Synthesis of Fuel Grade Molecules from Hydroprocessing of Biomass-Derived Compounds Catalyzed by Magnetic Fe(NiFe)O4-SiO2 Nanoparticles
Abstract
1. Introduction
2. Experimental
2.1. Catalyst Preparation and Heat Treatments
2.2. Catalyst Characterization
2.3. Catalytic Activity Studies
3. Results and Discussion
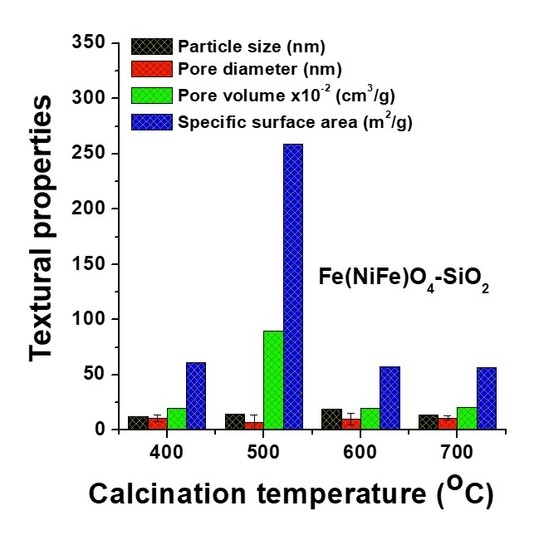
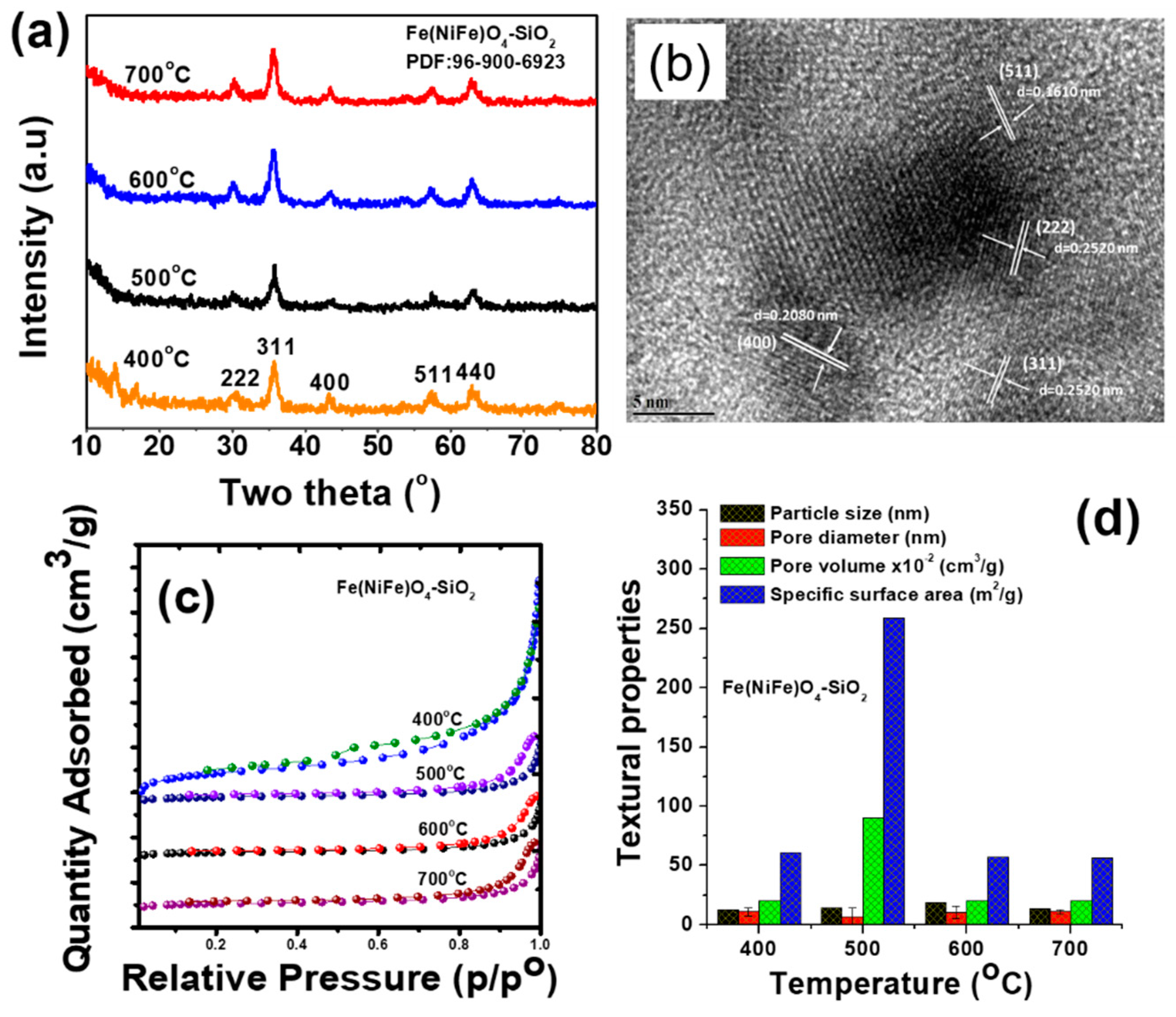
3.1. Effect of Temperature on Textural Properties of Fe(NiFe)O4−SiO2 Catalyst
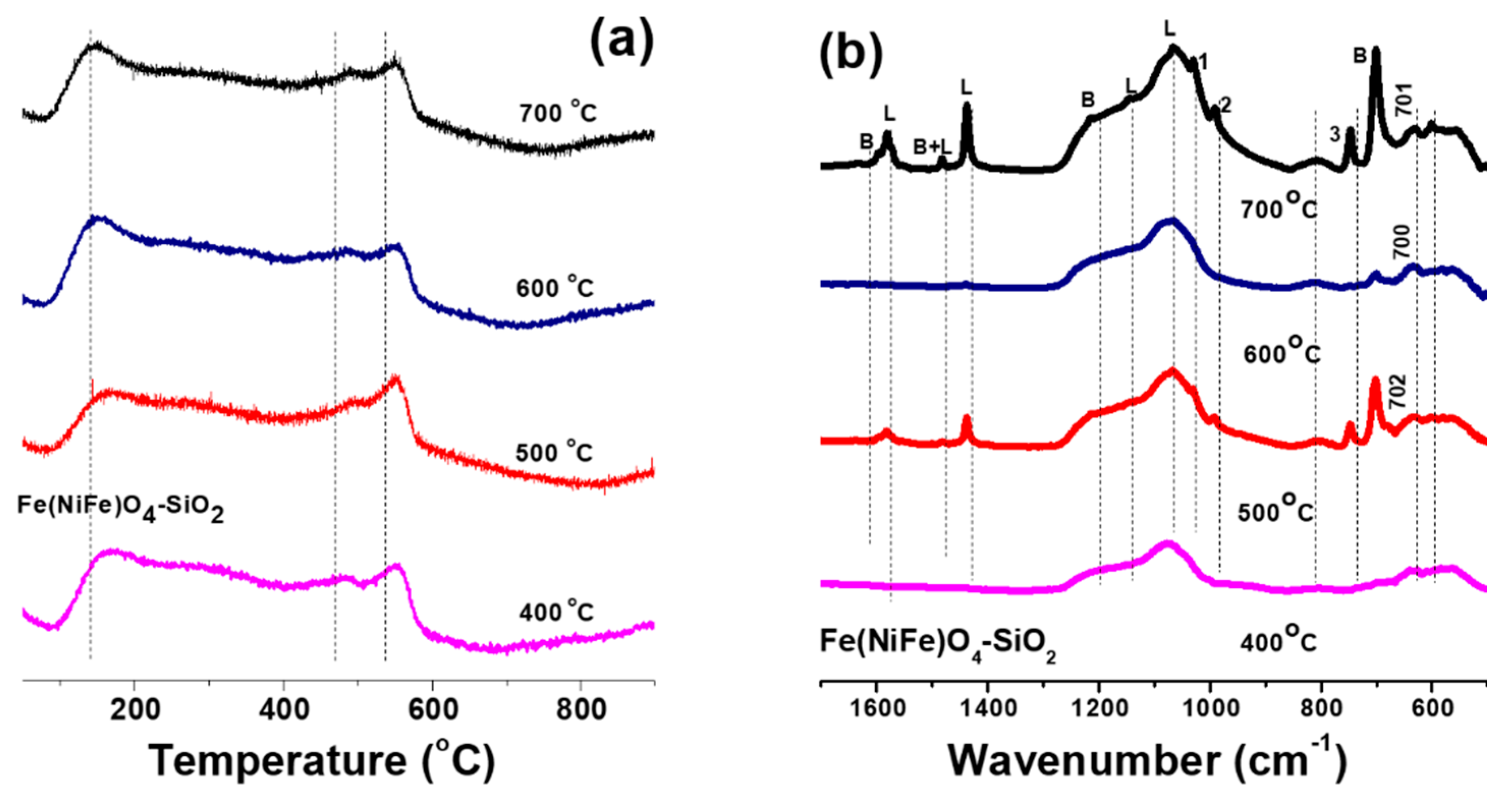
3.2. Effect of Calcination Temperature on Acid Properties of Fe(NiFe)O4-SiO2 Catalysts
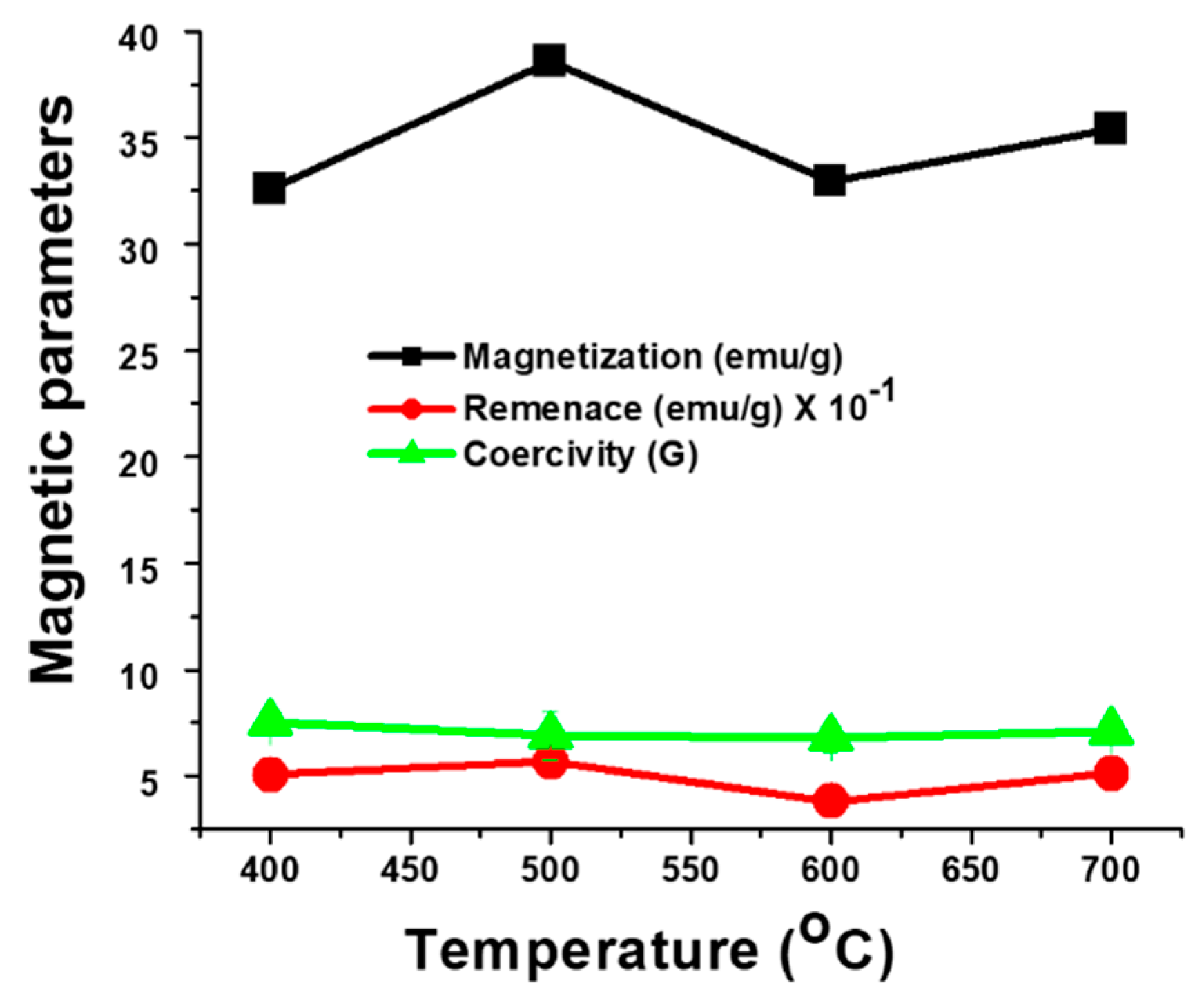
3.3. Effect of Calcination Temperature on Magnetic Properties of Fe(NiFe)O4-SiO2 Catalyst
3.4. Activity Studies of Magnetic Fe(NiFe)O4-SiO2 Catalyst
3.4.1. Hydrodeoxygenation of Vanillin
3.4.2. Hydrodeoxygenation of Furfural
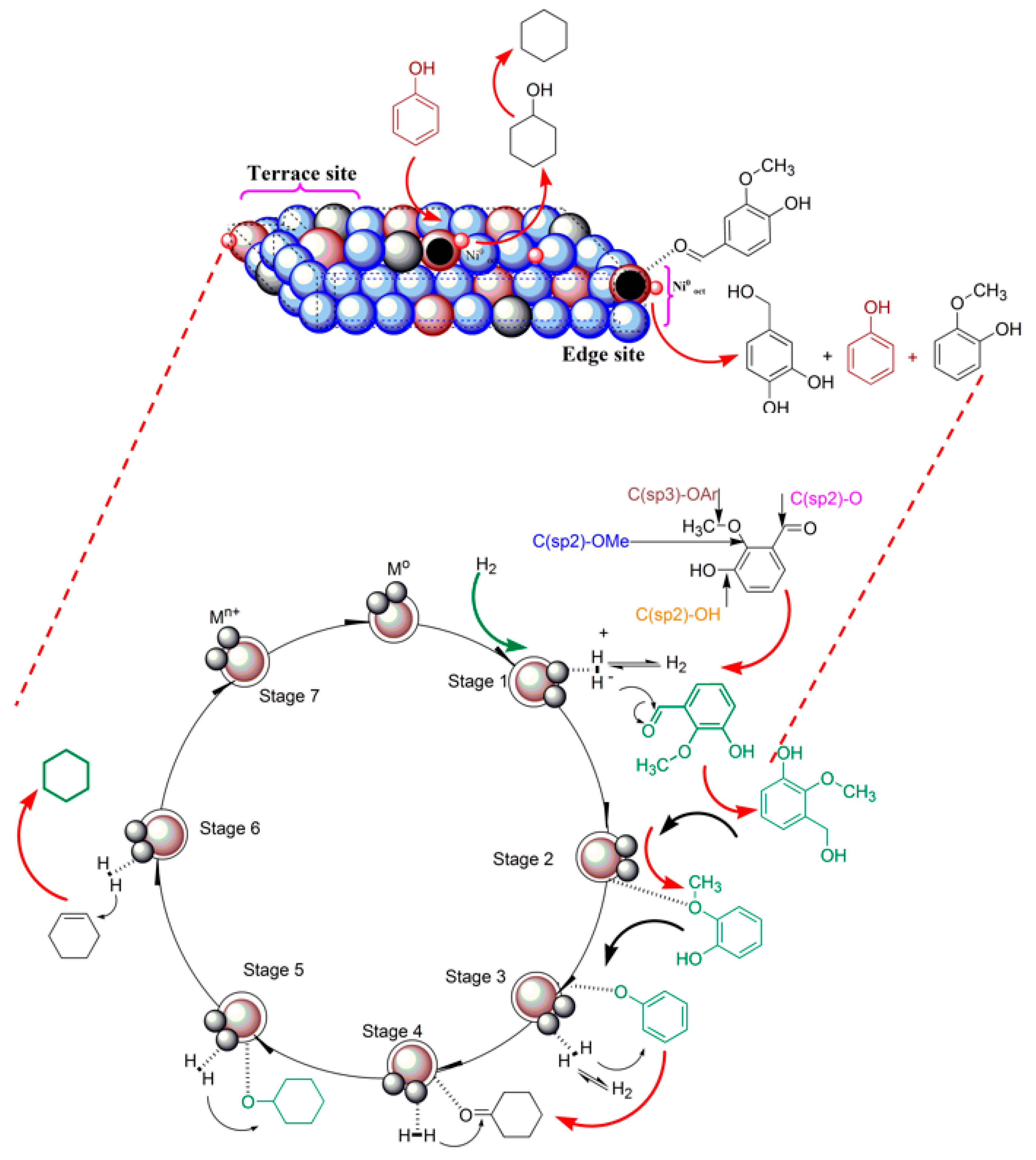
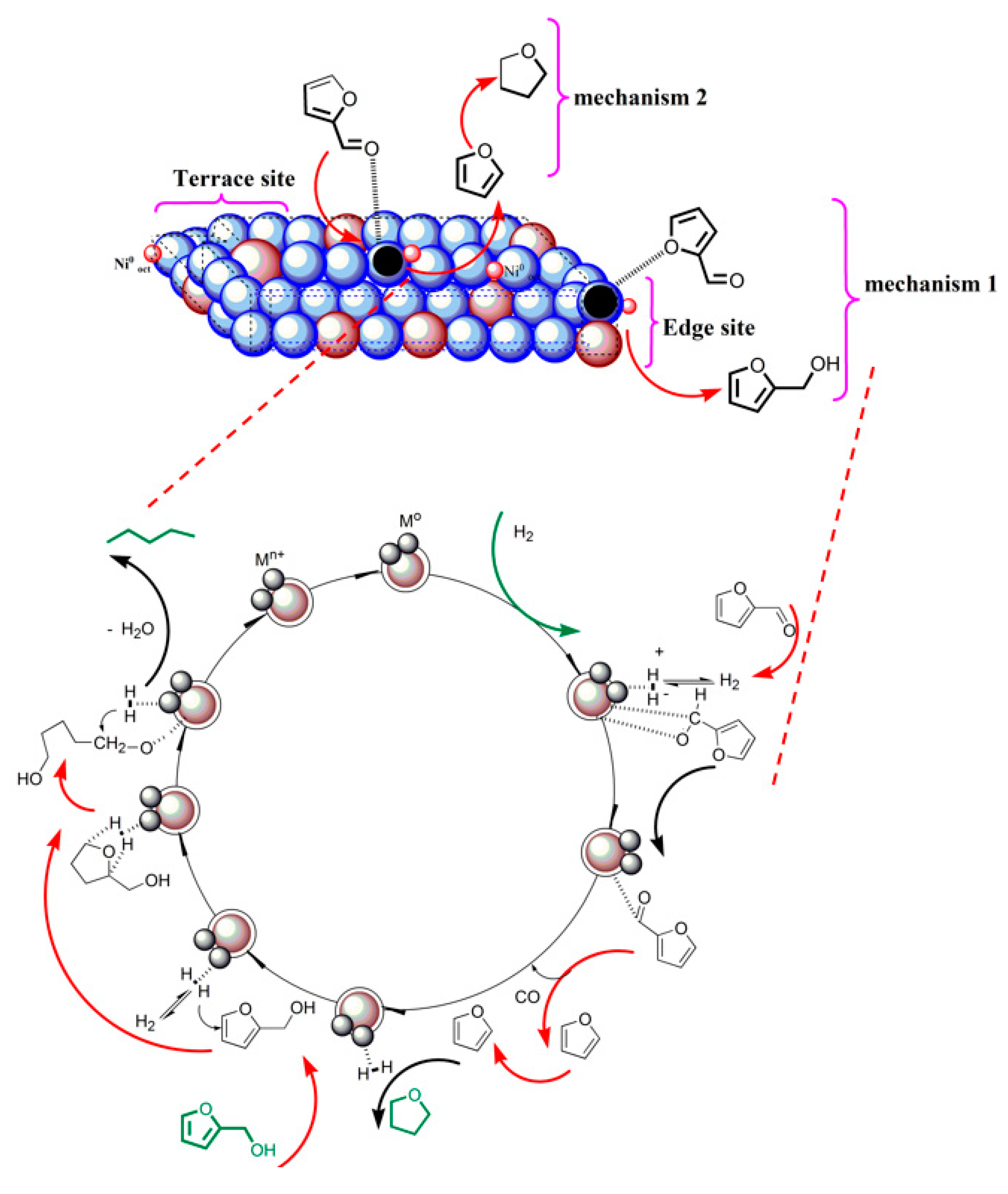
3.5. Possible Reaction Pathways
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Halilu, A. Is there a realistic solution to upgrading bio-oil to fuel and chemicals? Inform 2017, 28, 22–24. [Google Scholar] [CrossRef]
- Sudarsanam, P.; Zhong, R.; van den Bosch, S.; Coman, S.M.; Parvulescu, V.I.; Sels, B.F. Functionalised heterogeneous catalysts for sustainable biomass valorisation. Chem. Soc. Rev. 2018, 47, 8349–8402. [Google Scholar] [CrossRef]
