Simulated Breathing: Application of Molecular Dynamics Simulations to Pulmonary Lung Surfactant
Abstract
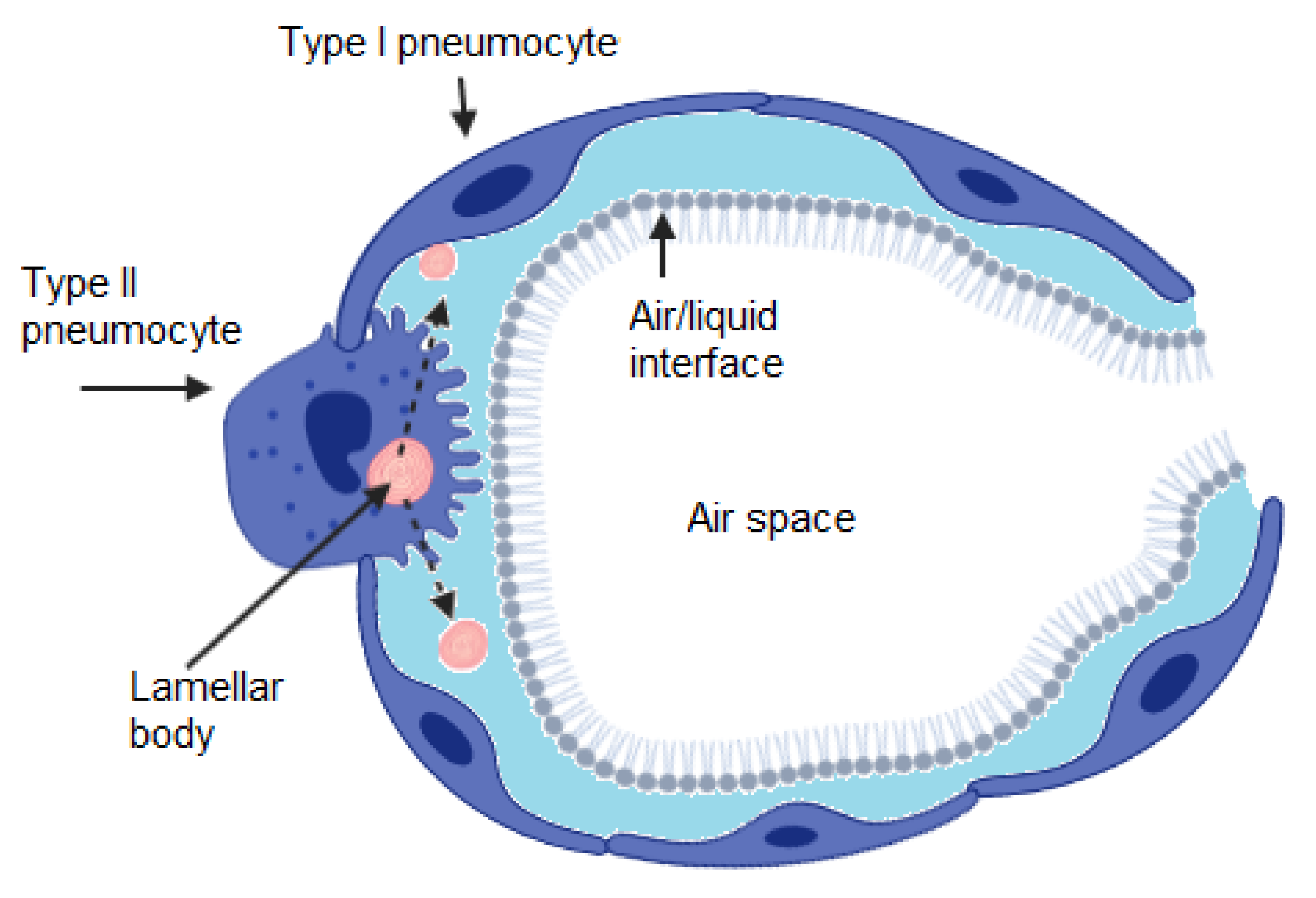
1. Introduction
2. Surfactant Composition
| Lipid | Relative Lipid Content |
|---|---|
| Phosphatidylcholine (PC) | 70–85% |
| DPPC | (≈40–55%) |
| Palmitoyl-myristoyl PC | (≈9–12%) |
| Palmitoyl-palmitoleoyl PC | (≈8%) |
| Palmitoyl-oleoyl PC | (≈10%) |
| Palmitoyl-linoleoyl PC | (≈6%) |
| Phosphatidylglycerol (PG) | 5–10% |
| Phosphatidylethanolamine (PE) | 0–5% |
| Phosphatidylinositol (PI) | 0–3% |
| Cholesterol | 5–10% |
2.1. Cholesterol
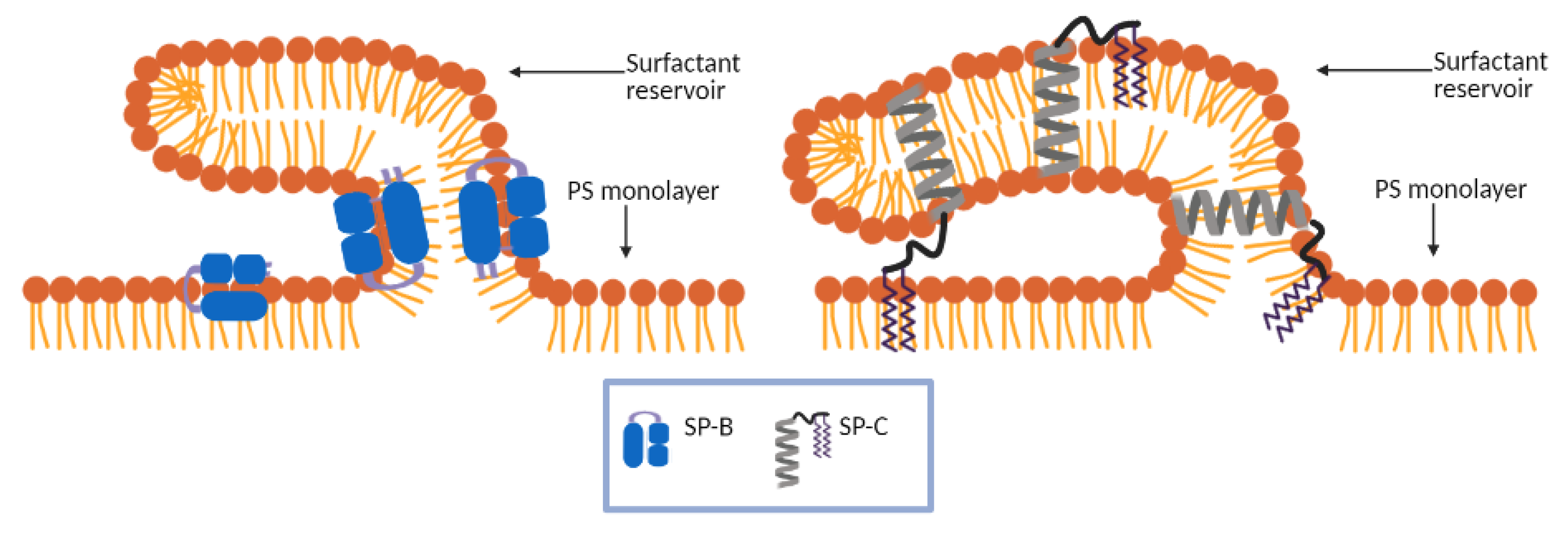
2.2. Surfactant Proteins
2.2.1. Hydrophilic Proteins
2.2.2. Hydrophobic Proteins
2.3. Surfactant Function
2.4. Role of Oxidative Stress
2.5. Proposed Mechanism of Function
2.5.1. Squeeze-Out Theory
2.5.2. Supercompression Model
3. Molecular Dynamics
4. Molecular Dynamics of Pulmonary Surfactant
4.1. Choosing Lipids to Build a Monolayer
4.2. Tuning the Phase Transition
4.3. Looking beyond the Interface
4.4. Supercompression through the In Silico Lens
4.5. Adding Surfactant Proteins
4.6. SP-D in Immunology
4.7. Impact of Foreign Nanoparticles
4.8. Oxidative Stress Impact on PS Lipids
5. Putting It Together
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Martinez-Calle, M.; Prieto, M.; Olmeda, B.; Fedorov, A.; Loura, L.M.S.; Pérez-Gil, J. Pulmonary surfactant protein SP-B nanorings induce the multilamellar organization of surfactant complexes. Biochim. Biophys. Acta (BBA) Biomembr. 2020, 1862, 183216. [Google Scholar] [CrossRef]
- Griese, M. Pulmonary surfactant in health and human lung diseases: State of the art. Eur. Respir. J. 1999, 13, 1455–1476. [Google Scholar] [CrossRef]
- Agudelo, C.W.; Kumley, B.K.; Area-Gomez, E.; Xu, Y.; Dabo, A.J.; Geraghty, P.; Campos, M.; Foronjy, R.; Garcia-Arcos, I. Decreased surfactant lipids correlate with lung function in chronic obstructive pulmonary disease (COPD). PLoS ONE 2020, 15, e0228279. [Google Scholar] [CrossRef]
- Nkadi, P.O.; Merritt, T.A.; Pillers, D.A.M. An overview of pulmonary surfactant in the neonate: Genetics, metabolism, and the role of surfactant in health and disease. Mol. Genet. Metab. 2009, 97, 95–101. [Google Scholar] [CrossRef]
- King, R.J. Pulmonary surfactant. J. Appl. Physiol. 1982, 53, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Weaver, T.E.; Conkright, J.J. Function of surfactant proteins B and C. Annu. Rev. Physiol. 2001, 63, 555–578. [Google Scholar] [CrossRef] [PubMed]
- Stahlman, M.T.; Gray, M.P.; Falconieri, M.W.; Whitsett, J.A.; Weaver, T.E. Lamellar body formation in normal and surfactant protein B-deficient fetal mice. Lab. Investig. 2000, 80, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Hawgood, S.; Derrick, M.; Poulain, F. Structure and properties of surfactant protein B. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 1998, 1408, 150–160. [Google Scholar] [CrossRef]
- Han, S.; Mallampalli, R.K. The role of surfactant in lung disease and host defense against pulmonary infections. Ann. Am. Thorac. Soc. 2015, 12, 765–774. [Google Scholar] [CrossRef]
