Effect of Sulfur Content on Copper Recovery in the Reduction Smelting Process
Abstract
:1. Introduction
2. Experimental Section
2.1. Preparation of Synthetic Slag
2.2. Thermodynamic Calculation Method
2.3. Experimental Method
2.4. Test and Conversion Methods
3. Results and Discussion
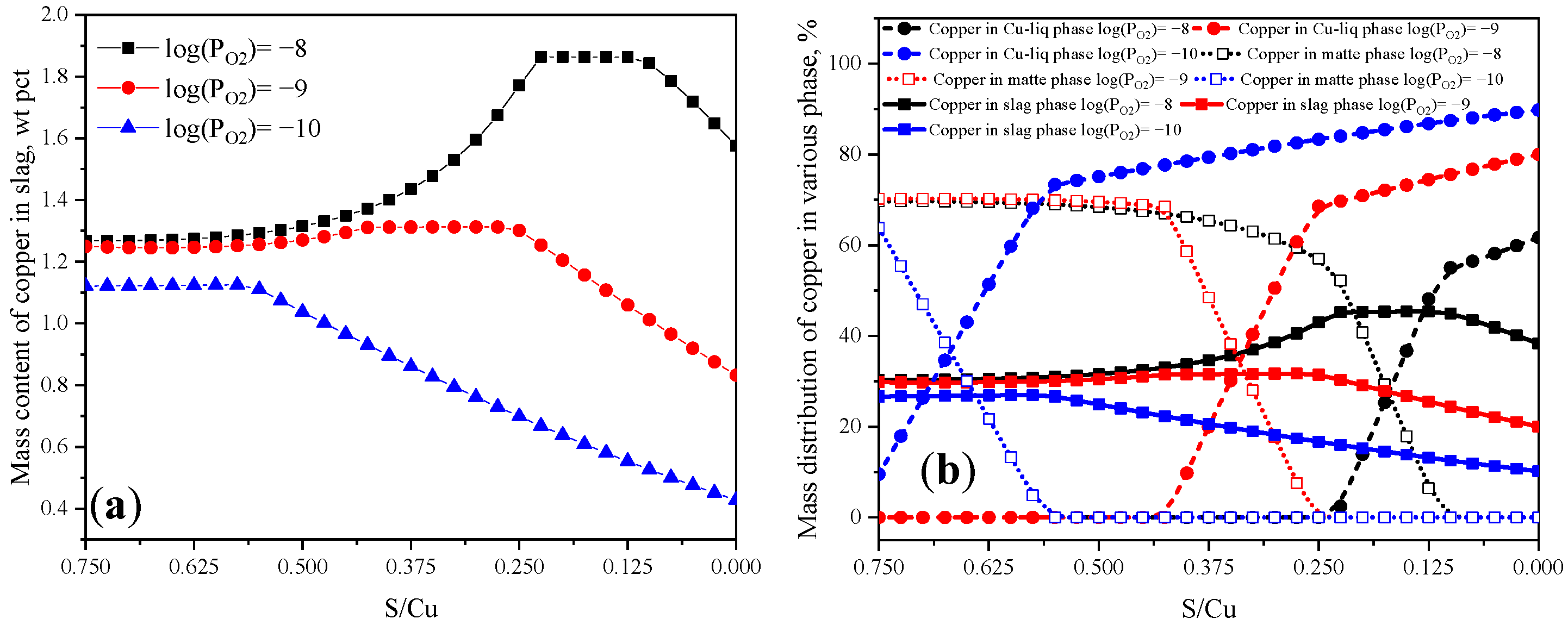
3.1. Calculation Results
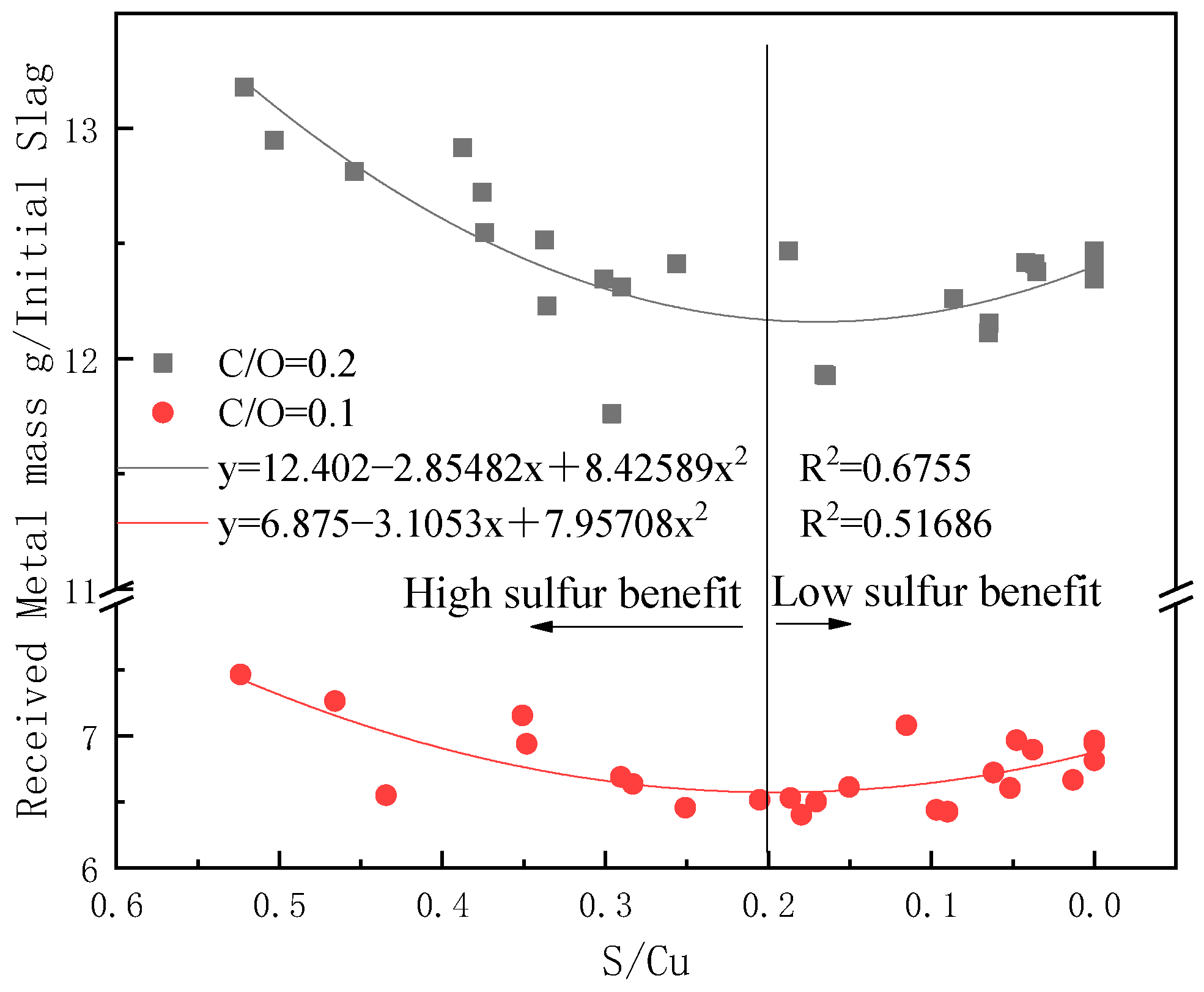
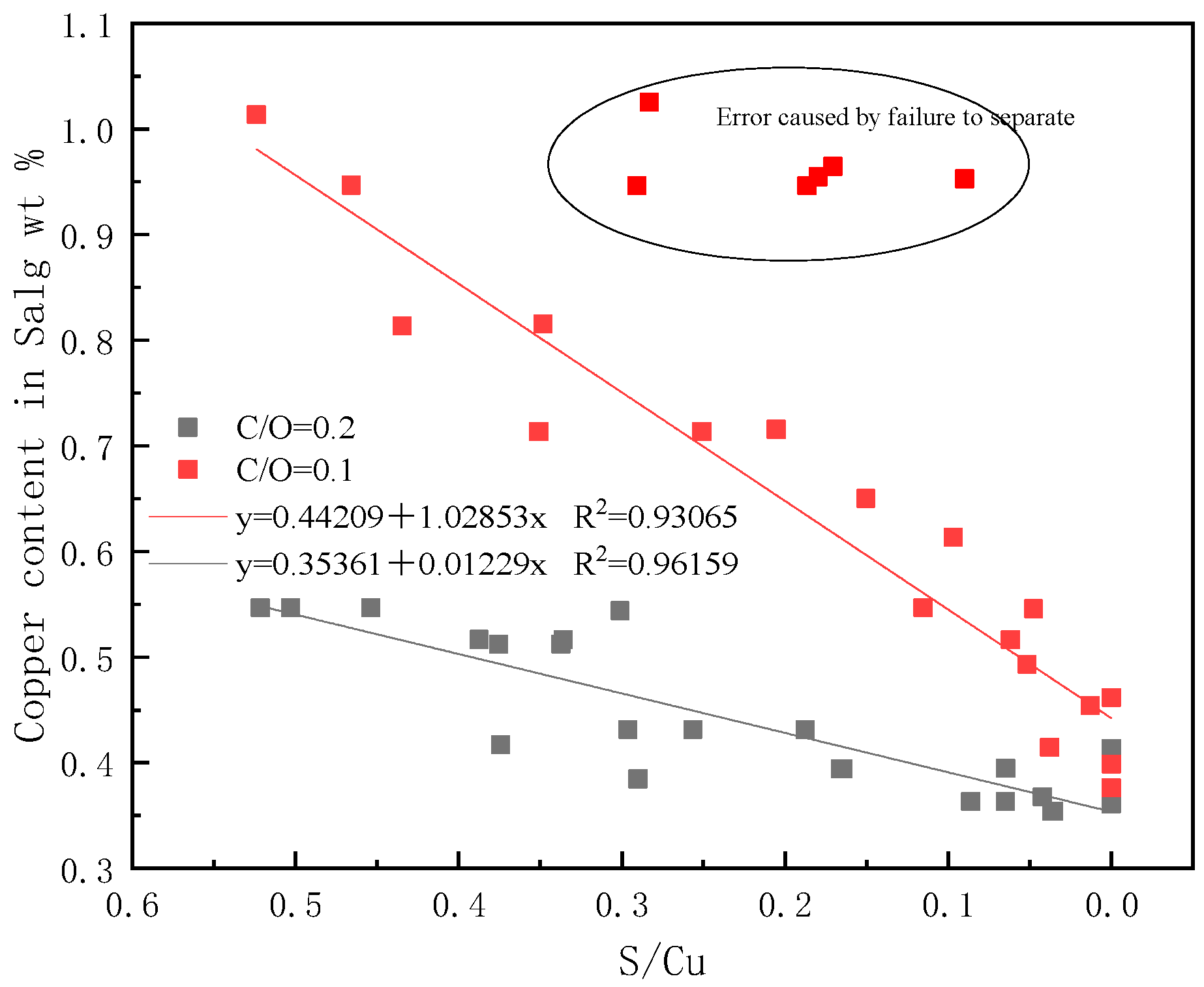
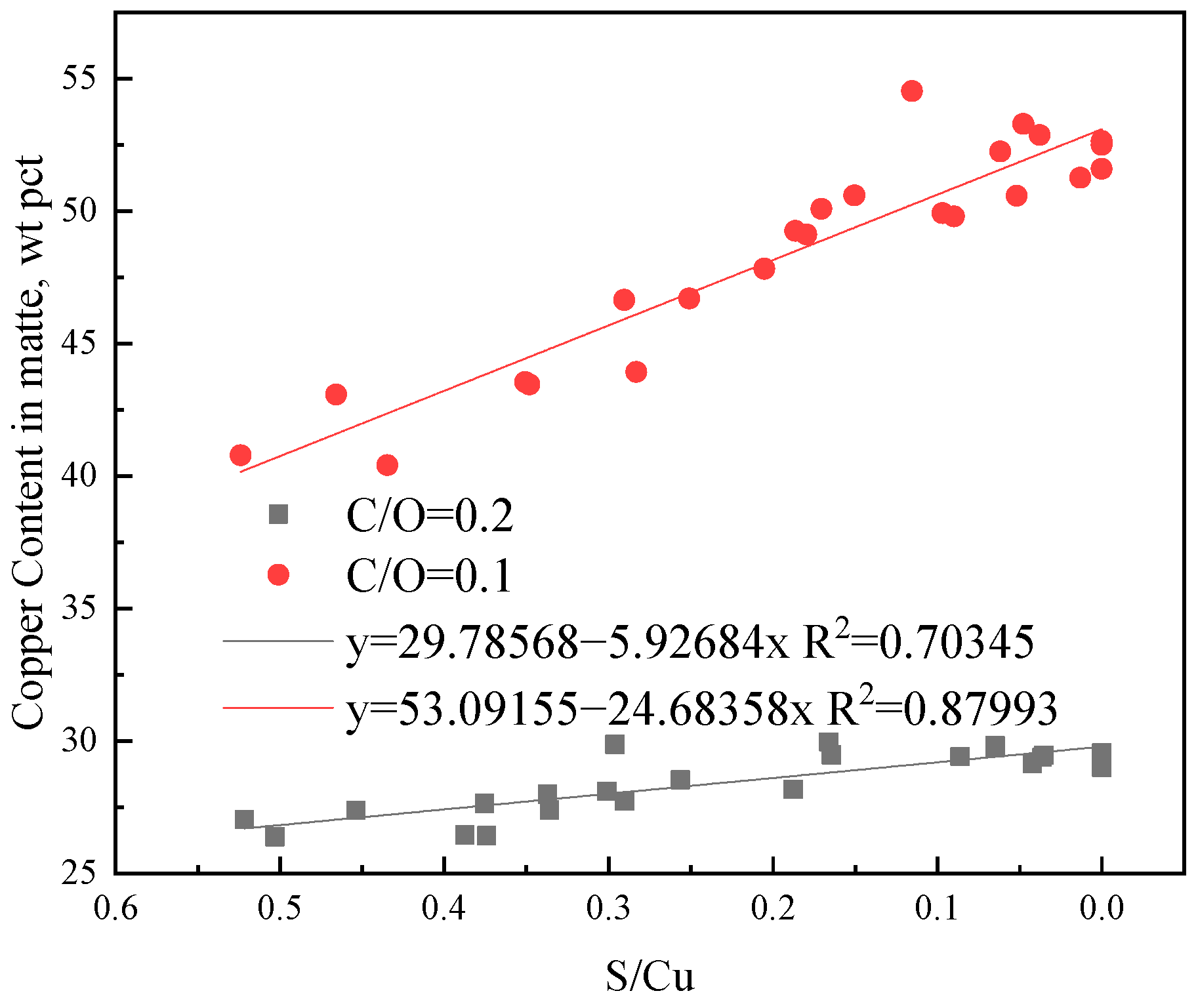
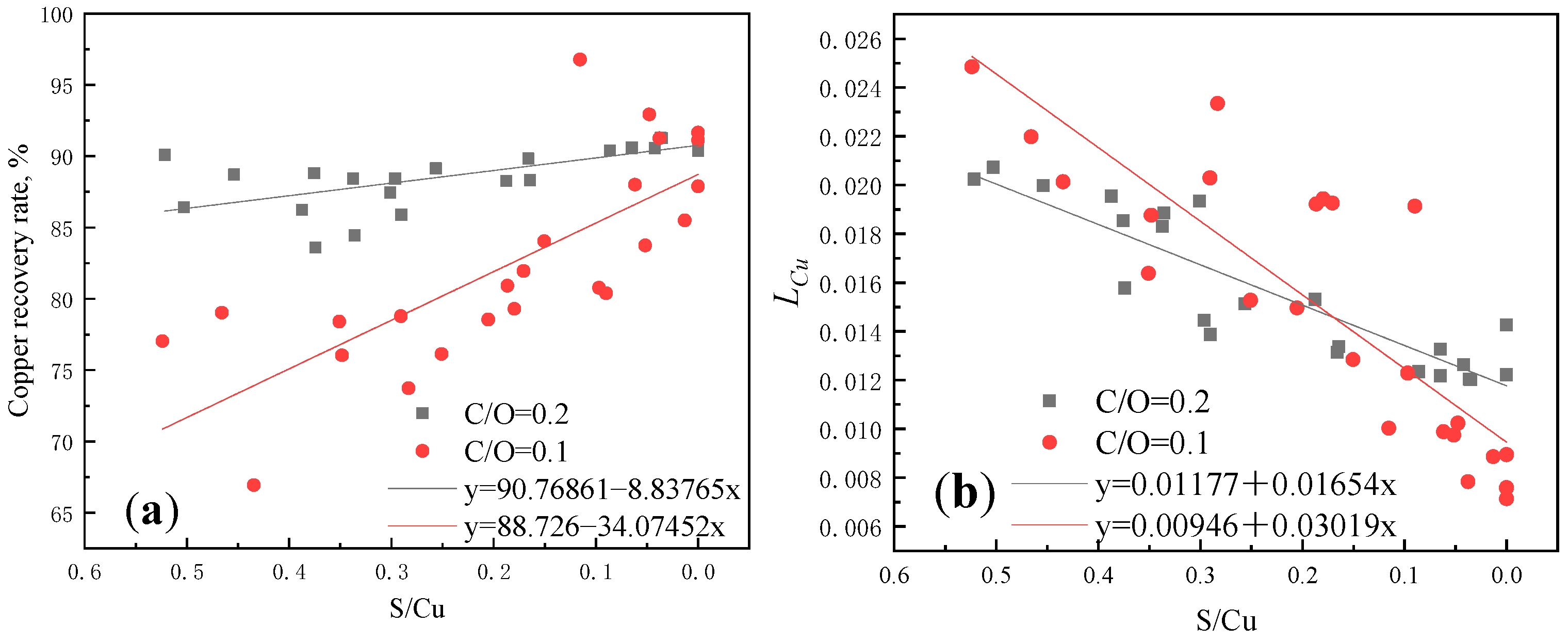
3.2. Experimental Results
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Phiri, T.C.; Singh, P.; Nikoloski, A.N. The potential for copper slag waste as a resource for a circular economy: A review—Part II. Miner. Eng. 2021, 172, 107150. [Google Scholar] [CrossRef]
- Gorai, B.; Jana, R.K. Premchand, Characteristics and utilisation of copper slag—A review. Resour. Conserv. Recycl. 2003, 39, 299–313. [Google Scholar] [CrossRef]
- Guo, X.; Tian, M.; Wang, S.; Yan, S.; Wang, Q.; Yuan, Z.; Tian, Q.; Tang, D.; Li, Z. Element Distribution in Oxygen-Enriched Bottom-Blown Smelting of High-Arsenic Copper Dross. JOM 2019, 71, 3941–3948. [Google Scholar] [CrossRef]
- Reddy, R.G.; Prabhu, V.L.; Mantha, D. Recovery of copper from copper blast furnace slag. Min. Metall. Explor. 2006, 23, 97–103. [Google Scholar] [CrossRef]
- Lin-nan, Z.; Li, Z.; Ming-yu, W.; Zhi-tong, S. Research on Selective Reducing Impoverishment Process of Copper Slag. Nonferrous Met. 2005, 57, 44–47. [Google Scholar] [CrossRef]