- Ambursa, M.M.; Sudarsanam, P.; Voon, L.H.; Hamid, S.B.A.; Bhargava, S.K. Bimetallic Cu-Ni catalysts supported on MCM-41 and Ti-MCM-41 porous materials for hydrodeoxygenation of lignin model compound into transportation fuels. Fuel Process. Technol. 2017, 162, 87–97. [Google Scholar] [CrossRef]
- Ambursa, M.M.; Ali, T.H.; Lee, H.V.; Sudarsanam, P.; Bhargava, S.K.; Hamid, S.B.A. Hydrodeoxygenation of dibenzofuran to bicyclic hydrocarbons using bimetallic Cu–Ni catalysts supported on metal oxides. Fuel 2016, 180, 767–776. [Google Scholar] [CrossRef]
- Halilu, A.; Ali, T.H.; Atta, A.Y.; Sudarsanam, P.; Bhargava, S.K.; Hamid, S.B.A. Highly Selective Hydrogenation of Biomass-Derived Furfural into Furfuryl Alcohol Using a Novel Magnetic Nanoparticles Catalyst. Energy Fuels 2016, 30, 2216–2226. [Google Scholar] [CrossRef]
- Sudarsanam, P.; Peeters, E.; Makshina, E.V.; Parvulescu, V.I.; Sels, B.F. Advances in porous and nanoscale catalysts for viable biomass conversion. Chem. Soc. Rev. 2019. [Google Scholar] [CrossRef] [PubMed]
- Polshettiwar, V.; Luque, R.; Fihri, A.; Zhu, H.; Bouhrara, M.; Basset, J.-M. Magnetically recoverable nanocatalysts. Chem. Rev. 2011, 111, 3036–3075. