The Effect of Ultra-Fast Heating on the Microstructure, Grain Size and Texture Evolution of a Commercial Low-C, Medium-Mn DP Steel
Abstract
1. Introduction
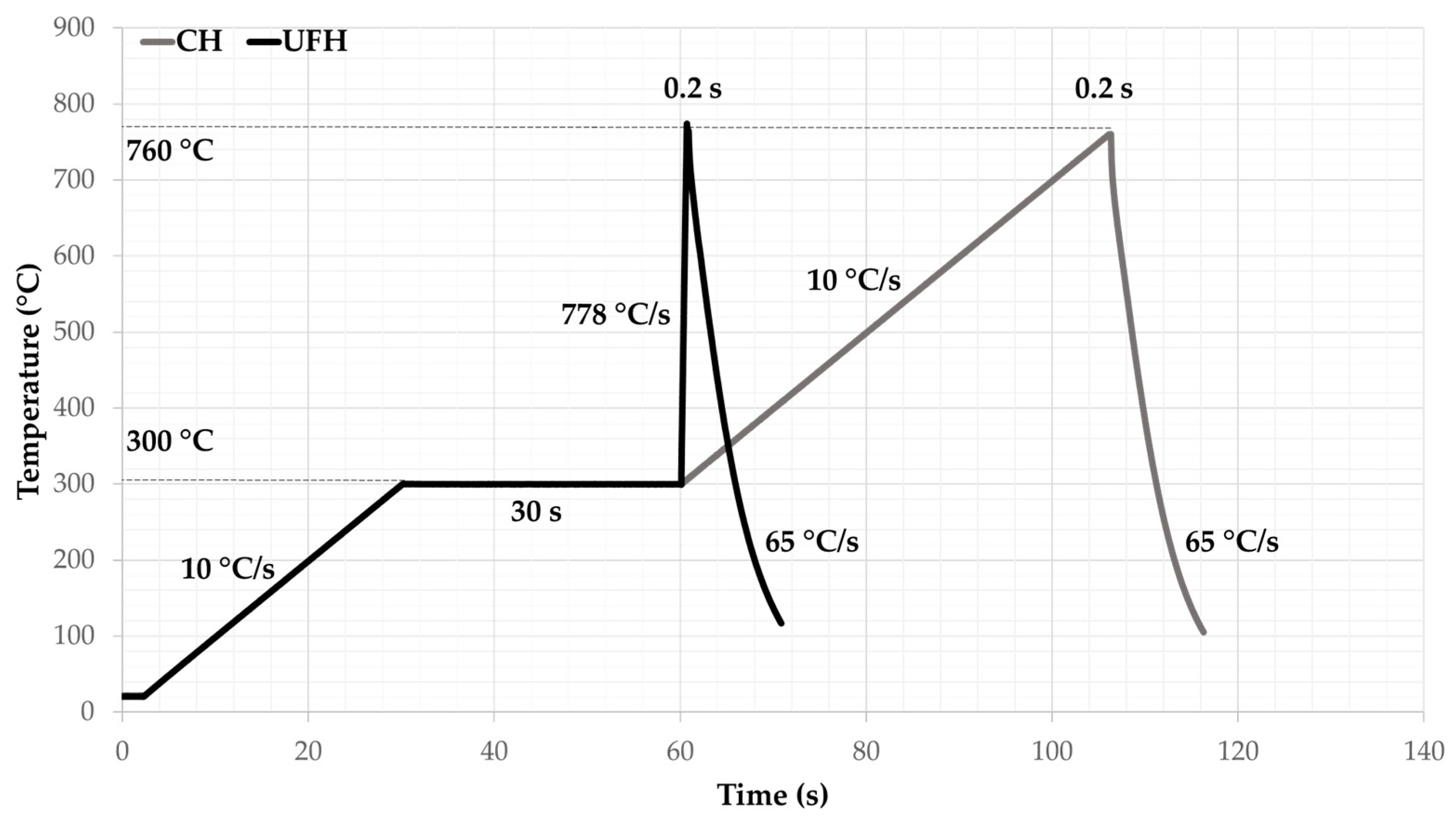
2. Materials and Methods
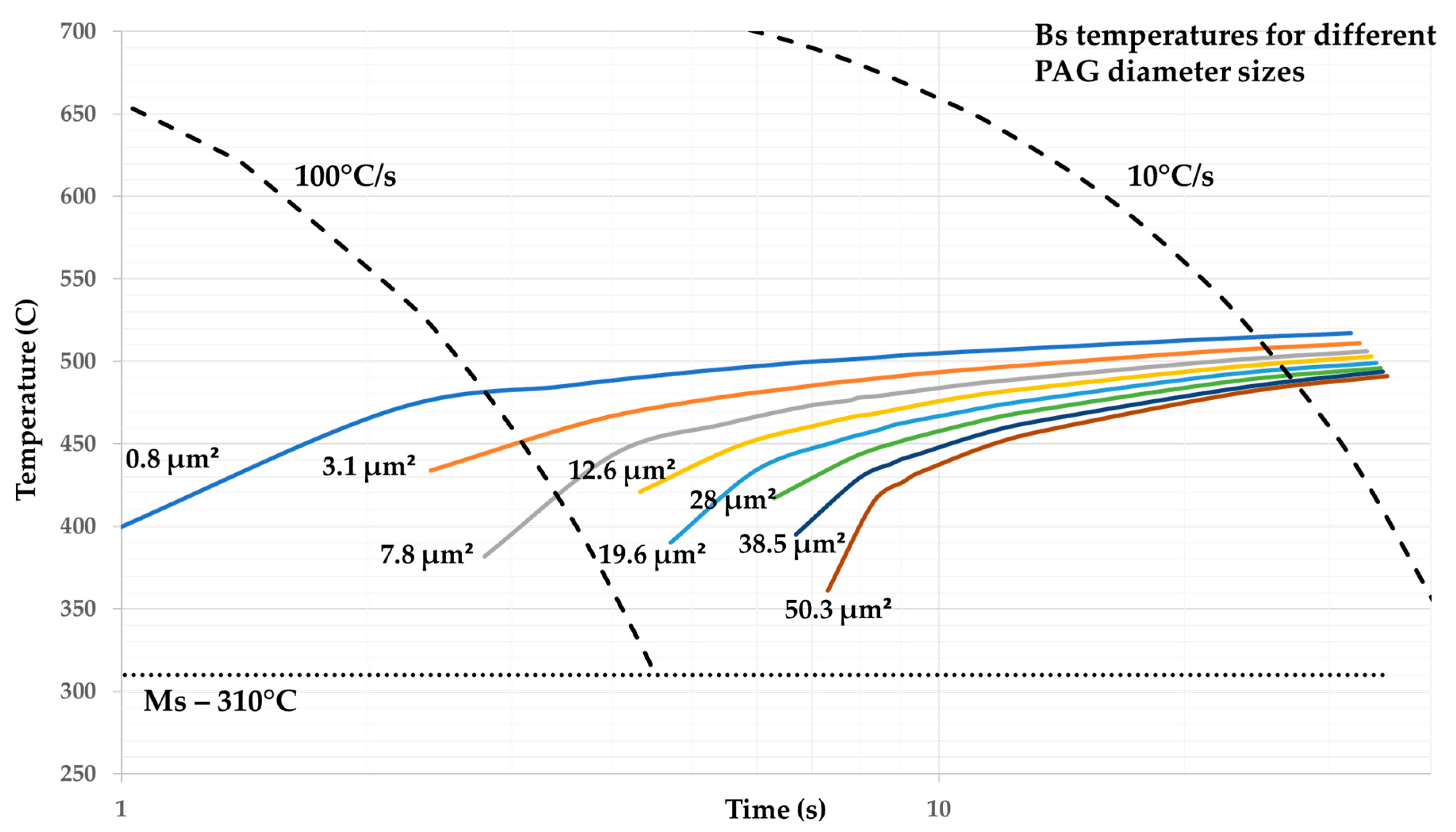
3. Results and Discussion
3.1. Microstructure Comparison
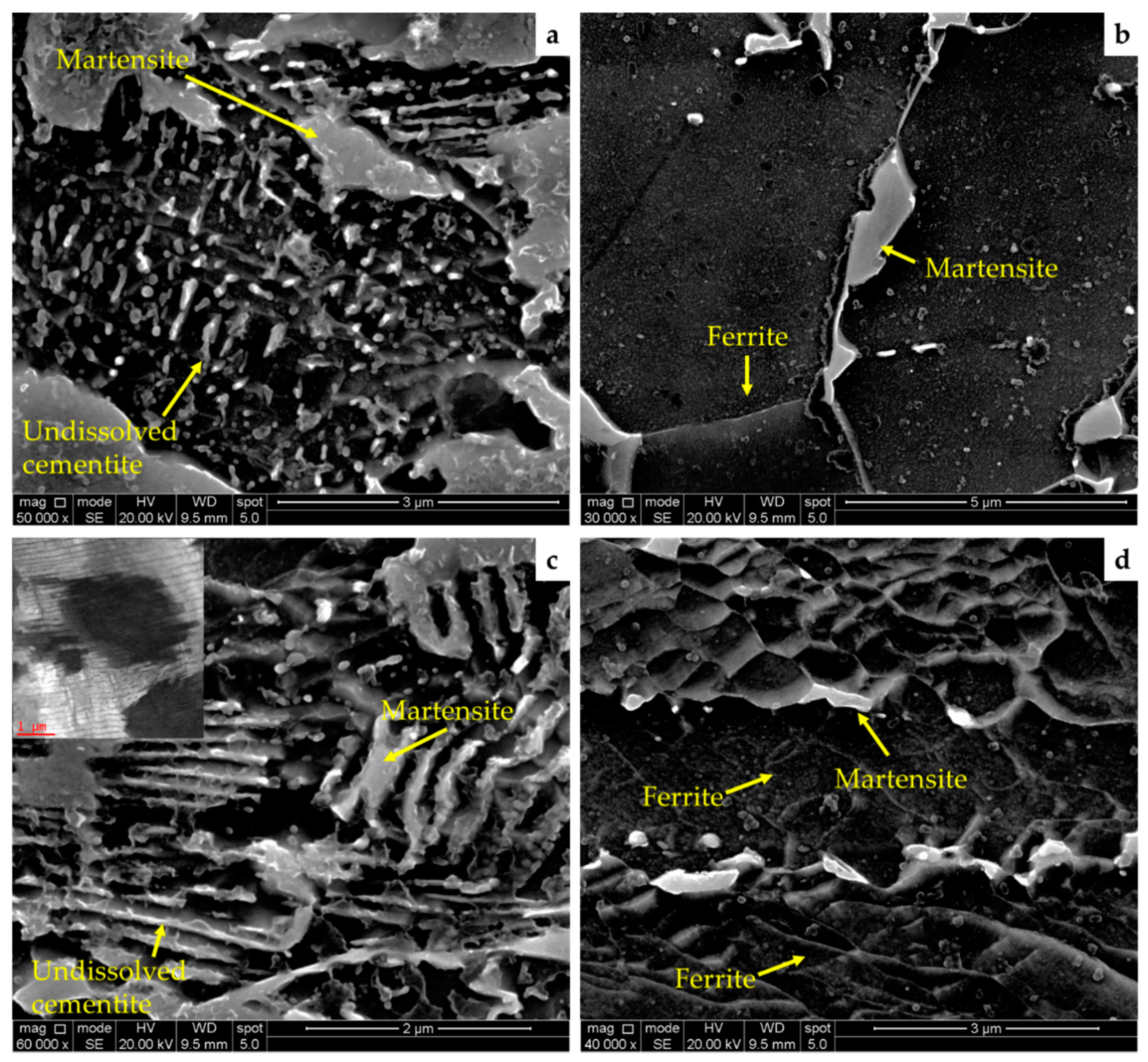
3.2. Microstructural Constituents Analysis
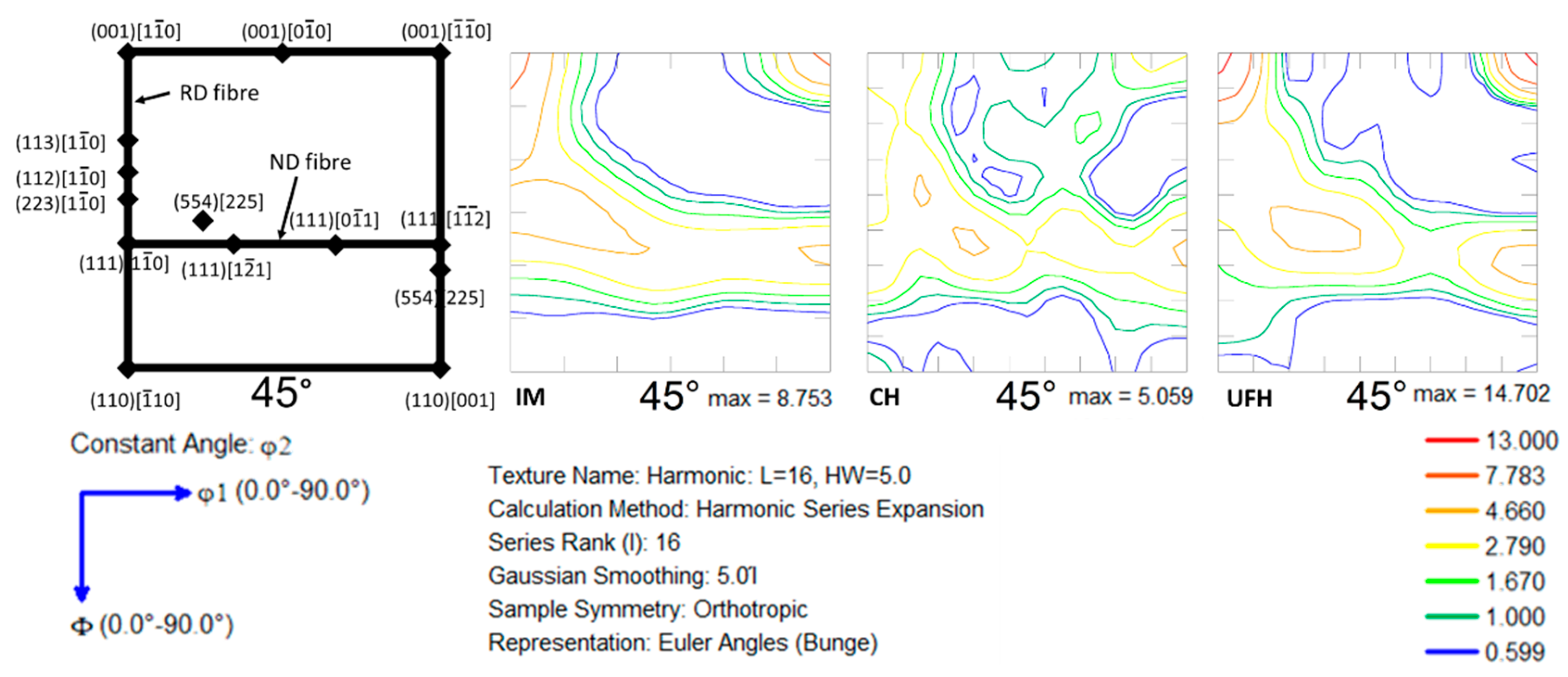
3.3. Texture and Recrystallization Analysis
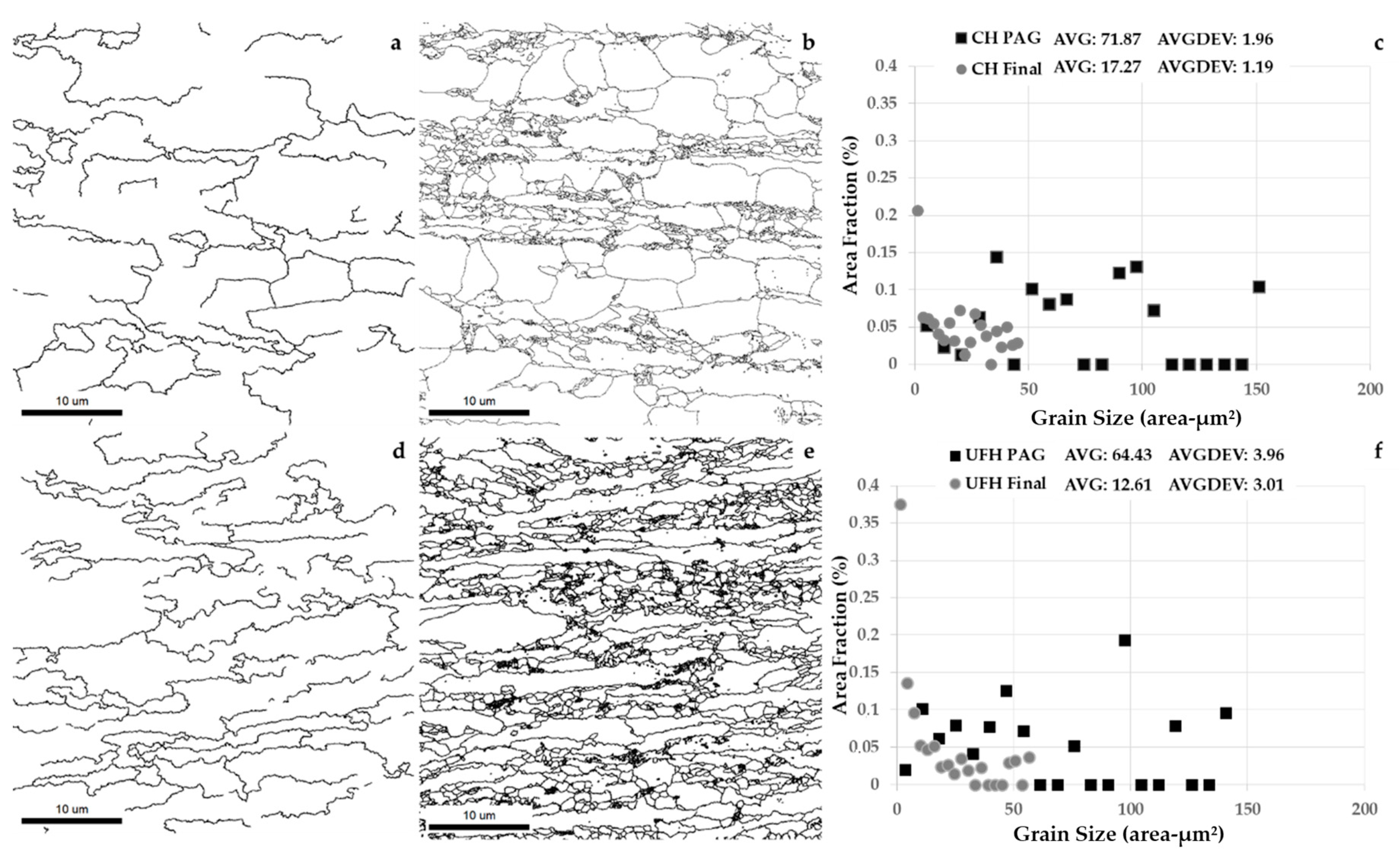
3.4. Grain Size Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gaspar, M.; Balogh, A. GMAW experiments for advanced (Q+T) high strength steels. Prod. Prof. Syst. 2013, 6, 9–24. [Google Scholar]
- Samei, J.; Zhou, L.; Kang, J.; Wilkinson, D.S. Microstructural analysis of ductility and fracture in fine-grained and ultrafine-grained vanadium-added DP1300 steels. Int. J. Plast. 2019, 117, 58–70. [Google Scholar] [CrossRef]
- Zaefferer, S.; Ohlert, J.; Bleck, W. A study of microstructure, transformation mechanisms and correlation between microstructure and mechanical properties of a low alloyed TRIP steel. Acta Mater. 2004, 52, 2765–2778. [Google Scholar] [CrossRef]
- Speer, J.; Matlock, D.; De Cooman, B.; Schroth, J. Carbon partitioning into austenite after martensite transformation. Acta Mater. 2003, 51, 2611–2622. [Google Scholar] [CrossRef]
