Evaluation of the Efficiency of Arundo donax L. Leaves as Biomonitors for Atmospheric Element Concentrations in an Urban and Industrial Area of Central Italy
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Sampling Sites
2.2. Arundo donax Leaves and River Water Samples Collection and Preparation
2.3. PM10 Samples Collection and Preparation
2.4. ICP-MS Analysis
2.5. Data Elaboration
2.6. Statistical Analysis
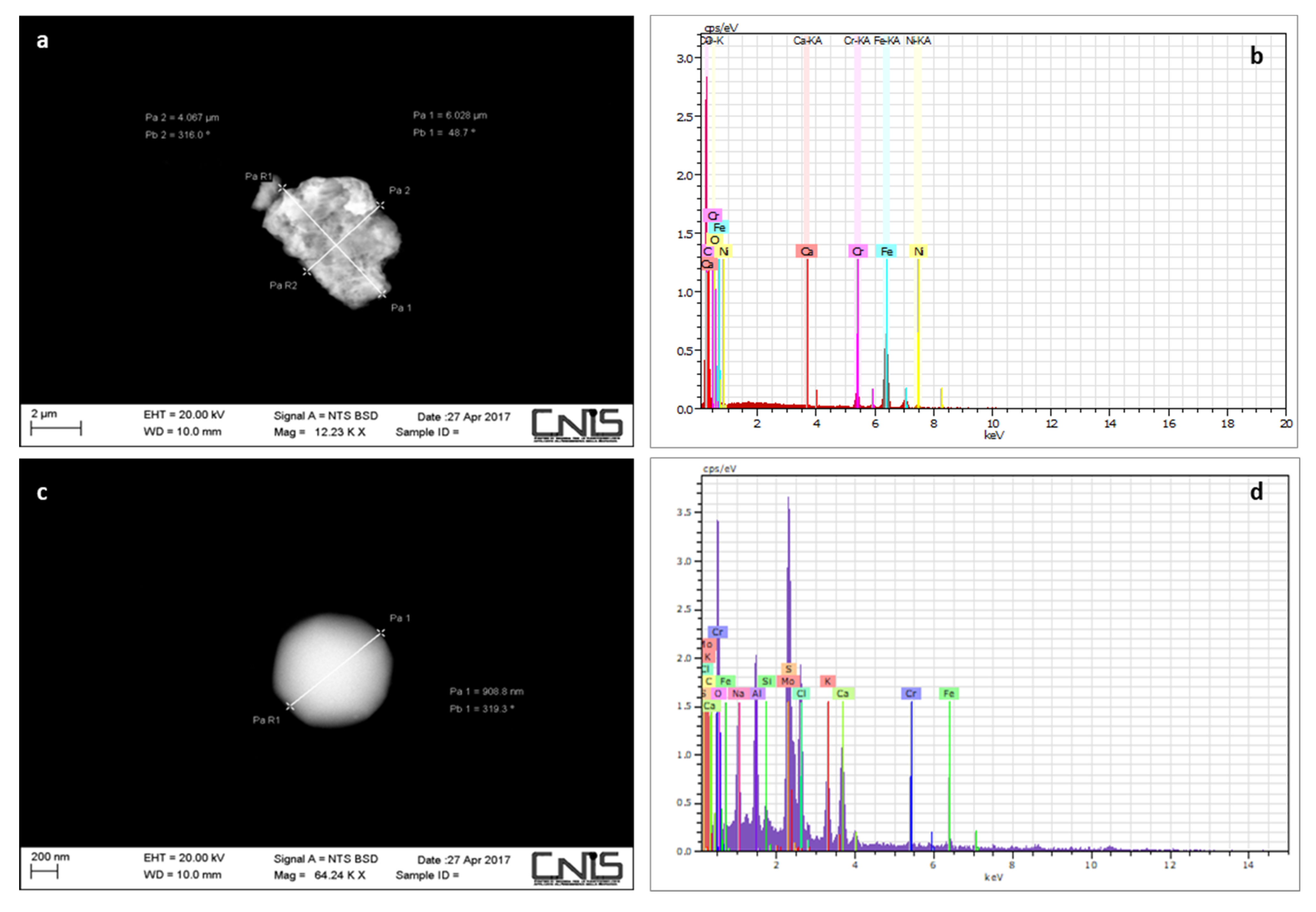
2.7. Scanning Electron Microscopy (SEM) Analysis of PM10 Samples
3. Results and Discussion
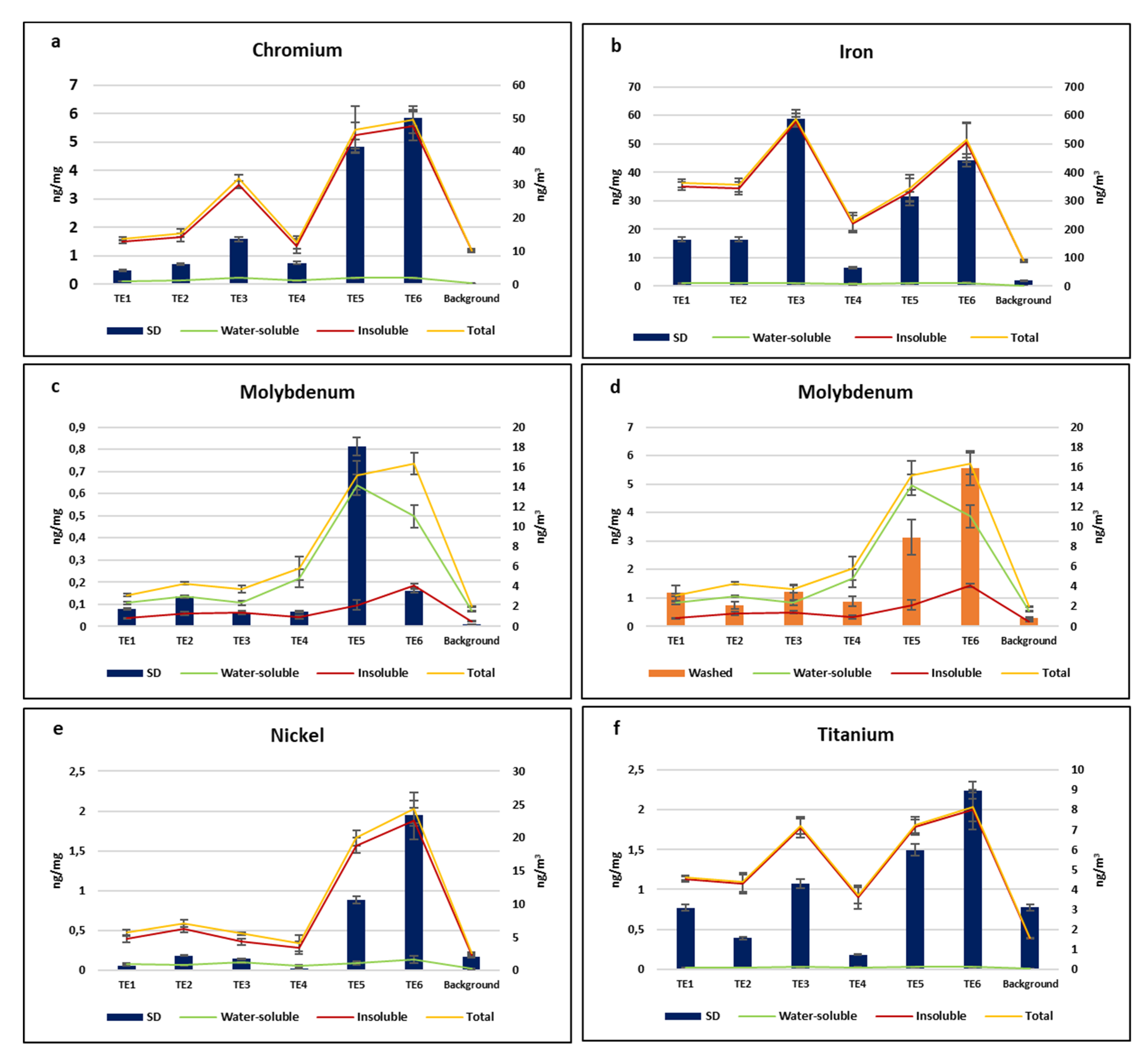
3.1. Element Concentrations in Arundo donax Leaves
3.2. Comparison between Leaf Deposition and PM10 Element Concentrations
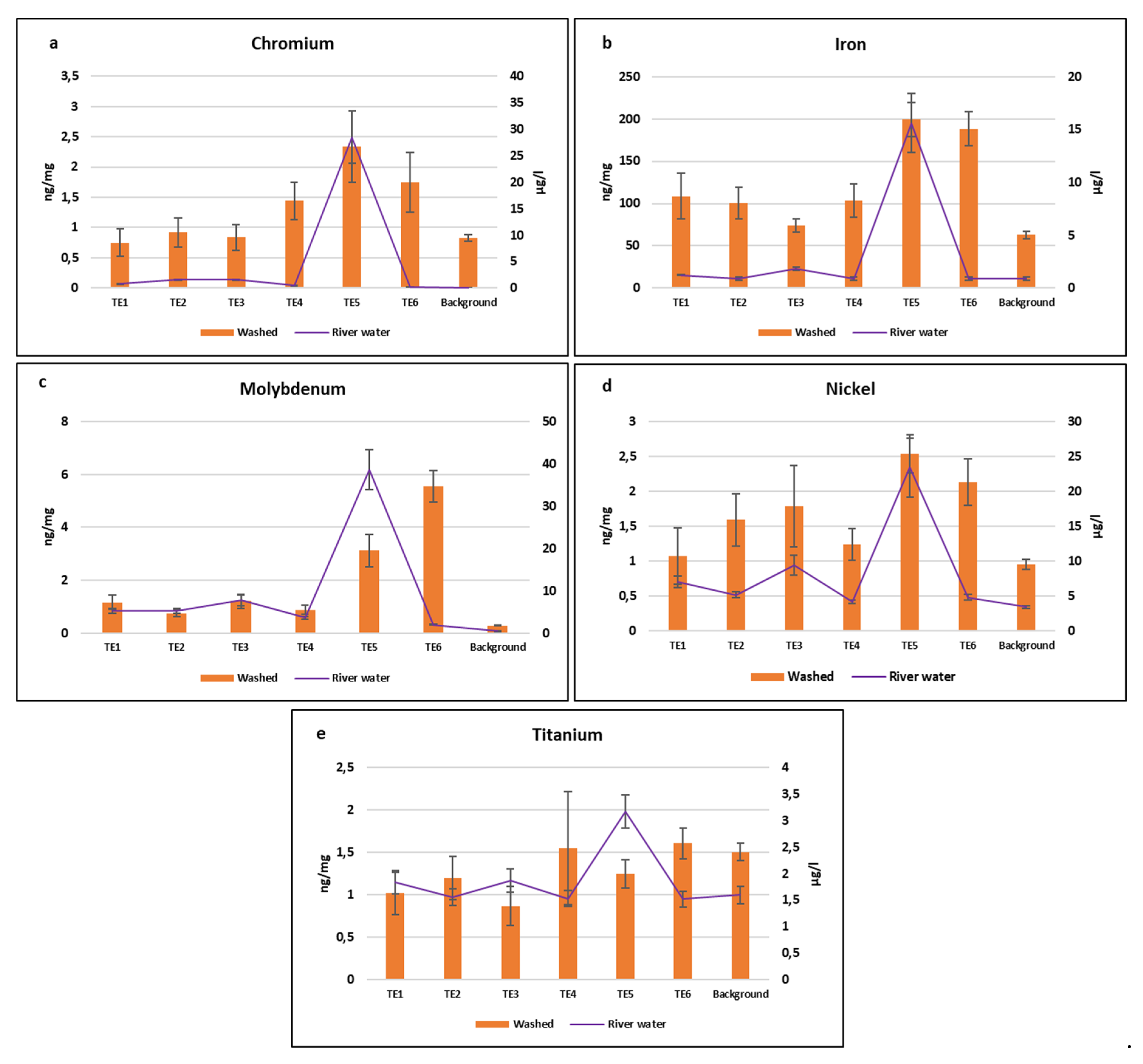
3.3. Comparison between River Water Samples and A. donax Leaves Elemental Content
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Perdue, R.E. Arundo donax—Source of musical reeds and industrial cellulose. Econ. Bot. 1958, 12, 368. [Google Scholar] [CrossRef]
- Bell, G.P. Ecology and management of Arundo donax, and approaches to riparian habitat restoration in Southern California. In Plant Invasions: Studies from North America and Europe; Backhuys Publishers: Leiden, The Netherlands, 1997; pp. 103–113. [Google Scholar]
- Papazoglou, E.G.; Karantounias, G.A.; Vemmos, S.N.; Bouranis, D.L. Photosynthesis and growth responses of giant reed (Arundo donax L.) to the heavy metals Cd and Ni. Environ. Int. 2005, 31, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Nsanganwimana, F.; Marchand, L.; Douay, F.; Mench, M. Arundo donax L., a candidate for phytomanaging water and soils contaminated by trace elements and producing plant-based feedstock. A review. Int. J. Phytoremediation 2014, 16, 982–1017. [Google Scholar] [CrossRef] [PubMed]
- Bonanno, G. Comparative performance of trace element bioaccumulation and biomonitoring in the plant species Typha domingensis, Phragmites australis and Arundo donax. Ecotoxicol. Environ. Saf. 2013, 97, 124–130. [Google Scholar] [CrossRef]
- Bonanno, G.; Borg, J.A.; Di Martino, V. Levels of heavy metals in wetland and marine vascular plants and their biomonitoring potential: A comparative assessment. Sci. Total Environ. 2017, 576, 796–806. [Google Scholar] [CrossRef]
