Antibacterial Activities of Herbal Toothpastes Combined with Essential Oils against Streptococcus mutans
Abstract
1. Introduction
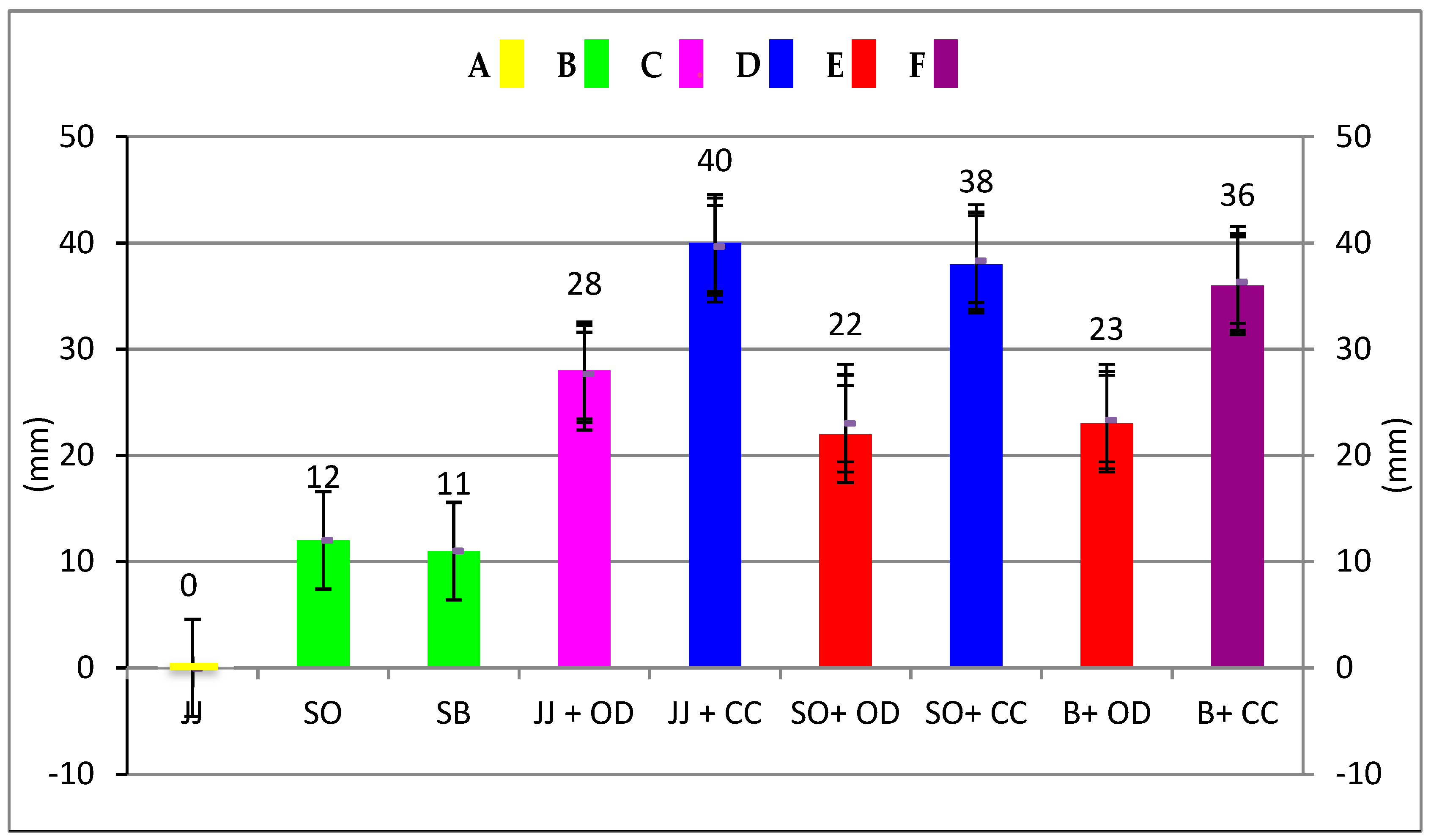
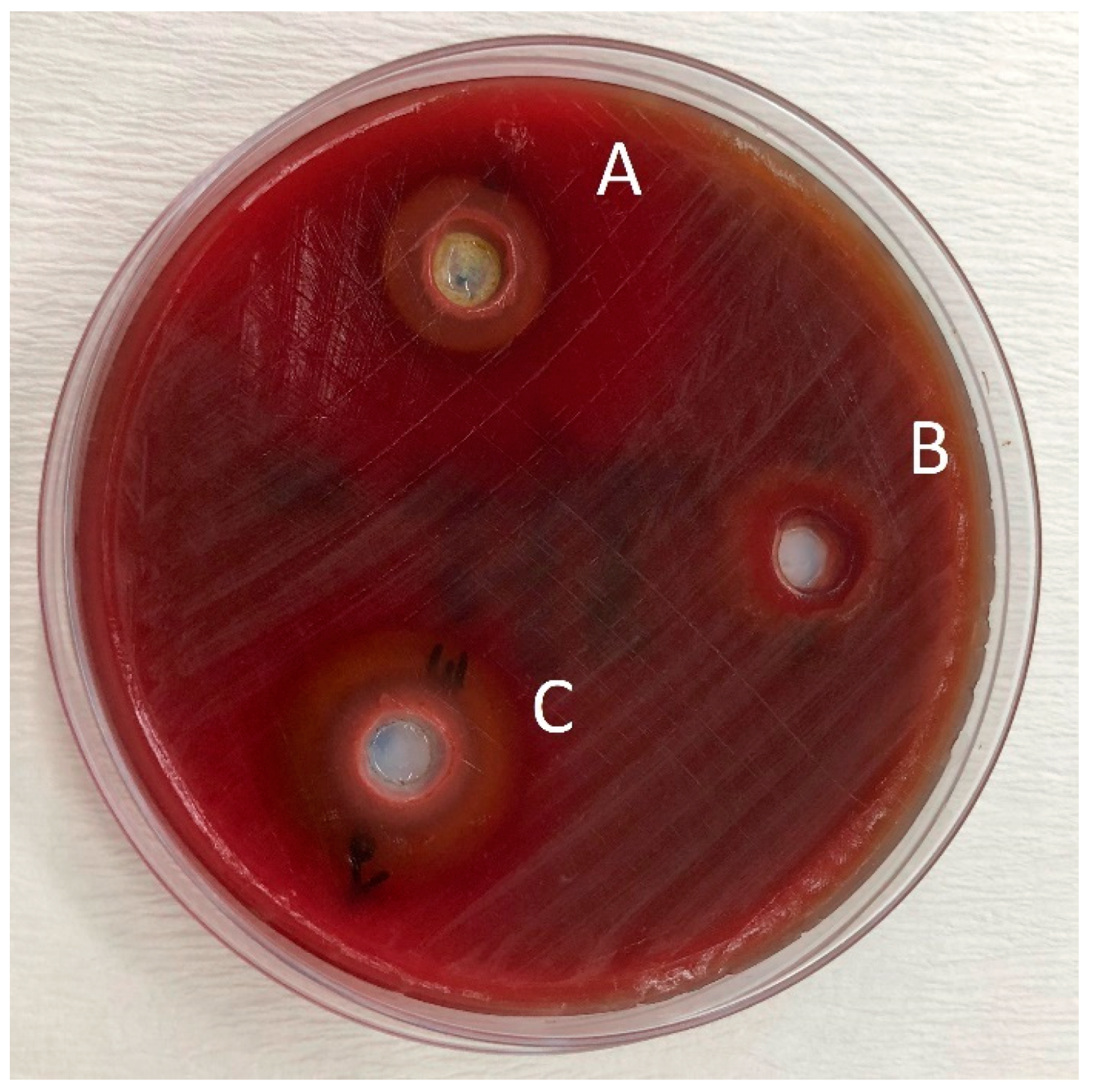
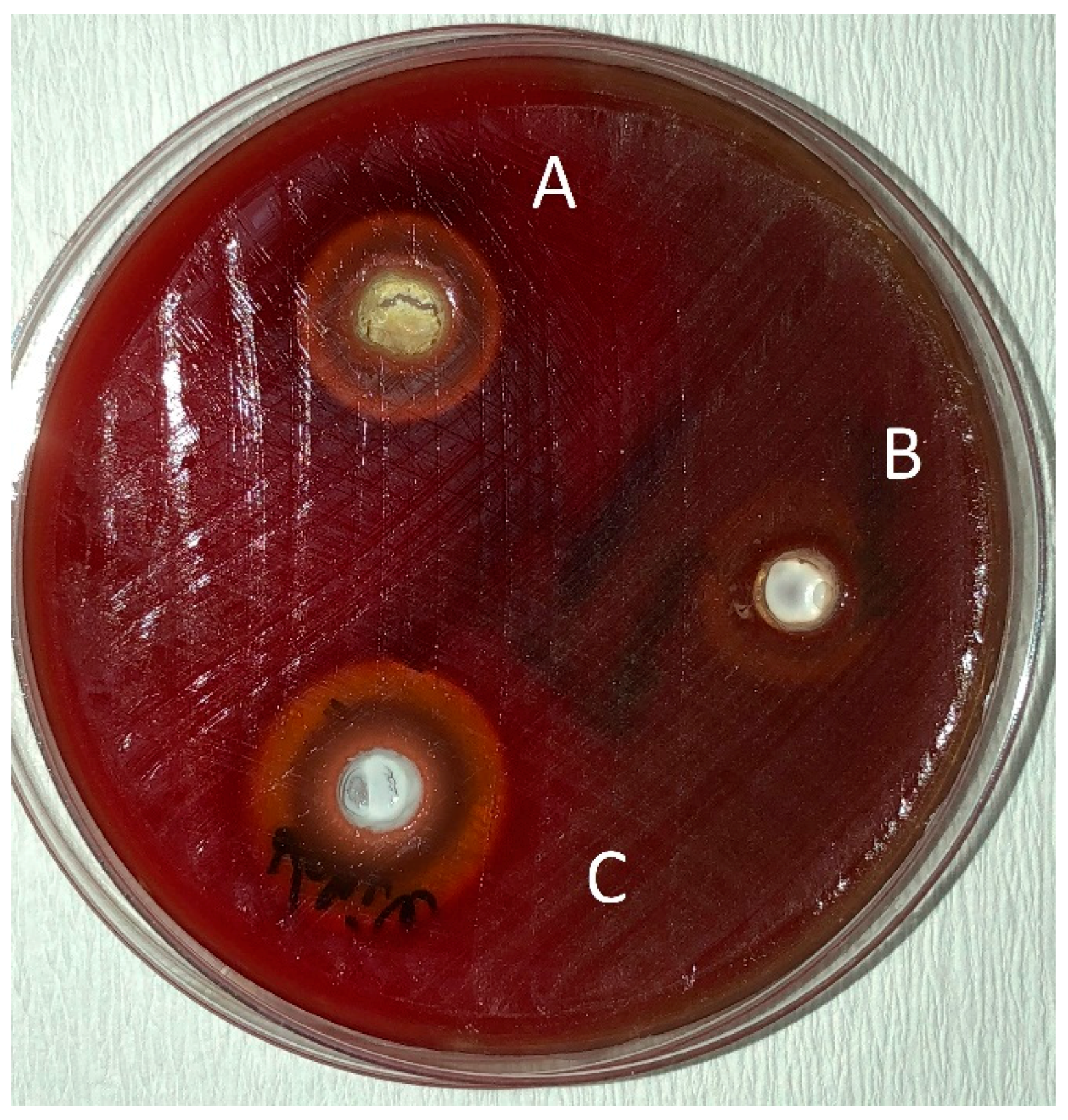
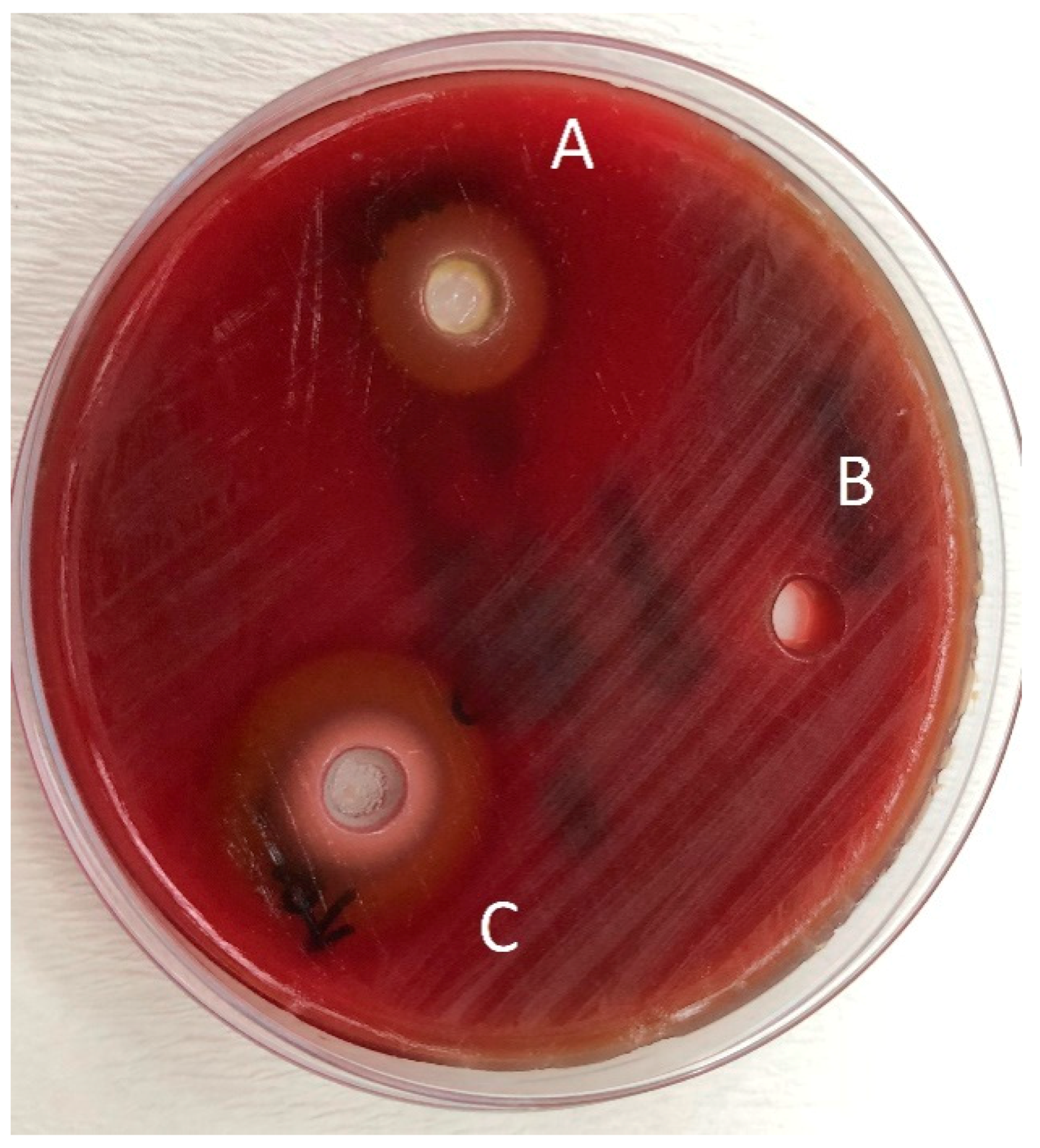
2. Results
3. Discussion
4. Materials and Methods
4.1. Obtaining Essential Oils
4.2. GC/MS Analysis of Essential Oils
4.2.1. GC/MS Analysis
4.2.2. GC Analysis
Determination of the Antibacterial Effect of Oils
4.3. Determination of the Antibacterial Effect of Toothpastes
4.4. Mixing the Herbal Toothpastes with Essential Oils and Determination of the Antibacterial Effect of Toothpastes Combined with Essential Oils
4.5. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Petersen, P.E. The World Oral Health Report 2003: Continuous improvement of oral health in the 21st century—The approach of the WHO Global Oral Health Programme. Community Dent. Oral Epidemiol. 2003, 31, 3–24. [Google Scholar] [CrossRef] [PubMed]
- Zero, D.T. Dental Caries process. Dent. Clin. N. Am. 1999, 43, 635–664. [Google Scholar] [PubMed]
- Selwitz, R.H.; Ismail, A.I.; Pitts, N.B. Dental caries. Lancet 2007, 369, 51–59. [Google Scholar] [CrossRef]
- Van Houte, J. Role of micro-organisms in caries etiology. J. Dent. Res. 1994, 73, 672–681. [Google Scholar] [CrossRef] [PubMed]
- Ritz, H.L. Microbial population shifts in developing human dental plaque. Arch. Oral Biol. 1967, 12, 1561–1568. [Google Scholar] [CrossRef]
- Gazzaneo, L.R.S.; De Lucena, R.F.P.; de Albuquerque, U.P. Knowledge and use of medicinal plants by local specialists in an region of Atlantic Forest in the state of Pernambuco (Northeastern Brazil). J. Ethnobiol. Ethnomed. 2005, 1, 9. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, P.; Das, J.; Foley, I. Biofilm susceptibility to antimicrobials. Adv. Dent. Res. 1997, 11, 160–167. [Google Scholar] [CrossRef]
- Projan, S.J.; Youngman, P.J. Antimicrobials: New solutions badly needed. Curr. Opin. Microbiol. 2002, 5, 463–465. [Google Scholar] [CrossRef]
- Mandel, I.D. Chemotherapeutic agents for controlling plaque and gingivitis. J. Clin. Periodontol. 1988, 15, 488–498. [Google Scholar] [CrossRef]
- Baehni, P.C.; Takeuchi, Y. Anti-plaque agents in the prevention of biofilm-associated oral diseases. Oral Dis. 2003, 9, 23–29. [Google Scholar] [CrossRef]
- Marinho, V.C.; Higgins, J.; Logan, S.; Sheiham, A. Fluoride toothpastes for preventing dental caries in children and adolescents. Cochrane Database Syst. Rev. 2003, CD002278. [Google Scholar] [CrossRef] [PubMed]
