Mulder, A.C.; Hofhuis, A.; Jacobs, R.; Heck, M.; Verbruggen, A.; van den Kerkhof, H.; Slegers-Fitz-James, I.; et al. Outbreak of Salmonella Bovismorbificans associated with the consumption of uncooked ham products, the Netherlands, 2016 to 2017. Eurosurveillance 2018, 23. [Google Scholar] [CrossRef]
- Jansen, A.; Frank, C.; Prager, R.; Oppermann, H.; Stark, K. Nation-wide outbreak of Salmonella Give in Germany, 2004. Z. Gastroenterol. 2005, 43, 707–713. [Google Scholar] [CrossRef]
- Horváth, J.K.; Mengel, M.; Krisztalovics, K.; Nogrady, N.; Pászti, J.; Lenglet, A.; Takkinen, J. Investigation into an unusual increase of human cases of Salmonella Goldcoast infection in Hungary in 2009. Eurosurveillance 2013, 18, 20422. [Google Scholar] [CrossRef] [PubMed]
- Scavia, G.; Ciaravino, G.; Luzzi, I.; Lenglet, A.; Ricci, A.; Barco, L.; Pavan, A.; Zaffanella, F.; Dionisi, A.M. A multistate epidemic outbreak of Salmonella Goldcoast infection in humans, June 2009 to March 2010: The investigation in Italy. Eurosurveillance 2013, 18, 20424. [Google Scholar] [CrossRef] [PubMed]
- Wegener, H.C.; Baggesen, D.L. Investigation of an outbreak of human salmonellosis caused by Salmonella enterica ssp. enterica serovar Infantis by use of pulsed field gel electrophoresis. Int. J. Food Microbiol. 1996, 32, 125–131. [Google Scholar] [CrossRef]
- Chironna, M.; Tafuri, S.; Gallone, M.S.; Sallustio, A.; Martinelli, D.; Prato, R.; Germinario, C. Outbreak of Salmonella Infantis gastroenteritis among people who had eaten at a hash house in southern Italy. Public Health 2014, 128, 438–443. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, S.; Harries, M.; Prager, R.; Hofig, A.; Ahrens, B.; Hoffmann, L.; Rabsch, W.; Mertens, E.; Rimek, D. A prolonged outbreak of Salmonella Infantis associated with pork products in central Germany, April–October 2013. Epidemiol. Infect. 2016, 144, 1429–1439. [Google Scholar] [CrossRef] [PubMed]
- Noël, H.; Dominguez, M.; Weill, F.X.; Brisabois, A.; Duchazeaubeneix, C.; Kerouanton, A.; Delmas, G.; Pihier, N.; Couturier, E. Outbreak of Salmonella enterica serotype Manhattan infection associated with meat products, France, 2005. Eurosurveillance 2006, 11, 270–273. [Google Scholar] [CrossRef]
- Schielke, A.; Rabsch, W.; Prager, R.; Simon, S.; Fruth, A.; Helling, R.; Schnabel, M.; Siffczyk, C.; Wieczorek, S.; Schroeder, S.; et al. Two consecutive large outbreaks of Salmonella Muenchen linked to pig farming in Germany, 2013 to 2014: Is something missing in our regulatory framework? Eurosurveillance 2017, 22, 30528. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, S.; Dierick, K.; Heylen, K.; De Baere, T.; Pochet, B.; Robesyn, E.; Lokietek, S.; Van Meervenne, E.; Imberechts, H.; De Zutter, L.; et al. Lessons learned from the management of a national outbreak of Salmonella Ohio linked to pork meat processing and distribution. J. Food Prot. 2010, 73, 529–534. [Google Scholar] [CrossRef] [PubMed]
- Fabrega, A.; Vila, J. Salmonella enterica Serovar Typhimurium Skills to Succeed in the Host: Virulence and Regulation. Clin. Microbiol. Rev. 2013, 26, 308–341. [Google Scholar] [CrossRef]
- Chaudhuri, R.R.; Morgan, E.; Peters, S.E.; Pleasance, S.J.; Hudson, D.L.; Davies, H.M.; Wang, J.; van Diemen, P.M.; Buckley, A.M.; Bowen, A.J.; et al. Comprehensive Assignment of Roles for Salmonella Typhimurium Genes in Intestinal Colonization of Food-Producing Animals. PLoS Genet. 2013, 9, e1003456. [Google Scholar] [CrossRef] [PubMed]
- Garaizar, J.; Porwollik, S.; Echeita, A.; Rementeria, A.; Herrera, S.; Wong, R.M.-Y.; Frye, J.; Usera, M.A.; McClelland, M. DNA Microarray-Based Typing of an Atypical Monophasic Salmonella enterica Serovar. J. Clin. Microbiol. 2002, 40, 2074–2078. [Google Scholar] [CrossRef]
- Patchanee, P.; Eiamsam-ang, T.; Vanaseang, J.; Boonkhot, P.; Tadee, P. Determination of regional relationships among Salmonella spp. isolated from retail pork circulating in the Chiang Mai municipality area using a WGS data approach. Int. J. Food Microbiol. 2017, 254, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Wannaprasat, W.; Padungtod, P.; Chuanchuen, R. Class 1 integrons and virulence genes in Salmonella enterica isolates from pork and humans. Int. J. Antimicrob. Agents 2011, 37, 457–461. [Google Scholar] [CrossRef]
- Del Cerro, A.; Soto, S.; Mendoza, M. Virulence and antimicrobial-resistance gene profiles determined by PCR-based procedures for Salmonella isolated from samples of animal origin. Food Microbiol. 2003, 20, 431–438. [Google Scholar] [CrossRef]
- Guerra, B.; Laconcha, I.; Soto, S.M.; Gonzãlez-Hevia, M.Ã.; Mendoza, M.C. Molecular characterisation of emergent multiresistant Salmonella enterica serotype [4,5,12:i:-] organisms causing human salmonellosis. FEMS Microbiol. Lett. 2000, 190, 341–347. [Google Scholar] [CrossRef]
- Rajtak, U.; Boland, F.; Leonard, N.; Bolton, D.; Fanning, S. Roles of Diet and the Acid Tolerance Response in Survival of Common Salmonella Serotypes in Feces of Finishing Pigs. Appl. Environ. Microbiol. 2012, 78, 110–119. [Google Scholar] [CrossRef]
- Hayward, M.R.; AbuOun, M.; La Ragione, R.M.; Tchórzewska, M.A.; Cooley, W.A.; Everest, D.J.; Petrovska, L.; Jansen, V.A.A.; Woodward, M.J. SPI-23 of S. Derby: Role in Adherence and Invasion of Porcine Tissues. PLoS ONE 2014, 9, e107857. [Google Scholar] [CrossRef] [PubMed]
- Sévellec, Y.; Vignaud, M.-L.; Granier, S.A.; Lailler, R.; Feurer, C.; Le Hello, S.; Mistou, M.-Y.; Cadel-Six, S. Polyphyletic Nature of Salmonella enterica Serotype Derby and Lineage-Specific Host-Association Revealed by Genome-Wide Analysis. Front. Microbiol. 2018, 9, 891. [Google Scholar] [CrossRef]
- Mourão, J.; Novais, C.; Machado, J.; Peixe, L.; Antunes, P. Metal tolerance in emerging clinically relevant multidrug-resistant Salmonella enterica serotype 4,[5],12:i:- clones circulating in Europe. Int. J. Antimicrob. Agents 2015, 45, 610–616. [Google Scholar] [CrossRef]
- Petrovska, L.; Mather, A.E.; AbuOun, M.; Branchu, P.; Harris, S.R.; Connor, T.; Hopkins, K.L.; Underwood, A.; Lettini, A.A.; Page, A.; et al. Microevolution of Monophasic Salmonella Typhimurium during Epidemic, United Kingdom, 2005–2010. Emerg. Infect. Dis. 2016, 22, 617–624. [Google Scholar] [CrossRef]
- Aarestrup, F.M. The livestock reservoir for antimicrobial resistance: A personal view on changing patterns of risks, effects of interventions and the way forward. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140085. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). WHO Guidelines on Use of Medically Important Antimicrobials in Food-Producing Animals; World Health Organization: Geneva, Switzerland, 2017; Available online: http://apps.who.int/iris/bitstream/handle/10665/258970/9789241550130-eng.pdf?sequence=1 (accessed on 25 May 2018).