- Liekkinen, J.; de Santos Moreno, B.; Paananen, R.O.; Vattulainen, I.; Monticelli, L.; De La Serna, J.B.; Javanainen, M. Understanding the Functional Properties of Lipid Heterogeneity in Pulmonary Surfactant Monolayers at the Atomistic Level. Front. Cell Dev. Biol. 2020, 8, 581016. [Google Scholar] [CrossRef]
- Perez-Gil, J.; Weaver, T.E. Pulmonary surfactant pathophysiology: Current models and open questions. Physiology 2010, 25, 132–141. [Google Scholar] [CrossRef]
- Goerke, J. Pulmonary surfactant: Functions and molecular composition. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 1998, 1408, 79–89. [Google Scholar] [CrossRef]
- Gunasekara, L.; Schürch, S.; Schoel, W.M.; Nag, K.; Leonenko, Z.; Haufs, M.; Amrein, M. Pulmonary surfactant function is abolished by an elevated proportion of cholesterol. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2005, 1737, 27–35. [Google Scholar] [CrossRef]
- Brown, A.J.; Galea, A.M. Cholesterol as an evolutionary response to living with oxygen. Evolution 2010, 64, 2179–2183. [Google Scholar] [CrossRef]
- Goerke, J.; Gonzales, J. Temperature dependence of dipalmitoyl phosphatidylcholine monolayer stability. J. Appl. Physiol. 1981, 51, 1108–1114. [Google Scholar] [CrossRef] [PubMed]
- Veldhuizen, R.; Nag, K.; Orgeig, S.; Possmayer, F. The role of lipids in pulmonary surfactant. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 1998, 1408, 90–108. [Google Scholar] [CrossRef]
- Hidalgo, A.; Cruz, A.; Pérez-Gil, J. Barrier or carrier? Pulmonary surfactant and drug delivery. Eur. J. Pharm. Biopharm. 2015, 95, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Hook, G.E.R.; Spalding, J.W.; Ortner, M.J.; Tombropoulos, E.G.; Chignell, C.F. Investigation of phospholipids of the pulmonary extracellular lining by electron paramagnetic resonance. The effects of phosphatidylglycerol and unsaturated phosphatidylcholines on the fluidity of dipalmitoyl phosphatidylcholine. Biochem. J. 1984, 223, 533–542. [Google Scholar] [CrossRef]
- Meban, C. Effect of lipids and other substances on the adsorption of dipalmitoyl phosphatidylcholine. Pediatr. Res. 1981, 15, 1029–1031. [Google Scholar] [CrossRef]
- Hallman, M.; Enhorning, G.; Possmayer, F. Composition and surface activity of normal and phosphatidylglycerol-deficient lung surfactant. Pediatr. Res. 1985, 19, 286–292. [Google Scholar] [CrossRef]
- Baatz, J.E.; Elledge, B.; Whitsett, J.A. Surfactant protein SP-B induces ordering at the surface of model membrane bilayers. Biochemistry 1990, 29, 6714–6720. [Google Scholar] [CrossRef]
- Frerking, I.; Günther, A.; Seeger, W.; Pison, U. Pulmonary surfactant: Functions, abnormalities and therapeutic options. Intensive Care Med. 2001, 27, 1699–1717. [Google Scholar] [CrossRef]
- Cañadas, O.; Olmeda, B.; Alonso, A.; Pérez-Gil, J. Lipid–protein and protein–protein interactions in the pulmonary surfactant system and their role in lung homeostasis. Int. J. Mol. Sci. 2020, 21, 3708. [Google Scholar] [CrossRef]
