Free-Water Imaging in White and Gray Matter in Parkinson’s Disease
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Acquisition of MRI Data
2.3. Diffusion MRI Preprocessing
2.4. Voxel-Wise Analysis
2.4.1. TBSS
2.4.2. GBSS
2.5. Region-of-Interest Analysis
2.6. Voxel-Based Morphometry
2.7. Statistical Analysis
3. Results
3.1. WM Alterations
3.1.1. TBSS
3.1.2. ROI
3.2. GM Alterations
3.2.1. GBSS
3.2.2. ROI
3.3. WM and GM Volumetry
3.4. Correlation Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Recasens, A.; Dehay, B. Alpha-synuclein spreading in Parkinson’s disease. Front. Neuroanat. 2014, 8, 159. [Google Scholar] [CrossRef] [PubMed]
- Duffy, M.F.; Collier, T.J.; Patterson, J.R.; Kemp, C.J.; Luk, K.C.; Tansey, M.G.; Paumier, K.L.; Kanaan, N.M.; Fischer, D.L.; Polinski, N.K.; et al. Lewy body-like alpha-synuclein inclusions trigger reactive microgliosis prior to nigral degeneration. J. Neuroinflamm. 2018, 15, 129. [Google Scholar] [CrossRef] [PubMed]
- Atkinson-Clement, C.; Pinto, S.; Eusebio, A.; Coulon, O. Diffusion tensor imaging in Parkinson’s disease: Review and meta-analysis. Neuroimage Clin. 2017, 16, 98–110. [Google Scholar] [CrossRef] [PubMed]
- Alexander, A.L.; Lee, J.E.; Lazar, M.; Field, A.S. Diffusion tensor imaging of the brain. Neurotherapeutics 2007, 4, 316–329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Assaf, Y.; Pasternak, O. Diffusion tensor imaging (DTI)-based white matter mapping in brain research: A review. J. Mol. Neurosci. 2008, 34, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Metzler-Baddeley, C.; O’Sullivan, M.J.; Bells, S.; Pasternak, O.; Jones, D.K. How and how not to correct for CSF-contamination in diffusion MRI. Neuroimage 2012, 59, 1394–1403. [Google Scholar] [CrossRef] [PubMed]
- Pasternak, O.; Sochen, N.; Gur, Y.; Intrator, N.; Assaf, Y. Free water elimination and mapping from diffusion MRI. Magn. Reson. Med. 2009, 62, 717–730. [Google Scholar] [CrossRef] [PubMed]
- Oestreich, L.K.L.; Lyall, A.E.; Pasternak, O.; Kikinis, Z.; Newell, D.T.; Savadjiev, P.; Bouix, S.; Shenton, M.E.; Kubicki, M.; Australian Schizophrenia Research Bank; et al. Characterizing white matter changes in chronic schizophrenia: A free-water imaging multi-site study. Schizophr. Res. 2017, 189, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.L.; Rumpel, H.; Yap, K.; Lee, E.; Loo, H.V.; Ho, G.L.; Fook-Chong, S.; Yuen, Y.; Tan, E.K. Case control study of diffusion tensor imaging in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2007, 78, 1383–1386. [Google Scholar] [CrossRef]