- Barton, M.D. Impact of antibiotic use in the swine industry. Curr. Opin. Microbiol. 2014, 19, 9–15. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA) Panel on Biological Hazards (BIOHAZ). Scientific Opinion on the public health risks of bacterial strains producing extended-spectrum β-lactamases and/or AmpC β-lactamases in food and food-producing animals. EFSA J. 2011, 9, 2322. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA) Panel on Biological Hazards (BIOHAZ). Joint Opinion on antimicrobial resistance (AMR) focused on zoonotic infections. EFSA J. 2009, 7, 1372. [Google Scholar] [CrossRef]
- Van Boeckel, T.P.; Brower, C.; Gilbert, M.; Grenfell, B.T.; Levin, S.A.; Robinson, T.P.; Teillant, A.; Laxminarayan, R. Global trends in antimicrobial use in food animals. Proc. Natl. Acad. Sci. USA 2015, 112, 5649–5654. [Google Scholar] [CrossRef]
- European Medicines Agency (EMA). Sales of Veterinary Antimicrobial Agents in 30 European Countries in 2015. 2017. Available online: https://www.ema.europa.eu/documents/report/seventh-esvac-report-sales-veterinary-antimicrobial-agents-30-european-countries-2015_en.pdf (accessed on 25 May 2018).
- Delsol, A.A.; Anjum, M.; Woodward, M.J.; Sunderland, J.; Roe, J.M. The effect of chlortetracycline treatment and its subsequent withdrawal on multi-resistant Salmonella enterica serovar Typhimurium DT104 and commensal Escherichia coli in the pig. J. Appl. Microbiol. 2003, 95, 1226–1234. [Google Scholar] [CrossRef]
- Wiuff, C.; Lykkesfeldt, J.; Svendsen, O.; Aarestrup, F. The effects of oral and intramuscular administration and dose escalation of enrofloxacin on the selection of quinolone resistance among Salmonella and coliforms in pigs. Res. Vet. Sci. 2003, 75, 185–193. [Google Scholar] [CrossRef]
- Emborg, H.-D.; Vigre, H.; Jensen, V.F.; Vieira, A.R.P.; Baggesen, D.L.; Aarestrup, F.M. Tetracycline Consumption and Occurrence of Tetracycline Resistance in Salmonella Typhimurium Phage Types from Danish Pigs. Microb. Drug Resist. 2007, 13, 289–294. [Google Scholar] [CrossRef] [PubMed]
- The National Antimicrobial Resistance Monitoring System (NARMS). NARMS 2015 Integrated Report. 2017. Available online: https://www.fda.gov/downloads/AnimalVeterinary/SafetyHealth/AntimicrobialResistance/NationalAntimicrobialResistanceMonitoringSystem/UCM581468.pdf (accessed on 2 June 2018).
- European Food Safety Authority (EFSA) and European Centre for Disease Prevention and Control (ECDC). Antimicrobial Resistance in Europe Resistance of Salmonella and E. coli in Food, Animals and Humans, Country by Country. Available online: https://www.efsa.europa.eu/en/interactive_pages/AMR_Report_2015 (accessed on 2 June 2018).
- U.S. Food and Drug Administration (FDA)—NARMS Now: Integrated Data. Available online: https://www.fda.gov/AnimalVeterinary/SafetyHealth/AntimicrobialResistance/NationalAntimicrobialResistanceMonitoringSystem/ucm416741.htm (accessed on 2 June 2018).
- Lee, H.-Y.; Su, L.-H.; Tsai, M.-H.; Kim, S.-W.; Chang, H.-H.; Jung, S.-I.; Park, K.-H.; Perera, J.; Carlos, C.; Tan, B.H.; et al. High Rate of Reduced Susceptibility to Ciprofloxacin and Ceftriaxone among Nontyphoid Salmonella Clinical Isolates in Asia. Antimicrob. Agents Chemother. 2009, 53, 2696–2699. [Google Scholar] [CrossRef] [PubMed]
- Chiou, C.-S.; Chen, Y.-T.; Wang, Y.-W.; Liu, Y.-Y.; Kuo, H.-C.; Tu, Y.-H.; Lin, A.-C.; Liao, Y.-S.; Hong, Y.-P. Dissemination of mcr-1-Carrying Plasmids among Colistin-Resistant Salmonella Strains from Humans and Food-Producing Animals in Taiwan. Antimicrob. Agents Chemother. 2017, 61, e00338-17. [Google Scholar] [CrossRef]
- Van, T.T.H.; Nguyen, H.N.K.; Smooker, P.M.; Coloe, P.J. The antibiotic resistance characteristics of non-typhoidal Salmonella enterica isolated from food-producing animals, retail meat and humans in South East Asia. Int. J. Food Microbiol. 2012, 154, 98–106. [Google Scholar] [CrossRef]
- Bai, L.; Lan, R.; Zhang, X.; Cui, S.; Xu, J.; Guo, Y.; Li, F.; Zhang, D. Prevalence of Salmonella Isolates from Chicken and Pig Slaughterhouses and Emergence of Ciprofloxacin and Cefotaxime Co-Resistant S. enterica Serovar Indiana in Henan, China. PLoS ONE 2015, 10, e0144532. [Google Scholar] [CrossRef]
- Ellerbroek, L.; Narapati, D.; Tai, N.P.; Poosaran, N.; Pinthong, R.; Sirimalaisuwan, A.; Tshering, P.; Fries, R.; Zessin, K.-H.; Baumann, M.; et al. Antibiotic Resistance in Salmonella Isolates from Imported Chicken Carcasses in Bhutan and from Pig Carcasses in Vietnam. J. Food Prot. 2010, 73, 376–379. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Lai, J.; Wang, Y.; Liu, S.; Li, Y.; Liu, K.; Shen, J.; Wu, C. Prevalence and characterization of Salmonella species isolated from pigs, ducks and chickens in Sichuan Province, China. Int. J. Food Microbiol. 2013, 163, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Morales, A.S.; Fragoso de Araújo, J.; de Moura Gomes, V.T.; Reis Costa, A.T.; dos Prazeres Rodrigues, D.; Porfida Ferreira, T.S.; de Lima Filsner, P.H.N.; Felizardo, M.R.; Micke Moreno, A. Colistin Resistance in Escherichia coli and Salmonella enterica Strains Isolated from Swine in Brazil. Sci. World J. 2012, 2012, 109795. [Google Scholar] [CrossRef]
