A Broad-Based Mosquito Yeast Interfering RNA Pesticide Targeting Rbfox1 Represses Notch Signaling and Kills Both Larvae and Adult Mosquitoes
Abstract
1. Introduction
2. Results and Discussion
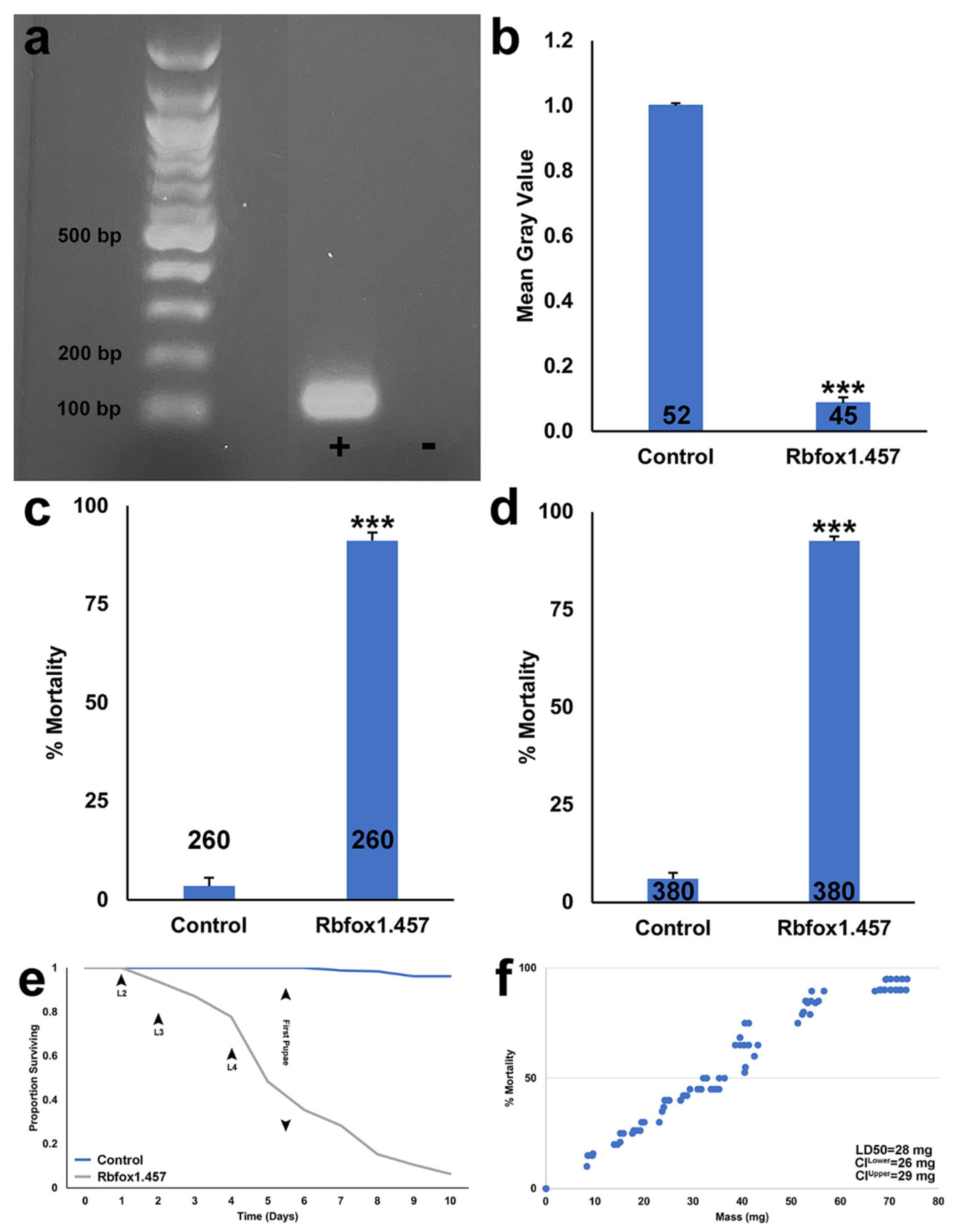
2.1. Silencing Rbfox1 Results in A. aegypti Larval Mortality
2.2. Sugar Baited Delivery of Rbfox1.457 siRNA
2.3. Development of an RNAi-Based Yeast Pesticide That Can Be Deployed as an ATSB
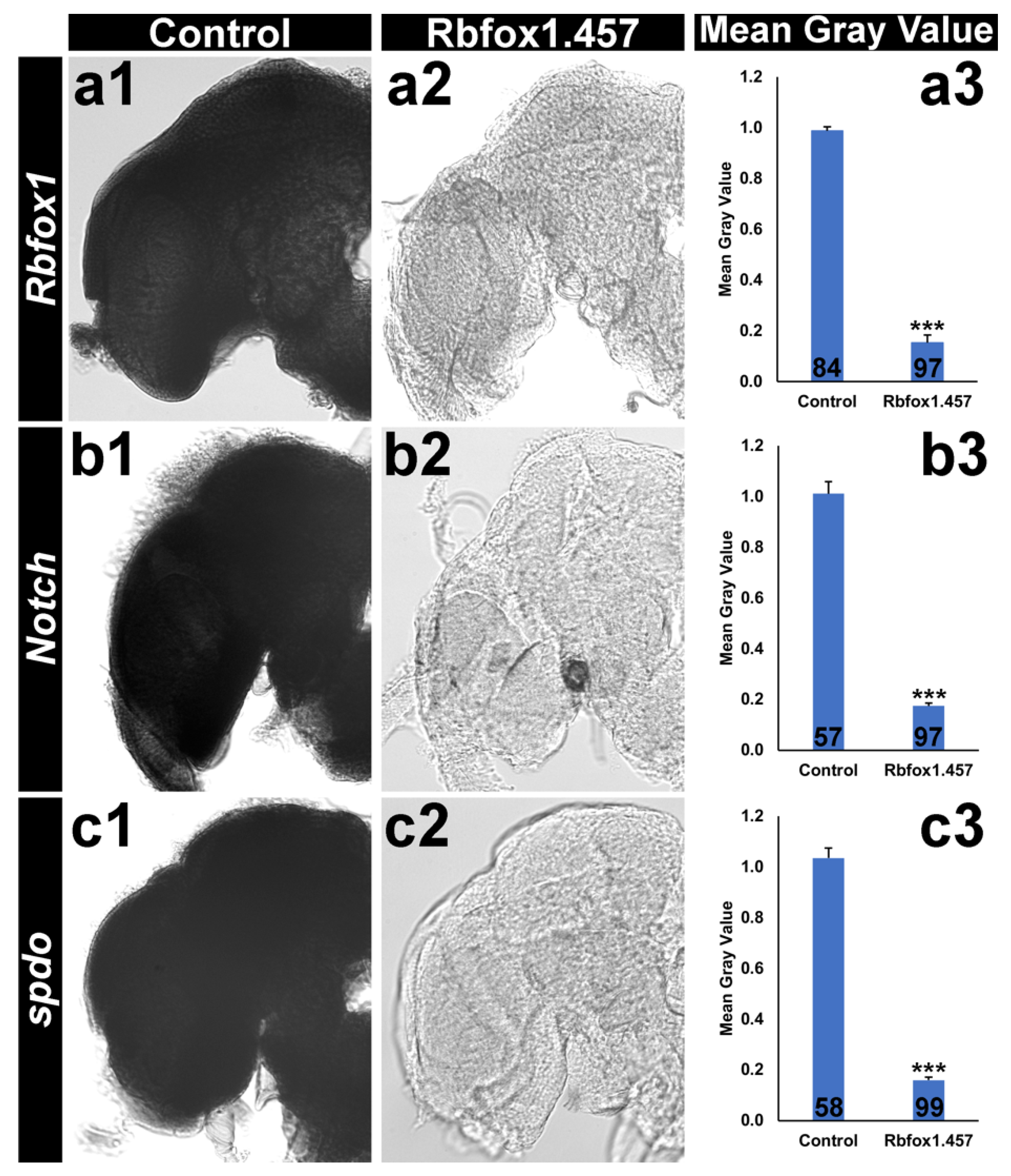
2.4. Silencing Rbfox1 Represses Notch Signaling
2.5. Rbfox1.457 Yeast Pesticides Function as Broad-Range Mosquito Insecticides but Are Not Found to Be Toxic to Select Non-Target Arthropods
3. Materials and Methods
3.1. Mosquito Rearing
3.2. Identification of siRNA #457
3.3. Adult siRNA-ATSB Trials
3.4. Generation of Yeast Interfering RNA Larvicide Strains and Yeast Culturing
3.5. Larvicide Trials
3.5.1. Laboratory Assays
3.5.2. Semi-Field Trials
3.5.3. Yeast ATSB Simulated Field Trials
3.6. Whole Mount In Situ Hybridization
3.7. Evaluation of Non-target Species
3.7.1. O. fasciatus
3.7.2. H. convergens
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Airs, P.M.; Bartholomay, L.C. RNA interference for mosquito and mosquito-borne disease control. Insects 2017, 8, 4. [Google Scholar] [CrossRef]
- Environmental Protection Agency. Pesticides. Available online: https://www.epa.gov/pesticides (accessed on 1 March 2019).
