Ovarian Transcriptomic Analyses in the Urban Human Health Pest, the Western Black Widow Spider
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection and RNA Extraction
2.2. Illumina Library Prep and Sequencing
2.3. De Novo Assembly of Ovary Transcriptome
2.4. Functional Annotation
3. Results
3.1. Sequencing and De Novo Assembly
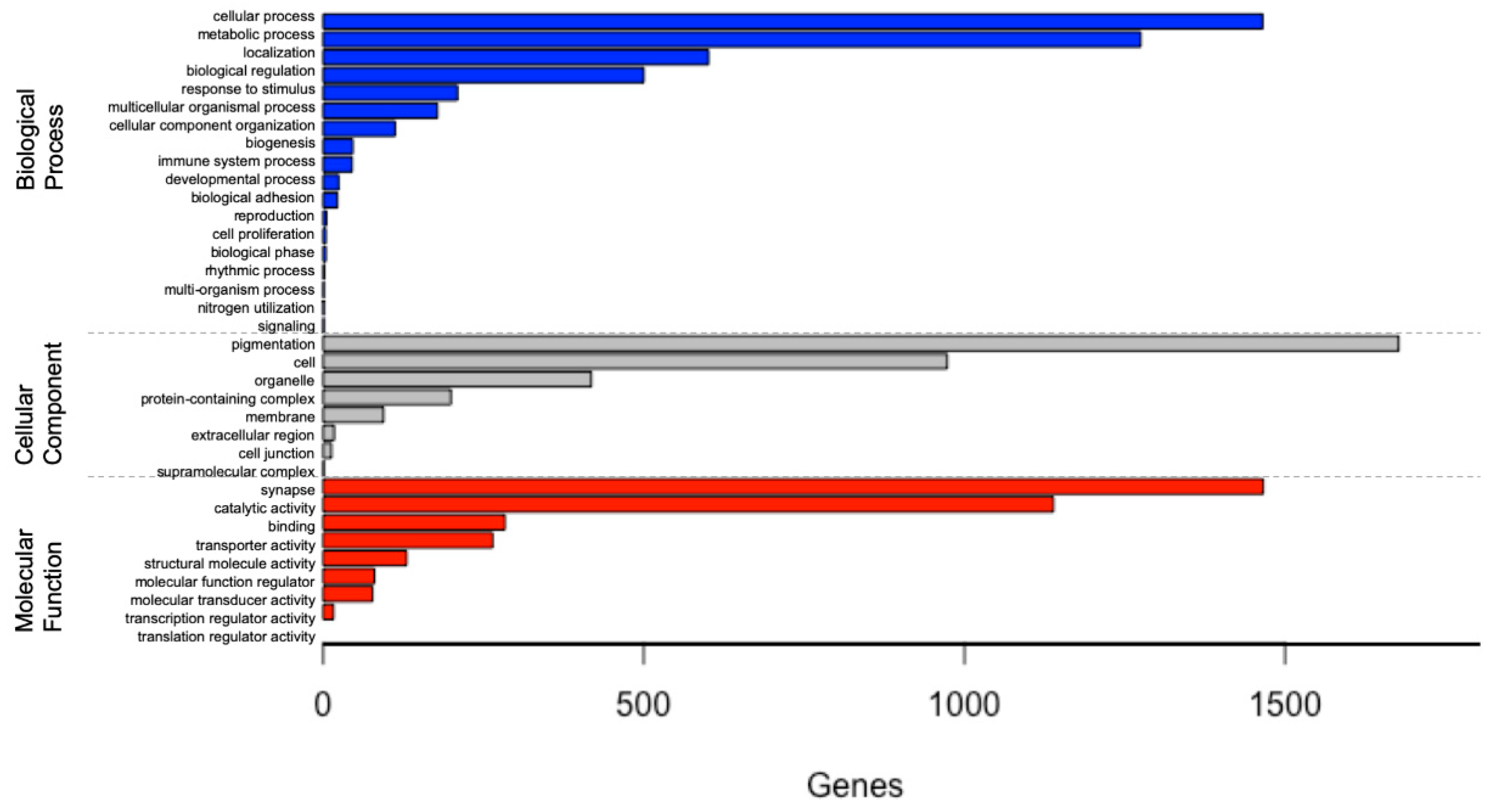
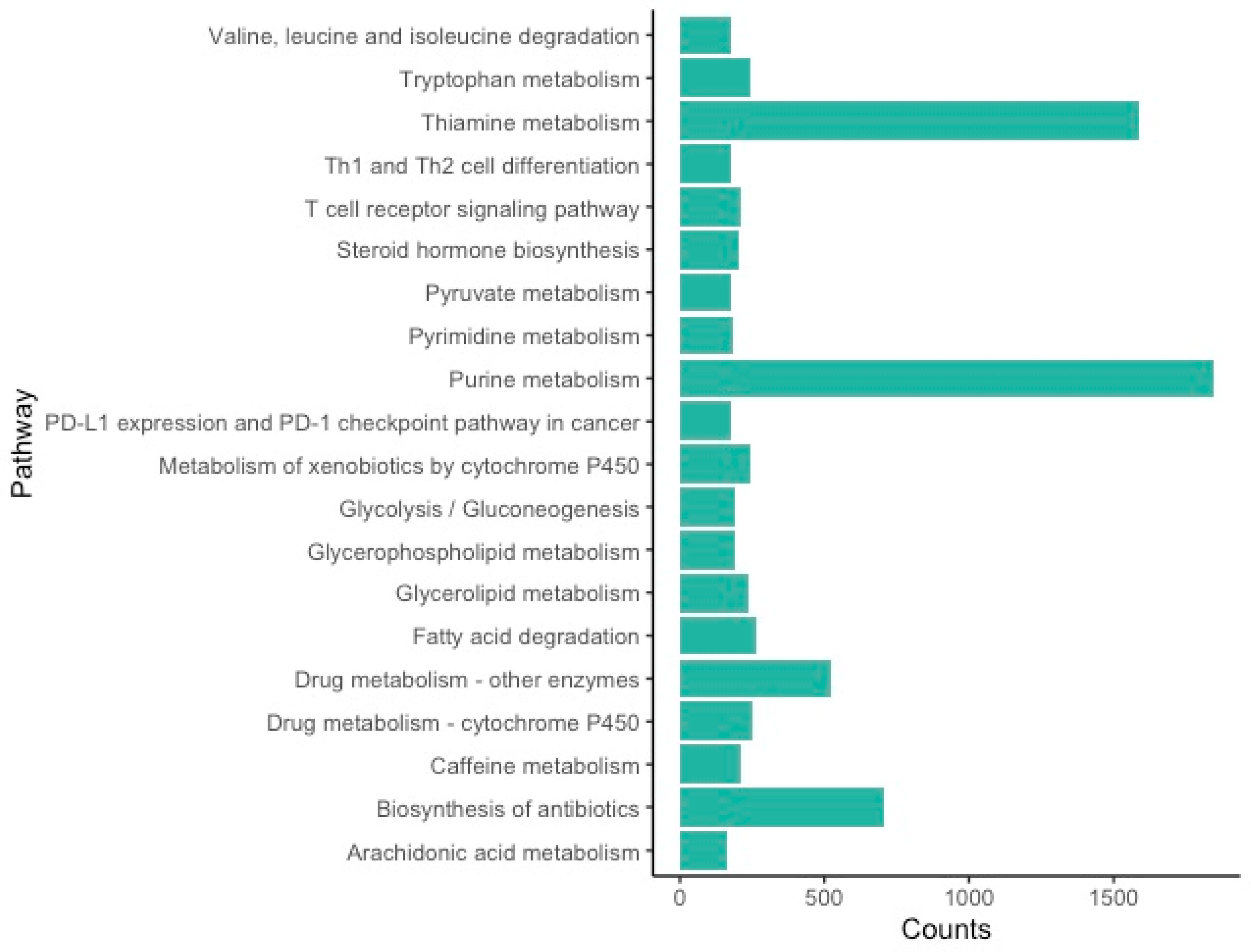
3.2. Functional Annotation and Classification
3.3. Spider Ovary Genes of Interest
4. Discussion
4.1. Western Black Widow Spider Ovarian Gene and Pathway Categories
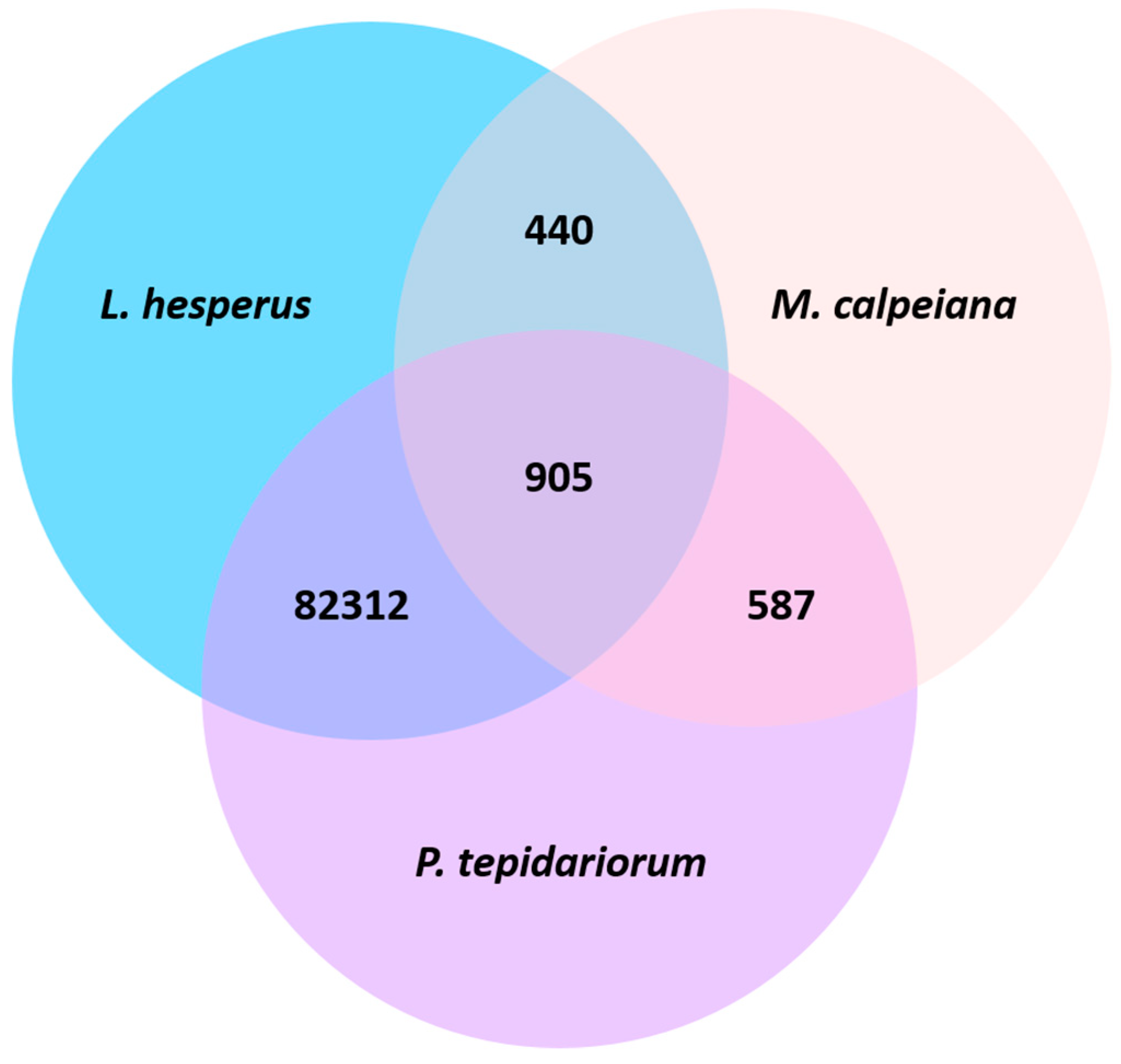
4.2. Species Comparisons of Spider Ovarian Transcriptomes
4.3. Western Black Widow Spider Genes Associated with Fecundity
4.4. Fecundity and Human Pest Adaptation to Urban Environments
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lewontin, R.C. The Effects of Population Density and Composition on Viability in Drosophila melanogaster. Evolution 1955, 9, 27–41. [Google Scholar] [CrossRef]
- Dawson, P.S. Life History Strategy and Evolutionary History of Tribolium Flour Beetles. Evolution 1977, 31, 226–229. [Google Scholar] [CrossRef] [PubMed]
- Scriber, J.M.; Slansky, F. The nutritional ecology of immature insects. Annu. Rev. Entomol. 1981, 26, 183–211. [Google Scholar] [CrossRef]
- Leather, S.R. Size, Reproductive Potential and Fecundity in Insects: Things aren’t as Simple as They Seem. Oikos 1988, 51, 386–389. [Google Scholar] [CrossRef]
- Barnes, A.I.; Wigby, S.; Boone, J.M.; Partridge, L.; Chapman, T. Feeding, fecundity and lifespan in female Drosophila melanogaster. Proc. R. Soc. B Biol. Sci. 2008, 275, 1675–1683. [Google Scholar] [CrossRef]
- Lister, B.C.; Garcia, A. Climate-driven declines in arthropod abundance restructure a rainforest food web. Proc. Natl. Acad. Sci. USA 2018, 115, E10397–E10406. [Google Scholar] [CrossRef]
- Li, B.; Predel, R.; Neupert, S.; Hauser, F.; Tanaka, Y.; Cazzamali, G.; Williamson, M.; Arakane, Y.; Verleyen, P.; Schoofs, L.; et al. Genomics, transcriptomics, and peptidomics of neuropeptides and protein hormones in the red flour beetle Tribolium castaneum. Genome Res. 2008, 18, 113–122. [Google Scholar] [CrossRef]
- Tan, A.; Palli, S.R. Edysone receptor isoforms play distinct roles in controlling molting and metamorphosis in the red flour beetle, Tribolium castaneum. Mol. Cell. Endocrinol. 2008, 291, 42–49. [Google Scholar] [CrossRef]
- Shukla, J.N.; Palli, S.R. Doublesex target genes in the red flour beetle, Tribolium castaneum. Sci. Rep. 2012, 2, 948. [Google Scholar] [CrossRef]
- Van Emden, H.F.; Harrington, R. Aphids as Crop Pests; Cabi: Wallingford, UK, 2017. [Google Scholar]
- Constantino, R. The pest termites of South America: Taxonomy, distribution and status. J. Appl. Entomol. 2002, 126, 355–365. [Google Scholar] [CrossRef]
- Kumar, D.; Pardeshi, M. Biodiversity of termites in agro-ecosystem and relation between their niche breadth and pest status. J. Entomol. 2011, 8, 250–258. [Google Scholar] [CrossRef]