- Presti, F.T. The role of cholesterol in regulating membrane fluidity. Membr. Fluidity Biol. 1985, 4, 97–145. [Google Scholar]
- Orgeig, S.; Daniels, C.B. The roles of cholesterol in pulmonary surfactant: Insights from comparative and evolutionary studies. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2001, 129, 75–89. [Google Scholar] [CrossRef]
- Hadley, N.F. Adaptive Role of Lipids in Biological Systems; Wiley: Hoboken, NJ, USA, 1985. [Google Scholar]
- Taneva, S.; Keough, K.M.W. Cholesterol modifies the properties of surface films of dipalmitoylphosphatidylcholine plus pulmonary surfactant-associated protein B or C spread or adsorbed at the air-water interface. Biochemistry 1997, 36, 912–922. [Google Scholar] [CrossRef]
- Evans, R.W.; Williams, M.A.; Tinoco, J. Surface viscosities of phospholipids alone and with cholesterol in monolayers at the air-water interface. Lipids 1980, 15, 524–533. [Google Scholar] [CrossRef]
- Rubio, S.; Lacaze-Masmonteil, T.; Chailley-Heu, B.; Kahn, A.; Bourbon, J.R.; Ducroc, R. Pulmonary surfactant protein A (SP-A) is expressed by epithelial cells of small and large intestine. J. Biol. Chem. 1995, 270, 12162–12169. [Google Scholar] [CrossRef] [PubMed]
- Khubchandani, K.R.; Snyder, J.M. Surfactant protein A (SP-A): The alveolus and beyond. FASEB J. 2001, 15, 59–69. [Google Scholar] [CrossRef]
- Guagliardo, R.; Pérez-Gil, J.; De Smedt, S.; Raemdonck, K. Pulmonary surfactant and drug delivery: Focusing on the role of surfactant proteins. J. Control. Release 2018, 291, 116–126. [Google Scholar] [CrossRef]
- Cabré, E.J.; Martínez-Calle, M.; Prieto, M.; Fedorov, A.; Olmeda, B.; Loura, L.M.S.; Pérez-Gil, J. Homo-and hetero-oligomerization of hydrophobic pulmonary surfactant proteins SP-B and SP-C in surfactant phospholipid membranes. J. Biol. Chem. 2018, 293, 9399–9411. [Google Scholar] [CrossRef]
- Vorbroker, D.K.; Voorhout, W.F.; Weaver, T.E.; Whitsett, J.A. Posttranslational processing of surfactant protein C in rat type II cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 1995, 269, L727–L733. [Google Scholar] [CrossRef]
- Mulugeta, S.; Beers, M.F. Surfactant protein C: Its unique properties and emerging immunomodulatory role in the lung. Microbes Infect. 2006, 8, 2317–2323. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, R.; Martin-Gonzalez, A.; Echaide, M.; Jain, A.; Brondyk, W.H.; Rosenbaum, J.; Moreno-Herrero, F.; Perez-Gil, J. Supramolecular assembly of human pulmonary surfactant protein SP-D. J. Mol. Biol. 2018, 430, 1495–1509. [Google Scholar] [CrossRef]
- Martínez-Calle, M.; Olmeda, B.; Dietl, P.; Frick, M.; Pérez-Gil, J. Pulmonary surfactant protein SP-B promotes exocytosis of lamellar bodies in alveolar type II cells. FASEB J. 2018, 32, 4600–4611. [Google Scholar] [CrossRef]
- Taneva, S.G.; Keough, K.M.W. Adsorption of pulmonary surfactant protein SP-A to monolayers of phospholipids containing hydrophobic surfactant protein SP-B or SP-C: Potential differential role for tertiary interaction of lipids, hydrophobic proteins, and SP-A. Biochemistry 2000, 39, 6083–6093. [Google Scholar] [CrossRef] [PubMed]