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, P.; Li, H.; Wang, L. A recyclable magnetic nanoparticles supported palladium catalyst for the Hiyama reaction of aryltrialkoxysilanes with aryl halides. Catal. Sci. Tech. 2012, 2, 1859–1864. [Google Scholar] [CrossRef]
- Gawande, M.B.; Branco, P.S.; Varma, R.S. Nano-magnetite (Fe3O4) as a support for recyclable catalysts in the development of sustainable methodologies. Chem. Soc. Rev. 2013, 42, 3371–3393. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Astruc, D. Fast-Growing Field of Magnetically Recyclable Nanocatalysts. Chem. Rev. 2014, 114, 6949–6985. [Google Scholar] [CrossRef]
- Sharma, R.K.; Dutta, S.; Sharma, S.; Zboril, R.; Varma, R.S.; Gawande, M.B. Fe3O4 (iron oxide)-supported nanocatalysts: Synthesis, characterization and applications in coupling reactions. Green Chem. 2016, 18, 3184–3209. [Google Scholar] [CrossRef]
- Gawande, M.B.; Monga, Y.; Zboril, R.; Sharma, R.K. Silica-decorated magnetic nanocomposites for catalytic applications. Coord. Chem. Rev. 2015, 288, 118–143. [Google Scholar] [CrossRef]
- Gawande, M.B.; Rathi, A.K.; Branco, P.S.; Nogueira, I.D.; Velhinho, A.; Shrikhande, J.J.; Indulkar, U.U.; Jayaram, R.V.; Ghumman, C.A.A.; Bundaleski, N.; et al. Regio- and Chemoselective Reduction of Nitroarenes and Carbonyl Compounds over Recyclable Magnetic Ferrite–Nickel Nanoparticles (Fe3O4–Ni) by Using Glycerol as a Hydrogen Source. Chem. Eur. J. 2012, 18, 12628–12632. [Google Scholar] [CrossRef] [PubMed]