- Al-Quran, S. Ethnobotanical survey of folk toxic plants in southern part of Jordan. Toxicon 2005, 46, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Knoll-Köhler, E.; Stiebel, J. Amine fluoride gel affects the viability and the generation of superoxide anions in human polymorphonuclear leukocytes: An in vitro study. Eur. J. Oral Sci. 2002, 110, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Neumegen, R.A.; Fernández-Alba, A.R.; Chisti, Y. Toxicities of triclosan, phenol, and copper sulfate in activated sludge. Environ. Toxicol. 2005, 20, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, F.; Lehmann, M.; doAmaral, V.S.; Reguly, M.L.; de Andrade, H.H.R. Genotoxicity of three mouthwash products, Cepacol®, Periogard®, and Plax®, in the Drosophila wing-spot test. Environ. Mol. Mutagen. 2007, 48, 644–649. [Google Scholar] [CrossRef] [PubMed]
- McCullough, M.J.; Farah, C.S. The role of alcohol in oral carcinogenesis with particular reference to alcohol-containing mouthwashes. Aust. Dent. J. 2008, 53, 302–305. [Google Scholar] [CrossRef] [PubMed]
- Palombo, E.A. Traditional medicinal plant extracts and natural products with activity against oral bacteria: Potential application in the prevention and treatment of oral diseases. J.Evid. Based Complement. Altern. Med. 2011, 2011, 1–15. [Google Scholar] [CrossRef]
- Prabu, G.R.; Gnanamani, A.; Sadulla, S. Guaijaverin—A plant flavonoid as potential antiplaque agent against Streptococcus mutans. J. Appl. Microbiol. 2006, 101, 487–495. [Google Scholar] [CrossRef]
- Chitme, H.R.; Chandra, R.; Kaushik, S. Studies on anti-diarrhoeal activity of Calotropis giganteaRBR in experimental animals. J. Pharm. Pharm. Sci. 2004, 7, 70–75. [Google Scholar]
- Steinberg, D.; Feldman, M.; Ofek, I.; Weiss, E. Effect of a high-molecular-weight component of cranberry on constituents of dental biofilm. J. Antimicrob. Chemother. 2004, 54, 86–89. [Google Scholar] [CrossRef]
- Balunas, M.J.; Kinghorn, A.D. Drug discovery from medicinal plants. Life Sci. 2005, 78, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Barnes, N.A.; Arnold, P.T. Investigation of Antibacterial Effects of Southern Appalachian Plants: Phytolaccaamericana and Juglans nigra on Selected Bacteria. Ga. J. Sci. 2018, 76, 45. [Google Scholar]
- Loesche, W.J. Role of Streptococcus mutans in human dental decay. Microbiol. Rev. 1986, 50, 353–380. [Google Scholar] [PubMed]
- Botelho, M.A.; Nogueira, N.A.; Bastos, G.M.; Fonseca, S.G.; Lemos, T.L.; Matos, F.J.; Montenegro, D.; Heukelbach, J.; Rao, V.S.; Brito, G.A. Antimicrobial activity of the essential oil from Lippiasidoides, carvacrol and thymol against oral pathogens. Braz. J. Med. Biol. Res. 2007, 40, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Furletti, V.F.; Teixeira, I.P.; Obando-Pereda, G.; Mardegan, R.C.; Sartoratto, A.; Figueira, G.M.; Duarte, R.M.; Rehder, V.L.; Duarte, M.C.; Höfling, J.F. Action of Coriandrum sativum L. essential oil upon oral Candida albicans biofilm formation. Evid. Based Complement. Altern. Med. 2011, 2011, 985832. [Google Scholar] [CrossRef] [PubMed]