- Guo, Z.; Pan, J.; Zhu, D.; Zhang, F. Green and efficient utilization of waste ferric-oxide desulfurizer to clean waste copper slag by the smelting reduction-sulfurizing process. J. Clean. Prod. 2018, 199, 891–899. [Google Scholar] [CrossRef]
- Wang, H.-Y.; Zhang, G.-H.; Chou, K.-C. Recovery of high-grade copper matte by selective sulfurization of CuO–Fe2O3–SiO2–CaO system. J. Mater. Res. Technol. 2021, 13, 1676–1683. [Google Scholar] [CrossRef]
- Zhou, H.; Liu, G.; Zhang, L.; Zhou, C. Mineralogical and morphological factors affecting the separation of copper and arsenic in flash copper smelting slag flotation beneficiation process. J. Hazard. Mater. 2021, 401, 123293. [Google Scholar] [CrossRef]
- Roy, S.; Datta, A.; Rehani, S. Flotation of copper sulphide from copper smelter slag using multiple collectors and their mixtures. Int. J. Miner. Process. 2015, 143, 43–49. [Google Scholar] [CrossRef]
- Tian, H.; Guo, Z.; Pan, J.; Zhu, D.; Yang, C.; Xue, Y.; Li, S.; Wang, D. Comprehensive review on metallurgical recycling and cleaning of copper slag. Resour. Conserv. Recycl. 2021, 168, 105366. [Google Scholar] [CrossRef]
- Rajcevic, H.P.; Opie, W.R. Development of Electric Furnace Slag Cleaning at a Secondary Copper Smelter. JOM 1982, 34, 54–56. [Google Scholar] [CrossRef]
- Banks, C.C.; Harrison, D.A. The recovery of non-ferrous metals from secondary copper smelter discard slags. Can. Metall. Q. 1975, 14, 183–190. [Google Scholar] [CrossRef]
- Coursol, P.; Valencia, N.C.; Mackey, P.; Bell, S.; Davis, B. Minimization of Copper Losses in Copper Smelting Slag During Electric Furnace Treatment. JOM 2012, 64, 1305–1313. [Google Scholar] [CrossRef] [Green Version]