- Yoshikawa, K.; Nakata, Y.; Yamada, K.; Nakagawa, M. Early pathological changes in the parkinsonian brain demonstrated by diffusion tensor MRI. J. Neurol. Neurosurg. Psychiatry 2004, 75, 481–484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duncan, G.W.; Firbank, M.J.; Yarnall, A.J.; Khoo, T.K.; Brooks, D.J.; Barker, R.A.; Burn, D.J.; O’Brien, J.T. Gray and white matter imaging: A biomarker for cognitive impairment in early Parkinson’s disease? Mov. Disord. 2016, 31, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Melzer, T.R.; Watts, R.; MacAskill, M.R.; Pitcher, T.L.; Livingston, L.; Keenan, R.J.; Dalrymple-Alford, J.C.; Anderson, T.J. White matter microstructure deteriorates across cognitive stages in Parkinson disease. Neurology 2013, 80, 1841–1849. [Google Scholar] [CrossRef] [PubMed]
- Pasternak, O.; Westin, C.F.; Dahlben, B.; Bouix, S.; Kubicki, M. The extent of diffusion MRI markers of neuroinflammation and white matter deterioration in chronic schizophrenia. Schizophr. Res. 2015, 161, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Ofori, E.; Pasternak, O.; Planetta, P.J.; Burciu, R.; Snyder, A.; Febo, M.; Golde, T.E.; Okun, M.S.; Vaillancourt, D.E. Increased free water in the substantia nigra of Parkinson’s disease: A single-site and multi-site study. Neurobiol. Aging 2015, 36, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Ofori, E.; Krismer, F.; Burciu, R.G.; Pasternak, O.; McCracken, J.L.; Lewis, M.M.; Du, G.; McFarland, N.R.; Okun, M.S.; Poewe, W.; et al. Free water improves detection of changes in the substantia nigra in parkinsonism: A multisite study. Mov. Disord. 2017, 32, 1457–1464. [Google Scholar] [CrossRef]
- Postuma, R.B.; Berg, D.; Stern, M.; Poewe, W.; Olanow, C.W.; Oertel, W.; Obeso, J.; Marek, K.; Litvan, I.; Lang, A.E.; et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov. Disord. 2015, 30, 1591–1601. [Google Scholar] [CrossRef]
- Matsuda, H.; Murata, M.; Mukai, Y.; Sako, K.; Ono, H.; Toyama, H.; Inui, Y.; Taki, Y.; Shimomura, H.; Nagayama, H.; et al. Japanese multicenter database of healthy controls for [(123)I]FP-CIT SPECT. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 1405–1416. [Google Scholar] [CrossRef]
- Stebbins, G.T.; Goetz, C.G.; Burn, D.J.; Jankovic, J.; Khoo, T.K.; Tilley, B.C. How to identify tremor dominant and postural instability/gait difficulty groups with the movement disorder society unified Parkinson’s disease rating scale: Comparison with the unified Parkinson’s disease rating scale. Mov. Disord. 2013, 28, 668–670. [Google Scholar] [CrossRef]
- Postuma, R.B.; Arnulf, I.; Hogl, B.; Iranzo, A.; Miyamoto, T.; Dauvilliers, Y.; Oertel, W.; Ju, Y.E.; Puligheddu, M.; Jennum, P.; et al. A single-question screen for rapid eye movement sleep behavior disorder: A multicenter validation study. Mov. Disord. 2012, 27, 913–916. [Google Scholar] [CrossRef]
- Andersson, J.L.; Sotiropoulos, S.N. An integrated approach to correction for off-resonance effects and subject movement in diffusion MR imaging. Neuroimage 2016, 125, 1063–1078. [Google Scholar] [CrossRef]
- Smith, S.M.; Jenkinson, M.; Johansen-Berg, H.; Rueckert, D.; Nichols, T.E.; Mackay, C.E.; Watkins, K.E.; Ciccarelli, O.; Cader, M.Z.; Matthews, P.M.; et al. Tract-based spatial statistics: Voxelwise analysis of multi-subject diffusion data. Neuroimage 2006, 31, 1487–1505. [Google Scholar] [CrossRef] [PubMed]
