Electrocoagulation as a Pretreatment for Electroxidation of E. coli
Abstract
1. Introduction
2. Materials and Methods
2.1. E. coli
2.2. Water Matrices
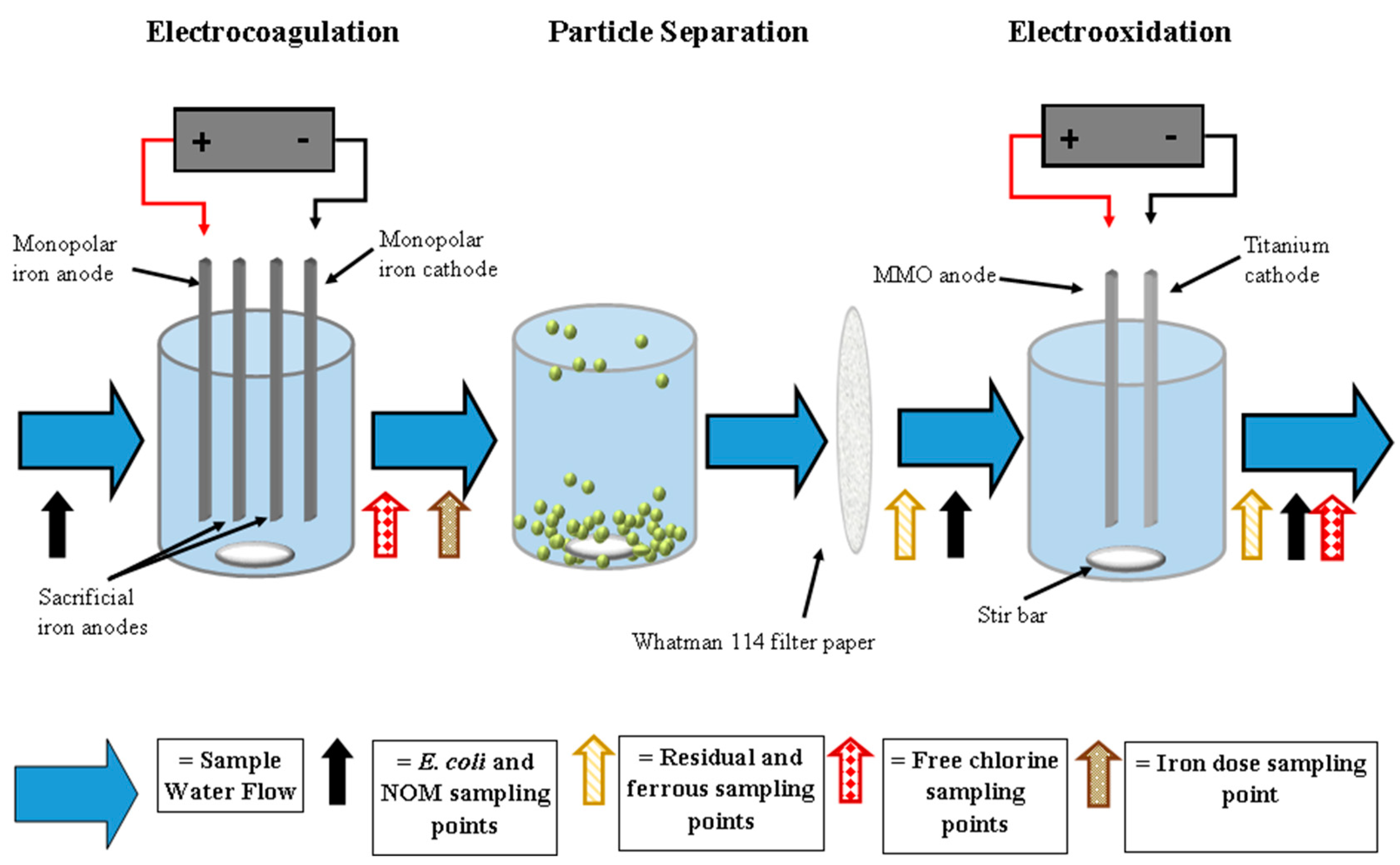
2.3. Testing Apparatus and Operation
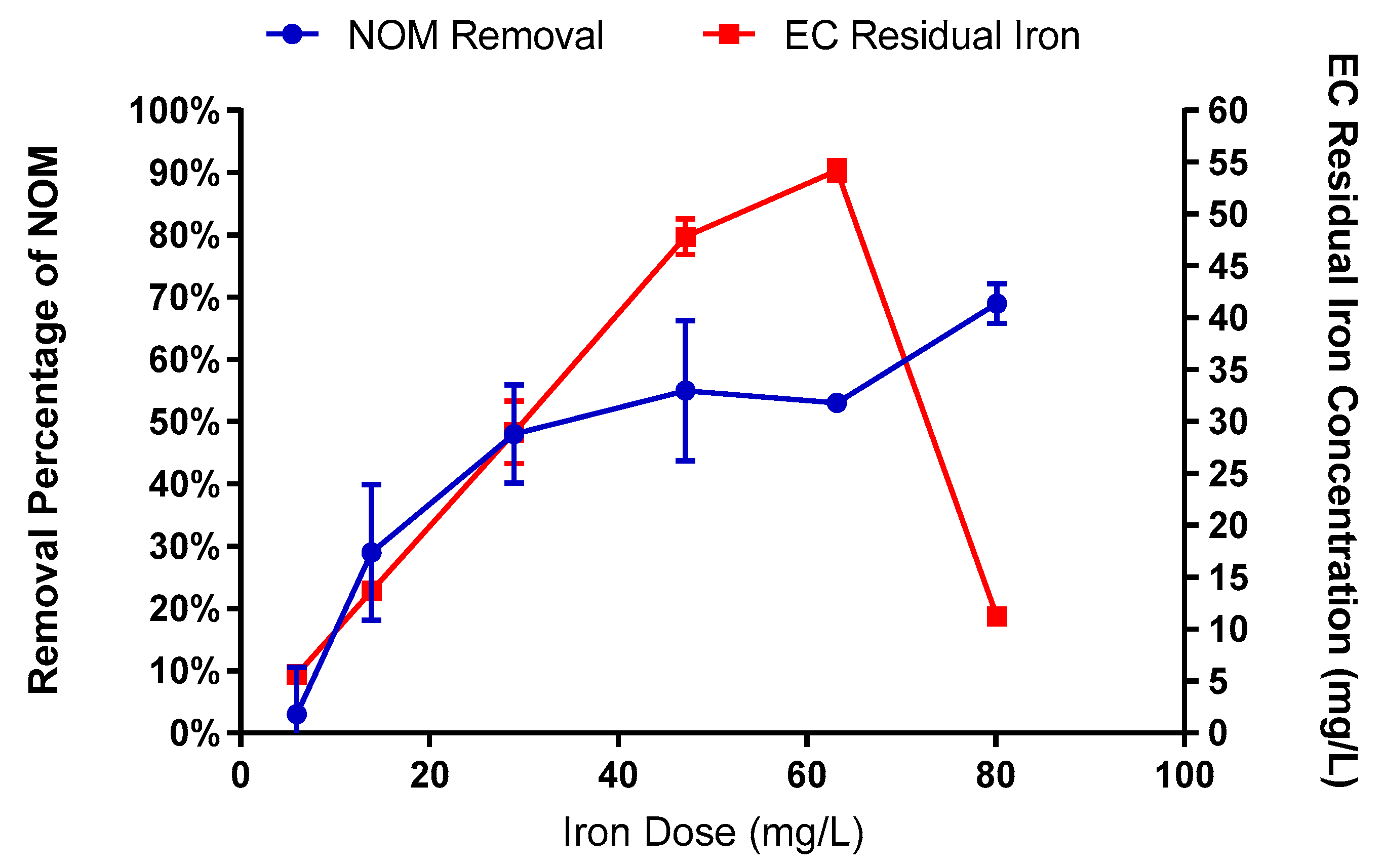
2.3.1. Electrocoagulation
2.3.2. Particle Separation
2.3.3. Electrooxidation
2.4. Analytical
2.5. Data Analysis and Quality Control
3. Results
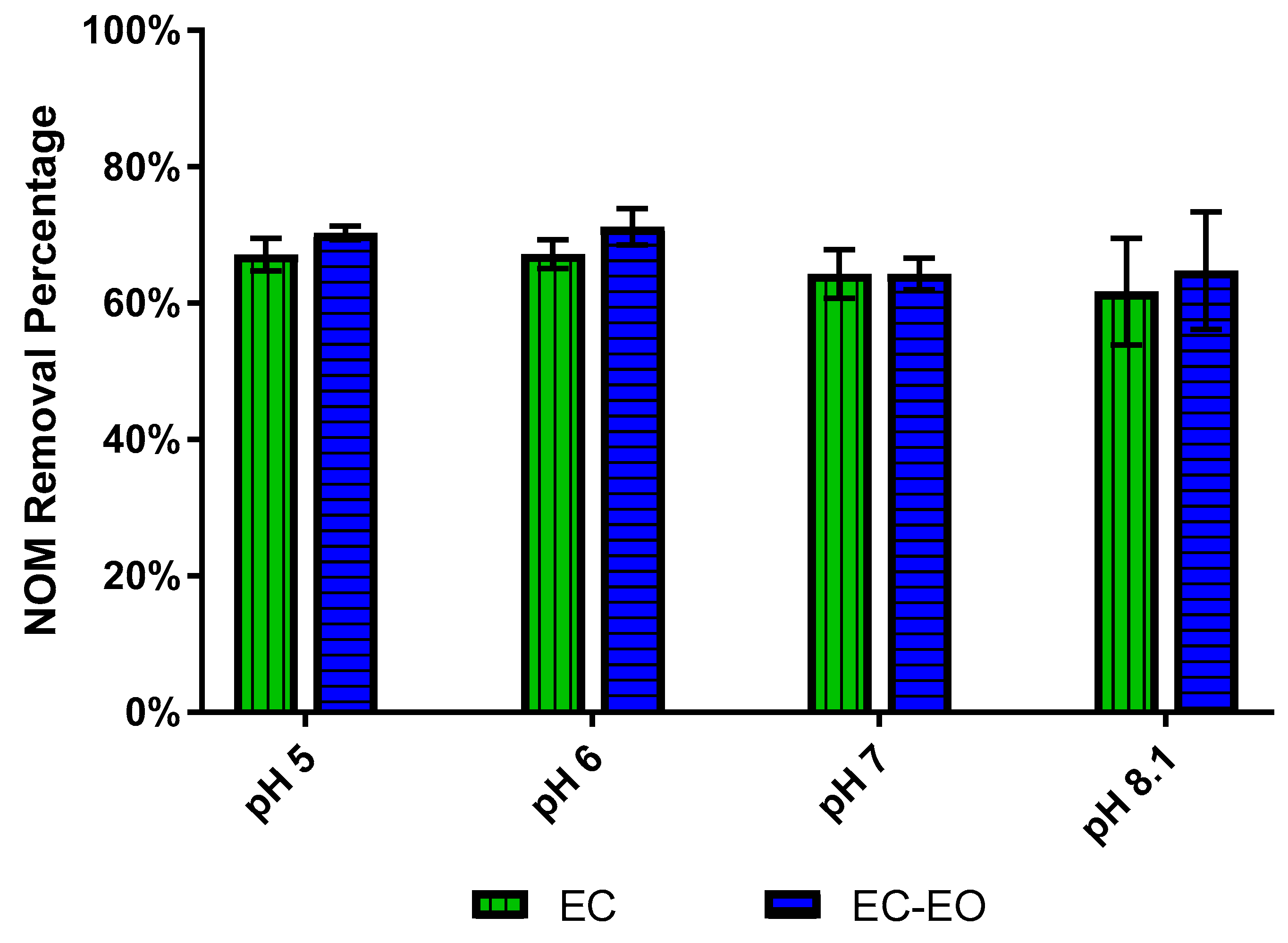
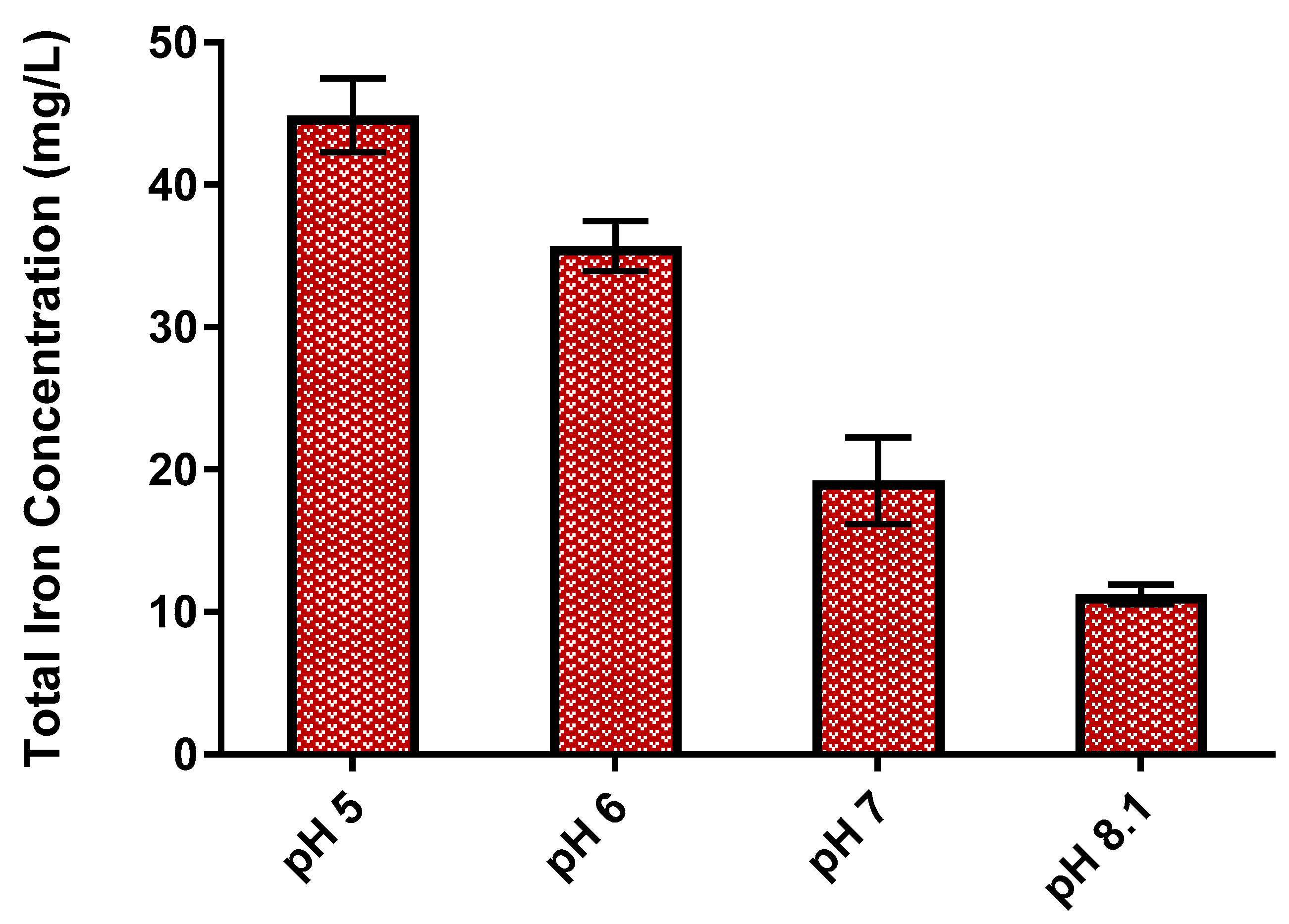
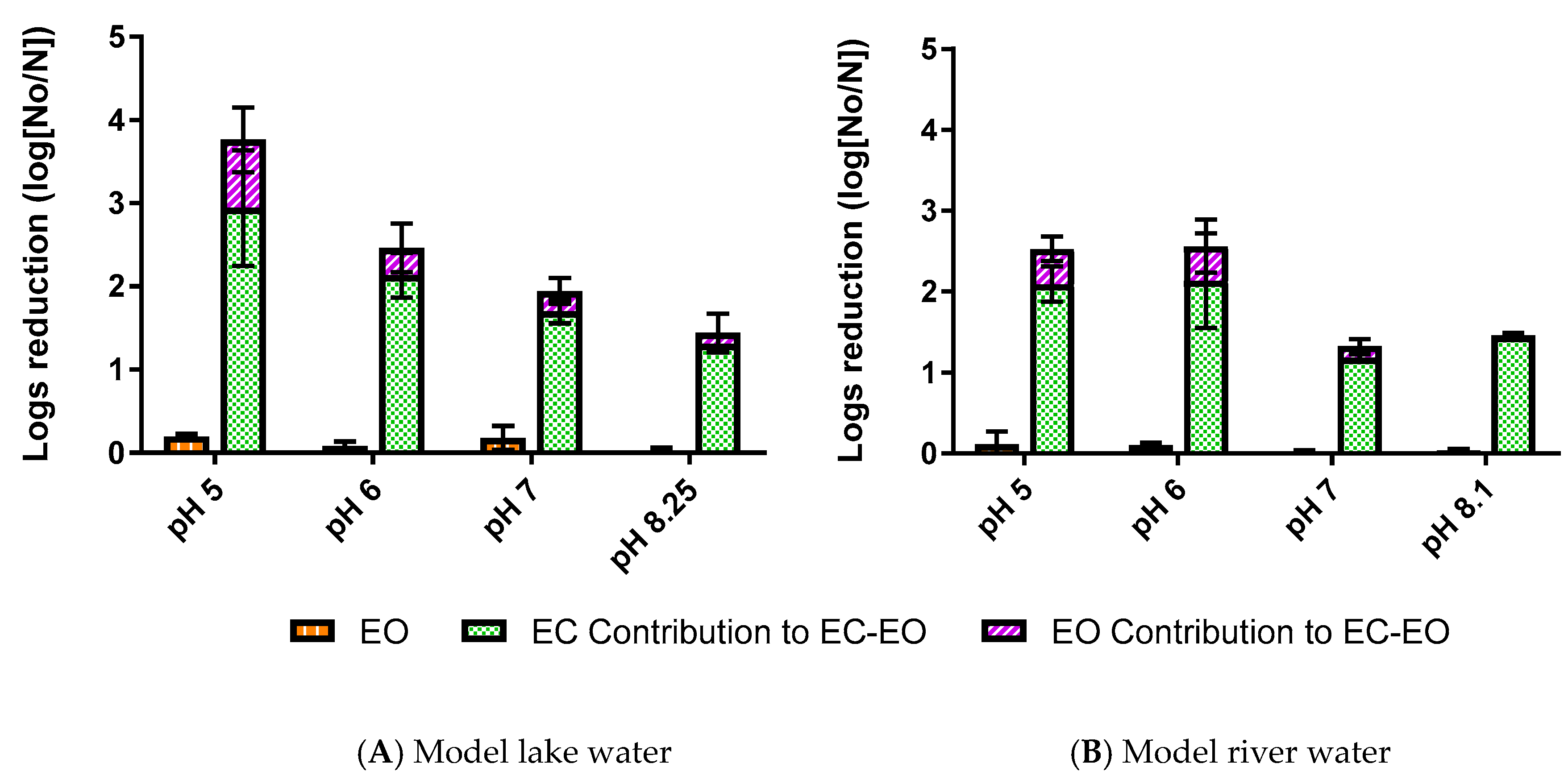