- Yang, Y.-Q.; Zhang, A.-Y.; Ma, S.-Z.; Kong, L.-H.; Li, Y.-X.; Liu, J.-X.; Davis, M.A.; Guo, X.-Y.; Liu, B.-H.; Lei, C.-W.; et al. Co-occurrence of mcr-1 and ESBL on a single plasmid in Salmonella enterica. J. Antimicrob. Chemother. 2016, 71, 2336–2338. [Google Scholar] [CrossRef]
- Li, X.-P.; Fang, L.-X.; Song, J.-Q.; Xia, J.; Huo, W.; Fang, J.-T.; Liao, X.-P.; Liu, Y.-H.; Feng, Y.; Sun, J. Clonal spread of mcr-1 in PMQR-carrying ST34 Salmonella isolates from animals in China. Sci. Rep. 2016, 6, 38511. [Google Scholar] [CrossRef] [PubMed]
- Yi, L.; Wang, J.; Gao, Y.; Liu, Y.; Doi, Y.; Wu, R.; Zeng, Z.; Liang, Z.; Liu, J.-H. mcr-1-Harboring Salmonella enterica Serovar Typhimurium Sequence Type 34 in Pigs, China. Emerg. Infect. Dis. 2017, 23, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Rhouma, M.; Beaudry, F.; Thériault, W.; Letellier, A. Colistin in Pig Production: Chemistry, Mechanism of Antibacterial Action, Microbial Resistance Emergence, and One Health Perspectives. Front. Microbiol. 2016, 7, 1789. [Google Scholar] [CrossRef]
- Poirel, L.; Nordmann, P. Emerging plasmid-encoded colistin resistance: The animal world as the culprit? J. Antimicrob. Chemother. 2016, 71, 2326–2327. [Google Scholar] [CrossRef]
- Wang, R.; van Dorp, L.; Shaw, L.P.; Bradley, P.; Wang, Q.; Wang, X.; Jin, L.; Zhang, Q.; Liu, Y.; Rieux, A.; et al. The global distribution and spread of the mobilized colistin resistance gene mcr-1. Nat. Commun. 2018, 9, 1179. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Zeng, X.; Li, X.-P.; Liao, X.-P.; Liu, Y.-H.; Lin, J. Plasmid-mediated colistin resistance in animals: Current status and future directions. Anim. Heal. Res. Rev. 2017, 18, 136–152. [Google Scholar] [CrossRef] [PubMed]
- McGlone, J. The Future of Pork Production in the World: Towards Sustainable, Welfare-Positive Systems. Animals 2013, 3, 401–415. [Google Scholar] [CrossRef] [PubMed]
- Rozwandowicz, M.; Brouwer, M.S.M.; Fischer, J.; Wagenaar, J.A.; Gonzalez-Zorn, B.; Guerra, B.; Mevius, D.J.; Hordijk, J. Plasmids carrying antimicrobial resistance genes in Enterobacteriaceae. J. Antimicrob. Chemother. 2018, 73, 1121–1137. [Google Scholar] [CrossRef]
- Jacoby, G.A. Plasmid-Mediated Quinolone Resistance. In Antimicrobial Drug Resistance; Springer International Publishing: Cham, Switzerland, 2017; pp. 265–268. [Google Scholar]
- Veldman, K.; Cavaco, L.M.; Mevius, D.; Battisti, A.; Franco, A.; Botteldoorn, N.; Bruneau, M.; Perrin-Guyomard, A.; Cerny, T.; De Frutos Escobar, C.; et al. International collaborative study on the occurrence of plasmid-mediated quinolone resistance in Salmonella enterica and Escherichia coli isolated from animals, humans, food and the environment in 13 European countries. J. Antimicrob. Chemother. 2011, 66, 1278–1286. [Google Scholar] [CrossRef] [PubMed]
- Campos, J.; Cristino, L.; Peixe, L.; Antunes, P. MCR-1 in multidrug-resistant and copper-tolerant clinically relevant Salmonella 1,4,[5],12:i:- and S. Rissen clones in Portugal, 2011 to 2015. Eurosurveillance 2016, 21, 30270. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, R.; Card, R.M.; Nunez, J.; Pomba, C.; Mendonça, N.; Anjum, M.F.; Da Silva, G.J. Detection of an mcr-1-encoding plasmid mediating colistin resistance in Salmonella enterica from retail meat in Portugal. J. Antimicrob. Chemother. 2016, 71, 2338–2340. [Google Scholar] [CrossRef] [PubMed]
- Barco, L.; Ramon, E.; Cortini, E.; Longo, A.; Dalla Pozza, M.C.; Lettini, A.A.; Dionisi, A.M.; Olsen, J.E.; Ricci, A. Molecular Characterization of Salmonella enterica Serovar 4,[5],12:i:- DT193 ASSuT Strains from Two Outbreaks in Italy. Foodborne Pathog. Dis. 2014, 11, 138–144. [Google Scholar] [CrossRef]
- Gallati, C.; Stephan, R.; Hächler, H.; Malorny, B.; Schroeter, A.; Nüesch-Inderbinen, M. Characterization of Salmonella enterica Subsp. enterica Serovar 4,[5],12:i:- Clones Isolated from Human and Other Sources in Switzerland Between 2007 and 2011. Foodborne Pathog. Dis. 2013, 10, 549–554. [Google Scholar]
- Scientific Committee on Emerging and Newly Identified Health Risks (SCENIHR). Assessment of the Antibiotic Resistance Effects of Biocides. 2009. Available online: http://ec.europa.eu/health/ph_risk/committees/04_scenihr/docs/scenihr_o_021.pdf (accessed on 1 April 2018).
