Evaluation of Lactiplantibacillus plantarum KAU007 against Low-Pathogenic Avian Influenza Virus (H9N2)
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacteria Culture and CFCS Preparation
2.2. Cell Culture, Cytotoxicity, and Antiviral Activity of CFCS of L. plantarum
2.3. Hemagglutination Assay
2.4. Estimation of Cytokines and Antioxidant Activity after CFCS L. plantarum KAU007
2.5. Statistical Analysis
3. Results and Discussion
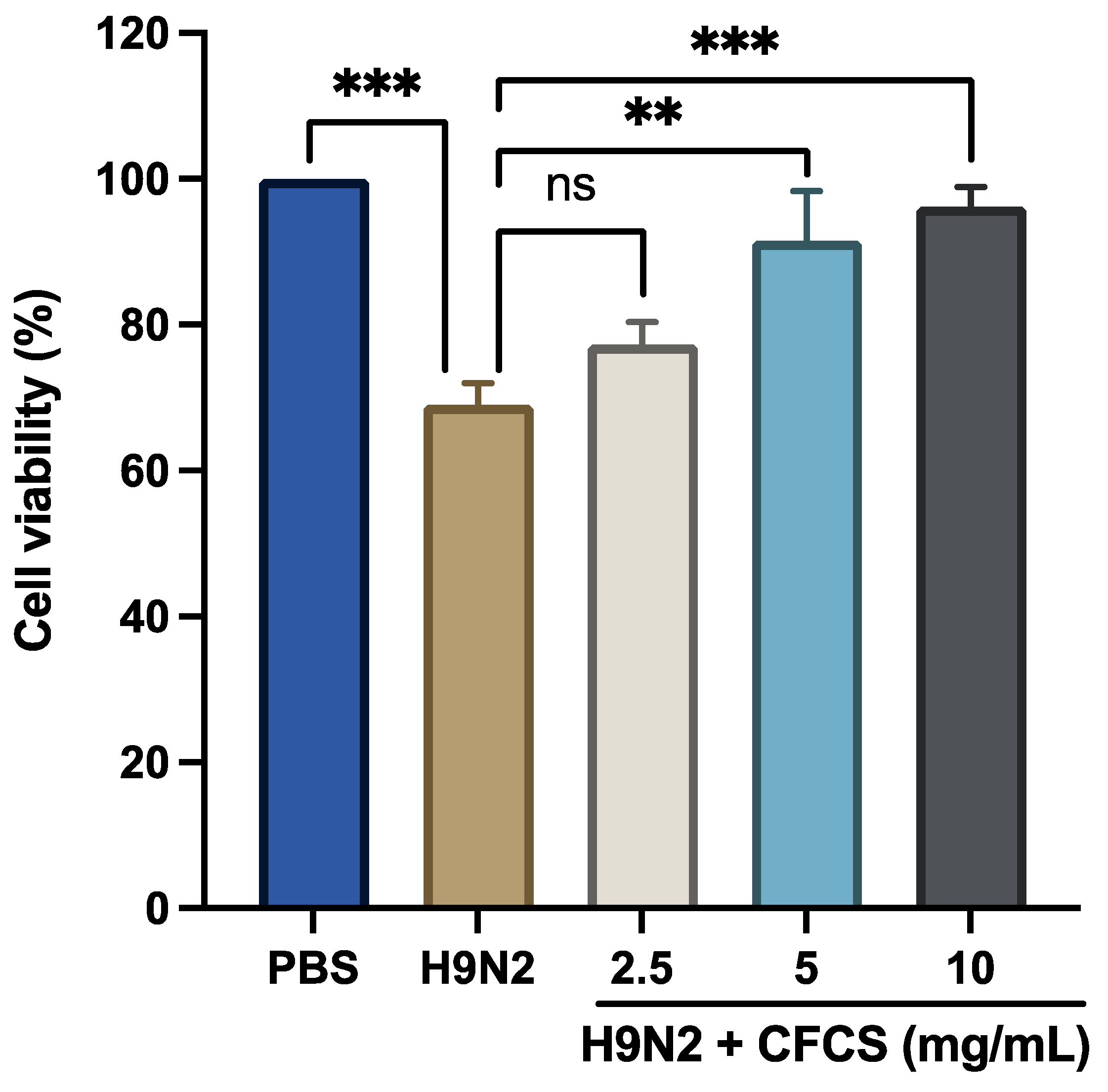
3.1. To Evaluate the Antiviral Effect of CFCS of L. plantarum against H9N2 in MDCK Cells
3.2. Evaluation of Antiviral Activity of L. plantarum CFCS against H9N2 Using Embryonated Eggs and Hemagglutination Assay
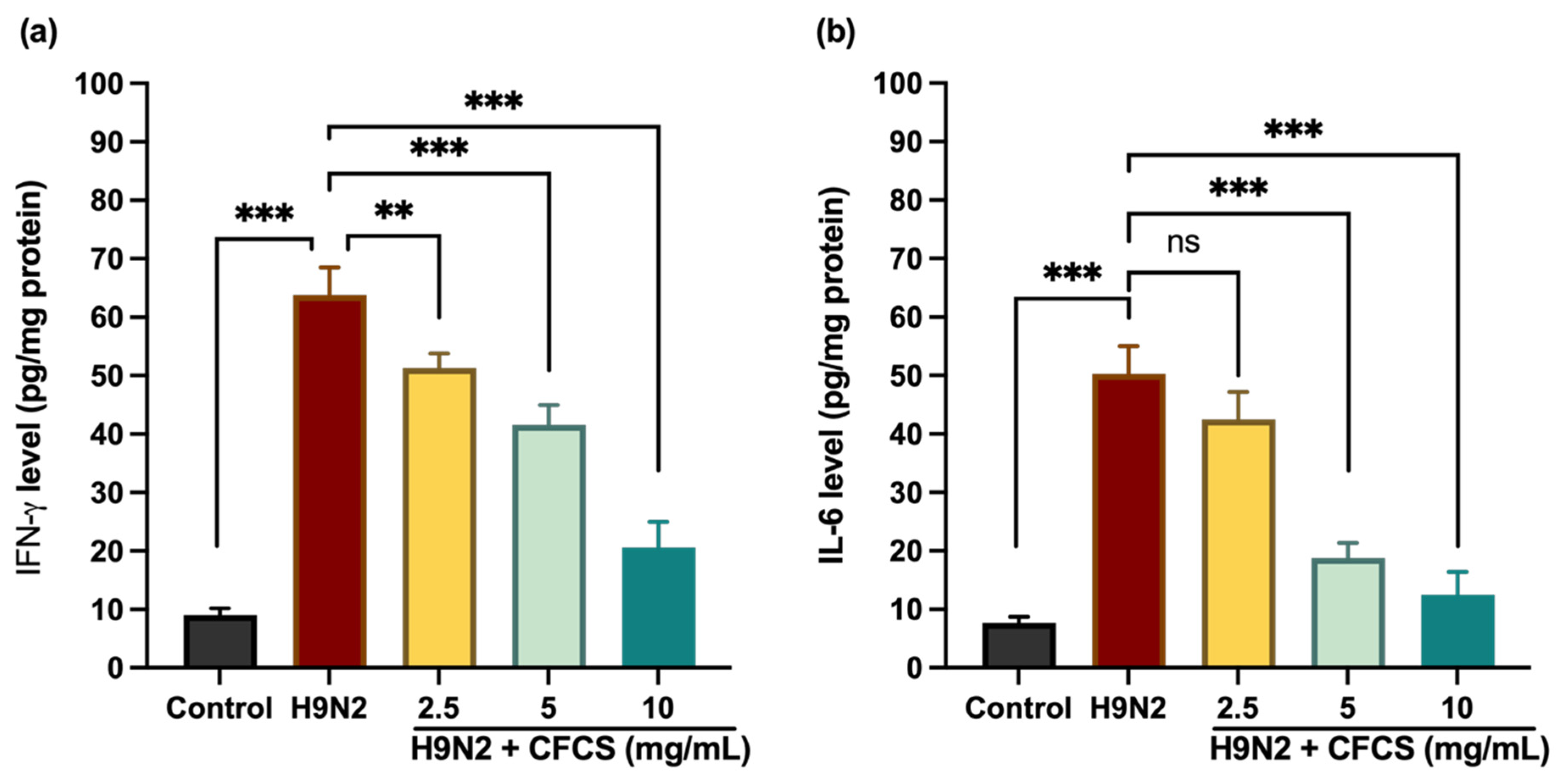
3.3. Effect of L. plantarum CFCS on Proinflammatory Cytokines
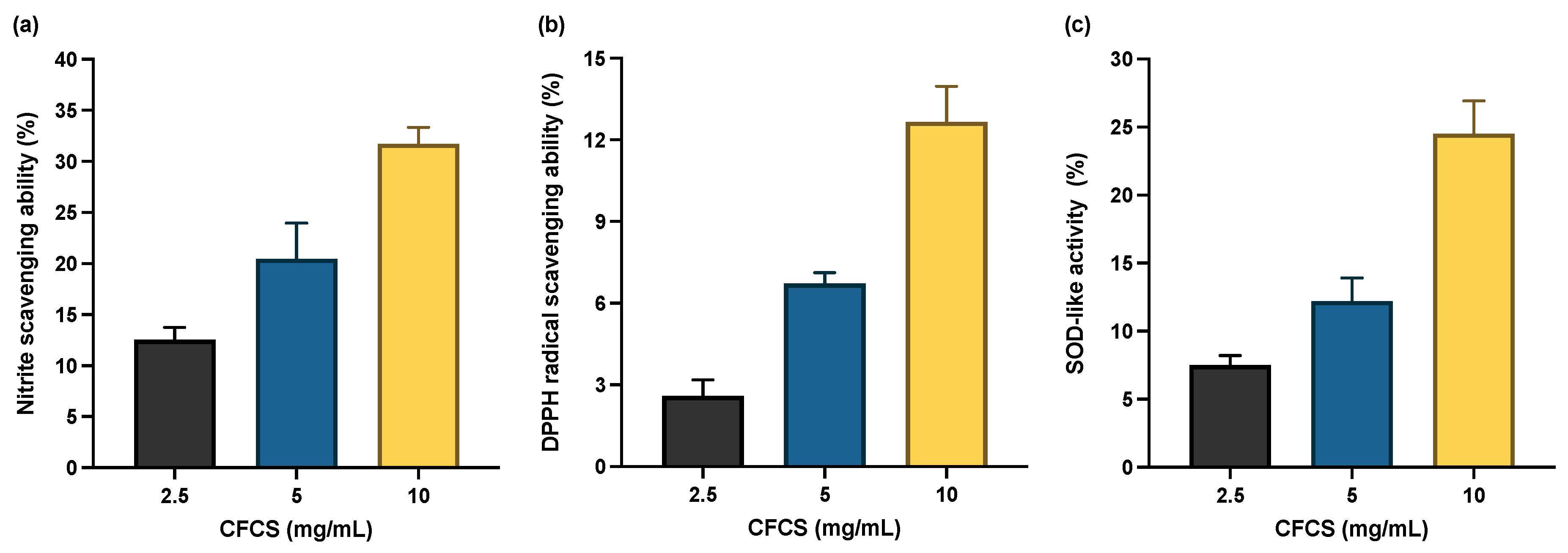
3.4. Effect of L. plantarum CFCS on Antioxidant System
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Carlson, C.J.; Albery, G.F.; Merow, C.; Trisos, C.H.; Zipfel, C.M.; Eskew, E.A.; Olival, K.J.; Ross, N.; Bansal, S. Climate change increases cross-species viral transmission risk. Nature 2022, 607, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Bi, Y.; Wong, G.; Gray, G.C.; Gao, G.F.; Li, S. Epidemiology, Evolution, and Recent Outbreaks of Avian Influenza Virus in China. J. Virol. 2015, 89, 8671–8676. [Google Scholar] [CrossRef] [PubMed]
- Arafat, N.; Eladl, A.H.; Marghani, B.H.; Saif, M.A.; El-Shafei, R. Enhanced infection of avian influenza virus H9N2 with infectious laryngeotracheitis vaccination in chickens. Vet. Microbiol. 2018, 219, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Barberis, A.; Boudaoud, A.; Gorrill, A.; Loupias, J.; Ghram, A.; Lachheb, J.; Alloui, N.; Ducatez, M.F. Full-length genome sequences of the first H9N2 avian influenza viruses isolated in the Northeast of Algeria. Virol. J. 2020, 17, 108. [Google Scholar] [CrossRef] [PubMed]
- Fouchier, R.A.M.; Munster, V.; Wallensten, A.; Bestebroer, T.M.; Herfst, S.; Smith, D.; Rimmelzwaan, G.F.; Olsen, B.; Osterhaus, A.D.M.E. Characterization of a Novel Influenza A Virus Hemagglutinin Subtype (H16) Obtained from Black-Headed Gulls. J. Virol. 2005, 79, 2814–2822. [Google Scholar] [CrossRef]