- Evans, T.A.; Iqbal, N. Termite (order Blattodea, infraorder Isoptera) baiting 20 years after commercial release. Pest Manag. Sci. 2015, 71, 897–906. [Google Scholar] [CrossRef] [PubMed]
- Byrne, K.; Nichols, R.A. Culex pipiens in London underground tunnels: Differentiation between surface and subterranean populations. Heredity 1999, 82, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Vargo, E.L.; Crissman, J.R.; Booth, W.; Santangelo, R.G.; Mukha, D.V.; Schal, C. Hierarchical genetic analysis of German cockroach (Blattella germanica) populations from within buildings to across continents. PLoS ONE 2014, 9, e102321. [Google Scholar] [CrossRef][Green Version]
- Booth, W.; Balvín, O.; Vargo, E.L.; Vilímová, J.; Schal, C. Host association drives genetic divergence in the bed bug, Cimex lectularius. Mol. Ecol. 2015, 24, 980–992. [Google Scholar] [CrossRef]
- Rivkin, L.R.; Santangelo, J.S.; Alberti, M.; Aronson, M.F.J.; de Keyzer, C.W.; Diamond, S.E.; Fortin, M.J.; Frazee, L.J.; Gorton, A.J.; Hendry, A.P.; et al. A roadmap for urban evolutionary ecology. Evol. Appl. 2019, 12, 384–398. [Google Scholar] [CrossRef]
- Adrian, A.B.; Comeron, J.M. The Drosophila early ovarian transcriptome provides insight to the molecular causes of recombination rate variation across genomes. BMC Genom. 2013, 14, 794. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, J.; Han, T.; Liu, T.; Wang, C.; Xiao, J.; Mu, C.; Li, R.; Yu, F.; Shi, H. Ovarian transcriptome analysis of Portunus trituberculatus provides insights into genes expressed during phase III and IV development. PLoS ONE 2015, 10, e0138862. [Google Scholar] [CrossRef]
- Uengwetwanit, T.; Ponza, P.; Sangsrakru, D.; Wichadakul, D.; Ingsriswang, S.; Leelatanawit, R.; Klinbunga, S.; Tangphatsornruang, S.; Karoonuthaisiri, N. Transcriptome-based discovery of pathways and genes related to reproduction of the black tiger shrimp (Penaeus monodon). Mar. Genom. 2018, 37, 69–73. [Google Scholar] [CrossRef]
- Blair, R.B. Land use and avian species diversity along an urban gradient. Ecol. Appl. 1996, 6, 506–519. [Google Scholar] [CrossRef]
- McKinney, M.L.; Lockwood, J.L. Biotic homogenization: A few winners replacing many losers in the next mass extinction. Trends Ecol. Evol. 1999, 14, 450–453. [Google Scholar] [CrossRef]
- McIntyre, N.E.; Rango, J.; Fagan, W.F.; Faeth, S.H. Ground arthropod community structure in a heterogeneous urban environment. Landsc. Urban Plan. 2001, 52, 257–274. [Google Scholar] [CrossRef]
- Kark, S.; Iwaniuk, A.; Schalimtzek, A.; Banker, E. Living in the city: Can anyone become an ‘urban exploiter’? J. Biogeogr. 2007, 34, 638–651. [Google Scholar] [CrossRef]
- Johnson, J.C.; Trubl, P.J.; Miles, L.S. Black widows in an urban desert: City-living compromises spider fecundity and egg investment despite urban prey abundance. Am. Midl. Nat. 2012, 168, 333–341. [Google Scholar] [CrossRef]
- Johnson, J.C.; Miles, L.S.; Trubl, P.J.; Hagenmaier, A. Maternal effects on egg investment and offspring performance in black widow spiders. Anim. Behav. 2014, 91, 67–73. [Google Scholar] [CrossRef]