- World Health Organization. Dengue Guidelines for Diagnosis, Treatment, Prevention and Control; WHO: Geneva, Switzerland, 2009. [Google Scholar]
- Wiltshire, R.M.; Duman-Scheel, M. Advances in oral RNAi for disease vector mosquito research and control. Curr. Opin. Insect Sci. 2020, 40, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Khan, S.A.; Heckel, D.G.; Bock, R. Next-generation insect-resistant plants: RNAi-mediated crop protection. Trends Biotechnol. 2017, 35, 871–882. [Google Scholar] [CrossRef] [PubMed]
- Hapairai, L.K.; Mysore, K.; Chen, Y.; Harper, E.I.; Scheel, M.P.; Lesnik, A.M.; Sun, L.; Severson, D.W.; Wei, N.; Duman-Scheel, M. Lure-and-kill yeast interfering RNA larvicides targeting neural genes in the human disease vector mosquito Aedes aegypti. Sci. Rep. 2017, 7, 13223. [Google Scholar] [CrossRef] [PubMed]
- Mysore, K.; Hapairai, L.K.; Sun, L.; Harper, E.I.; Chen, Y.; Eggleson, K.K.; Realey, J.S.; Scheel, N.D.; Severson, D.W.; Wei, N.; et al. Yeast interfering RNA larvicides targeting neural genes induce high rates of Anopheles larval mortality. Malar. J. 2017, 16, 461. [Google Scholar] [CrossRef] [PubMed]
- Hapairai, L.K.; Mysore, K.; Sun, L.; Li, P.; Wang, C.W.; Scheel, N.D.; Lesnik, A.; Scheel, M.P.; Igiede, J.; Wei, N.; et al. Characterization of an adulticidal and larvicidal interfering RNA pesticide that targets a conserved sequence in mosquito G protein-coupled dopamine 1 receptor genes. Insect Biochem. Mol. Biol. 2020, 120, 103359. [Google Scholar] [CrossRef]
- Mysore, K.; Hapairai, L.K.; Sun, L.; Li, P.; Wang, C.W.; Scheel, N.D.; Lesnik, A.; Igiede, J.; Scheel, M.P.; Wei, N.; et al. Characterization of a dual-action adulticidal and larvicidal interfering RNA pesticide targeting the Shaker gene of multiple disease vector mosquitoes. PLoS Negl. Trop. Dis. 2020, 14, e0008479. [Google Scholar] [CrossRef]
- Mysore, K.; Li, P.; Wang, C.W.; Hapairai, L.K.; Scheel, N.D.; Realey, J.S.; Sun, L.; Roethele, J.B.; Severson, D.W.; Wei, N.; et al. Characterization of a yeast interfering RNA larvicide with a target site conserved in the synaptotagmin gene of multiple disease vector mosquitoes. PLoS Negl. Trop. Dis. 2019, 13, e0007422. [Google Scholar] [CrossRef]
- Mysore, K.; Li, P.; Wang, C.W.; Hapairai, L.K.; Scheel, N.D.; Realey, J.S.; Sun, L.; Severson, D.W.; Wei, N.; Duman-Scheel, M. Characterization of a broad-based mosquito yeast interfering RNA larvicide with a conserved target site in mosquito semaphorin-1a genes. Parasit. Vectors 2019, 12, 256. [Google Scholar] [CrossRef]
- Conboy, J.G. Developmental regulation of RNA processing by Rbfox proteins. Wiley Interdiscip. Rev. RNA 2017, 8, e1398. [Google Scholar] [CrossRef]
- Shukla, J.P.; Deshpande, G.; Shashidhara, L.S. Ataxin 2-binding protein 1 is a context-specific positive regulator of Notch signaling during neurogenesis in Drosophila melanogaster. Development 2017, 144, 905–915. [Google Scholar] [CrossRef] [PubMed]
- Nikonova, E.; Kao, S.Y.; Ravichandran, K.; Wittner, A.; Spletter, M.L. Conserved functions of RNA-binding proteins in muscle. Int. J. Biochem. Cell Biol. 2019, 110, 29–49. [Google Scholar] [CrossRef]
- Shibata, H.; Huynh, D.P.; Pulst, S.M. A novel protein with RNA-binding motifs interacts with ataxin-2. Hum. Mol. Genet. 2000, 9, 1303–1313. [Google Scholar] [CrossRef]
- Gazzara, M.R.; Mallory, M.J.; Roytenberg, R.; Lindberg, J.P.; Jha, A.; Lynch, K.W.; Barash, Y. Ancient antagonism between CELF and RBFOX families tunes mRNA splicing outcomes. Genome Res. 2017, 27, 1360–1370. [Google Scholar] [CrossRef] [PubMed]
- Kucherenko, M.M.; Shcherbata, H.R. Stress-dependent miR-980 regulation of Rbfox1/A2bp1 promotes ribonucleoprotein granule formation and cell survival. Nat. Commun. 2018, 9, 312. [Google Scholar] [CrossRef] [PubMed]