- Baig, R.B.N.; Varma, R.S. Magnetically retrievable catalysts for organic synthesis. Chem. Commun. 2013, 49, 752–770. [Google Scholar] [CrossRef] [PubMed]
- Sobhani, S.; Ramezani, Z. Synthesis of arylphosphonates catalyzed by Pd-imino-Py-[gamma]-Fe2O3 as a new magnetically recyclable heterogeneous catalyst in pure water without requiring any additive. RSC Adv. 2016, 6, 29237–29244. [Google Scholar] [CrossRef]
- Hamid, S.B.A.; Halilu, A.; Gbadamasi, S.; Hakim, L. Opportunities in Utilization of Lignocellulosic Biomass Oil to Bio-Esters. In Renewable Energy and Sustainable Developments; Al-Douri, Y., Ed.; Scientific & Academic Publishing: Rosemead, CA, USA, 2014. [Google Scholar]
- Amin, M.H.; Putla, S.; Hamid, S.B.A.; Bhargava, S.K. Understanding the role of lanthanide promoters on the structure–activity of nanosized Ni/γ-Al2O3 catalysts in carbon dioxide reforming of methane. Appl. Catal. A 2015, 492, 160–168. [Google Scholar] [CrossRef]
- Otake, K.-i.; Cui, Y.; Buru, C.T.; Li, Z.; Hupp, J.T.; Farha, O.K. Single-Atom-Based Vanadium Oxide Catalysts Supported on Metal–Organic Frameworks: Selective Alcohol Oxidation and Structure–Activity Relationship. J. Am. Chem. Soc. 2018, 140, 8652–8656. [Google Scholar] [CrossRef] [PubMed]