3.1. Impact of Water Quality on E. coli Mitigation
3.1.1. E. coli Mitigation by EO
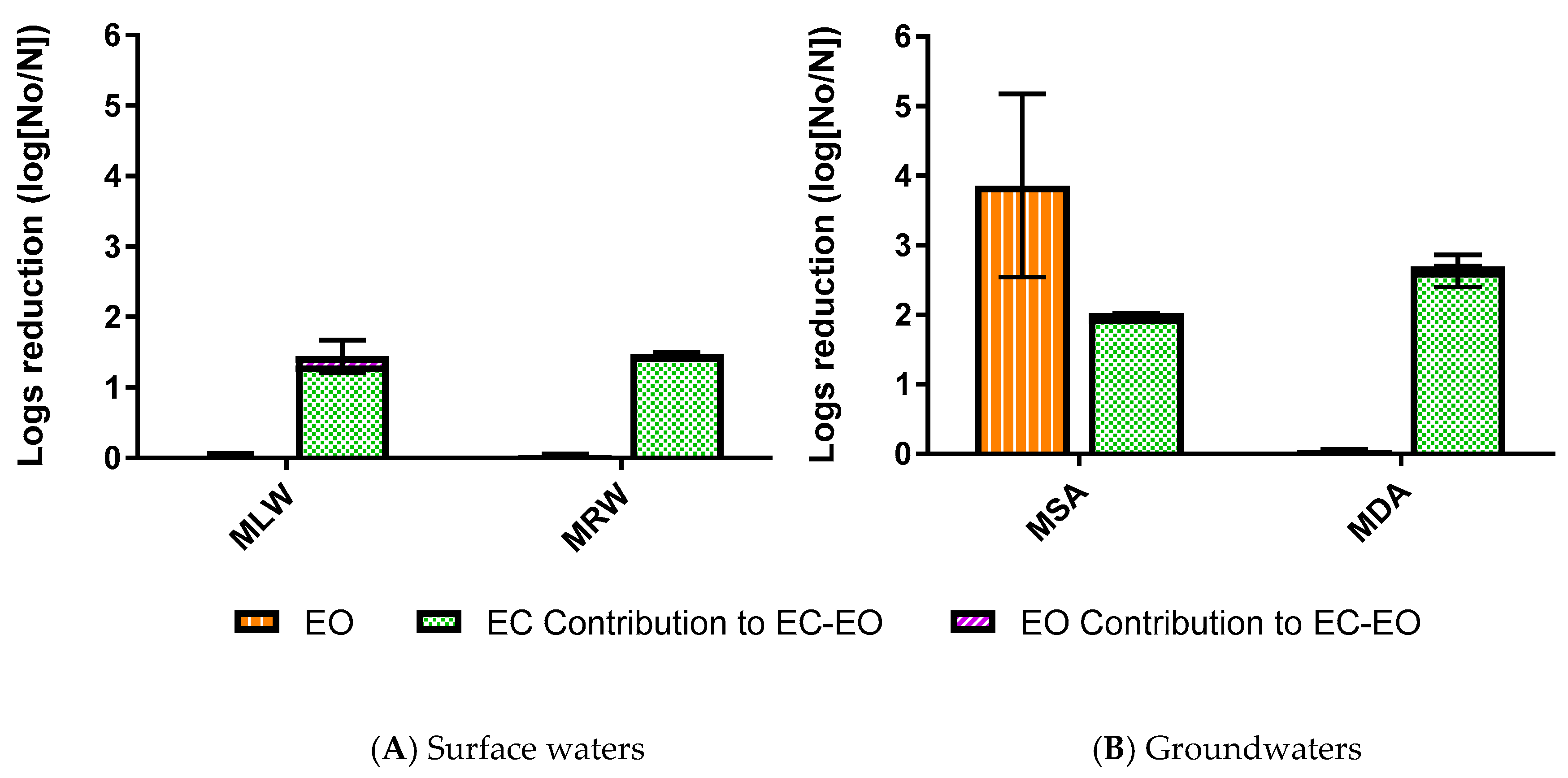
3.1.2. E. coli Mitigation by EC-EO
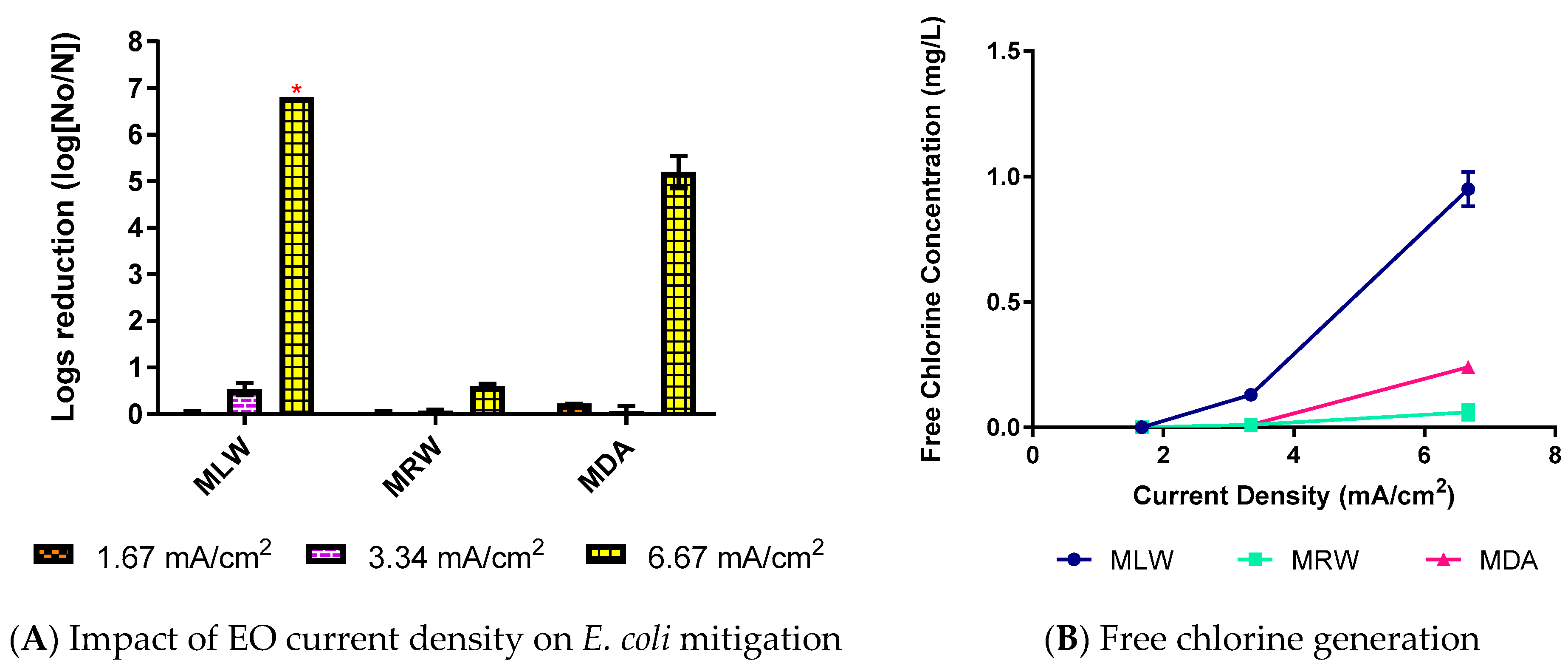
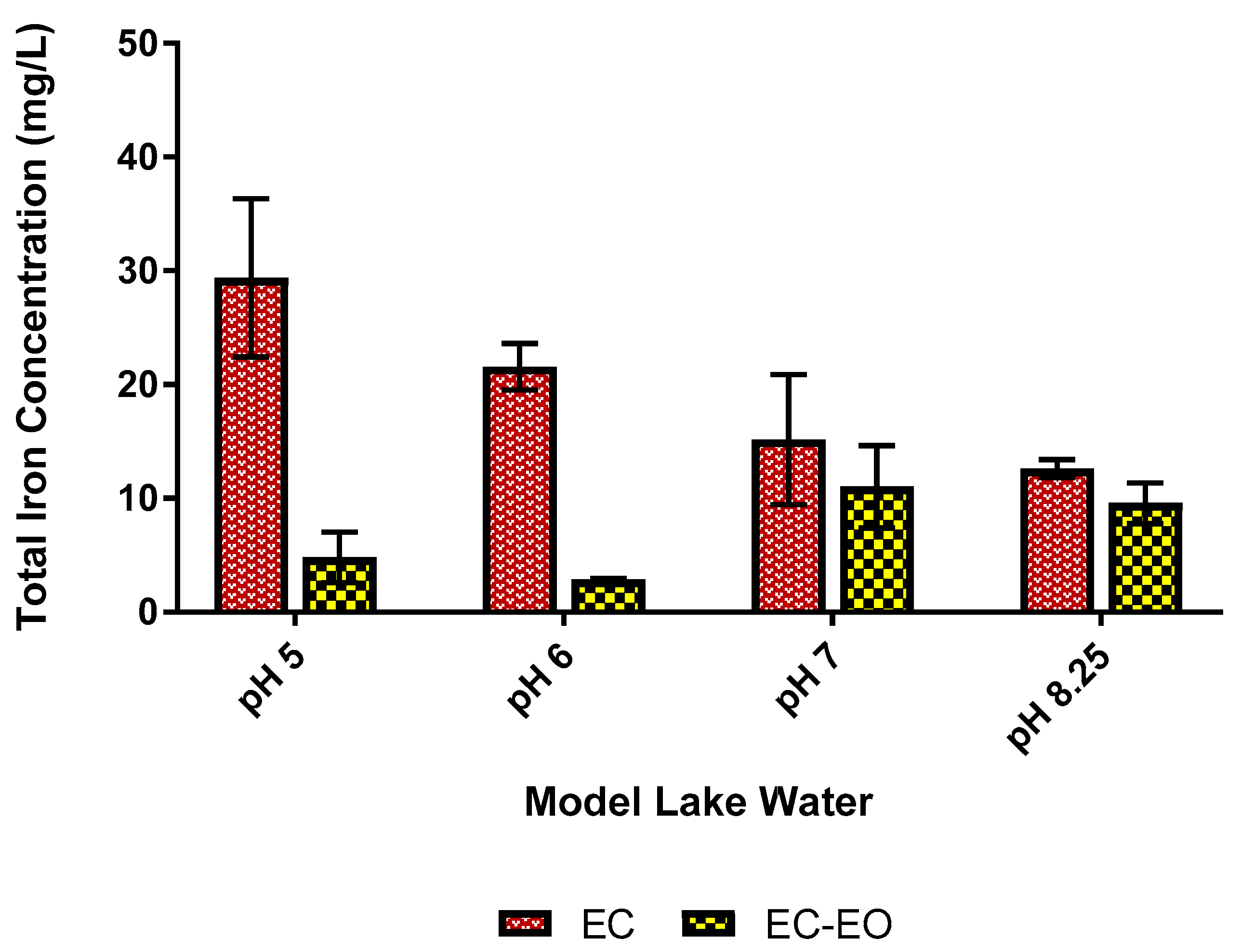
3.2. Impact of Enhanced EC-EO for E. coli Mitigation
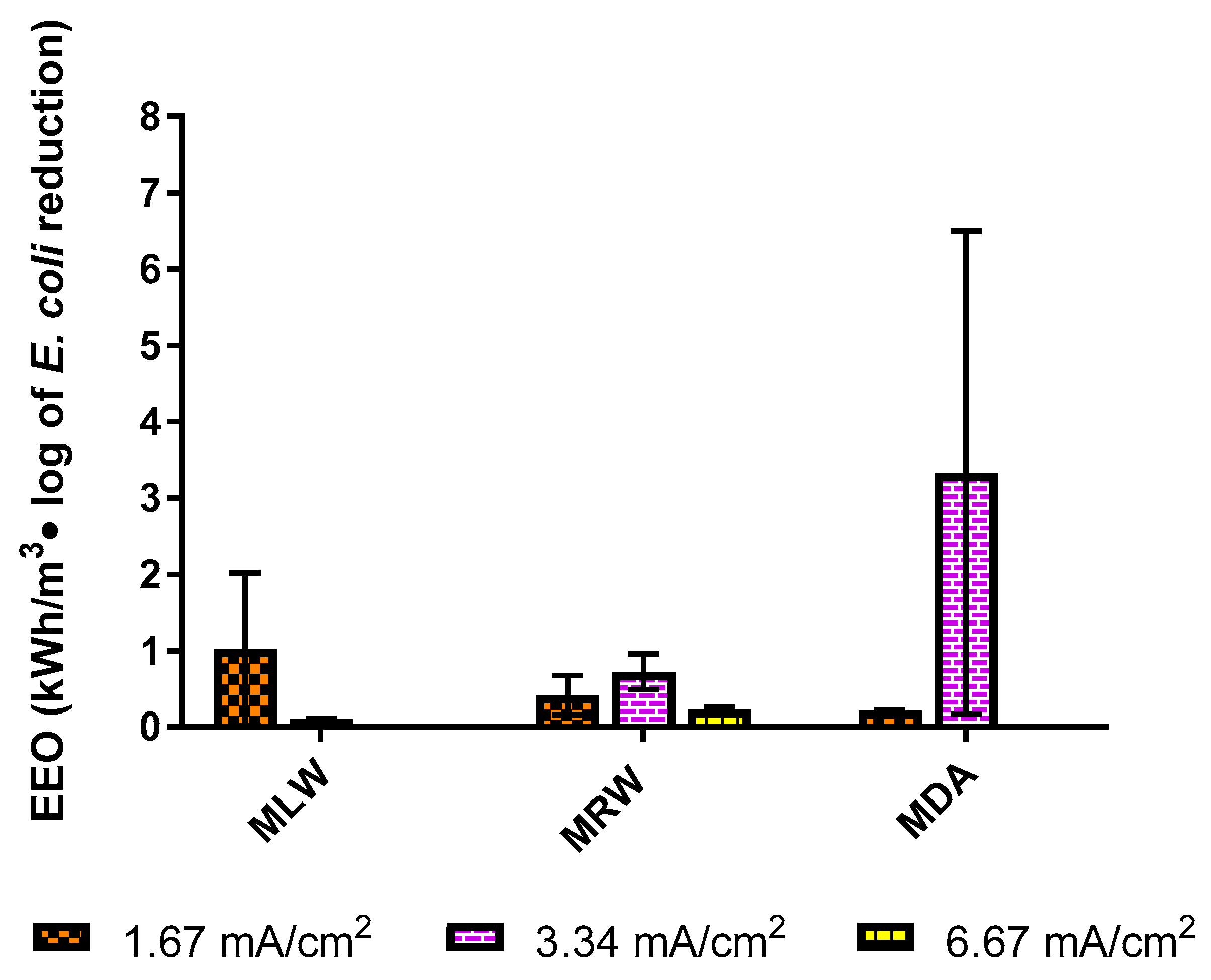
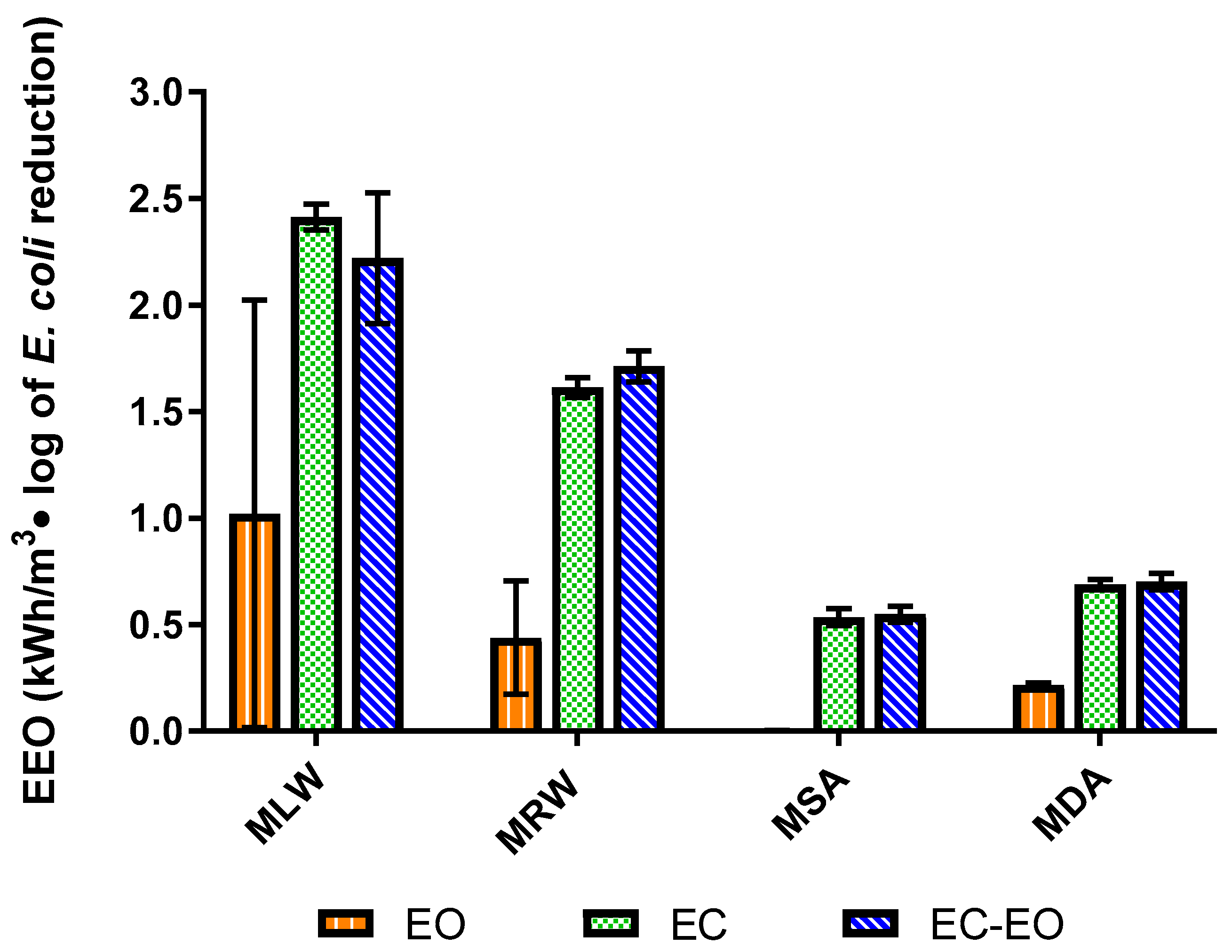
3.3. Electrical Efficiency of Electrochemical E. coli Mitigation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Treatment Process | Parameter | Water Matrix | |||
