Slc4 Gene Family in Spotted Sea Bass (Lateolabrax maculatus): Structure, Evolution, and Expression Profiling in Response to Alkalinity Stress and Salinity Changes
Abstract
1. Introduction
2. Materials and methods
2.1. Ethics Statement
2.2. Genome-Wide Identification of Spotted Sea Bass Slc4 Genes
2.3. Phylogenetic and Syntenic Analysis
2.4. Gene Structure and Domain Analysis
2.5. Selective Pressure Analysis
2.6. Carbonate Alkalinity Exposure Experiment
2.7. Freshwater and Seawater Transition Experiments
2.8. RNA Extraction and Quantitative Real-Time PCR (qPCR) Analysis
2.9. Statistical Analysis
3. Results
3.1. Characterizations of Slc4 Gene Sequences in Spotted Sea Bass
3.2. Phylogenetic, Syntenic, and Chromosomal Distribution Analysis of slc4 Genes
3.3. Gene Structure Analysis of Spotted Sea Bass slc4 Genes
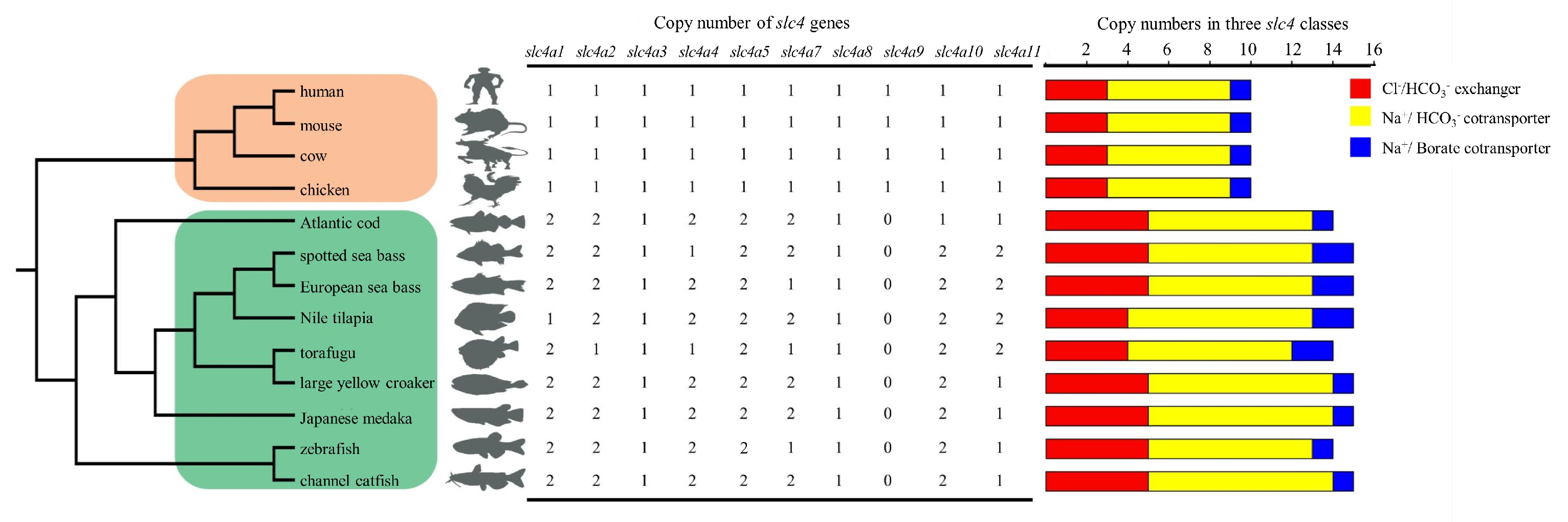
3.4. Copy Number Comparison of slc4 Genes in Selected Vertebrates
3.5. Selective Pressure Analysis
3.6. Expression Patterns of slc4 Genes in Response to Alkalinity Stress
3.7. Branchial Expression Patterns of slc4 Genes During FW and SW Acclimation
4. Discussion
4.1. Features of slc4 Genes in Spotted Sea Bass
4.2. Function Studies of slc4 Genes of Spotted Sea Bass in Response to Salinity and Alkalinity Stresses
4.2.1. Anion Exchanger Proteins (AEs)
4.2.2. Na+-Coupled Bicarbonate Transporters (NCBTs)
4.2.3. Na+-Coupled Borate Transporter (NaBC)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Romero, M.F. Molecular pathophysiology of SLC4 bicarbonate transporters. Curr. Opin. Nephrol. Hypertens. 2005, 14, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Romero, M.F.; Chen, A.P.; Parker, M.D.; Boron, W.F. The SLC4 family of bicarbonate (HCO3−) transporters. Mol. Aspects Med. 2013, 34, 159–182. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yang, J.; Chen, L.M. Structure and function of SLC4 family HCO3- transporters. Front. Physiol. 2015, 6, 355. [Google Scholar] [CrossRef] [PubMed]
- Alper, S.L. Molecular physiology and genetics of Na+-independent SLC4 anion exchangers. J. Exp. Biol. 2009, 212, 1672–1683. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Xu, J.Y.; Wang, D.K.; Wang, L.; Chen, L.M. Cloning and identification of two novel NBCe1 splice variants from mouse reproductive tract tissues: A comparative study of NCBT genes. Genomics 2011, 98, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Tsuganezawa, H.; Kobayashi, K.; Iyori, M.; Araki, T.; Koizumi, A.; Watanabe, S.; Kaneko, A.; Fukao, T.; Monkawa, T.; Yoshida, T.; et al. A new member of the HCO3- transporter superfamily is an apical anion exchanger of β-intercalated cells in the kidney. J. Biol. Chem. 2001, 276, 8180–8189. [Google Scholar] [CrossRef]
- Pena-Munzenmayer, G.; George, A.T.; Shull, G.E.; Melvin, J.E.; Catalan, M.A. Catalan, Ae4 (Slc4a9) is an electroneutral monovalent cation-dependent Cl-/HCO3− exchanger. J. Gen. Physiol. 2016, 147, 423–436. [Google Scholar] [CrossRef]