- Nagamori, M. Metal loss to slag: Part I. Sulfidic and oxidic dissolution of copper in fayalite slag from low grade matte. Metall. Mater. Trans. B 1974, 5, 531–538. [Google Scholar] [CrossRef]
- Nagamori, M.; Mackey, P.J.; Tarassoff, P. Copper solubility in FeO−Fe2O3−SiO2−Al2O3 slag and distribution equilibria of Pb, Bi, Sb and as between slag and metallic copper. Metall. Trans. B 1975, 6, 295–301. [Google Scholar] [CrossRef]
- Takeda, Y. Copper Solubility in SiO2-CaO-FeOx Slag Equilibrated with Matte. High Temp. Mater. Process. 2001, 20, 279–284. [Google Scholar] [CrossRef]
- Xuewei, S.; Yang, W.; Jianhang, H. The Dilution Effect and Slag Shape Structure of Copper Slag Optimized by Melting Step Reduction Method. Mater. Rev. 2019, 33, 505–511. [Google Scholar]
- Wang, H.; Zhu, R.; Dong, K.; Zhang, S.; Zhao, R.; Jiang, Z.; Lan, X. An experimental comparison: Horizontal evaluation of valuable metal extraction and arsenic emission characteristics of tailings from different copper smelting slag recovery processes. J. Hazard. Mater. 2022, 430, 128493. [Google Scholar] [CrossRef]
- Zhang, H.; Li, B.; Wei, Y.; Wang, H.; Yang, Y.; McLean, A. Reduction of Magnetite from Copper Smelting Slag in the Presence of a Graphite Rod. Metall. Mater. Trans. B 2020, 51, 2663–2672. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, R.; Chen, Q.; Wei, G.; Hu, S.; Guo, Y. Recovery of Fe, Ni, Co, and Cu from Nickel Converter Slag through Oxidation and Reduction. ISIJ Int. 2018, 58, 2191–2199. [Google Scholar] [CrossRef] [Green Version]
