Coastal Aquifer Salinization in Semi-Arid Regions: The Case of Grombalia (Tunisia)
Abstract
1. Introduction
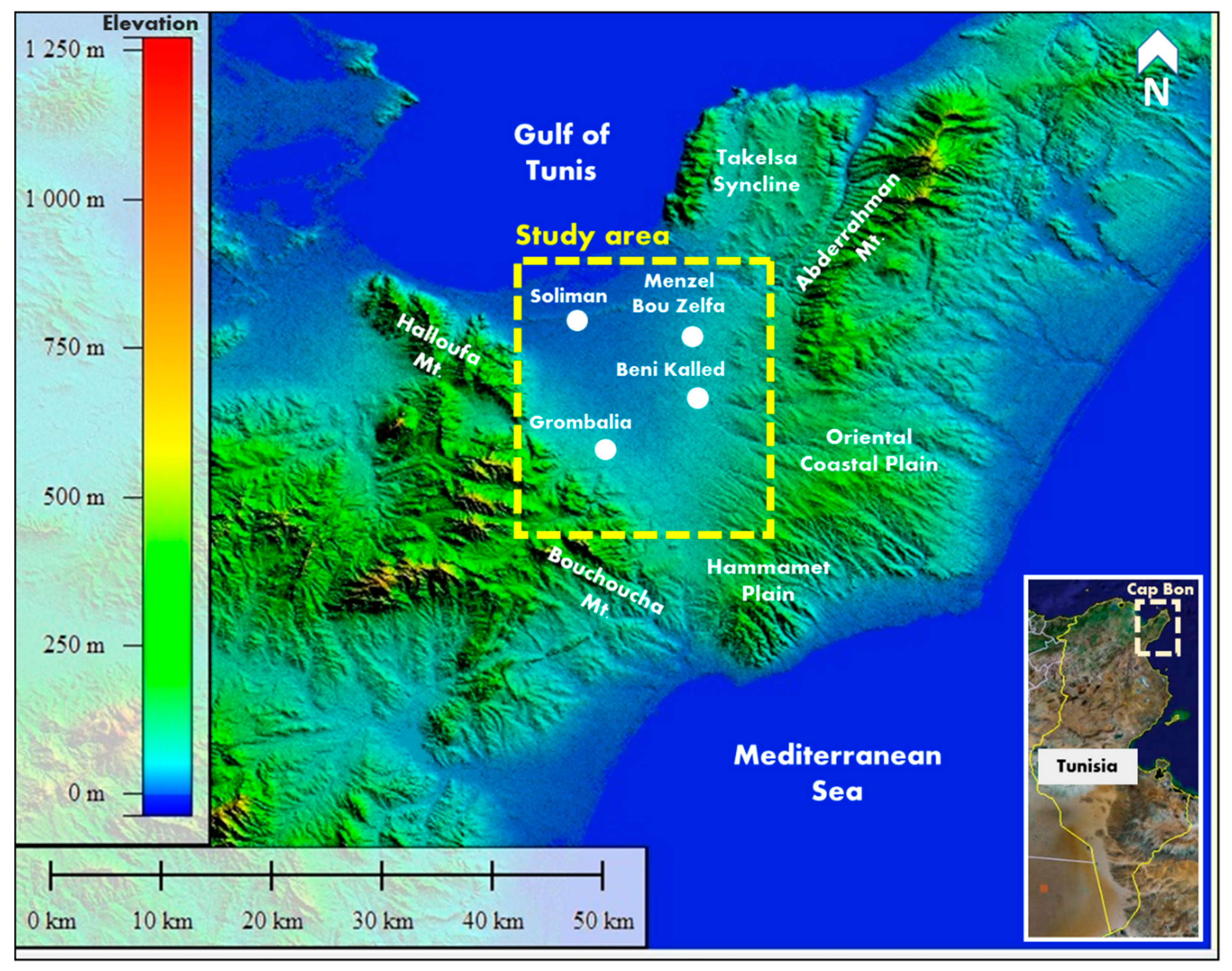
2. Site Description
3. Materials and Methods
4. Results and Discussion
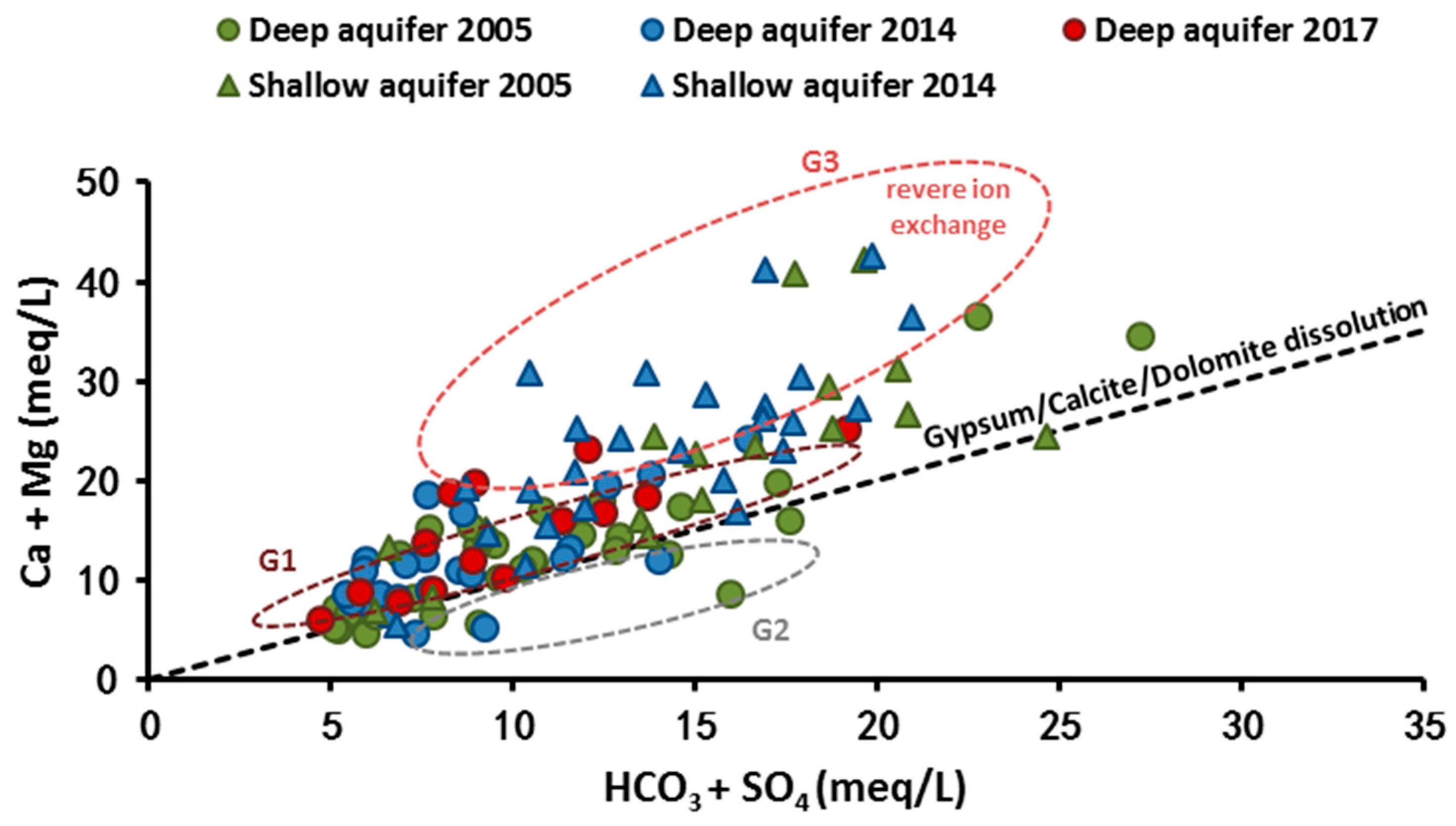
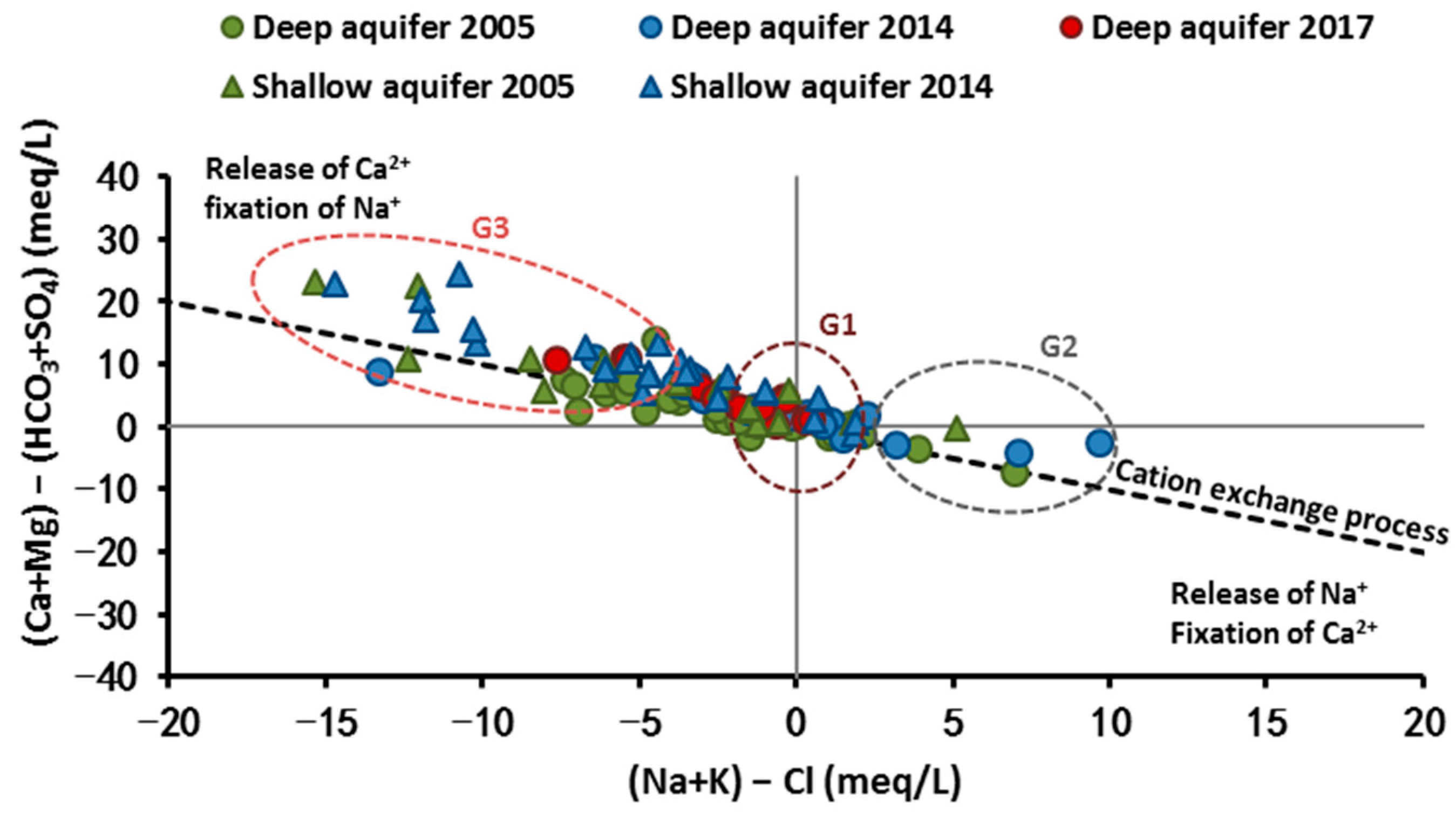
4.1. Mineralization Origin
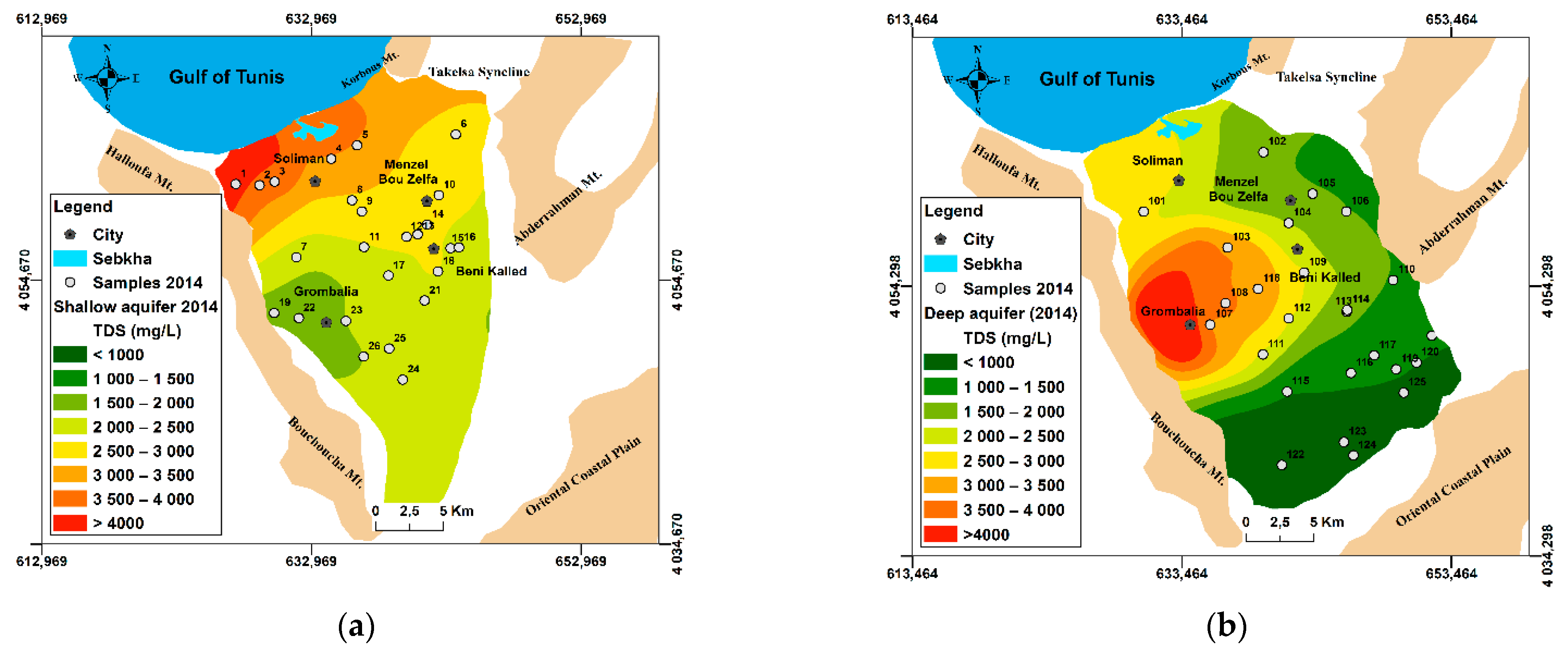
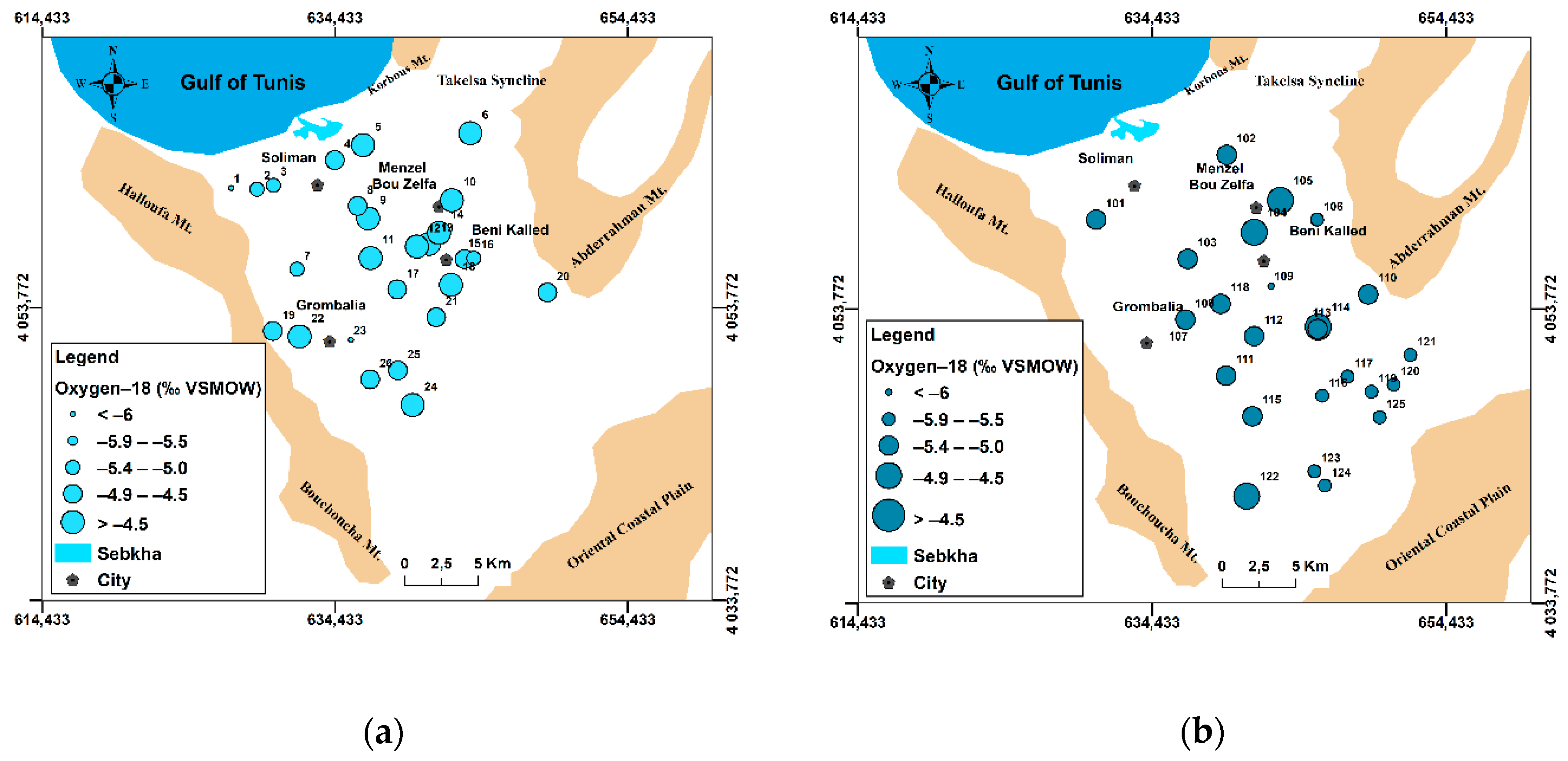
4.1.1. Salinity Evolution: Application of Geostatistical Mapping Tools
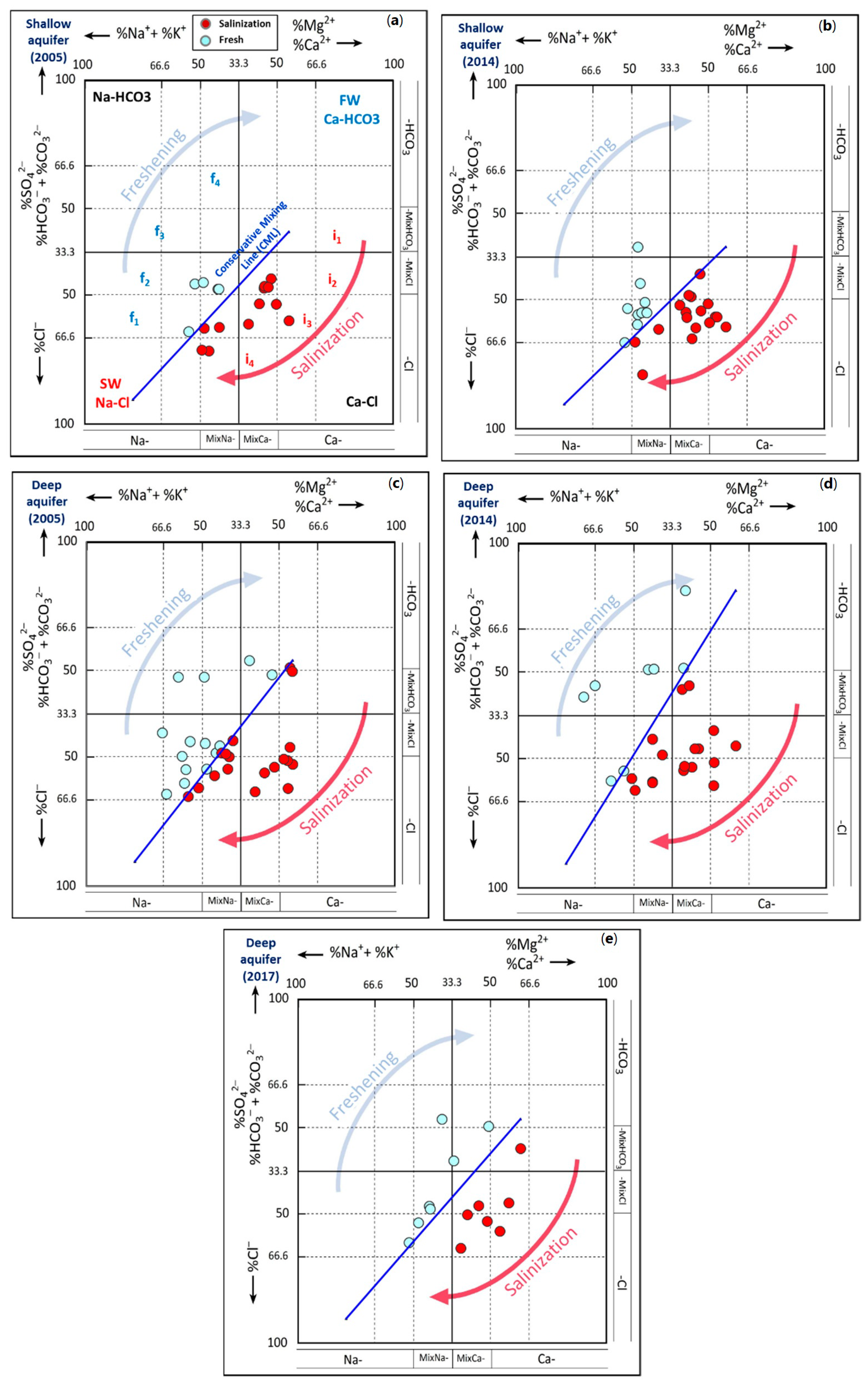
4.1.2. Salinity Origin: Use of Hydrochemical Facies Evolution Diagram
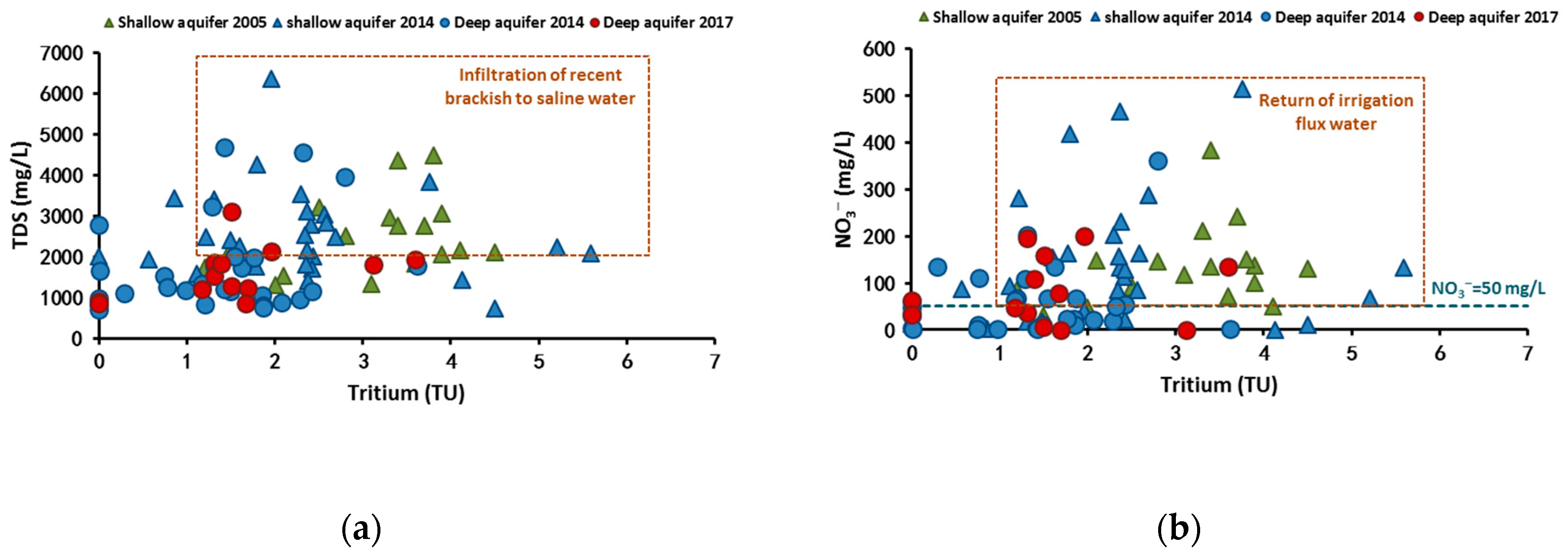
4.2. Recharge Processes and Water Quality Variation
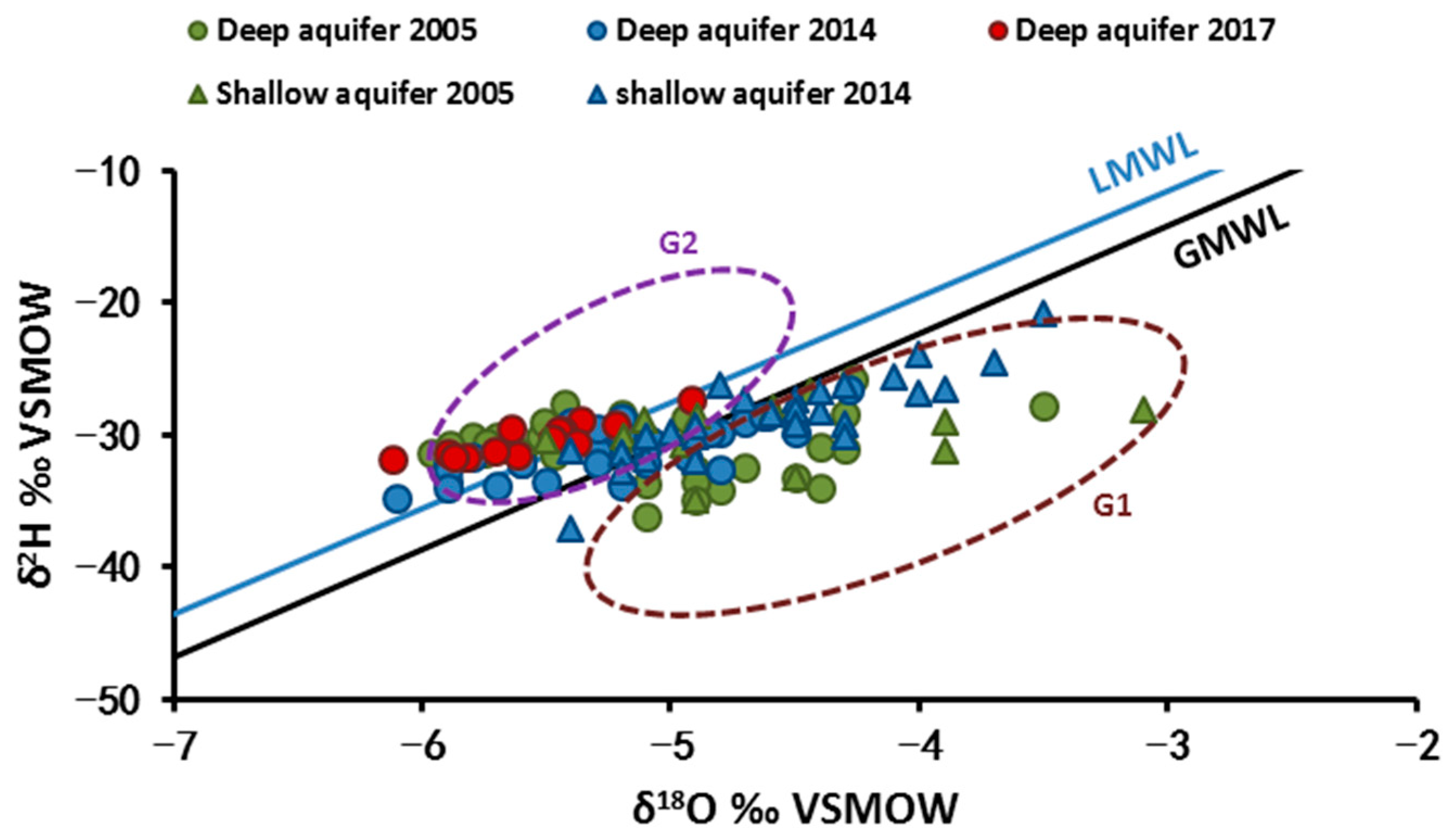
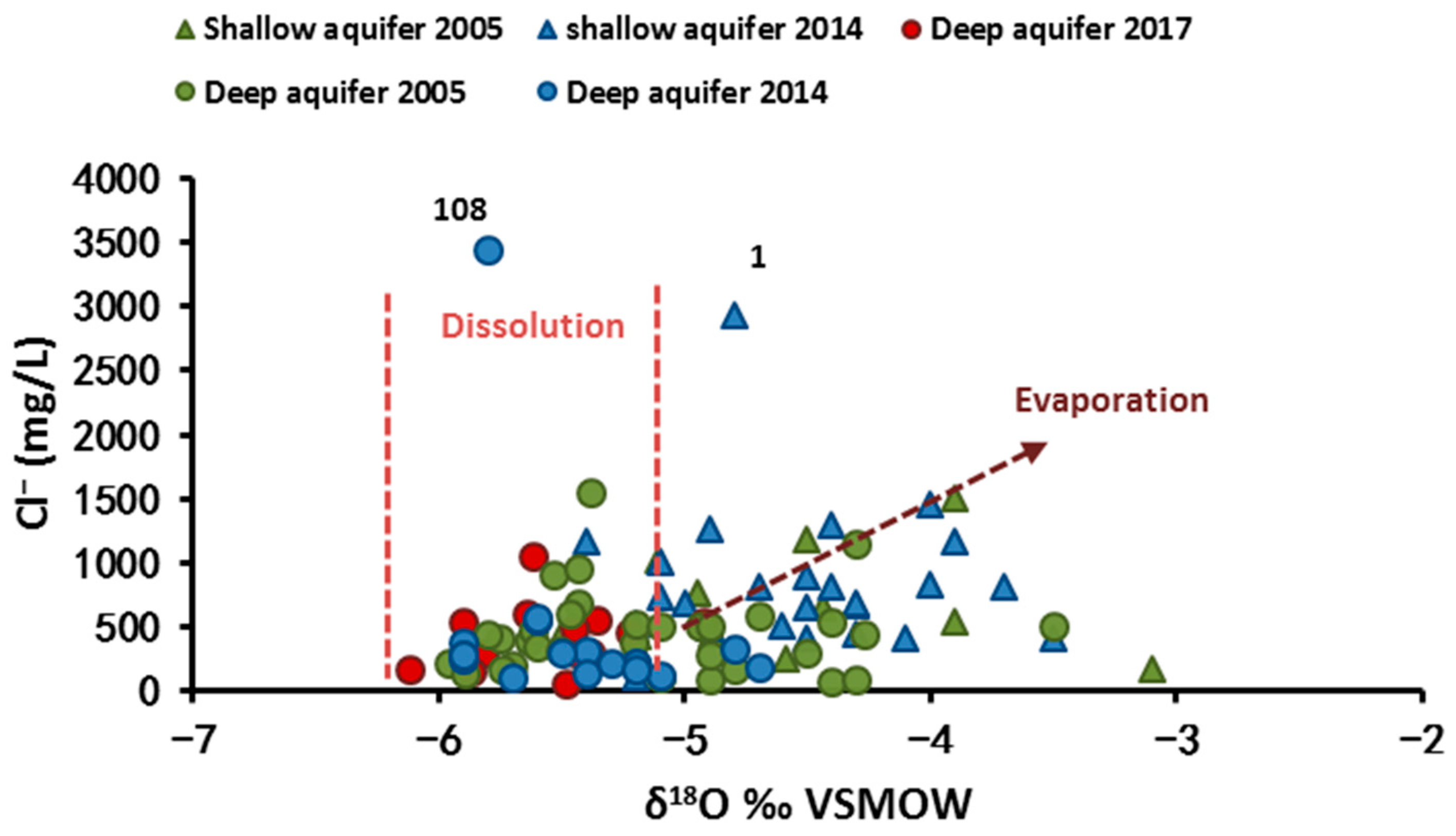
4.2.1. Water Stable Isotopes (δ18O and δ2H)
4.2.2. Tritium Isotope
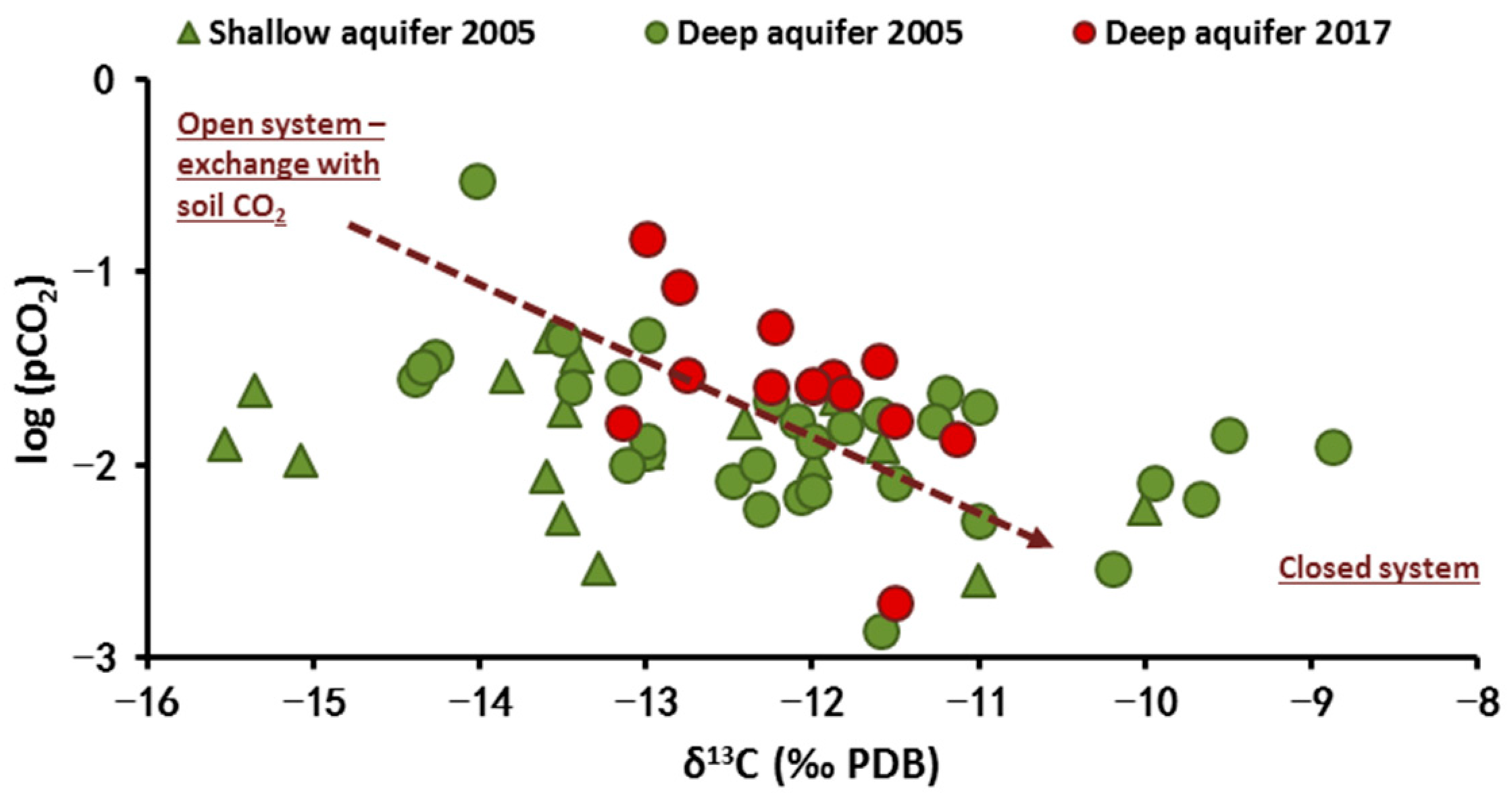
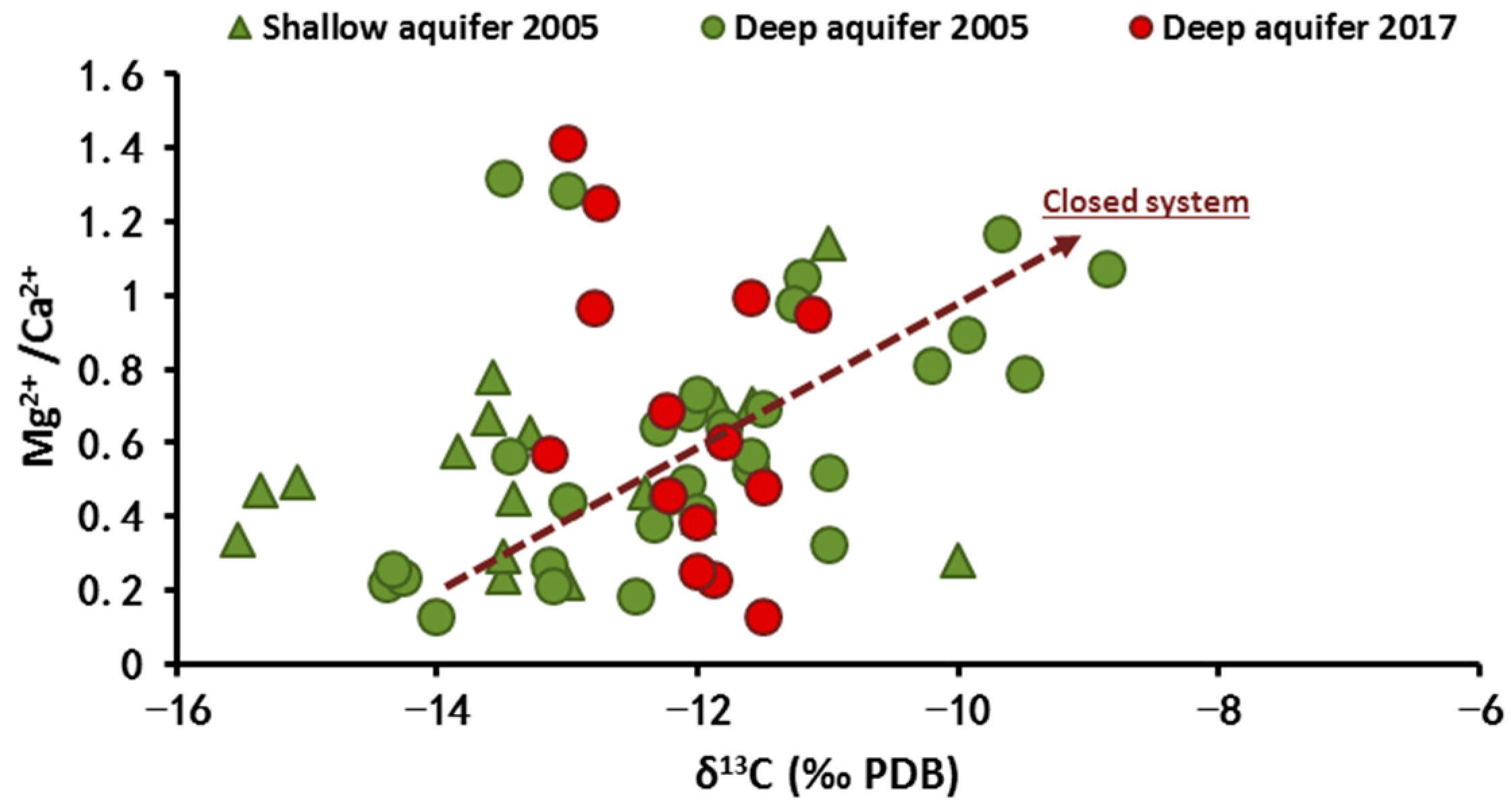
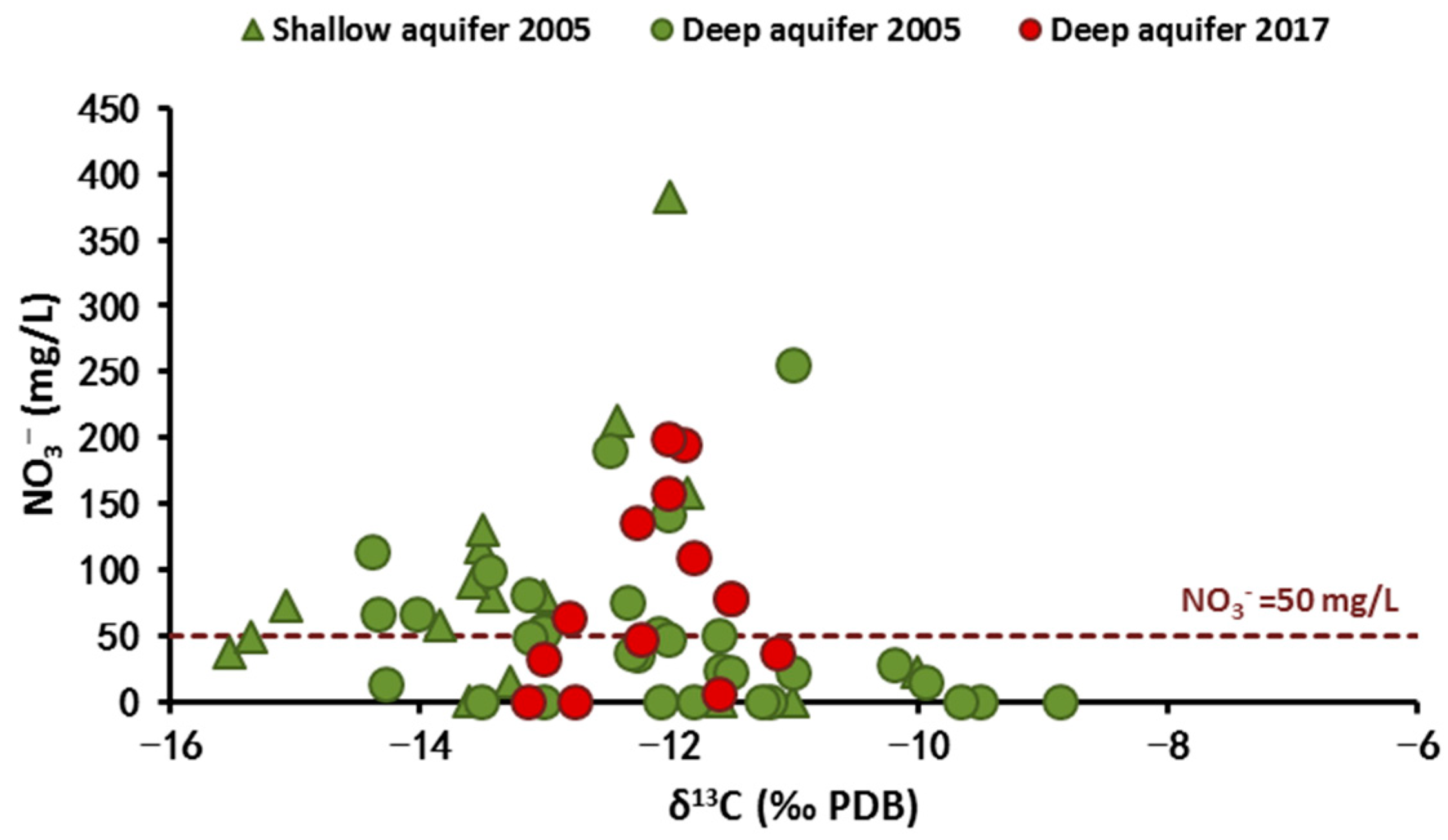
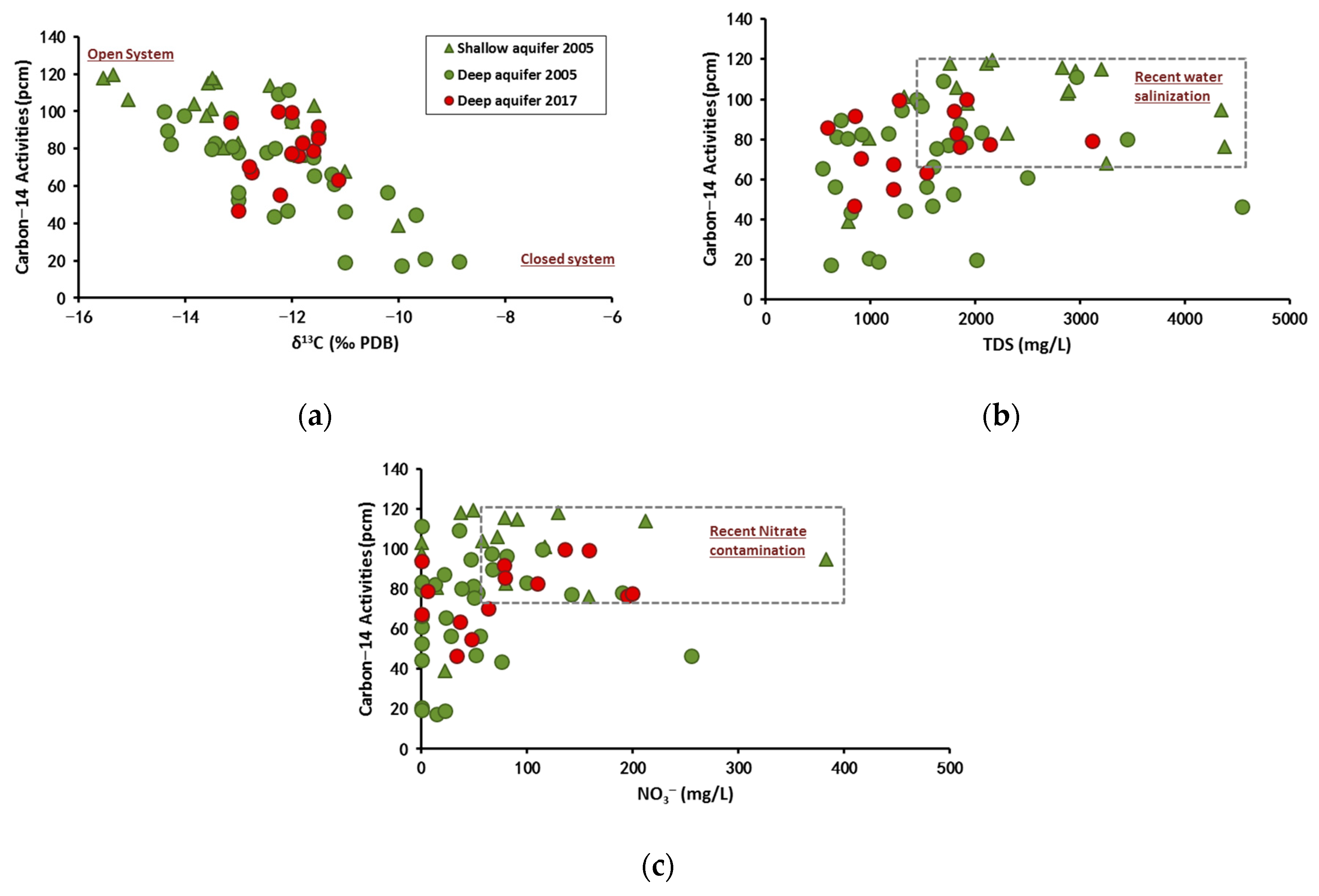
4.2.3. Carbon-13 Isotope
