Evidence of Ozone-Induced Visible Foliar Injury in Hong Kong Using Phaseolus Vulgaris as a Bioindicator
Abstract
1. Introduction
2. Experiments
2.1. Plant Materials and Gardening
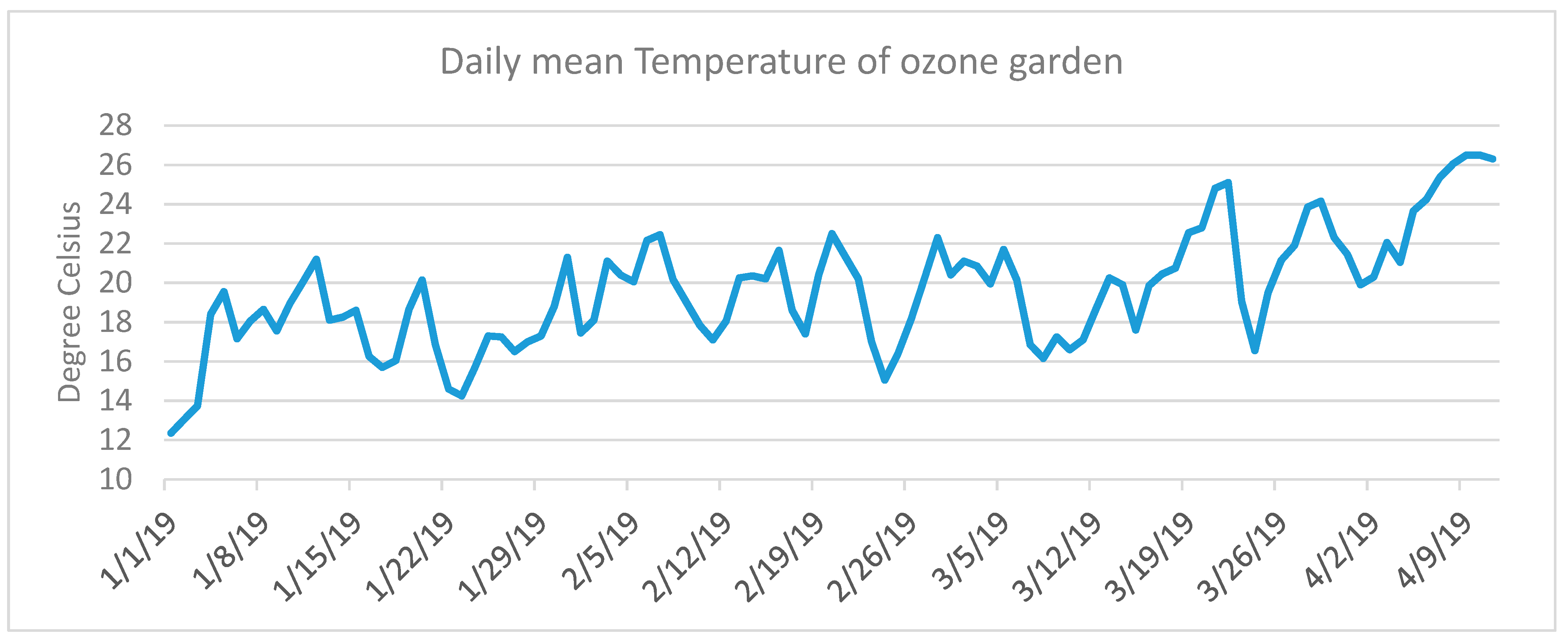
2.2. Ozone Monitoring and Meteorological Measurements
2.3. Insect Injury Monitoring
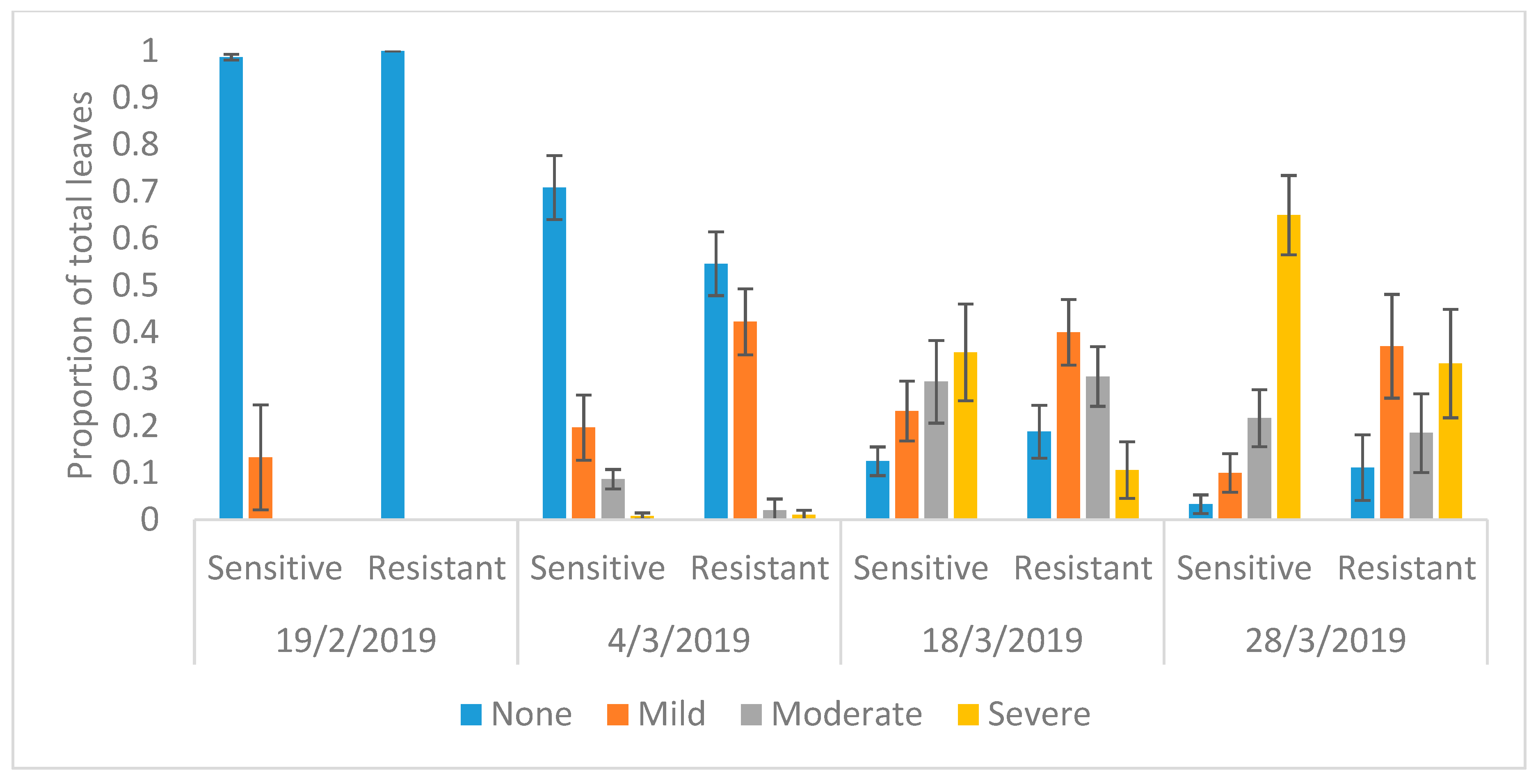
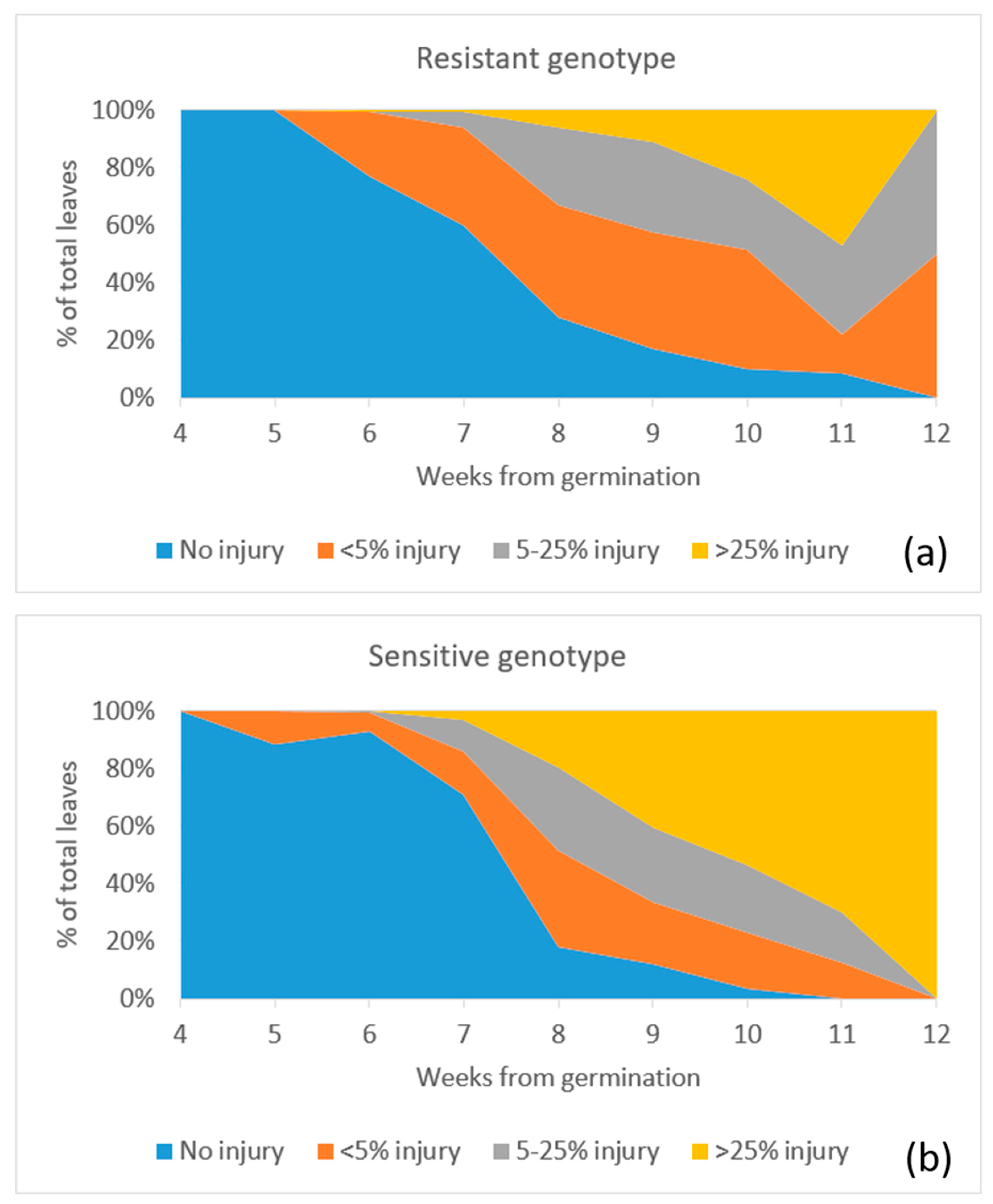
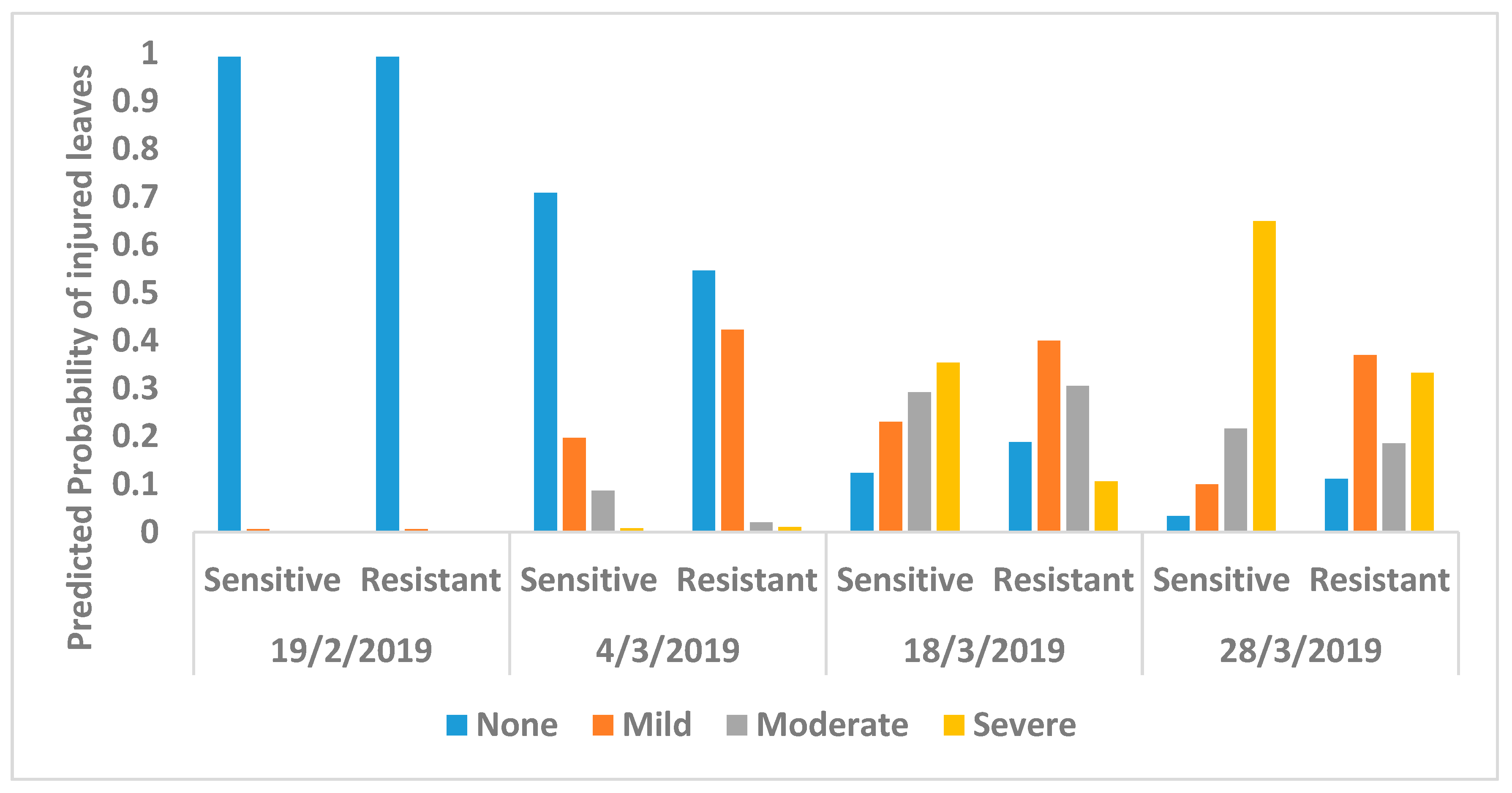
2.4. Ozone Injury Monitoring
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Month | Average Relative Humidity % | Total Rainfall (mm) |
|---|---|---|
| January | 76 | 4.7 |
| February | 82.5 | 68.7 |
| March | 85 | 186.5 |
| April | 80.5 | 185.8 |

| Dates | Multinom (Injury~Genotype) | Multinom (Injury~1) |
|---|---|---|
| 19 February 2019 | 22.62 | 18.04 |
| 4 March 2019 | 378.12 | 388.55 |
| 18 March 2019 | 528.96 | 542.21 |
| 28 March 2019 | 196.29 | 202.56 |