- Seiler, C.; Berendonk, T.U. Heavy metal driven co-selection of antibiotic resistance in soil and water bodies impacted by agriculture and aquaculture. Front. Microbiol. 2012, 3, 399. [Google Scholar] [CrossRef]
- Yu, Z.; Gunn, L.; Wall, P.; Fanning, S. Antimicrobial resistance and its association with tolerance to heavy metals in agriculture production. Food Microbiol. 2017, 64, 23–32. [Google Scholar] [CrossRef]
- De Jong, A.; Smet, A.; Ludwig, C.; Stephan, B.; De Graef, E.; Vanrobaeys, M.; Haesebrouck, F. Antimicrobial susceptibility of Salmonella isolates from healthy pigs and chickens (2008–2011). Vet. Microbiol. 2014, 171, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, I.; Barownick, W.; Helmuth, R.; Mendoza, M.C.; Rodicio, M.R.; Schroeter, A.; Guerra, B. Extended-spectrum {beta}-lactamases and AmpC {beta}-lactamases in ceftiofur-resistant Salmonella enterica isolates from food and livestock obtained in Germany during 2003–07. J. Antimicrob. Chemother. 2009, 64, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.-H.; Lin, X.-Y.; Xu, L.; Gu, X.-X.; Yang, L.; Li, W.; Ren, S.-Q.; Liu, Y.-H.; Zeng, Z.-L.; Jiang, H.-X. CTX-M-27 Producing Salmonella enterica Serotypes Typhimurium and Indiana Are Prevalent among Food-Producing Animals in China. Front. Microbiol. 2016, 7, 436. [Google Scholar] [CrossRef] [PubMed]
- O’Mahony, R.; Quinn, T.; Drudy, D.; Walsh, C.; Whyte, P.; Mattar, S.; Fanning, S. Antimicrobial resistance in nontyphoidal Salmonella from food sources in Colombia: Evidence for an unusual plasmid-localized class 1 integron in serotypes Typhimurium and Anatum. Microb. Drug Resist. 2006, 12, 269–277. [Google Scholar] [CrossRef]
- Zaidi, M.B.; Leon, V.; Canche, C.; Perez, C.; Zhao, S.; Hubert, S.K.; Abbott, J.; Blickenstaff, K.; McDermott, P.F. Rapid and widespread dissemination of multidrug-resistant blaCMY-2 Salmonella Typhimurium in Mexico. J. Antimicrob. Chemother. 2007, 60, 398–401. [Google Scholar] [CrossRef]
- Glenn, L.M.; Lindsey, R.L.; Folster, J.P.; Pecic, G.; Boerlin, P.; Gilmour, M.W.; Harbottle, H.; Zhao, S.; McDermott, P.F.; Fedorka-Cray, P.J.; et al. Antimicrobial resistance genes in multidrug-resistant Salmonella enterica isolated from animals, retail meats, and humans in the United States and Canada. Microb. Drug Resist. 2013, 19, 175–184. [Google Scholar] [CrossRef]
- Lee, K.-E.; Lim, S.-I.; Choi, H.-W.; Lim, S.-K.; Song, J.-Y.; An, D.-J. Plasmid-mediated AmpC β-lactamase (CMY-2) gene in Salmonella Typhimurium isolated from diarrheic pigs in South Korea. BMC Res. Notes 2014, 7, 329. [Google Scholar] [CrossRef]
- Lin, D.; Chen, K.; Chan, E.W.-C.; Chen, S. Increasing prevalence of ciprofloxacin-resistant food-borne Salmonella strains harboring multiple PMQR elements but not target gene mutations. Sci. Rep. 2015, 5, 14754. [Google Scholar] [CrossRef]
- Li, L.; Liao, X.; Yang, Y.; Sun, J.; Li, L.; Liu, B.; Yang, S.; Ma, J.; Li, X.; Zhang, Q.; et al. Spread of oqxAB in Salmonella enterica serotype Typhimurium predominantly by IncHI2 plasmids. J. Antimicrob. Chemother. 2013, 68, 2263–2268. [Google Scholar] [CrossRef]
- Quesada, A.; Ugarte-Ruiz, M.; Iglesias, M.R.; Porrero, M.C.; Martínez, R.; Florez-Cuadrado, D.; Campos, M.J.; García, M.; Píriz, S.; Sáez, J.L.; et al. Detection of plasmid mediated colistin resistance (MCR-1) in Escherichia coli and Salmonella enterica isolated from poultry and swine in Spain. Res. Vet. Sci. 2016, 105, 134–135. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Baloch, Z.; Zou, M.; Dong, Y.; Peng, Z.; Hu, Y.; Xu, J.; Yasmeen, N.; Li, F.; Fanning, S. Complete Genomic Analysis of a Salmonella enterica Serovar Typhimurium Isolate Cultured from Ready-to-Eat Pork in China Carrying One Large Plasmid Containing mcr-1. Front. Microbiol. 2018, 9, 616. [Google Scholar] [CrossRef] [PubMed]
- Campos, J.; Cristino, L.; Ribeiro, S.; Mourão, J.; Peixe, L.A.; Antunes, P. MCR-1 dispersion in clinically relevant clones of multidrug-resistant and copper-tolerant Salmonella from Portugal. In Proceedings of the 27th European Congress of Clinical Microbiology and Infectious Diseases (ECCMID), Vienna, Austria, 22–25 April 2017; Available online: https://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&ved=2ahUKEwjy75L79LjfAhVNSRUIHbiDC5gQFjAAegQIAhAC&url=https%3A%2F%2Fwww.escmid.org%2Fescmid_publications%2Fescmid_elibrary%2Fmaterial%2F%3Fmid%3D52654&usg=AOvVaw1Ywvlmi8Ktp9Vguytd3P_m (accessed on 1 February 2018).