- Cola, G. Properties of bainite nucleated by water quenching in 80 ms. In Proceedings of the 1st International Symposium on Steel Science, Kyoto, Japan, 16–19 May 2007; Furuhara, T., Tsuzaki, K., Eds.; Iron and Steel Institute of Japan: Tokyo, Japan, 2007; pp. 187–190. [Google Scholar]
- Lolla, S.V.T. Understanding Microstructure Evolution in Rapid Thermal Processing of AISI 8620 Steel. Master’s Thesis, The Ohio State University, Columbus, OH, USA, 2009. [Google Scholar]
- Lolla, S.V.T.; Alexandrov, B.; Babu, S.; Cola, G. Towards Understanding the Microstructure Development during Heating and Cooling of Steels. In Proceedings of the International Conference on Processing and Manufacturing of Advanced Materials-THERMEC’2009, Berlin, Germany, 25–29 August 2009. [Google Scholar]
- Lolla, T.; Cola, G.; Narayanan, B.; Alexandrov, B.; Babu, S.S. Development of rapid heating and cooling (flash processing) process to produce advanced high strength steel microstructures. Mater. Sci. Technol. 2011, 27, 863–875. [Google Scholar] [CrossRef]
- Papaefthymiou, S.; Banis, A.; Bouzouni, M.; Petrov, R.H. Effect of Ultra-Fast Heat Treatment on the Subsequent Formation of Mixed Martensitic/Bainitic Microstructure with Carbides in a CrMo Medium Carbon Steel. Metals 2019, 9, 312. [Google Scholar] [CrossRef]
- Lan, L.; Chang, Z.; Fan, P. Exploring the Difference in Bainite Transformation with Varying the Prior Austenite Grain Size in Low Carbon Steel. Metals 2018, 8, 988. [Google Scholar] [CrossRef]