4.2.4. Carbon-14 Isotope
- The 14C activity of the TDIC of the samples (measured value)
- The 14C activity of soil gas (A 14Cg = 116 pmc)
- The 14C activity of the matrix (A 14Cm = 0 pmc)
- The 13C content of the TDIC of the samples (measured value)
- The 13C content of soil gas (δ13Cg = −16‰ V-PDB)
- The 13C content of the matrix (δ13Cm = 0‰ V-PDB)
- Physico-chemical parameters: temperature, pH and alkalinity
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ouhamdouch, S.; Bahir, M.; Ouazar, D.; Carreira, P.M.; Zouari, K. Evaluation of climate change impact on groundwater from semi-arid environment (Essaouira Basin, Morocco) using integrated approaches. Environ. Earth Sci. 2019, 78, 449. [Google Scholar] [CrossRef]
- Rakib, M.A.; Sasaki, J.; Matsuda, H.; Quraishi, S.B.; Mahmud, M.J.; Bodrud, D.M.; Bhuiyan, M.A. Groundwater salinization and associated co-contamination risk increase severe drinking water vulnerabilities in the southwestern coast of Bangladesh. Chemosphere J. 2020, 246, 125646. [Google Scholar] [CrossRef] [PubMed]
- Bani, A.; Daghari, I.; Hatira, A.; Chaabane, A.; Daghari, H. Sustainable management of a cropping system under salt stress conditions (Korba, Cap-Bon, Tunisia). Environ. Sci. Pollut. Res. 2020, 1–8. [Google Scholar] [CrossRef]
- Krishan, G. Groundwater Salinity. Curr. World Environ. 2019, 14, 186. [Google Scholar] [CrossRef]
- Bahir, M.; Ouazar, D.; Ouhamdouch, S.; Zouari, K. Assessment of groundwater mineralization of alluvial coastal aquifer of essaouira basin (Morocco) using the hydrochemical facies evolution diagram (HFE-Diagram). Groundw. Sustain. Dev. 2020, 11, 100487. [Google Scholar] [CrossRef]
- Gana, A.; Fouillen, B. Concurrences et Conflits Pour L’usage de L’eau en Tunisie: Étude de cas Dans la Région du Cap Bon, Le Carnet de l’IRMC, 21 February 2014. Available online: http://irmc.hypotheses.org/1412 (accessed on 21 February 2014).
- Gaaloul, N.; Candela, L.; Chebil, A.; Soussi, A.; Tamoh, K. Groundwater flow simulation at the Grombalia phreatic aquifer (Cap Bon, Northeastern Tunisia). Desalin. Water Treat. 2014, 52, 1997–2008. [Google Scholar] [CrossRef]