- Sachs, M.; Sprick, R.S.; Pearce, D.; Hillman, S.A.; Monti, A.; Guilbert, A.A.; Brownbill, N.J.; Dimitrov, S.; Shi, X.; Blanc, F. Understanding structure-activity relationships in linear polymer photocatalysts for hydrogen evolution. Nat. Commun. 2018, 9, 4968. [Google Scholar] [CrossRef]
- Hillary, B.; Sudarsanam, P.; Amin, M.H.; Bhargava, S.K. Nanoscale Cobalt–Manganese Oxide Catalyst Supported on Shape-Controlled Cerium Oxide: Effect of Nanointerface Configuration on Structural. Redox, and Catalytic Properties. Langmuir 2017, 33, 1743–1750. [Google Scholar] [CrossRef] [PubMed]
- Sudarsanam, P.; Hillary, B.; Mallesham, B.; Rao, B.G.; Amin, M.H.; Nafady, A.; Alsalme, A.M.; Reddy, B.M.; Bhargava, S.K. Designing CuOx Nanoparticle-Decorated CeO2 Nanocubes for Catalytic Soot Oxidation: Role of the Nanointerface in the Catalytic Performance of Heterostructured Nanomaterials. Langmuir 2016, 32, 2208–2215. [Google Scholar] [CrossRef]
- Sudarsanam, P.; Hillary, B.; Amin, M.H.; Hamid, S.B.A.; Bhargava, S.K. Structure-activity relationships of nanoscale MnOx/CeO2 heterostructured catalysts for selective oxidation of amines under eco-friendly conditions. Appl. Catal. B Environ. 2016, 185, 213–224. [Google Scholar] [CrossRef]