- Banis, A.; Papaefthymiou, S. Microstructure Characterization of an Ultra-Fast Heated Medium Carbon Chromium-Manganese High Strength Steel. Int. J. Met. Met. Phys. 2018, 3, 1–14. [Google Scholar] [CrossRef]
- Cerda, F.C.; Schulz, B.; Celentano, D.; Monsalve, A.; Sabirov, I.; Petrov, R. Exploring the microstructure and tensile properties of cold-rolled low and medium carbon steels after ultrafast heating and quenching. Mater. Sci. Eng. A 2019, 745, 509–516. [Google Scholar] [CrossRef]
- Papaefthymiou, S.; Bouzouni, M.; Petrov, R.H. Study of Carbide Dissolution and Austenite Formation during Ultra–Fast Heating in Medium Carbon Chromium Molybdenum Steel. Metals 2018, 8, 646. [Google Scholar] [CrossRef]
- Bouzouni, M. Modeling of the Steel Microstructure Gained after the Application of an Ultra-Fast Heat Treatment. J. Nanosci. Adv. Technol. 2017, 2, 15–19. [Google Scholar] [CrossRef]
- Bouzouni, M.; Papaefthymiou, S. Preliminary Study of Carbide Dissolution during an Ultra-Fast Heat Treatment in Chromium Molybdenum Steel. Int. J. Met. Met. Phys. 2017, 2, 1–7. [Google Scholar] [CrossRef]
- Kaluba, W.; Taillard, R.; Foct, J. The bainitic mechanism of austenite formation during rapid heating. Acta Mater. 1998, 46, 5917–5927. [Google Scholar] [CrossRef]
- Kaluba, W.; Taillard, R.; Foct, J. A reply to “discussion to “the bainitic mechanism of austenite formation during rapid heating. Scr. Mater. 2000, 42, 511–516. [Google Scholar] [CrossRef]