- Bailey, E.S.; Choi, J.Y.; Fieldhouse, J.K.; Borkenhagen, L.K.; Zemke, J.; Zhang, D.; Gray, G.C. The continual threat of influenza virus infections at the human–animal interface. Evol. Med. Public Health 2018, 2018, 192–198. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, J. H9N2 influenza virus in China: A cause of concern. Protein Cell 2015, 6, 18–25. [Google Scholar] [CrossRef]
- Wang, S.; Jiang, N.; Shi, W.; Yin, H.; Chi, X.; Xie, Y.; Hu, J.; Zhang, Y.; Li, H.; Chen, J.-L. Co-infection of H9N2 Influenza A Virus and Escherichia coli in a BALB/c Mouse Model Aggravates Lung Injury by Synergistic Effects. Front. Microbiol. 2021, 12, 670688. [Google Scholar] [CrossRef]
- Pan, Q.; Liu, A.; Zhang, F.; Ling, Y.; Ou, C.; Hou, N.; He, C. Co-infection of broilers with Ornithobacterium rhinotracheale and H9N2 avian influenza virus. BMC Veter. Res. 2012, 8, 104. [Google Scholar] [CrossRef]
- Carnaccini, S.; Perez, D.R. H9 Influenza Viruses: An Emerging Challenge. Cold Spring Harb. Perspect. Med. 2020, 10, a038588. [Google Scholar] [CrossRef]
- Peiris, M.; Yuen, K.; Leung, C.; Chan, K.; Ip, P.; Lai, R.; Orr, W.; Shortridge, K. Human infection with influenza H9N2. Lancet 1999, 354, 916–917. [Google Scholar] [CrossRef]
- Shanmuganatham, K.; Feeroz, M.M.; Jones-Engel, L.; Walker, D.; Alam, S.M.R.; Hasan, M.K.; McKenzie, P.; Krauss, S.; Webby, R.J.; Webster, R.G. Genesis of Avian Influenza H9N2 in Bangladesh. Emerg. Microbes Infect. 2014, 3, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.P.; Shaw, M.; Gregory, V.; Cameron, K.; Lim, W.; Klimov, A.; Subbarao, K.; Guan, Y.; Krauss, S.; Shortridge, K.; et al. Avian-to-human transmission of H9N2 subtype influenza A viruses: Relationship between H9N2 and H5N1 human isolates. Proc. Natl. Acad. Sci. USA 2000, 97, 9654–9658. [Google Scholar] [CrossRef]
- World Health Organization Influenza at the Human-Animal Interface Summary and Assessment. Available online: https://shorturl.hk/bUFDbU (accessed on 21 January 2022).
- The SJCEIRS H9 Working Group; Schultz-Cherry, S.; Thomas, P. Assessing the fitness of distinct clades of influenza A (H9N2) viruses. Emerg. Microbes Infect. 2013, 2, e75. [Google Scholar] [CrossRef]
- Nagy, A.; Mettenleiter, T.C.; Abdelwhab, E.M. A brief summary of the epidemiology and genetic relatedness of avian influenza H9N2 virus in birds and mammals in the Middle East and North Africa. Epidemiol. Infect. 2017, 145, 3320–3333. [Google Scholar] [CrossRef]