- Yun, W.; Rong, Z.; Qizhou, C. Experimental Study on Oxidative Desulfurization and Selective Reduction of Molten Copper Slag. In TMS 2018: 9th International Symposium on High-Temperature Metallurgical Processing; The Minerals, Metals & Materials Series; Springer: Berlin/Heidelberg, Germany, 2018; pp. 233–243. [Google Scholar]
- Wang, H.; Zhu, R.; Wang, Y.; Mi, Y. Experimental Study on Oxidative Desulfurization of Molten Copper Slag by Different Oxidants. Metall. Mater. Trans. B 2020, 51, 543–557. [Google Scholar] [CrossRef]
- Mukai, K.; Matsushita, T.; Seetharaman, S. Motion of fine particles in liquid caused by interfacial tension gradient in relation to metals separation technologies. Scand. J. Metall. 2005, 34, 137–142. [Google Scholar] [CrossRef]
- Kosyakov, V.I. Topological analysis of the solid-liquid diagram for the system Cu-Fe-S. Russ. J. Inorg. Chem. 2008, 53, 946–952. [Google Scholar] [CrossRef]
- Peacey, J.G. Copper metallurgy. In AccessScience; McGraw-Hill Education: New York, NY, USA, 2020. [Google Scholar] [CrossRef]
- Mehta, U.; Yadav, S.K.; Koirala, I.; Koirala, R.P.; Shrestha, G.K.; Adhikari, D. Study of surface tension and viscosity of Cu–Fe–Si ternary alloy using a thermodynamic approach. Heliyon 2020, 6, e04674. [Google Scholar] [CrossRef] [PubMed]
- Jing, L.; Kexiong, H.; Pengcheng, L.; Xinmin, C. Research on density and surface tension of Cu-Fe-S system matte. Nonferrous Met. 1989, 04, 72–76. [Google Scholar]