- Aaronson, H.; Nie, J. Discussion to “the bainitic mechanism of austenite formation during rapid heating”. Scr. Mater. 2000, 42, 505–509. [Google Scholar] [CrossRef]
- Savran, V.I. Austenite Formation in C-Mn Steel. Ph.D. Thesis, TU Delft, Delft, The Netherlands, 2009. [Google Scholar]
- Castro Cerda, F.M. Third Generation Advanced High Strength Steels via Ultrafast Heating. Ph.D. Thesis, Ghent University, Ghent, Belgium, 2017. [Google Scholar]
- Speich, G.; Szirmae, A.; Richards, M. Formation of austenite from ferrite and ferrite-carbide aggregates. Trans. TMS-AIME 1969, 245, 1063–1074. [Google Scholar]
- Judd, R.R.; Paxton, H.W. Kinetics of Austenite Formation from a Spheroidized Ferrite-Carbide Aggregates. Trans. TMS-AIME 1968, 242, 206–215. [Google Scholar]
- Liu, Z.-K.; Ågren, J. Morphology of cementite decomposition in an fe-cr-c alloy. Met. Mater. Trans. A 1991, 22, 1753–1759. [Google Scholar] [CrossRef]
- Liu, Z.-K.; Höglund, L.; Jonsson, B.; Ågren, J. An experimental and theoretical study of cementite dissolution in an Fe-Cr-C alloy. Met. Mater. Trans. A 1991, 22, 1745–1752. [Google Scholar] [CrossRef]
- Gouné, M.; Maugis, P.; Drillet, J. A Criterion for the Change from Fast to Slow Regime of Cementite Dissolution in Fe–C–Mn Steels. J. Mater. Sci. Technol. 2012, 28, 728–736. [Google Scholar] [CrossRef]