- Trubl, P.; Gburek, T.; Miles, L.; Johnson, J.C. Black widow spiders in an urban desert: Population variation in an arthropod pest across metropolitan Phoenix, AZ. Urban Ecosyst. 2012, 15, 599–609. [Google Scholar] [CrossRef]
- Miles, L.S.; Johnson, J.C.; Dyer, R.J.; Verrelli, B.C. Urbanization as a facilitator of gene flow in a human health pest. Mol. Ecol. 2018, 27, 3219–3230. [Google Scholar] [CrossRef]
- Miles, L.S.; Rivkin, L.R.; Johnson, M.T.J.; Munshi-South, J.; Verrelli, B.C. Gene flow and genetic drift in urban environments. Mol. Ecol. 2019, 28, 4138–4151. [Google Scholar] [CrossRef]
- Miles, L.S.; Dyer, R.J.; Verrelli, B.C. Urban hubs of connectivity: Contrasting patterns of gene flow within and among cities in the western black widow spider. Proc. R. Soc. B Biol. Sci. 2018, 285, 20181224. [Google Scholar] [CrossRef]
- Mattila, T.M.; Bechsgaard, J.S.; Hansen, T.T.; Schierup, M.H.; Bilde, T. Orthologous genes identified by transcriptome sequencing in the spider genus Stegodyphus. BMC Genom. 2012, 13, 70. [Google Scholar] [CrossRef]
- Posnien, N.; Zeng, V.; Schwager, E.E.; Pechmann, M.; Hilbrant, M.; Keefe, J.D.; Damen, W.G.; Prpic, N.M.; McGregor, A.P.; Extavour, C.G. A comprehensive reference transcriptome resource for the common house spider Parasteatoda tepidariorum. PLoS ONE 2014, 9, e104885. [Google Scholar] [CrossRef] [PubMed]
- Sanggaard, K.W.; Bechsgaard, J.S.; Fang, X.; Duan, J.; Dyrlund, T.F.; Gupta, V.; Jiang, X.; Cheng, L.; Fan, D.; Feng, Y.; et al. Spider genomes provide insight into composition and evolution of venom and silk. Nat. Commun. 2014, 5, 3765. [Google Scholar] [CrossRef] [PubMed]
- Clarke, T.H.; Garb, J.E.; Hayashi, C.Y.; Haney, R.A.; Lancaster, A.K.; Corbett, S.; Ayoub, N.A. Multi-tissue transcriptomics of the black widow spider reveals expansions, co-options, and functional processes of the silk gland gene toolkit. BMC Genom. 2014, 15, 365. [Google Scholar] [CrossRef] [PubMed]
- Clarke, T.H.; Garb, J.E.; Hayashi, C.Y.; Arensburger, P.; Ayoub, N.A. Spider transcriptomes identify ancient large-scale gene duplication event potentially important in silk gland evolution. Genome Biol. Evol. 2015, 7, 1856–1870. [Google Scholar] [CrossRef] [PubMed]
- Clarke, T.H.; Garb, J.E.; Haney, R.A.; Chaw, R.C.; Hayashi, C.Y.; Ayoub, N.A. Evolutionary shifts in gene expression decoupled from gene duplication across functionally distinct spider silk glands. Sci. Rep. 2017, 7, 8393. [Google Scholar] [CrossRef] [PubMed]
- Haney, R.A.; Clarke, T.H.; Gadgil, R.; Fitzpatrick, R.; Hayashi, C.Y.; Ayoub, N.A.; Garb, J.E. Effects of gene duplication, positive selection, and shifts in gene expression on the evolution of the venom gland transcriptome in widow spiders. Genome Biol. Evol. 2016, 8, 228–242. [Google Scholar] [CrossRef] [PubMed]
- Garb, J.E.; Haney, R.A.; Schwager, E.E.; Gregorič, M.; Kuntner, M.; Agnarsson, I.; Blackledge, T.A. The transcriptome of Darwin’s bark spider silk glands predicts proteins contributing to dragline silk toughness. Commun. Biol. 2019, 2, 275. [Google Scholar] [CrossRef]