- Trabelsi, R.; Zouari, K.; Kammoun, S.; Trigui, M.R. Recharge and paleo-recharge of groundwater in different basins in Tunisia. Quat. Int. 2020, 547, 152–165. [Google Scholar] [CrossRef]
- Zaidi, F.K.; Nazzal, Y.; Jafri, M.K.; Naeem, M.; Ahmed, I. Reverse ion exchange as a major process controlling the groundwater chemistry in an arid environment: A case study from northwestern Saudi Arabia. Environ. Monit. Assess. 2015, 187, 607. [Google Scholar] [CrossRef]
- Kammoun, S.; Trabelsi, R.; Re, V.; Zouari, K.; Henchiri, J. Groundwater Quality Assessment in Semi-Arid Regions Using Integrated Approaches: The Case of Grombalia Aquifer (NE Tunisia). Environ. Monit. Assess. 2018, 190, 87. [Google Scholar] [CrossRef]
- Kammoun, S.; Re, V.; Trabelsi, R.; Zouari, K.; Daniele, S. Assessing Seasonal Variations and Aquifer Vulnerability in Coastal Aquifers of Semi-Arid Regions Using a Multi-Tracer Isotopic Approach: The Case of Grombalia (Tunisia). Hydrogeol. J. 2018, 26, 2575–2594. [Google Scholar] [CrossRef]