- Bates, S.R.; Dodia, C.; Tao, J.Q.; Fisher, A.B. Surfactant protein-A plays an important role in lung surfactant clearance: Evidence using the surfactant protein-A gene-targeted mouse. Am. J. Physiol. Lung Cell. Mol. Physiol. 2008, 294, L325–L333. [Google Scholar] [CrossRef]
- Hiansen, J.Q.; Keating, E.; Aspros, A.; Yao, L.J.; Bosma, K.J.; Yamashita, C.M.; Lewis, J.F.; Veldhuizen, R.A.W. Cholesterol-mediated surfactant dysfunction is mitigated by surfactant protein A. Biochim. Biophys. Acta (BBA) Biomembr. 2015, 1848, 813–820. [Google Scholar] [CrossRef]
- Liekkinen, J.; Enkavi, G.; Javanainen, M.; Olmeda, B.; Pérez-Gil, J.; Vattulainen, I. Pulmonary Surfactant Lipid Reorganization Induced by the Adsorption of the Oligomeric Surfactant Protein B Complex. J. Mol. Biol. 2020, 432, 3251–3268. [Google Scholar] [CrossRef]
- Schürch, D.; Ospina, O.L.; Cruz, A.; Pérez-Gil, J. Combined and independent action of proteins SP-B and SP-C in the surface behavior and mechanical stability of pulmonary surfactant films. Biophys. J. 2010, 99, 3290–3299. [Google Scholar] [CrossRef]
- Ingenito, E.P.; Mora, R.; Mark, L. Pivotal role of anionic phospholipids in determining dynamic behavior of lung surfactant. Am. J. Respir. Crit. Care Med. 2000, 161, 831–838. [Google Scholar] [CrossRef] [PubMed]
- Ikegami, M.; Na, C.L.; Korfhagen, T.R.; Whitsett, J.A. Surfactant protein D influences surfactant ultrastructure and uptake by alveolar type II cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 2005, 288, L552–L561. [Google Scholar] [CrossRef][Green Version]
- Fukuzawa, T.; Ishida, J.; Kato, A.; Ichinose, T.; Ariestanti, D.M.; Takahashi, T.; Ito, K.; Abe, J.; Suzuki, T.; Wakana, S. Lung surfactant levels are regulated by Ig-Hepta/GPR116 by monitoring surfactant protein D. PLoS ONE 2013, 8, e69451. [Google Scholar] [CrossRef]
- Sorensen, G.L. Surfactant protein D in respiratory and non-respiratory diseases. Front. Med. 2018, 5, 18. [Google Scholar] [CrossRef]
- Kishore, U.; Greenhough, T.J.; Waters, P.; Shrive, A.K.; Ghai, R.; Kamran, M.F.; Bernal, A.L.; Reid, K.B.M.; Madan, T.; Chakraborty, T. Surfactant proteins SP-A and SP-D: Structure, function and receptors. Mol. Immunol. 2006, 43, 1293–1315. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Barbero, F.; Rivas, G.; Steinhilber, W.; Casals, C. Structural and functional differences among human surfactant proteins SP-A1, SP-A2 and co-expressed SP-A1/SP-A2: Role of supratrimeric oligomerization. Biochem. J. 2007, 406, 479–489. [Google Scholar] [CrossRef] [PubMed]
- van de Wetering, J.K.; van Golde, L.M.G.; Batenburg, J.J. Collectins. Eur. J. Biochem. 2004, 271, 1229–1249. [Google Scholar] [CrossRef]
- Ikegami, M.; Korfhagen, T.R.; Whitsett, J.A.; Bruno, M.D.; Wert, S.E.; Wada, K.; Jobe, A.H. Characteristics of surfactant from SP-A-deficient mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 1998, 275, L247–L254. [Google Scholar] [CrossRef]