- Ball, G.; Srinivasan, L.; Aljabar, P.; Counsell, S.J.; Durighel, G.; Hajnal, J.V.; Rutherford, M.A.; Edwards, A.D. Development of cortical microstructure in the preterm human brain. Proc. Natl. Acad. Sci. USA 2013, 110, 9541–9546. [Google Scholar] [CrossRef] [Green Version]
- Jenkinson, M.; Bannister, P.; Brady, M.; Smith, S. Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage 2002, 17, 825–841. [Google Scholar] [CrossRef] [PubMed]
- Jenkinson, M.; Beckmann, C.F.; Behrens, T.E.; Woolrich, M.W.; Smith, S.M. Fsl. Neuroimage 2012, 62, 782–790. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Brady, M.; Smith, S. Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Trans. Med. Imaging 2001, 20, 45–57. [Google Scholar] [CrossRef]
- Braak, H.; Del Tredici, K. Invited Article: Nervous system pathology in sporadic Parkinson disease. Neurology 2008, 70, 1916–1925. [Google Scholar] [CrossRef]
- Dickson, D.W.; Uchikado, H.; Fujishiro, H.; Tsuboi, Y. Evidence in favor of Braak staging of Parkinson’s disease. Mov. Disord. 2010, 25, S78–S82. [Google Scholar] [CrossRef]
- Kamagata, K.; Zalesky, A.; Hatano, T.; Ueda, R.; Di Biase, M.A.; Okuzumi, A.; Shimoji, K.; Hori, M.; Caeyenberghs, K.; Pantelis, C.; et al. Gray Matter Abnormalities in Idiopathic Parkinson’s Disease: Evaluation by Diffusional Kurtosis Imaging and Neurite Orientation Dispersion and Density Imaging. Hum. Brain. Mapp. 2017, 38, 3704–3722. [Google Scholar] [CrossRef]
- Ashburner, J.; Friston, K.J. Voxel-based morphometry-The methods. Neuroimage 2000, 11, 805–821. [Google Scholar] [CrossRef]
- Ashburner, J. A fast diffeomorphic image registration algorithm. Neuroimage 2007, 38, 95–113. [Google Scholar] [CrossRef]
- Cohen, J. A power primer. Psychol. Bull. 1992, 112, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Q.; Haldar, J.P.; Yeh, F.C.; Xie, M.; Sun, P.; Tu, T.W.; Trinkaus, K.; Klein, R.S.; Cross, A.H.; et al. Quantification of increased cellularity during inflammatory demyelination. Brain 2011, 134, 3590–3601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Q.; Liu, Y.; Zhou, J. Neuroinflammation in Parkinson’s disease and its potential as therapeutic target. Transl. Neurodegener. 2015, 4, 19. [Google Scholar] [CrossRef] [PubMed]
- Reid, B.E.; Di Biase, M.A.; Pomper, M.G.; Shenton, M.E.; Du, Y.; Coughlin, J.M.; Pasternak, O. Examining Links Between Free Water and a TSPO-PET Marker of Neuroinflammation. In Proceedings of the ISMRM 27th Annual Meeting and Exhibition, Montreal, QC, Canada, 11–16 May 2019. [Google Scholar]
- Cochrane, C.J.; Ebmeier, K.P. Diffusion tensor imaging in parkinsonian syndromes: A systematic review and meta-analysis. Neurology 2013, 80, 857–864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luk, K.C.; Lee, V.M. Modeling Lewy pathology propagation in Parkinson’s disease. Parkinsonism Relat. Disord. 2014, 20, S85–S87. [Google Scholar] [CrossRef]