- Fiorentino, N.; Ventorino, V.; Rocco, C.; Cenvinzo, V.; Agrelli, D.; Gioia, L.; Di Mola, L.; Adamo, P.; Pepe, O.; Fagnano, M. Giant reed growth and effects on soil biological fertility in assisted phytoremediation of an industrial polluted soil. Sci. Total Environ. 2017, 575, 1375–1383. [Google Scholar] [CrossRef]
- Bonanno, G. Arundo donax as a potential biomonitor of trace element contamination in water and sediment. Ecotoxicol. Environ. Saf. 2012, 80, 20–27. [Google Scholar] [CrossRef]
- Szczepaniak, K.; Biziuk, M. Review: Ecotoxicology, Aspects of the biomonitoring studies using mosses and lichens as indicators of metal pollution. Environ. Res. 2003, 93, 221–230. [Google Scholar] [CrossRef]
- Abril, G.A.; Wannaz, E.D.; Mateos, A.C.; Pignata, M.L. Biomonitoring of airborne particulate matter emitted from a cement plant and comparison with dispersion modelling results. Atmos. Environ. 2014, 8, 154–163. [Google Scholar] [CrossRef]
- Massimi, L.; Conti, M.E.; Mele, G.; Ristorini, M.; Astolfi, M.L.; Canepari, S. Lichen Transplants as Indicators of Atmospheric Element Concentrations: A High Spatial Resolution Comparison with PM10 Samples in a Polluted Area (Central Italy). Ecol. Indic. 2019, 101, 759–769. [Google Scholar] [CrossRef]
- Urbat, M.; Lehndorff, E.; Schwark, L. Biomonitoring of air quality in the Cologne conurbation using pine needles as a passive sampler—Part I: Magnetic properties. Atmos. Environ. 2004, 38, 3781–3792. [Google Scholar] [CrossRef]
- Lin, V.S. Research highlights: Natural passive samplers–plants as biomonitors. Environ. Sci. Process. Impacts 2015, 17, 1137–1140. [Google Scholar] [CrossRef] [PubMed]
- Sgrigna, G.; Baldacchini, C.; Esposito, R.; Calandrelli, R.; Tiwary, A.; Calfapietra, C. Characterization of leaf-level particulate matter for an industrial city using electron microscopy and X-ray microanalysis. Sci. Total Environ. 2016, 548–549, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Janhall, S. Review on urban vegetation and particle air pollution e Deposition and dispersion. Atmos. Environ. 2015, 105, 130–137. [Google Scholar] [CrossRef]
- Gratani, L.; Crescente, M.F.; Varone, L. Long-term monitoring of metal pollution by urban trees. Atmos. Environ. 2008, 42, 8273–8277. [Google Scholar] [CrossRef]
- Baldacchini, C.; Sgrigna, G.; Clarke, W.; Tallis, M.; Calfapietra, C. An ultra-spatially resolved method to quali-quantitative monitor particulate matter in urban environment. Environ. Sci. Pollut. Res. Int. 2019, 26, 18719–18729. [Google Scholar] [CrossRef]