- Rice, W.R.; Ross, G.F.; Singleton, F.M.; Dingle, S.; Whitsett, J.A. Surfactant-associated protein inhibits phospholipid secretion from type II cells. J. Appl. Physiol. 1987, 63, 692–698. [Google Scholar] [CrossRef]
- Mikerov, A.N.; Umstead, T.M.; Huang, W.; Liu, W.; Phelps, D.S.; Floros, J. SP-A1 and SP-A2 variants differentially enhance association of Pseudomonas aeruginosa with rat alveolar macrophages. Am. J. Physiol. Lung Cell. Mol. Physiol. 2005, 288, L150–L158. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lopez-Rodriguez, E.; Pascual, A.; Arroyo, R.; Floros, J.; Perez-Gil, J. Human pulmonary surfactant protein SP-A1 provides maximal efficiency of lung interfacial films. Biophys. J. 2016, 111, 524–536. [Google Scholar] [CrossRef]
- Crouch, E.C. Collectins and pulmonary host defense. Am. J. Respir. Cell Mol. Biol. 1998, 19, 177–201. [Google Scholar] [CrossRef]
- Kuroki, Y.; Voelker, D.R. Pulmonary surfactant proteins. J. Biol. Chem. 1994, 269, 25943–25946. [Google Scholar] [CrossRef]
- Barriga, A.; Morán-Lalangui, M.; Castillo-Sánchez, J.C.; Mingarro, I.; Pérez-Gil, J.; García-Álvarez, B. Role of pulmonary surfactant protein Sp-C dimerization on membrane fragmentation: An emergent mechanism involved in lung defense and homeostasis. Biochim. Biophys. Acta (BBA) Biomembr. 2021, 1863, 183572. [Google Scholar] [CrossRef]
- Keller, A.; Eistetter, H.R.; Voss, T.; Schäfer, K.P. The pulmonary surfactant protein C (SP-C) precursor is a type II transmembrane protein. Biochem. J. 1991, 277, 493–499. [Google Scholar] [CrossRef]
- Melton, K.R.; Nesslein, L.L.; Ikegami, M.; Tichelaar, J.W.; Clark, J.C.; Whitsett, J.A.; Weaver, T.E. SP-B deficiency causes respiratory failure in adult mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 2003, 285, L543–L549. [Google Scholar] [CrossRef]
- Qanbar, R.; Cheng, S.; Possmayer, F.; Schurch, S. Role of the palmitoylation of surfactant-associated protein C in surfactant film formation and stability. Am. J. Physiol. Lung Cell. Mol. Physiol. 1996, 271, L572–L580. [Google Scholar] [CrossRef]
- Lee, H.; Kandasamy, S.K.; Larson, R.G. Molecular dynamics simulations of the anchoring and tilting of the lung-surfactant peptide SP-B1-25 in palmitic acid monolayers. Biophys. J. 2005, 89, 3807–3821. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tokieda, K.; Whitsett, J.A.; Clark, J.C.; Weaver, T.E.; Ikeda, K.; McConnell, K.B.; Jobe, A.H.; Ikegami, M.; Iwamoto, H.S. Pulmonary dysfunction in neonatal SP-B-deficient mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 1997, 273, L875–L882. [Google Scholar] [CrossRef]
- Glasser, S.W.; Burhans, M.S.; Korfhagen, T.R.; Na, C.L.; Sly, P.D.; Ross, G.F.; Ikegami, M.; Whitsett, J.A. Altered stability of pulmonary surfactant in SP-C-deficient mice. Proc. Natl. Acad. Sci. USA 2001, 98, 6366–6371. [Google Scholar] [CrossRef]
- Creuwels, L.; Van Golde, L.M.G.; Haagsman, H.P. The pulmonary surfactant system: Biochemical and clinical aspects. Lung 1997, 175, 1–39. [Google Scholar] [CrossRef]