- Frías-López, C.; Almeida, F.C.; Guirao-Rico, S.; Vizueta, J.; Sánchez-Gracia, A.; Arnedo, M.A.; Rozas, J. Comparative analysis of tissue-specific transcriptomes in the funnel-web spider Macrothele calpeiana (Araneae, Hexathelidae). PeerJ 2015, 3, e1064. [Google Scholar] [CrossRef]
- Haney, R.A.; Garb, J.E.; Forsyth, F.S.; Matte, T. Alternative transcription at venom genes and its role as a complementary mechanism for the generation of venom complexity. Front. Ecol. Evol. 2019, 7, 85. [Google Scholar] [CrossRef]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644. [Google Scholar] [CrossRef]
- Haas, B.J.; Papanicolaou, A.; Yassour, M.; Grabherr, M.; Blood, P.D.; Bowden, J.; Couger, M.B.; Eccles, D.; Li, B.; Lieber, M.; et al. De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat. Protoc. 2013, 8, 1494. [Google Scholar] [CrossRef] [PubMed]
- Simão, F.A.; Waterhouse, R.M.; Ioannidis, P.; Kriventseva, E.V.; Zdobnov, E.M. BUSCO: Assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 2015, 31, 3210–3212. [Google Scholar] [CrossRef]
- Waterhouse, R.M.; Seppey, M.; Simão, F.A.; Manni, M.; Ioannidis, P.; Klioutchnikov, G.; Kriventseva, E.V.; Zdobnov, E.M. BUSCO applications from quality assessments to gene prediction and phylogenomics. Mol. Biol. Evol. 2017, 35, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Patro, R.; Duggal, G.; Love, M.I.; Irizarry, R.A.; Kingsford, C. Salmon provides fast and bias-aware quantification of transcript expression. Nat. Methods 2017, 14, 417. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Madden, T.L.; Schäffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef] [PubMed]
- Mi, H.; Huang, X.; Muruganujan, A.; Tang, H.; Mills, C.; Kang, D.; Thomas, P.D. PANTHER version 11: Expanded annotation data from Gene Ontology and Reactome pathways, and data analysis tool enhancements. Nucleic Acids Res. 2016, 45, D183–D189. [Google Scholar] [CrossRef]
- Bryant, D.M.; Johnson, K.; DiTommaso, T.; Tickle, T.; Couger, M.B.; Payzin-Dogru, D.; Lee, T.J.; Leigh, N.D.; Kuo, T.H.; Davis, F.G.; et al. A tissue-mapped axolotl de novo transcriptome enables identification of limb regeneration factors. Cell Rep. 2017, 18, 762–776. [Google Scholar] [CrossRef]
- Götz, S.; García-Gómez, J.M.; Terol, J.; Williams, T.D.; Nagaraj, S.H.; Nueda, M.J.; Robles, M.; Talón, M.; Dopazo, J.; Conesa, A. High-throughput functional annotation and data mining with the Blast2GO suite. Nucleic Acids Res. 2008, 36, 3420–3435. [Google Scholar] [CrossRef]
- Downs, S.M.; Eppig, J.J. Induction of mouse oocyte maturation in vivo by perturbants of purine metabolism. Biol. Reprod. 1987, 36, 431–437. [Google Scholar] [CrossRef]
- Downs, S.M. Purine control of mouse oocyte maturation: Evidence that nonmetabolized hypoxanthine maintains meiotic arrest. Mol. Reprod. Dev. 1993, 35, 82–94. [Google Scholar] [CrossRef]
- Gillett, J.D. Variation in the time of release of the ovarian development hormone in Aedes aegypti. Nature 1957, 180, 656–657. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, A.; Nakamura, T.; Shibata, K. Effects of mild and severe vitamin B1 deficiencies on the meiotic maturation of mice oocytes. Nutr. Metab. Insights 2017, 10. [Google Scholar] [CrossRef]
