Intercomparison of Multiple UV-LIF Spectrometers Using the Aerosol Challenge Simulator
Abstract
1. Introduction
1.1. Overview of PBAP Measurement Techniques
1.2. UV-LIF Usage
1.3. UV-LIF Discrimination
1.4. Scope
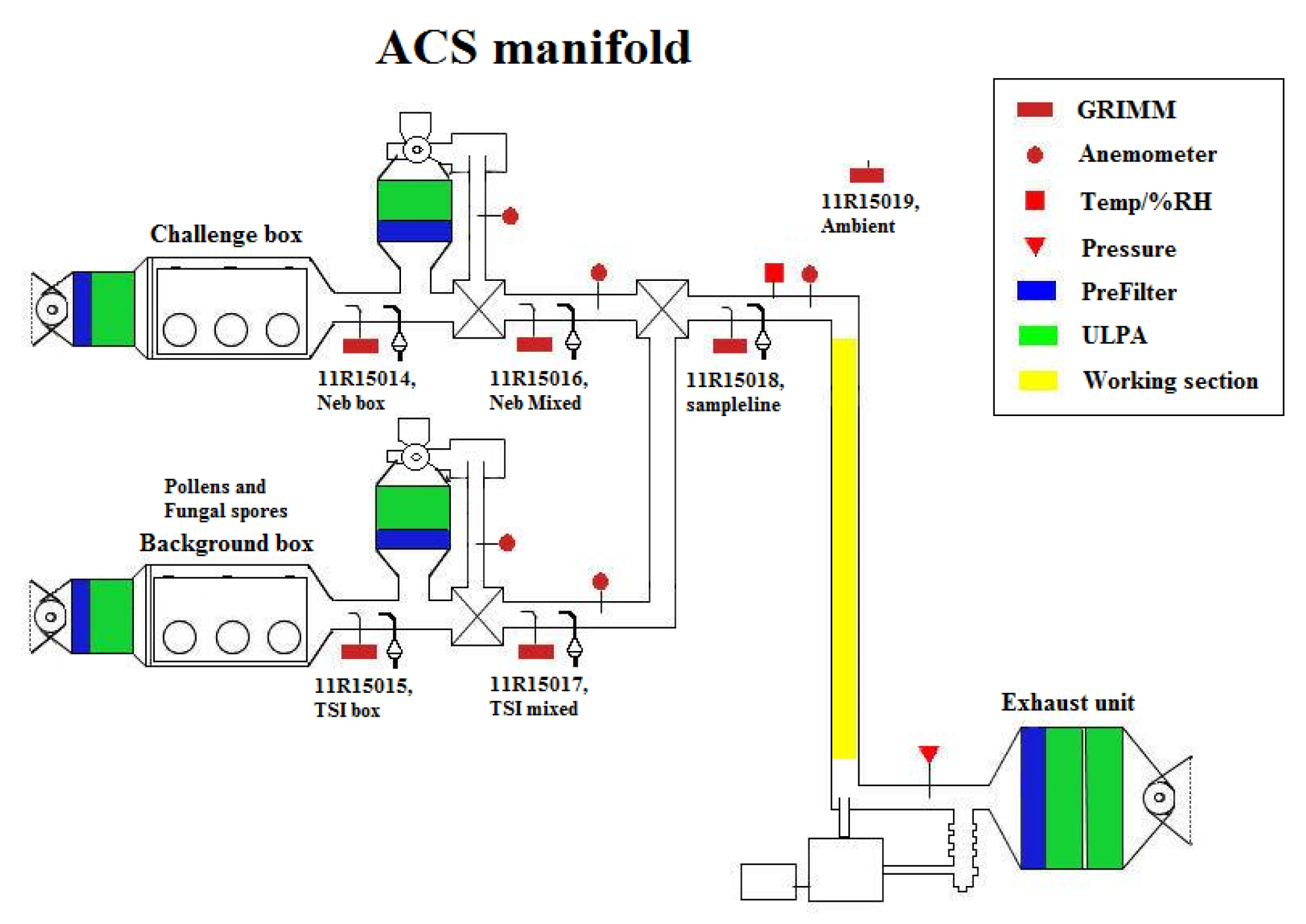
2. The Aerosol Challenge Simulator (ACS)
2.1. ACS Concentration Monitoring
2.2. Aerosolisation Methods
2.3. Challenge Particle List
2.4. Scanning Electron Microscopy
3. UV-LIF Instrumentation
3.1. WIBS-4
3.2. WIBS-NEO
3.3. Multiparameter Bioaerosol Spectrometer (MBS)
4. Data Format and Analysis Protocol
4.1. Data Format
4.1.1. Forced Trigger (FT) Data
4.2. Data Pre-Processing
4.2.1. WIBS Pre-Processing
4.2.2. MBS Pre-Processing
5. Results
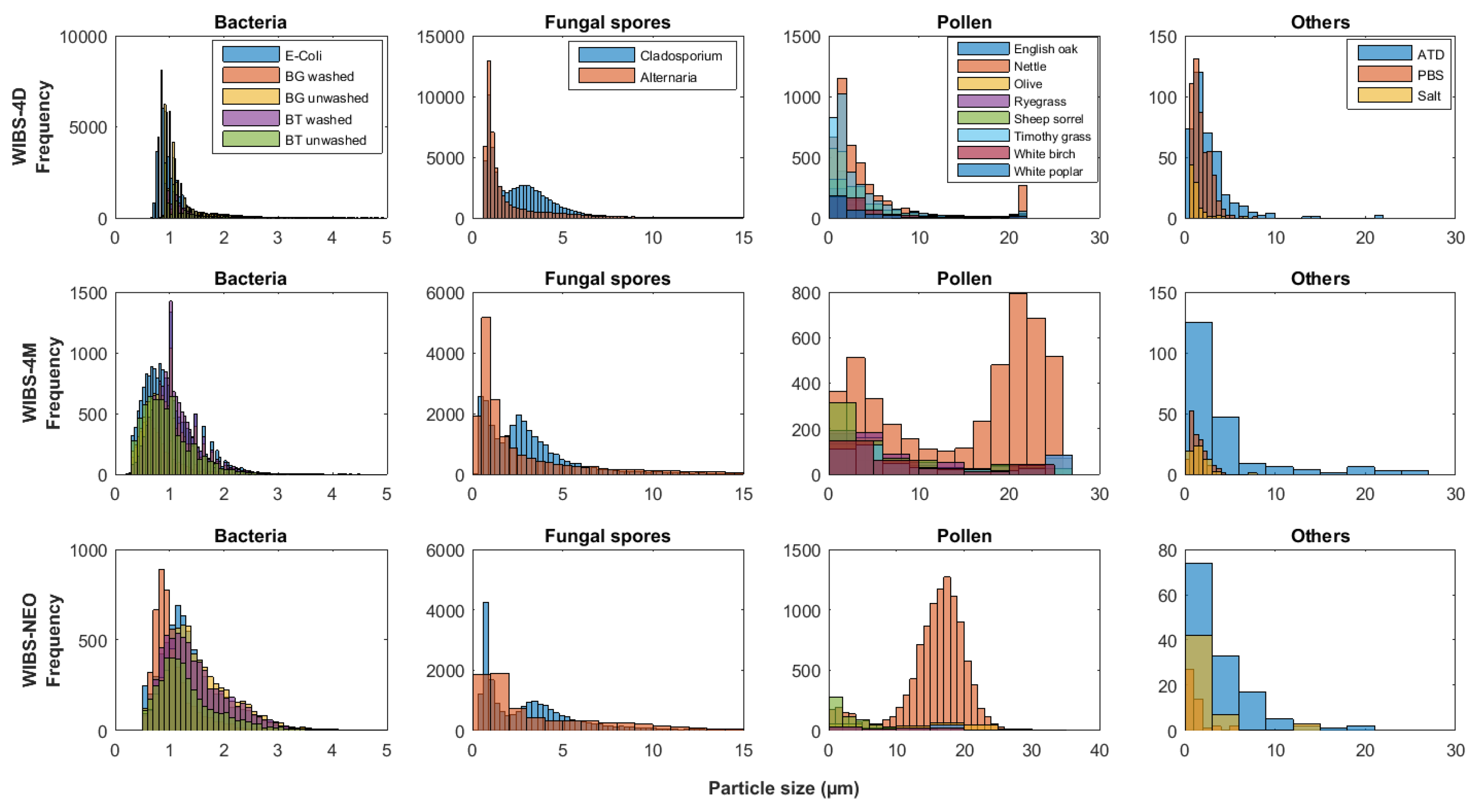
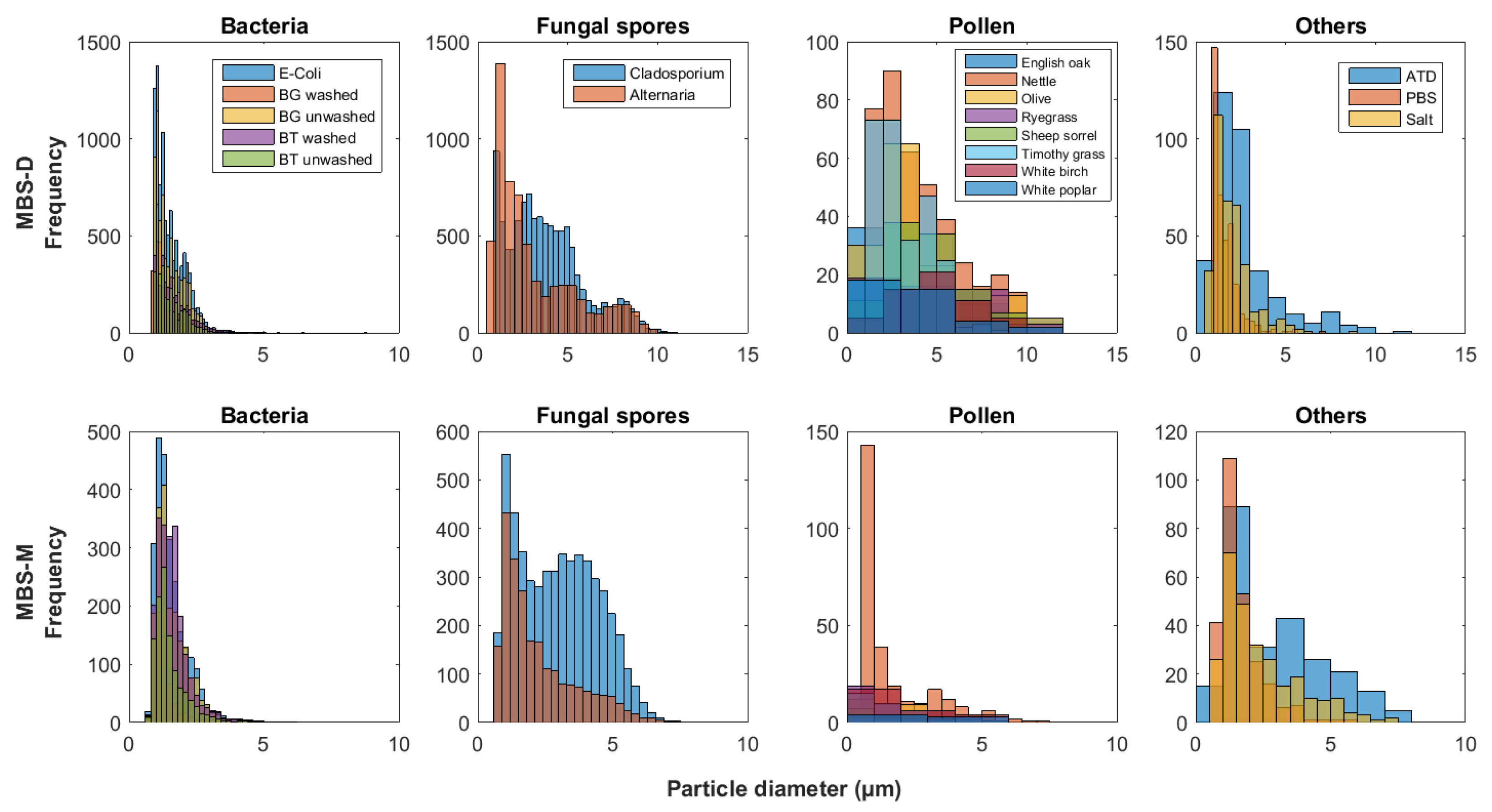
5.1. Particle Fluorescence Profiles
5.1.1. Particle Fragmentation
MBS Response to Cladosporium and Alternaria Material
WIBS Response to Pollen Samples
5.2. Particle Type Differences and Instrument Variation
WIBS
MBS
5.2.1. Bacteria
WIBS
MBS
5.2.2. Fungal Spore Material
WIBS
MBS
5.2.3. Pollen and Pollen Fragments
WIBS
MBS
5.2.4. Non-Biological Samples
WIBS
MBS
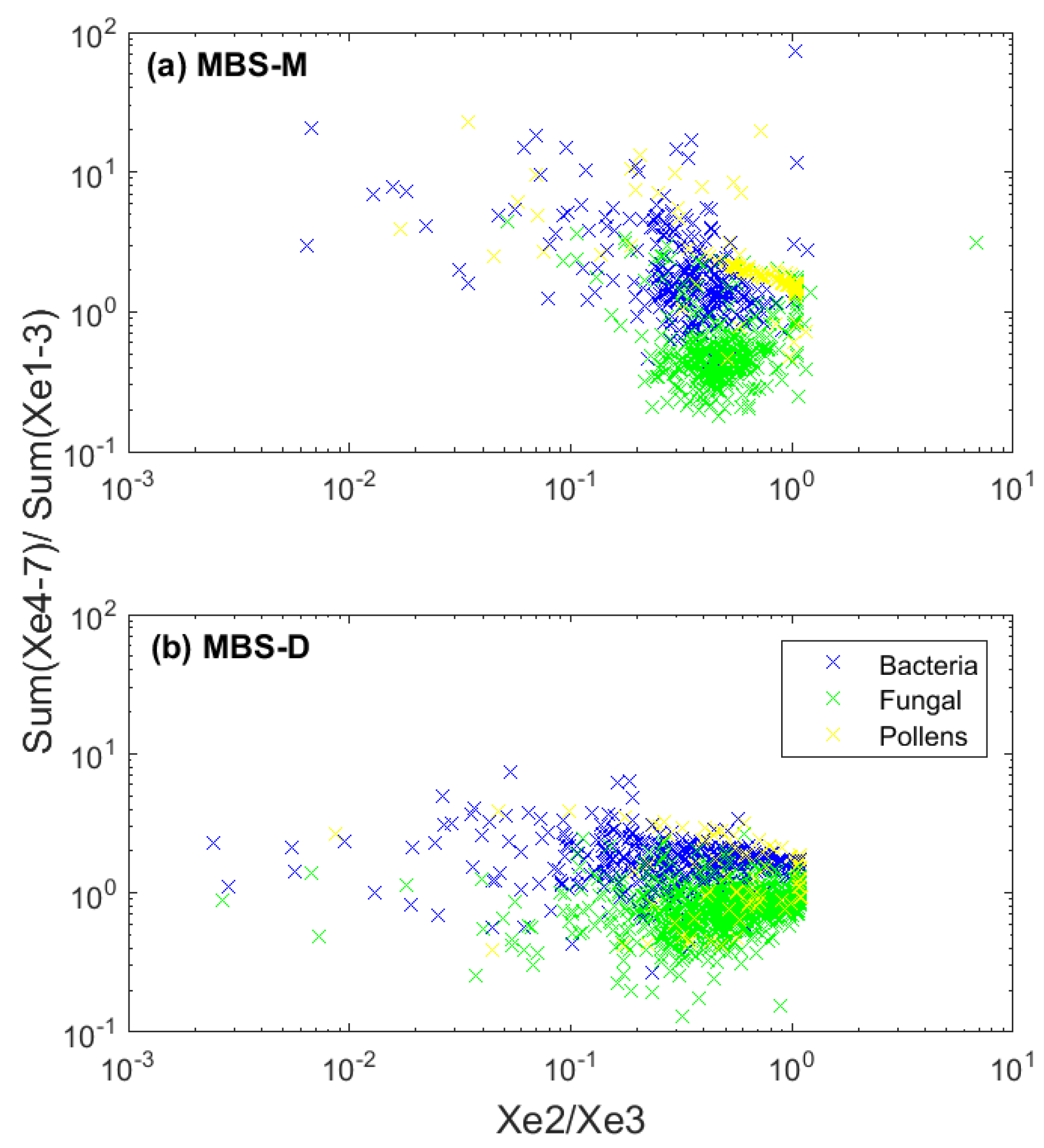
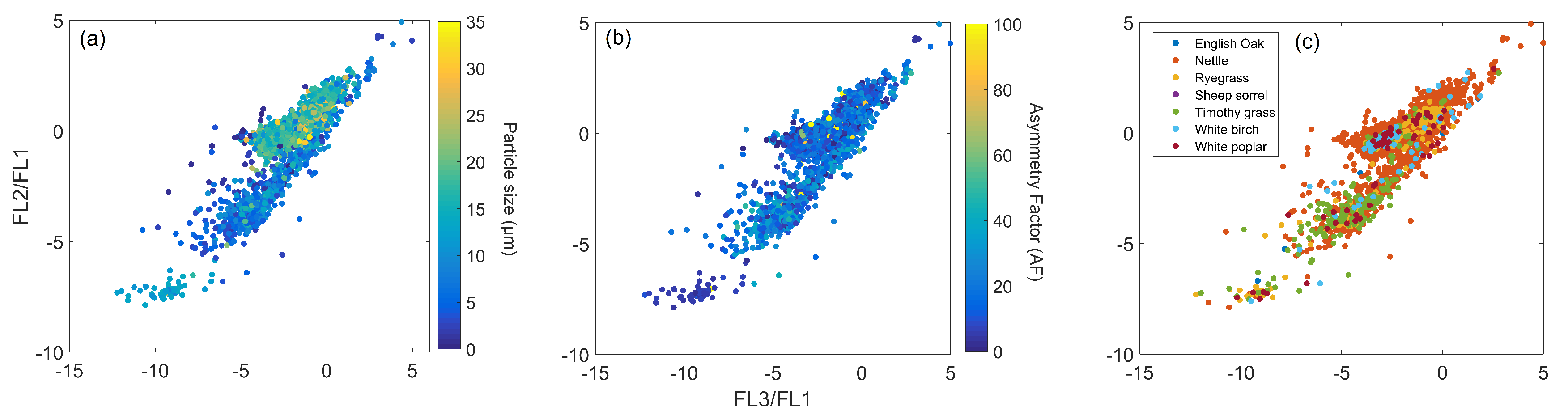
5.3. Relationship between Particle Size, Shape, and Fluorescence
5.3.1. Relationship between Particle Size and Shape
WIBS
MBS
5.4. Response to Dust–Bacterial Mixtures
WIBS
MBS
6. Conclusions and Recommendations
- The number of particles remaining following the removal of FT + 3SD presented a notable difference between the WIBS and MBS. A generally consistent greater number of interferent/non-biological samples were detected by the two MBS instruments especially when compared to the WIBS-4M and WIBS-NEO, and a considerably lower number of pollen particles were detected by the MBS instruments compared to the WIBS (Table S3).
- Pollen samples were the most variable in size and fluorescence response, and often sized smaller than expected, as shown by the WIBS-4D and both MBS instruments (Section 5.2.3). The variability in detected size is suggested to result from the influence of particle morphology affecting light scattering, as identified from SEM images of the samples. Such variability illustrates the potential difficulties in using laboratory data for ambient data interpretation.
- A clear defining trend in fluorescence response could not be identified between the different biological groups (Section 5.2). Additionally, the differences in fluorescent intensities were not always apparent, and often dependent on the instrument used. Although most non-biological particles generally presented lower fluorescence intensities compared to the biological samples, the WIBS-NEO presented higher fluorescent intensities for non-biological particles than some biological particles sampled (Section 5.2.4 and Table 6) due to the detector configuration which may require careful determination of thresholds for subsequent analysis.