- Tenzer, R. Influence of particle size on the coercive force of barium ferrite powders. J. Appl. Phys. 1963, 34, 1267–1268. [Google Scholar] [CrossRef]
- Sjögren, C.E.; Johansson, C.; Nævestad, A.; Sontum, P.C.; Briley-Sæbø, K.; Fahlvik, A.K. Crystal size and properties of superparamagnetic iron oxide (SPIO) particles. Magn. Reson. Imaging 1997, 15, 55–67. [Google Scholar] [CrossRef]
- Mistura, G.; Pozzato, A.; Grenci, G.; Bruschi, L.; Tormen, M. Continuous adsorption in highly ordered porous matrices made by nanolithography. Nat. Commun. 2013, 4, 2966. [Google Scholar] [CrossRef] [PubMed]
- Farneth, W.; Gorte, R. Methods for characterizing zeolite acidity. Chem. Rev. 1995, 95, 615–635. [Google Scholar] [CrossRef]
- Hutchings, G.J.; Lee, D.F. Control of product selectivity for the epoxidation of allyl alcohol by variation of the acidity of the catalyst TS-1. Chem. Commun. 1994, 9, 1095–1096. [Google Scholar] [CrossRef]
- Sumiya, S.; Oumi, Y.; Uozumi, T.; Sano, T. Characterization of AlSBA-15 prepared by post-synthesisalumination with trimethylaluminium. J. Mater. Chem. 2001, 11, 1111–1115. [Google Scholar] [CrossRef]
- Rana, M.S.; Srinivas, B.N.; Maity, S.K.; Dhar, G.M.; Rao, T.S.R.P. Origin of Cracking Functionality of Sulfided (Ni) CoMo/SiO2–ZrO2 Catalysts. J. Catal. 2000, 195, 31–37. [Google Scholar] [CrossRef]