- Haney, R.A.; Ayoub, N.; Clarke, T.H.; Hayashi, C.Y.; Garb, J.E. Dramatic expansion of the black widow toxin arsenal uncovered by multi-tissue transcriptomics and venom proteomics. BMC Genom. 2014, 15, 366. [Google Scholar] [CrossRef] [PubMed]
- Mikhailov, A.T.; Torrado, M. Carboxylesterases moonlight in the male reproductive tract: A functional shift pivotal for male fertility. Front. Biosci. 2000, 5, E53–E62. [Google Scholar] [PubMed]
- Wen, J.; Xie, J.; Liu, S.; Gui, J. Differential expression and characterization analysis of a new gene with WD domains in fish oogenesis. Sci. China C Life Sci. 2001, 44, 541–553. [Google Scholar] [CrossRef] [PubMed]
- Danshina, P.V.; Geyer, C.B.; Dai, Q.; Goulding, E.H.; Willis, W.D.; Kitto, G.B.; McCarrey, J.R.; Eddy, E.M.; O’Brien, D.A. Phosphoglycerate kinase 2 (PGK2) is essential for sperm function and male fertility in mice. Biol. Reprod. 2010, 82, 136–145. [Google Scholar] [CrossRef]
- Hashiyama, K.; Hayashi, Y.; Kobayashi, S. Drosophila sex lethal gene initiates female development in germline progenitors. Science 2011, 333, 885–888. [Google Scholar] [CrossRef]
- Van Doren, M. Determining sexual identity. Science 2011, 333, 829–830. [Google Scholar] [CrossRef]
- Wei, L.; Cheng, D.; Li, D.; Meng, M.; Peng, L.; Tang, L.; Pan, M.; Xiang, Z.; Xia, Q.; Lu, C. Identification and characterization of Sox genes in the silkworm, Bombyx mori. Mol. Biol. Rep. 2011, 38, 3573–3584. [Google Scholar] [CrossRef]
- Jiang, T.; Hou, C.C.; She, Z.Y.; Yang, W.X. The SOX gene family: Function and regulation in testis determination and male fertility maintenance. Mol. Biol. Rep. 2013, 40, 2187–2194. [Google Scholar] [CrossRef]
- Zhai, Y.; Zhang, J.; Sun, Z.; Dong, X.; He, Y.; Kang, K.; Liu, Z.; Zhang, W. Proteomic and transcriptomic analyses of fecundity in the brown planthopper Nilaparvata lugens (Stal). J. Proteome Res. 2013, 12, 5199–5212. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y.; Sun, Z.; Zhang, J.; Kang, K.; Chen, J.; Zhang, W. Activation of the TOR signalling pathway by glutamine regulates insect fecundity. Sci. Rep. 2015, 5, 10694. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.X.; Zhai, Y.F.; Zhang, J.Q.; Kang, K.; Cai, J.H.; Fu, Y.; Qiu, J.Q.; Shen, J.W.; Zhang, W.Q. The genetic basis of population fecundity prediction across multiple field populations of Nilaparvata lugens. Mol. Ecol. 2015, 24, 771–784. [Google Scholar] [CrossRef] [PubMed]
- Qiu, J.; He, Y.; Zhang, J.; Kang, K.; Li, T.; Zhang, W. Discovery and functional identification of fecundity-related genes in the brown planthopper by large-scale RNA interference. Insect Mol. Biol. 2016, 25, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Tufail, M.; Takeda, M. Molecular characteristics of insect vitellogenins. J. Insect Physiol. 2008, 54, 1447–1458. [Google Scholar] [CrossRef]