- Anjum, M.F.; Duggett, N.A.; AbuOun, M.; Randall, L.; Nunez-Garcia, J.; Ellis, R.J.; Rogers, J.; Horton, R.; Brena, C.; Williamson, S.; et al. Colistin resistance in Salmonella and Escherichia coli isolates from a pig farm in Great Britain. J. Antimicrob. Chemother. 2016, 71, 2306–2313. [Google Scholar] [CrossRef] [PubMed]
- Cui, M.; Zhang, J.; Zhang, C.; Li, R.; Chan, E.W.-C.; Wu, C.; Wu, C.; Chen, S. Distinct mechanisms of acquisition of mcr-1–bearing plasmid by Salmonella strains recovered from animals and food samples. Sci. Rep. 2017, 7, 13199. [Google Scholar] [CrossRef]
- Freire Martín, I.; AbuOun, M.; Reichel, R.; La Ragione, R.M.; Woodward, M.J. Sequence analysis of a CTX-M-1 IncI1 plasmid found in Salmonella 4,5,12:i:-, Escherichia coli and Klebsiella pneumoniae on a UK pig farm. J. Antimicrob. Chemother. 2014, 69, 2098–2101. [Google Scholar] [CrossRef]
- Rodríguez, I.; Jahn, S.; Schroeter, A.; Malorny, B.; Helmuth, R.; Guerra, B. Extended-spectrum β-lactamases in German isolates belonging to the emerging monophasic Salmonella enterica subsp. enterica serovar Typhimurium 4,[5],12:i:- European clone. J. Antimicrob. Chemother. 2012, 67, 505–508. [Google Scholar]
- Clemente, L.; Manageiro, V.; Ferreira, E.; Jones-Dias, D.; Correia, I.; Themudo, P.; Albuquerque, T.; Caniça, M. Occurrence of extended-spectrum β-lactamases among isolates of Salmonella enterica subsp. enterica from food-producing animals and food products, in Portugal. Int. J. Food Microbiol. 2013, 167, 221–228. [Google Scholar] [CrossRef]
- Tyson, G.H.; Tate, H.P.; Zhao, S.; Li, C.; Dessai, U.; Simmons, M.; McDermott, P.F. Identification of Plasmid-Mediated Quinolone Resistance in Salmonella Isolated from Swine Ceca and Retail Pork Chops in the United States. Antimicrob. Agents Chemother. 2017, 61, e01318-17. [Google Scholar] [CrossRef]
- Rebelo, A.R.; Bortolaia, V.; Kjeldgaard, J.S.; Pedersen, S.K.; Leekitcharoenphon, P.; Hansen, I.M.; Guerra, B.; Malorny, B.; Borowiak, M.; Hammerl, J.A.; et al. Multiplex PCR for detection of plasmid-mediated colistin resistance determinants, mcr-1, mcr-2, mcr-3, mcr-4 and mcr-5 for surveillance purposes. Eurosurveillance 2018, 23. [Google Scholar] [CrossRef]
- Garcia-Graells, C.; De Keersmaecker, S.C.J.; Vanneste, K.; Pochet, B.; Vermeersch, K.; Roosens, N.; Dierick, K.; Botteldoorn, N. Detection of Plasmid-Mediated Colistin Resistance, mcr-1 and mcr-2 Genes, in Salmonella spp. Isolated from Food at Retail in Belgium from 2012 to 2015. Foodborne Pathog. Dis. 2018, 15. [Google Scholar] [CrossRef]
- Carattoli, A.; Villa, L.; Feudi, C.; Curcio, L.; Orsini, S.; Luppi, A.; Pezzotti, G.; Magistrali, C.F. Novel plasmid-mediated colistin resistance mcr-4 gene in Salmonella and Escherichia coli, Italy 2013, Spain and Belgium, 2015 to 2016. Eurosurveillance 2017, 22, 30589. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.H.Y.; Chen, S. First detection of oqxAB in Salmonella spp. isolated from food. Antimicrob. Agents Chemother. 2013, 57, 658–660. [Google Scholar] [CrossRef] [PubMed]
- Webb, H.E.; Granier, S.A.; Marault, M.; Millemann, Y.; den Bakker, H.C.; Nightingale, K.K.; Bugarel, M.; Ison, S.A.; Scott, H.M.; Loneragan, G.H. Dissemination of the mcr-1 colistin resistance gene. Lancet. Infect. Dis. 2016, 16, 144–145. [Google Scholar] [CrossRef]
- Riaño, I.; Moreno, M.A.; Teshager, T.; Sáenz, Y.; Domínguez, L.; Torres, C. Detection and characterization of extended-spectrum beta-lactamases in Salmonella enterica strains of healthy food animals in Spain. J. Antimicrob. Chemother. 2006, 58, 844–847. [Google Scholar] [CrossRef]
- Luk-In, S.; Pulsrikarn, C.; Bangtrakulnonth, A.; Chatsuwan, T.; Kulwichit, W. Occurrence of a novel class 1 integron harboring qnrVC4 in Salmonella Rissen. Diagn. Microbiol. Infect. Dis. 2017, 88, 282–286. [Google Scholar] [CrossRef]
- Keelara, S.; Thakur, S. Dissemination of plasmid-encoded AmpC β-lactamases in antimicrobial resistant Salmonella serotypes originating from humans, pigs and the swine environment. Vet. Microbiol. 2014, 173, 76–83. [Google Scholar] [CrossRef] [PubMed]
- El Garch, F.; de Jong, A.; Bertrand, X.; Hocquet, D.; Sauget, M. mcr-1- like detection in commensal Escherichia coli and Salmonella spp. from food-producing animals at slaughter in Europe. Vet. Microbiol. 2018, 213, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Wasyl, D.; Hoszowski, A.; Zając, M. Prevalence and characterisation of quinolone resistance mechanisms in Salmonella spp. Vet. Microbiol. 2014, 171, 307–314. [Google Scholar] [CrossRef]
- Winokur, P.L.; Brueggemann, A.; De Salvo, D.L.; Hoffmann, L.; Apley, M.D.; Uhlenhopp, E.K.; Pfaller, M.A.; Doern, G.V. Animal and human multidrug-resistant, cephalosporin-resistant Salmonella isolates expressing a plasmid-mediated CMY-2 AmpC beta-lactamase. Antimicrob. Agents Chemother. 2000, 44, 2777–2783. [Google Scholar] [CrossRef]