- Gaaloul, N. Gestion intégrée des ressources en eaux souterraines: Cas de la plaine du Cap Bon. La Houille Blanche 2008, 5, 38–44. [Google Scholar] [CrossRef]
- Charfi, S.; Zouari, K.; Feki, S.; Mami, E. Study of variation in groundwater quality in a coastal aquifer in north-eastern Tunisia using multivariate factor analysis. Quat. Int. 2013, 302, 199–209. [Google Scholar] [CrossRef]
- Lachaal, F.; Chekirbane, A.; Chargui, S.; Sellami, H.; Tsujimura, M.; Hezzi, H.; Mlayah, A. Water resources management strategies and its implications on hydrodynamic and hydrochemical changes of costal groundwater: Case of Grombalia shallow aquifer, NE Tunisia. J. Afr. Earth Sci. 2016, 124, 171–188. [Google Scholar] [CrossRef]
- Lachaal, F.; Chargui, S.; Messaoud, R.B.; Chekirbane, A.; Tsujimura, M.; Mlayah, A.; Leduc, C. Impacts of global changes on groundwater resources in north-east Tunisia: The case of the Grombalia phreatic aquifer. In Groundwater and Global Change in the Western Mediterranean Area; Springer: Cham, Switzerland, 2018; pp. 179–188. [Google Scholar]
- Moussa, A.B.; Zouari, K. Hydrochemical Investigation of Groundwater Contamination in the Grombalia Shallow Aquifer, Cap Bon Peninsula, Tunisia: Impact of Irrigation with Industrial Waste Water; Tech Open Access: Rijeka, Croatia, 2011; pp. 239–252. [Google Scholar]
- Charfi, S.; Trabelsi, R.; Zouari, K.; Chkir, N.; Charfi, H.; Rekaia, M. Isotopic and hydrochemical investigation of the Grombalia deep aquifer system, northeastern Tunisia. Carbonates Evaporites 2013, 28, 281–295. [Google Scholar] [CrossRef]
- CRDA. Annuaire de L’exploitation des Nappes Souterraines de L’année 2014 [Yearbook of Groundwater Exploitation of the Year 2014]; Technical Report; CRDA: Tunis, Tunisia, 2014. [Google Scholar]
- Simler, R.; Diagrammes. Laboratoire D’hydrogeologie D’avignon, Université D’avignon, Logiciel D’hydrochimie Multilangage en Distribution Libre. 2004. Available online: http://www.lha.univ-avignon.fr (accessed on 20 October 2020).