- Tastan, O.Y.; Maines, J.Z.; Li, Y.; McKearin, D.M.; Buszczak, M. Drosophila ataxin 2-binding protein 1 marks an intermediate step in the molecular differentiation of female germline cysts. Development 2010, 137, 3167–3176. [Google Scholar] [CrossRef] [PubMed]
- Carreira-Rosario, A.; Bhargava, V.; Hillebrand, J.; Kollipara, R.K.; Ramaswami, M.; Buszczak, M. Repression of Pumilio protein expression by Rbfox1 promotes germ cell differentiation. Dev. Cell 2016, 36, 562–571. [Google Scholar] [CrossRef]
- Bajpai, R.; Sambrani, N.; Stadelmayer, B.; Shashidhara, L.S. Identification of a novel target of D/V signaling in Drosophila wing disc: Wg-independent function of the organizer. Gene Expr. Patterns 2004, 5, 113–121. [Google Scholar] [CrossRef]
- Usha, N.; Shashidhara, L.S. Interaction between Ataxin-2 Binding Protein 1 and Cubitus-interruptus during wing development in Drosophila. Dev. Biol. 2010, 341, 389–399. [Google Scholar] [CrossRef][Green Version]
- Guven-Ozkan, T.; Busto, G.U.; Schutte, S.S.; Cervantes-Sandoval, I.; O’Dowd, D.K.; Davis, R.L. MiR-980 is a memory suppressor microRNA that regulates the autism-susceptibility gene A2bp1. Cell Rep. 2016, 14, 1698–1709. [Google Scholar] [CrossRef][Green Version]
- Nazario-Toole, A.E.; Robalino, J.; Okrah, K.; Corrada-Bravo, H.; Mount, S.M.; Wu, L.P. The splicing factor RNA-binding Fox Protein 1 mediates the cellular immune response in Drosophila melanogaster. J. Immunol. 2018, 201, 1154–1164. [Google Scholar] [CrossRef]
- Koizumi, K.; Higashida, H.; Yoo, S.; Islam, M.S.; Ivanov, A.I.; Guo, V.; Pozzi, P.; Yu, S.H.; Rovescalli, A.C.; Tang, D.; et al. RNA interference screen to identify genes required for Drosophila embryonic nervous system development. Proc. Natl. Acad. Sci. USA 2007, 104, 5626–5631. [Google Scholar] [CrossRef] [PubMed]
- Tunstall, N.E.; Herr, A.; de Bruyne, M.; Warr, C.G. A screen for genes expressed in the olfactory organs of Drosophila melanogaster identifies genes involved in olfactory behaviour. PLoS ONE 2012, 7, e35641. [Google Scholar] [CrossRef] [PubMed]
- Giraldo-Calderon, G.I.; Emrich, S.J.; MacCallum, R.M.; Maslen, G.; Dialynas, E.; Topalis, P.; Ho, N.; Gesing, S.; VectorBase, C.; Madey, G.; et al. VectorBase: An updated bioinformatics resource for invertebrate vectors and other organisms related with human diseases. Nucleic Acids Res. 2015, 43, D707–D713. [Google Scholar] [CrossRef]
- Sayers, E.W.; Beck, J.; Bolton, E.E.; Bourexis, D.; Brister, J.R.; Canese, K.; Comeau, D.C.; Funk, K.; Kim, S.; Klimke, W.; et al. Database resources of the National Center for Biotechnology Information. Nucleic Acids Res. 2021, 49, D10–D17. [Google Scholar] [CrossRef]
- Sousa-Paula, L.C.; Otranto, D.; Dantas-Torres, F. Lutzomyia longipalpis (Sand Fly). Trends Parasitol. 2020, 36, 796–797. [Google Scholar] [CrossRef] [PubMed]
- Fiorenzano, J.M.; Koehler, P.G.; Xue, R.D. Attractive toxic sugar bait (ATSB) for control of mosquitoes and its impact on non-target organisms: A review. Int. J. Environ. Res. Public Health 2017, 14, 398. [Google Scholar] [CrossRef]
- Faraji, A.; Unlu, I. The eye of the tiger, the thrill of the gight: Effective larval and adult control measures against the Asian tiger mosquito, Aedes albopictus (Diptera: Culicidae), in North America. J. Med. Entomol. 2016, 53, 1029–1047. [Google Scholar] [CrossRef]
- Duman-Scheel, M. Saccharomyces cerevisiae (baker’s yeast) as an interfering RNA expression and delivery system. Curr. Drug Targets 2019, 20, 942–952. [Google Scholar] [CrossRef]
- Mysore, K.; Hapairai, L.K.; Li, P.; Roethele, J.B.; Sun, L.; Igiede, J.; Misenti, J.K.; Duman-Scheel, M. A functional requirement for sex-determination M/m locus region lncRNA genes in Aedes aegypti female larvae. Sci. Rep. 2021, 11, 10657. [Google Scholar] [CrossRef]