- Shiraishi, Y.; Ikeda, K.; Tamura, A.; Saitô, T. On the Viscosity and Density of the Molten FeO-SiO2 System. Trans. Jpn. Inst. Met. 1978, 19, 264–274. [Google Scholar] [CrossRef] [Green Version]
- Kondratiev, A.; Jak, E. A quasi-chemical viscosity model for fully liquid slags in the Al2O3-CaO-‘FeO’-SiO2 system. Metall. Mater. Trans. B 2005, 36, 623–638. [Google Scholar] [CrossRef]
- Hara, S.; Yamamoto, H.; Tateishi, S.; Gaskell, D.R.; Ogino, K. Surface Tension of Melts in the FeO-Fe2O3-CaO and FeO-Fe2O3-2CaO·SiO2 Systems under Air and CO2 Atmosphere, Materials Transactions. JIM 1991, 32, 829–836. [Google Scholar] [CrossRef] [Green Version]
- Sun, H.; Nakashima, K.; Mori, K. Influence of Slag Composition on Slag-Iron Interfacial Tension. ISIJ Int. 2006, 46, 407–412. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Huang, K.; Chen, X. Interfacial tension between slag and matte. Acta Metall. Sin. 1989, 25, 96–101. [Google Scholar]
- Zhang, H.; Li, B.; Wei, Y.; Wang, H. The settling behavior of matte particles in copper slag and the new technology of copper slag cleaning. J. Mater. Res. Technol. 2021, 15, 6216–6230. [Google Scholar] [CrossRef]
- Yamashita, T.; Hayes, P. Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials. Appl. Surf. Sci. 2008, 254, 2441–2449. [Google Scholar] [CrossRef]
- Bale, C.W.; Bélisle, E.; Chartrand, P.; Decterov, S.A.; Eriksson, G.; Gheribi, A.E.; Hack, K.; Jung, I.H.; Kang, Y.B.; Melançon, J.; et al. FactSage thermochemical software and databases, 2010–2016. Calphad 2016, 54, 35–53. [Google Scholar] [CrossRef] [Green Version]