- Compared to the WIBS, the MBS presented higher shape values for all particles sampled, especially for pollen particles (Section 5.3.1). To some degree, the larger variation in shape values between the particle groups would enable the pollen samples to be segregated from the other samples, making this potentially useful as an additional classification parameter.
- While only differences in fluorescence intensities could be seen for the different size modes of fragmented particles (Section 5.1.1), the WIBS-4M was the only instrument to display a different fluorescence profile for each size mode of Nettle pollen. The difference in fluorescence between the modes detected by the WIBS-4M requires consideration when interpreting ambient datasets.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Instrument Version
| Instrument | Software Version | Firmware Version |
|---|---|---|
| WIBS-4M | N/a | N/a |
| WIBS-4D | N/a | N/a |
| WIBS-NEO | 2.3.3.16 | 42 |
| MBS-M | 4.3.0.3 | N/a |
| MBS-D | 4.5.0.9 | N/a |
References
- Després, V.R.; Alex Huffman, J.; Burrows, S.M.; Hoose, C.; Safatov, A.S.; Buryak, G.; Fröhlich-Nowoisky, J.; Elbert, W.; Andreae, M.O.; Pöschl, U.; et al. Primary biological aerosol particles in the atmosphere: A review. Tellus Ser. Chem. Phys. Meteorol. 2012, 64. [Google Scholar] [CrossRef]
- Fröhlich-Nowoisky, J.; Kampf, C.J.; Weber, B.; Huffman, J.A.; Pöhlker, C.; Andreae, M.O.; Lang-Yona, N.; Burrows, S.M.; Gunthe, S.S.; Elbert, W.; et al. Bioaerosols in the Earth System: Climate, Health, and Ecosystem Interactions. Atmos. Res. 2016, 182, 346–376. [Google Scholar] [CrossRef]
- Fisher, M.C.; Henk, D.A.; Briggs, C.J.; Brownstein, J.S.; Madoff, L.C.; McCraw, S.L.; Gurr, S.J. Emerging fungal threats to animal, plant and ecosystem health. Nature 2012, 484, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Polymenakou, P.N.; Mandalakis, M.; Stephanou, E.G.; Tselepides, A. Particle size distribution of airborne microorganisms and pathogens during an intense African dust event in the eastern Mediterranean. Environ. Health Perspect. 2008, 116, 292–296. [Google Scholar] [CrossRef]
- Taylor, P.E.; Flagan, R.C.; Valenta, R.; Glovsky, M.M. Release of allergens as respirable aerosols: A link between grass pollen and asthma. J. Allergy Clin. Immunol. 2002, 109, 51–56. [Google Scholar] [CrossRef]
- Pope, F.D. Pollen grains are efficient cloud condensation nuclei. Environ. Res. Lett. 2010, 5, 044015. [Google Scholar] [CrossRef]
- Hoose, C.; Möhler, O. Heterogeneous ice nucleation on atmospheric aerosols: A review of results from laboratory experiments. Atmos. Chem. Phys. 2012, 12, 9817–9854. [Google Scholar] [CrossRef]
- Cziczo, D.J.; Froyd, K.D.; Hoose, C.; Jensen, E.J.; Diao, M.; Zondlo, M.A.; Smith, J.B.; Twohy, C.H.; Murphy, D.M. Clarifying the Dominant Sources and Mechanisms of Cirrus Cloud Formation. Am. Assoc. Adv. Sci. 2013, 340, 1320–1324. [Google Scholar] [CrossRef]
- Robertson, C.E.; Baumgartner, L.K.; Harris, J.K.; Peterson, K.L.; Stevens, M.J.; Frank, D.N.; Pace, N.R. Culture-independent analysis of aerosol microbiology in a metropolitan subway system. Appl. Environ. Microbiol. 2013, 79, 3485–3493. [Google Scholar] [CrossRef]
- Kaye, P.; Stanley, W.R.; Hirst, E.; Foot, E.V.; Baxter, K.L.; Barrington, S.J. Single particle multichannel bio-aerosol fluorescence sensor. Opt. Express 2005, 13, 3583–3593. [Google Scholar] [CrossRef]
- Pöhlker, C.; Huffman, J.A.; Pöschl, U. Autofluorescence of atmospheric bioaerosols—Fluorescent biomolecules and potential interferences. Atmos. Meas. Tech. 2012, 5, 37–71. [Google Scholar] [CrossRef]
- Hill, S.C.; Mayo, M.W.; Chang, R.K. Fluorescence of Bacteria, Pollens, and Naturally Occurring Airborne Particles: Excitation/Emission Spectra; Army Research Laboratory: Adelphi, MD, USA, 2009. [Google Scholar]
- Lakowicz, J.R. (Ed.) Principles of Fluorescence Spectroscopy, 3rd ed.; Springer: Berlin, Germany, 2006; p. 954. [Google Scholar] [CrossRef]
- Foot, V.E.; Kaye, P.H.; Stanley, W.R.; Barrington, S.J.; Gallagher, M.; Gabey, A. Low-Cost real-time multiparameter bio-aerosol sensors. In Proceedings of the Optically Based Biological and Chemical Detection for Defence IV, Cardiff, Wales, UK, 15–18 September 2008; Volume 7116, p. 71160I. [Google Scholar] [CrossRef]
- Karlsson, A.; He, J. Optics of Biological Particles; Springer Science & Business Media: Berlin, Germany, 2005; Volume 238. [Google Scholar] [CrossRef]
- Kiselev, D.; Bonacina, L.; Wolf, J.P. Individual bioaerosol particle discrimination by multi-photon excited fluorescence. Opt. Express 2011, 19, 24516. [Google Scholar] [CrossRef] [PubMed]
- Kiselev, D.; Bonacina, L.; Wolf, J.P. A flash-lamp based device for fluorescence detection and identification of individual pollen grains. Rev. Sci. Instruments 2013, 84. [Google Scholar] [CrossRef] [PubMed]
- Nasir, Z.A.; Rolph, C.; Collins, S.; Stevenson, D.; Gladding, T.L.; Hayes, E.; Williams, B.; Khera, S.; Jackson, S.; Bennett, A.; et al. A controlled study on the characterisation of bioaerosols emissions from compost. Atmosphere 2018, 9, 379. [Google Scholar] [CrossRef]
- Nasir, Z.A.; Hayes, E.; Williams, B.; Gladding, T.; Rolph, C.; Khera, S.; Jackson, S.; Bennett, A.; Collins, S.; Parks, S.; et al. Scoping studies to establish the capability and utility of a real-time bioaerosol sensor to characterise emissions from environmental sources. Sci. Total. Environ. 2019, 648, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Könemann, T.; Savage, N.; Klimach, T.; Walter, D.; Fröhlich-Nowoisky, J.; Su, H.; Pöschl, U.; Huffman, A.; Pöhlker, C. Spectral Intensity Bioaerosol Sensor (SIBS): A new Instrument for Spectrally Resolved Fluorescence Detection of Single Particles in Real-Time. Atmos. Meas. Tech. Discuss. 2018, 1–46. [Google Scholar] [CrossRef]