- Simões, M. Antimicrobial strategies effective against infectious bacterial biofilms. Curr. Med. Chem. 2011, 18, 2129–2145. [Google Scholar] [CrossRef]
- Al-Mariri, A.; Safi, M. In vitro antibacterial activity of several plant extracts and oils against some gram-negative bacteria. Iran. J. Med. Sci. 2014, 39, 36–43. [Google Scholar]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef]
- Randall, J.P.; Seow, W.K.; Walsh, L.J. Antibacterial activity of fluoride compounds and herbal toothpastes on Streptococcus mutans: An in vitro study. Aust. Dent. J. 2015, 60, 368–374. [Google Scholar] [CrossRef]
- Bhattacharjee, S.; Nath, S.; Bhattacharjee, P.; Chouhan, M.; Deb, B. Efficacy of Toothpastes on Bacteria Isolated from Oral Cavity. Int. J. Med. Public Health 2018, 8, 89–92. [Google Scholar] [CrossRef]
- Fine, D.H.; Furgang, D.; Barnett, M.L.; Drew, C.; Steinberg, L.; Charles, C.H.; Vincent, J.W. Effect of an essential oil-containing antiseptic mouthrinse on plaque and salivary Streptococcus mutans levels. J. Clin. Periodontol. 2000, 27, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, G.; Bottaro, M.; Nilson, A.C. Effect of the Thymus vulgaris essential oil on the growth of Streptococcus mutans. Revista de Ciências Farmacêuticas Básica e Aplicada 2012, 32, 375–380. [Google Scholar]
- Albuquerque, Y.E.; Danelon, M.; Salvador, M.J.; Koga-Ito, C.Y.; Botazzo Delbem, A.C.; Ramirez-Rueda, R.Y.; Lacerda Guntijo, A.V.; Brighenti, F.L. Mouthwash containing Croton doctoris essential oil: In vitro study using a validated model of caries induction. Future Microbiol. 2018, 13, 631–643. [Google Scholar] [CrossRef] [PubMed]
- Morris, A.J.; Steele, J.; White, D.A. Adult dental health survey: The oral cleanliness and periodontal health of U.Kadults in 1998. Br. Dent. J. 2001, 191, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Chaknis, P.; DeVizio, W.; Petrone, M.; Panagakos, F.S.; Proskin, H.M. A Clinical investigation of the efficacy of three commercially available dentifrices for controlling established gingivitis and supragingival plaque. J. Clin. Dent. 2010, 21, 105–110. [Google Scholar] [PubMed]
- Kim, S.H.; Hyun, S.H.; Choung, S.Y. Anti-diabetic effect of cinnamon extract on blood glucose in db/db mice. J. Ethnopharmacol. 2006, 104, 119–123. [Google Scholar] [CrossRef]
- Brierley, S.M.; Kelber, O. Use of natural products in gastrointestinal therapies. Curr. Opin. Pharmacol. 2011, 11, 604–611. [Google Scholar] [CrossRef]
- Al-Jiffri, O.; El-Sayed, Z.; Al-Sharif, F.M. Urinary tract infection with Esherichia coli and antibacterial activity of some plants extracts. Int. J. Microbiol. Res. 2011, 2, 1–7. [Google Scholar]