- Fallah-Mehrjardi, A.; Hayes, P.C.; Jak, E. The Effect of CaO on Gas/Slag/Matte/Tridymite Equilibria in Fayalite-Based Copper Smelting Slags at 1473 K (1200 °C) and P(SO2) = 0.25 Atm. Metall. Mater. Trans. B 2018, 49, 602–609. [Google Scholar] [CrossRef]
- Fallah-Mehrjardi, A.; Hidayat, T.; Hayes, P.C.; Jak, E. Experimental Investigation of Gas/Slag/Matte/Tridymite Equilibria in the Cu-Fe-O-S-Si System in Controlled Atmospheres: Development of Technique. Metall. Mater. Trans. B 2017, 48, 3002–3016. [Google Scholar] [CrossRef]
- Yazawa, A. Thermodynamic considerations of copper smelting. Can. Metall. Q. 1974, 13, 443–453. [Google Scholar] [CrossRef]
- Toguri, J.M.; Santander, N.H. The solubility of copper in fayalite slags at 1300 °C. Can. Metall. Q. 1969, 8, 167–171. [Google Scholar] [CrossRef]
- Isaksson, J.; Vikström, T.; Lennartsson, A.; Samuelsson, C. Influence of Process Parameters on Copper Content in Reduced Iron Silicate Slag in a Settling Furnace. Metals 2021, 11, 992. [Google Scholar] [CrossRef]
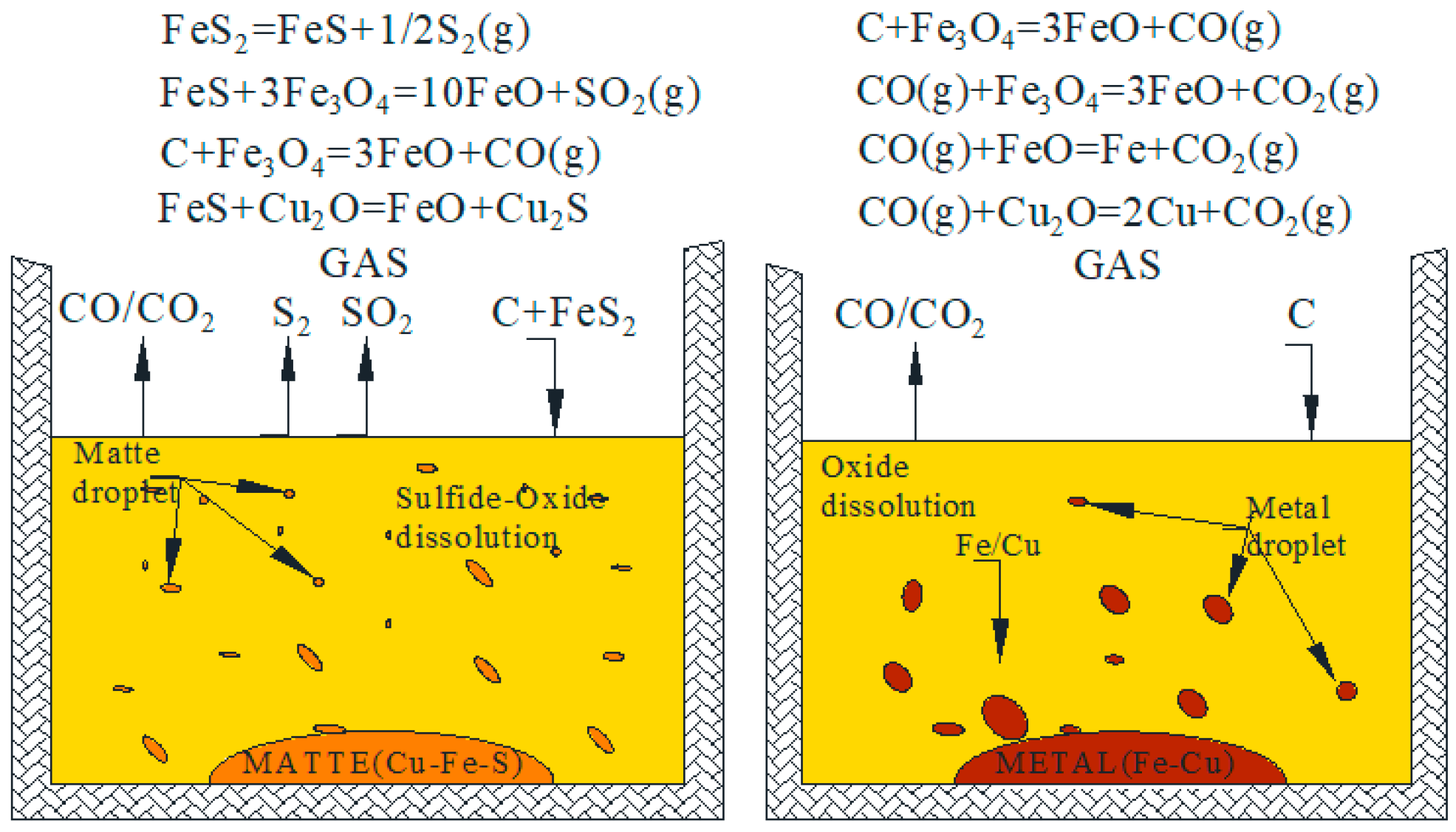
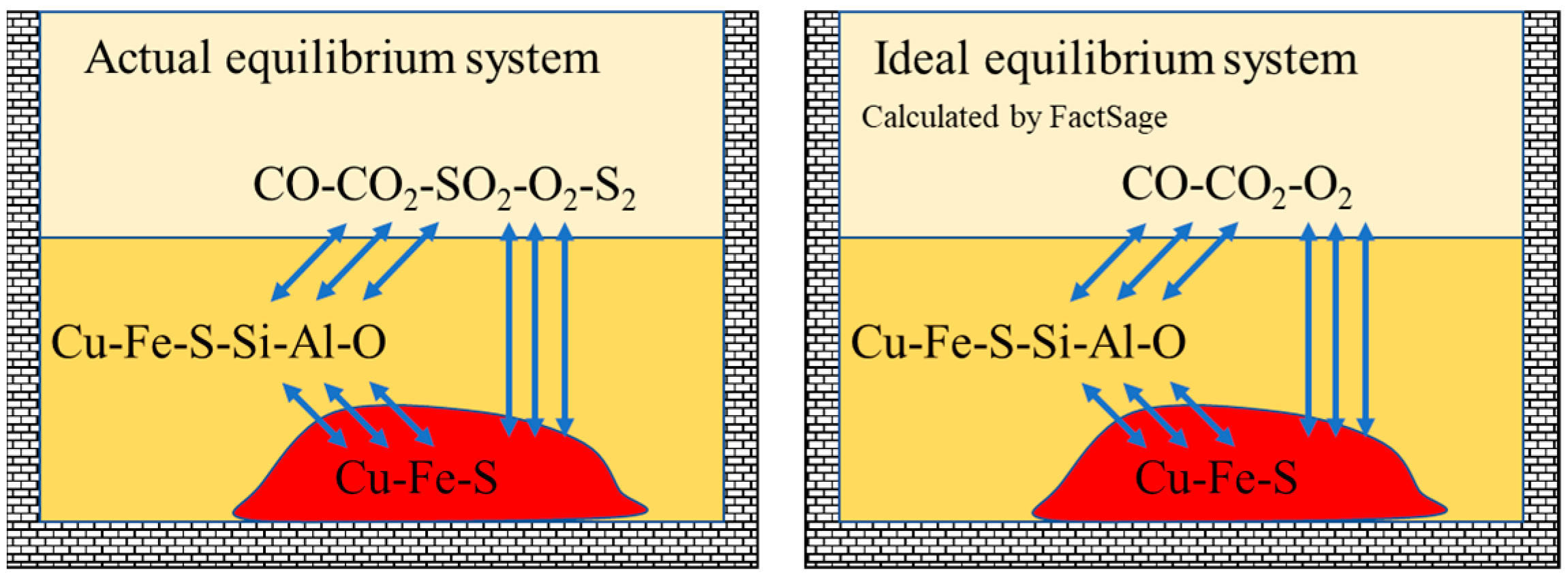
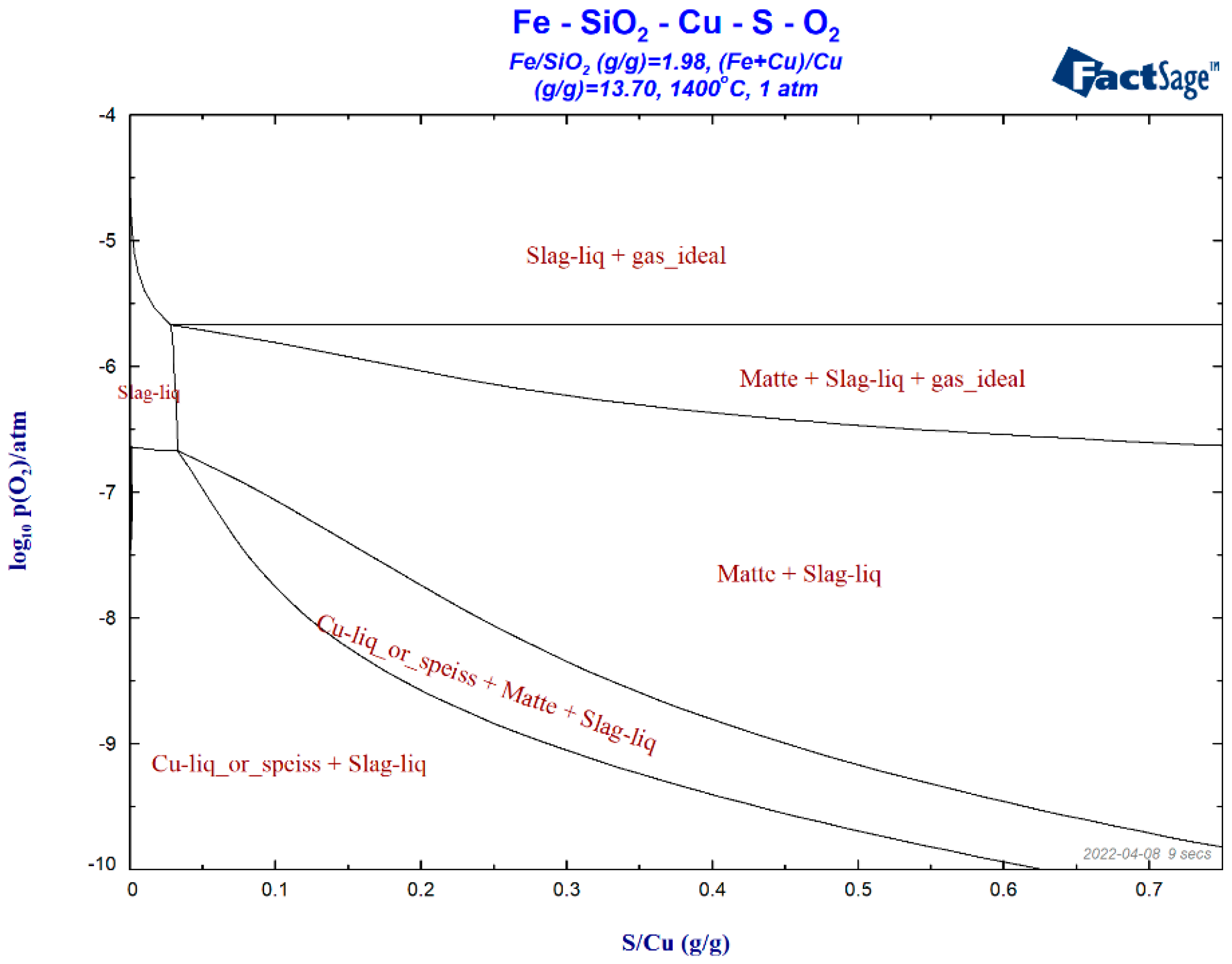
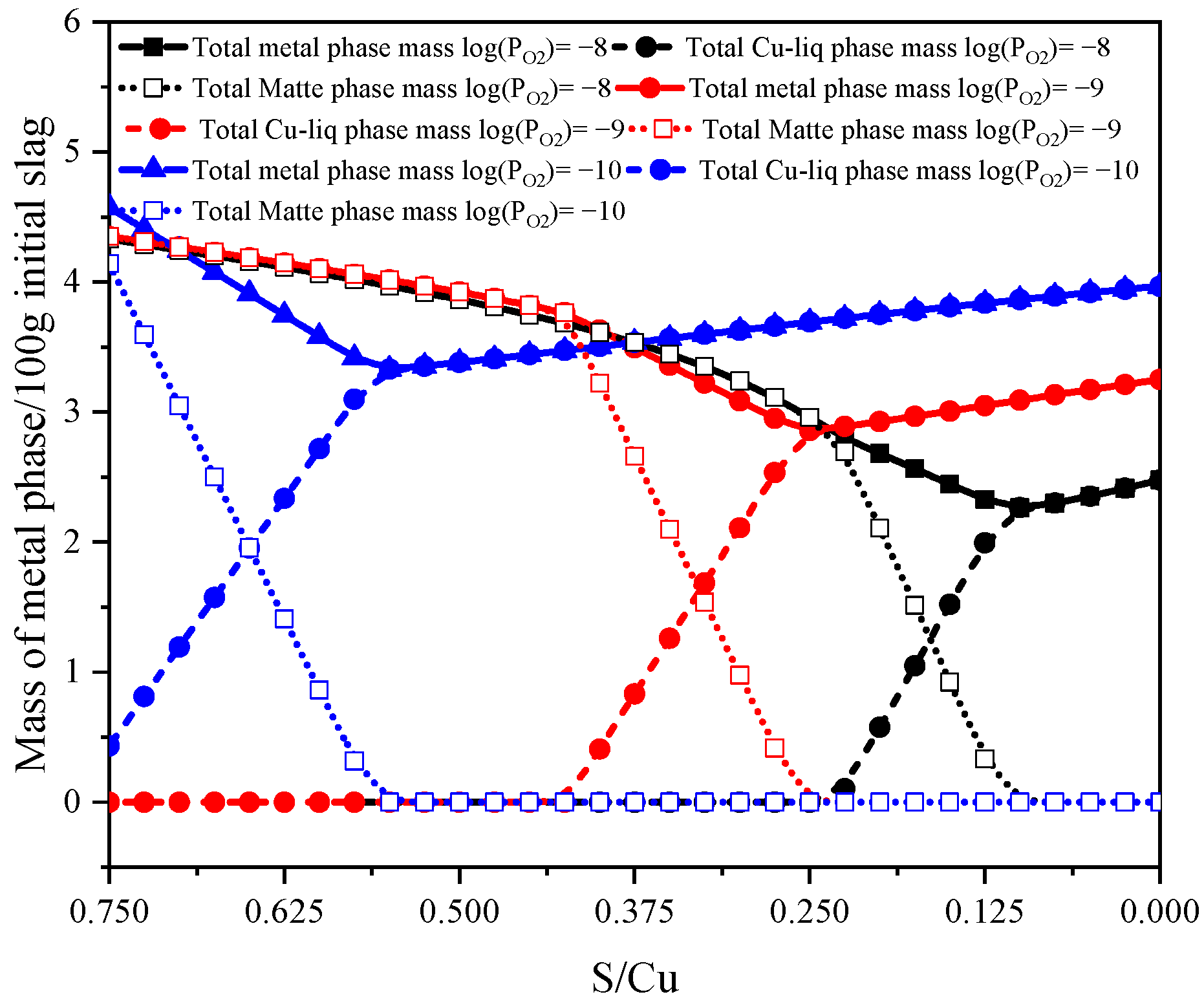

| Density | Viscosity | Surface Tension | |
|---|---|---|---|
| Metal | 7.1–8.0 g/cm3 [25] | 1.1–1.5 N/m (1200–1500 °C) [26] | |
| Matte | 4.13–4.98 g/cm3 [27] | 0.32–0.38 N/m (1200 °C) [27] | |
| Slag | 3.5–4.0 g/cm3 (20–40 mol% SiO2 at 1400 °C) [28] | 0.07–0.14 Pa·s; Fe/SiO2 = 1.67 (1200–1300 °C) [28,29] | 0.5–0.4 N/m (20–40 mol% SiO2 at 1400 °C) [30] |
| Interfacial tension | |||
| Interfacial tension between metal and slag | 0.96–1.67 N/m (estimated data) [31] | ||