- Gabey, A.M.; Vaitilingom, M.; Freney, E.; Boulon, J.; Sellegri, K.; Gallagher, M.W.; Crawford, I.P.; Robinson, N.H.; Stanley, W.R.; Kaye, P.H. Observations of fluorescent and biological aerosol at a high-altitude site in central France. Atmos. Chem. Phys. 2013, 13, 7415–7428. [Google Scholar] [CrossRef]
- Whitehead, J.D.; Darbyshire, E.; Brito, J.; Barbosa, H.M.; Crawford, I.; Stern, R.; Gallagher, M.W.; Kaye, P.H.; Allan, J.D.; Coe, H.; et al. Biogenic cloud nuclei in the central Amazon during the transition from wet to dry season. Atmos. Chem. Phys. 2016, 16, 9727–9743. [Google Scholar] [CrossRef]
- Crawford, I.; Gallagher, M.W.; Bower, K.N.; Choularton, T.W.; Flynn, M.J.; Ruske, S.; Listowski, C.; Brough, N.; Lachlan-Cope, T.; Flemming, Z.L.; et al. Real Time Detection of Airborne Bioparticles in Antarctica. Atmos. Chem. Phys. Discuss. 2017, 1–21. [Google Scholar] [CrossRef]
- Gabey, A.M.; Stanley, W.R.; Gallagher, M.W.; Kaye, P.H. The fluorescence properties of aerosol larger than 0.8 μ in urban and tropical rainforest locations. Atmos. Chem. Phys. 2011, 11, 5491–5504. [Google Scholar] [CrossRef]
- Huffman, J.A.; Prenni, A.J.; Demott, P.J.; Pöhlker, C.; Mason, R.H.; Robinson, N.H.; Fröhlich-Nowoisky, J.; Tobo, Y.; Després, V.R.; Garcia, E.; et al. High concentrations of biological aerosol particles and ice nuclei during and after rain. Atmos. Chem. Phys. 2013, 13, 6151–6164. [Google Scholar] [CrossRef]
- Gosselin, M.; Rathnayake, C.M.; Crawford, I.; Pöhlker, C.; Fröhlich-Nowoisky, J.; Schmer, B.; Després, V.R.; Engling, G.; Gallagher, M.; Stone, E.; et al. Fluorescent bioaerosol particle, molecular tracer, and fungal spore concentrations during dry and rainy periods in a semi-arid forest. Atmos. Chem. Phys. 2016, 16, 15165–15184. [Google Scholar] [CrossRef]
- Yue, S.; Ren, H.; Fan, S.; Wei, L.; Zhao, J.; Bao, M.; Hou, S.; Zhan, J.; Zhao, W.; Ren, L.; et al. High Abundance of Fluorescent Biological Aerosol Particles in Winter in Beijing, China. Acs Earth Space Chem. 2017, 1, 493–502. [Google Scholar] [CrossRef]
- Forde, E.; Gallagher, M.; Foot, V.; Sarda-Esteve, R.; Crawford, I.; Kaye, P.; Stanley, W.; Topping, D. Characterisation and source identification of biofluorescent aerosol emissions over winter and summer periods in the United Kingdom. Atmos. Chem. Phys. 2019, 19, 1665–1684. [Google Scholar] [CrossRef]
- Healy, D.A.; O’Connor, D.J.; Burke, A.M.; Sodeau, J.R. A laboratory assessment of the Waveband Integrated Bioaerosol Sensor (WIBS-4) using individual samples of pollen and fungal spore material. Atmos. Environ. 2012, 60, 534–543. [Google Scholar] [CrossRef]
- Toprak, E.; Schnaiter, M. Fluorescent biological aerosol particles measured with the Waveband Integrated Bioaerosol Sensor WIBS-4: Laboratory tests combined with a one year field study. Atmos. Chem. Phys. 2013, 13, 225–243. [Google Scholar] [CrossRef]
- Pan, Y.L. Detection and characterization of biological and other organic-carbon aerosol particles in atmosphere using fluorescence. J. Quant. Spectrosc. Radiat. Transf. 2015, 150, 12–35. [Google Scholar] [CrossRef]
- Pan, Y.L.; Hill, S.C.; Santarpia, J.L.; Brinkley, K.; Sickler, T.; Coleman, M.; Williamson, C.; Gurton, K.; Felton, M.; Pinnick, R.G.; et al. Spectrally-resolved fluorescence cross sections of aerosolized biological live agents and simulants using five excitation wavelengths in a BSL-3 laboratory. Opt. Express 2014, 22, 6191–6208. [Google Scholar] [CrossRef]
- Hernandez, M.; Perring, A.E.; McCabe, K.; Kok, G.; Granger, G.; Baumgardner, D. Chamber catalogues of optical and fluorescent signatures distinguish bioaerosol classes. Atmos. Meas. Tech. 2016, 9, 3283–3292. [Google Scholar] [CrossRef]
- Savage, N.J.; Krentz, C.E.; Könemann, T.; Han, T.T.; Mainelis, G.; Pöhlker, C.; Huffman, J.A. Systematic characterization and fluorescence threshold strategies for the wideband integrated bioaerosol sensor (WIBS) using size-resolved biological and interfering particles. Atmos. Meas. Tech. 2017, 10, 4279–4302. [Google Scholar] [CrossRef]
- Yu, X.; Wang, Z.; Zhang, M.; Kuhn, U.; Xie, Z.; Cheng, Y.; Pöschl, U.; Su, H. Ambient measurement of fluorescent aerosol particles with a WIBS in the Yangtze River Delta of China: Potential impacts of combustion-related aerosol particles. Atmos. Chem. Phys. 2016, 16, 11337–11348. [Google Scholar] [CrossRef]
- Pöschl, U.; Shiraiwa, M. Multiphase Chemistry at the Atmosphere - Biosphere Interface Influencing Climate and Public Health in the Anthropocene. Chem. Rev. 2015, 115, 4440–4475. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M. Dust clouds implicated in spread of infection. Lancet 2001, 358, 478. [Google Scholar] [CrossRef]
- Soleimani, Z.; Goudarzi, G.; Sorooshian, A.; Marzouni, M.B.; Maleki, H. Impact of Middle Eastern dust storms on indoor and outdoor composition of bioaerosol. Atmos. Environ. 2016, 138, 135–143. [Google Scholar] [CrossRef]
- Perring, A.E.; Schwarz, J.P.; Baumgardner, D.; Hernandez, M.T.; Spracklen, D.V.; Heald, C.L.; Gao, R.S.; Kok, G.; Mcmeeking, G.R.; Mcquaid, J.B.; et al. Airborne observations of regional variation in fluorescence aerosol across the United States. J. Geophys. Res. Atmos. 2014, 120, 1153–1170. [Google Scholar] [CrossRef]
- Gabey, A.M.; Gallagher, M.W.; Whitehead, J.; Dorsey, J.R.; Kaye, P.H.; Stanley, W.R. Measurements and comparison of primary biological aerosol above and below a tropical forest canopy using a dual channel fluorescence spectrometer. Atmos. Chem. Phys. 2010, 10, 4453–4466. [Google Scholar] [CrossRef]
- Kaye, P.P. Multiparameter Bioaerosol Sensor User Manual; Centre for Atmospheric & Instrumentation Research (CAIR), The University of Hertfordshire: Hertfordshire, UK, 2013; pp. 1–32. [Google Scholar]
Sample Availability: The UV-LIF datasets will be accessible via Zenodo (10.5281/zenodo.3567097), and the analysis scripts produced to process the UV-LIF data are available at: https://github.com/elizabethforde/UV-LIF. |