- Gu, M.; Xu, L.; Wang, X.; Liu, X. Current situation of H9N2 subtype avian influenza in China. Vet. Res. 2017, 48, 49. [Google Scholar] [CrossRef]
- Sitaras, I.; Rousou, X.; Kalthoff, D.; Beer, M.; Peeters, B.; de Jong, M.C.M. Role of vaccination-induced immunity and antigenic distance in the transmission dynamics of highly pathogenic avian influenza H5N1. J. R. Soc. Interface 2016, 13, 20150976. [Google Scholar] [CrossRef] [PubMed]
- Pu, J.; Wang, S.; Yin, Y.; Zhang, G.; Carter, R.A.; Wang, J.; Xu, G.; Sun, H.; Wang, M.; Wen, C.; et al. Evolution of the H9N2 influenza genotype that facilitated the genesis of the novel H7N9 virus. Proc. Natl. Acad. Sci. USA 2015, 112, 548–553. [Google Scholar] [CrossRef]
- Poland, G.A. Influenza Vaccine Failure: Failure to Protect or Failure to Understand? Expert Rev. Vaccines 2018, 17, 495–502. [Google Scholar] [CrossRef]
- Tiwari, S.K.; Dicks, L.M.T.; Popov, I.V.; Karaseva, A.; Ermakov, A.M.; Suvorov, A.; Tagg, J.R.; Weeks, R.; Chikindas, M.L. Probiotics at War Against Viruses: What Is Missing from the Picture? Front. Microbiol. 2020, 11, 1877. [Google Scholar] [CrossRef]
- AL Kassaa, I.; Hober, D.; Hamze, M.; Chihib, N.E.; Drider, D. Antiviral Potential of Lactic Acid Bacteria and Their Bacteriocins. Probiotics Antimicrob. Proteins 2014, 6, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Drider, D.; Bendali, F.; Naghmouchi, K.; Chikindas, M.L. Bacteriocins: Not Only Antibacterial Agents. Probiotics Antimicrob. Proteins 2016, 8, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Chikindas, M.L.; Weeks, R.; Drider, D.; Chistyakov, V.A.; Dicks, L.M. Functions and emerging applications of bacteriocins. Curr. Opin. Biotechnol. 2018, 49, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Kanmani, P.; Albarracin, L.; Kobayashi, H.; Hebert, E.M.; Saavedra, L.; Komatsu, R.; Gatica, B.; Miyazaki, A.; Ikeda-Ohtsubo, W.; Suda, Y.; et al. Genomic Characterization of Lactobacillus delbrueckii TUA4408L and Evaluation of the Antiviral Activities of its Extracellular Polysaccharides in Porcine Intestinal Epithelial Cells. Front. Immunol. 2018, 9, 2178. [Google Scholar] [CrossRef] [PubMed]