- Cerda, F.M.C.; Kestens, L.A.I.; Monsalve, A.; Petrov, R.H.; Cerda, F.C. The Effect of Ultrafast Heating in Cold-Rolled Low Carbon Steel: Recrystallization and Texture Evolution. Metals 2016, 6, 288. [Google Scholar] [CrossRef]
- Cerda, F.M.C.; Kestens, L.A.I.; Petrov, R.H. “Flash” Annealing in a Cold-Rolled Low Carbon Steel Alloyed with Cr, Mn, Mo, and Nb: Part II-Anisothermal Recrystallization and Transformation Textures. Steel Res. Int. 2018, 90, 1–13. [Google Scholar]
- Landheer, H. Nucleation of Ferrite in Austenite: The Role of Crystallography. Ph.D. Thesis, TU Delft, Delft, The Netherlands, 2010. [Google Scholar]
- Cayron, C. ARPGE: A computer program to automatically reconstruct the parent grains from electron backscatter diffraction data. J. Appl. Crystallogr. 2007, 40, 1183–1188. [Google Scholar] [CrossRef] [PubMed]
- Kirkaldy, J.S.; Venugopolan, D. Phase Transformations in Ferrous Alloys; Marder, A.R., Goldstein, J.I., Eds.; AIME: Warrendale, PA, USA, 1984; p. 125. [Google Scholar]
- Kirkaldy, J.S.; Thomson, B.A.; Baganis, E.A. Hardenability Concepts with Applications to Steel; Kirkaldy, J.S., Doane, D.V., Eds.; AIME: Warrendale, PA, USA, 1978; p. 82. [Google Scholar]
- Bhadeshia, H.K.D.H. The Driving Force for Martensitic Transformation in Steels. Met. Sci. 1981, 15, 175–177. [Google Scholar] [CrossRef]
- Tian, Y.L.; Kraft, R.W. Mechanisms of Pearlite Spheroidization. Met. Mater. Trans. A 1987, 18, 1403–1414. [Google Scholar] [CrossRef]