| Parameter | WIBS-4M | WIBS-4D | WIBS-NEO | MBS-M | MBS-D |
|---|---|---|---|---|---|
| Size range | 0.5–20 m | 0.5–20 m | 0.5–30 m | 0.5–20 m | 0.5–20 m |
| Total Flow Rate | 2.5 L/min | 2.5 L/min | 2.1 L/min | 1.5 L/min | 1.5 L/min |
| Sample Flow Rate | 0.38 L/min | 1.2 L/min (usually 1 L/min) | 0.3 L/min | 0.2 L/min | 0.2 L/min |
| Size/shape | 635 nm laser | 635 nm laser | 635 nm laser | 635 nm (size) 637 nm (shape) | 635 nm (size) 637 nm (shape) |
| Size/shape detection | Quadrant PMT | Quadrant PMT | Quadrant PMT | CMOS linear arrays | CMOS linear arrays |
| Fl. Excitation | 280 nm, 370 nm | 280 nm, 370 nm | 280 nm, 370 nm | 280 nm | 280 nm |
| Fl. Detection | 310–400 nm, 420–650 nm | 310–400 nm, 420–650 nm | 310–400 nm, 420–650 nm | 8 channel (310–630 nm) | 8 channel detection (310–630 nm) |
| MBS-M | XE1 | XE2 | XE3 | XE4 | XE5 | XE6 | XE7 | XE8 | SIZE (m) | SHAPE |
|---|---|---|---|---|---|---|---|---|---|---|
| PSLs | ||||||||||
| 3 m | 507.8 ± 0.0 | 274.9 ± 612.6 | 523.1 ± 576.5 | 77.4 ± 719.1 | 387.0 ± 507.7 | 207.1 ± 290.1 | 266.9 ± 0.0 | 411.6 ± 0.0 | 3.2 ± 1.2 | 12.2 ± 6.6 |
| 2 m blue | 279.9 ± 71.9 | 287.7 ± 67.2 | 347.9 ± 73.6 | 1538.3 ± 435.8 | 1893.1 ± 298.6 | 1921.3 ± 273.5 | 1448.7 ± 218.0 | 487.9 ± 90.7 | 2.0 ± 0.4 | 12.2 ± 6.6 |
| 1 m blue | 12.5 ± 15.2 | 18.8 ± 50.6 | 20.9 ± 98.7 | 193.0 ± 100.2 | 1742.8 ± 275.9 | 708.1 ± 221.6 | 95.0 ± 59.3 | 15.0 ± 19.0 | 1.3 ± 0.2 | 12.7 ± 4.6 |
| 3 m green | 361.1 ± 46.4 | 531.0 ± 86.2 | 629.1 ± 130.9 | 477.1 ± 51.1 | 1895.6 ± 295.7 | 1923.1 ± 213.7 | 1952.3 ± 217.8 | 1806.5 ± 342.2 | 3.2 ± 0.7 | 11.4 ± 7.9 |
| 2 m green | 113.5 ± 31.8 | 160.6 ± 52.3 | 182.0 ± 74.4 | 119.4 ± 49.9 | 1819.3 ± 120.0 | 1919.1 ± 0.3 | 1955.4 ± 151.7 | 795.8 ± 111.9 | 1.8 ± 0.3 | 10.0 ± 4.0 |
| 1 m green | 9.9 ± 16.3 | 17.1 ± 23.3 | 18.3 ± 24.1 | 17.4 ± 26.0 | 248.7 ± 130.5 | 819.7 ± 220.2 | 270.1 ± 139.5 | 33.2 ± 58.8 | 1.2 ± 0.3 | 12.5 ± 5.8 |
| 23 m | 268.5 ± 0.0 | 903.6 ± 1151.9 | 895.4 ± 1107.3 | 620.0 ± 0.0 | 117.0 ± 157.8 | 13.0 ± 0.0 | 31.1 ± 0.0 | 1.0 ± 0.2 | 15.0 ± 21.2 | |
| 11 m | 52.2 ± 944.6 | 148.4 ± 671.2 | 160.0 ± 577.1 | 154.9 ± 562.3 | 34.8 ± 407.7 | 58.0 ± 305.7 | 26.6 ± 378.9 | 18.6 ± 847.2 | 1.2 ± 1.2 | 17.5 ± 13.0 |
| Pollen | ||||||||||
| Timothy grass (Pheleum pratense) | 226.1 ± 573.1 | 789.7 ± 779.8 | 916.6 ± 721.1 | 741.5 ± 775.7 | 399.0 ± 840.9 | 261.6 ± 895.6 | 1937.3 ± 953.4 | 344.0 ± 904.2 | 1.1 ± 1.2 | 21.4 ± 13.0 |
| Nettle (Urtica dioica) | 1262.4 ± 441.4 | 1858.5 ± 751.7 | 1809.8 ± 755.9 | 1818.6 ± 740.3 | 1893.1 ± 816.8 | 1917.6 ± 833.8 | 1944.8 ± 827.3 | 1774.7 ± 674.8 | 0.9 ± 1.3 | 30.0 ± 12.5 |
| Sheep Sorrel (Rumex acetosella) | 1108.5 ± 533.3 | 1668.5 ± 821.5 | 1796.7 ± 590.0 | 1822.3 ± 588.8 | 1887.5 ± 765.5 | 1921.6 ± 687.6 | 1942.1 ± 577.2 | 1769.7 ± 521.1 | 0.9 ± 0.7 | 34.6 ± 13.6 |
| Ryegrass (Lolium perenne) | 1225.1 ± 568.0 | 1637.5 ± 627.5 | 1808.4 ± 442.7 | 1817.3 ± 424.0 | 1904.2 ± 732.2 | 1916.6 ± 542.9 | 1960.6 ± 842.6 | 1883.5 ± 571.0 | 0.9 ± 1.2 | 32.4 ± 11.8 |
| White Poplar (Populus alba) | 605.7 ± 835.5 | 124.8 ± 837.3 | 276.2 ± 714.6 | 347.8 ± 739.3 | 216.8 ± 733.9 | 149.5 ± 729.3 | 92.7 ± 828.3 | 70.4 ± 934.2 | 2.9 ± 1.9 | 35.4 ± 21.5 |
| European white birch (Betula pendula) | 100.8 ± 627.9 | 183.9 ± 858.1 | 1049.1 ± 879.0 | 1819.6 ± 868.0 | 1313.0 ± 887.2 | 129.9 ± 862.4 | 256.1 ± 877.2 | 574.3 ± 853.4 | 1.4 ± 1.5 | 32.7 ± 13.6 |
| Olive (Olea europaea) | 51.4 ± 496.0 | 186.3 ± 679.5 | 337.9 ± 677.1 | 202.6 ± 712.9 | 1272.2 ± 829.8 | 1212.7 ± 763.9 | 695.7 ± 607.4 | 266.5 ± 638.2 | 1.3 ± 0.7 | 14.5 ± 11.0 |
| English Oak (Quercus robur) | 812.9 ± 1113.0 | 140.1 ± 1023.7 | 198.9 ± 752.0 | 67.9 ± 490.2 | 33.3 ± 660.1 | 34.8 ± 729.3 | 9.9 ± 726.9 | 465.1 ± 932.3 | 2.6 ± 0.9 | 13.4 ± 10.2 |
| Fungal spores | ||||||||||
| Cladosporium herbarium | 10.6 ± 80.5 | 113.1 ± 155.8 | 228.0 ± 311.0 | 104.6 ± 153.8 | 44.8 ± 110.1 | 29.6 ± 99.1 | 16.3 ± 80.0 | 12.6 ± 103.6 | 2.9 ± 1.5 | 18.9 ± 10.3 |
| Alternaria alternata | 8.5 ± 178.9 | 29.5 ± 200.7 | 65.1 ± 279.7 | 42.9 ± 326.2 | 27.3 ± 335.6 | 19.4 ± 344.0 | 13.6 ± 293.7 | 10.8 ± 297.2 | 1.8 ± 1.3 | 17.6 ± 10.7 |
| Bacteria | ||||||||||
| Esherishia coli(E. coli) | 12.4 ± 25.1 | 69.4 ± 148.5 | 112.0 ± 312.6 | 83.0 ± 269.8 | 77.8 ± 216.8 | 63.7 ± 143.7 | 26.4 ± 66.3 | 18.3 ± 43.2 | 1.4 ± 0.6 | 12.2 ± 4.8 |
| Bacillus atrophaeus (BG) (washed) | 9.3 ± 11.3 | 16.9 ± 63.2 | 31.4 ± 101.2 | 15.7 ± 73.2 | 14.3 ± 28.6 | 8.2 ± 24.7 | 8.0 ± 14.4 | 10.2 ± 18.2 | 1.2 ± 0.6 | 11.7 ± 5.5 |
| Bacillus atrophaeus (BG) (unwashed) | 8.8 ± 11.6 | 29.0 ± 47.6 | 76.2 ± 132.8 | 84.9 ± 209.6 | 87.7 ± 197.4 | 53.6 ± 124.9 | 29.9 ± 56.4 | 18.0 ± 25.1 | 1.4 ± 0.6 | 11.8 ± 4.2 |
| Bacillus thuringensis (BT) (washed) | 5.5 ± 10.0 | 20.3 ± 27.2 | 34.8 ± 49.9 | 15.2 ± 26.4 | 12.6 ± 14.6 | 11.7 ± 12.9 | 6.0 ± 8.3 | 6.2 ± 9.6 | 1.5 ± 0.6 | 12.2 ± 5.0 |
| Bacillus thuringensis (BT) (unwashed) | 5.6 ± 13.1 | 16.5 ± 23.2 | 34.2 ± 75.9 | 75.9 ± 191.2 | 63.8 ± 231.7 | 53.7 ± 181.9 | 41.3 ± 70.9 | 16.2 ± 30.0 | 1.3 ± 0.6 | 12.4 ± 5.0 |
| Others | ||||||||||
| Arizona test dust | 4.2 ± 6.0 | 8.5 ± 31.0 | 12.7 ± 141.7 | 11.5 ± 160.3 | 10.9 ± 75.3 | 9.1 ± 26.1 | 8.8 ± 29.0 | 5.8 ± 9.5 | 2.6 ± 1.8 | 18.5 ± 12.2 |
| PBS | 6.0 ± 9.1 | 8.1 ± 22.6 | 15.6 ± 123.5 | 14.9 ± 70.5 | 9.5 ± 8.1 | 8.0 ± 7.9 | 4.4 ± 9.4 | 8.5 ± 8.5 | 1.4 ± 0.8 | 11.9 ± 5.7 |
| Salt (NaCl) | 4.8 ± 8.5 | 13.5 ± 0.7 | 12.2 ± 14.5 | 10.5 ± 8.0 | 9.8 ± 11.2 | 7.8 ± 11.6 | 8.8 ± 10.4 | 7.8 ± 17.9 | 1.9 ± 1.4 | 14.7 ± 9.4 |
| Mixture | 6.0 ± 112.8 | 18.0 ± 63.1 | 44.0 ± 121.3 | 47.2 ± 135.1 | 61.1 ± 162.9 | 46.4 ± 372.5 | 33.7 ± 163.2 | 19.6 ± 133.0 | 1.3 ± 1.0 | 12.2 ± 7.8 |
| MBS-D | XE1 | XE2 | XE3 | XE4 | XE5 | XE6 | XE7 | XE8 | SIZE (m) | SHAPE |
|---|---|---|---|---|---|---|---|---|---|---|
| PSLs | ||||||||||
| 3 m | 35.7 ± 41.6 | 364.6 ± 198.1 | 1220.9 ± 582.3 | 105.4 ± 124.2 | 63.4 ± 86.7 | 34.4 ± 54.9 | 23.5 ± 24.3 | 16.9 ± 26.0 | 2.8 ± 1.0 | 15.4 ± 21.6 |