- del Rio, B.; Dattwyler, R.J.; Aroso, M.; Neves, V.; Meirelles, L.; Seegers, J.F.M.L.; Gomes-Solecki, M. Oral Immunization with Recombinant Lactobacillus plantarum Induces a Protective Immune Response in Mice with Lyme Disease. Clin. Vaccine Immunol. 2008, 15, 1429–1435. [Google Scholar] [CrossRef]
- Rather, I.A.; Kamli, M.R.; Sabir, J.S.M.; Paray, B.A. Potential Antiviral Activity of Lactiplantibacillus plantarum KAU007 against Influenza Virus H1N1. Vaccines 2022, 10, 456. [Google Scholar] [CrossRef] [PubMed]
- Majumder, R.; Rather, I.A.; Bajpai, V.K.; Park, Y.-H. In vitro antiviral activity of Lactobacillus plantarum using SPF embryonated eggs and hemagglutination assay. Bangladesh J. Pharmacol. 2015, 10, 688–691. [Google Scholar] [CrossRef]
- Rather, I.A.; Choi, K.-H.; Bajpai, V.K.; Park, Y.-H. Antiviral mode of action of Lactobacillus plantarum YML009 on Influenza virus H1N1. Bangladesh J. Pharmacol. 2015, 10, 475–482. [Google Scholar] [CrossRef]
- Seo, B.; Rather, I.; Kumar, V.; Choi, U.; Moon, M.; Lim, J.; Park, Y. Evaluation of Leuconostoc mesenteroides YML003 as a probiotic against low-pathogenic avian influenza (H9N2) virus in chickens. J. Appl. Microbiol. 2012, 113, 163–171. [Google Scholar] [CrossRef]
- Choi, D.; Cho, K.-A.; Na, M.-S.; Choi, H.-S.; Kim, Y.-O.; Lim, D.-H.; Cho, S.J.; Cho, H. Effect of bamboo oil on antioxidative activity and nitrite scavenging activity. J. Ind. Eng. Chem. 2008, 14, 765–770. [Google Scholar] [CrossRef]
- Bajpai, V.K.; Rather, I.A.; Park, Y.-H. Partially Purified Exo-Polysaccharide from Lactobacillus Sakei Probio 65 with Antioxidant, α-Glucosidase and Tyrosinase Inhibitory Potential. J. Food Biochem. 2016, 40, 264–274. [Google Scholar] [CrossRef]
- Bajpai, V.K.; Alam, M.B.; Quan, K.T.; Kwon, K.-R.; Ju, M.-K.; Choi, H.-J.; Lee, J.S.; Yoon, J.-I.; Majumder, R.; Rather, I.A.; et al. Antioxidant efficacy and the upregulation of Nrf2-mediated HO-1 expression by (+)-lariciresinol, a lignan isolated from Rubia philippinensis, through the activation of p38. Sci. Rep. 2017, 7, 46035. [Google Scholar] [CrossRef] [PubMed]
- Oh, N.; Kwon, H.; Lee, H.; Joung, J.; Lee, J.; Lee, K.; Shin, Y.; Baick, S.; Park, M.; Kim, Y.; et al. Preventive effect of fermented Maillard reaction products from milk proteins in cardiovascular health. J. Dairy Sci. 2014, 97, 3300–3313. [Google Scholar] [CrossRef] [PubMed]