|---|---|---|---|---|---|
| Model Lake Water | Model River Water | Model Shallow Aquifer | Model Deep Aquifer | ||
| EC | Final pH | 9.59 ± 0.13 | 9.55 ± 0.07 | 8.93 ± 0.14 | 9.13 ± 0.07 |
| NOM Removal % | 8.2 ± 4.1 | 61.7 ± 7.8 | N/A | N/A | |
| Free Chlorine (mg/L) | BDL | BDL | BDL | BDL | |
| Total Residual Iron (mg/L) | 12.6 ± 0.8 | 11.2 ± 0.7 | 17.1 ± 0.8 | 15.2 ± 4.0 | |
| EO | Final pH | 8.01 ± 0.03 | 7.98 ± 0.07 | 7.78 ± 0.02 | 7.77 ± 0.01 |
| NOM Removal % | 14.1 ± 0.9 | 2.2 ± 1.7 | N/A | N/A | |
| Free Chlorine (mg/L) | BDL | BDL | 0.074 ± 0.008 | BDL | |
| EC-EO | Final pH | 9.53 ± 0.09 | 9.47 ± 0.03 | 8.90 ± 0.15 | 9.02 ± 0.07 |
| NOM Removal % | 9.1 ± 5.4 | 64.8 ± 8.6 | N/A | N/A | |
| Free Chlorine (mg/L) | BDL | BDL | 0.010 ± 0.014 | BDL | |
| Initial pH | Model Lake Water | Model River Water | ||||
|---|---|---|---|---|---|---|
| EC | EO | EC-EO | EC | EO | EC-EO | |
| Baseline | 9.59 ± 0.13 | 8.01 ± 0.03 | 9.53 ± 0.09 | 9.55 ± 0.07 | 7.98 ± 0.07 | 9.47 ± 0.03 |