- Mysore, K.; Sun, L.; Roethele, J.B.; Li, P.; Igiede, J.; Misenti, J.K.; Duman-Scheel, M. A conserved female-specific larval requirement for MtnB function facilitates sex separation in multiple species of disease vector mosquitoes. Parasit. Vectors 2021, 14, 338. [Google Scholar] [CrossRef]
- Skeath, J.B.; Doe, C.Q. Sanpodo and Notch act in opposition to Numb to distinguish sibling neuron fates in the Drosophila CNS. Development 1998, 125, 1857–1865. [Google Scholar] [CrossRef] [PubMed]
- Mulla, M.S. Larvicides and larvicidal formulations for the control of Culex pipiens fatigans. Bull. World Health Organ. 1967, 37, 311–315. [Google Scholar] [PubMed]
- World Health Organization. Larval Source Management: A Supplementary Measure for Malaria Vector Control: An Operational Manual; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Center for Disease Control. Larvicides. Available online: https://www.cdc.gov/mosquitoes/mosquito-control/community/larvicides.html (accessed on 3 July 2021).
- Afrane, Y.A.; Mweresa, N.G.; Wanjala, C.L.; Gilbreath III, T.M.; Zhou, G.; Lee, M.C.; Githeko, A.K.; Yan, G. Evaluation of long-lasting microbial larvicide for malaria vector control in Kenya. Malar. J. 2016, 15, 577. [Google Scholar] [CrossRef] [PubMed]
- Kahindi, S.C.; Muriu, S.; Derua, Y.A.; Wang, X.; Zhou, G.; Lee, M.C.; Mwangangi, J.; Atieli, H.; Githeko, A.K.; Yan, G. Efficacy and persistence of long-lasting microbial larvicides against malaria vectors in western Kenya highlands. Parasit. Vectors 2018, 11, 438. [Google Scholar] [CrossRef] [PubMed]
- Derua, Y.A.; Kweka, E.J.; Kisinza, W.N.; Githeko, A.K.; Mosha, F.W. Bacterial larvicides used for malaria vector control in sub-Saharan Africa: Review of their effectiveness and operational feasibility. Parasit. Vectors 2019, 12, 426. [Google Scholar] [CrossRef]
- Derua, Y.A.; Kahindi, S.C.; Mosha, F.W.; Kweka, E.J.; Atieli, H.E.; Zhou, G.; Lee, M.C.; Githeko, A.K.; Yan, G. Susceptibility of Anopheles gambiae complex mosquitoes to microbial larvicides in diverse ecological settings in western Kenya. Med. Vet. Entomol. 2019, 33, 220–227. [Google Scholar] [CrossRef]
- Getachew, D.; Balkew, M.; Tekie, H. Anopheles larval species composition and characterization of breeding habitats in two localities in the Ghibe River Basin, southwestern Ethiopia. Malar. J. 2020, 19, 65. [Google Scholar] [CrossRef]
- Khallaayoune, K.; Qualls, W.A.; Revay, E.E.; Allan, S.A.; Arheart, K.L.; Kravchenko, V.D.; Xue, R.D.; Schlein, Y.; Beier, J.C.; Muller, G.C. Attractive toxic sugar baits: Control of mosquitoes with the low-risk active ingredient dinotefuran and potential impacts on nontarget organisms in Morocco. Environ. Entomol. 2013, 42, 1040–1045. [Google Scholar] [CrossRef]
- Qualls, W.A.; Muller, G.C.; Revay, E.E.; Allan, S.A.; Arheart, K.L.; Beier, J.C.; Smith, M.L.; Scott, J.M.; Kravchenko, V.D.; Hausmann, A.; et al. Evaluation of attractive toxic sugar bait (ATSB)-barrier for control of vector and nuisance mosquitoes and its effect on non-target organisms in sub-tropical environments in Florida. Acta Trop. 2014, 131, 104–110. [Google Scholar] [CrossRef]
- Fulcher, A.; Scott, J.M.; Qualls, W.A.; Muller, G.C.; Xue, R.D. Attractive toxic sugar baits mixed with pyriproxyfen sprayed on plants against adult and larval Aedes albopictus (Diptera: Culicidae). J. Med. Entomol. 2014, 51, 896–899. [Google Scholar] [CrossRef]
- Revay, E.E.; Muller, G.C.; Qualls, W.A.; Kline, D.L.; Naranjo, D.P.; Arheart, K.L.; Kravchenko, V.D.; Yefremova, Z.; Hausmann, A.; Beier, J.C.; et al. Control of Aedes albopictus with attractive toxic sugar baits (ATSB) and potential impact on non-target organisms in St. Augustine, Florida. Parasitol. Res. 2014, 113, 73–79. [Google Scholar] [CrossRef]