- Catalán, M.A.; José Viveros, J.; Fernandez, F.; Flores, L.; Brauchi, S.; Peña-Münzenmayer, G. Exploring the transport mechanism of the human AE4 (SLC4A9) Cl-/HCO3− exchanger. Biophys. J. 2018, 114, 330a. [Google Scholar] [CrossRef]
- Pushkin, A.; Kurtz, I. SLC4 base (HCO3−, CO32−) transporters: Classification, function, structure, genetic diseases, and knockout models. Am. J. Physiol. Renal Physiol. 2006, 290, 580–599. [Google Scholar] [CrossRef]
- Damkier, H.H.; Nielsen, S.; Praetorius, J. Molecular expression of SLC4-derived Na+-dependent anion transporters in selected human tissues. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 293, 2136–2146. [Google Scholar] [CrossRef]
- Parker, M.D. Mouse models of SLC4-linked disorders of HCO3−-transporter dysfunction. Am. J. Physiol. Cell Physiol. 2018, 314, 569–588. [Google Scholar] [CrossRef] [PubMed]
- Shmukler, B.E.; Kurschat, C.E.; Ackermann, G.E.; Jiang, L.; Zhou, Y.; Barut, B.; Stuart-Tilley, A.K.; Zhao, J.; Zon, L.I.; Drummond, I.A.; et al. Zebrafish slc4a2/ae2 anion exchanger: cDNA cloning, mapping, functional characterization, and localization. Am. J. Physiol. Renal Physiol. 2005, 289, 835–849. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Jarabo, I.; Gregorio, S.F.; Gaetano, P.; Trischitta, F.; Fuentes, J. High rates of intestinal bicarbonate secretion in seawater tilapia (Oreochromis mossambicus). Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2017, 207, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Hwang, P.P. Ion uptake and acid secretion in zebrafish (Danio rerio). J. Exp. Biol. 2009, 212, 1745–1752. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.M.; Tang, R.; Dou, X.J.; Tao, R.; Sun, X.W.; Liang, L.Q. Transcriptome and expression profiling analysis of Leuciscus waleckii: An exploration of the alkali-adapted mechanisms of a freshwater teleost. Mol. Biosyst. 2014, 10, 491–504. [Google Scholar] [CrossRef] [PubMed]
- Gregorio, S.F.; Carvalho, E.S.; Encarnacao, S.; Wilson, J.M.; Power, D.M.; Canario, A.V.; Fuentes, J. Adaptation to different salinities exposes functional specialization in the intestine of the sea bream (Sparus aurata L.). J. Exp. Biol. 2013, 216, 470–479. [Google Scholar] [CrossRef] [PubMed]
- Kurita, Y.; Nakada, T.; Kato, A.; Doi, H.; Mistry, A.C.; Chang, M.H.; Romero, M.F.; Hirose, S. Identification of intestinal bicarbonate transporters involved in formation of carbonate precipitates to stimulate water absorption in marine teleost fish. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, 1402–1412. [Google Scholar] [CrossRef]
- An, H.S.; Kim, H.Y.; Kim, J.B.; Chang, D.S.; Park, K.D.; Lee, J.W.; Myeong, J.I.; An, C.M. Genetic characterization of hatchery populations of Korean spotted sea bass (Lateolabrax maculatus) using multiplex polymerase chain reaction assays. Genet. Mol. Res. 2014, 13, 6701–6715. [Google Scholar] [CrossRef]