- Andrysiak, A.K.; Olson, A.B.; Tracz, D.M.; Dore, K.; Irwin, R.; Ng, L.-K.; Gilmour, M.W.; Canadian Integrated Program for Antimicrobial Resistance Surveillance Collaborative. Genetic characterization of clinical and agri-food isolates of multi drug resistant Salmonella enterica serovar Heidelberg from Canada. BMC Microbiol. 2008, 8, 89. [Google Scholar] [CrossRef]
- Jiang, H.-X.; Song, L.; Liu, J.; Zhang, X.-H.; Ren, Y.-N.; Zhang, W.-H.; Zhang, J.-Y.; Liu, Y.-H.; Webber, M.A.; Ogbolu, D.O.; et al. Multiple transmissible genes encoding fluoroquinolone and third-generation cephalosporin resistance co-located in non-typhoidal Salmonella isolated from food-producing animals in China. Int. J. Antimicrob. Agents 2014, 43, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Fischer, J.; Rodríguez, I.; Schmoger, S.; Friese, A.; Roesler, U.; Helmuth, R.; Guerra, B. Salmonella enterica subsp. enterica producing VIM-1 carbapenemase isolated from livestock farms. J. Antimicrob. Chemother. 2013, 68, 478–480. [Google Scholar] [CrossRef] [PubMed]
- Clemente, L.; Manageiro, V.; Jones-Dias, D.; Correia, I.; Themudo, P.; Albuquerque, T.; Geraldes, M.; Matos, F.; Almendra, C.; Ferreira, E.; et al. Antimicrobial susceptibility and oxymino-β-lactam resistance mechanisms in Salmonella enterica and Escherichia coli isolates from different animal sources. Res. Microbiol. 2015, 166, 574–583. [Google Scholar] [CrossRef] [PubMed]
- Kylla, H.; Kumar Dutta, T.; Roychoudhury, P.; Subudhi, P.K. Salmonella enterica serovar Miami Possessing Both Virulence and Extended-Spectrum β-Lactamase Resistant Genes Isolated from Diarrhoeic Piglets of North East India (Mizoram). Int. J. Curr. Microbiol. Appl. Sci. 2018, 7, 1451–1456. [Google Scholar] [CrossRef]
| Serotype | Country(ies) 2 | Source | Year(s) | No. of 3 | Reference | |
|---|---|---|---|---|---|---|
| (Subtyping features/molecular markers, when available) 1 | Cases | Death(s) | ||||
| Typhimurium | ||||||
| (DT104A) | Italy | Pork salami | 2004 | 63 | 0 | [91] |
| (DT12) | Denmark | Pork products | 2005 | 26 | 0 | [92] |
| (MLVA-type 3-12-4-13-2) | Denmark, Norway, Sweden | Danish pork meat/minced meat | 2008 | 37/10/4 | 4/0/0 | [93] |
| (DT193; MLVA-type 3-14-12-NA-211) | Denmark | Pork salami | 2010 | 20 | NS | [94] |
| (DT120; MLVA-type 3-11-14-NA-211) | Denmark | Imported smoked pork tenderloin | 2011 | 22 | 0 | [95] |
| (DT193) | Spain | Dried pork sausage | 2011 | 8 | 0 | [96] |
| Australia | Barbecued pork | 2010 | 4 | NS | [97] | |
| Australia | Pork spit roast | 2011 | 5 | NS | [97] | |
| Australia | Cooked pork hock | 2014 | 4 | NS | [97] | |
| Australia | Pork dish | 2015 | 10 | NS | [97] | |
| (ST19) | Denmark 4 | Spanish salami | 2017 | NS | NS | [98] |
| Sweden | Spanish salami | 2017 | NS | NS | [98] | |
| 1,4,[5],12:i:- | ||||||
| (DT193; PFGE-type STYMXB.0131; ASSuT) | Luxemburg | Pork meat | 2006 | 133 | 1 | [99] |
| (MLVA-Type 3-13-15-NA-211) | France | Dried pork sausage | 2010 | 69 | 0 | [100] |
| (PFGE-type XTYM-159; MLVA-type 3-13-9-NA-211; ASSuT) | France | Dried pork sausage | 2011 | 337 | 0 | [101] |
| (PFGE-type STYMXB.0131/ STYMXB.0083; MLVA-type 3-13-9-NA-211; ASSuT) | Italy | Pork salami | 2012–2015 | NS | NS | [102] |
| (DT138; PFGE-type XbaI.0005/ STYMXB.0083; BlnI.00X; ASSuT) | Spain | Pork chorizo | 2014 | 6 | 0 | [103] |
| (DT138) | Spain | Dried pork sausage | 2011 | 38 | 0 | [96] |
| USA | Pork meat | 2015 | 188 | 0 | [60] | |
| Sweden 4 | Italian chilled truffle salami | 2018 | NS | NS | [98] | |
| Derby | ||||||
| Spain | Dried pork sausage | 2011 | 3 | 0 | [96] | |
| (PT53; ST682) | Germany | Raw fermented pork | 2013–2014 | 145 | 0 | [104] |
| Rissen | ||||||
| (PFGE-type TEEX01.0017.DK) | Denmark 4 | Imported pork products | 2000–2005 | NS | NS | [27] |
| Bovismorbificans | ||||||
| (PT24) | Germany | Pork minced meat | 2004–2005 | 525 | 1 | [105] |
| (ST142) | The Netherlands, Belgium, France | Pork ham products | 2016-2017 | 54/NS/NS | 0/NS/NS | [106] |
| Give | Germany | Minced pork meat | 2004 | 115 | 1 | [107] |
| Goldcoast | ||||||
| (PFGE-type SCG Xba3) | Hungary | Pork | 2009–2010 | 44 | 0 | [108] |
| Italy | Pork salami | 2009–2010 | 79 | 0 | [109] | |
| Infantis | ||||||
| (PFGE-type EPI-type) | Denmark | Pork meat | 1992–1993 | >500 | 0 | [110] |
| (ST32) | Italy | Pork meat—porchetta | 2011 | 23 | 0 | [111] |
| (PT29; PFGE-type XB27) | Germany | Raw pork | 2013 | 267 | 0 | [112] |
| Australia | Pork rools | 2013 | 2 | NS | [61] | |
| USA | Pork meat | 2015 | 5 | 0 | [60] | |
| Manhattan | France | Pork products | 2005–2006 | 69 | 0 | [113] |
| Muenchen | ||||||
| Germany | Raw pork | 2013 | 203 | 0 | [114] | |
| Germany | Raw pork | 2014 | 247 | 0 | [114] | |
| Ohio | ||||||
| (PFGE-type BlnI-A) | Belgium | Pork meat | 2005 | 6o | 0 | [115] |