- Thorpe, A.; Harrison, R.M. Sources and properties of non-exhaust particulate matter from road traffic: A review. Sci. Total Environ. 2008, 400, 270–282. [Google Scholar] [CrossRef]
- Slezakova, K.; Pires, J.C.M.; Pereira, M.C.; Martins, F.G.; Alvim-Ferraz, M.C. Influence of traffic emissions on the composition of atmospheric particles of different sizes—Part 2: SEM–EDS characterization. J. Atmos. Chem. 2008, 60, 221–236. [Google Scholar] [CrossRef]
- Pope, C.A., III; Burnett, R.T.; Thun, M.J.; Calle, E.E.; Krewski, D.; Ito, K.; Thurston, G.D. Lung cancer, cardiopulmonary mortality, and long-term exposure to fine particulate air pollution. Jama 2002, 287, 1132–1141. [Google Scholar] [CrossRef]
- Ebelt, S.T.; Petkau, A.J.; Vedal, S.; Fisher, T.V.; Brauer, M. Exposure of chronic obstructive pulmonary disease patients to particulate matter: Relationships between personal and ambient air concentrations. J. Air Waste Manag. Assoc. 2000, 50, 1081–1094. [Google Scholar] [CrossRef]
- Chen, L.C.; Lippmann, M. Effects of Metals within Ambient Air Particulate Matter (PM) on Human Health. Inhal. Toxicol. 2009, 21, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.O.; Thundiyil, J.G.; Stolbach, A. Clearing the Air: A Review of the Effects of Particulate Matter Air Pollution on Human Health. J. Med. Toxicol. 2012, 8, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Pope, C.A., III; Dockery, D.W. Acute Health Effects of PM10 Pollution on Symptomatic and Asymptomatic Children. AJRCCM Issues 1992, 145. [Google Scholar] [CrossRef]
- Donaldson, K.; MacNee, W. Potential mechanisms of adverse pulmonary and cardiovascular effects of particulate air pollution (PM10). Int. J. Hyg. Environ. Health 2010, 203, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Sæbø, A.; Popek, R.; Nawrot, B.; Hanslin, H.M.; Gawronska, H.; Gawronski, S.W. Plant species differences in particulate matter accumulation on leaf surfaces. Sci. Total Environ. 2012, 427–428, 347–354. [Google Scholar] [CrossRef]
- Petroff, A.; Mailliat, A.; Amielh, M.; Anselmet, F. Aerosol dry deposition on vegetative canopies. Part I: Review of present knowledge. Atmos. Environ. 2008, 42, 3625–3653. [Google Scholar] [CrossRef]
- Terzaghi, E.; Wild, E.; Zacchello, G.; Cerabolini, B.E.L.; Jones, K.C.; Di Guardo, A. Forest Filter Effect: Role of leaves in capturing/releasing air particulate matter and its associated PAHs. Atmos. Environ. 2013, 74, 378–384. [Google Scholar] [CrossRef]