- Bateman, A.; Bennett, H.P. Granulins: The structure and function of an emerging family of growth factors. J. Endocrinol. 1998, 158, 145–151. [Google Scholar] [CrossRef]
- Li, L.T.; Zhu, Y.B.; Ma, J.F.; Li, Z.Y.; Dong, Z.P. An analysis of the Athetis lepigone transcriptome from four developmental stages. PLoS ONE 2013, 8, e73911. [Google Scholar] [CrossRef]
- Do Nascimento, A.R.; Fresia, P.; Cônsoli, F.L. Comparative transcriptome analysis of lufenuron-resistant and susceptible strains of Spodoptera frugiperda (Lepidoptera: Noctuidae). BMC Genom. 2015, 16, 985. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, B.; Dong, S.; Cao, D.; Dong, J.; Walker, W.B.; Liu, Y.; Wang, G. Antennal transcriptome analysis and comparison of chemosensory gene families in two closely related noctuidae moths, Helicoverpa armigera and H. assulta. PLoS ONE 2015, 10, e0117054. [Google Scholar] [CrossRef]
- Rendón-Anaya, M.; Delaye, L.; Possani, L.D.; Herrera-Estrella, A. Global transcriptome analysis of the scorpion Centruroides noxius: New toxin families and evolutionary insights from an ancestral scorpion species. PLoS ONE 2012, 7, e43331. [Google Scholar] [CrossRef]
- Vizueta, J.; Frías-López, C.; Macías-Hernández, N.; Arnedo, M.A.; Sánchez-Gracia, A.; Rozas, J. Evolution of chemosensory gene families in arthropods: Insight from the first inclusive comparative transcriptome analysis across spider appendages. Genome Biol. Evol. 2017, 9, 178–196. [Google Scholar] [CrossRef] [PubMed]
- Bian, H.X.; Chen, D.B.; Zheng, X.X.; Ma, H.F.; Li, Y.P.; Li, Q.; Xia, R.X.; Wang, H.; Jiang, Y.R.; Liu, Y.Q.; et al. Transcriptomic analysis of the prothoracic gland from two lepidopteran insects, domesticated silkmoth Bombyx mori and wild silkmoth Antheraea pernyi. Sci. Rep. 2019, 9, 5313. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Weng, S.; Chen, Y.; Yu, X.; Lü, L.; Zhang, H.; He, J.; Xu, X. Analysis of Litopenaeus vannamei transcriptome using the next-generation DNA sequencing technique. PLoS ONE 2012, 7, e47442. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Lee, B.Y.; Won, E.J.; Han, J.; Hwang, D.S.; Park, H.G.; Lee, J.S. Identification of xenobiotic biodegradation and metabolism-related genes in the copepod Tigriopus japonicus whole transcriptome analysis. Mar. Genom. 2015, 24, 207–208. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Zhang, B.; Cai, S.; Zeng, X.; Ye, Q.; Mao, X.; Zhang, S.; Zeng, X.; Ye, C.; Qiao, S. Metabolic disorder of amino acids, fatty acids and purines reflects the decreases in oocyte quality and potential in sows. J. Proteom. 2019, 200, 134–143. [Google Scholar] [CrossRef]
- Sirard, M.A.; First, N.L. In vitro inhibition of oocyte nuclear maturation in the bovine. Biol. Reprod. 1988, 39, 229–234. [Google Scholar] [CrossRef]
- Carta, M.C.; Mattana, A.; Camici, M.; Allegrini, S.; Tozzi, M.G.; Sgarrella, F. Catabolism of exogenous deoxyinosine in cultured epithelial amniotic cells. Biochim. Biophys. Acta (BBA) Gen. Subj. 2001, 1528, 74–80. [Google Scholar] [CrossRef]
- Rio, R.V.; Attardo, G.M.; Weiss, B.L. Grandeur alliances: Symbiont metabolic integration and obligate arthropod hematophagy. Trends Parasitol. 2016, 32, 739–749. [Google Scholar] [CrossRef]