- Penna, D.; Stenni, B.; Wrede, S.; Bogaard, T.A.; Gobbi, A.; Borga, M.; Fischer, B.M.C.; Bonazza, M.; Charova, Z. On the reproducibility and repeatability of laser absorption spectroscopy measurements for [delta] 2H and [delta] 18O isotopic analysis. Hydrol. Earth Syst. Sci. 2010, 14, 1551. [Google Scholar] [CrossRef]
- Taylor, C.B. IAEA Isotope Hydrology Laboratory Technical Procedure; Note No.19; International Atomic Energy Agency: Vienna, Austria, 1976; pp. 1–30. [Google Scholar]
- Fontes, J.C. Un ensemble destiné à la mesure de l’activité du radiocarbone naturel par scintillation liquide. Rev. Géog. Phys. Géol. Dyn. 1971, 13, 67–86. [Google Scholar]
- WHO, World Health Organisation. Guidelines for Drinking-Water Quality, 4th ed.; Incorporating First Addendum: Geneva, Switzerland, 2017; ISBN 978-92-4-254995-9. [Google Scholar]
- Tlili, Z.B.; Hamzaoui, A.F.; Gueddari, M.; Bouhlila, R. Geochemistry and quality assessment of groundwater using graphical and multivariate statistical methods. A case study: Grombalia phreatic aquifer (Northeastern Tunisia). Arab. J. Geosci. 2013, 6, 3545–3561. [Google Scholar] [CrossRef]
- Re, V.; Sacchi, E.; Kammoun, S.; Tringali, C.; Trabelsi, R.; Zouari, K.; Daniele, S. Integrated socio-hydrogeological approach to tackle nitrate contamination in groundwater resources. The case of Grombalia Basin (Tunisia). Sci. Total Environ. 2017, 593, 664–676. [Google Scholar] [CrossRef]
- Fisher, R.S.; Mullican, W.F. Hydrochemical evolution of sodium-sulphate and sodium-chloride groundwater beneath the Northern Chihuahuan desert, Trans-Pecos, Texas, USA. Hydrogeol. J. 1997, 5, 4–16. [Google Scholar] [CrossRef]
- Rajmohan, N.; Elango, L. Identification and evolution of hydrogeochemical processes in the groundwater environment in an area of the Palar and Cheyyar River Basins, Southern India. Environ. Geol. 2004, 46, 47–61. [Google Scholar] [CrossRef]
- Garcia, G.M.; Hidalgo, M.D.V.; Blesa, M.A. Geochemistry of groundwater in the alluvial plain of Tucuman province, Argentina. Hydrogeol. J. 2001, 9, 597–610. [Google Scholar] [CrossRef]
- Lean, W.; Jankowski, J.; Lavitt, N. Groundwater quality and sustainability in an alluvial aquifer, Australia. In Groundwater, Past Achievements and Future Challenges; A. A. Balkema Publishers: Rotterdam, The Netherlands, 2000; pp. 567–573. [Google Scholar]
- Giménez, F.E. Dynamic of sea water interface using hydrochemical facies evolution diagram. Groundwater 2010, 48, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Song, X.; Currell, M.J. Identification of anthropogenic and natural inputs of SO4 into a karstic coastal groundwater system in northeast China, Evidence from major ions, δ13 CDIC and δ34 SSO4. Hydrol. Earth Syst. Sci. 2016, 20, 1983–1999. [Google Scholar] [CrossRef]
- Van, L.T.T.; Lertsirivorakul, R.; Bui, T.V.; Schulmeister, M.K. An application of HFE-D for evaluating seawater intrusion in coastal aquifers of Southern Vietnam. Groundwater 2020. [Google Scholar] [CrossRef]
- Kumar, P.S. Deciphering the groundwater–saline water interaction in a complex coastal aquifer in South India using statistical and hydrochemical mixing models. Model. Earth Syst. Environ. 2016, 2, 1–11. [Google Scholar] [CrossRef]
- Giménez, F.E. Use of the Hydrochemical Facies Diagram (HFE-D) for the evaluation of salinization by seawater intrusion in the coastal Oropesa Plain: Comparative analysis with the coastal Vinaroz Plain, Spain. HydroResearch 2019, 2, 76–84. [Google Scholar] [CrossRef]
- Appelo, C.A.J.; Willemsen, A. Geochemical calculations and observations on salt water intrusions, I. A combined geochemical/minxing cell model. J. Hydrol. 1987, 94, 313–330. [Google Scholar] [CrossRef]
- Fontes, J.C. Isotopes du Milieu et Cycle des Eaux Naturelles: Quelques Aspects; Thèse de Doctorat en Sciences, Université de Paris: Paris, France, 1976. [Google Scholar]
- Rozanski, K.; Araguás, A.L.; Gonfiantini, R. Isotopic patterns in modern global precipitation. Geophys. Monogr. 1993, 78, 1–36. [Google Scholar]
- Zouari, K.; Aranyossy, J.F.; Mamou, A.; Fontes, J.C. Etude isotopique et géochimique des mouvements et de l’évolution des solutions de la zone aérée des sols sous climat semi-aride (Sud tunisien); Stable and Radioactive Isotopes in the Study of the Unsaturated Soil Zone; IAEA-TECDOC-357: Vienna, Austria, 1985; pp. 121–144. [Google Scholar]
- GNIP. Global network of isotopes in precipitation. In Atlas of Isotopes Hydrology; IAEA: Vienna, Austria, 2007; pp. 89–93. [Google Scholar]
- Dulinski, M.; Rozanski, K.; Kania, J.; Karlikowska, J.; Korczynski, J.M.; Witczak, S.; Mochalski, P.; Opoka, M.; Sliwka, I.; Zuber, A. Groundwater Dating with Sulfur Hexafluoride: Methodology and Field Comparison with Tritium and Hydrodynamic Methods; International Atomic Energy Agency: Vienna, Austria, 2003. [Google Scholar]
- Lucas, L.L.; Unterweger, M.P. Comprehensive Review and critical Evaluation of the Half-Life of Tritium. J. Res. Natl. Inst. Stand. Technol. 2000, 105, 541–549. [Google Scholar] [CrossRef]
- Clark, I.; Fritz, P. Environmental Isotopes in Hydrogeology; CRC Press: Boca Raton, FL, USA, 1997; Volume I, pp. 104–328. [Google Scholar]
- Driscoll, F.G. Groundwater and Wells; Jonhson Division Publishers: Saint-Paul, MN, USA, 1986; p. 1089. [Google Scholar]