- Lu, H.; Jensen, J.H.; Ramani, A.; Helpern, J.A. Three-dimensional characterization of non-gaussian water diffusion in humans using diffusion kurtosis imaging. NMR Biomed. 2006, 19, 236–247. [Google Scholar] [CrossRef]
- Lee, J.E.; Chung, M.K.; Lazar, M.; DuBray, M.B.; Kim, J.; Bigler, E.D.; Lainhart, J.E.; Alexander, A.L. A study of diffusion tensor imaging by tissue-specific, smoothing-compensated voxel-based analysis. Neuroimage 2009, 44, 870–883. [Google Scholar] [CrossRef] [Green Version]
- Rathi, Y.; Pasternak, O.; Savadjiev, P.; Michailovich, O.; Bouix, S.; Kubicki, M.; Westin, C.F.; Makris, N.; Shenton, M.E. Gray matter alterations in early aging: A diffusion magnetic resonance imaging study. Hum. Brain Mapp. 2014, 35, 3841–3856. [Google Scholar] [CrossRef]
- Taylor, K.I.; Sambataro, F.; Boess, F.; Bertolino, A.; Dukart, J. Progressive Decline in Gray and White Matter Integrity in de novo Parkinson’s Disease: An Analysis of Longitudinal Parkinson Progression Markers Initiative Diffusion Tensor Imaging Data. Front. Aging Neurosci. 2018, 10, 318. [Google Scholar] [CrossRef]
- Agosta, F.; Canu, E.; Stojkovic, T.; Pievani, M.; Tomic, A.; Sarro, L.; Dragasevic, N.; Copetti, M.; Comi, G.; Kostic, V.S.; et al. The topography of brain damage at different stages of Parkinson’s disease. Hum. Brain Mapp. 2013, 34, 2798–2807. [Google Scholar] [CrossRef]
- Rektor, I.; Svatkova, A.; Vojtisek, L.; Zikmundova, I.; Vanicek, J.; Kiraly, A.; Szabo, N. White matter alterations in Parkinson’s disease with normal cognition precede grey matter atrophy. PLoS ONE 2018, 13, e0187939. [Google Scholar] [CrossRef] [PubMed]
- Hattori, T.; Orimo, S.; Aoki, S.; Ito, K.; Abe, O.; Amano, A.; Sato, R.; Sakai, K.; Mizusawa, H. Cognitive status correlates with white matter alteration in Parkinson’s disease. Hum. Brain Mapp. 2012, 33, 727–739. [Google Scholar] [CrossRef] [PubMed]
- Mukaetova-Ladinska, E.B.; McKeith, I.G. Pathophysiology of synuclein aggregation in Lewy body disease. Mech. Ageing Dev. 2006, 127, 188–202. [Google Scholar] [CrossRef] [PubMed]
- Rub, U.; Del Tredici, K.; Schultz, C.; Ghebremedhin, E.; de Vos, R.A.; Jansen Steur, E.; Braak, H. Parkinson’s disease: The thalamic components of the limbic loop are severely impaired by alpha-synuclein immunopositive inclusion body pathology. Neurobiol. Aging 2002, 23, 245–254. [Google Scholar] [CrossRef]
- Salsone, M.; Cerasa, A.; Arabia, G.; Morelli, M.; Gambardella, A.; Mumoli, L.; Nistico, R.; Vescio, B.; Quattrone, A. Reduced thalamic volume in Parkinson disease with REM sleep behavior disorder: Volumetric study. Parkinsonism Relat. Disord. 2014, 20, 1004–1008. [Google Scholar] [CrossRef] [PubMed]