- Petrov, R.; Kestens, L.; Wasilkowska, A.; Houbaert, Y. Microstructure and texture of a lightly deformed TRIP-assisted steel characterized by means of the EBSD technique. Mater. Sci. Eng. A 2007, 447, 285–297. [Google Scholar] [CrossRef]
- Pinard, P.T.; Schwedt, A.; Ramazani, A.; Prahl, U.; Richter, S. Characterization of Dual-Phase Steel Microstructure by Combined Submicrometer EBSD and EPMA Carbon Measurements. Microsc. Microanal. 2013, 19, 996–1006. [Google Scholar] [CrossRef] [PubMed]
- Cerda, F.M.C.; Goulas, C.; Sabirov, I.; Papaefthymiou, S.; Monsalve, A.; Petrov, R. Microstructure, texture and mechanical properties in a low carbon steel after ultrafast heating. Mater. Sci. Eng. A 2016, 672, 108–120. [Google Scholar] [CrossRef]
- Calcagnotto, M.; Ponge, D.; Raabe, D. On the Effect of Manganese on Grain Size Stability and Hardenability in Ultra-Fine-Grained Ferrite-Martensite DualPhase Steels. Metall. Mater. Trans. A 2012, 43, 37–42. [Google Scholar] [CrossRef]
- Peranio, N.; Li, Y.; Roters, F.; Raabe, D. Microstructure and texture evolution in dual-phase steels: Competition between recovery, recrystallization, and phase transformation. Mater. Sci. Eng. A 2010, 527, 4161–4168. [Google Scholar] [CrossRef]
- Humphreys, F.J.; Hatherly, M. Recrystallization and Related Annealing Phenomena; Elsevier: Oxford, UK, 2004. [Google Scholar]