- Hofman, J.; Wuyts, K.; Van Wittenberghe, S.; Brackx, M.; Samson, R. On the link between biomagnetic monitoring and leaf-deposited dust load of urban trees: Relationships and spatial variability of different particle size fractions. Environ. Poll. 2014, 189, 63–72. [Google Scholar] [CrossRef]
- Sgrigna, G.; Sæbø, A.; Gawronski, S.; Popek, R.; Calfapietra, C. Particulate matter deposition on Quercus ilex leaves in an industrial city of central Italy. Environ. Pollut. 2015, 197, 187–194. [Google Scholar] [CrossRef]
- Massimi, L.; Ristorini, M.; Eusebio, M.; Florendo, D.; Adeyemo, A.; Brugnoli, D.; Canepari, S. Monitoring and evaluation of Terni (Central Italy) air quality through spatially resolved analyses. Atmosphere 2017, 8, 200. [Google Scholar] [CrossRef]
- Moroni, B.; Ferrero, L.; Crocchianti, S.; Perrone, M.G.; Sangiorgi, G.; Bolzacchini, E.; Cappelletti, D. Aerosol dynamics upon Terni basin (Central Italy): Results of integrated vertical profile measurements and electron microscopy analyses. Rend. Lincei 2013, 24, 319–328. [Google Scholar] [CrossRef]
- Manigrasso, M.; Protano, C.; Astolfi, M.L.; Massimi, L.; Avino, P.; Vitali, M.; Canepari, S. Evidences of copper nanoparticle exposure in indoor environments: Long-term assessment, high-resolution field emission scanning electron microscopy evaluation, in silico respiratory dosimetry study and possible health implications. Sci. Total Environ. 2017, 653, 1192–1203. [Google Scholar] [CrossRef] [PubMed]
- Zona., A.; Pasetto, R.; Fazzo, L.; Iavarone, I.; Bruno, C.; Pirastu, R.; Comba, P. Fifth report S.E.N.T.I.E.R.I 2019, Epidemiologia & Prevenzione, Rivista dell’Associazione Italiana di Epidemiologia 2-3, 43, Supplement 1. Available online: http://www.epiprev.it/materiali/2019/EP2-3_Suppl1/SENTIERI_FullText.pdf (accessed on 7 January 2020).
- Arpa Umbria: Agenzia Regionale per la Protezione Ambientale dell’ Umbria. Available online: https://www.arpa.umbria.it/monitoraggi/aria/Default.aspx (accessed on 7 January 2020).
- APHA. Standard Methods for Examination of Water and Waste Water, 20th ed.; American Public Health Association: Washington, DC, USA, 1998. [Google Scholar]