| Interfacial tension between matte and slag | 0.04–0.09 N/m; Fe/SiO2 = 1.0–1.65 (1600 °C) [32] | ||
| Proportion, % | Mass Content, wt% | Mass Ratio | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cu2S | Cu2O | SiO2 | FeS | FeO-SiO2 | Total S | Total Cu | Total Fe | Fe/Cu | Fe/SiO2 | S/Cu |
| 0.00 | 4.50 | 0.00 | 0.00 | 95.50 | 0.00 | 4.00 | 50.81 | 12.70 | 1.98 | 0.000 |
| 1.00 | 3.60 | 0.00 | 0.00 | 95.50 | 0.20 | 4.00 | 50.76 | 12.70 | 1.98 | 0.050 |
| 2.50 | 2.25 | 0.00 | 0.00 | 95.50 | 0.50 | 3.99 | 50.68 | 12.70 | 1.98 | 0.125 |
| 5.00 | 0.00 | 0.00 | 0.00 | 95.50 | 1.00 | 3.98 | 50.56 | 12.70 | 1.98 | 0.250 |
| 5.00 | 0.00 | 0.45 | 1.38 | 93.84 | 1.49 | 3.97 | 50.47 | 12.70 | 1.98 | 0.375 |
| 5.00 | 0.00 | 0.89 | 2.75 | 92.20 | 1.98 | 3.97 | 50.39 | 12.70 | 1.98 | 0.500 |
| 5.00 | 0.00 | 1.33 | 4.13 | 90.55 | 2.48 | 3.96 | 50.30 | 12.70 | 1.98 | 0.625 |
| 5.00 | 0.00 | 1.77 | 5.50 | 88.91 | 2.97 | 3.95 | 50.22 | 12.70 | 1.98 | 0.750 |
| Cu2S | Cu2O | FeS | FeO | Fe2O3 | SiO2 | Al2O3 | ∑ |
|---|---|---|---|---|---|---|---|
| 0.00 | 4.50 | 0.00 | 62.25 | 3.48 | 25.66 | 4.12 | 100.00 |
| 1.00 | 3.60 | 0.00 | 62.19 | 3.47 | 25.63 | 4.11 | 100.00 |
| 2.49 | 2.24 | 0.00 | 62.09 | 3.47 | 25.60 | 4.11 | 100.00 |
| 4.98 | 0.00 | 0.00 | 61.94 | 3.46 | 25.53 | 4.10 | 100.00 |
| 4.97 | 0.00 | 1.37 | 60.76 | 3.39 | 25.49 | 4.02 | 100.00 |
| 4.96 | 0.00 | 2.73 | 59.60 | 3.33 | 25.45 | 3.94 | 100.00 |
| 4.95 | 0.00 | 4.09 | 58.43 | 3.26 | 25.40 | 3.86 | 100.00 |
| 4.94 | 0.00 | 5.44 | 57.28 | 3.20 | 25.36 | 3.79 | 100.00 |