- Valdes-Tabernero, M.A.; Vercruysse, F.; Sabirov, I.; Petrov, R.H.; Monclús, M.A.; Molina-Aldareguía, J.M. Effect of Ultrafast Heating on the Properties of the Microconstituents in a Low-Carbon Steel. Met. Mater. Trans. A 2018, 49, 3145–3150. [Google Scholar] [CrossRef]
- De Knijf, D.; Puype, A.; Föjer, C.; Petrov, R. The influence of ultra-fast annealing prior to quenching and partitioning on the microstructure and mechanical properties. Mater. Sci. Eng. A 2015, 627, 182–190. [Google Scholar] [CrossRef]
- Oliveira, M.A.F.; Jorge, A.M., Jr.; Balancin, O. Influence of strain-induced nucleation on the kinetics of phase transformation in a forging steel during warm working. Scr. Mater. 2004, 50, 1157. [Google Scholar] [CrossRef]
- Porter, D.A.; Easterling, K.E. Phase Transformations in Metals and Alloys; Van Nostrand Reinhold: Wokingham, UK, 1981. [Google Scholar]
- Love, G.R. Dislocation Pipe Diffusion. Acta Metall. 1964, 12, 731–737. [Google Scholar] [CrossRef]
- Cerda, F.C.; Goulas, C.; Sabirov, I.; Kestens, L.; Petrov, R. The effect of the pre-heating stage on the microstructure and texture of a cold rolled FeCMnAlSi steel under conventional and ultrafast heating. Mater. Charact. 2017, 130, 188–197. [Google Scholar] [CrossRef]
- Meshkov, Y.Y.; Pereloma, E.V. 17—The effect of heating rate on reverse transformations in steels and Fe-Ni-based alloys. In Phase Transformations in Steels; Pereloma, E., Edmonds, D.V., Eds.; Woodhead Publishing: Cambridge, UK, 2012; pp. 581–618. [Google Scholar]