| 2 m blue | 264.4 ± 138.3 | 468.7 ± 217.2 | 1536.7 ± 350.6 | 1555.7 ± 310.6 | 1626.1 ± 198.7 | 1660.7 ± 148.7 | 1860.4 ± 258.0 | 651.7 ± 214.8 | 2.0 ± 0.4 | 8.0 ± 5.4 |
| 1 m blue | 24.2 ± 28.7 | 33.5 ± 59.0 | 85.0 ± 116.0 | 1311.3 ± 385.9 | 1598.5 ± 145.8 | 1586.9 ± 359.7 | 145.7 ± 120.9 | 20.9 ± 39.6 | 1.0 ± 0.2 | 12.6 ± 5.9 |
| 3 m green | 365.9 ± 109.9 | 814.5 ± 236.9 | 1495.5 ± 311.5 | 1587.5 ± 157.8 | 1624.3 ± 0.0 | 1696.4 ± 214.8 | 1868.6 ± 151.5 | 1844.8 ± 107.5 | 2.9 ± 0.5 | 13.1 ± 110.6 |
| 2 m green | 115.1 ± 71.7 | 189.5 ± 107.7 | 438.0 ± 215.0 | 1430.1 ± 442.6 | 1604.4 ± 25.6 | 1684.6 ± 82.5 | 1860.1 ± 63.0 | 1466.8 ± 322.4 | 1.7 ± 0.2 | 8.1 ± 6.2 |
| 1 m green | 19.2 ± 24.6 | 36.6 ± 40.4 | 47.3 ± 58.2 | 75.8 ± 91.8 | 949.3 ± 348.5 | 1690.1 ± 258.6 | 574.9 ± 250.4 | 50.3 ± 71.2 | 1.0 ± 0.2 | 13.4 ± 11.1 |
| 23 m | 81.1 ± 175.8 | 446.3 ± 430.6 | 1451.5 ± 494.4 | 304.5 ± 350.6 | 56.3 ± 74.0 | 59.0 ± 65.8 | 14.8 ± 9.9 | 2.0 ± 0.1 | 3.0 ± 1.4 | 24.1 ± 95.4 |
| 11 m | 79.0 ± 328.5 | 290.4 ± 468.7 | 689.4 ± 644.7 | 208.0 ± 532.4 | 147.8 ± 522.5 | 139.4 ± 429.7 | 32.5 ± 403.6 | 16.9 ± 284.7 | 2.8 ± 1.9 | 26.9 ± 233.9 |
| Pollen | ||||||||||
| Timothy grass (Pheleum pratense) | 36.6 ± 57.8 | 147.0 ± 296.0 | 246.7 ± 524.8 | 157.6 ± 419.6 | 134.0 ± 350.3 | 135.7 ± 211.7 | 60.4 ± 76.0 | 17.6 ± 32.0 | 2.5 ± 1.6 | 22.6 ± 21.5 |
| Nettle (Urtica dioica) | 101.3 ± 236.7 | 227.9 ± 408.6 | 733.5 ± 595.4 | 413.4 ± 568.9 | 350.8 ± 593.1 | 287.2 ± 603.0 | 136.5 ± 581.0 | 95.2 ± 447.9 | 3.4 ± 2.3 | 29.2 ± 21.1 |
| Sheep Sorrel (Rumex acetosella) | 28.1 ± 104.6 | 258.5 ± 468.1 | 804.0 ± 605.6 | 347.4 ± 505.4 | 157.6 ± 476.1 | 137.1 ± 445.6 | 62.0 ± 213.8 | 64.7 ± 32.5 | 4.2 ± 2.1 | 32.6 ± 40.4 |
| Ryegrass (Lolium perenne) | 121.6 ± 206.6 | 307.6 ± 551.8 | 391.3 ± 611.6 | 276.4 ± 600.7 | 244.0 ± 596.6 | 209.1 ± 501.0 | 136.8 ± 454.8 | 31.7 ± 375.5 | 4.6 ± 2.7 | 38.3 ± 77.9 |
| White Poplar (Populus alba) | 75.9 ± 570.0 | 398.9 ± 464.8 | 382.2 ± 577.1 | 468.1 ± 554.9 | 330.4 ± 604.4 | 201.4 ± 581.8 | 103.5 ± 478.2 | 51.6 ± 267.3 | 3.4 ± 2.5 | 45.6 ± 1214.1 |
| European white birch (Betula pendula) | 61.6 ± 163.7 | 409.6 ± 378.6 | 712.9 ± 637.3 | 390.2 ± 523.8 | 192.3 ± 499.1 | 118.9 ± 535.3 | 66.0 ± 645.3 | 38.0 ± 525.4 | 5.1 ± 2.1 | 40.5 ± 34.2 |
| Olive (Olea europaea) | 53.8 ± 98.4 | 256.4 ± 297.4 | 774.9 ± 588.1 | 207.5 ± 473.4 | 161.3 ± 415.0 | 122.4 ± 450.8 | 66.0 ± 431.7 | 47.4 ± 461.0 | 3.1 ± 2.5 | 20.0 ± 14.5 |
| English Oak (Quercus robur) | 49.2 ± 100.3 | 182.6 ± 489.6 | 649.8 ± 562.7 | 171.1 ± 478.4 | 105.8 ± 353.3 | 45.7 ± 440.4 | 35.9 ± 485.7 | 34.3 ± 618.0 | 2.7 ± 2.9 | 26.7 ± 25.8 |
| Fungal spores | ||||||||||
| Cladosporium herbarium | 49.2 ± 109.8 | 238.2 ± 326.5 | 858.1 ± 579.5 | 335.4 ± 423.9 | 147.5 ± 251.2 | 92.0 ± 160.6 | 46.9 ± 91.5 | 23.0 ± 91.4 | 3.5 ± 2.1 | 23.5 ± 12.0 |
| Alternaria alternata | 29.6 ± 73.2 | 92.3 ± 249.3 | 198.3 ± 445.2 | 166.0 ± 380.3 | 141.0 ± 322.2 | 93.2 ± 224.2 | 47.9 ± 126.7 | 27.1 ± 51.0 | 2.2 ± 2.3 | 23.7 ± 30.8 |
| Bacteria | ||||||||||
| Esherishia coli (E. coli) | 33.1 ± 70.2 | 113.6 ± 256.3 | 328.0 ± 488.2 | 253.7 ± 441.0 | 209.6 ± 407.0 | 129.5 ± 305.0 | 56.6 ± 142.8 | 32.0 ± 52.7 | 1.4 ± 0.5 | 17.5 ± 9.7 |
| Bacillus atrophaeus (BG) (washed) | 21.7 ± 22.6 | 43.6 ± 64.3 | 120.4 ± 165.9 | 41.6 ± 63.4 | 40.9 ± 42.6 | 25.9 ± 40.4 | 15.4 ± 31.1 | 15.4 ± 18.4 | 1.1 ± 0.5 | 13.6 ± 15.6 |
| Bacillus atrophaeus (BG) (unwashed) | 16.5 ± 26.2 | 47.9 ± 93.1 | 153.5 ± 317.4 | 186.2 ± 382.9 | 176.4 ± 372.9 | 117.6 ± 282.1 | 47.1 ± 114.8 | 24.9 ± 40.1 | 1.3 ± 0.5 | 14.2 ± 371.5 |
| Bacillus thuringensis (BT) (washed) | 18.0 ± 29.3 | 42.1 ± 72.4 | 109.4 ± 164.3 | 50.0 ± 88.4 | 38.4 ± 68.8 | 38.7 ± 53.8 | 16.6 ± 28.5 | 17.5 ± 19.7 | 1.4 ± 0.6 | 14.5 ± 8.4 |
| Bacillus thuringensis (BT) (unwashed) | 21.0 ± 29.7 | 37.8 ± 90.7 | 109.5 ± 236.0 | 175.6 ± 363.3 | 229.4 ± 424.2 | 164.4 ± 353.4 | 69.2 ± 145.8 | 25.6 ± 38.8 | 1.3 ± 0.5 | 14.3 ± 8.9 |
| Others | ||||||||||
| Arizona test dust | 13.8 ± 91.9 | 31.9 ± 117.6 | 38.1 ± 274.0 | 31.5 ± 191.6 | 34.1 ± 229.5 | 19.2 ± 354.5 | 18.2 ± 226.0 | 25.2 ± 296.3 | 2.0 ± 1.8 | 19.9 ± 12.3 |
| PBS | 14.8 ± 134.4 | 19.9 ± 207.0 | 52.2 ± 395.0 | 40.5 ± 313.2 | 27.8 ± 302.5 | 27.4 ± 116.8 | 11.0 ± 35.5 | 16.3 ± 22.4 | 1.4 ± 0.8 | 16.6 ± 8.6 |
| Salt (NaCl) | 17.6 ± 20.8 | 20.9 ± 31.4 | 29.5 ± 33.0 | 25.6 ± 38.9 | 18.2 ± 27.6 | 21.4 ± 29.7 | 15.9 ± 17.9 | 16.6 ± 28.1 | 1.7 ± 1.1 | 16.0 ± 9.4 |
| Mixture | 18.4 ± 29.1 | 43.4 ± 80.4 | 113.2 ± 223.4 | 136.6 ± 281.7 | 181.6 ± 418.2 | 142.3 ± 621.8 | 67.7 ± 364.7 | 35.6 ± 96.4 | 1.3 ± 0.8 | 16.1 ± 9.2 |
| WIBS-4D | FL1 | FL2 | FL3 | SIZE (m) | AF |
|---|---|---|---|---|---|
| PSLs | |||||
| 3 m | 83.3 ± 189.9 | 45.4 ± 139.2 | 27.6 ± 152.3 | 1.1 ± 1.2 | 7.1 ± 5.6 |
| 2 m blue | 58.3 ± 96.3 | 1954.4 ± 535.5 | 1806.6 ± 416.3 | 1.8 ± 0.5 | 6.2 ± 5.0 |
| 1 m blue | 7.3 ± 13.6 | 1954.4 ± 63.9 | 1806.6 ± 31.6 | 0.9 ± 0.2 | 13.4 ± 5.7 |
| 3 m green | 386.3 ± 86.1 | 1954.4 ± 209.3 | 1806.6 ± 48.4 | 3.0 ± 0.5 | 4.4 ± 2.8 |
| 2 m green | 108.3 ± 31.7 | 1954.4 ± 102.3 | 1806.6 ± 37.2 | 1.9 ± 0.3 | 5.8 ± 3.7 |
| 1 m green | 21.3 ± 10.8 | 1954.4 ± 70.5 | 1735.6 ± 138.8 | 0.9 ± 0.2 | 13.9 ± 5.7 |
| 23 m | 359.8 ± 347.6 | 97.9 ± 148.6 | 63.6 ± 160.4 | 3.1 ± 1.8 | 9.2 ± 15.9 |
| 11 m | 38.3 ± 474.2 | 89.4 ± 439.6 | 107.6 ± 487.2 | 1.1 ± 2.9 | 12.5 ± 14.4 |
| Pollens (complete) | |||||
| Timothy grass (Pheleum pratense) | 33.3 ± 288.5 | 118.4 ± 506.4 | 184.6 ± 573.6 | 1.7 ± 4.0 | 23.0 ± 19.6 |
| Nettle (Urtica dioica) | 97.3 ± 623.6 | 423.4 ± 749.3 | 620.6 ± 718.9 | 2.4 ± 5.5 | 25.1 ± 18.0 |
| Sheep Sorrel (Rumex acetosella) | 53.3 ± 516.4 | 238.4 ± 652.0 | 357.6 ± 646.2 | 2.9 ± 4.5 | 24.9 ± 18.0 |
| Ryegrass (Lolium perenne) | 77.3 ± 555.7 | 436.4 ± 727.5 | 437.6 ± 689.9 | 2.7 ± 5.2 | 24.7 ± 19.3 |