- Hawkes, C.H.; Del Tredici, K.; Braak, H. A timeline for Parkinson’s disease. Parkinsonism Relat. Disord. 2010, 16, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.C.; Ulane, C.M.; Burke, R.E. Clinical progression in Parkinson disease and the neurobiology of axons. Ann. Neurol. 2010, 67, 715–725. [Google Scholar] [CrossRef] [PubMed]
- Lenfeldt, N.; Hansson, W.; Larsson, A.; Nyberg, L.; Birgander, R.; Forsgren, L. Diffusion tensor imaging and correlations to Parkinson rating scales. J. Neurol. 2013, 260, 2823–2830. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, I.W.; Buckley, S.; Coffey, C.S.; Foster, E.; Mendick, S.; Seibyl, J.; Schuff, N. Diffusion tensor imaging of the nigrostriatal fibers in Parkinson’s disease. Mov. Disord. 2015, 30, 1229–1236. [Google Scholar] [CrossRef]
- Wen, M.C.; Heng, H.S.; Ng, S.Y.; Tan, L.C.; Chan, L.L.; Tan, E.K. White matter microstructural characteristics in newly diagnosed Parkinson’s disease: An unbiased whole-brain study. Sci. Rep. 2016, 6, 35601. [Google Scholar] [CrossRef]
- Andica, C.; Kamagata, K.; Hatano, T.; Okuzumi, A.; Saito, A.; Nakazawa, M.; Ueda, R.; Motoi, Y.; Kamiya, K.; Suzuki, M.; et al. Neurite orientation dispersion and density imaging of the nigrostriatal pathway in Parkinson’s disease: Retrograde degeneration observed by tract-profile analysis. Parkinsonism Relat. Disord. 2018, 51, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Perlmutter, J.S. Assessment of Parkinson disease manifestations. Curr. Protoc. Neurosci. 2009, 49, 10.1.1–10.1.14. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, G.; Caligiuri, M.E.; Arabia, G.; Cherubini, A.; Lupo, A.; Nistico, R.; Salsone, M.; Novellino, F.; Morelli, M.; Cascini, G.L.; et al. Structural connectivity differences in motor network between tremor-dominant and nontremor Parkinson’s disease. Hum. Brain Mapp. 2017, 38, 4716–4729. [Google Scholar] [CrossRef] [PubMed]
- Iranzo, A.; Santamaria, J.; Rye, D.B.; Valldeoriola, F.; Marti, M.J.; Munoz, E.; Vilaseca, I.; Tolosa, E. Characteristics of idiopathic REM sleep behavior disorder and that associated with MSA and PD. Neurology 2005, 65, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Hashimoto, M.; Yoshioka, M.; Murakami, M.; Kawasaki, K.; Urashima, M. The odor stick identification test for Japanese differentiates Parkinson’s disease from multiple system atrophy and progressive supra nuclear palsy. BMC Neurol. 2011, 11, 157. [Google Scholar] [CrossRef] [PubMed]

| HC | All PD | Right-Sided Onset PD | Left-Sided Onset PD | P Value (HC vs. All PD | P Value (R vs. L Onset PD) | P Value (HC vs. R Onset PD) | P Value (HC vs. L Onset PD) | |
|---|---|---|---|---|---|---|---|---|
| Number | 20 | 20 | 12 | 8 | — | — | — | — |
| Sex, N (male/female) ° | 12/8 | 11/9 | 5/7 | 6/2 | 0.75 | 0.14 | 0.31 | 0.45 |
| Age (mean years ± SD) * | 67.15 ± 1.18 | 65.05 ± 10.9 | 63.50 ± 11.13 | 67.38 ± 8.43 | 0.52 | 0.16 | 0.15 | 0.91 |
| Disease duration (mean years ± SD) | — | 6.95 ± 3.93 | 7.5 ± 3.63 | 6.13 ± 4.45 | — | 0.46 | — | — |