- Rassem, H.H.; Nour, A.H. Techniques for extraction of essential oils from plants: A review. Aust. J. Basic Appl. Sci. 2016, 10, 117–127. [Google Scholar]
- Meyer-Warnod, B. Natural essential oils: Extraction processes and application to some major oils. Perfum. Flavorist 1984, 9, 93–104. [Google Scholar]
- Calsamiglia, M.; Busquet, P.W.; Cardozo, L.; Castillejos, L.; Ferret, A. Invited review: Essential oils as modifiers of rumen microbial fermentation. J. Dairy Sci. 2007, 90, 2580–2595. [Google Scholar] [CrossRef]
- Daferera, D.J.; Ziogas, B.N.; Polissiou, M.G. GC-MS analysis of essential oils from some Greek aromatic plants and their fungitoxicity on Penicillium digitatum. J. Agric. Food Chem. 2000, 48, 2576–2581. [Google Scholar] [CrossRef]
- McLafferty, F.W.; Stauffer, D.B. (Eds.) The Wiley/NBS Registry of Mass Spectral Data; J Wiley and Sons: Hoboken, NJ, USA; Wiley Interscience Publication: New York, NY, USA, 1989. [Google Scholar]
- Koenig, W.A.; Joulain, D.; Hochmuth, D.H. Terpenoids and Related Constituents of Essential Oils. Mass Finder 3; EB-Verlag: Hamburg, Germany, 2004. [Google Scholar]
- Joulain, D.; Koenig, W.A. The Atlas of Spectra Data of Sesquiterpene Hydrocarbons; EB-Verlag: Hamburg, Germany, 1998. [Google Scholar]
- ESO 2000. The Complete Database of Essential Oils; Boelens Aroma Chemical Information Service: Huizen, The Netherlands, 1999. [Google Scholar]
- Baser, K.H.C. The Turkish Origanum Species. In Oregano, the Genera Origanum and Lippia; Kintzios, S.E., Ed.; Taylor and Francis: London, UK, 2002. [Google Scholar]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Oke, F.; Aslim, B. Biological potentials and cytotoxicity of various extracts from endemic Origanumminutiflorum O. Schwarz, PH Davis. Food Chem. Toxicol. 2010, 48, 1728–1733. [Google Scholar] [CrossRef] [PubMed]
- Karioti, A.; Vrahimi-Hadjilouca, T.; Droushiotis, D.; Rancic, A.; Hadjipavlou-Litina, D.; Skaltsa, H. Analysis of the essential oil of Origanum dubium growing wild in Cyprus. Investigation of its antioxidant capacity and antimicrobial activity. Planta Medica 2006, 72, 1–4. [Google Scholar] [CrossRef]
- Liolios, C.C.; Gortzi, O.; Lalas, S.; Tsaknis, J.; Chinou, I. Liposomal incorporation of carvacrol and thymol isolated from the essential oil of Origanum dictamnus L. and in vitro antimicrobial activity. Food Chem. 2009, 112, 77–83. [Google Scholar] [CrossRef]
- Ozkalp, B.; Sevgi, F.; Ozcan, M.; Ozcan, M.M. The antibacterial activity of essential oil of oregano (Origanum vulgare L.). J. Food Agric. Environ. 2010, 8, 272–274. [Google Scholar]
- Chorianopoulos, N.; Kalpoutzakis, E.; Aligianis, N.; Mitaku, S.; Nychas, G.J.; Haroutounian, S. Essential oils of Satureja, Origanum and Thymus species chemical composition and antibacterial activities against foodborne pathogen. J. Agric. Food Chem. 2004, 52, 8261–8267. [Google Scholar] [CrossRef] [PubMed]