- Junnila, A.; Revay, E.E.; Muller, G.C.; Kravchenko, V.; Qualls, W.A.; Xue, R.D.; Allen, S.A.; Beier, J.C.; Schlein, Y. Efficacy of attractive toxic sugar baits (ATSB) against Aedes albopictus with garlic oil encapsulated in beta-cyclodextrin as the active ingredient. Acta Trop. 2015, 152, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Seeger, K.E.; Scott, J.M.; Muller, G.C.; Qualls, W.A.; Xue, R.D. Effect of common species of Florida landscaping plants on the efficacy of attractive toxic sugar baits against Aedes albopictus. J. Am. Mosq. Control. Assoc. 2017, 33, 139–141. [Google Scholar] [CrossRef]
- Sippy, R.; Rivera, G.E.; Sanchez, V.; Heras, F.; Morejon, B.; Beltran, E.; Hikida, R.S.; Lopez-Latorre, M.A.; Aguirre, A.; Stewart-Ibarra, A.M.; et al. Ingested insecticide to control Aedes aegypti: Developing a novel dried attractive toxic sugar bait device for intra-domiciliary control. Parasit. Vectors 2020, 13, 78. [Google Scholar] [CrossRef] [PubMed]
- Muller, G.C.; Junnila, A.; Schlein, Y. Effective control of adult Culex pipiens by spraying an attractive toxic sugar bait solution in the vegetation near larval habitats. J. Med. Entomol. 2010, 47, 63–66. [Google Scholar] [CrossRef]
- Muller, G.C.; Junnila, A.; Qualls, W.; Revay, E.E.; Kline, D.L.; Allan, S.; Schlein, Y.; Xue, R.D. Control of Culex quinquefasciatus in a storm drain system in Florida using attractive toxic sugar baits. Med. Vet. Entomol. 2010, 24, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Qualls, W.A.; Scott-Fiorenzano, J.; Muller, G.C.; Arheart, K.L.; Beier, J.C.; Xue, R.D. Evaluation and adaptation of attractive toxic sugar baits for Culex tarsalis and Culex quinquefasciatus control in the Coachella Valley, Southern California. J. Am. Mosq. Control. Assoc. 2016, 32, 292–299. [Google Scholar] [CrossRef]
- Beier, J.C.; Muller, G.C.; Gu, W.; Arheart, K.L.; Schlein, Y. Attractive toxic sugar bait (ATSB) methods decimate populations of Anopheles malaria vectors in arid environments regardless of the local availability of favoured sugar-source blossoms. Malar. J. 2012, 11, 31. [Google Scholar] [CrossRef]
- Qualls, W.A.; Muller, G.C.; Traore, S.F.; Traore, M.M.; Arheart, K.L.; Doumbia, S.; Schlein, Y.; Kravchenko, V.D.; Xue, R.D.; Beier, J.C. Indoor use of attractive toxic sugar bait (ATSB) to effectively control malaria vectors in Mali, West Africa. Malar. J. 2015, 14, 301. [Google Scholar] [CrossRef]
- Tenywa, F.C.; Kambagha, A.; Saddler, A.; Maia, M.F. The development of an ivermectin-based attractive toxic sugar bait (ATSB) to target Anopheles arabiensis. Malar. J. 2017, 16, 338. [Google Scholar] [CrossRef]
- Furnival-Adams, J.E.C.; Camara, S.; Rowland, M.; Koffi, A.A.; Ahoua Alou, L.P.; Oumbouke, W.A.; N’Guessan, R. Indoor use of attractive toxic sugar bait in combination with long-lasting insecticidal net against pyrethroid-resistant Anopheles gambiae: An experimental hut trial in Mbe, central Cote d’Ivoire. Malar. J. 2020, 19, 11. [Google Scholar] [CrossRef] [PubMed]
- Traore, M.M.; Junnila, A.; Traore, S.F.; Doumbia, S.; Revay, E.E.; Kravchenko, V.D.; Schlein, Y.; Arheart, K.L.; Gergely, P.; Xue, R.D.; et al. Large-scale field trial of attractive toxic sugar baits (ATSB) for the control of malaria vector mosquitoes in Mali, West Africa. Malar. J. 2020, 19, 72. [Google Scholar] [CrossRef] [PubMed]
- Diarra, R.A.; Traore, M.M.; Junnila, A.; Traore, S.F.; Doumbia, S.; Revay, E.E.; Kravchenko, V.D.; Schlein, Y.; Arheart, K.L.; Gergely, P.; et al. Testing configurations of attractive toxic sugar bait (ATSB) stations in Mali, West Africa, for improving the control of malaria parasite transmission by vector mosquitoes and minimizing their effect on non-target insects. Malar. J. 2021, 20, 184. [Google Scholar] [CrossRef]
- MonSanto. Docket ID: EPA-HQ-OPP-2013-0485. Available online: https://www.apsnet.org/members/outreach/ppb/Documents/Monsanto%20posted%20written%20comment%20Jan%202014.pdf (accessed on 5 May 2014).