- Tian, Y.; Wen, H.; Qi, X.; Mao, X.; Shi, Z.; Li, J.; He, F.; Yang, W.; Zhang, X.; Li, Y. Analysis of apolipoprotein multigene family in spotted sea bass (Lateolabrax maculatus) and their expression profiles in response to Vibrio harveyi infection. Fish Shellfish Immunol. 2019, 92, 111–118. [Google Scholar] [CrossRef]
- Tian, Y.; Wen, H.; Qi, X.; Zhang, X.; Li, Y. Identification of mapk gene family in Lateolabrax maculatus and their expression profiles in response to hypoxia and salinity challenges. Gene 2019, 684, 20–29. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Yang, L.; Jiang, H.; Wang, Y.; Lei, Y.; Chen, J.; Sun, N.; Lv, W.; Wang, C.; Near, T.J.; He, S. Expansion of vomeronasal receptor genes (OlfC) in the evolution of fright reaction in Ostariophysan fishes. Commun. Biol. 2019, 2, 235. [Google Scholar] [CrossRef] [PubMed]
- Anisimova, M.; Kosiol, C. Investigating protein-coding sequence evolution with probabilistic codon substitution models. Mol. Biol. Evol. 2009, 26, 255–271. [Google Scholar] [CrossRef]
- Wang, H.; Wen, H.; Li, Y.; Zhang, K.; Liu, Y. Evaluation of potential reference genes for quantitative RT-PCR analysis in spotted sea bass (Lateolabrax maculatus) under normal and salinity stress conditions. PeerJ 2018, 6, e5631. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.C.; Yan, J.J.; Cruz, S.A.; Horng, J.L.; Hwang, P.P. Anion exchanger 1b, but not sodium-bicarbonate cotransporter 1b, plays a role in transport functions of zebrafish H+-ATPase-rich cells. Am. J. Physiol. Cell Physiol. 2011, 300, 295–307. [Google Scholar] [CrossRef] [PubMed]
- Boron, W.F.; Chen, L.; Parker, M.D. Modular structure of sodium-coupled bicarbonate transporters. J. Exp. Biol. 2009, 212, 1697–1706. [Google Scholar] [CrossRef]
- Zhang, D.; Kiyatkin, A.; Bolin, J.T.; Low, P.S. Crystallographic structure and functional interpretation of the cytoplasmic domain of erythrocyte membrane band 3. Blood 2000, 96, 2925–2933. [Google Scholar] [CrossRef] [PubMed]
- Meyer, A.; Van de Peer, Y. From 2R to 3R: Evidence for a fish-specific genome duplication (FSGD). Bioessays 2005, 27, 937–945. [Google Scholar] [CrossRef] [PubMed]
- Glasauer, S.M.; Neuhauss, S.C. Whole-genome duplication in teleost fishes and its evolutionary consequences. Mol. Genet. Genom. 2014, 289, 1045–1060. [Google Scholar] [CrossRef]
- Li, Y.; Liu, S.; Qin, Z.; Yao, J.; Jiang, C.; Song, L.; Dunham, R.; Liu, Z. The serpin superfamily in channel catfish: Identification, phylogenetic analysis and expression profiling in mucosal tissues after bacterial infections. Dev. Comp. Immunol. 2015, 49, 267–277. [Google Scholar] [CrossRef]
- Fan, H.; Zhou, Y.; Wen, H.; Zhang, X.; Zhang, K.; Qi, X.; Xu, P.; Li, Y. Genome-wide identification and characterization of glucose transporter (glut) genes in spotted sea bass (Lateolabrax maculatus) and their regulated hepatic expression during short-term starvation. Comp. Biochem. Physiol. Part D Genom. Proteom. 2019, 30, 217–229. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.D.; Wang, G.D.; Irwin, D.M.; Zhang, Y.P. A profound role for the expansion of trypsin-like serine protease family in the evolution of hematophagy in mosquito. Mol. Biol. Evol. 2009, 26, 2333–2341. [Google Scholar] [CrossRef] [PubMed]