- Skoula, M.; Gotsiou, P.; Naxakis, G.; Johnson, C.B.A. Chemosystematic investigation on the mono- and sesqui terpenoids in the genus Origanum (Labiatae). Phytochemistry 1999, 52, 649–657. [Google Scholar] [CrossRef]
- Davis, P.H.; Mill, R.R.; Tan, K. Flora of Turkey and the East Aegean Islands; University Press: Edinburgh, UK, 1988; Volume 10. [Google Scholar]
- Guner, A.; Ozhatay, N.; Ekim, T.; Baser, K.H.C. Flora of Turkey and the East Aegean Islands; University Press: Edinburgh, UK, 2000; Volume 11. [Google Scholar]
- Sivropoulou, A.; Papanikolaou, E.; Nikolaou, C.; Kokkini, S.; Lanaras, T.; Arsenakis, M. Antimicrobial and cytotoxic activities of Origanum essential oils. J. Agric. Food Chem. 1996, 44, 1202–1205. [Google Scholar] [CrossRef]
- De Souza, E.L.; de Barros, J.C.; de Oliveira, C.E.V.; da Conceição, M.L. Influence of Origanum vulgare L. essential oil on enterotoxin production, membrane permeability and surface characteristics of Staphylococcus aureus. Int. J. FoodMicrobiol. 2010, 137, 308–311. [Google Scholar]
- Szabó, M.Á.; Varga, G.Z.; Hohmann, J.; Schelz, Z.; Szegedi, E.; Amaral, L.; Molnár, J. Inhibition of quorum-sensing signals by essential oils. Phytother. Res. 2010, 24, 782–786. [Google Scholar] [CrossRef] [PubMed]
- Hyldgaard, M.; Mygind, T.; Meyer, R.L. Essential oils in food preservation: Mode of action, synergies, and interactions with food matrix components. Front. Microbiol. 2012, 3, 12. [Google Scholar] [CrossRef]
- Alvarez, M.V.; Ortega-Ramirez, L.A.; Gutierrez-Pacheco, M.M.; Bernal-Mercado, A.T.; Rodriguez-Garcia, I.; Gonzalez-Aguilar, G.A.; Ponce, A.; del Remoreina, M.; Roura, S.I.; Ayala-Zavala, J.F. Oregano essential oil-pectin edible films as anti-quorum sensing and food antimicrobial agents. Front. Microbiol. 2014, 5, 699. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Hernández, E.; Regalado-González, C.; Vázquez-Landaverde, P.; Guerrero-Legarreta, I.; García-Almendárez, B.E. Microencapsulation, chemical characterization, and antimicrobial activity of Mexican (Lippiagraveolens HBK) and European (Origanum vulgare L.) oregano essential oils. Sci. World J. 2014, 2014, 641814. [Google Scholar] [CrossRef] [PubMed]
- Pesavento, G.; Calonico, C.; Bilia, A.R.; Barnabei, M.; Calesini, F.; Addona, R.; Mencarelli, L.; Carmagnini, L.; Di Martino, M.C.; Nostro, A.L. Antibacterial activity of Oregano, Rosmarinus and Thymus essential oils against Staphylococcus aureus and Listeria monocytogenes in beef meatballs. Food Control 2015, 54, 188–199. [Google Scholar] [CrossRef]