| Serotype | Clinically Relevant Gene(s) 1 (no. isolates) | Source | Human Concomitant Presence 2 | Country/year(s) | Antibiotic Resistance Phenotype 3 | Gene Location (PL-Inc Group/Chr) | Reference |
|---|---|---|---|---|---|---|---|
| Typhimurium | blaTEM-52 (n = 2) | Pigs | Belgium/2009 | NR | NR | [166] | |
| blaCTX-M-1 (n = 1) | Pork meat | Germany/2007 | K-N-S-Su-T-Tm-Sxt | PL-N | [167] | ||
| blaCTX-M-14+blaSHV-1+oqxAB+ aac(6′)-Ib-cr (n = 2) | Pigs | Yes | China/2014 | NS | PL-FIB, N | [168] | |
| blaCTX-M-27+blaSHV-1+ oqxAB+ aac(6′)-Ib-cr (n = 1) | Pig | China/2014 | NS | PL-P | [168] | ||
| blaCTX-M-27+qnrB+oqxAB+ aac(6′)-Ib-cr (n = 1) | Pig | China/2014 | NS | PL-NT | [168] | ||
| blaCTX-M-55+qnrA+qnrB (n = 4) | Pigs | China/2016 | A-(Cp-E-F-L-Na-N-T) | NR | [58] | ||
| blaCTX-M-65+oqxAB (n = 1) | Pig | China/2014 | NS | PL-FIB | [168] | ||
| blaCMY-2 (n = 52) | Pork | Colombia/UN | A-T | NR | [169] | ||
| Pork and pig | Yes | Mexico/2002–2005 | C-S-Su-T-(G-K-Na-Sxt) | NR | [170] | ||
| Pigs | Mexico/NR | A-Caz-Ctx-Fox-T-(G) | NR | [46] | |||
| Pigs | USA/2007 | A-C-Cro-Fox-G-K-S-Su-Sxt-T-Ti | PL-A/C, FIB, I1 | [171] | |||
| Pigs | Belgium/2009 | NR | NR | [166] | |||
| Diarrheic pigs | South Korea/2011–2012 | A-Cef-C-F-Fox-G-Na-Sxt-T-Ti | PL-A/C, FIB | [172] | |||
| qnrS1+oqxAB+aac(6′)-Ib-cr (n = 4) | Pork | China/2012–2013 | A-C-Cp-G-K-Na-O-S-Su-T | NR | [173] | ||
| oqxAB (n = 3) | Pigs | China/2010 | A-C-F-Na-O-Su-T-(G) | PL-F | [174] | ||
| Pork | China/2012–2013 | A-C-Cp-K-Na-O-S-Su-T-(G) | NR | [173] | |||
| oqxAB+aac(6′)-Ib-cr (n = 6) | Pigs | China/2008–2010 | A-Na-O-Sxt-T-(C-Cp-F-G) | PL-HI2 | [174] | ||
| Pork | China/2012 | A-C-Cp-G-K-Na-O-S-Su-T | NR | [173] | |||
| mcr-1 (n = 13) | Pigs | Spain/2009–2011 | Col | PL-NR | [175] | ||
| Ready-to-eat pork | China/2014 | C-Cp-Col-F-G-Na-S-Sxt-T | PL-HI2 | [176] | |||
| Pork carcass/ Pork product | Portugal/2016 | A-Col-S-Su-T-Tp-(C-Cp-Fox) | PL-HI2 | [177] | |||
| Pig | Yes | Great Britain/NR | A-C-Col-Fox-Su-T-Tm-Tyg | PL-I2 | [178] | ||
| mcr-1+blaCTX-M-1 (n = 1) | Pig | Portugal/2011 | A-At-C-Caz-Col-Ctx-Fep-Fox-G-T-Tob | PL-HI2 | [160] | ||
| mcr-1+blaCTX-M-14 (n = 2) | Pork | China/2015 | A-C-Caz- Col-Ctx-Fos-G-Su-T | PL-HI2 | [179] | ||
| mcr-1+oqxAB (n = 17) | Pigs | China/2013–2014 | Cp-Col-F-O-Sxt-T-(A-G-S) | PL-HI2-F4:A-:B5 4 | [150] | ||
| mcr-1+oqxAB+aac(6′)-Ib-cr (n = 4) | Pigs | China/2008–2009 | A-Col-F-G-Na-O-S-Sxt-T-(Cp) | PL-I2, HI2 | [149] | ||
| mcr-1+aac(6′)-Ib-cr (n = 1) | Pig | China/2008–2009 | A-Col-F-G-Na-O-S-Sxt-T | PL-I2 | [149] | ||
| Pigs | China/2015 | A-Cp-Col-G-(C-Na-Sxt) | PL-HI2 | [179] | |||
| 1,4,[5],12:i:- | blaCTX-M-1 (n = 15) | Pigs | UK/2009 | A-Ctx-Su-(C) | PL-I1-γ | [180] | |
| Pigs | Germany/2007, 2009–2010 | A-At-Cro-Ctx-Cef-Cxm-P-Tc-Ti-(Fep-S) | PL-N, I1 | [181] | |||
| blaCTX-M-14 (n = 1) | Pork | Portugal/2010 | A-C-Ctx-T-Tm | NR | [182] | ||
| blaCTX-M-15+blaSHV-12 (n = 1) | Pork | Portugal/2011 | A-C-Ctx-S-Su | NR | [182] | ||
| blaCTX-M-32 (n = 1) | Pork | Portugal/2011 | A-C-Ctx-G-S-Su-T | NR | [182] | ||
| qnrB19 (n = 1) | Pig | USA/2014 | Cp | PL-NR | [183] | ||
| mcr-1 (n = 24) | Pigs | Yes | Italy/2012–2015 | A-Col-S-T-(C-Cp-F) | NR | [75] | |
| Pork | Yes | Italy/2013–2015 | A-Col-S-T-(C-F) | NR | [75] | ||
| Pork carcass/ Pork meat | Yes | Portugal/2014–2015 | A-Col-S-Su-T-(C-Cp-Tm) | PL-X4, HI2 | [159] | ||
| Slaughterhouse/ Pork sausage | Portugal/2015–2016 | A-Col-S-Su-T-(Cp-Tm) | PL-X4, HI2 | [177] | |||
| Pork carcass | France/2016 | Col | PL-NR | [184] | |||
| Pork carcass | Belgium/2012 | A-Col-S-Su-T | PL-X4 | [185] | |||
| mcr-4 (n = 1) | Pig | Italy/2014 | A-Col-S-Su-T | PL-ColE | [186] | ||
| Derby | blaCTX-M-1 (n = 3) | Pigs | Belgium/2009 | NR | NR | [166] | |
| Pork sector | France/2014 | A-Cef-Ctx-Caz | NR | [125] | |||
| qnrA (n = 2) | Pigs | China/2016 | A | NR | [58] | ||
| qnrB19 (n = 3) | Pigs | USA/2014 | Cp | PL-NR | [183] | ||
| Pork chops | USA/2014–2015 | Cp | PL-NR | [183] | |||
| qnrB+qnrS1+oqxAB (n = 1) | Pork | China/2013 | A-C-Cp-G-K-Na-O-S-Su-T-(Az) | NR | [173] | ||
| qnB8+qnrS2+oqxAB+ aac(6′)-Ib-cr (n = 1) | Pork | China/2012 | A-C-Cp-G-K-Na-O-S-Su-T | NR | [173] | ||
| qnrS2 (n = 1) | Pork chops | USA/2014 | Cp | NR | [183] | ||
| qnrS2+oqxAB+aac(6′)-Ib-cr (n = 11) | Pork | China/2012–2013 | A-C-Cp-G-K-Na-O-S-Su-T-(Az) | NR | [173] | ||
| oqxAB (n = 3) | Pork | China/NR | C-Na-O-T | Chr | [187] | ||
| Pork | China/2013 | A-C-Cp-G-K-Na-OLA-S-Su-T | NR | [173] | |||
| mcr-1 (n = 13) | Pigs | Italy/2012–2015 | C-Col-S-Su-Sxt-T | NR | [75] | ||
| Pork sausage | France/2013 | Col-(A-S-T) | PL-P | [188] | |||
| Pork | China/2015 | A-C-Col-T | PL-X4 | [179] | |||
| mcr-2 (n = 1) | Pork carcass | Belgium/2012 | C-Col-Su-Tm | PL-X4 | [185] | ||
| Rissen | blaCTX-M-1 (n = 1) | Pig | Belgium/2009 | NR | NR | [166] | |
| blaCTX-M-55 (n = 1) | Pork carcass | Thailand/2014–2015 | A-C-Caz-Ctx-Cpd-G-S-Su-T | PL-NR | [50] | ||