| Date | Genotype | None (0%) | Mild (<5%) | Moderate (5% to 25%) | Severe (>25%) |
|---|---|---|---|---|---|
| 19/02/2019 | Sensitive | 74 | 1 | 0 | 0 |
| Resistant | 77 | 0 | 0 | 0 | |
| 4/03/2019 | Sensitive | 90 | 25 | 11 | 1 |
| Resistant | 53 | 41 | 2 | 1 | |
| t-value | −8.24 | 13.14 | −4.55 | −0.39 | |
| p-value | <0.01 | <0.01 | <0.01 | 0.7 | |
| 18/03/2019 | Sensitive | 14 | 26 | 33 | 40 |
| Resistant | 16 | 34 | 26 | 9 | |
| t-value | 4.58 | 8.39 | 1.34 | −20.14 | |
| p-value | <0.01 | <0.01 | 0.21 | <0.01 | |
| 28/03/2019 | Sensitive | 2 | 6 | 13 | 39 |
| Resistant | 3 | 10 | 5 | 9 | |
| t-value | 5.5 | 11.5 | −2.48 | −6.97 | |
| p-value | <0.01 | <0.01 | 0.03 | <0.01 |
References
- Tai, A.P.K.; Martin, M.V.; Heald, C.L. Threat to future global food security from climate change and ozone air pollution. Nat. Clim. Chang. 2014, 4, 817–821. [Google Scholar] [CrossRef]