- Xinghua, Z.; Tiejun, W.; Longlong, M.; Chuangzhi, W. Aqueous-phase catalytic process for production of pentane from furfural over nickel-based catalysts. Fuel 2010, 89, 2697–2702. [Google Scholar] [CrossRef]
- Mortensen, P.M.; Grunwaldt, J.-D.; Jensen, P.A.; Jensen, A.D. Screening of Catalysts for Hydrodeoxygenation of Phenol as a Model Compound for Bio-oil. ACS Catal. 2013, 3, 1774–1785. [Google Scholar] [CrossRef]
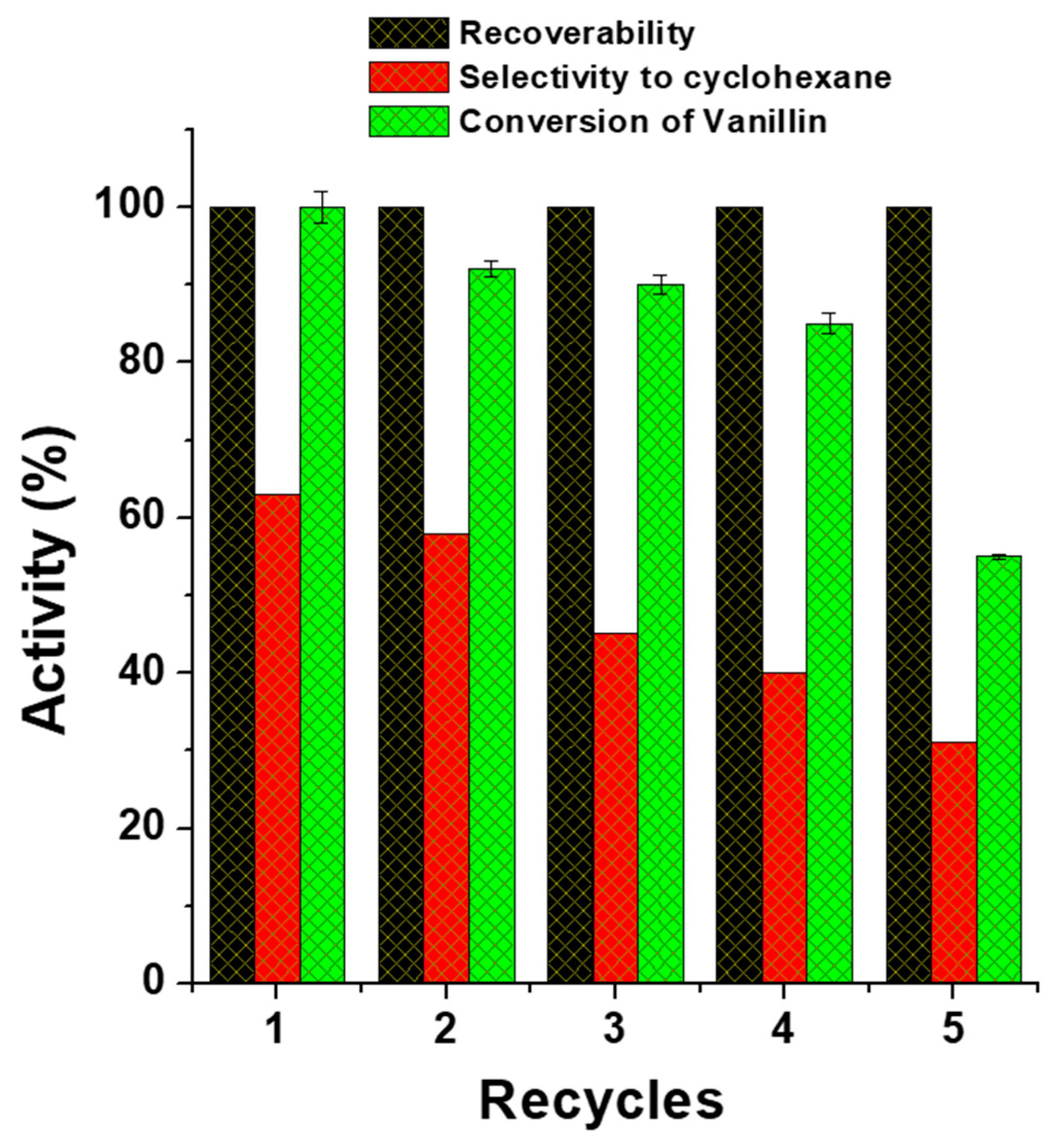
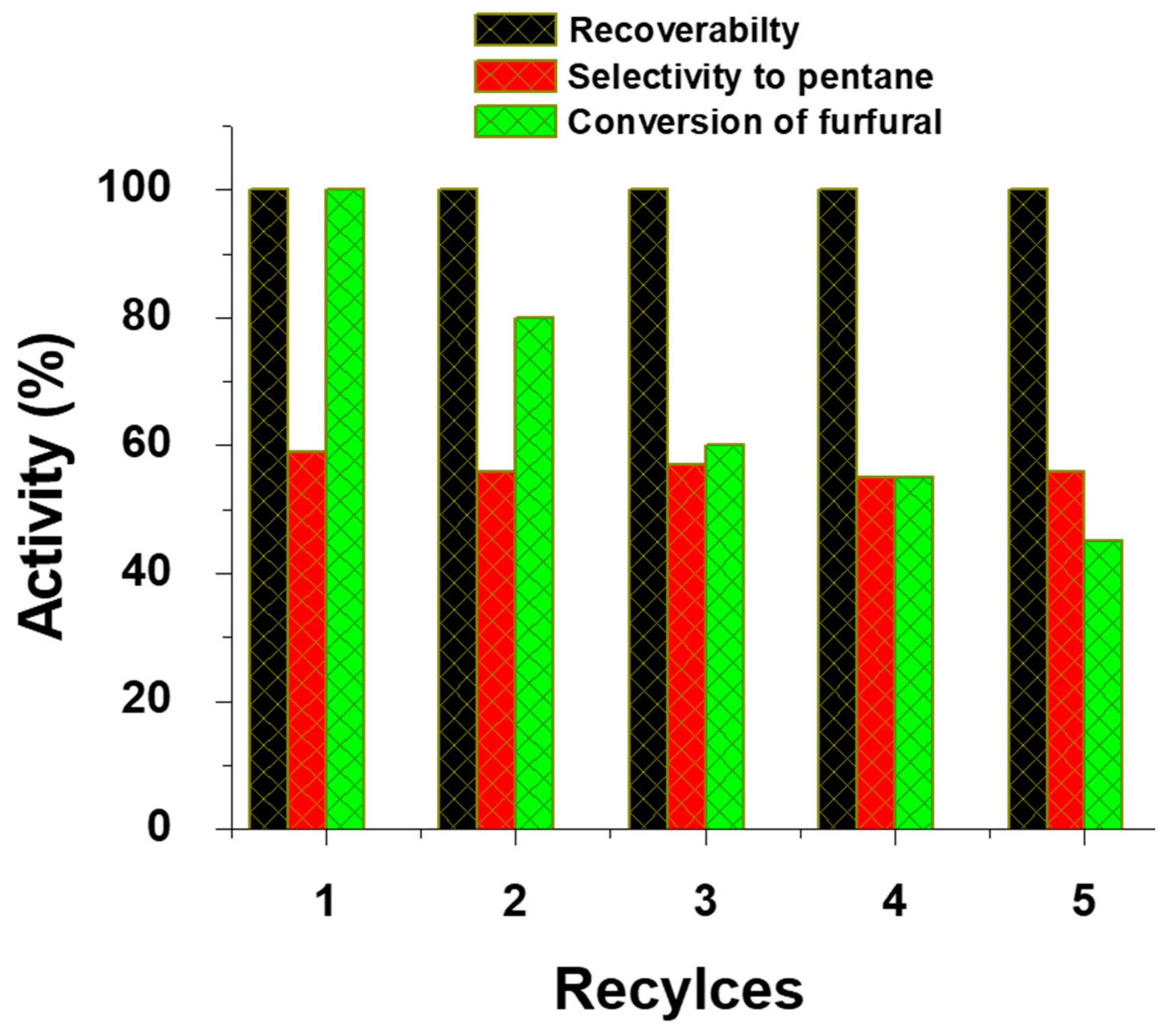
| Catalyst | Vanillin Conversion (%) | Selectivity (%) | % HDO | |||||
|---|---|---|---|---|---|---|---|---|
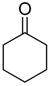 | 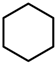 | 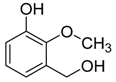 | 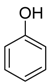 | 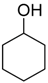 | 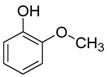 | |||
| Fe(NiFe)O4-SiO2 (400 °C) | 100 | 56 | 21 | 2 | 4 | - | 17 | 17.89 |
| Fe(NiFe)O4-SiO2 (500 °C) | 100 | 14 | 63 | - | 5 | 10 | 8 | 20.20 |
| Fe(NiFe)O4-SiO2 (600 °C) | 100 | 5 | 54 | - | 2 | 20 | 19 | 20.08 |
| Fe(NiFe)O4-SiO2 (700 °C) | 100 | 4 | 53 | - | 1 | 33 | 9 | 19.51 |
| Catalyst | Furfural Conversion (%) | Selectivity (%) | % HDO | ||
|---|---|---|---|---|---|
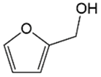 | 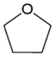 |  | |||
| Fe(NiFe)O4-SiO2 (400 °C) | 100 | 12 | 42 | 46 | 29.78 |