| White Poplar (Populus alba) | 34.3 ± 539.4 | 378.4 ± 683.7 | 334.6 ± 721.8 | 1.5 ± 4.8 | 20.4 ± 17.0 |
| European white birch (Betula pendula) | 83.3 ± 508.5 | 191.4 ± 552.9 | 297.6 ± 612.9 | 2.6 ± 4.1 | 26.1 ± 18.2 |
| Olive (Olea europaea) | 202.3 ± 547.9 | 87.4 ± 536.0 | 309.6 ± 585.5 | 1.1 ± 3.9 | 12.6 ± 15.6 |
| English Oak (Quercus robur) | 10.3 ± 504.4 | 91.4 ± 491.2 | 608.6 ± 650.2 | 1.1 ± 2.9 | 11.9 ± 11.5 |
| Fungal spores | |||||
| Cladosporium herbarium | 110.3 ± 247.7 | 210.4 ± 319.2 | 213.6 ± 331.4 | 2.3 ± 2.2 | 29.3 ± 20.5 |
| Alternaria alternata | 29.3 ± 155.1 | 84.4 ± 451.7 | 202.6 ± 594.3 | 1.2 ± 2.5 | 19.9 ± 18.9 |
| Bacteria | |||||
| Esherishia coli(E. coli) (unwashed) | 46.3 ± 149.7 | 228.4 ± 472.4 | 257.6 ± 529.1 | 1.0 ± 0.4 | 11.6 ± 5.7 |
| Bacillus atrophaeus (BG) (washed) | 35.3 ± 30.4 | 19.4 ± 65.6 | 29.6 ± 132.7 | 0.9 ± 0.3 | 12.7 ± 5.5 |
| Bacillus atrophaeus (BG) (unwashed) | 13.3 ± 44.8 | 163.4 ± 404.6 | 191.6 ± 462.4 | 1.0 ± 0.4 | 11.7 ± 5.7 |
| Bacillus thuringensis (BT) (washed) | 32.3 ± 27.5 | 23.4 ± 49.4 | 30.6 ± 103.3 | 1.1 ± 0.3 | 11.5 ± 5.2 |
| Bacillus thuringensis (BT) (unwashed) | 8.3 ± 26.7 | 300.4 ± 495.4 | 247.6 ± 485.2 | 1.0 ± 0.3 | 12.0 ± 5.8 |
| Others | |||||
| Arizona test dust (ATD) | 1.3 ± 241.8 | 52.4 ± 343.1 | 96.6 ± 413.1 | 1.9 ± 2.4 | 23.6 ± 16.1 |
| Phosphate-buffered saline (PBS) | 3.3 ± 123.5 | 120.4 ± 260.9 | 35.1 ± 46.0 | 1.6 ± 1.0 | 12.7 ± 6.8 |
| Salt (NaCl) | 0.3 ± 20.4 | 55.9 ± 53.0 | 39.6 ± 41.0 | 1.1 ± 0.9 | 11.0 ± 9.6 |
| Mixture | 17.3 ± 35.3 | 214.4 ± 572.6 | 209.6 ± 515.7 | 0.9 ± 0.3 | 12.7 ± 6.4 |
| WIBS-4M | FL1 | FL2 | FL3 | SIZE (m) | AF |
|---|---|---|---|---|---|
| PSLs | |||||
| 3 m | 312.1 ± 146.7 | 6.1 ± 22.3 | 2.9 ± 56.4 | 2.4 ± 1.8 | 4.9 ± 3.8 |
| 2 m blue | 50.1 ± 76.6 | 2063.1 ± 490.0 | 1962.9 ± 322.6 | 1.6 ± 0.4 | 5.2 ± 4.6 |
| 1 m blue | 4.1 ± 6.6 | 821.1 ± 168.1 | 1891.9 ± 135.9 | 0.7 ± 0.2 | 6.3 ± 2.3 |
| 3 m green | 277.1 ± 35.7 | 2063.1 ± 99.0 | 1962.9 ± 162.2 | 2.4 ± 0.3 | 5.0 ± 1.9 |
| 2 m green | 77.1 ± 19.4 | 2063.1 ± 48.2 | 1962.9 ± 93.2 | 1.2 ± 0.3 | 5.1 ± 3.4 |
| 1 m green | 13.1 ± 6.9 | 420.1 ± 88.8 | 237.9 ± 51.7 | 0.8 ± 0.2 | 5.8 ± 2.3 |
| 23 m | 256.1 ± 248.2 | 7.1 ± 14.9 | 9.9 ± 10.7 | 2.5 ± 1.1 | 6.9 ± 12.8 |
| 11 m | 2099.1 ± 976.8 | 160.6 ± 511.5 | 70.9 ± 543.7 | 4.9 ± 5.6 | 6.9 ± 13.3 |
| Pollens (complete) | |||||
| Timothy grass (Pheleum pratense) | 113.1 ± 773.7 | 124.1 ± 852.7 | 198.4 ± 847.7 | 3.9 ± 7.2 | 21.3 ± 19.7 |
| Nettle (Urtica dioica) | 1658.6 ± 814.4 | 2063.1 ± 912.6 | 1962.9 ± 833.8 | 18.8 ± 8.5 | 24.6 ± 15.1 |
| Sheep Sorrel (Rumex acetosella) | 551.1 ± 923.4 | 222.1 ± 872.1 | 233.9 ± 846.0 | 5.9 ± 8.0 | 21.5 ± 17.6 |
| Ryegrass (Lolium perenne) | 713.1 ± 905.4 | 277.1 ± 894.7 | 408.9 ± 867.0 | 5.8 ± 8.3 | 23.1 ± 17.2 |
| White Poplar (Populus alba) | 422.1 ± 921.3 | 151.6 ± 843.0 | 203.9 ± 838.2 | 4.9 ± 7.6 | 20.6 ± 16.3 |
| European white birch (Betula pendula) | 209.1 ± 798.1 | 231.1 ± 856.2 | 351.9 ± 881.8 | 6.0 ± 8.7 | 25.5 ± 17.1 |
| Olive (Olea europaea) | |||||
| English Oak (Quercus robur) | 233.1 ± 844.4 | 86.6 ± 844.2 | 199.9 ± 749.3 | 3.0 ± 7.8 | 11.8 ± 18.2 |
| Fungal spores | |||||
| Cladosporium herbarium | 106.1 ± 222.7 | 36.1 ± 124.3 | 32.9 ± 162.8 | 2.6 ± 2.9 | 26.6 ± 21.1 |
| Alternaria alternata | 25.1 ± 237.2 | 30.1 ± 284.2 | 95.9 ± 416.8 | 1.3 ± 4.4 | 13.2 ± 20.5 |
| Bacteria | |||||
| Esherishia coli(E. coli) (unwashed) | 30.1 ± 92.5 | 34.1 ± 93.2 | 45.9 ± 150.9 | 0.9 ± 0.5 | 5.9 ± 2.5 |
| Bacillus atrophaeus (BG) (washed) | 28.1 ± 21.8 | 3.1 ± 7.4 | 6.9 ± 15.7 | 0.7 ± 0.4 | 5.9 ± 2.6 |
| Bacillus atrophaeus (BG) (unwashed) | 11.1 ± 39.0 | 28.1 ± 80.8 | 36.9 ± 113.8 | 0.9 ± 0.5 | 5.7 ± 2.4 |
| Bacillus thuringensis (BT) (washed) | 24.1 ± 19.4 | 4.1 ± 9.1 | 5.9 ± 12.6 | 1.0 ± 0.4 | 5.8 ± 2.7 |
| Bacillus thuringensis (BT) (unwashed) | 6.1 ± 48.8 | 47.1 ± 116.8 | 41.9 ± 127.8 | 0.9 ± 0.5 | 5.8 ± 2.4 |
| Others | |||||
| Arizona test dust (ATD) | 4.1 ± 491.3 | 13.1 ± 326.3 | 44.9 ± 387.7 | 2.2 ± 5.4 | 15.1 ± 16.2 |
| Phosphate-buffered saline (PBS) | 2.6 ± 202.7 | 7.1 ± 113.3 | 14.9 ± 128.3 | 1.3 ± 0.9 | 7.5 ± 8.4 |
| Salt (NaCl) | 0.1 ± 2.2 | 4.1 ± 42.7 | 16.9 ± 25.5 | 1.3 ± 1.2 | 7.8 ± 11.7 |
| Mixture | 14.1 ± 100.7 | 31.1 ± 166.4 | 43.9 ± 137.4 | 0.9 ± 1.1 | 5.8 ± 3.8 |
| WIBS-NEO | FL1 | FL2 | FL3 | SIZE (m) | AF |
|---|---|---|---|---|---|
| PSLs | |||||
| 3 m | 949,644.1 ± 1,124,295.0 | 18,072.8 ± 91,258.2 | 6,957.8 ± 44,726.5 | 1.4 ± 1.5 | 4.4 ± 15.1 |
| 2 m blue | 285,459.1 ± 959,733.5 | 14,661,725.8 ± 4,806,105.5 | 31,613,101.8 ± 9,115,405.0 | 2.4 ± 0.9 | 4.1 ± 7.3 |
| 1 m blue | 111,156.1 ± 2,240,213.6 | 1,228,305.8 ± 366,340.7 | 2,837,835.8 ± 808,337.6 | 1.1 ± 0.2 | 5.9 ± 3.5 |
| 3 m green | 2,802,084.1 ± 720,841.7 | 24,688,733.8 ± 3,960,189.6 | 7,907,798.8 ± 1,398,652.5 | 3.6 ± 0.5 | 4.1 ± 4.5 |
| 2 m green | 343,505.1 ± 305,872.0 | 9,877,597.8 ± 1,677,830.1 | 3,186,115.8 ± 603,016.7 | 2.2 ± 0.5 | 4.0 ± 5.0 |
| 1 m green | 84,830.1 ± 3,416,291.3 | 931,367.8 ± 249,472.0 | 272,009.8 ± 88,497.9 | 1.1 ± 0.2 | 4.5 ± 2.4 |
| 23 m | 8,751,868.1 ± 8,404,649.6 | 26,167.8 ± 78,786.4 | 23,797.8 ± 61,137.1 | 3.7 ± 1.4 | 5.8 ± 10.7 |
| 11 m | 508,279,832.1 ± 205,576,657.8 | 223,525.8 ± 1,396,336.4 | 32,137.8 ± 1,747,277.7 | 12.0 ± 3.3 | 5.8 ± 9.2 |
| Pollen | |||||
| Timothy grass (Pheleum pratense) | 1,624,865.1 ± 83,236,645.9 | 205,293.8 ± 32,108,331.7 | 170,059.8 ± 5,338,021.6 | 2.8 ± 5.2 | 13.8 ± 13.9 |
| Nettle (Urtica dioica) | 86,296,748.1 ± 52,817,534.7 | 69,711,965.8 ± 27,976,180.0 | 5,169,435.8 ± 4,291,601.9 | 16.5 ± 5.0 | 15.7 ± 13.1 |
| Sheep Sorrel (Rumex acetosella) | 9,652,980.1 ± 131,420,881.2 | 406,330.8 ± 35,407,618.0 | 610,315.8 ± 3,402,720.3 | 7.3 ± 6.4 | 15.4 ± 14.4 |
| Ryegrass (Lolium perenne) | 41,175,556.1 ± 144,108,873.7 | 57,473,117.8 ± 49,485,478.9 | 9,724,077.8 ± 13,311,509.2 | 15.6 ± 8.4 | 16.8 ± 16.5 |