| MDS-UPDRS part I (mean ± SD) * | — | 5.45 ± 2.93 | 5.25 ± 2.45 | 5.75 ± 3.69 | — | 0.72 | — | — |
| MDS-UPDRS part I subscores (mean ± SD) * | ||||||||
| Cognitive (I.1) | — | 0.15 ± 0.37 | 0.17 ± 0.39 | 0.13 ± 0.35 | — | 0.81 | — | — |
| Neuropsychiatric (I.2–I.6) | — | 1.20 ± 1.32 | 1.00 ± 1.04 | 1.50 ± 1.69 | — | 0.42 | — | — |
| Sleep disorder (I.7, I.8) | — | 1.45 ± 1.28 | 1.25 ± 1.22 | 1.75 ± 1.39 | — | 0.41 | — | — |
| Sensory and others (I.9, I.13) | — | 0.90 ± 1.02 | 1.83 ± 1.11 | 1.63 ± 0.74 | — | 0.61 | — | — |
| Autonomic (I.10–I.12) | — | 1.75 ± 0.97 | 1.83 ± 1.11 | 1.63 ± 0.74 | — | 0.65 | — | — |
| MDS-UPDRS part III (mean ± SD) * | — | 11.05 ± 5.22 | 11.25 ± 4.86 | 10.75 ± 6.04 | — | 0.84 | — | — |
| Hoehn and Yahr staging (mean ± SD) * | — | 1.85 ± 0.37 | 1.75 0.45 | 2 ± 0 | 0.14 | — | — | |
| 1, N (%) | — | 3 (15%) | 3 (25%) | 0 (0%) | — | — | — | — |
| 2, N (%) | — | 17 (85%) | 9 (75%) | 8 (100%) | — | — | — | — |
| LED (mean ± SD) * | — | 862.25 ± 596.50 | 898.75 ± 607.84 | 807.50 ± 616.02 | — | 0.75 | — | — |
| Mean SBR (mean ± SD) | — | 3.28 ± 1.27 | — | — | — | — | — |
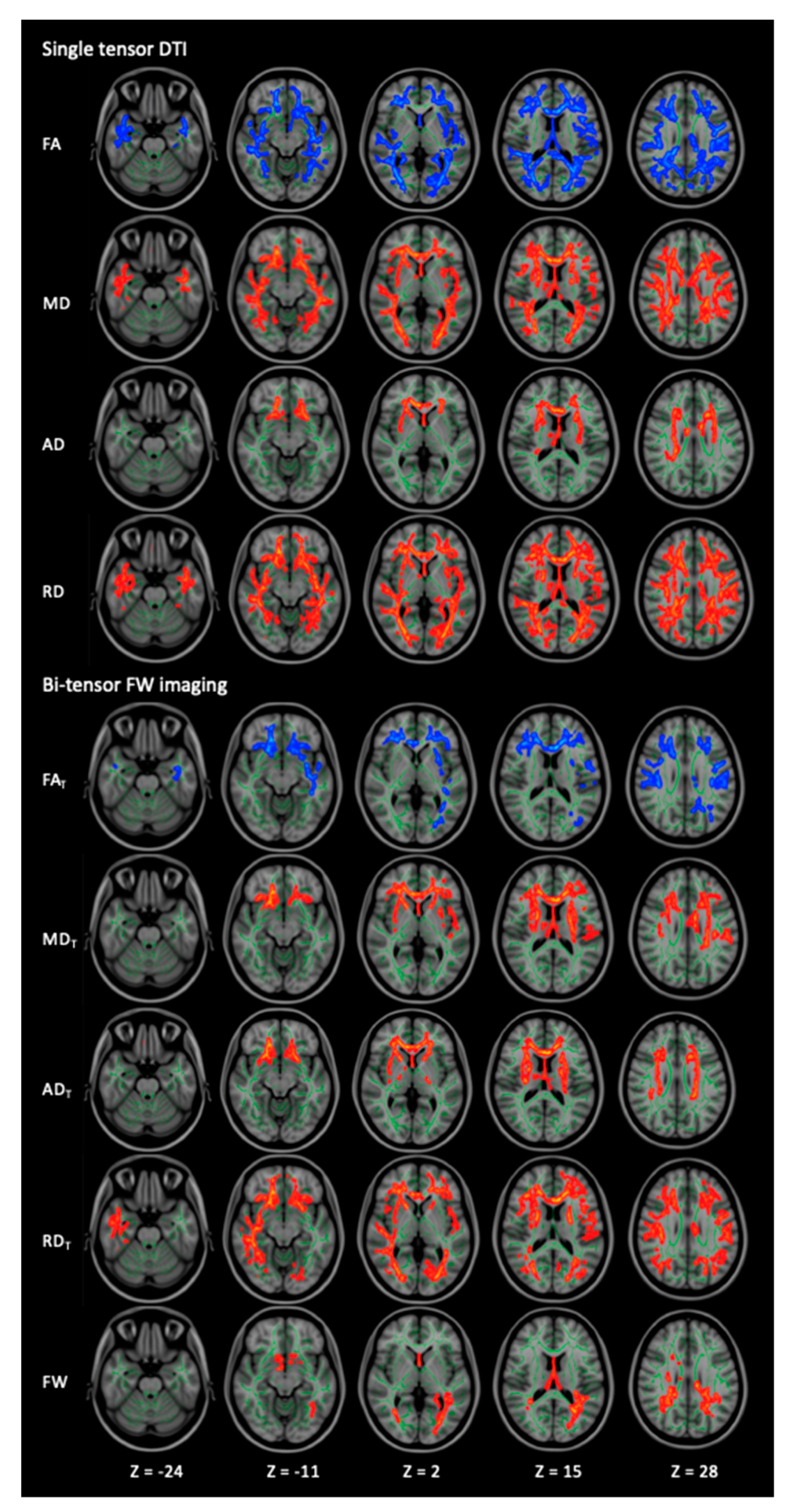
| Modality | Contrast | Cluster Size | Anatomical Region | Peak t-Value | Peak MNI Coordinates (X, Y, Z) |
|---|---|---|---|---|---|
| Single-tensor DTI | |||||
| FA | HC > PD | 46483 | Bilateral ATR, CST, CgH, IFOF, ILF, SLF, UF, temporal part of the SLF, retrolenticular part of the IC, ACR, SCR, PCR, PTR, sagittal stratum, external capsule, tapatum; Lt-CCG; Rt-PLIC; fornix, forceps major and minor, genu, body and splenium of CC | 6.81 | 133, 124, 44 |
| MD | HC < PD | 39448 | Bilateral ATR, CST, IFOF, ILF, SLF, UF, temporal part of the SLF, ALIC, PLIC, retrolenticular part of the IC, ACR, SCR, PCR, PTR, sagittal stratum, external capsule, SFOF, tapatum; genu, body and splenium of CC, fornix and forceps major and minor | 5.9 | 113, 160, 76 |
| AD | HC < PD | 8520 | Bilateral ATR, CST, IFOF, UF, ALIC, ACR, SCR, external capsule, SFOF; Lt-PLIC; Rt-SLF, PCR, retrolenticular part of the IC, fornix; genu and body of CC, forceps minor | 5.49 | 113, 160, 76 |
| RD | HC < PD | 54131 | Bilateral ATR, CST, CgH, IFOF, ILF, SLF, UF, temporal part of the SLF, medial lemniscus, ICP, SCP, ALIC, PLIC, retrolenticular part of the IC, ACR, SCR, PCR, PTR, sagittal stratum, external capsule, SFOF, tapatum; Lt-CCG; genu, body and splenium of CC, fornix, forceps major and minor and MCP | 5.95 | 121, 106, 64 |
| Bi-tensor FW imaging | |||||
| FAT | HC > PD | 22185 | Bilateral ATR, CST, IFOF, ILF, SLF, ACR, SCR, PCR, external capsule; Lt-CCG, UF, retrolenticular part of the IC, PTR, sagittal stratum; forceps major and minor, genu, body and splenium of CC, fornix | 5.37 | 45, 125, 47 |
| MDT | HC < PD | 18356 | Bilateral ATR, CST, IFOF, SLF, UF, ALIC, PLIC, ACR, SCR, external capsule, SFOF; Lt-CCG, retrolenticular part of the IC, PCR; forceps minor, genu, body and splenium of CC; fornix | 5.73 | 119, 94, 120 |