- Canepari, S.; Cardarelli, E.; Giuliano, A.; Pietrodangelo, A. Determination of metals, metalloids and non-volatile ions in airborne particulate matter by a new two-step sequential leaching procedure Part A: Experimental design and optimization. Talanta 2006, 69, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Canepari, S.; Cardarelli, E.; Pietrodangelo, A.; Strincone, M. Determination of metals, metalloids and non-volatile ions in airborne particulate matter by a new two-step sequential leaching procedure Part B: Validation on equivalent real samples. Talanta 2006, 69, 588–595. [Google Scholar] [CrossRef]
- Canepari, S.; Pietrodangelo, A.; Perrino, C.; Astolfi, M.L.; Marzo, M.L. Enhancement of source traceability of atmospheric PM by elemental chemical fractionation. Atmos. Environ. 2009, 43, 4754–4765. [Google Scholar] [CrossRef]
- Piacentini, D.; Falasca, G.; Canepari, S.; Massimi, L. Potential of PM-selected components to induce oxidative stress and root system alteration in a plant model organism. Environ. Int. 2019, 132, 105094. [Google Scholar] [CrossRef]
- Astolfi, M.L.; Marconi, E.; Protano, C.; Vitali, M.; Schiavi, E.; Mastromarino, P.; Canepari, S. Optimization and validation of a fast digestion method for the determination of major and trace elements in breast milk by ICP-MS. Anal. Chim. Acta 2018, 1040, 49–62. [Google Scholar] [CrossRef]
- Cressie, N. Spatial Prediction and Ordinary Kriging. Math. Geol. 1988, 20, 405–421. [Google Scholar] [CrossRef]
- Cressie, N. The origins of kriging. Math. Geol. 1990, 22, 239–252. [Google Scholar] [CrossRef]
- Blair, M.; Stevens, T.L. Steel Castings Handbook, 6th ed.; Steel Founders’ Society and ASM International: Novelty, OH, USA, 1995; pp. 2–34. [Google Scholar]
- Adachi, K.; Tainosho, Y. Characterization of heavy metal particles embedded in tire dust. Environ. Int. 2004, 30, 1009–1017. [Google Scholar] [CrossRef]
- Birmili, W.; Allen, A.G.; Bary, F.; Harrison, R.M. Trace Metal Concentrations and Water Solubility in Size-Fractionated Atmospheric Particles and Influence of Road Traffic. Environ. Sci. Technol. 2006, 40, 1144–1153. [Google Scholar] [CrossRef] [PubMed]
- Litschke, T.; Kuttler, W. On the reduction of urban particle concentration by vegetation–A review. Meteorol. Z. 2008, 17, 229–240. [Google Scholar] [CrossRef]
- Morakinyo, T.E.; Lam, Y.F. Simulation study of dispersion and removal of particulate matter from traffic by road-side vegetation barrier. Environ. Sci. Pollut. Res. 2016, 23, 6709–6722. [Google Scholar] [CrossRef] [PubMed]