- Hormiga, G.; Griswold, C.E. Systematics, phylogeny, and evolution of orb-weaving spiders. Annu. Rev. Entomol. 2014, 59, 487–512. [Google Scholar] [CrossRef]
- Gendreau, K.L.; Haney, R.A.; Schwager, E.E.; Wierschin, T.; Stanke, M.; Richards, S.; Garb, J.E. House spider genome uncovers evolutionary shifts in the diversity and expression of black widow venom proteins associated with extreme toxicity. BMC Genom. 2017, 18, 178. [Google Scholar] [CrossRef]
- Garrison, N.L.; Rodriguez, J.; Agnarsson, I.; Coddington, J.A.; Griswold, C.E.; Hamilton, C.A.; Hedin, M.; Kocot, K.M.; Ledford, J.M.; Bond, J.E. Spider phylogenomics: Untangling the Spider Tree of Life. PeerJ 2016, 4, e1719. [Google Scholar] [CrossRef] [PubMed]
- Sawadro, M.; Bednarek, A.; Babczyńska, A. The current state of knowledge on the neuroactive compounds that affect the development, mating and reproduction of spiders (Araneae) compared to insects. Invertebr. Neurosci. 2017, 2017, 17. [Google Scholar] [CrossRef]
- Nolte, R.T.; Conlin, R.M.; Harrison, S.C.; Brown, R.S. Differing roles for zinc fingers in DNA recognition: Structure of a six-finger transcription factor IIIA complex. Proc. Natl. Acad. Sci. USA 1998, 95, 2938–2943. [Google Scholar] [CrossRef] [PubMed]
- Emerson, R.O.; Thomas, J.H. Adaptive evolution in zinc finger transcription factors. PLoS Genet. 2009, 5, e1000325. [Google Scholar] [CrossRef] [PubMed]
- Thompson, D.M.; Khalil, S.M.S.; Jeffers, L.A.; Sonenshine, D.E.; Mitchel, R.D.; Osgood, C.J.; Michael, R.R. Sequence and the developmental and tissue-specific regulation of the first complete vitellogenin messenger RNA from ticks responsible for heme sequestration. Insect Biochem. Mol. Biol. 2007, 37, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wu, X.; Liu, Z.; Zheng, H.; Cheng, Y. Insights into hepatopancreatic functions for nutrition metabolism and ovarian development in the crab Portunus trituberculatus: Gene discovery in the comparative transcriptome of different hepatopancreas stages. PLoS ONE 2014, 9, e84921. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Wang, L.; Wu, H.; Cao, Y.; Xiao, R.; Lai, X.; Liu, J.; Yi, J.; Zhang, G. Molecular characterization and expression of vitellogenin genes from the wolf spider Pardosa pseudoannulata (Araneae: Lycosidae). Physiol. Entomol. 2018, 43, 295–305. [Google Scholar] [CrossRef]
- Bednarek, A.W.; Sawadro, M.K.; Nicewicz, Ł.; Babczyńska, A.I. Vitellogenins in the spider Parasteatoda tepidariorum–expression profile and putative hormonal regulation of vitellogenesis. BMC Dev. Biol. 2019, 19, 4. [Google Scholar] [CrossRef]
- Jean André, C.R. The ultrastructure of the vitelline body in the oocyte of the spider Tegenaria parietina. J. Biophys. Biochem. Cytol. 1957, 3, 977–1000. [Google Scholar] [CrossRef]
- Sotello, J.R.; Oscar, T.-C. Electron microscope study of the vitelline body of some spider oocytes. J. Biophys. Biochem. Cytol. 1957, 3, 301–317. [Google Scholar] [CrossRef]