| ADT | HC < PD | 11610 | Bilateral ATR, CST, IFOF, UF, ALIC, PLIC, retrolenticular part of the IC, ACR, SCR, PCR, external capsule, SFOF; Rt- SLF; genu, body and splenium of the CC and forceps minor | 5.52 | 80, 158, 77 |
| RDT | HC < PD | 33504 | Bilateral ATR, CST, IFOF, ILF, SLF, UF, ALIC, PLIC, ACR, SCR, PCR, PTR, sagittal stratum, external capsule, SFOF; Lt-CCG; temporal part of the Rt-SLF, retrolenticular part of the IC, UF, tapatum; forceps major and minor, genu, body and splenium of CC, fornix | 5.64 | 143, 99, 99 |
| FW | HC < PD | 5716 | Bilateral ATR, CST, IFOF, ILF, SLF, SLF temporal part, SCR, PCR, PTR, tapatum; Lt-retrolenticular part of the IC, sagittal stratum; Rt-ACR; forceps major and minor, genu, body and splenium of CC, fornix | 5.45 | 89, 133, 74 |
| WM Areas | HC | PD | P Value | t-Value | Cohen’s d | |||
|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | |||||
| DTI | ||||||||
| FA | Anterior | 0.48 | 0.014 | 0.47 | 0.018 | 0.019 * | 2.46 | 0.78 |
| Posterior | 0.61 | 0.021 | 0.60 | 0.021 | 0.011 * | 2.69 | 0.85 | |
| MD | Anterior | 0.80 | 0.035 | 0.83 | 0.048 | 0.014 * | −2.58 | 0.81 |
| Posterior | 0.82 | 0.029 | 0.85 | 0.047 | 0.036 ° | −2.18 | 0.69 | |
| AD | Anterior | 1.25 | 0.043 | 1.28 | 0.058 | 0.038 ° | −2.16 | 0.68 |
| Posterior | 1.49 | 0.038 | 1.51 | 0.058 | 0.32 | −1.00 | 0.32 | |
| RD | Anterior | 0.57 | 0.032 | 0.61 | 0.046 | 0.010 * | −2.71 | 0.86 |
| Posterior | 0.49 | 0.032 | 0.52 | 0.045 | 0.012 * | −2.65 | 0.84 | |
| FW imaging | ||||||||
| FAT | Anterior | 0.64 | 0.026 | 0.61 | 0.020 | 0.0021 * | 3.30 | 1.04 |
| Posterior | 0.75 | 0.018 | 0.75 | 0.019 | 0.40 | 0.85 | 0.27 | |
| MDT | Anterior | 0.56 | 0.039 | 0.60 | 0.036 | 0.0014 * | −3.45 | 1.09 |
| Posterior | 0.61 | 0.014 | 0.62 | 0.022 | 0.25 | −1.16 | 0.37 | |
| ADT | Anterior | 1.00 | 0.053 | 1.05 | 0.053 | 0.0025 * | −3.24 | 1.02 |
| Posterior | 1.29 | 0.027 | 1.29 | 0.041 | 0.71 | −0.38 | 0.12 | |
| RDT | Anterior | 0.34 | 0.033 | 0.37 | 0.031 | 0.0016 * | −3.40 | 1.22 |
| Posterior | 0.27 | 0.020 | 0.28 | 0.024 | 0.26 | −1.14 | 0.36 | |
| FW | Anterior | 0.21 | 0.016 | 0.23 | 0.042 | 0.079 | −1.81 | 0.57 |
| Posterior | 0.21 | 0.024 | 0.24 | 0.042 | 0.020 * | −2.44 | 0.77 | |
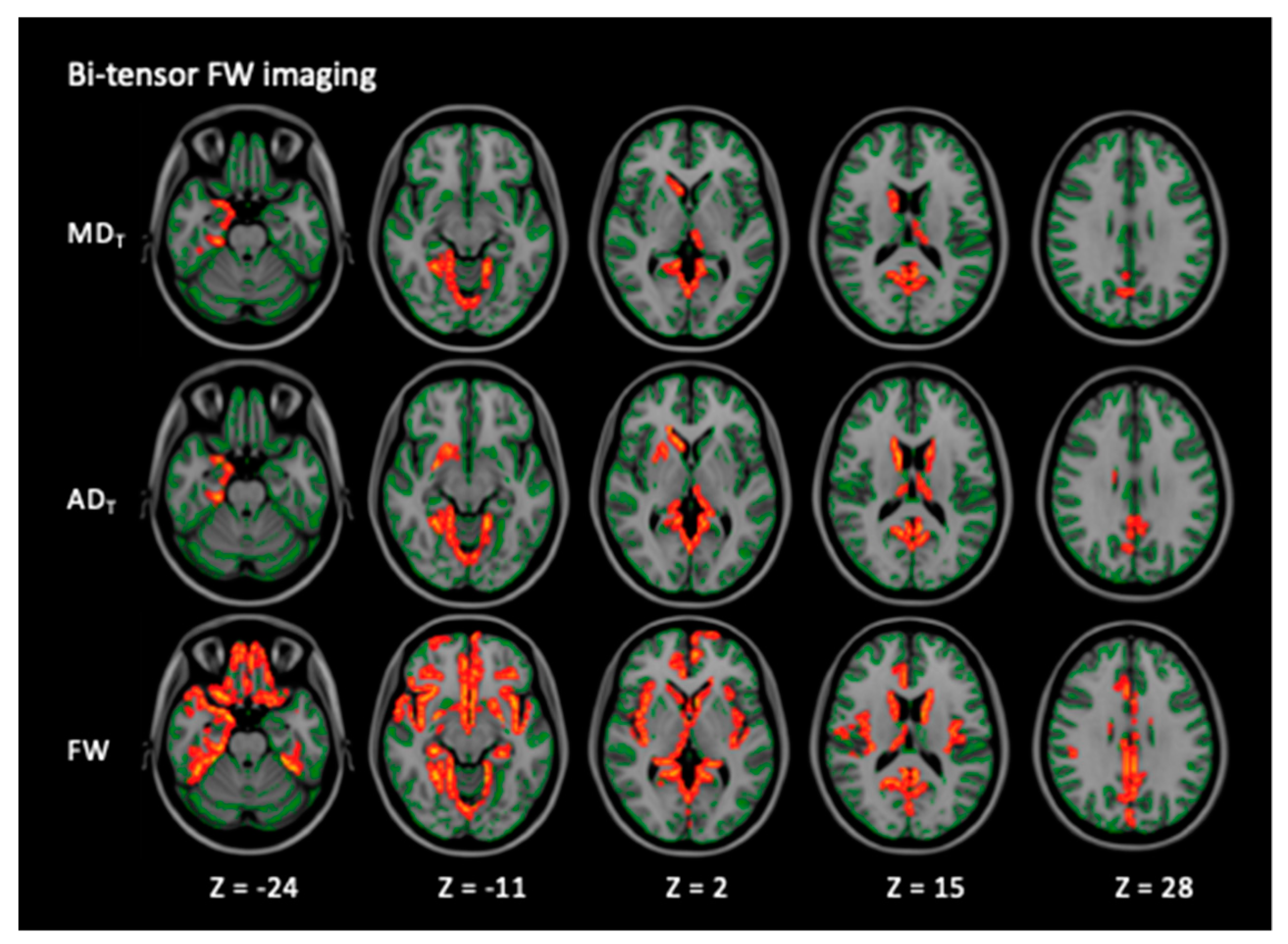
| Modality | Contrast | Cluster Size | Anatomical Region | Peak t-Value | Peak MNI Coordinates (X, Y, Z) | |
|---|---|---|---|---|---|---|
| MDT | HC < PD | 4.9 | 38, 60, 46 | |||
| — | Frontal | — | ||||
| 42 | Temporal | Bilateral fusiform, Rt-entorhinal and temporal pole | ||||
| 43 | Parietal | Bilateral precuneus | ||||
| 23 | Occipital | Bilateral lingual | ||||
| 102 | Limbic and para-limbic | Bilateral isthmus cingulate and para-hippocampal; Rt-hippocampus | ||||
| 49 | Deep GM | Lt-thalamus; Rt-caudate and putamen | ||||
| ADT | HC < PD | 5.05 | 38, 60, 46 | |||