- Zuo, Y.Y.; Veldhuizen, R.A.W.; Neumann, A.W.; Petersen, N.O.; Possmayer, F. Current perspectives in pulmonary surfactant—Inhibition, enhancement and evaluation. Biochim. Biophys. Acta (BBA) Biomembr. 2008, 1778, 1947–1977. [Google Scholar] [CrossRef]
- Rodríguez-Capote, K.; Manzanares, D.; Haines, T.; Possmayer, F. Reactive oxygen species inactivation of surfactant involves structural and functional alterations to surfactant proteins SP-B and SP-C. Biophys. J. 2006, 90, 2808–2821. [Google Scholar] [CrossRef] [PubMed]
- Sabatini, K.; Mattila, J.P.; Megli, F.M.; Kinnunen, P.K.J. Characterization of two oxidatively modified phospholipids in mixed monolayers with DPPC. Biophys. J. 2006, 90, 4488–4499. [Google Scholar] [CrossRef]
- Stachowicz-Kuśnierz, A.; Cwiklik, L.; Korchowiec, J.; Rogalska, E.; Korchowiec, B. The impact of lipid oxidation on the functioning of a lung surfactant model. Phys. Chem. Chem. Phys. 2018, 20, 24968–24978. [Google Scholar] [CrossRef] [PubMed]
- Possmayer, F.; Nag, K.; Rodriguez, K.; Qanbar, R.; Schürch, S. Surface activity in vitro: Role of surfactant proteins. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2001, 129, 209–220. [Google Scholar] [CrossRef]
- Ding, J.; Takamoto, D.Y.; Von Nahmen, A.; Lipp, M.M.; Lee, K.Y.C.; Waring, A.J.; Zasadzinski, J.A. Effects of lung surfactant proteins, SP-B and SP-C, and palmitic acid on monolayer stability. Biophys. J. 2001, 80, 2262–2272. [Google Scholar] [CrossRef]
- Diemel, R.V.; Snel, M.M.E.; Waring, A.J.; Walther, F.J.; van Golde, L.M.G.; Putz, G.; Haagsman, H.P.; Batenburg, J.J. Multilayer formation upon compression of surfactant monolayers depends on protein concentration as well as lipid composition: An atomic force microscopy study. J. Biol. Chem. 2002, 277, 21179–21188. [Google Scholar] [CrossRef]
- Rugonyi, S.; Biswas, S.C.; Hall, S.B. The biophysical function of pulmonary surfactant. Respir. Physiol. Neurobiol. 2008, 163, 244–255. [Google Scholar] [CrossRef]
- Parra, E.; Pérez-Gil, J. Composition, structure and mechanical properties define performance of pulmonary surfactant membranes and films. Chem. Phys. Lipids 2015, 185, 153–175. [Google Scholar] [CrossRef]
- Zuo, Y.Y.; Possmayer, F. How does pulmonary surfactant reduce surface tension to very low values? J. Appl. Physiol. 2007, 102, 1733–1734. [Google Scholar] [CrossRef][Green Version]
- Hansson, T.; Oostenbrink, C.; van Gunsteren, W. Molecular dynamics simulations. Curr. Opin. Struct. Biol. 2002, 12, 190–196. [Google Scholar] [CrossRef]
- Agrahari, A.K.; Pieroni, E.; Gatto, G.; Kumar, A. The impact of missense mutation in PIGA associated to paroxysmal nocturnal hemoglobinuria and multiple congenital anomalies-hypotonia-seizures syndrome 2: A computational study. Heliyon 2019, 5, e02709. [Google Scholar] [CrossRef]
- Kumar, A.; Sechi, L.A.; Caboni, P.; Marrosu, M.G.; Atzori, L.; Pieroni, E. Dynamical insights into the differential characteristics of Mycobacterium avium subsp. paratuberculosis peptide binding to HLA-DRB1 proteins associated with multiple sclerosis. New J. Chem. 2015, 39, 1355–1366. [Google Scholar] [CrossRef]