| C | Si | Mn | S | P | Cr | Mo | Nb + Ti | Fe |
|---|---|---|---|---|---|---|---|---|
| 0.14 | 0.24 | 1.7 | 0.004 | 0.014 | 0.25 | 0.25 | 0.05 | Rest |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Banis, A.; Hernandez Duran, E.; Bliznuk, V.; Sabirov, I.; Petrov, R.H.; Papaefthymiou, S. The Effect of Ultra-Fast Heating on the Microstructure, Grain Size and Texture Evolution of a Commercial Low-C, Medium-Mn DP Steel. Metals 2019, 9, 877. https://doi.org/10.3390/met9080877
Banis A, Hernandez Duran E, Bliznuk V, Sabirov I, Petrov RH, Papaefthymiou S. The Effect of Ultra-Fast Heating on the Microstructure, Grain Size and Texture Evolution of a Commercial Low-C, Medium-Mn DP Steel. Metals. 2019; 9(8):877. https://doi.org/10.3390/met9080877
Chicago/Turabian StyleBanis, Alexandros, Eliseo Hernandez Duran, Vitaliy Bliznuk, Ilchat Sabirov, Roumen H. Petrov, and Spyros Papaefthymiou. 2019. "The Effect of Ultra-Fast Heating on the Microstructure, Grain Size and Texture Evolution of a Commercial Low-C, Medium-Mn DP Steel" Metals 9, no. 8: 877. https://doi.org/10.3390/met9080877
APA StyleBanis, A., Hernandez Duran, E., Bliznuk, V., Sabirov, I., Petrov, R. H., & Papaefthymiou, S. (2019). The Effect of Ultra-Fast Heating on the Microstructure, Grain Size and Texture Evolution of a Commercial Low-C, Medium-Mn DP Steel. Metals, 9(8), 877. https://doi.org/10.3390/met9080877