| — | Frontal | — | ||||
| 62 | Temporal | Bilateral fusiform; Rt-enthorinal, inferior temporal and temporal pole | ||||
| 53 | Parietal | Bilateral precuneus | ||||
| 27 | Occipital | Bilateral lingual | ||||
| 146 | Limbic and para-limbic | Bilateral isthmus cingulate, para-hippocampal and hippocampus; Rt-amygdala and accumbens | ||||
| 162 | Deep GM | Bilateral thalamus, caudate; Rt-putamen | ||||
| FW | HC < PD | 7.37 | 43, 46, 54 | |||
| 534 | Frontal | Bilateral lateral orbitofrontal, medial orbitofrontal, pars opercularis, pars orbitalis, pars triangularis, superior frontal, frontal pole and precentral; Rt-paracentral and rostral middle frontal | ||||
| 235 | Temporal | Bilateral fusiform and superior temporal; Lt-transverse temporal; Rt-entorhinal, fusiform, inferior temporal and temporal pole | ||||
| 132 | Parietal | Bilateral post-central, precuneus; Rt-supramarginal | ||||
| 47 | Occipital | Bilateral lingual; Lt-cuneus, lateral-occipital and pericalcarine | ||||
| 1106 | Limbic and para-limbic | Bilateral isthmus cingulate, caudal anterior cingulate, posterior cingulate, rostral anterior cingulate, insula, para-hippocampal, accumbens and hippocampus; Rt-amygdala | ||||
| 133 | Deep GM | Bilateral thalamus, caudate and putamen | ||||
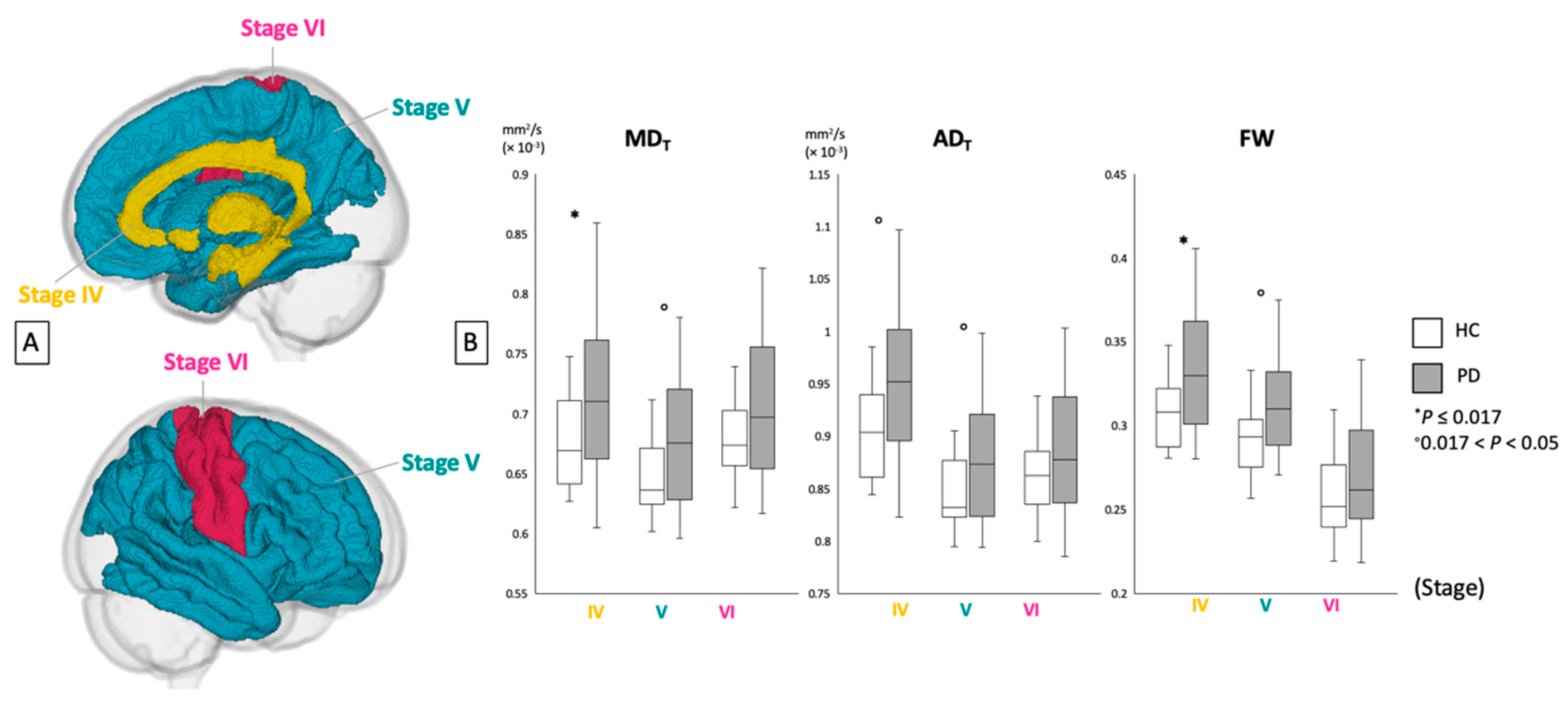
| Braak Stage | HC | PD | P Value | t-Value | Cohen’s d | |||
|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | |||||
| MDT | IV | 0.56 | 0.039 | 0.60 | 0.036 | 0.016 * | −2.51 | 0.79 |
| V | 0.61 | 0.014 | 0.62 | 0.022 | 0.036 ° | −2.18 | 0.69 | |
| VI | 0.34 | 0.033 | 0.37 | 0.031 | 0.073 | −1.85 | 0.35 | |
| ADT | IV | 0.90 | 0.043 | 0.95 | 0.078 | 0.018 ° | −2.48 | 0.78 |
| V | 0.84 | 0.032 | 0.88 | 0.059 | 0.044 ° | −2.08 | 0.66 | |
| VI | 0.86 | 0.037 | 0.88 | 0.064 | 0.20 | −1.30 | 0.41 | |
| FW | VI | 0.31 | 0.020 | 0.33 | 0.037 | 0.0059 * | −2.92 | 0.92 |
| V | 0.29 | 0.020 | 0.31 | 0.030 | 0.021 ° | −2.41 | 0.76 | |
| VI | 0.26 | 0.025 | 0.27 | 0.036 | 0.13 | −1.56 | 0.49 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andica, C.; Kamagata, K.; Hatano, T.; Saito, A.; Uchida, W.; Ogawa, T.; Takeshige-Amano, H.; Zalesky, A.; Wada, A.; Suzuki, M.; et al. Free-Water Imaging in White and Gray Matter in Parkinson’s Disease. Cells 2019, 8, 839. https://doi.org/10.3390/cells8080839
Andica C, Kamagata K, Hatano T, Saito A, Uchida W, Ogawa T, Takeshige-Amano H, Zalesky A, Wada A, Suzuki M, et al. Free-Water Imaging in White and Gray Matter in Parkinson’s Disease. Cells. 2019; 8(8):839. https://doi.org/10.3390/cells8080839
Chicago/Turabian StyleAndica, Christina, Koji Kamagata, Taku Hatano, Asami Saito, Wataru Uchida, Takashi Ogawa, Haruka Takeshige-Amano, Andrew Zalesky, Akihiko Wada, Michimasa Suzuki, and et al. 2019. "Free-Water Imaging in White and Gray Matter in Parkinson’s Disease" Cells 8, no. 8: 839. https://doi.org/10.3390/cells8080839