- Sironi, M.; Cagliani, R.; Forni, D.; Clerici, M. Evolutionary insights into host-pathogen interactions from mammalian sequence data. Nat. Rev. Genet. 2015, 16, 224–236. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.H.; Piermarini, P.M.; Choe, K.P. The multifunctional fish gill: Dominant site of gas exchange, osmoregulation, acid-base regulation, and excretion of nitrogenous waste. Physiol. Rev. 2005, 85, 97–177. [Google Scholar] [CrossRef]
- Liu, S.T.; Horng, J.L.; Chen, P.Y.; Hwang, P.P.; Lin, L.Y. Salt secretion is linked to acid-base regulation of ionocytes in seawater-acclimated medaka: New insights into the salt-secreting mechanism. Sci. Rep. 2016, 6, 31433. [Google Scholar] [CrossRef]
- Wilson, J.M.; Whiteley, N.M.; Randall, D.J. Ionoregulatory changes in the gill epithelia of coho salmon during seawater acclimation. Physiol. Biochem. Zool. 2002, 75, 237–249. [Google Scholar] [CrossRef]
- Tang, C.H.; Lee, T.H. The novel correlation of carbonic anhydrase II and anion exchanger 1 in gills of the spotted green pufferfish, Tetraodon nigrovirids. J. Exp. Zool. A Ecol. Genet. Physiol. 2007, 307, 411–418. [Google Scholar] [CrossRef]
- Shmukler, B.E.; Reimold, F.R.; Heneghan, J.F.; Chen, C.; Zhao, T.; Paw, B.H.; Alper, S.L. Molecular cloning and functional characterization of zebrafish Slc4a3/Ae3 anion exchanger. Pflugers Arch. 2014, 466, 1605–1618. [Google Scholar] [CrossRef]
- Aalkjaer, C.; Boedtkjer, E.; Choi, I.; Lee, S. Cation-coupled bicarbonate transporters. Compr. Physiol. 2014, 4, 1605–1637. [Google Scholar] [CrossRef]
- Ishiguro, H.; Steward, M.C.; Lindsay, A.R.; Case, R.M. Accumulation of intracellular HCO3− by Na+-HCO3− cotransport in interlobular ducts from guinea-pig pancreas. J. Physiol. 1996, 495, 169–178. [Google Scholar] [CrossRef]
- Romero, M.F.; Fulton, C.M.; Boron, W.F. The SLC4 family of HCO3- transporters. Pflugers Arch. 2004, 447, 495–509. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.H.; Plata, C.; Kurita, Y.; Kato, A.; Hirose, S.; Romero, M.F. Euryhaline pufferfish NBCe1 differs from nonmarine species NBCe1 physiology. Am. J. Physiol. Cell Physiol. 2012, 302, 1083–1095. [Google Scholar] [CrossRef]
- Perry, S.F.; Furimsky, M.; Bayaa, M.; Georgalis, T.; Shahsavarani, A.; Nickerson, J.G.; Moon, T.W. Integrated responses of Na+/HCO3- cotransporters and V-type H+-ATPases in the fish gill and kidney during respiratory acidosis. Biochim. Biophys. Acta 2003, 1618, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Alves, A.; Gregorio, S.F.; Egger, R.C.; Fuentes, J. Molecular and functional regionalization of bicarbonate secretion cascade in the intestine of the European sea bass (Dicentrarchus labrax). Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2019, 233, 53–64. [Google Scholar] [CrossRef]