- Donsì, F.; Ferrari, G. Essential oil nanoemulsions as antimicrobial agents in food. J. Biotechnol. 2016, 233, 106–120. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, L.K.D.; Jawale, B.A.; Kumar, H.S.C.D.M.; Kulkarni, P.A. Antimicrobial activity of commercially available essential oils against Streptococcus mutans. J. Contemp. Dent. Pract. 2012, 13, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Berber, I.; Avsar, C.; Cine, N.; Bozkurt, N.; Elmas, E. Determination of antibacterial and antifungal activities of methanolic extracts of some plants growing in Sinop. Karaelmas Sci. Eng. J. 2013, 3, 10–16. [Google Scholar] [CrossRef]
- Ranasinghe, P.; Jayawardana, R.; Galappaththy, P.; Constantine, G.R.; de Vas Gunawardana, N.; Katulanda, P. Efficacy and safety of ‘true’ cinnamon (Cinnamomum zeylanicum) as a pharmaceutical agent in diabetes: A systematic review and meta-analysis. Diabet. Med. 2012, 29, 1480–1492. [Google Scholar] [CrossRef] [PubMed]
- Nabavi, S.F.; Di Lorenzo, A.; Izadi, M.; Sobarzo-Sánchez, E.; Daglia, M.; Nabavi, S.M. Antibacterial effects of cinnamon: From farm to food, cosmetic and pharmaceutical industries. Nutrients 2015, 7, 7729–7748. [Google Scholar] [CrossRef] [PubMed]
- Aneja, K.R.; Joshi, R.; Sharma, C. Antimicrobial activity of Dalchini (Cinnamomum zeylanicum bark) extracts on some dental caries pathogens. J. Pharm. Res. 2009, 2, 1387–1390. [Google Scholar]
- Ribeiro, M.; Malheiro, J.; Grenho, L.; Fernandes, M.H.; Simões, M. Cytotoxicity and antimicrobial action of selected phytochemicals against planktonic and sessile Streptococcus mutans. PeerJ 2018, 6, e4872. [Google Scholar] [CrossRef] [PubMed]
- Choi, O.; Cho, S.K.; Kim, J.; Park, C.G.; Kim, J. In vitro antibacterial activity and major bioactive components of Cinnamomum verum essential oils against cariogenic bacteria, Streptococcus mutans and Streptococcus sobrinus. Asian Pac. J. Trop. Biomed. 2016, 6, 308–314. [Google Scholar] [CrossRef]
- Ooi, L.S.; Li, Y.; Kam, S.L.; Wang, H.; Wong, E.Y.; Ooi, V.E. Antimicrobial activities of cinnamon oil and cinnamaldehyde from the Chinese medicinal herb Cinnamomum cassia Blume. Am. J. Chin. Med. 2006, 34, 511–522. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.L.; Chen, X.P.; Wei, X.J. Study on chemical component of essential oil from Cinnamomum cassia presl and Cinnamomum cassia petrophilum leaves in Guangxi. Technol. Dev. Enterp. 2003, 6, 6–8. [Google Scholar]
- Singh, G.; Maurya, S.; deLampasana, M.P.; Catalan, C.A. A comparison of chemical, antioxidant and antimicrobial studies of cinnamon leaf and bark volatile oils, oleoresins and their constituents. Food Chem. Toxicol. 2007, 45, 1650–1661. [Google Scholar] [CrossRef]
- The European Antimicrobial Susceptibility Testing. Calibration and Validation. Available online: http://www.eucast.org/ (accessed on 26 August 2013).