- Tai, A.P.K.; Val Martin, M. Impacts of ozone air pollution and temperature extremes on crop yields: Spatial variability, adaptation and implications for future food security. Atmos. Environ. 2017, 169, 11–21. [Google Scholar] [CrossRef]
- Ainsworth, E.A.; Yendrek, C.R.; Sitch, S.; Collins, W.J.; Emberson, L.D. The Effects of Tropospheric Ozone on Net Primary Productivity and Implications for Climate Change. Annu. Rev. Plant Biol. 2012, 63, 637–661. [Google Scholar] [CrossRef] [PubMed]
- Huntingford, C.; Cox, P.M.; Mercado, L.M.; Sitch, S.; Bellouin, N.; Boucher, O.; Gedney, N. Highly contrasting effects of different climate forcing agents on terrestrial ecosystem services. Philos. Trans. A. Math. Phys. Eng. Sci. 2011, 369, 2026–2037. [Google Scholar] [CrossRef] [PubMed]
- Sadiq, M.; Tai, A.P.K.; Lombardozzi, D.; Val Martin, M. Effects of ozone–vegetation coupling on surface ozone air quality via biogeochemical and meteorological feedbacks. Atmos. Chem. Phys. 2017, 17, 3055–3066. [Google Scholar] [CrossRef]
- Zhao, C.; Wang, Y.; Zeng, T. East China plains: A “basin” of ozone pollution. Environ. Sci. Technol. 2009, 43, 1911–1915. [Google Scholar] [CrossRef]
- Lu, X.; Hong, J.; Zhang, L.; Cooper, O.R.; Schultz, M.G.; Xu, X.; Wang, T.; Gao, M.; Zhao, Y.; Zhang, Y. Severe Surface Ozone Pollution in China: A Global Perspective. Environ. Sci. Technol. Lett. 2018, 5, 487–494. [Google Scholar] [CrossRef]
- Ng, E.; Chen, L.; Wang, Y.; Yuan, C. A study on the cooling effects of greening in a high-density city: An experience from Hong Kong. Build. Environ. 2012, 47, 256–271. [Google Scholar] [CrossRef]
- The Royal Society Ground-level ozone in the 21st century: Future trends, impacts and policy implications. Sci. Policy 2008, 15, 1–148.