| Fe(NiFe)O4-SiO2 (500 °C) | 100 | 8 | 33 | 59 | 29.78 |
| Fe(NiFe)O4-SiO2 (600 °C) | 100 | 13 | 34 | 53 | 29.78 |
| Fe(NiFe)O4-SiO2 (700 °C) | 100 | 8 | 58 | 34 | 29.78 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Halilu, A.; Hussein Ali, T.; Sudarsanam, P.; Bhargava, S.K. Synthesis of Fuel Grade Molecules from Hydroprocessing of Biomass-Derived Compounds Catalyzed by Magnetic Fe(NiFe)O4-SiO2 Nanoparticles. Symmetry 2019, 11, 524. https://doi.org/10.3390/sym11040524
Halilu A, Hussein Ali T, Sudarsanam P, Bhargava SK. Synthesis of Fuel Grade Molecules from Hydroprocessing of Biomass-Derived Compounds Catalyzed by Magnetic Fe(NiFe)O4-SiO2 Nanoparticles. Symmetry. 2019; 11(4):524. https://doi.org/10.3390/sym11040524
Chicago/Turabian StyleHalilu, Ahmed, Tammar Hussein Ali, Putla Sudarsanam, and Suresh K. Bhargava. 2019. "Synthesis of Fuel Grade Molecules from Hydroprocessing of Biomass-Derived Compounds Catalyzed by Magnetic Fe(NiFe)O4-SiO2 Nanoparticles" Symmetry 11, no. 4: 524. https://doi.org/10.3390/sym11040524
APA StyleHalilu, A., Hussein Ali, T., Sudarsanam, P., & Bhargava, S. K. (2019). Synthesis of Fuel Grade Molecules from Hydroprocessing of Biomass-Derived Compounds Catalyzed by Magnetic Fe(NiFe)O4-SiO2 Nanoparticles. Symmetry, 11(4), 524. https://doi.org/10.3390/sym11040524