- Valgas, C.; Souza, S.M.D.; Smânia, E.F.; Smânia, A., Jr. Screening methods to determine antibacterial activity of natural products. Braz. J. Microbiol. 2007, 38, 369–380. [Google Scholar] [CrossRef]
- Chaudhry, N.M.A.; Tariq, P. Anti-microbial activity of Cinnamomum cassia against diverse microbial flora with its nutritional and medicinal impacts. Pak. J. Bot. 2006, 38, 169–174. [Google Scholar]
- Magaldi, S.; Mata-Essayag, S.; De Capriles, C.H.; Perez, C.; Colella, M.T.; Olaizola, C.; Ontiveros, Y. Well diffusion for antifungal susceptibility testing. Int. J. Infect. Dis. 2004, 8, 39–45. [Google Scholar] [CrossRef]
- Wiwattanarattanabut, K.; Choonharuangdej, S.; Srithavaj, T. In vitro anti-cariogenic plaque effects of essential oils extracted from culinary herbs. J. Clin. Diagn. Res. 2017, 11, DC30–DC35. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.A. Concern regarding herbal toxicities: Case reports and counseling tips. Ann. Pharmacother. 1996, 30, 79–80. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, T.; Dweck, A.C. Aloe vera leaf gel: A review update. J. Ethnopharmacol. 1999, 68, 3–37. [Google Scholar] [CrossRef]
- Vogler, B.K.; Ernst, E. Aloe vera: A systemic review of its clinical effectiveness. Br. J. Gen.Pract. 1999, 49, 823–828. [Google Scholar] [PubMed]
- Ndhlala, A.R.; Amoo, S.O.; Stafford, G.I.; Finnie, J.F.; Van Staden, J. Antimicrobial, anti-inflammatory and mutagenic investigation of the South African tree Aloe (Aloe barberae). J. Ethnopharmacol. 2009, 124, 404–408. [Google Scholar] [CrossRef] [PubMed]
- Pandey, R.; Mishra, A. Antibacterial activities of crude extract of Aloe barbadensis to clinically isolated bacterial pathogens. Appl. Biochem. Biotechnol. 2010, 160, 1356–1361. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.S.; Zhang, W.U.; Li, Y. The antimicrobial potential of 14 natural herbal dentifrices: Results of an in vitro diffusion method study. J. Am. Dent. Assoc. 2004, 135, 1133–1141. [Google Scholar] [CrossRef] [PubMed]
- Fani, M.; Kohanteb, J. Inhibitory activity of Aloe vera gel on some clinically isolated cariogenic and periodontopathic bacteria. J. Oral Sci. 2012, 54, 15–21. [Google Scholar] [CrossRef]
- George, D.; Bhat, S.S.; Antony, B. Comparative evaluation of the antimicrobial efficacy of Aloe vera tooth gel and two popular commercial toothpastes: An in vitro study. Gen. Dent. 2009, 57, 238–241. [Google Scholar]
- Basting, R.T.; Gonçalves, F.R.; França, F.M.G.; do Amaral, F.L.B.; Flório, F.M. Antimicrobial potential of papain chemomechanical agent on Streptococcus mutans and Lactobacillus casei followed by the use of self-etching adhesive systems. J. Clin.Pediatr. Dent. 2016, 40, 62–68. [Google Scholar] [CrossRef]
- Mahmood, A.A.; Sidik, K.; Salmah, I. Wound healing activity of Carica papaya L. aqueous leaf extract in rats. Int. J. Mol. Med. 2005, 1, 398–401. [Google Scholar]
- Gartika, M.; Sasmita, I.S.; Satari, M.H.; Chairulfattah, A.; Hilmanto, D. Antibacterial activity of papain against Streptococcus mutans ATCC 25175. Int. J. Dev.Res. 2014, 4, 2075–2077. [Google Scholar]
- Silva, Z.S., Jr.; Huang, Y.Y.; De Freitas, L.F.; França, C.M.; Botta, S.B.; Ana, P.A.; Mesquita-Ferrari, R.A.; Fernandes, K.P.S.; Deana, A.; Leal, C.R.L.; et al. Papain gel containing methylene blue for simultaneous caries removal and antimicrobial photoinactivation against Streptococcus mutans biofilms. Sci.Rep. 2016, 6, 33270. [Google Scholar] [CrossRef] [PubMed]
- Casarin, M.; Pazinatto, J.; Santos, R.C.V.; Zanatta, F.B. Melaleuca alternifolia and its application against dental plaque and periodontal diseases: A systematic review. Phytother. Res. 2018, 32, 230–242. [Google Scholar] [CrossRef] [PubMed]
- Nikolić, M.M.; Jovanović, K.K.; Marković, T.L.; Marković, D.L.; Gligorijević, N.N.; Radulović, S.S.; Kostic, M.; Glouoclija, J.M.; Soković, M.D. Antimicrobial synergism and cytotoxic properties of Citrus limon L., Piper nigrum L. and Melaleuca alternifolia (Maiden and Betche) Cheel essential oils. J. Pharm.Pharmacol. 2017, 69, 1606–1614. [Google Scholar] [PubMed]
- Haffajee, A.D.; Yaskell, T.; Socransky, S.S. Antimicrobial effectiveness of an herbal mouthrinse compared with an essential oil and a chlorhexidine mouthrinse. J. Am. Dent. Assoc. 2008, 139, 606–611. [Google Scholar] [CrossRef]
- Oza, S.; Patel, K.; Bhosale, S.; Mitra, R.; Gupta, R.; Choudhary, D. To determine the effect of chewing gum containing xylitol and sorbitol on mutans streptococci and Lactobacilli count in saliva, plaque, and gingival health and to compare the efficacy of chewing gums. J. Int. Soc. Prev. Community Dent. 2018, 8, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, E.; Mazaheri, R.; Tahmourespour, A. Effect of probiotic yogurt and xylitol-containing chewing gums on salivary S. mutans count. J. Clin.Pediatr. Dent. 2017, 41, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Eskandarian, T.; Motamedifar, M.; Arasteh, P.; Eghbali, S.S.; Adib, A.; Abdoli, Z. Comparison of antimicrobial effects of titanium tetrafluoride, chlorhexidine, xylitol and sodium fluoride on Streptococcus mutans: An in-vitro study. Electron. Phys. 2017, 9, 4042–4047. [Google Scholar] [CrossRef]