| 7 | 9.23 ± 0.09 | 7.23 ± 0.03 | 9.10 ± 0.17 | 8.75 ± 0.18 | 7.21 ± 0.02 | 8.69 ± 0.20 |
| 6 | 7.11 ± 0.01 | 6.29 ± 0.04 | 6.93 ± 0.01 | 7.06 ± 0.02 | 6.29 ± 0.03 | 6.80 ± 0.06 |
| 5 | 6.88 ± 0.03 | 5.49 ± 0.03 | 6.65 ± 0.08 | 6.82 ± 0.04 | 5.34 ± 0.04 | 6.40 ± 0.04 |
References
- Latham, M.; Impellitteri, C. Small Drinking Water Systems Research and Development; United States Environmental Protection Agency: Washington, DC, USA, 2016.
- National Research Council. Safe Water From Every Tap; National Academies Press: Washington, DC, USA, 1997; ISBN 978-0-309-05527-7. [Google Scholar]
- Allaire, M.; Wu, H.; Lall, U. National trends in drinking water quality violations. Proc. Natl. Acad. Sci. 2018, 115, 2078–2083. [Google Scholar] [CrossRef] [PubMed]
- USEPA Revised Total Coliform Rule. Fed. Regist. 2013. Available online: https://www.govinfo.gov/content/pkg/FR-2013-02-13/pdf/2012-31205.pdf (accessed on 27 November 2019).
- USEPA National primary drinking water regulations: Long Term 1 Enhanced Surface Water Treatment Rule. Final rule. Fed. Regist. 2002, 67, 1811–1844.