- Umbrìa, A.; Galan, M.; Munoz, M.J.; Martìn, R. Characterization of atmospheric particles: Analysis of particles in the Campo de Gibraltar. Atmosfera 2004, 17, 191–206. [Google Scholar]
- Bouhsina, S.; Cazier, F.; Noual, H.; Dewaele, D.; Delbende, A.; Courcot, D.; Aboukais, A. Characteristics of suspended particulate matter emitted from an iron and steel company–A multi technique approach for search of tracers. Chem. Eng. Trans. 2008, 16, 79. [Google Scholar]
- Tunno, B.J.; Dalton, R.; Michanowicz, D.R.; Shmool, J.L.C.; Kinnee, E.; Tripathy, S.; Cambal, L.; Clougherty, J.E. Spatial patterning in PM2.5 constituents under an inversion-focused sampling design across an urban area of complex terrain. J. Expo. Sci. Environ. Epidemiol. 2016, 26, 385–396. [Google Scholar] [CrossRef]
- Thurston, G.D.; Ito, K.; Lall, R. A source apportionment of U.S. fine particulate matter air pollution. Atmos. Environ. 2011, 45, 3924–3936. [Google Scholar] [CrossRef]
- Song, F.; Gao, Y. Size distributions of trace elements associated with ambient particular matter in the affinity of a major highway in the New Jersey–New York metropolitan area. Atmos. Environ. 2011, 45, 6714–6723. [Google Scholar] [CrossRef]
- Canepari, S.; Perrino, C.; Olivieri, F.; Astolfi, M.L. Characterisation of the traffic sources of PM through size-segregated sampling, sequential leaching and ICP analysis. Atmos. Environ. 2008, 42, 8161–8175. [Google Scholar] [CrossRef]
- Shakoor, S.A.; Soodan, A.M.; Kumar, K. Morphological Diversity and Frequency of Phytolith Types in Gaint Reed Arundo donax (L.). World Appl. Sci. J. 2014, 29, 926–932. [Google Scholar] [CrossRef]
- Simonetti, G.; Buiarelli, F.; Di Filippo, P.; Pomata, D.; Riccardi, C.; Ristorini, M.; Astolfi, M.L.; Canepari, S. Spatial Distribution of Levoglucosan and Alternative Biomass Burning Tracers in an Urban and Industrial Hot-spot of Central Italy. Atmos. Res. 2020, 239, 104904. [Google Scholar]
- Minguillón, M.C.; Querol, X.; Baltensperger, U.; Prévôt, A.S.H. Fine and coarse PM composition and sources in rural and urban sites in Switzerland: Local or regional pollution? Sci. Total Environ. 2012, 427–428, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Gietl, J.K.; Lawrence, L.; Thorpe, A.J.; Harrison, R.M. Identification of brake wear particles and derivation of a quantitative tracer for brake dust at a major road. Atmos. Environ. 2010, 44, 141–146. [Google Scholar] [CrossRef]