| RRI | Compound Name | Relative Percentage Amounts (%) A% |
|---|---|---|
| 1020 | α-Pinene | 0.3 |
| 1024 | α-Thujene | 0.5 |
| 1172 | Myrcene | 0.4 |
| 1177 | α-Phellandrene | 0.2 |
| 1192 | α-Terpinene | 0.9 |
| 1211 | Limonene | 0.1 |
| 1223 | β-Phellandrene | 0.1 |
| 1260 | γ-Terpinene | 2.7 |
| 1288 | p-Cymene | 3.8 |
| 1299 | Terpinolene | 0.1 |
| 1478 | trans-sabinene hydrate | 0.4 |
| 1556 | Linalool | 0.1 |
| 1565 | cis-sabinene hydrate | 0.1 |
| 1625 | Terpinene-4-ol | 0.7 |
| 1629 | β-Caryophyllene | 0.1 |
| 1639 | trans-dihydrocarvone | Tr |
| 1718 | α-Terpineol | 0.5 |
| 1728 | Borneol | 0.1 |
| 1771 | Carvone | Tr |
| 2108 | Elemol | 0.1 |
| 2159 | Spathunelol | 0.1 |
| 2210 | Thymol | 0.2 |
| 2243 | Carvacrol | 88.3 |
| 2273 | β-Eudesmol | 0.1 |
| Total | 100.0 |
| LRI | Compound | Relative Percentage Amounts (%) B% |
|---|---|---|
| 1021 | α-pinene | 0.61 |
| 1073 | Camphene | 0.67 |
| 1119 | β-pinene | 0.17 |
| 1212 | Limonene | 0.23 |
| 1221 | 1,8-cineole | 1.18 |
| 1288 | p-cymene | 0.08 |
| 1515 | α-cubebene | 0.07 |
| 1556 | Benzaldehyde | 0.46 |
| 1605 | Bornyl acetate | 0.54 |
| 1625 | Terpinen-4-ol | 0.48 |
| 1629 | β-caryophyllene | 0.05 |
| 1718 | α-terpineol | 0.83 |
| 1728 | Borneol | 0.19 |
| 1787 | δ-cadinene | 0.03 |
| 1818 | Benzenepropanal (=phenylpropyl aldehyde) | 0.34 |
| 1940 | (Z)-cinnamaldehyde | 0.06 |
| 2091 | (E)-cinnamaldehyde | 91.79 |
| 2104 | 1-epi-cubenol | 1.16 |
| 2188 | Cinnamyl acetate | 0.72 |
| 2242 | Carvacrol | 0.01 |
| 2514 | Coumarin | 0.19 |
| Total | 99.84 | |
| Groups | Min | Max | Mean ± SD | |
|---|---|---|---|---|
| O. dubium Oil | - | 30 | 31 | 30.33 ± 0.58 a |
| C. cassia Oil | - | 38 | 40 | 38.67 ± 1.15 b |
| Colgate Total | - | 12 | 12 | 12.00 ± 0.00 c |
| Jack N’ Jill | - | 0 | 0 | 0.00 ± 0.00 d |
| +O. dubium Oil | 27 | 28 | 27.67 ± 0.58 c | |
| +C. cassia Oil | 39 | 40 | 39.67 ± 0.58 b | |
| Splat Organic | - | 12 | 12 | 12.00 ± 0.00 c |
| +O. dubium Oil | 22 | 24 | 23.00 ± 1.00 f | |
| +C. cassia Oil | 38 | 39 | 38.33 ± 0.58 b | |
| Splat Biocalcium | - | 11 | 11 | 11.00 ± 0.00 c |
| +O. dubium Oil | 23 | 24 | 23.33 ± 0.58 f | |
| +C. cassia Oil | 36 | 37 | 36.33 ± 0.58 g | |
| Distilled Water | - | 0 | 0 | 0.00 ± 0.00 d |
| TP | EO | TP + EO | |
|---|---|---|---|
| Inhibition Zone | 0.000 | 0.000 | 0.000 |
| Partial eta Squared | 0.772 | 0.999 | 0.983 |
| Toothpaste | Ingredients |
|---|---|
| Colgate Total | Sodium Fluoride (0.24% (0.14% w/v Fluoride Ion)), Triclosan (0.30%), Water Hydrated Silica, Glycerine, Sorbitol, PVM/MA Copolymer, Sodium Lauryl Sulphate |
| Splat Organic | Cellulose Gum, Flavour, Sodium Hydroxide, Carrageenan, Propylene Glycol, Sodium Saccharin, Titanium Dioxide Hydrogenated Starch Hydrolysate, Aqua, Hydrated Silica, PEG-8, Sodium Lauryl Sulphate, Aroma, Glycerine, Calcium Lactate, Aloe barbadensis Leaf Extract, Xanthan Gum, Melaleuca alternifolia Leaf Oil, Sodium Methylparaben, o-Cymen-5-ol, Papain, Citric Acid, Sodium Benzoate, CI 19140, CI 42090, Limonene. |
| Splat Biocalcium | Calcium Lactate, Sodium Bicarbonate, Hydroxyapatite, PVP, Fish Oil, Papain. |
| Jack N’ Jill | Xylitol, Purified water, Gylicerin, Silica, Xanthan gum, Organic Calendula officinalis extract, Potassium sorbate, Citric acid |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karadağlıoğlu, Ö.İ.; Ulusoy, N.; Başer, K.H.C.; Hanoğlu, A.; Şık, İ. Antibacterial Activities of Herbal Toothpastes Combined with Essential Oils against Streptococcus mutans. Pathogens 2019, 8, 20. https://doi.org/10.3390/pathogens8010020
Karadağlıoğlu Öİ, Ulusoy N, Başer KHC, Hanoğlu A, Şık İ. Antibacterial Activities of Herbal Toothpastes Combined with Essential Oils against Streptococcus mutans. Pathogens. 2019; 8(1):20. https://doi.org/10.3390/pathogens8010020
Chicago/Turabian StyleKaradağlıoğlu, Özgü İlkcan, Nuran Ulusoy, Kemal Hüsnü Can Başer, Azmi Hanoğlu, and İrem Şık. 2019. "Antibacterial Activities of Herbal Toothpastes Combined with Essential Oils against Streptococcus mutans" Pathogens 8, no. 1: 20. https://doi.org/10.3390/pathogens8010020
APA StyleKaradağlıoğlu, Ö. İ., Ulusoy, N., Başer, K. H. C., Hanoğlu, A., & Şık, İ. (2019). Antibacterial Activities of Herbal Toothpastes Combined with Essential Oils against Streptococcus mutans. Pathogens, 8(1), 20. https://doi.org/10.3390/pathogens8010020