- Pal, R.; Kumar, A.; Misra, G. Exploring TEAD2 as a drug target for therapeutic intervention of cancer: A multi-computational case study. Brief. Bioinform. 2021. [Google Scholar] [CrossRef]
- Ammarah, U.; Kumar, A.; Pal, R.; Bal, N.C.; Misra, G. Identification of new inhibitors against human Great wall kinase using in silico approaches. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef]
- May, A.; Pool, R.; van Dijk, E.; Bijlard, J.; Abeln, S.; Heringa, J.; Feenstra, K.A. Coarse-grained versus atomistic simulations: Realistic interaction free energies for real proteins. Bioinformatics 2014, 30, 326–334. [Google Scholar] [CrossRef]
- Rose, D.; Rendell, J.; Lee, D.; Nag, K.; Booth, V. Molecular dynamics simulations of lung surfactant lipid monolayers. Biophys. Chem. 2008, 138, 67–77. [Google Scholar] [CrossRef]
- Duncan, S.L.; Dalal, I.S.; Larson, R.G. Molecular dynamics simulation of phase transitions in model lung surfactant monolayers. Biochim. Biophys. Acta (BBA) Biomembr. 2011, 1808, 2450–2465. [Google Scholar] [CrossRef]
- Baoukina, S.; Tieleman, D.P. Computer simulations of lung surfactant. Biochim. Biophys. Acta (BBA) Biomembr. 2016, 1858, 2431–2440. [Google Scholar] [CrossRef]
- Pocivavsek, L.; Dellsy, R.; Kern, A.; Johnson, S.; Lin, B.; Lee, K.Y.C.; Cerda, E. Stress and fold localization in thin elastic membranes. Science 2008, 320, 912–916. [Google Scholar] [CrossRef]
- Baoukina, S.; Monticelli, L.; Amrein, M.; Tieleman, D.P. The molecular mechanism of monolayer-bilayer transformations of lung surfactant from molecular dynamics simulations. Biophys. J. 2007, 93, 3775–3782. [Google Scholar] [CrossRef]
- Baoukina, S.; Rozmanov, D.; Mendez-Villuendas, E.; Tieleman, D.P. The mechanism of collapse of heterogeneous lipid monolayers. Biophys. J. 2014, 107, 1136–1145. [Google Scholar] [CrossRef]
- Johansson, J.; Curstedt, T. Molecular structures and interactions of pulmonary surfactant components. Eur. J. Biochem. 1997, 244, 675–693. [Google Scholar] [CrossRef]
- Lee, K.Y.C.; Lipp, M.M.; Zasadzinski, J.A.; Waring, A.J. Effects of lung surfactant specific protein SP-B and model SP-B peptide on lipid monolayers at the air-water interface. Colloids Surf. A Physicochem. Eng. Asp. 1997, 128, 225–242. [Google Scholar] [CrossRef]
- Krüger, P.; Schalke, M.; Wang, Z.; Notter, R.H.; Dluhy, R.A.; Lösche, M. Effect of hydrophobic surfactant peptides SP-B and SP-C on binary phospholipid monolayers. I. Fluorescence and dark-field microscopy. Biophys. J. 1999, 77, 903–914. [Google Scholar] [CrossRef][Green Version]
- Freites, J.A.; Choi, Y.; Tobias, D.J. Molecular dynamics simulations of a pulmonary surfactant protein B peptide in a lipid monolayer. Biophys. J. 2003, 84, 2169–2180. [Google Scholar] [CrossRef][Green Version]
- Goh, B.C.; Rynkiewicz, M.J.; Cafarella, T.R.; White, M.R.; Hartshorn, K.L.; Allen, K.; Crouch, E.C.; Calin, O.; Seeberger, P.H.; Schulten, K. Molecular mechanisms of inhibition of influenza by surfactant protein D revealed by large-scale molecular dynamics simulation. Biochemistry 2013, 52, 8527–8538. [Google Scholar] [CrossRef]