| S/Cu | Quality of Synthetic Slag | Cu2O | Fe2O3 | FeO | Graphite | C/O |
|---|---|---|---|---|---|---|
| 0.000 | 100 | 4.50 | 3.48 | 62.25 | 2.31 | 0.2 |
| 0.050 | 3.60 | 3.47 | 62.19 | 2.29 | ||
| 0.125 | 2.24 | 3.47 | 62.09 | 2.26 | ||
| 0.250 | 0.00 | 3.46 | 61.94 | 2.22 | ||
| 0.375 | 0.00 | 3.39 | 60.76 | 2.18 | ||
| 0.500 | 0.00 | 3.33 | 59.60 | 2.14 | ||
| 0.625 | 0.00 | 3.26 | 58.43 | 2.09 | ||
| 0.750 | 0.00 | 3.20 | 57.28 | 2.05 | ||
| 0.000 | 100 | 4.50 | 3.48 | 62.25 | 1.15 | 0.1 |
| 0.050 | 3.60 | 3.47 | 62.19 | 1.14 | ||
| 0.125 | 2.24 | 3.47 | 62.09 | 1.13 | ||
| 0.250 | 0.00 | 3.46 | 61.94 | 1.11 | ||
| 0.375 | 0.00 | 3.39 | 60.76 | 1.09 | ||
| 0.500 | 0.00 | 3.33 | 59.60 | 1.07 | ||
| 0.725 | 0.00 | 3.26 | 58.43 | 1.05 | ||
| 0.750 | 0.00 | 3.20 | 57.28 | 1.03 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, L.; Wang, H.; Dong, K. Effect of Sulfur Content on Copper Recovery in the Reduction Smelting Process. Metals 2022, 12, 857. https://doi.org/10.3390/met12050857
Wu L, Wang H, Dong K. Effect of Sulfur Content on Copper Recovery in the Reduction Smelting Process. Metals. 2022; 12(5):857. https://doi.org/10.3390/met12050857
Chicago/Turabian StyleWu, Long, Hongyang Wang, and Kai Dong. 2022. "Effect of Sulfur Content on Copper Recovery in the Reduction Smelting Process" Metals 12, no. 5: 857. https://doi.org/10.3390/met12050857
APA StyleWu, L., Wang, H., & Dong, K. (2022). Effect of Sulfur Content on Copper Recovery in the Reduction Smelting Process. Metals, 12(5), 857. https://doi.org/10.3390/met12050857