- Osaki, H. Electron microscope studies on developing oocytes of the spider Plexippus paykulli. Annot. Zool. Jpn. 1972, 45, 187–200. [Google Scholar]
- Trabalon, M.; Bautz, A.M.; Moriniere, M.; Porcheron, P. Ovarian development and correlated changes in hemolymphatic ecdysteroid levels in two spiders, Coelotes terrestris and Tegenaria domestics (araneae, agelenidae). Gen. Comp. Endocrinol. 1992, 88, 128–136. [Google Scholar] [CrossRef]
- Yan, S.; Wang, X. Recent advances in research on widow spider venoms and toxins. Toxins 2015, 7, 5055–5067. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.C.; Urcuyo, J.; Moen, C.; Stevens, D.R. Urban heat island conditions experienced by the Western black widow spider (Latrodectus hesperus): Extreme heat slows development but results in behavioral accommodations. PLoS ONE 2019, 14, e0220153. [Google Scholar] [CrossRef] [PubMed]
- Trubl, P.; Johnson, J.C. Ecological stoichiometry of the black widow spider and its prey from desert, urban and laboratory populations. J. Arid Environ. 2019, 163, 18–25. [Google Scholar] [CrossRef]
| Gene. | Function | Count * |
|---|---|---|
| Vitellogenin | oocyte development | 181 |
| Vitelline membrane outer layer protein | oocyte development | 174 |
| Estrogen | ovary development | 3397 |
| 3 beta-hydroxysteroid dehydrogenase (3-β-HSD) | ovary development | 405 |
| Mandibular organ-inhibiting hormone (MOIH) | ovary development | 100 |
| Lutropin-choriogonadotropic hormone receptor (LSHR) | ovary development | 30 |
| Follicle-stimulating hormone receptor (FSHR) | ovary development | 5 |
| Zinc-Finger protein (ZFP) | vitellogenin regulation | 315938 |
| phosphoglycerate kinase | vitellogenin regulation | 5424 |
| carboxylesterase | vitellogenin regulation | 2610 |
| C-terminal-binding protein (CtBP) | vitellogenin regulation | 584 |
| protein geranylgeranyl transferase | vitellogenin regulation | 297 |
| fizzy (fzy) | vitellogenin regulation | 244 |
| SRY related HMG-Box-11 (SOX-11) | vitellogenin regulation | 145 |
| Sex-lethal (Sxl) | vitellogenin regulation | 5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miles, L.S.; Ayoub, N.A.; Garb, J.E.; Haney, R.A.; Verrelli, B.C. Ovarian Transcriptomic Analyses in the Urban Human Health Pest, the Western Black Widow Spider. Genes 2020, 11, 87. https://doi.org/10.3390/genes11010087
Miles LS, Ayoub NA, Garb JE, Haney RA, Verrelli BC. Ovarian Transcriptomic Analyses in the Urban Human Health Pest, the Western Black Widow Spider. Genes. 2020; 11(1):87. https://doi.org/10.3390/genes11010087
Chicago/Turabian StyleMiles, Lindsay S., Nadia A. Ayoub, Jessica E. Garb, Robert A. Haney, and Brian C. Verrelli. 2020. "Ovarian Transcriptomic Analyses in the Urban Human Health Pest, the Western Black Widow Spider" Genes 11, no. 1: 87. https://doi.org/10.3390/genes11010087
APA StyleMiles, L. S., Ayoub, N. A., Garb, J. E., Haney, R. A., & Verrelli, B. C. (2020). Ovarian Transcriptomic Analyses in the Urban Human Health Pest, the Western Black Widow Spider. Genes, 11(1), 87. https://doi.org/10.3390/genes11010087