- Rather, I.A.; Sabir, J.S.M.; Asseri, A.H.; Ali, S. Antifungal Activity of Human Cathelicidin LL-37, a Membrane Disrupting Peptide, by Triggering Oxidative Stress and Cell Cycle Arrest in Candida auris. J. Fungi 2022, 8, 204. [Google Scholar] [CrossRef]
- Al Malki, A.; Yoon, S.-H.; Firoz, A.; Ali, H.M.; Park, Y.-H.; Hor, Y.-Y.; Rather, I.A. Characterization of New Probiotic Isolates from Fermented Ajwa Dates of Madinah and Their Anti-Inflammatory Potential. Appl. Sci. 2022, 12, 5082. [Google Scholar] [CrossRef]
- Ooi, M.F.; Foo, H.L.; Loh, T.C.; Mohamad, R.; Rahim, R.A.; Ariff, A. A refined medium to enhance the antimicrobial activity of postbiotic produced by Lactiplantibacillus plantarum RS5. Sci. Rep. 2021, 11, 7617. [Google Scholar] [CrossRef]
- Motevaseli, E.; Shirzad, M.; Akrami, S.M.; Mousavi, A.-S.; Mirsalehian, A.; Modarressi, M.H. Normal and tumour cervical cells respond differently to vaginal lactobacilli, independent of pH and lactate. J. Med. Microbiol. 2013, 62, 1065–1072. [Google Scholar] [CrossRef]
- Chuah, L.-O.; Foo, H.L.; Loh, T.C.; Alitheen, N.B.M.; Yeap, S.K.; Mutalib, N.E.A.; Rahim, R.A.; Yusoff, K. Postbiotic metabolites produced by Lactobacillus plantarum strains exert selective cytotoxicity effects on cancer cells. BMC Complement. Altern. Med. 2019, 19, 114. [Google Scholar] [CrossRef]
- Sunmola, A.A.; Ogbole, O.O.; Faleye, T.O.C.; Adetoye, A.; Adeniji, J.A.; Ayeni, F.A. Antiviral potentials of Lactobacillus plantarum, Lactobacillus amylovorus, and Enterococcus hirae against selected Enterovirus. Folia Microbiol. 2019, 64, 257–264. [Google Scholar] [CrossRef]
- Kozlov, E.; Ivanova, E.; Grechko, A.; Wu, W.-K.; Starodubova, A.; Orekhov, A. Involvement of Oxidative Stress and the Innate Immune System in SARS-CoV-2 Infection. Diseases 2021, 9, 17. [Google Scholar] [CrossRef]
- Jeon, H.-J.; Choi, H.-S.; Lee, O.-H.; Jeon, Y.-J.; Lee, B.-Y. Inhibition of Reactive Oxygen Species (ROS) and Nitric Oxide (NO) by Gelidium elegans Using Alternative Drying and Extraction Conditions in 3T3-L1 and RAW 264.7 Cells. Prev. Nutr. Food Sci. 2012, 17, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.M.; Foo, H.L.; Loh, T.C.; Lim, E.T.C.; Mutalib, N.E.A. Comparative Studies of Inhibitory and Antioxidant Activities, and Organic Acids Compositions of Postbiotics Produced by Probiotic Lactiplantibacillus plantarum Strains Isolated from Malaysian Foods. Front. Vet. Sci. 2021, 7, 602280. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Yang, W.; Song, J.; Wu, Y.; Ni, B. Hepatitis B Virus X Protein-Induced Aberrant Epigenetic Modifications Contributing to Human Hepatocellular Carcinoma Pathogenesis. Mol. Cell. Biol. 2013, 33, 2810–2816. [Google Scholar] [CrossRef] [PubMed]