- Bagga, A.; Chellam, S.; Clifford, D.A. Evaluation of iron chemical coagulation and electrocoagulation pretreatment for surface water microfiltration. J. Memb. Sci. 2008, 309, 82–93. [Google Scholar] [CrossRef]
- Comninellis, C.; Chen, G. Electroflotation. In Electrochemistry for the Environment; Chen, X., Ed.; Springer: Berlin, Germany, 2010; pp. 263–277. [Google Scholar]
- Delaire, C.; Van Genuchten, C.M.; Nelson, K.L.; Amrose, S.E.; Gadgil, A.J. Escherichia coli attenuation by Fe electrocoagulation in synthetic Bengal groundwater: Effect of pH and natural organic matter. Environ. Sci. Technol. 2015, 49, 9945–9953. [Google Scholar] [CrossRef] [PubMed]
- Ghernaout, D.; Badis, A.; Kellil, A.; Ghernaout, B. Application of electrocoagulation in Escherichia coli culture and two surface waters. Desalination 2008, 219, 118–125. [Google Scholar] [CrossRef]
- Ndjomgoue-Yossa, A.C.; Nanseu-Njiki, C.P.; Kengne, I.M.; Ngameni, E. Effect of electrode material and supporting electrolyte on the treatment of water containing Escherichia coli by electrocoagulation. Int. J. Environ. Sci. Technol. 2015, 12, 2103–2110. [Google Scholar] [CrossRef]
- Matilainen, A.; Vepsäläinen, M.; Sillanpää, M. Natural organic matter removal by coagulation during drinking water treatment: A review. Adv. Colloid Interface Sci. 2010, 159, 189–197. [Google Scholar] [CrossRef]
- Dubrawski, K.L.; Mohseni, M. In-situ identification of iron electrocoagulation speciation and application for natural organic matter (NOM) removal. Water Res. 2013, 47, 5371–5380. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. Enhanced Coagulation and Enhanced Precipitative Softening Guidance Manual; United States Environmental Protection Agency: Washington, DC, USA, 1999.
- Vepsäläinen, M.; Ghiasvand, M.; Selin, J.; Pienimaa, J.; Repo, E.; Pulliainen, M.; Sillanpää, M. Investigations of the effects of temperature and initial sample pH on natural organic matter (NOM) removal with electrocoagulation using response surface method (RSM). Sep. Purif. Technol. 2009, 69, 255–261. [Google Scholar] [CrossRef]
- Aguilar, Z.G.; Coreño, O.; Salazar, M.; Sirés, I.; Brillas, E.; Nava, J.L. Ti|Ir–Sn–Sb oxide anode: Service life and role of the acid sites content during water oxidation to hydroxyl radicals. J. Electroanal. Chem. 2018, 820, 82–88. [Google Scholar] [CrossRef]
- Chen, G. Electrochemical technologies in wastewater treatment. Sep. Purif. Technol. 2004, 38, 11–41. [Google Scholar] [CrossRef]
- Ghernaout, D.; Naceur, M.W.; Aouabed, A. On the dependence of chlorine by-products generated species formation of the electrode material and applied charge during electrochemical water treatment. Desalination 2011, 270, 9–22. [Google Scholar] [CrossRef]
- Jeong, J.; Kim, J.Y.; Cho, M.; Choi, W.; Yoon, J. Inactivation of Escherichia coli in the electrochemical disinfection process using a Pt anode. Chemosphere 2007, 67, 652–659. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Huang, Z.-H.; Lim, T.-T. Recent development of mixed metal oxide anodes for electrochemical oxidation of organic pollutants in water. Appl. Catal. A Gen. 2014, 480, 58–78. [Google Scholar] [CrossRef]
- Kraft, A. Electrochemical water disinfection: A short review. Platin. Met. Rev. 2008, 52, 177–185. [Google Scholar] [CrossRef]
- Kraft, A.; Stadelmann, M.; Blaschke, M.; Kreysig, D.; Sandt, B.; Schro, F.; Der, È.; Rennau, J. Electrochemical water disinfection Part I: Hypochlorite production from very dilute chloride solutions. J. Appl. Electrochem. 1999, 29, 859–866. [Google Scholar] [CrossRef]
- Aquino Neto, S.; de Andrade, A.R. Electrooxidation of glyphosate herbicide at different DSA® compositions: pH, concentration and supporting electrolyte effect. Electrochim. Acta 2009, 54, 2039–2045. [Google Scholar] [CrossRef]
- Martínez-Huitle, C.A.; Brillas, E. Electrochemical alternatives for drinking water disinfection. Angew. Chemie Int. Ed. 2008, 47, 1998–2005. [Google Scholar] [CrossRef]
- Hu, S.; Hu, J.; Liu, B.; Wang, D.; Wu, L.; Xiao, K.; Liang, S.; Hou, H.; Yang, J. In situ generation of zero valent iron for enhanced hydroxyl radical oxidation in an electrooxidation system for sewage sludge dewatering. Water Res. 2018, 145, 162–171. [Google Scholar] [CrossRef]
- Anfruns-Estrada, E.; Bruguera-Casamada, C.; Salvadó, H.; Brillas, E.; Sirés, I.; Araujo, R.M. Inactivation of microbiota from urban wastewater by single and sequential electrocoagulation and electro-Fenton treatments. Water Res. 2017, 126, 450–459. [Google Scholar] [CrossRef]
- Yeon Kim, J.; Lee, C.; Love, D.C.; Sedlak, D.L.; Yoon, J.; Nelson, K.L. Inactivation of MS2 Coliphage by Ferrous Ion and Zero-Valent Iron Nanoparticles. Environ. Sci. Technol. 2011, 45, 6978–6984. [Google Scholar]
- Jeong, J.; Kim, C.; Yoon, J. The effect of electrode material on the generation of oxidants and microbial inactivation in the electrochemical disinfection processes. Water Res. 2009, 43, 895–901. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, C.E.; Andaya, C.; Urtiaga, A. Assessment of disinfection and by-product formation during electrochemical treatment of surface water using a Ti/IrO2 anode. Chem. Eng. J. 2015, 264, 411–416. [Google Scholar] [CrossRef]
- Linares-Hernández, I.; Barrera-Díaz, C.; Bilyeu, B.; Juárez-GarcíaRojas, P.; Campos-Medina, E. A combined electrocoagulation–electrooxidation treatment for industrial wastewater. J. Hazard. Mater. 2010, 175, 688–694. [Google Scholar] [CrossRef]
- Cotillas, S.; Llanos, J.; Cañizares, P.; Mateo, S.; Rodrigo, M.A. Optimization of an integrated electrodisinfection/electrocoagulation process with Al bipolar electrodes for urban wastewater reclamation. Water Res. 2013, 47, 1741–1750. [Google Scholar] [CrossRef]
- Llanos, J.; Cotillas, S.; Cañizares, P.; Rodrigo, M.A. Effect of bipolar electrode material on the reclamation of urban wastewater by an integrated electrodisinfection/electrocoagulation process. Water Res. 2014, 53, 329–338. [Google Scholar] [CrossRef]
- Thurston-Enriquez, J.A.; Haas, C.N.; Gerba, C.P.; Jacangelo, J. Chlorine inactivation of adenovirus 40 and feline calicivirus. Appl. Environ. Microbiol. 2003, 69, 3979–3985. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. Guide Standard and Protocol for Testing Microbiological Water Purifiers; United States Environmental Protection Agency: Washington, DC, USA, 1987.