- Gregorio, S.F.; Ruiz-Jarabo, I.; Carvalho, E.M.; Fuentes, J. Increased intestinal carbonate precipitate abundance in the sea bream (Sparus aurata L.) in response to ocean acidification. PLoS ONE 2019, 14, e0218473. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Ma, D.; Zhu, H.; Liu, Z.; Gao, F. Transcriptomic response to three osmotic stresses in gills of hybrid tilapia (Oreochromis mossambicus female x O. urolepis hornorum male). BMC Genom. 2020, 21, 110. [Google Scholar] [CrossRef]
- Felder, R.A.; Jose, P.A.; Xu, P.; Gildea, J.J. The renal sodium bicarbonate cotransporter NBCe2: Is it a major contributor to sodium and pH homeostasis? Curr. Hypertens. Rep. 2016, 18, 71. [Google Scholar] [CrossRef]
- Park, M.; Li, Q.; Shcheynikov, N.; Zeng, W.; Muallem, S. NaBC1 is a ubiquitous electrogenic Na+ -coupled borate transporter essential for cellular boron homeostasis and cell growth and proliferation. Mol. Cell 2004, 16, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Ogando, D.G.; Bonanno, J.A.; Obukhov, A.G. Human SLC4A11 is a novel NH3/H+ co-transporter. J. Biol. Chem. 2015, 290, 16894–16905. [Google Scholar] [CrossRef] [PubMed]







| Classification | Gene Name | CDS Length (bp) | Predicted Protein Length (Amino Acid) | Molecular Weight (kDa) | Isoelectric Point (pI) | Accession Number of NCBI |
|---|---|---|---|---|---|---|
| Cl−/HCO3− exchanger | slc4a1a | 2766 | 921 | 103.29 | 5.31 | MN909728 |
| slc4a1b | 2523 | 840 | 94.05 | 8.68 | MN909729 | |
| slc4a2a | 3417 | 1138 | 126.82 | 5.99 | MN909730 | |
| slc4a2b | 3678 | 1225 | 137.11 | 6.09 | MN909731 | |
| slc4a3 | 3702 | 1234 | 139.71 | 5.85 | MN909732 | |
| Na+/HCO3− cotransporter | slc4a4 | 3213 | 1070 | 120.51 | 6.23 | MN909733 |
| slc4a5a | 2586 | 861 | 96.92 | 5.80 | MN909734 | |
| slc4a5b | 3423 | 1140 | 129.03 | 6.63 | MN909735 | |
| slc4a7a | 2799 | 932 | 103.90 | 6.82 | MN909736 | |
| slc4a7b | 3015 | 1004 | 111.26 | 6.66 | MN909737 | |
| slc4a8 | 3363 | 1120 | 125.37 | 6.03 | MN909738 | |
| slc4a10a | 3261 | 1086 | 122.58 | 6.18 | MN909739 | |
| slc4a10b | 4023 | 1340 | 149.04 | 5.83 | MN909740 | |
| Na+/Borate cotransporter | slc4a11a | 2529 | 842 | 94.91 | 6.11 | MN909741 |
| slc4a11b | 2316 | 771 | 86.84 | 6.73 | MN909742 |
| Model | np | LnL | Models Compared | 2△LnL | df | p | Positively Selected Sites |
|---|---|---|---|---|---|---|---|
| Null model A—slc4a1a | 43 | −27215.89837 | MA vs. NMA | 0.00000 | 1 | 1.00000 | |
| Model A—slc4a1a | 44 | −27215.89837 | |||||
| Null model A—slc4a1b | 43 | −27204.09798 | MA vs. NMA | 42.52516 | 1 | 0.00000 | 138S * 141T ** 148N ** 152T ** 470V ** 471N ** |
| Model A—slc4a1b | 44 | −27182.83540 | |||||
| Null model A—slc4a2a | 43 | −16901.01428 | MA vs. NMA | 2.0 × 10−5 | 1 | 0.99643 | |
| Model A—slc4a2a | 44 | −16901.01429 | |||||