- Park, C.W.; Youn, M.; Jung, Y.-M.; Kim, H.; Jeong, Y.; Lee, H.-K.; Kim, H.O.; Lee, I.; Lee, S.W.; Kang, K.H.; et al. New Functional Probiotic Lactobacillus sakei Probio 65 Alleviates Atopic Symptoms in the Mouse. J. Med. Food 2008, 11, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Parvez, S.; Malik, K.A.; Ah Kang, S.; Kim, H.-Y. Probiotics and their fermented food products are beneficial for health. J. Appl. Microbiol. 2006, 100, 1171–1185. [Google Scholar] [CrossRef]
- Drouault, S. Health Effects of Lactic Acid Bacteria Ingested in Fermented Milk. Vet. Res. 2001, 32, 101–117. [Google Scholar] [CrossRef]
- Apás, A.L.; Arena, M.E.; Elias, A.; González, F.; González, S.N. Beneficial Effects of Fermented Sugarcane Residue with Goat Probiotic on Gut Health. Int. J. Agric. Innov. Res. 2015, 3, 1–8. [Google Scholar]
- Adedokun, E.O.; Rather, I.A.; Bajpai, V.K.; Choi, K.-H.; Park, Y.-H. Isolation and Characterization of Lactic Acid Bacteria from Nigerian Fermented Foods and Their Antimicrobial Activity. J. Pure Appl. Microbiol. 2014, 8, 3411–3420. [Google Scholar]
- Nagao, F.; Nakayama, M.; Muto, T.; Okumura, K. Effects of a Fermented Milk Drink ContainingLactobacillus caseiStrain Shirota on the Immune System in Healthy Human Subjects. Biosci. Biotechnol. Biochem. 2000, 64, 2706–2708. [Google Scholar] [CrossRef]
- Makino, S.; Ikegami, S.; Kume, A.; Horiuchi, H.; Sasaki, H.; Orii, N. Reducing the risk of infection in the elderly by dietary intake of yoghurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1. Br. J. Nutr. 2010, 104, 998–1006. [Google Scholar] [CrossRef]
- Bajpai, V.K.; Rather, I.A.; Majumder, R.; Alshammari, F.H.; Nam, G.-J.; Park, Y.-H. Characterization and Antibacterial Mode of Action of Lactic Acid BacteriumLeuconostoc mesenteroidesHJ69 from Kimchi. J. Food Biochem. 2017, 41, e12290. [Google Scholar] [CrossRef]