- Conklin, P.L.; Barth, C. Ascorbic acid, a familiar small molecule intertwined in the response of plants to ozone, pathogens, and the onset of senescence. Plant. Cell Environ. 2004, 27, 959–970. [Google Scholar] [CrossRef]
- Feng, Z.; Pang, J.; Nouchi, I.; Kobayashi, K.; Yamakawa, T.; Zhu, J. Apoplastic ascorbate contributes to the differential ozone sensitivity in two varieties of winter wheat under fully open-air field conditions. Environ. Pollut. 2010, 158, 3539–3545. [Google Scholar] [CrossRef] [PubMed]
- Turcsányi, E.; Lyons, T.; Plöchl, M.; Barnes, J. Does ascorbate in the mesophyll cell walls form the first line of defence against ozone? Testing the concept using broad bean (Vicia faba L.). J. Exp. Bot. 2000, 51, 901–910. [Google Scholar] [CrossRef] [PubMed]
- Ashmore, M.R. Assessing the future global impacts of ozone on vegetation. Plant, Cell Environ. 2005, 28, 949–964. [Google Scholar] [CrossRef]
- Emberson, L.D.; Büker, P.; Ashmore, M.R. Assessing the risk caused by ground level ozone to European forest trees: A case study in pine, beech and oak across different climate regions. Environ. Pollut. 2007, 147, 454–466. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Mane, S.P.; Sioson, A.A.; Robinet, C.V.; Heath, L.S.; Bohnert, H.J.; Grene, R. Effects of chronic ozone exposure on gene expression in Arabidopsis thaliana ecotypes and in Thellungiella halophila. Plant Cell Environ. 2006, 29, 854–868. [Google Scholar] [CrossRef] [PubMed]
- Wrzaczek, M.; Brosché, M.; Salojärvi, J.; Kangasjärvi, S.; Idänheimo, N.; Mersmann, S.; Robatzek, S.; Karpiński, S.; Karpińska, B.; Kangasjärvi, J. Transcriptional regulation of the CRK/DUF26 group of receptor-like protein kinases by ozone and plant hormones in Arabidopsis. BMC Plant Biol. 2010, 10, 95. [Google Scholar] [CrossRef]
- Vahisalu, T.; Puzõrjova, I.; Brosché, M.; Valk, E.; Lepiku, M.; Moldau, H.; Pechter, P.; Wang, Y.-S.; Lindgren, O.; Salojärvi, J.; et al. Ozone-triggered rapid stomatal response involves the production of reactive oxygen species, and is controlled by SLAC1 and OST1. Plant J. 2010, 62, 442–453. [Google Scholar] [CrossRef]
- Wilkinson, S.; Davies, W.J. Drought, ozone, ABA and ethylene: New insights from cell to plant to community. Plant. Cell Environ. 2010, 33, 510–525. [Google Scholar] [CrossRef]
- Lindroth, R.L. Impacts of elevated atmospheric CO2 and O3 on forests: Phytochemistry, trophic interactions, and ecosystem dynamics. J. Chem. Ecol. 2010, 36, 2–21. [Google Scholar] [CrossRef]
- Hayes, F.; Sharps, K.; Harmens, H.; Roberts, I.; Mills, G. Tropospheric ozone pollution reduces the yield of African crops. J. Agron. Crop Sci. 2019, 12376. [Google Scholar] [CrossRef]
- Piraino, F.; Aina, R.; Palin, L.; Prato, N.; Sgorbati, S.; Santagostino, A.; Citterio, S. Air quality biomonitoring: Assessment of air pollution genotoxicity in the Province of Novara (North Italy) by using Trifolium repens L. and molecular markers. Sci. Total Environ. 2006, 372, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Klumpp, A.; Ansel, W.; Klumpp, G.; Belluzzo, N.; Calatayud, V.; Chaplin, N.; Garrec, J.P.; Gutsche, H.J.; Hayes, M.; Hentze, H.W.; et al. EuroBionet: A Pan-European biomonitoring Network for Urban Air Quality Assessment. Environ. Sci. Pollut. Res. 2002, 9, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Scerbo, R.; Ristori, T.; Possenti, L.; Lampugnani, L.; Barale, R.; Barghigiani, C. Lichen (Xanthoria parietina) biomonitoring of trace element contamination and air quality assessment in Pisa Province (Tuscany, Italy). Sci. Total Environ. 2002, 286, 27–40. [Google Scholar] [CrossRef]