- Casanova, C.; Andrighetti, M.T.; Sampaio, S.M.; Marcoris, M.L.; Colla-Jacques, F.E.; Prado, A.P. Larval breeding sites of Lutzomyia longipalpis (Diptera: Psychodidae) in visceral leishmaniasis endemic urban areas in Southeastern Brazil. PLoS Negl. Trop. Dis. 2013, 7, e2443. [Google Scholar] [CrossRef]
- Vivero, R.J.; Torres-Gutierrez, C.; Bejarano, E.E.; Pena, H.C.; Estrada, L.G.; Florez, F.; Ortega, E.; Aparicio, Y.; Muskus, C.E. Study on natural breeding sites of sand flies (Diptera: Phlebotominae) in areas of Leishmania transmission in Colombia. Parasit. Vectors 2015, 8, 116. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, T.N.; Pita-Pereira, D.; Costa, S.G.; Brazil, R.P.; Moraes, C.S.; Diaz-Albiter, H.M.; Genta, F.A. Transmission blocking sugar baits for the control of Leishmania development inside sand flies using environmentally friendly beta-glycosides and their aglycones. Parasit. Vectors 2018, 11, 614. [Google Scholar] [CrossRef] [PubMed]
- McDermott, E.G.; Morris, E.K.; Garver, L.S. Sodium ascorbate as a potential toxicant in attractive sugar baits for control of adult mosquitoes (Diptera: Culicidae) and sand flies (Diptera: Psychodidae). J. Med. Entomol. 2019, 56, 1359–1367. [Google Scholar] [CrossRef]
- Clemons, A.; Mori, A.; Haugen, M.; Severson, D.W.; Duman-Scheel, M. Culturing and egg collection of Aedes aegypti. Cold Spring Harb. Protoc. 2010, 2010, pdb-prot5507. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.D.; Wong, S.; Ryan, C.P.; Whyard, S. Oral delivery of double-stranded RNA in larvae of the yellow fever mosquito, Aedes aegypti: Implications for pest mosquito control. J. Insect Sci. 2013, 13, 69. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Laboratory and Field Testing of Mosquito Larvicides; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]
- Tomchaney, M.; Mysore, K.; Sun, L.; Li, P.; Emrich, S.J.; Severson, D.W.; Duman-Scheel, M. Examination of the genetic basis for sexual dimorphism in the Aedes aegypti (dengue vector mosquito) pupal brain. Biol. Sex. Differ. 2014, 5, 10. [Google Scholar] [CrossRef]
- van Dijken, J.P.; Bauer, J.; Brambilla, L.; Duboc, P.; Francois, J.M.; Gancedo, C.; Giuseppin, M.L.; Heijnen, J.J.; Hoare, M.; Lange, H.C.; et al. An interlaboratory comparison of physiological and genetic properties of four Saccharomyces cerevisiae strains. Enzyme Microb. Technol. 2000, 26, 706–714. [Google Scholar] [CrossRef]
- Mysore, K.; Hapairai, L.K.; Wei, N.; Realey, J.S.; Scheel, N.D.; Severson, D.W.; Duman-Scheel, M. Preparation and use of a yeast shRNA delivery system for gene silencing in mosquito larvae. Methods Mol. Biol. 2019, 1858, 213–231. [Google Scholar] [CrossRef]
- Patel, N.H. In situ hybridization to whole mount Drosophila embryos. In A Laboratory Guide to RNA: Isolation, Analysis, and Synthesis; Krieg, P.A., Ed.; Wiley-Liss: New York, NY, USA, 1996; pp. 357–370. [Google Scholar]
- Haugen, M.; Tomchaney, M.; Kast, K.; Flannery, E.; Clemons, A.; Jacowski, C.; Simanton Holland, W.; Le, C.; Severson, D.; Duman-Scheel, M. Whole-mount in situ hybridization for analysis of gene expression during Aedes aegypti development. Cold Spring Harb. Protoc. 2010, 2010, pdb-prot5509. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2019, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Mysore, K.; Sun, L.; Tomchaney, M.; Sullivan, G.; Adams, H.; Piscoya, A.S.; Severson, D.W.; Syed, Z.; Duman-Scheel, M. siRNA-Mediated silencing of doublesex during female development of the dengue vector mosquito Aedes aegypti. PLoS Negl. Trop. Dis. 2015, 9, e0004213. [Google Scholar] [CrossRef]