- Dongarrà, G.; Manno, E.; Varrica, D. Possible markers of traffic-related emissions. Environ. Monit. Assess. 2008, 154, 117. [Google Scholar] [CrossRef] [PubMed]
- Mirza, N.; Pervez, A.; Mahmood, Q.; Shah, M.M.; Shafqat, M.N. Ecological restoration of arsenic contaminated soil by Arundo donax L. Ecol. Eng. 2011, 37, 1949–1956. [Google Scholar] [CrossRef]
- Barbosa, B.; Boléo, S.; Sidella, S.; Costa, J.; Duarte, M.; Mendes, B.; Cosentino, S.L.; Fernando, A.L. Phytoremediation of Heavy Metal-Contaminated Soils Using the Perennial Energy Crops Miscanthus spp. and Arundo donax L. Bioenerg. Res. 2015, 8, 1500–1511. [Google Scholar] [CrossRef]
- Carranza-Álvarez, C.; Alonso-Castro, A.J.; Alfaro-De La Torre, M.C.; García-De La Cruz, R.F. Accumulation and Distribution of Heavy Metals in Scirpus americanus and Typha latifolia from an Artificial Lagoon in San Luis Potosí, México. Water Air Soil Pollut. 2007, 188, 297–309. [Google Scholar] [CrossRef]
- Kabata-Pendias, A.; Mukherjee, A.B. Trace Elements from Soil to Human; Springer: Cham, Switzerland, 2007; ISBN 978-3-540-32714-1. [Google Scholar]
- Capelli, L.; Sironi, S.; Del Rosso, R.; Céntola, P.; Rossi, A.; Austeri, C. Olfactometric approach for the evaluation of citizens’ exposure to industrial emissions in the city of Terni, Italy. Sci. Total Environ. 2011, 409, 595–603. [Google Scholar] [CrossRef]
- Sabeen, M.; Mahmood, Q.; Irshad, M.; Fareed, I.; Khan, A.; Ullah, F.; Hussain, J.; Hayat, Y.; Tabassum, S. Cadmium Phytoremediation by Arundo donax L. from Contaminated Soil and Water. BioMed. Res. Int. 2013. [Google Scholar] [CrossRef]
- Atma, W.; Larouci, M.; Meddah, B.; Benabdeli, K.; Sonnet, P. Evaluation of the phytoremediation potential of Arundo donax L. for nickel-contaminated soil. Int. J. Phytoremediat. 2017, 19, 377–386. [Google Scholar] [CrossRef]
- Fiorentino, N.; Fagnano, M.; Adamo, P.; Impagliazzo, A.; Mori, M.; Pepe, O.; Ventorino, V.; Zoina, A. Assisted phytoextraction of heavy metals: Compost and Trichoderma effects on giant reed (Arundo donax L.) uptake and soil N-cycle microflora. Italian J. Agron. 2013, 8, 244–254. [Google Scholar] [CrossRef]
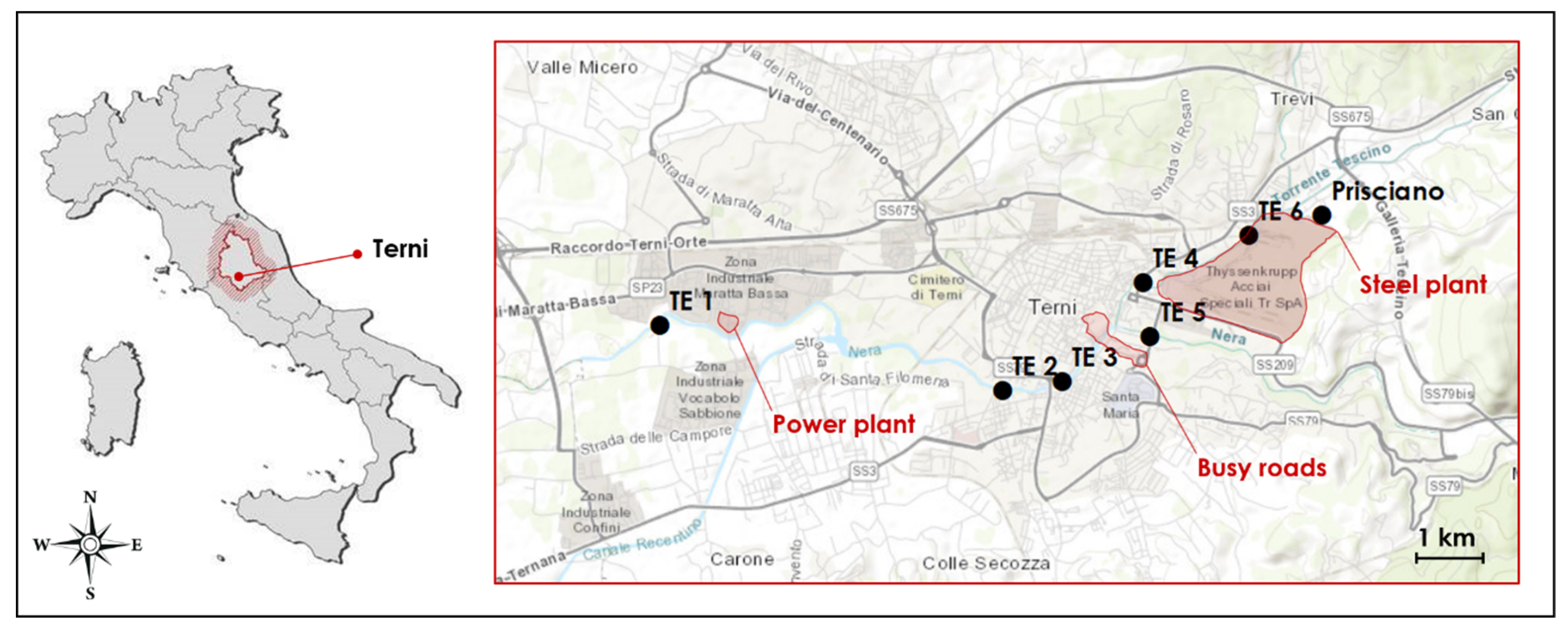

| SAMPLING SITES | LATITUDE | LONGITUDE |
|---|---|---|
| TE1 | 42°33′43.84″ N | 12°35′47.18″ E |
| TE2 | 42°33′22.27″ N | 12°38′21.02″ E |
| TE3 | 42°33′25.29″ N | 12°38′47.81″ E |
| TE4 | 42°33′58.02″ N | 12°39′24.08″ E |