- Urbat, M.; Lehndorff, E.; Schwark, L. Biomonitoring of air quality in the Cologne conurbation using pine needles as a passive sampler—Part I: Magnetic properties. Atmos. Environ. 2004, 38, 3781–3792. [Google Scholar] [CrossRef]
- Harmens, H.; Norris, D.; Mills, G. Heavy Metals and Nitrogen in Mosses: Spatial Patterns in 2010/2011 and Long-Term Temporal Trends in Europe; NERC/Centre for Ecology & Hydrology: Bangor, UK, 2013. [Google Scholar]
- Kandziora-Ciupa, M.; Nadgórska-Socha, A.; Barczyk, G.; Ciepał, R. Bioaccumulation of heavy metals and ecophysiological responses to heavy metal stress in selected populations of Vaccinium myrtillus L. and Vaccinium vitis-idaea L. Ecotoxicology 2017, 26, 966–980. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.; Sun, J.; Wan, W.; Hu, E.; Calatayud, V. Evidence of widespread ozone-induced visible injury on plants in Beijing, China. Environ. Pollut. 2014, 193, 296–301. [Google Scholar] [CrossRef]
- Klumpp, A.; Ansel, W.; Klumpp, G.; Calatayud, V.; Pierre Garrec, J.; He, S.; Peñuelas, J.; Ribas, À.; Ro-Poulsen, H.; Rasmussen, S.; et al. Ozone pollution and ozone biomonitoring in European cities. Part I: Ozone concentrations and cumulative exposure indices at urban and suburban sites. Atmos. Environ. 2006, 40, 7963–7974. [Google Scholar] [CrossRef]
- Elagöz, V.; Manning, W.J. Responses of sensitive and tolerant bush beans (Phaseolus vulgaris L.) to ozone in open-top chambers are influenced by phenotypic differences, morphological characteristics, and the chamber environment. Environ. Pollut. 2005, 136, 371–383. [Google Scholar] [CrossRef]
- Krupa, S.V.; Manning, W.J. Atmospheric Ozone: Formation and Effects on Vegetation. Eur. PMC. 1988, 50, 101–137. [Google Scholar] [CrossRef]
- Krupa, S.; McGrath, M.T.; Andersen, C.P.; Booker, F.L.; Burkey, K.O.; Chappelka, A.H.; Chevone, B.I.; Pell, E.J.; Zilinskas, B.A. Ambient Ozone and Plant Health. Plant Dis. 2001, 85, 4–12. [Google Scholar] [CrossRef]
- Sitch, S.; Cox, P.M.; Collins, W.J.; Huntingford, C. Indirect radiative forcing of climate change through ozone effects on the land-carbon sink. Nature 2007, 448, 791–794. [Google Scholar] [CrossRef] [PubMed]
- Lombardozzi, D.; Levis, S.; Bonan, G.; Hess, P.G.; Sparks, J.P.; Lombardozzi, D.; Levis, S.; Bonan, G.; Hess, P.G.; Sparks, J.P. The Influence of Chronic Ozone Exposure on Global Carbon and Water Cycles. J. Clim. 2015, 28, 292–305. [Google Scholar] [CrossRef]
- Hayes, F.; Mills, G.; Harmens, H.; Norris, D. Evidence of Widespread Ozone Damage to Vegetation in Europe (1990–2006); ICP Vegetation Programme Coordination Centre: CEH Bangor, UK, 2007. [Google Scholar]
- Mills, G.; Hayes, F.; Simpson, D.; Emberson, L.; Norris, D.; Harmens, H.; Büker, P. Evidence of widespread effects of ozone on crops and (semi-)natural vegetation in Europe (1990–2006) in relation to AOT40- and flux-based risk maps. Glob. Chang. Biol. 2011, 17, 592–613. [Google Scholar] [CrossRef]
- Schaub, M.; Calatayud, V.; Ferreti, M.; Brunialti, G.; Lovblad, G.; Krause, G.; Sanz, M.J. Assessment of Ozone Injury. Manual on Methods and Criteria for Harmonized Sampling, Assessment, Monitoring and Analysis of the Effects of Air Pollution on Forests; UNECE ICP Forests Programme Co-ordinating Centre: Hamburg, Germany, 2010; Volume 1. [Google Scholar]
- Manning, W.J.; Godzik, B. Bioindicator plants for ambient ozone in Central and Eastern Europe. Environ. Pollut. 2004, 130, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Umponstira, C.; Pimpa, W.; Nanegrungsun, S. Physiological and biochemical responses of cowpea (Vigna unguiculata (L.) Walp) to ozone. Songklanakarin J. Sci. Technol. 2006, 28, 861–869. [Google Scholar]
- Sanz, M.J.; Calvo, E. Ozone injury in crops. Available online: http://ozoneinjury.org (accessed on 17 January 2020).