| blaSHV-12 (n = 1) | Pig | Spain/1999 | A-At-Caz-Cef-Ctx-S-Su-T | NR | [189] | ||
| qnrB19 (n = 1) | Pig | USA/2013 | Cp | PL-NR | [183] | ||
| qnrS1 (n = 1) | Pig | Korea/2012–2013 | Cp-Na | NR | [57] | ||
| qnrVC4 (n = 1) | Pig | Thailand/2007 | A-C-Cp-Na-S | PL-Q1 | [190] | ||
| oqxAB (n = 1) | Pork | China/2013 | A-C-Cp-K-Na-O-S-Su-T | NR | [173] | ||
| mcr-1 (n = 3) | Pig | Spain/2009–2011 | Col | PL-NR | [175] | ||
| Pork carcass | Portugal/2014–2015 | A-C-Col-S-Su-Tm-(T) | PL-X4 | [159] | |||
| Anatum | blaCMY-2 (n = 4) | Pigs | USA/2008-2011 | A-Cro-Fox-Su-T-Ti | PL-I1-γ | [191] | |
| qnrB19 (n = 5) | Pigs | USA/2014 | Cp | PL-NR | [183] | ||
| mcr-1+blaCMY-2 (n = 3) | Pigs | Taiwan/2013 | A-C-Caz-Cp-Col-Ctx-Fox-Na-S-Su-Sxt-T | PL-NR | [142] | ||
| Adelaide | qnrB19 (n = 1) | Pig | USA/2014 | Cp | PL-NR | [183] | |
| Bovismorbificans | blaCTX-M-1 (n = 1) | Pig | UK/2009 | A-Ctx-Su | PL-I1-γ | [180] | |
| mcr-1 (n = 1) | Pork | Yes | Italy/2013-2015 | A-C-Cp-Col-S-T | NR | [75] | |
| Brandenburg | qnrB19 (n = 1) | Pigs | USA/2014 | Cp | PL-NR | [183] | |
| Concord | qnrB2 (n = 1) | Pig | Czech Republic/NR | NR | NR | [158] | |
| Dublin | mcr-1-like (n = 1) | Pig | France/NS | NS | NR | [192] | |
| Enteritidis | blaCTX-M-15 (n = 2) | Pigs | Korea/2012-2013 | A-Cep-G-Na-N-S-T-Ti | PL-HI2 | [57] | |
| blaCTX-M-55+qnrA+qnrB (n = 2) | Pigs | China/2016 | A-(Cp-F-Fos-Na-T-Ti) | NR | [58] | ||
| qnrA+qnrB (n = 1) | Pig | China/2016 | A-Cp-F-Na | NR | [58] | ||
| qnrS1/S3 (n = 1) | Pig | Poland/2008 | A-Cp | NR | [193] | ||
| Give | blaCMY-2 (n = 1) | Pig | USA/1998-1999 | A-At-C-Caz-Ctx-Fox-G-P-S-Su-T-Tc-Ti | PL-NR | [194] | |
| Goldcoast | qnrS1 (n = 1) | Pig | Belgium/NR | NR | NR | [158] | |
| Heidelberg | blaCMY-2 (n = 3) | Pigs | Canada/2004 | A-Cef-Fox-Ti | PL-NR | [195] | |
| Pigs | USA/1998-1999 | A-At-C-Caz-Ctx-Fox-G-P-S-Su-Sxt-T-Tc-Ti | PL-NR | [194] | |||
| mcr-1+oqxAB (n = 1) | Pig | China/2013-2014 | Cp-Col-F-O-S-Sxt | PL-HI2-F4:A-:B5 4 | [150] | ||
| Hinsingen | qnrD+aac(6′)-Ib-cr (n = 1) | Pig | China/2009-2010 | Cp-Na | PL-NR | [196] | |
| Indiana | blaCTX-M-27+blaSHV-1+oqxAB (n = 2) | Pigs | China/2014 | NS | PL-N, P | [168] | |
| blaCTX-M-65+blaSHV-1+oqxAB+ aac(6′)-Ib-cr (n = 1) | Pig | China/2014 | NS | PL-NT | [168] | ||
| blaCTX-M-65+oqxAB+ aac(6′)-Ib-cr (n = 7) | Pig | China/2011 | A-C-Caz-Cp-Ctx-G-Na-Sxt-T | PL-NR | [144] | ||
| Pork | China/2012 | A-Az-C-Cp-Cro-G-K-Na-O-S-Su-T | NR | [173] | |||
| Pig | China/2014 | NS | PL-N | [168] | |||
| qnrA+oqxA+aac(6′)-Ib-cr (n = 1) | Pig | China/2009-2010 | Cp-O-Na | PL-NR | [196] | ||
| oqxA (n = 2) | Pig | China/2009-2010 | Cp-O-Na | PL-NR | [196] | ||
| aac(6′)-Ib-cr (n = 1) | Pig | China/2009-2010 | Cp-Na | PL-NR | [196] | ||
| Infantis | blaCMY-2 (n = 2) | Pigs | Japan/2007-2008 | A-C-Caz-Cef- Fox-S-Su-T | PL-NR | [59] | |
| blaVIM-1+blaAAC-1 (n = 4) | Pigs | Germany/2011 | A-C-Caz-Cef-CRro-Ctx-Cxm-Fep-Fox-P-S-Su-Tc-Ti-Tm | PL-HI2 | [197] | ||
| Kedougou | mcr-4.1 (n = 1) | Pig carcass | Spain/2016 | Col | PL-NR | [184] | |
| London | blaCTX-M-14 (n = 1) | Pork | Portugal/2012-2013 | A-Ctx | NR | [198] | |
| qnrB19 (n = 1) | Pig | USA/2014 | Cp | PL-NR | [183] | ||
| mcr-1+oqxAB (n = 1) | Pig | China/2013-2014 | A-Cp-Col-F-G-O-S-Sxt | PL-HI2-F4:A-:B5 4 | [150] | ||
| Miami | blaCMY-2 (n = 2) | Diarrheic piglets | India/2014 | A-Cfl-Ctx-E-Fix-P | NR | [199] | |
| Muenchen | qnrB19 (n = 8) | Pigs | USA/2013-2014 | Cp | PL-NR | [183] | |
| Newport | mcr-1+blaTEM-135 (n = 5) | Pigs | China/2015 | A-C-Cp- Col-G | PL-HI2 | [179] | |
| Senftenberg | qnrB6+aac(6′)-Ib-cr (n = 1) | Pig | USA/2013 | Cp | NR | [183] | |
| Virchow | blaCTX-M-15 (n = 5) | Pigs | Yes | Korea/2012-2013 | A-Cef-G-Na-N-S-T-Ti | PL-HI2 | [57] |
| Weltevreden | mcr-1 (n = 1) | Pork | China/2015 | Col-T | PL-X4 | [179] | |
| Salmonellaspp. | mcr-1 (n = 3) | Pigs | Italy/2010-2011 | NR | NR | [192] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Campos, J.; Mourão, J.; Peixe, L.; Antunes, P. Non-typhoidal Salmonella in the Pig Production Chain: A Comprehensive Analysis of Its Impact on Human Health. Pathogens 2019, 8, 19. https://doi.org/10.3390/pathogens8010019
Campos J, Mourão J, Peixe L, Antunes P. Non-typhoidal Salmonella in the Pig Production Chain: A Comprehensive Analysis of Its Impact on Human Health. Pathogens. 2019; 8(1):19. https://doi.org/10.3390/pathogens8010019
Chicago/Turabian StyleCampos, Joana, Joana Mourão, Luísa Peixe, and Patrícia Antunes. 2019. "Non-typhoidal Salmonella in the Pig Production Chain: A Comprehensive Analysis of Its Impact on Human Health" Pathogens 8, no. 1: 19. https://doi.org/10.3390/pathogens8010019
APA StyleCampos, J., Mourão, J., Peixe, L., & Antunes, P. (2019). Non-typhoidal Salmonella in the Pig Production Chain: A Comprehensive Analysis of Its Impact on Human Health. Pathogens, 8(1), 19. https://doi.org/10.3390/pathogens8010019