| TE5 | 42°33′40.12″ N | 12°39′27.33″ E |
| TE6 | 42°34′13.53″ N | 12°40′11.41″ E |
| Pearson’s | Ba | Cd | Cr | Cs | Cu | Fe | Li | Mn | Mo | Ni | Pb | Rb | Sb | Sn | Sr | Ti |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SD/Water soluble PM10 | −0.06 | 0.31 | 0.74 | 0.31 | −0.39 | 0.49 | 0.62 | 0.77 | 0.78 | 0.77 | 0.21 | 0.00 | −0.65 | 0.34 | −0.56 | 0.79 |
| SD/Insoluble PM10 | −0.12 | 0.22 | 0.87 | 0.38 | −0.08 | 0.93 | 0.25 | 0.92 | 0.16 | 0.95 | −0.79 | 0.12 | −0.74 | 0.53 | −0.62 | 0.93 |
| Washed leaves/Water soluble PM10 | 0.01 | 0.18 | 0.49 | 0.10 | 0.08 | 0.31 | 0.67 | 0.73 | 0.90 | 0.63 | 0.02 | 0.51 | 0.45 | −0.43 | 0.19 | 0.17 |
| Washed leaves/Insoluble PM10 | −0.59 | 0.24 | 0.59 | −0.14 | 0.18 | 0.02 | −0.03 | 0.75 | 0.35 | 0.82 | 0.55 | −0.08 | 0.43 | −0.66 | 0.21 | 0.01 |
| Pearson’s | Ba | Cd | Cr | Cs | Cu | Fe | Li | Mn | Mo | Ni | Pb | Rb | Sb | Sn | Sr | Ti |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Washed leaves/ River Water | 0.38 | 0.81 | 0.76 | 0.18 | 0.34 | 0.53 | −0.20 | −0.55 | 0.17 | 0.71 | −0.10 | −0.35 | 0.06 | −0.20 | −0.44 | −0.23 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ristorini, M.; Astolfi, M.L.; Frezzini, M.A.; Canepari, S.; Massimi, L. Evaluation of the Efficiency of Arundo donax L. Leaves as Biomonitors for Atmospheric Element Concentrations in an Urban and Industrial Area of Central Italy. Atmosphere 2020, 11, 226. https://doi.org/10.3390/atmos11030226
Ristorini M, Astolfi ML, Frezzini MA, Canepari S, Massimi L. Evaluation of the Efficiency of Arundo donax L. Leaves as Biomonitors for Atmospheric Element Concentrations in an Urban and Industrial Area of Central Italy. Atmosphere. 2020; 11(3):226. https://doi.org/10.3390/atmos11030226
Chicago/Turabian StyleRistorini, Martina, Maria Luisa Astolfi, Maria Agostina Frezzini, Silvia Canepari, and Lorenzo Massimi. 2020. "Evaluation of the Efficiency of Arundo donax L. Leaves as Biomonitors for Atmospheric Element Concentrations in an Urban and Industrial Area of Central Italy" Atmosphere 11, no. 3: 226. https://doi.org/10.3390/atmos11030226
APA StyleRistorini, M., Astolfi, M. L., Frezzini, M. A., Canepari, S., & Massimi, L. (2020). Evaluation of the Efficiency of Arundo donax L. Leaves as Biomonitors for Atmospheric Element Concentrations in an Urban and Industrial Area of Central Italy. Atmosphere, 11(3), 226. https://doi.org/10.3390/atmos11030226