- Reinert, R.A.; Eason, G. Genetic Control of O3 Sensitivity in a Cross Between Two Cultivars of Snap Bean. J. Am. Soc. Hort. Sci. 2000, 125, 222–227. [Google Scholar] [CrossRef]
- Burkey, K.O.; Miller, J.E.; Fiscus, E.L. Assessment of Ambient Ozone Effects on Vegetation Using Snap Bean as a Bioindicator Species. Publ. J. Environ. Qual 2005, 34, 1081–1086. [Google Scholar] [CrossRef]
- ICP Vegetation New Ozone Garden Project. Available online: https://icpvegetation.ceh.ac.uk/new-ozone-garden-project-hong-kong (accessed on 27 November 2019).
- ICP Vegetation “Seeing is Believing” Ozone injury on Phaseolus vulgaris (common bean). In Bean Biomonitoring Protocol. 2018. Available online: https://icpvegetation.ceh.ac.uk/get-involved/ozone-injury (accessed on 3 January 2019).
- Teledyne Model T400 Photometric Ozone Analyzer Manual. Available online: http://www.teledyne-api.com/prod/Downloads/T400 Manual - 06870.pdf (accessed on 11 November 2018).
- R Core Team. R: A language and environment for statistical computing. In R Foundation for Statistical Computing; 2018; Vienna, Austria. Available online: https://repo.bppt.go.id/cran/web/packages/dplR/vignettes/intro-dplR.pdf (accessed on 1 November 2019).
- Veneables, W.N.; Ripley, B.D. Modern Applied Statistics with S, 4th ed.; Springer: New York, NY, USA, 2002; ISBN 0-387-95457-0. [Google Scholar]
- Zuur, A.; Leno, E.N.; Walker, N.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R, 1st ed.; Springer: New York, NY, USA, 2009; ISBN 978-0-387-87457-9. [Google Scholar]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference : A Practical Information-Theoretic Approach, 2nd ed.; Springer: New York, NY, USA, 2002; ISBN 9781441929730. [Google Scholar]
- Wada, K.C.; Takeno, K. Stress-induced flowering. Plant Signal. Behav. 2010, 5, 944–947. [Google Scholar] [CrossRef]
- Burkey, K.O.; Booker, F.L.; Ainsworth, E.A.; Nelson, R.L. Field assessment of a snap bean ozone bioindicator system under elevated ozone and carbon dioxide in a free air system. Environ. Pollut. 2012, 166, 167–171. [Google Scholar] [CrossRef]
- Anav, A.; Proietti, C.; Menut, L.; Carnicelli, S.; De Marco, A.; Paoletti, E. Sensitivity of stomatal conductance to soil moisture: Implications for tropospheric ozone. Atmos. Chem. Phys 2018, 18, 5747–5763. [Google Scholar] [CrossRef]
- Reiling, K.; Davison, A.W.; Son, V.I. Effects of ozone on stomatal conductance and photosynthesis in populations of Plantago major L. New Phytol. 1995, 129, 587–594. [Google Scholar] [CrossRef]
- Osborne, S.A.; Mills, G.; Hayes, F.; Ainsworth, E.A.; Büker, P.; Emberson, L. Has the sensitivity of soybean cultivars to ozone pollution increased with time? An analysis of published dose-response data. Glob. Chang. Biol. 2016, 22, 3097–3111. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, V.; Kumar, P.; Long, S.P. Decreasing, not increasing, leaf area will raise crop yields under global atmospheric change. Glob. Chang. Biol. 2017, 23, 1626–1635. [Google Scholar] [CrossRef] [PubMed]
- Pringle, J.; Yu, C.; Sachs, M.; Ellis, R. Assessing ozone damage to cutleaf coneflower in an ozone bioindicator garden. J. Franklin Inst. 2018, 355, 6152–6168. [Google Scholar] [CrossRef]
- Fishman, J.; Creilson, J.K.; Parker, P.A.; Ainsworth, E.A.; Vining, G.G.; Szarka, J.; Booker, F.L.; Xu, X. An investigation of widespread ozone damage to the soybean crop in the upper Midwest determined from ground-based and satellite measurements. Atmos. Environ. 2010, 44, 2248–2256. [Google Scholar] [CrossRef]
- Holland, M.; Kinghorn, S.; Emberson, L.; Cinderby, S.; Ashmore, M.; Mills, G.E.; Harmens, H. Development of a framework for probabilistic assessment of the economic losses caused by ozone damage to crops in Europe. ICP Veg. Rep. Defra 2006, 205, 1–62. [Google Scholar]
- NASA Ozone Bioindicator Garden. Available online: https://airquality.gsfc.nasa.gov/ozone-bioindicator-garden (accessed on 27 November 2019).