| White Poplar (Populus alba) | 48,761,290.1 ± 149,138,701.7 | 1,426,153.8 ± 38,419,997.0 | 3,307,277.8 ± 7,368,437.3 | 11.1 ± 7.0 | 15.0 ± 17.8 |
| European white birch (Betula pendula) | 64,080,214.1 ± 95,033,743.9 | 5,709,261.8 ± 36,638,859.2 | 3,614,843.8 ± 3,228,645.1 | 14.1 ± 7.2 | 17.0 ± 8.8 |
| Olive (Olea europaea) | - | - | - | - | - |
| English Oak (Quercus robur) | 27,085,424.1 ± 219,717,163.9 | 3,872,943.8 ± 40,315,365.6 | 365,373.8 ± 4,596,854.0 | 3.6 ± 8.1 | 7.5 ± 8.6 |
| Fungal spores | |||||
| Cladosporium herbarium | 2,532,342.1 ± 14,374,629.0 | 96,313.8 ± 2,247,766.3 | 48,979.8 ± 1,469,403.2 | 2.7 ± 3.0 | 14.5 ± 11.9 |
| Alternaria alternata | 878,026.1 ± 12,134,583.7 | 218,701.8 ± 3,286,191.6 | 163,927.8 ± 1,704,039.2 | 1.9 ± 4.2 | 13.4 ± 11.7 |
| Bacteria | |||||
| Esherishia coli(E. coli) (unwashed) | 1,395,704.1 ± 6,104,494.3 | 96,149.8 ± 400,039.7 | 67,455.8 ± 319,082.8 | 1.3 ± 0.6 | 6.6 ± 7.4 |
| Bacillus atrophaeus (BG) (washed) | 577,000.1 ± 947,362.0 | 13,085.8 ± 93,188.2 | 17,012.8 ± 88,797.1 | 1.0 ± 0.4 | 6.2 ± 4.3 |
| Bacillus atrophaeus (BG) (unwashed) | 818,344.1 ± 2,189,716.3 | 77,999.8 ± 361,893.6 | 58,953.8 ± 238,205.3 | 1.4 ± 0.6 | 6.1 ± 5.1 |
| Bacillus thuringensis (BT) (washed) | 565,390.1 ± 1,050,059.1 | 16,845.8 ± 107,775.4 | 33,609.8 ± 136,412.2 | 1.3 ± 0.6 | 7.7 ± 5.5 |
| Bacillus thuringensis (BT) (unwashed) | 462,951.1 ± 1,598,749.9 | 82,577.8 ± 336,102.9 | 59,279.8 ± 182,927.4 | 1.2 ± 0.6 | 6.7 ± 5.0 |
| Others | |||||
| Arizona test dust (ATD) | 678,868.1 ± 41,490,476.3 | 150,925.8 ± 15,352,219.3 | 69,581.8 ± 1,838,486.0 | 2.3 ± 3.7 | 19.0 ± 12.8 |
| Phosphate-buffered saline (PBS) | 213,514.1 ± 1,075,310.4 | 66,635.8 ± 197,479.3 | 73,015.8 ± 287,865.8 | 0.9 ± 1.2 | 13.0 ± 15.6 |
| Salt (NaCl) | 456,328.1 ± 5,554,114.6 | 188,697.8 ± 5,669,790.9 | 195,485.8 ± 2,063,667.0 | 1.3 ± 3.1 | 9.5 ± 15.3 |
| Mixture | 709,936.1 ± 3,444,655.9 | 114,789.8 ± 1,745,612.0 | 161,639.8 ± 437,517.3 | 1.3 ± 0.9 | 5.5 ± 4.4 |
| Material | FL1 | FL2 | FL3 | Size (m) | Af |
|---|---|---|---|---|---|
| Nettle >13 m (WIBS4M) | 1713.6 ± 790.4 | 2063.1 ± 890.3 | 1962.9 ± 815.1 | 19.4 ± 8.0 | 25.4 ± 14.7 |
| Nettle <13 m (WIBS4M) | 178.1 ± 646.2 | 83.1 ± 510.4 | 101.9 ± 542.2 | 4.0 ± 3.2 | 24.9 ± 19.2 |
| Nettle >7.5 m (NEO) | 89,245,846.1 ± 49,965,614.8 | 71,252,061.8 ± 23,212,541.3 | 5,267,931.8 ± 4,203,910.0 | 16.9 ± 3.2 | 15.8 ± 13.2 |
| Nettle <7.5 m (NEO) | 2,528,544.1 ± 21,418,950.3 | 206,275.8 ± 13,599,589.4 | 182,731.8 ± 3,204,266.1 | 2.5 ± 1.9 | 14.7 ± 12.7 |
| Sample | Instrument | FL1 | FL2 | FL3 | Size (m) | Shape |
|---|---|---|---|---|---|---|
| Complete | WIBS-4D | 17.3 ± 35.3 | 214.4 ± 572.6 | 209.6 ± 515.7 | 0.9 ± 0.3 | 12.7 ± 6.4 |
| Split | WIBS-4D | 21.3 ± 36.5 | 188.4 ± 356.4 | 174.6 ± 382.1 | 0.9 ± 0.3 | 12.5 ± 6.1 |
| ATD | WIBS-4D | 1.3 ± 241.8 | 52.4 ± 343.1 | 96.6 ± 413.1 | 1.9 ± 2.4 | 23.6 ± 16.1 |
| Complete | WIBS-4M | 14.1 ± 100.7 | 31.1 ± 166.4 | 43.9 ± 137.4 | 0.9 ± 1.1 | 5.8 ± 3.8 |
| Split | WIBS-4M | 17.1 ± 50.5 | 26.1 ± 62.9 | 30.9 ± 88.2 | 0.9 ± 0.9 | 5.8 ± 3.2 |
| ATD | WIBS-4M | 4.1 ± 491.3 | 13.1 ± 326.3 | 44.9 ± 387.7 | 2.2 ± 5.4 | 15.1 ± 16.2 |
| Complete | WIBS-NEO | 709,936.1 ± 3,444,655.9 | 114,789.8 ± 1,745,612.0 | 161,639.8 ± 437,517.3 | 1.3 ± 0.9 | 5.5 ± 4.4 |
| Split | WIBS-NEO | 708,300.1 ± 1,302,565.7 | 65,899.8 ± 232,694.7 | 51,677.8 ± 371,714.5 | 1.3 ± 0.7 | 5.7 ± 4.0 |
| ATD | WIBS-NEO | 678,868.1 ± 41,490,476.3 | 150,925.8 ± 15,352,219.3 | 69,581.8 ± 1,838,486.0 | 2.3 ± 3.7 | 19.0 ± 12.8 |
| Instrument | XE1 | XE2 | XE3 | XE4 | XE5 | XE6 | XE7 | XE8 | Size (m) | Shape | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Complete | MBS-M | 6.0 ± 112.8 | 18.0 ± 63.1 | 44.0 ± 121.3 | 47.2 ± 135.1 | 61.1 ± 162.9 | 46.4 ± 372.5 | 33.7 ± 163.2 | 19.6 ± 133.0 | 1.3 ± 1.0 | 12.2 ± 7.8 |
| Split | MBS-M | 5.0 ± 116.5 | 20.1 ± 90.6 | 53.7 ± 135.3 | 49.7 ± 136.0 | 48.4 ± 142.4 | 33.1 ± 112.3 | 15.1 ± 115.7 | 10.2 ± 276.4 | 1.4 ± 0.8 | 12.0 ± 6.5 |
| ATD | MBS-M | 4.2 ± 6.0 | 8.5 ± 31.0 | 12.7 ± 141.7 | 11.5 ± 160.3 | 10.9 ± 75.3 | 9.1 ± 26.1 | 8.8 ± 29.0 | 5.8 ± 9.5 | 2.6 ± 1.8 | 18.5 ± 12.2 |
| Complete | MBS-D | 18.4 ± 29.1 | 43.4 ± 80.4 | 113.2 ± 223.4 | 136.6 ± 281.7 | 181.6 ± 418.2 | 142.3 ± 621.8 | 67.7 ± 364.7 | 35.6 ± 96.4 | 1.3 ± 0.8 | 16.1 ± 9.2 |
| Split | MBS-D | 15.7 ± 37.7 | 51.4 ± 116.1 | 116.8 ± 222.8 | 142.2 ± 288.1 | 153.5 ± 299.1 | 106.3 ± 209.4 | 38.8 ± 113.8 | 18.5 ± 39.8 | 1.3 ± 0.6 | 17.2 ± 9.9 |
| ATD | MBS-D | 13.8 ± 91.9 | 31.9 ± 117.6 | 38.1 ± 274.0 | 31.5 ± 191.6 | 34.1 ± 229.5 | 19.2 ± 354.5 | 18.2 ± 226.0 | 25.2 ± 296.3 | 2.0 ± 1.8 | 19.9 ± 12.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Forde, E.; Gallagher, M.; Walker, M.; Foot, V.; Attwood, A.; Granger, G.; Sarda-Estève, R.; Stanley, W.; Kaye, P.; Topping, D. Intercomparison of Multiple UV-LIF Spectrometers Using the Aerosol Challenge Simulator. Atmosphere 2019, 10, 797. https://doi.org/10.3390/atmos10120797
Forde E, Gallagher M, Walker M, Foot V, Attwood A, Granger G, Sarda-Estève R, Stanley W, Kaye P, Topping D. Intercomparison of Multiple UV-LIF Spectrometers Using the Aerosol Challenge Simulator. Atmosphere. 2019; 10(12):797. https://doi.org/10.3390/atmos10120797
Chicago/Turabian StyleForde, Elizabeth, Martin Gallagher, Maurice Walker, Virginia Foot, Alexis Attwood, Gary Granger, Roland Sarda-Estève, Warren Stanley, Paul Kaye, and David Topping. 2019. "Intercomparison of Multiple UV-LIF Spectrometers Using the Aerosol Challenge Simulator" Atmosphere 10, no. 12: 797. https://doi.org/10.3390/atmos10120797
APA StyleForde, E., Gallagher, M., Walker, M., Foot, V., Attwood, A., Granger, G., Sarda-Estève, R., Stanley, W., Kaye, P., & Topping, D. (2019). Intercomparison of Multiple UV-LIF Spectrometers Using the Aerosol Challenge Simulator. Atmosphere, 10(12), 797. https://doi.org/10.3390/atmos10120797