- Chen, P.; Zhang, Z.; Gu, N.; Ji, M. Effect of the surface charge density of nanoparticles on their translocation across pulmonary surfactant monolayer: A molecular dynamics simulation. Mol. Simul. 2018, 44, 85–93. [Google Scholar] [CrossRef]
- Bakshi, M.S.; Zhao, L.; Smith, R.; Possmayer, F.; Petersen, N.O. Metal nanoparticle pollutants interfere with pulmonary surfactant function in vitro. Biophys. J. 2008, 94, 855–868. [Google Scholar] [CrossRef]
- Yue, K.; Sun, X.; Tang, J.; Wei, Y.; Zhang, X. A Simulation Study on the Interaction Between Pollutant Nanoparticles and the Pulmonary Surfactant Monolayer. Int. J. Mol. Sci. 2019, 20, 3281. [Google Scholar] [CrossRef] [PubMed]
- Olzyńska, A.; Delcroix, P.; Dolejšová, T.; Krzaczek, K.; Korchowiec, B.; Czogalla, A.; Cwiklik, L. Properties of lipid models of lung surfactant containing cholesterol and oxidized lipids: A mixed experimental and computational study. Langmuir 2020, 36, 1023–1033. [Google Scholar] [CrossRef] [PubMed]

| Protein | Size (kDa) | Formations | Nature | Possible Interactions | Primary Functions |
|---|---|---|---|---|---|
| SP-A | 32–38 | monomer trimer octadecameric complex | hydrophilic | SP-B, SP-B/PG, DPPC, Cholesterol, Phospholipase A2 | innate immune activation, structuring tubular myelin, enables lipid spreading, homeostasis of lipid recycling |
| SP-B | 8.7 | oligomerize into ring-shaped channels | hydrophobic | SP-B, SP-C, PG, cholesterol | supports stability of lipids, enhance respreading capabilities |
| SP-C | 3.7–4 | oligomerize into supramolecular complexes | hydrophobic | PG, Cholesterol | supports stability of lipids |
| SP-D | 43 | monomer trimer hexamer X-shaped dodecamer | hydrophilic | PI G-protein-coupled receptor 116 | innate immune activation, support type 2 pneumocytes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dziura, M.; Mansour, B.; DiPasquale, M.; Chandrasekera, P.C.; Gauld, J.W.; Marquardt, D. Simulated Breathing: Application of Molecular Dynamics Simulations to Pulmonary Lung Surfactant. Symmetry 2021, 13, 1259. https://doi.org/10.3390/sym13071259
Dziura M, Mansour B, DiPasquale M, Chandrasekera PC, Gauld JW, Marquardt D. Simulated Breathing: Application of Molecular Dynamics Simulations to Pulmonary Lung Surfactant. Symmetry. 2021; 13(7):1259. https://doi.org/10.3390/sym13071259
Chicago/Turabian StyleDziura, Maksymilian, Basel Mansour, Mitchell DiPasquale, P. Charukeshi Chandrasekera, James W. Gauld, and Drew Marquardt. 2021. "Simulated Breathing: Application of Molecular Dynamics Simulations to Pulmonary Lung Surfactant" Symmetry 13, no. 7: 1259. https://doi.org/10.3390/sym13071259
APA StyleDziura, M., Mansour, B., DiPasquale, M., Chandrasekera, P. C., Gauld, J. W., & Marquardt, D. (2021). Simulated Breathing: Application of Molecular Dynamics Simulations to Pulmonary Lung Surfactant. Symmetry, 13(7), 1259. https://doi.org/10.3390/sym13071259