- Weijden, C.H.; Pacheco, F.A.L. Hydrochemistry, weathering and weathering rates on Madeira Island. J. Hydrol. 2003, 283, 122–145. [Google Scholar] [CrossRef]
- Adams, S.; Tredoux, G.; Harris, C.; Titus, R.; Pietersen, K. Hydrochemical characteristics of aquifers near Sutherland in the Western Karoo, South Africa. J. Hydrol. 2001, 241, 91–103. [Google Scholar] [CrossRef]
- Awad, S. Analysing groundwater using the 13C isotope. Phys. Procedia 2014, 55, 35–40. [Google Scholar] [CrossRef]
- Mook, W.G.; Vries, J.J. Environmental Isotopes in Hydrogeological Cycle-Principles and Applications. In Technical Documents in Hydrology; International Atomic Energy Agency (UNESCO/IAEA): Paris, France, 2000; Volumes 1–6. [Google Scholar]
- Pearson, F.J.; Hanshaw, B.B. Sources of dissolved carbonate species in groundwater and their effects on carbon-14 dating. Isot. Hydrol. 1970, 1970, 271–285. [Google Scholar]
- Tamers, M.A. Validity of radiocarbon dates on groundwater. Geophys. Surv. 1975, 2, 217–239. [Google Scholar] [CrossRef]
- Fontes, J.C.; Garnier, J.M. Determination of the initial 14C activity of the total dissolved carbon. A review of the existing models and a new approach. Water Resour. Res. 1979, 12, 399–413. [Google Scholar] [CrossRef]
- Salem, O.; Visser, J.H.; Dray, M.; Gonfiantini, R. Groundwater flow patterns in the western Libyan Arab Jamahiriya. In Arid Zone Hydrogeology; Investigations with Isotope Techniques; IAEA: Vienna, Austria, 1980; pp. 165–179. [Google Scholar]
- Eichinger, E. A contribution to the interpretation of 14C groundwater ages considering the example of partially confined sandstone aquifer. Radiocarbon 1983, 25, 347–356. [Google Scholar] [CrossRef]
- Gillon, M.; Barbecot, F.; Gibert, E.; Alvarado, J.C.; Marlin, C.; Massault, M. Open to closed system transition traced through the TDIC isotopic signature at the aquifer recharge stage, implications for groundwater 14C dating. Geochim. Cosmochim. Acta 2009, 73, 6488–6501. [Google Scholar] [CrossRef]
- Fang, S.C. Study on 14C dating analysis of deep groundwater resources on islands. J. Environ. Radioact. 2019, 208, 105994. [Google Scholar] [CrossRef]
- Chakroun, A.; Zaghbib, T.D.; Miskovsky, J.C.; Davaud, E. Two Tyrrhenian transgressive cycles in coastal deposits of the Cap Bon Peninsula, Tunisia. Quaternaire. Rev. Assoc. Française Étude Quat. 2009, 20, 215–226. [Google Scholar] [CrossRef]
- Re, V.; Zuppi, G.M. Influence of precipitation and deep saline groundwater on the hydrological systems of Mediterranean coastal plains: A general overview. Hydrol. Sci. J. 2011, 56, 966–980. [Google Scholar] [CrossRef]
- Tringali, C.; Re, V.; Siciliano, G.; Chkir, N.; Tuci, C.; Zouari, K. Insights and participatory actions driven by a socio-hydrogeological approach for groundwater management: The Grombalia Basin case study (Tunisia). Hydrogeol. J. 2017, 25, 1241–1255. [Google Scholar] [CrossRef]
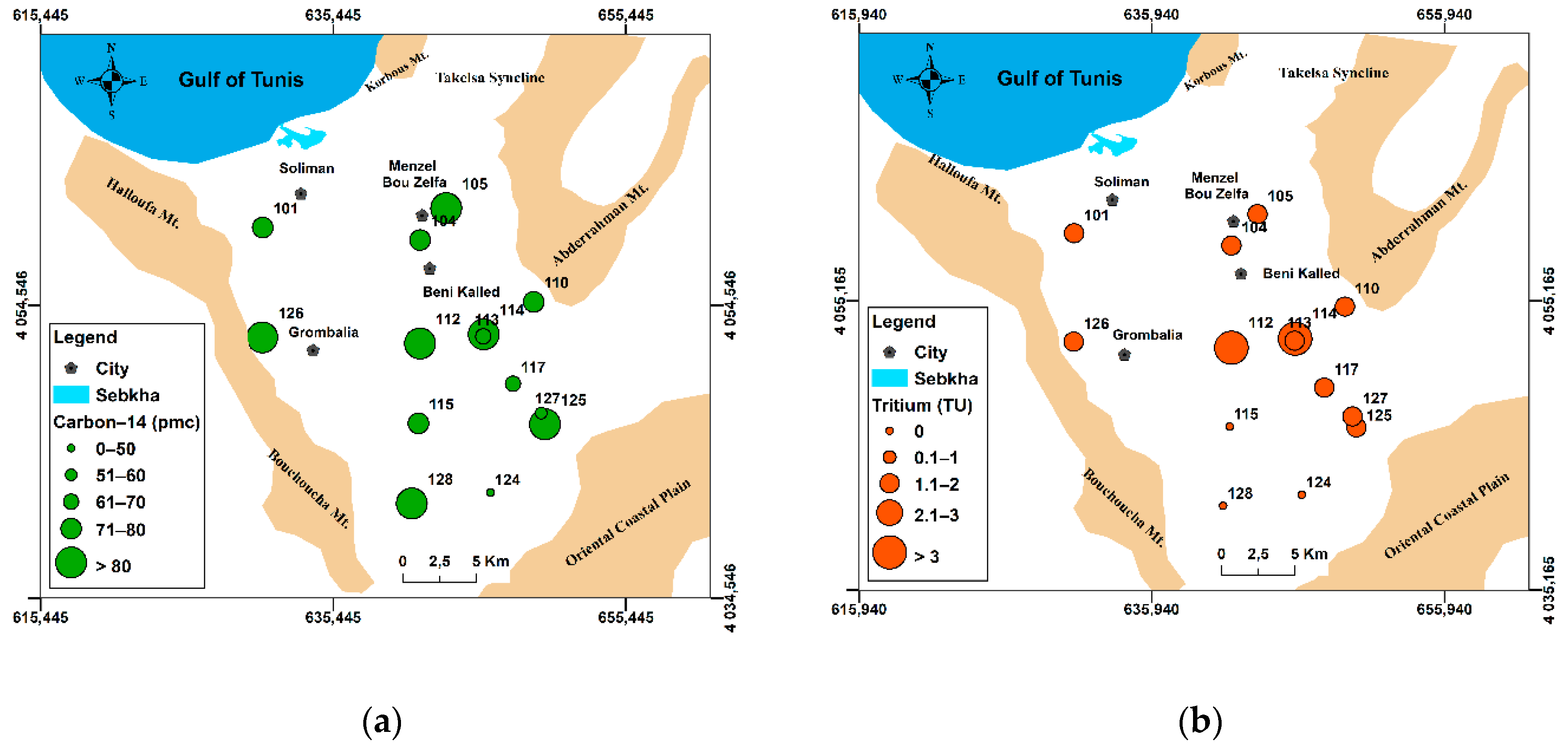
| Sample | 3H | 14C | 13C | Age | Age | Age | Age | Age | Age | Age | Age |
|---|---|---|---|---|---|---|---|---|---|---|---|
| TU | pmc | ‰ V-PDB | Apparent | Brut (Ao = 100) | Tamers | Pearson | F. & G. | AIEA | Evans | Eichinger | |
| 101 | 0 | 79.03 | −11.60 | 1890 | 3172 | Current | 514 | 1966 | 6796 | 380 | 133 |
| 104 | 1.31 | 76.46 | −11.88 | 2156 | 3446 | Current | 984 | 2558 | 7304 | 865 | 634 |
| 105 | 0.29 | 99.38 | −12 | Current | 1278 | Current | Current | 699 | 5195 | Current | Current |
| 110 | 0.77 | 77.67 | −12 | 2030 | 3316 | Current | 938 | 2764 | 7208 | 819 | 583 |
| 112 | 1.62 | 103.54 | −12.25 | Current | 940 | Current | Current | 724 | 5101 | Current | Current |
| 113 | 0.74 | 67.44 | −12.72 | 3165 | 4484 | Current | 2587 | 4843 | 8736 | 2489 | 2240 |
| 114 | 1.76 | 94.16 | −13.14 | 483 | 1724 | Current | 96 | 2916 | 6975 | 48 | Current |
| 115 | 0 | 70.43 | −12.80 | 2816 | 4125 | 1301 | 2280 | 3231 | 8381 | 2182 | 1917 |
| 117 | 2.07 | 63.44 | −11.13 | 3656 | 4990 | 29 | 1989 | 3971 | 8814 | 1887 | 1511 |
| 124 | 0 | 46.77 | −13 | 6105 | 7510 | 5249 | 5793 | 6406 | 12340 | 5726 | 5246 |
| 125 | 1.2 | 91.84 | −11.50 | 684 | 1931 | Current | Current | 1016 | 5570 | Current | Current |
| 126 | 0.98 | 82.88 | −11.80 | 1509 | 2779 | Current | 262 | 1996 | 6632 | 144 | Current |
| 127 | 1.17 | 55.10 | −12.23 | 4788 | 6155 | 2654 | 3933 | 5227 | 10378 | 3836 | 3595 |
| 128 | 1.2 | 85.77 | −11.50 | 1233 | 2496 | Current | Current | 2008 | 5784 | Current | Current |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kammoun, S.; Trabelsi, R.; Re, V.; Zouari, K. Coastal Aquifer Salinization in Semi-Arid Regions: The Case of Grombalia (Tunisia). Water 2021, 13, 129. https://doi.org/10.3390/w13020129
Kammoun S, Trabelsi R, Re V, Zouari K. Coastal Aquifer Salinization in Semi-Arid Regions: The Case of Grombalia (Tunisia). Water. 2021; 13(2):129. https://doi.org/10.3390/w13020129
Chicago/Turabian StyleKammoun, Siwar, Rim Trabelsi, Viviana Re, and Kamel Zouari. 2021. "Coastal Aquifer Salinization in Semi-Arid Regions: The Case of Grombalia (Tunisia)" Water 13, no. 2: 129. https://doi.org/10.3390/w13020129
APA StyleKammoun, S., Trabelsi, R., Re, V., & Zouari, K. (2021). Coastal Aquifer Salinization in Semi-Arid Regions: The Case of Grombalia (Tunisia). Water, 13(2), 129. https://doi.org/10.3390/w13020129