| Null model A—slc4a2b | 43 | −16900.85034 | MA vs. NMA | 0.00000 | 1 | 1.00000 | |
| Model A—slc4a2b | 44 | −16900.85034 | |||||
| Null model A—slc4a3 | 27 | −22702.55961 | MA vs. NMA | 2.6 × 10−4 | 1 | 0.4791 | |
| Model A—slc4a3 | 28 | −22702.55948 | |||||
| Null model A—slc4a4 | 43 | −27709.75538 | MA vs. NMA | 0.00000 | 1 | 1.00000 | |
| Model A—slc4a4 | 44 | −27709.75538 | |||||
| Null model A—slc4a5a | 45 | −22011.18613 | MA vs. NMA | 0.08432 | 1 | 0.77153 | |
| Model A—slc4a5a | 46 | −22011.14397 | |||||
| Null model A—slc4a5b | 45 | −22013.51479 | MA vs. NMA | 0.00000 | 1 | 1.00000 | |
| Model A—slc4a5b | 46 | −22013.51479 | |||||
| Null model A—slc4a7a | 39 | −3957.77188 | MA vs. NMA | 0.00000 | 1 | 1.00000 | |
| Model A—slc4a7a | 40 | −3957.77188 | |||||
| Null model A—slc4a7b | 39 | −3957.65280 | MA vs. NMA | 0.23816 | 1 | 0.62554 | |
| Model A—slc4a7b | 40 | −3957.77188 | |||||
| Null model A—slc4a8 | 27 | −7554.82350 | MA vs. NMA | 7.70544 | 1 | 0.00551 | 33L * 34S ** |
| Model A—slc4a8 | 28 | −7550.97078 | |||||
| Null model A—slc4a10a | 43 | −27358.78420 | MA vs. NMA | 0.28100 | 1 | 0.59605 | |
| Model A—slc4a10a | 44 | −27358.64370 | |||||
| Null model A—slc4a10b | 43 | −27346.85330 | MA vs. NMA | 31.10466 | 1 | 0.00000 | 200S * 214S ** 215A ** 216T ** 349R * |
| Model A—slc4a10b | 44 | −27331.30097 | |||||
| Null model A—slc4a11a | 35 | −6835.90728 | MA vs. NMA | 0.00000 | 1 | 1.00000 | |
| Model A—slc4a11a | 36 | −6835.90728 | |||||
| Null model A—slc4a11b | 35 | −6835.90728 | MA vs. NMA | 0.00000 | 1 | 1.00000 | |
| Model A—slc4a11b | 36 | −6835.90728 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.-Y.; Tian, Y.; Wen, H.-S.; Yu, P.; Liu, Y.; Qi, X.; Gao, Z.-C.; Zhang, K.-Q.; Li, Y. Slc4 Gene Family in Spotted Sea Bass (Lateolabrax maculatus): Structure, Evolution, and Expression Profiling in Response to Alkalinity Stress and Salinity Changes. Genes 2020, 11, 1271. https://doi.org/10.3390/genes11111271
Wang L-Y, Tian Y, Wen H-S, Yu P, Liu Y, Qi X, Gao Z-C, Zhang K-Q, Li Y. Slc4 Gene Family in Spotted Sea Bass (Lateolabrax maculatus): Structure, Evolution, and Expression Profiling in Response to Alkalinity Stress and Salinity Changes. Genes. 2020; 11(11):1271. https://doi.org/10.3390/genes11111271
Chicago/Turabian StyleWang, Ling-Yu, Yuan Tian, Hai-Shen Wen, Peng Yu, Yang Liu, Xin Qi, Zhi-Chao Gao, Kai-Qiang Zhang, and Yun Li. 2020. "Slc4 Gene Family in Spotted Sea Bass (Lateolabrax maculatus): Structure, Evolution, and Expression Profiling in Response to Alkalinity Stress and Salinity Changes" Genes 11, no. 11: 1271. https://doi.org/10.3390/genes11111271
APA StyleWang, L.-Y., Tian, Y., Wen, H.-S., Yu, P., Liu, Y., Qi, X., Gao, Z.-C., Zhang, K.-Q., & Li, Y. (2020). Slc4 Gene Family in Spotted Sea Bass (Lateolabrax maculatus): Structure, Evolution, and Expression Profiling in Response to Alkalinity Stress and Salinity Changes. Genes, 11(11), 1271. https://doi.org/10.3390/genes11111271