- Hunter, W.; Ellis, J.; Vanengelsdorp, D.; Hayes, J.; Westervelt, D.; Glick, E.; Williams, M.; Sela, I.; Maori, E.; Pettis, J.; et al. Large-scale field application of RNAi technology reducing Israeli acute paralysis virus disease in honey bees (Apis mellifera, Hymenoptera: Apidae). PloS Pathog. 2010, 6, e1001160. [Google Scholar] [CrossRef] [PubMed]


| Experiment 1 | % Mortality | p Value Control vs. Treatment | n |
|---|---|---|---|
| Larval soaking | |||
| Control siRNA | 0 ± 0 | 4.4−13 | 40 |
| Rbfox1 siRNA | 73 ± 2.5 | ||
| Adult microinjection | |||
| Control siRNA | 5 | 4.0−3 | 20 |
| Rbfox1 siRNA | 45 | ||
| ATSB/siRNA feeding | |||
| Control siRNA | 8 ± 5.3 | 3.5−10 | 37 |
| Rbfox1 siRNA | 77 ± 7.1 | 42 |
| Feeding Rate (%) | ||||
|---|---|---|---|---|
| Experiment 1 | Species | Control | Rbfox1.457 | n |
| siRNA ATSB | A. aegypti | 57 ± 7 | 63 ± 8.5 | 65 |
| Yeast ATSB | A. aegypti | 100 ± 0 | 100 ± 0 | 150 |
| A. gambiae | 100 ± 0 | 100 ± 0 | 75 | |
| C. quinquefasciatus | 100 ± 0 | 100 ± 0 | 225 | |
| A. albopictus | 89 ± 0.5 | 91 ± 1 | 225 | |
| Test Organism 1 | Control | Rbfox1.457 | n |
|---|---|---|---|
| D. magna adults | 100 ± 0 | 98 ± 3.5 | 40 |
| D. melanogaster larvae | 100 ± 0 | 100 ± 0 | 60 |
| D. melanogaster adults | 100 ± 0 | 99 ± 1 | 60 |
| H. convergens adults | 90 ± 0 | 90 ± 7 | 20 |
| O. fasciatus adults | 80 ± 7 | 77.5 ± 18 | 40 |
| T. castaneum adults | 100 ± 0 | 100 ± 0 | 60 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mysore, K.; Sun, L.; Hapairai, L.K.; Wang, C.-W.; Roethele, J.B.; Igiede, J.; Scheel, M.P.; Scheel, N.D.; Li, P.; Wei, N.; et al. A Broad-Based Mosquito Yeast Interfering RNA Pesticide Targeting Rbfox1 Represses Notch Signaling and Kills Both Larvae and Adult Mosquitoes. Pathogens 2021, 10, 1251. https://doi.org/10.3390/pathogens10101251
Mysore K, Sun L, Hapairai LK, Wang C-W, Roethele JB, Igiede J, Scheel MP, Scheel ND, Li P, Wei N, et al. A Broad-Based Mosquito Yeast Interfering RNA Pesticide Targeting Rbfox1 Represses Notch Signaling and Kills Both Larvae and Adult Mosquitoes. Pathogens. 2021; 10(10):1251. https://doi.org/10.3390/pathogens10101251
Chicago/Turabian StyleMysore, Keshava, Longhua Sun, Limb K. Hapairai, Chien-Wei Wang, Joseph B. Roethele, Jessica Igiede, Max P. Scheel, Nicholas D. Scheel, Ping Li, Na Wei, and et al. 2021. "A Broad-Based Mosquito Yeast Interfering RNA Pesticide Targeting Rbfox1 Represses Notch Signaling and Kills Both Larvae and Adult Mosquitoes" Pathogens 10, no. 10: 1251. https://doi.org/10.3390/pathogens10101251
APA StyleMysore, K., Sun, L., Hapairai, L. K., Wang, C.-W., Roethele, J. B., Igiede, J., Scheel, M. P., Scheel, N. D., Li, P., Wei, N., Severson, D. W., & Duman-Scheel, M. (2021). A Broad-Based Mosquito Yeast Interfering RNA Pesticide Targeting Rbfox1 Represses Notch Signaling and Kills Both Larvae and Adult Mosquitoes. Pathogens, 10(10), 1251. https://doi.org/10.3390/pathogens10101251