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater, 22nd ed.; Rice, E.W., Baird, R.B., Eaton, A.D., Clesceri, R.S., Eds.; APHA AWWA WEF: Washington, DC, USA, 2012; ISBN 9780875530130. [Google Scholar]
- Heffron, J.; Ryan, D.R.; Mayer, B.K. Sequential electrocoagulation-electrooxidation for virus mitigation in drinking water. Water Res. 2019, 160, 435–444. [Google Scholar] [CrossRef]
- Maher, E.K.; O’Malley, K.N.; Heffron, J.; Huo, J.; Mayer, B.K.; Wang, Y.; McNamara, P.J. Analysis of operational parameters, reactor kinetics, and floc characterization for the removal of estrogens via electrocoagulation. Chemosphere 2019, 220, 1141–1149. [Google Scholar] [CrossRef]
- Lakshmanan, D.; Clifford, D.A.; Samanta, G. Ferrous and ferric ion generation during iron electrocoagulation. Environ. Sci. Technol. 2009, 43, 3853–3859. [Google Scholar] [CrossRef]
- Gu, Z.; Liao, Z.; Schulz, M.; Davis, J.R.; Baygents, J.C.; Farrell, J. Estimating dosing rates and energy consumption for electrocoagulation using Iron and aluminum electrodes. Ind. Eng. Chem. Res. 2009, 48, 3112–3117. [Google Scholar] [CrossRef]
- Sasson, M.B.; Calmano, W.; Adin, A. Iron-oxidation processes in an electroflocculation (electrocoagulation) cell. J. Hazard. Mater. 2009, 171, 704–709. [Google Scholar] [CrossRef] [PubMed]
- Devilliers, D.; Mahé, E. Modified titanium electrodes: Application to Ti/TiO2/PbO2 dimensionally stable anodes. Electrochim. Acta 2010, 55, 8207–8214. [Google Scholar] [CrossRef]
- Jeong, J.; Kim, J.Y.; Yoon, J. The role of reactive oxygen species in the electrochemical inactivation of microorganisms. Environ. Sci. Technol. 2006, 40, 6117–6122. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. Method 3050B Acid Digestion of Sediments, Sludges, and Soils 1.0 Scope and Application; United States Environmental Protection Agency: Washington, DC, USA, 1996.
- Bolton, J.R.; Bircher, K.G.; Tumas, W.; Tolman, C.A. Figures-of-merit for the technical development and application of advanced oxidation processes. J. Adv. Oxid. Technol. 1996, 1, 13–17. [Google Scholar] [CrossRef]
- R Core Team R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing 2018. Available online: https://www.R-project.org/ (accessed on 17 October 2019).
- Kraft, A.; Blaschke, M.; Kreysig, D.; Sandt, B.; Schro, F.; Der, È.; Rennau, J. Electrochemical water disinfection. Part II: Hypochlorite production from potable water, chlorine consumption and the problem of calcareous deposits. Appl. Electochemistry 1999, 29, 895–902. [Google Scholar] [CrossRef]
- Tanneru, C.T.; Chellam, S. Mechanisms of virus control during iron electrocoagulation--microfiltration of surface water. Water Res. 2012, 46, 2111–2120. [Google Scholar] [CrossRef]
- Crittenden, J.C.; Trussell, R.R.; Hand, D.W.; Howe, K.J.; Tchobanoglous, G. MWH’s Water Treatment: Principles and Design: Third Edition; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012; ISBN 9780470405390. [Google Scholar]
- Koparal, A.S.; Yildiz, Y.Ş.; Keskinler, B.; Demircioǧlu, N. Effect of initial pH on the removal of humic substances from wastewater by electrocoagulation. Sep. Purif. Technol. 2008, 59, 175–182. [Google Scholar] [CrossRef]
- Delaire, C.; van Genuchten, C.M.; Amrose, S.E.; Gadgil, A.J. Bacteria attenuation by iron electrocoagulation governed by interactions between bacterial phosphate groups and Fe(III) precipitates. Water Res. 2016, 103, 74–82. [Google Scholar] [CrossRef]
- Stumm, W.; Lee, G.F. Oxygenation of Ferrous Iron. Ind. Eng. Chem. Res. 1961, 53, 143–146. [Google Scholar] [CrossRef]
- Heffron, J.; McDermid, B.; Mayer, B.K. Bacteriophage inactivation as a function of ferrous iron oxidation. Environ. Sci. Water Res. Technol. 2019, 5, 1309–1317. [Google Scholar] [CrossRef]
- Heffron, J.; McDermid, B.; Maher, E.; McNamara, P.J.; Mayer, B.K. Mechanisms of virus mitigation and suitability of bacteriophages as surrogates in drinking water treatment by iron electrocoagulation. Water Res. 2019, 163, 114877. [Google Scholar] [CrossRef] [PubMed]
| Water Type | Alkalinity (mg/L) a | Chloride (mg/L) b | Turbidity (NTU) c | DOC (mg/L) d | pH e | Approximate Conductivity (µS/cm) f | |
|---|---|---|---|---|---|---|---|
| Surface Waters | Model Lake Water (Lake Michigan) | 95.0 | 13.3 | 3.40 | ≈2.7 | 8.25 | 260 |
| Model River Water (Mississippi River) | 119 | 11.4 | 11.8 | ≈8.0 | 8.1 | 300 | |
| Ground Waters | Model Deep Aquifer (sandstone aquifer) | 178 | 3.80 | 0 | 0 | 7.5 | 415 |
| Model Shallow Aquifer (dolomite aquifer) | 226 | 70.4 | 2.00 | 0 | 7.5 | 775 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lynn, W.; Heffron, J.; Mayer, B.K. Electrocoagulation as a Pretreatment for Electroxidation of E. coli. Water 2019, 11, 2509. https://doi.org/10.3390/w11122509
Lynn W, Heffron J, Mayer BK. Electrocoagulation as a Pretreatment for Electroxidation of E. coli. Water. 2019; 11(12):2509. https://doi.org/10.3390/w11122509
Chicago/Turabian StyleLynn, William, Joe Heffron, and Brooke K. Mayer. 2019. "Electrocoagulation as a Pretreatment for Electroxidation of E. coli" Water 11, no. 12: 2509. https://doi.org/10.3390/w11122509
APA StyleLynn, W., Heffron, J., & Mayer, B. K. (2019). Electrocoagulation as a Pretreatment for Electroxidation of E. coli. Water, 11(12), 2509. https://doi.org/10.3390/w11122509