- Bajpai, V.K.; Han, J.-H.; Rather, I.A.; Park, C.; Lim, J.; Paek, W.K.; Lee, J.S.; Yoon, J.-I.; Park, Y.-H. Characterization and Antibacterial Potential of Lactic Acid Bacterium Pediococcus pentosaceus 4I1 Isolated from Freshwater Fish Zacco koreanus. Front. Microbiol. 2016, 7, 2037. [Google Scholar] [CrossRef] [PubMed]
- Rather, I.A.; Seo, B.J.; Kumar, V.J.R.; Choi, U.-H.; Choi, K.-H.; Lim, J.; Park, Y.-H. Biopreservative potential of Lactobacillus plantarum YML007 and efficacy as a replacement for chemical preservatives in animal feed. Food Sci. Biotechnol. 2014, 23, 195–200. [Google Scholar] [CrossRef]
- Ahmad Rather, I.; Seo, B.J.; Rejish Kumar, V.J.; Choi, U.-H.; Choi, K.-H.; Lim, J.H.; Park, Y.-H. Isolation and characterization of a proteinaceous antifungal compound from Lactobacillus plantarum YML007 and its application as a food preservative. Lett. Appl. Microbiol. 2013, 57, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Anwar, F.; Altayb, H.N.; Al-Abbasi, F.A.; Al-Malki, A.L.; Kamal, M.A.; Kumar, V. Antiviral effects of probiotic metabolites on COVID-19. J. Biomol. Struct. Dyn. 2021, 39, 4175–4184. [Google Scholar] [CrossRef]
- Youn, H.-N.; Lee, D.-H.; Lee, Y.-N.; Park, J.-K.; Yuk, S.-S.; Yang, S.-Y.; Lee, H.-J.; Woo, S.-H.; Kim, H.-M.; Lee, J.-B.; et al. Intranasal administration of live Lactobacillus species facilitates protection against influenza virus infection in mice. Antivir. Res. 2012, 93, 138–143. [Google Scholar] [CrossRef]
- Tomasik, P.; Tomasik, P. Probiotics, Non-Dairy Prebiotics and Postbiotics in Nutrition. Appl. Sci. 2020, 10, 1470. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; Rodrigues, C.; Stojanović-Radić, Z.; Dimitrijević, M.; Aleksić, A.; Neffe-Skocińska, K.; Zielińska, D.; Kołożyn-Krajewska, D.; Salehi, B.; Prabu, S.M.; et al. Probiotics: Versatile Bioactive Components in Promoting Human Health. Medicina 2020, 56, 433. [Google Scholar] [CrossRef]
- Rather, I.A.; Majumder, R.; Alshammari, F.H.; Park, J.G.; Bajpai, V.K. Review-Ulcerative colitis and probiotics: An overview. Pak. J. Pharm. Sci. 2016, 29, 1877–1880. [Google Scholar]
- Santosa, S.; Farnworth, E.; Jones, P.J.H. Probiotics and Their Potential Health Claims. Nutr. Rev. 2006, 64, 265–274. [Google Scholar] [CrossRef]
- Rather, I.A.; Bajpai, V.K.; Kumar, S.; Lim, J.; Paek, W.K.; Park, Y.-H. Probiotics and Atopic Dermatitis: An Overview. Front. Microbiol. 2016, 7, 507. [Google Scholar] [CrossRef] [PubMed]