| Effect | Mean for Resistant Genotype ± 1 σ SD | Number of Replicates for Resistant | Mean for Sensitive Genotype ± 1 σ SD | Number of Replicates for Sensitive | t-Value | p-Value |
|---|---|---|---|---|---|---|
| Days from germination to harvest | 75.1 ± 7.5 | 12 | 78.8 ± 5 | 12 | −1.5 | 0.146 |
| Maximum flowering day | 37.9 ± 7.1 | 12 | 40.3 ±5.2 | 12 | −1.04 | 0.309 |
| Bean per plant | 10.7 ± 5.9 | 12 | 11.3 ± 8.4 | 12 | −0.17 | 0.667 |
| Bean per pod * | 3 ± 1.2 | 56 | 2.6 ± 1.36 | 71 | 1.37 | 0.172 |
| Bean dry weight (g) | 0.21 ± 0.06 | 128 | 0.24 ± 0.05 | 136 | −4.47 | <0.001 |
| Total bean weight per plant (g) | 2.09 ± 1.55 | 12 | 2.69 ± 1.85 | 12 | −0.84 | 0.412 |
| Maximum number of flowers | 8.01 ± 2 | 12 | 12.7 ± 6 | 12 | −2.55 | <0.001 |
| Maximum flowering day | 37.9 ± 7 | 12 | 40.3 ± 5.2 | 12 | −1.04 | 0.309 |
| Pod per plant | 4.9 ± 2.35 | 12 | 5.9 ± 3.0 | 12 | −1.04 | 0.214 |
| Effect | Count for Resistant Genotype | Count for Sensitive Genotype | χ2 | p-Value |
|---|---|---|---|---|
| Successful and (unsucessful) fruit development | 58 (32) | 74 (81) | 6.39 | <0.05 |
| Live and (dead) pod | 43 (15) | 56 (18) | 0.0410 | 0.840 |
| Leaves with <5% and (>=5%) insect injury at MFD | 86 (12) | 115 (10) | 1.11 | 0.291 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leung, F.; Pang, J.Y.S.; Tai, A.P.K.; Lam, T.; Tao, D.K.C.; Sharps, K. Evidence of Ozone-Induced Visible Foliar Injury in Hong Kong Using Phaseolus Vulgaris as a Bioindicator. Atmosphere 2020, 11, 266. https://doi.org/10.3390/atmos11030266
Leung F, Pang JYS, Tai APK, Lam T, Tao DKC, Sharps K. Evidence of Ozone-Induced Visible Foliar Injury in Hong Kong Using Phaseolus Vulgaris as a Bioindicator. Atmosphere. 2020; 11(3):266. https://doi.org/10.3390/atmos11030266
Chicago/Turabian StyleLeung, Felix, Jacky Y. S. Pang, Amos P. K. Tai, Timothy Lam, Donald K. C. Tao, and Katrina Sharps. 2020. "Evidence of Ozone-Induced Visible Foliar Injury in Hong Kong Using Phaseolus Vulgaris as a Bioindicator" Atmosphere 11, no. 3: 266. https://doi.org/10.3390/atmos11030266
APA StyleLeung, F., Pang, J. Y. S., Tai, A. P. K., Lam, T., Tao, D. K. C., & Sharps, K. (2020). Evidence of Ozone-Induced Visible Foliar Injury in Hong Kong Using Phaseolus Vulgaris as a Bioindicator. Atmosphere, 11(3), 266. https://doi.org/10.3390/atmos11030266