- Reid, G.; Jass, J.; Sebulsky, M.T.; McCormick, J.K. Potential Uses of Probiotics in Clinical Practice. Clin. Microbiol. Rev. 2003, 16, 658–672. [Google Scholar] [CrossRef] [PubMed]
- Suez, J.; Zmora, N.; Segal, E.; Elinav, E. The pros, cons, and many unknowns of probiotics. Nat. Med. 2019, 25, 716–729. [Google Scholar] [CrossRef] [PubMed]
- Mangrolia, U.; Osborne, J.W. Probiotics in Counteracting the Role of Neutrophils in Cancer Metastasis. Vaccines 2021, 9, 1306. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, V.K.; Chandra, V.; Kim, N.-H.; Rai, R.; Kumar, P.; Kim, K.; Aeron, A.; Kang, S.C.; Maheshwari, D.K.; Na, M.; et al. Ghost probiotics with a combined regimen: A novel therapeutic approach against the Zika virus, an emerging world threat. Crit. Rev. Biotechnol. 2018, 38, 438–454. [Google Scholar] [CrossRef]
| Groups | Treatment | Survival/Total | Survival Percentage |
|---|---|---|---|
| Con | PBS | 2/2 | 100% |
| H9N2 | 107.5 EID50/0.1 mL | 5/9 | 55% |
| H9N2 + KAU007—I | CFCS (2.5 mg/mL) | 3/4 | 75% |
| H9N2 + KAU007—II | CFCS (5 mg/mL) | 5/6 | 83% |
| H9N2 + KAU007—III | CFCS (10 mg/mL) | 8/9 | 88.88% |
| Treatment | ||||
|---|---|---|---|---|
| Hemagglutinating activity | Con | KAU007 CFCS (2.5 mg/mL) | KAU007 CFCS (5 mg/mL) | KAU007 CFCS (10 mg/mL) |
| 0 | 1:4 | 1:8 | 1:8 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rather, I.A.; Kamli, M.R.; Sabir, J.S.M.; Ali, S. Evaluation of Lactiplantibacillus plantarum KAU007 against Low-Pathogenic Avian Influenza Virus (H9N2). Pathogens 2022, 11, 1246. https://doi.org/10.3390/pathogens11111246
Rather IA, Kamli MR, Sabir JSM, Ali S. Evaluation of Lactiplantibacillus plantarum KAU007 against Low-Pathogenic Avian Influenza Virus (H9N2). Pathogens. 2022; 11(11):1246. https://doi.org/10.3390/pathogens11111246
Chicago/Turabian StyleRather, Irfan A., Majid Rasool Kamli, Jamal S. M. Sabir, and Sajad Ali. 2022. "Evaluation of Lactiplantibacillus plantarum KAU007 against Low-Pathogenic Avian Influenza Virus (H9N2)" Pathogens 11, no. 11: 1246. https://doi.org/10.3390/pathogens11111246
APA StyleRather, I. A., Kamli, M. R., Sabir, J. S. M., & Ali, S. (2022). Evaluation of Lactiplantibacillus plantarum KAU007 against Low-Pathogenic Avian Influenza Virus (H9N2). Pathogens, 11(11), 1246. https://doi.org/10.3390/pathogens11111246







